Abstract
Background. To improve clinical and public health outcomes through early human immunodeficiency virus (HIV) detection, fourth-generation antigen/antibody immunoassay (4IA) and supplemental testing results must be returned rapidly.
Methods. We examined HIV testing data at Harborview Medical Center (HMC), Massachusetts General Hospital (MGH), and the Medical University of South Carolina (MUSC), which used 4IA and supplemental antibody and nucleic acid tests (NATs). At MGH and MUSC, HIV-1 Western blot (WB) and HIV-2 testing were conducted at a reference laboratory. We compared time from specimen collection to laboratory result for established (positive WB) and acute infections (reactive 4IA, negative/indeterminate WB, detectable NAT), and we calculated testing cost per positive-test result.
Results. From 3731 (MUSC) to 19 774 (MGH) tests were conducted; 0.01% (MGH) to 0.05% (HMC) were acute infections. Each laboratory had reactive 4IA, WB-negative, or indeterminate specimens without NAT (ie, potential acute infections). Time to result was 1.5 (HMC) to 5.2 days (MGH) for acute and 1.0 (HMC) to 5.2 days (MGH) for established infections. Costs were $1054 (MGH) to $1521 (MUSC).
Conclusions. Conducting supplemental testing in-house lowered turnaround times, which may be further reduced with rapid HIV-1/HIV-2 differentiation tests. Hospitals may benefit from quantitative NATs not requiring physician orders, so all potential acute infections receive NAT.
Keywords: acute infection, cost, HIV, laboratory
The accurate diagnosis of human immunodeficiency virus (HIV) infection is paramount in controlling the spread of infection. Four laboratory-based, fourth-generation HIV antigen/antibody screening assays are approved by the US Food and Drug Administration (FDA): ADVIA Centaur HIV Ag/Ab Combo (CHIV) Assay (Siemens, Tarrytown, NY), ARCHITECT HIV Ag/Ab Combo (Abbott Diagnostics, Chicago, IL) (ARCHITECT), BioPlex 2200 HIV Ag-Ab assay (Bio-Rad Laboratories, Inc., Hercules, CA), and GS HIV Combo Ag/Ab EIA (Bio-Rad Laboratories, Redmond, WA) (GS Combo).
These assays can detect acute infections that occur during a time associated with high potential for transmission [1–3], when antigen is present, but antibodies are not [4, 5]. Evidence suggests that there is a clinical benefit to providing antiretroviral treatment during all stages of HIV infection and that treatment substantially reduces transmissions and improves health outcomes [6–12].
In 2014, the Centers for Disease Control and Prevention (CDC) and the Association of Public Health Laboratories (APHL) recommended an algorithm in which an antigen/antibody immunoassay is followed, when reactive, with an HIV-1/HIV-2 antibody differentiation supplemental test. This is preferred over the alternative, the HIV-1 Western blot (WB), which does not distinguish HIV-2 from HIV-1 [13]. When the screening test and antibody supplemental test are positive, a person is considered to have an established infection. When the antibody supplemental test is negative or indeterminate, the recommended algorithm calls for a follow-up nucleic acid test (NAT), which can detect acute infections.
One NAT is FDA-approved for diagnostic use, the APTIMA HIV-1 RNA Qualitative Assay (Hologic, San Diego, CA). However, hospitals typically use an FDA-approved quantitative RNA viral load assay to assess patient prognosis and to monitor response to antiretroviral therapy [14]. Physicians can order these assays for patient monitoring, but a laboratory must perform an extensive validation to automatically conduct them for diagnostic purposes for a specimen with repeatedly reactive screening test results and negative or indeterminate antibody supplemental test results [15].
Routine HIV testing for adults and adolescents is recommended in US hospitals [16, 17], but reports on the use of fourth-generation assays and the proportion of acute infections identified in these settings are limited [4, 18, 19]. The time to reporting fourth-generation assay results should be minimized to capitalize on the benefit of identifying early infections so that persons who may be highly infectious can promptly be linked to treatment and partner services. Few studies have examined the cost of fourth-generation assays and supplemental tests in hospitals, although cost plays an important role in the uptake of this technology [20].
In this paper, we report the results of a study conducted in 3 US hospital laboratories using fourth-generation immunoassays to examine the proportion of acute infections, time to result, and laboratory testing cost per HIV infection identified.
METHODS
Hospital Human Immunodeficiency Virus Testing Algorithms
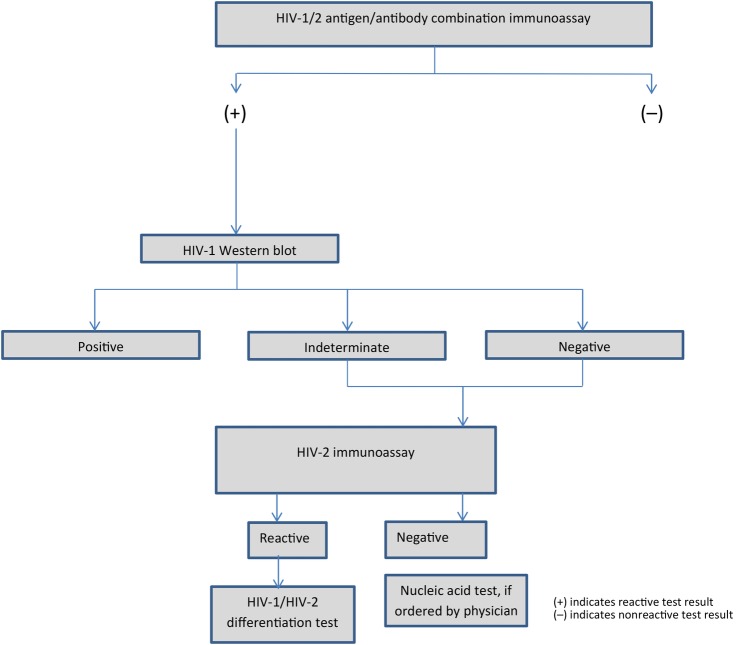
Three hospitals (Harborview Medical Center [HMC; Seattle, WA], Massachusetts General Hospital [MGH; Boston, MA], and the Medical University of South Carolina [MUSC; Charleston, SC]), which used fourth-generation antigen/antibody immunoassays, an antibody supplemental test, and a NAT, were included in this study. These hospitals adopted fourth-generation assays before the CDC's 2014 recommendations were issued [13]. Massachusetts General Hospital and MUSC used an algorithm based on the CDC's former recommendation to follow a reactive HIV-1/HIV-2 immunoassay with an HIV-1 WB, and, when negative or indeterminate, to conduct an HIV-2 immunoassay [21] (Figure 1 ). The MGH laboratory used the ARCHITECT as the screening test, and if it was repeatedly reactive the laboratory conducted a GS HIV-1 Western blot (Bio-Rad Laboratories, Redmond, WA). If the WB was negative or indeterminate, a GS HIV-2 EIA (Bio-Rad Laboratories, Redmond, WA) was conducted. When the HIV-2 EIA was reactive, the laboratory conducted the INNO-LIA HIV I/II Score test (Fujirebio Europe, Ghent, Belgium). A reference laboratory conducted the HIV-1 WB and HIV-2 immunoassays. The MGH hospital laboratory used the COBAS Ampliprep/COBAS TaqMan HIV-1 version 2.0 (Roche Molecular Systems, Branchburg, NJ), an HIV-1 RNA quantitative viral load test, when ordered by a physician.
Figure 1.
Human immunodeficiency virus (HIV) testing algorithm used at Massachusetts General Hospital and the Medical University of South Carolina.
At MUSC, when the GS Combo screening test was repeatedly reactive, the specimen was sent to a reference laboratory that conducted a GS HIV-1 WB (Bio-Rad Laboratories). If the WB was negative or indeterminate, the laboratory conducted a GS HIV-2 EIA (Bio-Rad Laboratories). If the HIV-2 IA was reactive, the Multispot HIV-1/HIV-2 Rapid Test (Multispot) (Bio-Rad Laboratories, Redmond, WA) was conducted. The hospital laboratory also used an HIV-1 viral load test, m2000rt RealTime HIV-1 RNA (Abbott Molecular, Inc., Des Plaines, IL), if ordered by the physician.
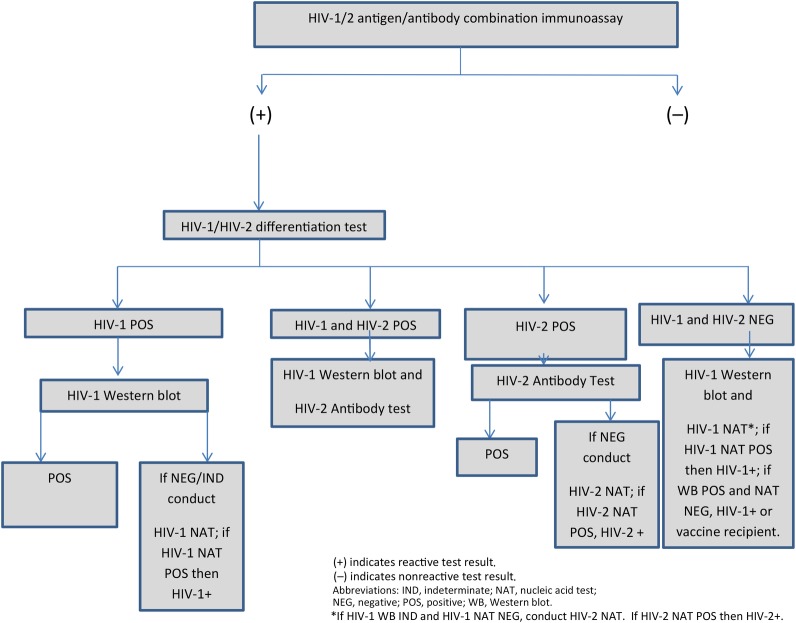
The Harborview laboratory used a modified algorithm (Figure 2). When the ARCHITECT was repeatedly reactive, a Multispot test was conducted. When Multispot was reactive for HIV-1 only, a GS HIV-1 WB (Bio-Rad Laboratories) was conducted. If the WB was negative or indeterminate, they ran an HIV-1 viral load test, m2000rt RealTime HIV-1 RNA (Abbott Molecular, Inc.), which they had independently validated for use in a diagnostic algorithm. If Multispot was reactive for both HIV-1 and HIV-2, the Harborview laboratory conducted an HIV-1 WB and forwarded the sample to a reference laboratory for an HIV-2 immunoblot [22]. If Multispot was reactive for HIV-2 only, the laboratory forwarded the sample to a reference laboratory for an HIV-2 immunoblot. If that was not reactive, the laboratory tested the specimen with a quantitative HIV-2 RNA real-time, independently validated in-house polymerase chain reaction assay [20]. If Multispot was HIV-1 and HIV-2 negative, an HIV-1 WB and an HIV-1 NAT were conducted. If the HIV-1 NAT was negative and the WB was indeterminate, an HIV-2 NAT was conducted.
Figure 2.
Human immunodeficiency virus (HIV) testing algorithm used by Harborview Medical Center.
Testing Outcomes
Results from routine diagnostic testing conducted in the hospital laboratory using specimens collected in the medical center (inpatient, emergency room, outpatient, affiliated primary care clinics) from July 1, 2012 to June 30, 2013 (HMC and MGH) and January 4, 2013 to July 4, 2013 (MUSC) were retrospectively reviewed and were categorized as fourth-generation nonreactive or repeatedly reactive. Specimens that were repeatedly reactive were further categorized as follows: HIV-1 antibody positive (ie, HIV-1 WB positive), acute HIV-1 infection (ie, antibody supplemental test negative or indeterminate, detectable HIV-1 NAT), fourth-generation assay false positive (ie, antibody supplemental test negative or indeterminate, undetectable HIV-1 NAT), HIV-2 positive, incomplete testing, or other.
Time to Reporting a Test Result
We describe (1) the time from specimen collection to receipt in the hospital laboratory and (2) time from receipt in the hospital laboratory to reporting of final HIV-1 negative or positive results into the laboratory information system. At MGH, we also used aggregate medical record data to evaluate time to patient receipt of HIV-1 antibody positive and acute infection results, although time to receipt of results was not collected for persons previously known to be HIV-positive and research specimens.
Laboratory Testing Cost per Human Immunodeficiency Virus Infection Detected
To obtain cost per infection at each hospital, we summed total laboratory testing and labor costs and divided by the number of specimens confirmed to be HIV-1 or HIV-2 positive. To calculate laboratory-testing costs, we collected information on the test kit brand, cost and size, typical run size, and frequency and number of controls per run. An adjusted cost per test was calculated by adding the total kit cost to the cost of controls not provided in the kit and dividing by the number of tests not used for controls [20]. This number was multiplied by the number of times the test was conducted during the study period. That product was summed for each test used to obtain total laboratory testing costs.
Labor costs were calculated for each test by obtaining the product of the labor hours per specimen, the total number of specimens tested, and the labor cost per hour. The labor hours were based on estimates from the literature when the laboratory technologist could not perform other tasks [20, 23, 24]. In addition to test processing times, they included time to prepare samples for testing as well as time to report results. These times were estimated to be 1.9 hours (ARCHITECT), 1.3 hours (manual GS Combo), 3 hours (WB), and 0.5 hours (Multispot). The labor cost per hour was based on mean hourly rates including benefits from the Bureau of Labor Statistics for medical and clinical laboratory technologists in the state where the hospital is located [25]. Labor costs were summed for each test. Reference laboratory test charges to the hospital included testing and labor costs. Shared laboratory overhead costs (such as utilities) or other costs (such as time used for counseling patients) were not included. In a separate computation, the laboratory testing cost per specimen screened was calculated using the same methods as described above except that the total laboratory testing and labor costs were divided by the number of screening tests, instead of the number of positive specimens. Data were analyzed using SAS 9.3 (Cary, NC) and Excel (cost data only).
The project was approved by each hospital's Institutional Review Board. Routinely collected, deidentified data were submitted to the CDC by the contractor, John Snow Inc., using a secure data network. The project was approved as postmarketing surveillance by the CDC.
RESULTS
Testing Outcomes
The hospitals tested 3731 (MUSC), 14 511 (Harborview), and 19 774 (MGH) specimens (Table 1). The proportion of acute infections ranged from 0.01% (MGH) to 0.05% (HMC). The prevalence of established antibody-positive HIV-1 infections was <1% in all hospitals. Human immunodeficiency virus-2 positive specimens were only identified at MGH (0.01% of total tests). Acute infections constituted 1.6% of HIV-1 infections at MGH, 5.6% at Harborview, and 5.9% at MUSC. Of the tests conducted, <0.2% had false-positive fourth-generation immunoassay results; the specificity of fourth-generation assays was 99.8% or greater. There were 4 specimens from MGH, 3 from MUSC, and 2 from Harborview with a repeatedly reactive immunoassay and negative or indeterminate WB that did not have an HIV-1 NAT result. It is not known whether these would represent false-positive fourth-generation results or acute infections.
Table 1.
Testing Outcomes for Specimens Tested in 3 US Hospitals That Use an HIV Fourth-Generation Immunoassay
| Test Results | Harborview Medical Center | Massachusetts General Hospital | Medical University of South Carolina |
|---|---|---|---|
| n (%) | n (%) | n (%) | |
| Fourth-generation IA | |||
| Nonreactive | 14357 (98.94) | 19630 (99.27) | 3711 (99.44) |
| Repeatedly reactive | 154 | 144 | 20 |
| Results of repeatedly reactive IAs | |||
| Established HIV-1a | 118 (0.81) | 121 (0.61) | 16 (0.43) |
| Acute infectionb | 7 (0.05) | 2 (0.01) | 1 (0.03) |
| IA false positivec | 26 (0.18) | 12 (0.06) | 0 |
| HIV-2 positived | 0 | 2 (0.01) | 0 |
| Incomplete testing | 2e (0.01) | 7f (0.04) | 3g (0.08) |
| Other | 1h (0.01) | 0 | 0 |
Abbreviations: HIV, human immunodeficiency virus; HMC, Harborview Medical Center; IA, immunoassay; MGH, Massachusetts General Hospital; MUSC, Medical University of South Carolina; NAT, nucleic acid test; WB, Western blot.
a Human immunodeficiency virus-1 positive by WB or Multispot.
b Western blot or HIV-1/HIV-2 rapid test negative or indeterminate, detectable NAT result.
c Western blot or HIV-1/HIV-2 rapid test negative or indeterminate, NAT negative. Fourth-generation specificity was 99.8% (HMC), 99.9% (MGH), 100% (MUSC).
d Positive for HIV-2 by HIV-1/HIV-2 rapid test, HIV-2 IA, HIV-2 immunoblot, or HIV-2 NAT.
e Western blot negative, NAT not done (n = 2).
f Western blot negative or indeterminate, NAT not done (n = 4); WB not done (n = 1); WB not done, reactive NAT (n = 1); WB not done, negative NAT (n = 1).
g Western blot negative or indeterminate, HIV-2 EIA negative, NAT not done (n = 3).
h Human immunodeficiency virus-1 positive by HIV-1/HIV-2 rapid test, WB indeterminate, NAT negative (n = 1).
Human Immunodeficiency Virus Testing Turnaround Times
Based on median times at each hospital, the initial fourth-generation assay result was available within 1 day of specimen collection (Table 2). The time from specimen receipt in the hospital laboratory to WB result was 1 day at HMC, which conducts the test in-house, and 5.0 and 5.1 days at MGH and MUSC, which use a reference laboratory. For specimens with negative or indeterminate WB results that had an HIV-1 NAT, HMC had a lower time from receipt of specimen to NAT result than MGH and MUSC (1.5 days compared with 4.0 and 5.0 days, respectively). At MGH, it took 4 days for persons to receive reactive WB results after test results were available. For the individual with acute HIV-1 infection at MGH, it took 12 days to receive test results.
Table 2.
Time to Test Result Report in 3 US Hospital Laboratories Using Fourth-Generation HIV Immunoassays
| Time Period | Median Days, Harborview Medical Center (Minimum, Maximum) | Median Days, Massachusetts General Hospital (Minimum, Maximum) | Median Days, Medical University of South Carolina (Minimum, Maximum) |
|---|---|---|---|
| Specimen collection to laboratory receipt | 1.0 (0, 58.0) | 0.1 (0, 2.9) | 0 (0, 3.0) |
| Laboratory receipt to nonreactive IA | 0 (0, 5.0) | 0.8 (0, 17.9) | 1.0 (0,7.0) |
| Receipt to positive WB reporta | 1.0 (0, 6.0) | 5.0 (2.0, 10.8) | 5.1 (3.2, 9.2) |
| Receipt to NAT report, acute | 1.5 (1.0, 5.0) | 5.0 (5.0, 5.0)b | 4.0c |
| Collection to positive WB report | 1 (0, 6) | 5.2 (2.3, 11) | 5.1 (3.2, 9.2) |
| Collection to NAT report, acute | 1.5 (1.0, 5.0) | 5.2 (5.0, 5.3) | 4.0 |
| Positive WB result to patient receipt of results | Not collected | 4.0 (0, 14)d,e | Not collected |
| Acute result in laboratory to patient receipt of results | Not collected | 14 (12 15)f | Not collected |
Abbreviations: HIV, human immunodeficiency virus; IA, immunoassay; NAT, nucleic acid test; WB, Western blot.
a Indicates an established HIV infection.
b Western blot results came back from the reference laboratory after NAT results, a median of 6.7 days after specimens were received in the hospital laboratory.
c Western blot results came back from the reference laboratory 4.4 days after the specimen was received in the hospital laboratory.
d Massachusetts General Hospital provided the median, minimum, and maximum days from test result to client receipt of results based on medical record documentation.
e For 75 specimens from persons with known infection and 24 research participants without a medical record number, time was not collected.
f Represents 2 specimens from 1 individual who got results back from the first specimen in 12 days.
Laboratory Cost per Human Immunodeficiency Virus Infection Identified
The laboratory cost per infection ranged from $1054 (MGH) to $1521 (MUSC) (Table 3). Human immunodeficiency virus screening tests accounted for the majority of HIV testing costs. Including labor costs, supplemental antibody testing using Multispot at Harborview was more costly than HIV-1 WB, but it was less costly than HIV-1 WB testing at the reference laboratories used by MGH and MUSC. At Harborview, WB strips were cut longitudinally to decrease cost, an off-label procedure that was validated at that laboratory. All hospitals incurred NAT costs for persons tested with at least 1 screening assay who did not have negative or indeterminate WB results. The laboratory testing cost per specimen screened was $6.66 (MGH), $6.93 (MUSC), and $9.73 (HMC).
Table 3.
Laboratory Testing and Labor Costs and Total Cost per HIV Infection Detected in 3 US Hospital Laboratories Using Fourth-Generation HIV Immunoassays
| Hospital and Test Type | Test Cost per Specimen ($)a | Adjusted Test Cost per Specimen ($)b | Labor Cost per Specimen ($)c | Reference Laboratory Fee ($)d | Total Specimens Testede | Total Costs ($)f | Cost per Infection Detected ($) |
|---|---|---|---|---|---|---|---|
| HMC | |||||||
| ARCHITECT | 6.19 | 7.17 | 1.27 | 14,819 | 125 062.88 | ||
| HIV-1 WB | 17.80 | 23.83 | 7.02 | 153 | 4720.79 | ||
| Multispot | 19.44 | 20.68 | 23.87 | 154 | 6861.31 | ||
| RealTime RNA | 43.99 | 48.95 | 1.46 | 82g | 4134.22 | ||
| HIV-2 NAT | 75.50 | 132.13 | 23.41 | 3 | 466.59 | ||
| Total costs | 141 245.79 | 1129.97h | |||||
| MGH | |||||||
| ARCHITECT | 4.20 | 4.78 | 1.10 | 20 062 | 118 065.64 | ||
| HIV-1 WB | 58.06 | 142 | 8244.52 | ||||
| HIV-2 EIA | 56.50 | 20 | 1130.00 | ||||
| HIV I/II test | 101.69 | 2 | 203.38 | ||||
| COBAS RNA | 43.18 | 49.35 | 3.44 | 77g | 4064.16 | ||
| Total costs | 131 707.69 | 1053.66h | |||||
| MUSC | |||||||
| GS Combo | 3.37 | 4.42 | 1.66 | 3774 | 22 969.30 | ||
| HIV-1 WB | 66.24 | 21 | 1391.04 | ||||
| RealTime RNA | 42.52 | 53.09 | 1.18 | 22g | 1194.08 | ||
| HIV-2 EIA | 72.90 | 4 | 291.60 | ||||
| Total costs | 25 846.01 | 1520.35i | |||||
Abbreviations: EIA, enzyme immunoassay; HIV, human immunodeficiency virus; HMC, Harborview Medical Center; MGH, Massachusetts General Hospital; MUSC, Medical University of South Carolina; NAT, nucleic acid test; WB, Western blot.
a Total kit cost divided by the number of tests per kit.
b Total kit cost plus cost of controls not provided in kit divided by number of tests not used as controls.
c The product of labor hours per specimen (data not shown) and labor costs per hour (Bureau of Labor Statistics data for medical and clinical laboratory technologists: $46.81 (HMC); $49.38 (MGH); $21.60 (MUSC).
d Amount charged to the hospital, which included testing and labor costs.
e Total specimens were multiplied by 3 if initial test reactive, and testing was repeated in duplicate.
f Sum of the laboratory testing costs (adjusted reagent cost per specimen × the total number of specimens) and labor costs (labor cost per specimen × the total number of specimens). Total costs are based on testing and labor costs, which were carried to additional digits not reflected in this table.
g Human immunodeficiency virus-1 NAT costs were included if at least 1 diagnostic test was done in conjunction with NAT.
h Total costs for all tests divided by 125 HIV-1- or HIV-2-positive specimens.
i Total costs for all tests divided by 17 HIV-1- or HIV-2-positive specimens.
DISCUSSION
Acute HIV infections were detected in all 3 hospitals using fourth-generation immunoassay screening and constituted from 1.6% to 5.9% of HIV infections identified. The identification of these infections would have been delayed or missed without supplemental testing including a NAT. Human immunodeficiency virus-1 NAT was indicated in 0.2% or fewer of specimens tested in all hospitals, a number that is low, in part, because of the high specificity of the fourth-generation assay. The number of acute infections may have been higher if NAT had been conducted on all specimens with repeatedly reactive fourth-generation results and negative or indeterminate WB results. Hospital laboratories may benefit from an FDA-approved diagnostic claim on a viral load test so that they can automatically conduct the test when discordant test results occur, without the need for a physician request. Conducting an independent validation of an existing HIV-1 viral load test for diagnostic testing is cost-prohibitive, and institutions are unlikely to make this investment [15, 26].
Providing timely HIV test results to the provider and patient can facilitate appropriate medical care and reduce transmissions, which are more likely during the highly infectious acute period [27, 28]. Most specimens were tested with an initial fourth-generation assay within 1 day of specimen collection. Random access automated tests can be conducted without batching, because specimens are received in the laboratory. One day elapsed from specimen receipt in the hospital laboratory to WB result at HMC, which had Multispot and WB onsite, compared with 5 days in the hospitals using a reference laboratory for HIV-1 WB testing. The CDC/APHL algorithm does not recommend additional testing after a repeatedly reactive fourth-generation immunoassay result and a reactive HIV-1 antibody result on the HIV-1/HIV-2 differentiation test [13].
During this study, in 2013, the Multispot was approved as a supplemental test by the FDA, making it easier for laboratories within and outside of hospitals to conduct supplemental testing onsite. Because HMC collected plasma initially and had conducted a validation to use their viral load test as a diagnostic test, they were able to conduct a reflexive NAT on the original specimen after a negative or indeterminate WB result. This may explain the shorter time to NAT results for persons with acute infection at HMC. At MGH and MUSC, for those with acute infection, NAT results were available from the hospital laboratory before WB results were provided by the reference laboratory.
There are factors to consider when using reflexive NATs from the specimen submitted for serological testing, including different requirements for specimen handling and volume. In addition, the analytical sensitivity of NATs is very high, so there is the possibility of sample cross-contamination with HIV RNA when specimens are handled in a central specimen processing area and tested first on immunoassay instruments. One option to overcome the latter is to separate the specimen initially to retain a separate tube to be used for NAT, as needed [29]. In addition to identifying delays in the testing process, testing programs should examine delays in the delivery of results. We were not able to evaluate why the person with an acute infection at MGH did not receive his or her test results until 12 days after they were available in the laboratory.
Although over 38 000 total tests were conducted, 2 HIV-2-positive specimens were identified. Identification of HIV-2 infection can be clinically important because some antiretroviral drugs used to treat HIV-1 are not effective for treatment of HIV-2 [30, 31]. When HIV-1 WBs are used after a repeatedly reactive fourth-generation assay result, and they are not positive, the CDC recommends using an HIV-1 NAT [32]. Additional HIV-2 testing is conducted if the HIV-1 NAT is negative. However, when the HIV-1/HIV-2 differentiation test is used as the initial supplemental test and it is HIV-2 positive, further testing is not recommended, because there is no FDA-approved HIV-2 NAT. At HMC, which used the HIV-1/HIV-2 differentiation test and the HIV-1 WB for supplemental testing, the differentiation assay cost more, especially when accounting for labor costs per specimen. In that case, the WB was run in a batch of approximately 15 specimens, so costs were spread over multiple specimens. Multispot is not batched, but it has a faster turnaround time, so the additional costs can be considered the price for obtaining a faster result. In addition, at HMC, WB strips were cut longitudinally, an off-label practice that reduced WB costs at that site. It is not known how the cost of the Geenius HIV-1/2 Supplemental Assay (Bio-Rad, Redmond, WA), the new FDA-approved HIV-1/HIV-2 differentiation test with automated reader, will compare [33]. One study examining testing costs in 17 laboratories found WB costs to be higher than Multispot costs even without accounting for the HIV-2 testing needed when HIV-1 WBs were used [20].
Screening assay costs drove laboratory-testing costs because most specimens did not need supplemental testing. Costs may decrease with additional fourth-generation assays in the US marketplace. This cost evaluation did not examine the cost savings of testing for multiple analytes, such as viral hepatitis, on the ARCHITECT platform. We also did not evaluate nonlaboratory components such as labor costs for specimen collection or release of results. Test costs change frequently, so costs in this study represent a snapshot in time. Costs are dependent on factors such as testing volume, whether the same manufacturer provides confirmatory test kits, and the frequency of running quality controls. As expected, cost per reported positive test result and cost per specimen screened were lowest in the hospital with the highest volume of tests, MGH. All hospitals incurred costs for NAT ordered at physician discretion for specimens other than those that were repeatedly reactive by the screening test and negative or indeterminate using the supplemental antibody test, likely in persons clinically suspected of being infected. However, they constituted a small proportion of total testing costs. Adjusted NAT costs per specimen were similar across hospitals. Overall, the cost per specimen was similar to what has been reported in public health laboratories using comparable analyses ($5 to $11), but cost per positive result was lower in public health laboratories ($208 to $336 per positive) than in hospitals, likely due to the higher proportion of infected persons tested by these laboratories.
The small number of hospitals and the variations in the testing algorithms are a study limitation. Turnaround time and cost may not be representative of US hospitals, and comparisons between hospitals are limited. The ADVIA Centaur HIV Ag/Ab Combo assay and the BioPlex 2200 HIV Ag-Ab assay were approved by the FDA in 2015 after this study was completed. Furthermore, analyses were conducted by specimen and not by person, which may have affected outcomes such as proportion of acute infections or HIV-2 infections if persons were tested more than once. In addition, labor hours were estimated from the literature and not measured directly at the hospitals, which may have impacted cost estimates.
CONCLUSIONS
Fourth-generation assays identified acute infections in all hospitals. With the use of these assays, there is a need to expedite the provision of results, because persons with acute infection can be highly infectious. Onsite supplemental rapid testing using an HIV-1/HIV-2 differentiation test could be used to accelerate the provision of results for hospitals that currently rely on reference laboratories and may reduce costs. Hospitals should also evaluate whether testing using plasma is feasible so that a separate specimen is not needed for quantitative NAT. Finally, hospital laboratories may benefit from a diagnostic claim for quantitative NATs, which would allow them to automatically conduct NAT on specimens that are reactive for antigen, but not antibody, instead of relying on physician ordering.
Acknowledgments
We acknowledge the work of the following individuals: Dr. J. Michael Kilby, former study Principal Investigator (Medical University of South Carolina Hospital); Dr. Noah G. Hoffman, Cathy Griffin, and Kim Tanka-Hill, (University of Washington, Department of Laboratory Medicine) for data extraction and report generation; Graham McGrath (Massachusetts General Hospital); Joyce Foster and Angela Rourk (Medical University of South Carolina Hospital); Jeanne Day, Amy Flynn, and Kristin Johnson (John Snow, Inc.); Emeka Oraka (ICF International); and Kristina Bowles (Centers for Disease Control and Prevention). Ramos et al presented on a subset of testing outcomes described in this paper but did not report on outcomes such as turnaround time and cost.
Disclaimer. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the Centers for Disease Control and Prevention or the US Department of Health and Human Services.
Financial support. This study was funded by the Centers for Disease Control and Prevention through a contract with John Snow, Inc.
Potential conflicts of interest. R. W. C. reports an advisory board membership with Roche Molecular Diagnostics and research support from Abbott Molecular, Inc. F. S. N. reports research support, advisory board, and honoraria from Abbott Molecular, Roche Diagnostics, and Ortho Diagnostics. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
References
- 1.Daar ES, Moudgil T, Meyer RD, Ho DD. Transient high levels of viremia in patients with primary human immunodeficiency virus type 1 infection. N Engl J Med 1991; 324:961–4. [DOI] [PubMed] [Google Scholar]
- 2.Morrison CS, Demers K, Kwok C et al. Plasma and cervical viral loads among Ugandan and Zimbabwean women during acute and early HIV-1 infection. AIDS 2010; 24:573–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pilcher CD, Joaki G, Hoffman IF et al. Amplified transmission of HIV-1: comparison of HIV-1 concentrations in semen and blood during acute and chronic infection. AIDS 2007; 21:1723–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dowshen N, Pierce VM, Zanno A et al. Acute HIV infection in a critically ill 15-year-old male. Pediatrics 2013; 131:e959–63. [DOI] [PubMed] [Google Scholar]
- 5.Masciotra S, Luo W, Youngpairoj AS et al. Performance of the Alere Determine HIV-1/2 Ag/Ab Combo Rapid Test with specimens from HIV-1 seroconverters from the US and HIV-2 infected individuals from Ivory Coast. J Clin Virol 2013; 58(Suppl 1):e54–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hogan CM, Degruttola V, Sun X et al. The setpoint study (ACTG A5217): effect of immediate versus deferred antiretroviral therapy on virologic set point in recently HIV-1-infected individuals. J Infect Dis 2012; 205:87–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Panel on Antiretroviral Guidelines for Adults and Adolescents. Guidelines for the use of antiretroviral agents in HIV-1-infected adults and adolescents. Department of Health and Human Services. Available at: http://www.aidsinfo.nih.gov/ContentFiles/AdultandAdolescentGL.pdf. Accessed 31 July 2014.
- 8.Saez-Cirion A, Bacchus C, Hocqueloux L et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI Study. PLoS Pathog 2013; 9:e1003211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hocqueloux L, Saez-Cirion A, Rouzioux C. Immunovirologic control 24 months after interruption of antiretroviral therapy initiated close to HIV seroconversion. JAMA Intern Med 2013; 173:475–6. [DOI] [PubMed] [Google Scholar]
- 10.Cohen MS, Chen YQ, McCauley M et al. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med 2011; 365:493–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.National Institute of Allergy and Infectious Diseases. Starting antiretroviral treatment early improves outcomes for HIV-infected individuals. In: National Institutes of Health: News Releases, 2015. Available at: http://www.niaid.nih.gov/news/newsreleases/2015/Pages/START.aspx. Accessed 1 August 2015. [Google Scholar]
- 12.The INSIGHT START Study Group. Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med 2015; 373:795–807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Centers for Disease Control and Prevention and Association of Public Health Laboratories. Laboratory Testing for the Diagnosis of HIV Infection: Updated Recommendations. Available at: http://www.cdc.gov/hiv/pdf/hivtestingalgorithmrecommendation-final.pdf. Accessed 1 August 2015.
- 14.Branson BM. HIV testing updates and challenges: when regulatory caution and public healthimperatives collide. Curr HIV/AIDS Rep 2015; 12:117–26. [DOI] [PubMed] [Google Scholar]
- 15.Centers for Medicare and Medicaid Services. Standard: establishment and verification of performance specifications. 42 CFR 5 § 493.1253., 2008. Available at: https://www.gpo.gov/fdsys/pkg/CFR-2009-title42-vol5/pdf/CFR-2009-title42-vol5-sec493-1252.pdf. Accessed 1 August 2015.
- 16.Branson BM, Handsfield HH, Lampe MA et al. Revised recommendations for HIV testing of adults, adolescents, and pregnant women in health-care settings. MMWR Recomm Rep 2006; 55:1–17; quiz CE1–4. [PubMed] [Google Scholar]
- 17.U.S. Preventive Services Task Force. Final Recommendation Statement, Human Immunodeficiency Virus (HIV) Infection: Screening. April 2013 ed, 2013. Available at: http://www.uspreventiveservicestaskforce.org/Page/Document/RecommendationStatementFinal/human-immunodeficiency-virus-hiv-infection-screening. Accessed 1 August 2015.
- 18.Ramos EM, Harb S, Dragavon J et al. Performance of an alternative HIV diagnostic algorithm using the ARCHITECT HIV Ag/Ab Combo assay and potential utility of sample-to-cutoff ratio to discriminate primary from established infection. J Clin Virol 2013; 58(Suppl 1):e38–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ramos EM, Harb S, Dragavon J, Coombs RW. Clinical performance of the Multispot HIV-1/HIV-2 rapid test to correctly differentiate HIV-2 from HIV-1 infection in screening algorithms using third and fourth generation assays and to identify cross reactivity with the HIV-1 Western Blot. J Clin Virol 2013; 58:e104–e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hutchinson AB, Ethridge SF, Wesolowski LG et al. Costs and outcomes of laboratory diagnostic algorithms for the detection of HIV. J Clin Virol 2013; 58(Suppl 1):e2–7. [DOI] [PubMed] [Google Scholar]
- 21.O'Brien TR, George JR, Epstein JS et al. Testing for antibodies to human immunodeficiency virus type 2 in the United States. MMWR Recomm Rep 1992; 41:1–9. [PubMed] [Google Scholar]
- 22.Chang M, Gottlieb GS, Dragavon JA et al. Validation for clinical use of a novel HIV-2 plasma RNA viral load assay using the Abbott m2000 platform. J Clin Virol 2012; 55:128–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Amendola A, Coen S, Belladonna S et al. Improving clinical laboratory efficiency: a time-motion evaluation of the Abbott m2000 RealTime and Roche COBAS AmpliPrep/COBAS TaqMan PCR systems for the simultaneous quantitation of HIV-1 RNA and HCV RNA. Clin Chem Lab Med 2011; 49:1283–8. [DOI] [PubMed] [Google Scholar]
- 24.Nasrullah M, Wesolowski LG, Ethridge SF et al. Acute infections, cost and time to reporting of HIV test results in U.S. State Public Health Laboratories. In: Conference on Retroviruses and Opportunistic Infections Boston: Massachusetts, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.United States Department of Labor. Occupational Employment Statistics. Available at: http://www.bls.gov/oes/tables.htm. Accessed 1 August 2015.
- 26.Association of Public Health Laboratories/Centers for Disease Control and Prevention STD Steering Committee. General Checklist for Establishment of Performance Specifications for Tests Not FDA-Cleared or Approved. Available at: http://www.aphl.org/aphlprograms/infectious/std/Documents/ID_2009Oct15_Checklist-STD-Performance-Specs.pdf. Accessed 1 August 2015.
- 27.Wawer MJ, Gray RH, Sewankambo NK et al. Rates of HIV-1 transmission per coital act, by stage of HIV-1 infection, in Rakai, Uganda. J Infect Dis 2005; 191:1403–9. [DOI] [PubMed] [Google Scholar]
- 28.Brenner BG, Roger M, Routy JP et al. High rates of forward transmission events after acute/early HIV-1 infection. J Infect Dis 2007; 195:951–9. [DOI] [PubMed] [Google Scholar]
- 29.CLSI. Criteria for Laboratory Testing and Diagnosis of Human Immunodeficiency Virus Infection; Approved Guideline. CLSI document M53A Wayne, PA: Clinical and Laboratory Standards Institute; 2011. [Google Scholar]
- 30.Ntemgwa ML, d'Aquin Toni T, Brenner BG et al. Antiretroviral drug resistance in human immunodeficiency virus type 2. Antimicrob Agents Chemother 2009; 53:3611–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hizi A, Tal R, Shaharabany M et al. Specific inhibition of the reverse transcriptase of human immunodeficiency virus type 1 and the chimeric enzymes of human immunodeficiency virus type 1 and type 2 by nonnucleoside inhibitors. Antimicrob Agents Chemother 1993; 37:1037–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Centers for Disease Control and Prevention. HIV-2 infection surveillance--United States, 1987–2009. MMWR Morb Mortal Wkly Rep 2011; 60:985–8. [PubMed] [Google Scholar]
- 33.Herssens N, Beelaert G, Fransen K. Discriminatory capacity between HIV-1 and HIV-2 of the new rapid confirmation assay Geenius. J Virol Methods 2014; 208:11–5. [DOI] [PubMed] [Google Scholar]