Abstract
A randomized, multi-center study of adult cigarette smokers switched to tobacco-heating cigarettes, snus or ultra-low machine yield tobacco-burning cigarettes (50/group) was conducted, and subjects’ experience with the products was followed for 24 weeks. Differences in biomarkers of tobacco exposure between smokers and never smokers at baseline and among groups relative to each other and over time were assessed. Results indicated reduced exposure to many potentially harmful constituents found in cigarette smoke following product switching. Findings support differences in exposure from the use of various tobacco products and are relevant to the understanding of a risk continuum among tobacco products (ClinicalTrials.gov Identifier: NCT02061917).
Keywords: Mouth level exposure, multi-center randomized study, risk continuum, tobacco and tobacco smoke constituents, yield-in-use
Introduction
Cigarette smoking is a leading cause of preventable deaths in the USA. Cigarette smoking significantly increases the risk of developing lung cancer, heart disease, chronic bronchitis, emphysema and other serious diseases and adverse health conditions (Adhikari et al., 2008). Quitting cigarette smoking significantly reduces the risk for cigarette smoking attributable diseases and adverse health conditions. For adult cigarette smokers who are unwilling or unable to quit, health risks might be lowered by reducing exposure to potentially harmful cigarette smoke constituents via switching to alternate, potentially-reduced exposure tobacco products. This concept is the basis for the principle of harm reduction and the existence of a risk continuum across a range of tobacco products (Nutt et al., 2014; Zeller et al., 2009).
With the reduction of exposure to tobacco toxicants, reduction in the risk of disease associated with tobacco-consumption is achievable (Stratton et al., 2001). This is supported by epidemiology data that have indicated that reducing exposure reduces the subsequent risk of smoking-related diseases (Lee, 2013; USDHHS, 1989, 2004). Understanding the differences in toxicant exposure between cigarettes and other tobacco products, including following a switch from cigarettes to alternate tobacco products, is relevant to the discussion of modified risk tobacco products under the United States Family Smoking Prevention and Tobacco Control Act (US Congress, 2009).
Measuring the levels of tobacco-related biomarkers of exposure is an appropriate metric for exposure assessment, as biomarkers indicate that both contact and uptake into the body have occurred (Sexton, 2006). Additionally, these biomarkers account for variations in smoking behaviors among consumers (e.g. puff number, puff volume, puff frequency), unlike the easily quantified and standardized machine-generated mainstream smoke constituent yields (Borgerding & Klus, 2005).
A randomized, multi-center study of adult cigarette smokers switched to tobacco-heating cigarettes, snus or ultra-low machine yield tobacco-burning cigarettes was conducted (ClinicalTrials.gov Identifier: NCT02061917) (Ogden et al., 2015b). Subjects’ experience with the randomized product was followed for 24 weeks. A group of never smokers was included for baseline comparisons. This paper documents the differences in biomarkers of tobacco exposure among cigarette smokers and never smokers at baseline, and the changes in biomarkers in the three groups switched to alternate tobacco products relative to each other and over time. Additionally, mouth level exposure (MLE) analyses [e.g. yield-in-use (YIU)] were used to evaluate the potential for clinical confinement effects, compensatory smoking and nicotine yield versus nicotine uptake in subjects switched to the ultra-low machine yield tobacco-burning cigarettes. Detailed study methodology and subject disposition, as well as study results for biomarkers of biological effect, are presented elsewhere (Ogden et al., 2015a,b).
Methods
Study conduct
The study was conducted in accordance with Good Clinical Practice (ICH, 1996) between February and November 2007. Study and subject materials including protocol, protocol amendments, informed consent forms, study product information and recruitment literature were reviewed and approved by Independent Investigational Review Board, Inc. (currently Shulman Associates IRB, Inc., Fort Lauderdale, FL). Written informed consent was obtained from each subject before protocol-specific procedures were carried out (Ogden et al., 2015b).
This study was conducted at five clinical research units (CRUs) in the USA managed by Covance Early Clinical Development (Madison, WI). Subjects were adult cigarette smokers randomly switched to a tobacco-heating cigarette (Eclipse, non-menthol or menthol variety depending on subject preference), snus (Camel SNUS, subject choice of Frost, Spice, and Mellow varieties) or an ultra-low machine yield tobacco-burning cigarette (5 mg Cambridge Filter Method “tar”; Camel or Salem, non-menthol and menthol, respectively, depending on subject preference). A fourth group of never smokers was included for baseline comparisons. For subjects switched to alternate tobacco products, experience with the products was followed for 24 weeks. Ogden et al. (2015b) have reported additional details of the study design and methodology.
Analytical methodology and statistical analysis
Smokers were confined to the CRU for three 24-h periods at weeks 0, 12, and 24 with sample collection; never smokers were confined to the CRU for 24 h at week 0 with sample collection. Subjects were provided with standardized meals during confinement periods. Samples for 24-h urine, spot urine and fasting blood were collected and analyzed for biomarkers of exposure including those for nicotine, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK), aromatic amines, polycyclic aromatic hydrocarbons (PAHs), acrylamide, 1,3-butadiene, crotonaldehyde, acrolein, benzene and carbon monoxide (Tables 1 and 2).
Table 1. Urinary biomarkers of exposure.
| Constituent | Biomarker | Abbreviation | Method | LOD a | LOQ a | Analytical lab | |||
|---|---|---|---|---|---|---|---|---|---|
| Nicotine | Unconjugated nicotine | NIC-U | LC–MS/MS (Meger et al., 2002) | 18.2 | 55 | RJRT | |||
| Unconjugated cotinine | COT-U | 1.12 | 3.38 | ||||||
| Unconjugated trans-3′-hydroxycotinine | OHCOT-U | 1.57 | 4.77 | ||||||
| Nicotine-N-glucuronide | NIC-G | 8.36 | 25.3 | ||||||
| Cotinine-N-glucuronide | COT-G | 1.53 | 4.63 | ||||||
| trans-3′-Hydroxycotinine-O-glucuronide | OHCOT-G | 3.49 | 10.6 | ||||||
| Cotinine-N-oxide | CNO | 1.2 | 3.65 | ||||||
| Nicotine-N-oxide | NNO | 4.88 | 14.78 | ||||||
| Norcotinine | NCOT | 3.9 | 11.8 | ||||||
| Nornicotine | NNIC | 2.21 | 6.71 | ||||||
| NICEq b | NICEq | 23.7 | 71.7 | ||||||
| NNK c | NNAL (free) | NNAL | LC–MS/MS(Byrd & Ogden, 2003) | 0.007 | 0.02 | RJRT | |||
| NNAL total | NNAL + NNAL-G | ||||||||
| NNAL conjugated | NNAL-G | ||||||||
|
Aromatic amines | |||||||||
| Toluidine | o-Toluidine | o-T | GC–MS(Riedel et al., 2006) | 0.83 ng/L | 10 ng/L | ABF | |||
| 2-Aminonaphthalene | 2-Aminonaphthalene | 2-AN | 0.57 ng/L | 1.7 ng/L | |||||
| Aminobiphenyl | 3-Aminobiphenyl | 3-ABP | 0.45 ng/L | 1.3 ng/L | |||||
| 4-Aminobiphenyl | 4-ABP | 0.51 ng/L | 1.5 ng/L | ||||||
|
Polycyclic aromatic hydrocarbons |
|||||||||
| Pyrene | Hydroxypyrene | 1-OHP | LC-fluorescence(Scherer et al. 2000a) | 0.003 | 0.01 | ABF | |||
| Naphthalene | 1-Naphthol | 1-Naphthol | 0.31 | 1 | |||||
| 2-Naphthol | 2-Naphthol | 0.33 | 1 | ||||||
| Fluorene | Hydroxyfluorene | 2-OHF | 0.08 | 0.25 | |||||
| Phenanthrene | Hydroxyphenanthrene | 1-/9-OHPh; 2-/3-OHPh | 0.01 | 0.02 | |||||
| Acrylamide | N-Acetyl-S-(2-carbamoylethyl)cysteine | AAMA | LC–MS/MS(Urban et al., 2006) | 3 | 10 | ABF | |||
| N-Acetyl-S-(2-hydroxy-2-carbamoylethyl)cysteine | GAMA | 0.3 | 1 | ||||||
| 1,3-Butadiene | Monohydroxybutenylmercapturic acids | MHBMA | LC-MS/MS(Urban et al., 2003) | 0.4 | 1.1 | ||||
| 1,2-Dihydroxybutylmercapturic acids | DHBMA | 5.1 | 15.4 | ||||||
| Crotonaldehyde | Hydroxyl-1-methylpropyl-mercapturic acid | HMPMA | LC–MS/MS(Scherer et al., 2010) | 20 | 52 | ||||
| Acrolein | 3-Hydroxypropylmercapturic acid | HPMA | LC-MS/MS(Mascher et al., 2001; Scherer et al., 2000b) | 6.2 | 20.4 | ||||
| Benzene | S-Phenylmercapturic acid | SPMA | LC–MS/MS(Scherer et al., 2001) | 0.03 | 0.09 | ||||
| “Mutagens” | Urine mutagenicity | UM | Ames, TA98 and YG1024 + S9(Kado et al., 1983) | – | – | CLL | |||
ABF, Analytisch-Biologisches Forschungslabor (Munich, Germany); CLL, Covance Laboratory Ltd. (Harrogate, UK); GC–MS, gas chromatography–mass spectrometry; LC, liquid chromatography; LC–MS/MS, liquid chromatography–tandem mass spectrometry; LOD, limit of detection; LOQ, limit of quantitation; RJRT, R.J. Reynolds Tobacco Company (Winston-Salem, NC); –, not available.
ang/mL except where noted.
bNIC-U and the 9 metabolites are converted to molar NIC-U equivalents and summed.
cNNK, 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone.
Table 2. Blood biomarkers of exposure.
| Constituent | Biomarker | Abbreviation | Method | LOD | LOQ | Analytical lab |
|---|---|---|---|---|---|---|
| Nicotine | Serum cotinine | Cotinine | LC–MS/MS(Byrd et al. 2005) | 6 nmol/L | 20 nmol/L | RJRT |
| Carbon monoxide | Carboxyhemoglobin | COHb | CO-oximeter a | 0.3% | – | CCLS |
| 4-Aminobiphenyl | 4-Aminobiphenyl-hemoglobin adducts | 4-ABP-Hb | GC–MS(Lewalter & Gries, 2000; Richter et al., 2001) | 2.3 pg/g Hb | 6.9 pg/g Hb | ABF |
ABF, Analytisch-Biologisches Forschungslabor (Munich, Germany); CCLS, Covance Central Laboratory Services (Indianapolis, IN); RJRT, R.J. Reynolds Tobacco Company (Winston-Salem, NC); LC–MS/MS, liquid chromatography–tandem mass spectrometry; GC–MS, gas chromatography–mass spectrometry; LOD, limit of detection; LOQ, limit of quantitation; –, not available.
CCLS methodology. Anticoagulated whole blood was aspirated into an oximeter. Sample was mixed with diluent, hemolyzed via surfactant, and brought to temperature in the cuvette. Absorbancies at 6 different wavelengths were measured and used by the oximeter to generate total hemoglobin and percent COHb.
Blood and urine samples were collected according to protocol-specified procedures. For consistency, samples at weeks 0, 12, and 24 were generally collected at approximately the same time of day. The 24-h urine collection period began after the spot urine collection, with the spot urine taken at the beginning of the study visit when the subjects checked in to the CRU (between 2:00 and 5:00 pm). Urine collections were aliquoted into tubes prepared by Covance Clinical Laboratory Services (CCLS, Indianapolis, IN) and were shipped to CCLS for storage at −70 °C until completion of the sample collection phase. Urine samples were then shipped in batch to the appropriate analytical laboratory for analysis. Results presented here are based on the 24-h collection. Blood samples were collected on the morning following subject check-in. Tubes prepared by CCLS were utilized, and samples were shipped to CCLS (per CCLS specifications), stored until the end of the study, and then shipped to the appropriate analytical laboratory for analysis.
The intent-to-treat sample was used for week 0 analyses and included all randomized subjects in the groups to which they were assigned, regardless of adherence with the compliance criteria, deviation from protocol, and subsequent withdrawal from the study. Sample sizes for the intent-to-treat sample were: 44 switched to tobacco-heating cigarettes, 43 switched to snus and 44 switched to ultra-low machine yield tobacco-burning cigarettes; 32 never smokers were available for week 0 analysis. The per-protocol sample was used for change-from-baseline analyses and product group comparisons at weeks 12 and 24, and was defined by mean cumulative compliance greater than 50% (i.e. percent of assigned product used out of total tobacco and nicotine-containing products used) over the 24 study weeks. Sample sizes for the per-protocol sample were: 33 switched to tobacco-heating cigarettes, 20 switched to snus and 35 switched to ultra-low machine yield tobacco-burning cigarettes (Ogden et al., 2015b).
Statistical analysis of all endpoints included descriptive statistics at each time point, with week 0 data stratified by smokers and never smokers, and smoker subject data further stratified into product groups (i.e. tobacco-heating cigarettes, snus and ultra-low machine yield tobacco-burning cigarettes). For all biomarkers, any value reported as less than the limit of detection (LOD) or less than the limit of quantitation (LOQ) was adjusted to equal one-half of the LOD or LOQ before analysis of the data. Mean levels of biomarkers for smokers and never smokers at week 0 were compared by using a t-test with unequal variances. Changes in biomarkers from baseline (at week 0) were calculated for weeks 12 and 24 and analyzed using a mixed model, treating subject within product group as a random effect. Product group (i.e. tobacco-heating cigarette, snus and ultra-low machine yield tobacco-burning cigarette), week (i.e. weeks 12 and 24) and their interaction were fixed effects in the model. Tests for significant changes from baseline for each group at weeks 12 and 24 were conducted by comparing the mean change from baseline to the value zero (i.e. “0.0”) for each randomization group at each time point with t-tests using the mixed model. Differences in changes from baseline among subjects randomized to different products were compared with contrast tests of randomization group by week interaction means using the mixed model. No adjustment for covariates was performed and no adjustment for multiple comparisons was made. Dropouts were not replaced and missing data were not imputed. p < 0.05 was required for statistical significance in all comparisons.
The random subject variance component was 0 or negative for some urinary biomarkers, as modeling change from baseline already accounted for a large part of variation among subjects in some cases; therefore, the random subject term was removed from the models for those affected urinary biomarkers only. Residuals and normality of the data were examined for the fitted models. The normality assumption was not met for some biomarkers, but no change was made from planned analyses due to the relative robustness of the applied statistical model. For some biomarkers, residual plots showed some extreme data points whose standardized residuals were out of the range of (−3, 3), but no observations were excluded from analyses. All calculations were performed using SAS® version 9.1 (Cary, NC).
MLE analyses
Non-invasive means to estimate MLE in tobacco-burning cigarette smokers can be conducted by assessing the amount of smoke that passes through the filter of a cigarette (i.e. YIU) (Nelson et al., 2011). In the current study, three topics were explored with MLE analyses: potential confinement effects (i.e. any tendency for smokers to smoke more or less intensively prior to confinement versus in-clinic); compensatory smoking (i.e. any tendency for smokers switched to a lower machine-generated yield product to smoke more or less intensively) and the relationship between nicotine yield measured by YIU and nicotine uptake measured by total nicotine equivalents (NICEq).
Used cigarette butts were collected for the 24-h period prior to each confinement check-in and for the 24 h of each confinement. Cigarette butts were collected from all smokers at week 0 and from the ultra-low machine yield tobacco-burning cigarette group subjects at weeks 12 and 24. Clinical staff counted and recorded the number of cigarette butts for each subject, visually assessed them for brand conformity, and measured and recorded the length of each butt to the nearest millimeter. Any butts determined to have been from a cigarette brand other than the subjects’ usual brand at week 0 or the protocol-specified brand at weeks 12 and 24 were counted, recorded as a product deviation, and discarded (i.e. not measured, cut or pooled with the remaining tips). Char line, a measure of smoking intensity, was measured in millimeters as the distance from the tip of the mouth-end of the cigarette to the burn line.
For determination of MLE of “tar” and nicotine (i.e. measures of maximum potential uptake), the clinical staff cut and retained the tip of each cigarette butt 1 cm from the mouth end and discarded the remainder. These cut cigarette tips for each subject were pooled (i.e. for each visit and separately for prior to and within confinement samples) into labeled 60 mL amber glass bottles, frozen at the CRU, and shipped to CCLS for storage (−70 °C) and distribution. Analysis (Arista Laboratories, Richmond, VA) consisted of methanol extraction of groups of cut cigarette tips (n=5 ± 1) from the study subjects as well as calibration cigarettes. The methanol extract was analyzed by UV spectrometry (i.e. UV surrogate of cigarette “tar”) and by gas chromatography (for nicotine). Absorbance and the concentration of nicotine were converted to MLE “tar” and nicotine, respectively, based on calibration curves generated from machine-smoked cigarettes. The methods used were consistent with those reported by Nelson et al. (2011).
Descriptive statistics were generated for char line, MLE “tar” (mg/cigarette, mg/day) and MLE nicotine (mg/cigarette, mg/day) for each time point. Week 0 data were broken out additionally by product group (i.e. tobacco-heating cigarette, snus and ultra-low machine yield tobacco-burning cigarettes). MLE means at week 0 for the assigned product groups were compared using analysis of variance (ANOVA), conducted separately for prior to and within confinement conditions for these five endpoints. In addition, YIU means were compared using a two-way ANOVA with test factors confinement (i.e. prior to and within) and week (i.e. weeks 0, 12 and 24). Confinement effects were tested for each week and changes over time (i.e. week 12 versus week 0, week 24 versus week 0 and week 24 versus week 12) and were tested for each confinement condition using contrasts of least square means. Separately, means for YIU nicotine per day (i.e. nicotine yield) and nicotine uptake (measured by NICEq recovered in urine) were presented for each week, with change from baseline and percent uptake calculated as the ratio of nicotine uptake to nicotine yield per day. p < 0.05 was required for statistical significance in all comparisons. All calculations were performed using SAS® version 9.1.
Results
Urine biomarkers
At baseline, measured levels of urine biomarkers of exposure were statistically significantly higher among cigarette smokers compared with never smokers (Table 3).
Table 3. Urine biomarkers, smokers versus never smokers, intent-to-treat sample, week 0.
| Smokers (n = 125) |
Never smokers (n = 29) |
||||
|---|---|---|---|---|---|
| Constituent/marker | Units per 24 h | Mean | SD | Mean | SD |
| Nicotine | |||||
| NIC-U | mg | 2.88 | 1.94 | 0.04 | 0.03 |
| COT-U | 3.22 | 1.72 | 0.001 | 0.01 | |
| OHCOT-U | 9.63 | 4.79 | 0.007 | 0.02 | |
| NIC-G b | 1.32 | 0.98 | 0.06 | 0.04 | |
| COT-G b | 4.81 | 2.76 | 0.03 | 0.01 | |
| OHCOT-G b | 3.69 | 2.36 | 0.002 | 0.01 | |
| CNO | 0.86 | 0.39 | 0.0004 | 0.002 | |
| NNO | 0.97 | 0.62 | 0.003 | 0.008 | |
| NCOT | 0.11 | 0.05 | 0 | 0 | |
| NNIC | 0.14 | 0.07 | 0.02 | 0.01 | |
| NICEq | 24.7 | 10.3 | 0.17 | 0.06 | |
| NNK | |||||
| NNAL total | ng | 683 a | 434 | 43.5 | 47.7 |
| NNAL-G b | 488 a | 335 | 35.5 | 46.7 | |
| NNAL free | 196 a | 127 | 8.17 | 3.59 | |
| Aromatic amines | |||||
| 3-ABP | ng | 12.5 | 7.53 | 0.59 | 0.90 |
| 4-ABP | 27.1 | 14.0 | 2.53 | 2.07 | |
| 2-AN | 38.5 | 20.6 | 1.19 | 1.62 | |
| o-T | 276 | 115 | 101 | 119 | |
| Polycyclic aromatic hydrocarbons | |||||
| 1-naphthol | μg | 56.2 | 139 | 18.8 | 29.3 |
| 2-naphthol | 29.7 | 15.4 | 5.87 | 3.74 | |
| 2-OHF | 3.74 a | 1.92 | 0.57 | 0.37 | |
| 1-/9-OHPh | 1.01 | 0.51 | 0.25 | 0.14 | |
| 2-/3-OHPh | 0.79 | 0.41 | 0.23 | 0.09 | |
| 1-OHP | 0.48 | 0.28 | 0.21 | 0.15 | |
| Acrylamide | |||||
| AAMA | μg | 349 | 145 | 132 | 73.4 |
| GAMA | 49.9 | 21.4 | 33.9 | 18.8 | |
| 1,3-Butadiene | |||||
| DHBMA | μg | 770 | 275 | 549 | 171 |
| MHBMA | 6.27 | 5.85 | 0.45 | 0.32 | |
| Crotonaldehyde | |||||
| HMPMA | mg | 8.65 | 3.89 | 1.47 | 0.69 |
| Acrolein | |||||
| HPMA | mg | 2.10 | 1.11 | 0.50 | 0.24 |
| Benzene | |||||
| SPMA | μg | 11.9 | 8.34 | 0.83 | 2.06 |
| Urine mutagenicty | |||||
| UM (TA98) | revertants | 1.82 × 107 c | 1.09 × 107 | 4.83 × 105 e | 6.05 × 105 |
| UM (YG1024) | revertants | 8.09 × 107 d | 4.70 × 107 | 4.17 × 106 f | 4.21 × 106 |
p<0.05 for all; see Table 1 for abbreviations. Values are rounded.
n = 124.
mass is representative of aglycon moiety.
n = 120.
n = 121.
n = 27.
n = 28.
Urine biomarker values and percent changes from baseline (week 0) to weeks 12 and 24 among the three product groups are presented in Supplemental Table 1. Quantified pairwise comparisons of changes in biomarkers between product groups in the per-protocol sample are presented in Supplemental Table 3.
Nicotine
Change from usual brand cigarette at baseline resulted in a statistically significantly reduced uptake of nicotine (i.e. NICEq) at weeks 12 and 24 in both the tobacco-heating (i.e. decreased approximately 13%) and the ultra-low machine yield tobacco-burning (i.e. decreased approximately 29%) cigarette groups (Figure 1). Change from usual brand at baseline to snus resulted in a non-statistically significant nicotine reduction of approximately 9% at both time points. In comparing pairs of product groups, the only statistically significant difference was a smaller reduction for the snus group than the ultra-low machine yield tobacco-burning cigarette group at week 12.
Figure 1.
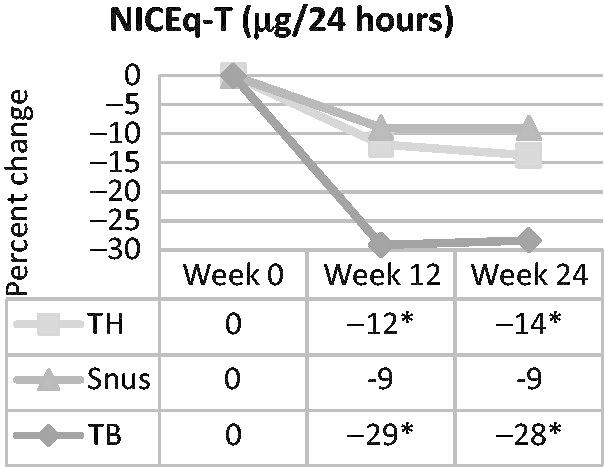
Percent change in urinary NICEq over time in smokers switched to tobacco-heating cigarettes (TH), snus or ultra-low machine yield tobacco-burning cigarettes (TB). *Statistically significant reduction (p<0.05) from week 0.
NNK
The NNK exposure (i.e. as measured by total NNAL) was statistically significantly decreased (i.e. 30–39%) in all three product groups at weeks 12 and 24 compared with week 0 (Figure 2). No statistically significant differential reductions were detected in pairwise product group comparisons.
Figure 2.
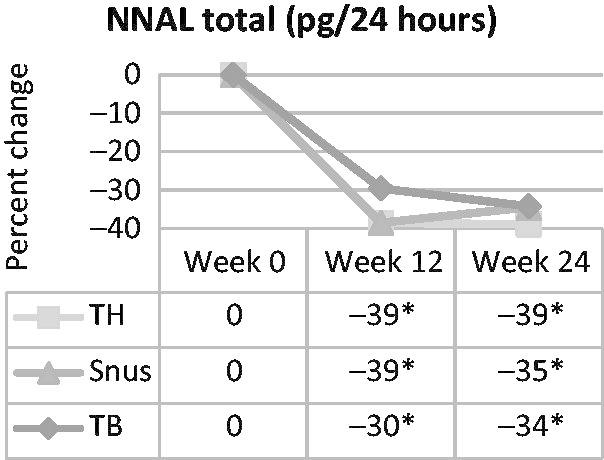
Percent change in urinary total NNAL, a marker of NNK exposure, over time in smokers switched to tobacco-heating cigarettes (TH), snus or ultra-low machine yield tobacco-burning cigarettes (TB). *Statistically significant reduction (p<0.05) from week 0.
Aromatic amines
Biomarkers of all four aromatic amines (i.e. 3-ABP, 4-ABP, 2-AN, and o-T) were statistically significantly reduced at weeks 12 and 24 in all three product groups. Among the three groups and at both time points, 3-ABP exposure in the tobacco-heating cigarette and snus groups showed statistically significantly greater reductions than the ultra-low machine yield tobacco-burning cigarette group, while reductions for the tobacco-heating cigarette and snus groups were not different. Similar patterns were observed for 2-AN and for o-T. For 4-ABP, the overall reduction from baseline was similar to the other aromatic amines. However, in pairwise product group comparisons, the only statistically significant difference was a greater reduction in the tobacco-heating group than the ultra-low machine yield tobacco-burning group at both weeks 12 and 24.
Polycyclic aromatic hydrocarbons
Among the PAH biomarkers, 2-naphthol was statistically significantly and similarly reduced from baseline in the three product groups at both weeks 12 and 24 (i.e. in the range of 25–42%), with no differences detected in the three product group pairwise comparisons. Similar patterns of reductions from baseline at both time points were observed in 2-OHF, 1-/9-OHPh, and 2-/3-OHPh in the tobacco-heating cigarette and snus groups only. Reductions in 2-OHF were statistically significantly greater for the tobacco-heating cigarette group than the ultra-low machine yield tobacco-burning cigarette group at both weeks 12 and 24. For 1-naphthol, no statistically significant differences were observed in any of the product groups at any time point relative to baseline, and there were no differences in any of the product group pairwise comparisons. For 1-OHP, there was a statistically significant increase in the tobacco-heating group at week 12 only (i.e. approximately 63%), but no other statistically significant changes from baseline for any groups at any time point were observed. In product group comparisons, the increase in 1-OHP was statistically significant in the tobacco-heating cigarette group relative to the snus group at weeks 12 and 24, and relative to the ultra-low machine yield tobacco-burning cigarette group at week 12 only.
Acrylamide
Acrylamide exposure, as assessed by AAMA and GAMA, was statistically significantly reduced in all three groups by approximately the same degree (approximately 33–39% for AAMA and approximately 18–32% for GAMA) with no statistically significant differential reductions observed in the three product group pairwise comparisons.
1,3-Butadiene
The 1,3-butadiene exposure, as assessed by MHBMA, was statistically significantly reduced from baseline (approximately 60%) for both the tobacco-heating cigarette and the snus groups, but was not different for the ultra-low machine yield tobacco-burning cigarette group (Figure 3). In pairwise group comparisons, reductions for both the tobacco-heating and snus group were statistically significantly greater than those of the ultra-low machine yield tobacco-burning group but not different from one another. DHBMA (another potential marker of 1,3-butadiene exposure) levels did not change statistically significantly from baseline in any of the three groups, at either time point. In pairwise product group comparisons, the increase in DHBMA from baseline for the tobacco-heating group and the decrease in DHBMA for the snus group resulted in a statistically significant difference between these groups at week 12. Similarly, a statistically significant difference was observed between the tobacco-heating cigarette group and the ultra-low machine yield tobacco-burning cigarette group at week 24.
Figure 3.
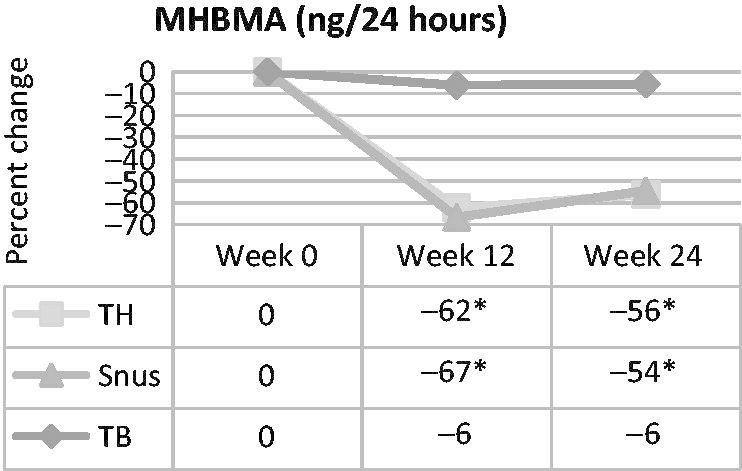
Percent change in urinary MHBMA, a marker of 1,3-butadiene exposure, over time in smokers switched to tobacco-heating cigarettes (TH), snus or ultra-low machine yield tobacco-burning cigarettes (TB). *Statistically significant reduction (p<0.05) from week 0.
Crotonaldehyde
For changes from baseline, HMPMA, a marker of crotonaldehyde exposure, was statistically significantly reduced in the tobacco-heating cigarette and snus groups (approximately 35% and 52%, respectively) at weeks 12 and 24. HMPMA was statistically significantly reduced (approximately 8%) in the ultra-low machine yield tobacco-burning cigarette group at week 12 but not at week 24. In the pairwise product group comparisons, HMPMA reductions in the tobacco-heating cigarette and snus groups were statistically significantly greater than those in the ultra-low machine yield tobacco-burning group, and reductions in the snus group were statistically significantly greater than reductions in the tobacco-heating group at both time points.
Acrolein
Acrolein exposure, as measured by HPMA, increased 20–27% in the tobacco-heating cigarette group, and decreased 41–45% in the snus group at weeks 12 and 24 (Figure 4). HPMA did not change statistically significantly from baseline for the ultra-low machine yield tobacco-burning cigarette group at either time point. HPMA changes from baseline were statistically significantly different for all three group pairwise comparisons at both time points.
Figure 4.
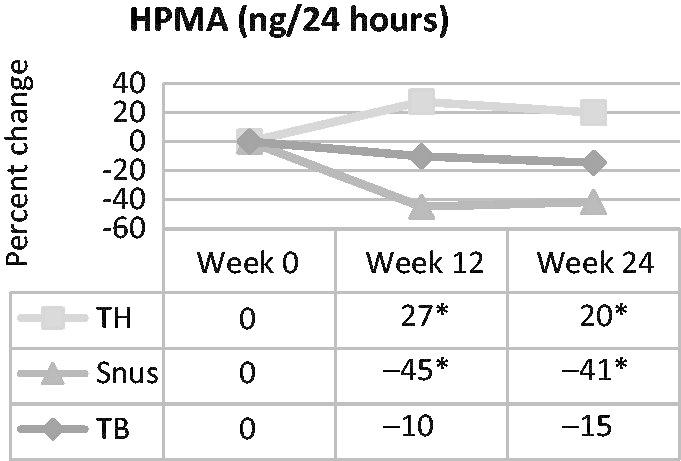
Percent change in urinary HPMA, a marker of acrolein exposure, over time in smokers switched to tobacco-heating cigarettes (TH), snus or ultra-low machine yield tobacco-burning cigarettes (TB). *Statistically significantly different (p<0.05) from week 0.
Benzene
Benzene, as assessed by the biomarker SPMA, was statistically significantly reduced from baseline (approximately 50–57%) for both the tobacco-heating cigarette and snus groups at both weeks 12 and 24, but did not change statistically significantly for the ultra-low machine yield tobacco-burning cigarette group at either time point. In pairwise product group comparisons, reductions in both the tobacco-heating cigarette and the snus groups were statistically significantly greater than those of the ultra-low machine yield tobacco-burning cigarette group. No statistically significant differences between the tobacco-heating cigarette and the snus groups in SPMA reductions were observed.
Urine mutagenicity
The uptake of mutagens, as assessed by the Ames assay with bacterial strains TA98 and YG1024, showed clear and consistent patterns of reduction from baseline in all three product groups and at both weeks 12 and 24. The TA98 and YG1024 count reductions were 37–46% for the tobacco-heating cigarette group, 53–63% for the snus group and 28–33% for the ultra-low machine yield tobacco-burning cigarette group. In pairwise comparisons, the TA98 reductions in the snus group were statistically significantly greater than those of the tobacco-heating cigarette and ultra-low machine yield tobacco-burning cigarette groups at both time points. In YG1024, reductions in the snus group were statistically significantly different from the ultra-low machine yield tobacco-burning group at week 12 only.
Blood biomarkers
Levels of biomarkers of exposure measured in blood, including serum cotinine, percent Carboxyhemoglobin (COHb) and aromatic amine (4-ABP-Hb) adducts, were statistically significantly higher in cigarette smokers compared with never smokers at baseline (Table 4).
Table 4. Blood biomarkers, smokers versus never smokers, intent-to-treat sample, week 0.
| Smokers |
Never smokers |
||||||
|---|---|---|---|---|---|---|---|
| Marker | Units | n | Mean | SD | n | Mean | SD |
| Cotinine | nmol/L | 108 | 1.60 | 0.62 | 27 | 0.004 | 0.002 |
| COHb | % saturation | 130 | 6.10 | 1.94 | 32 | 1.21 | 0.38 |
| 4-ABP-Hb | pg/g Hb | 116 | 63.6 | 29.2 | 31 | 11.9 | 15.1 |
p < 0.05 for all. See Table 2 for abbreviations.
Blood biomarker values and changes from baseline to weeks 12 and 24 among the three groups are presented in Supplemental Table 2. Quantitative findings from product group pairwise comparisons of blood biomarker changes are presented in Supplemental Table 4.
Nicotine (measured as serum cotinine)
In the tobacco-heating cigarette group, no statistically significant differences in serum cotinine were observed at either week 12 or 24 as compared with baseline. In the snus group, no statistically significant difference at week 12 compared with baseline was observed, but a statistically significant increase in serum cotinine was observed at week 24 (i.e. approximately 32%). Changes from baseline indicated a statistically significantly reduced serum cotinine at both weeks 12 and 24 for the ultra-low machine yield tobacco-burning group (i.e. approximately 23–29%). In the pairwise comparisons, statistically significant differences in serum cotinine levels were observed in the snus group relative to both the tobacco-heating and ultra-low machine yield tobacco-burning cigarette groups at week 24, with no other statistically significant differences in product group comparisons observed.
Carboxyhemoglobin
In the tobacco-heating cigarette group, a statistically significant reduction of approximately 16% in percent COHb at week 12 was observed, but there were no statistically significant differences at week 24 (Figure 5). Reduction from baseline was statistically significant in the snus group at both time points (i.e. approximately 59% at week 12 and approximately 37% at week 24). Percent COHb was not statistically significantly changed at either week 12 or 24 for the ultra-low machine yield tobacco-burning cigarette group. In pairwise product group comparisons, reductions in the snus group were statistically significantly greater than reductions in either the tobacco-heating or ultra-low machine yield tobacco-burning cigarette groups at both time points.
Figure 5.
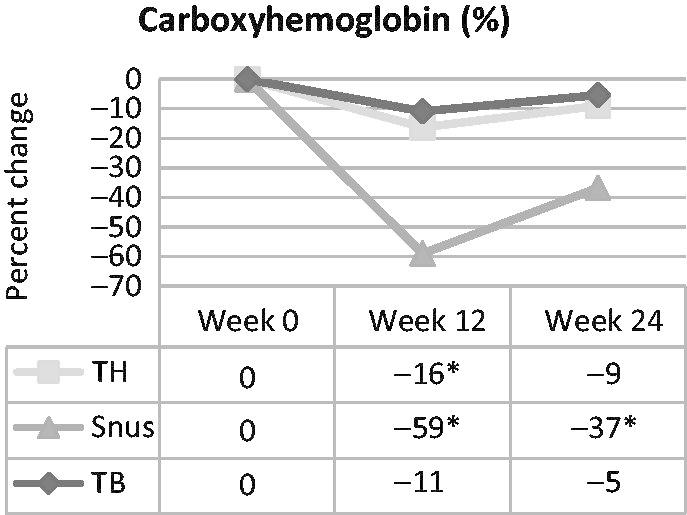
Carboxyhemoglobin over time in smokers switched to tobacco-heating cigarettes (TH), snus or ultra-low machine yield tobacco-burning cigarettes (TB). *Statistically significant reduction (p < 0.05) from week 0.
Aromatic amine
Statistically significant reductions (i.e. approximately 45%) in 4-ABP-Hb were observed in the tobacco-heating cigarette group at both weeks 12 and 24 (Figure 6). No other statistically significant differences were observed. In the product group pairwise comparisons, reductions in the tobacco-heating cigarette group were statistically significantly greater than those in the snus and ultra-low machine yield tobacco-burning cigarette groups at both time points.
Figure 6.
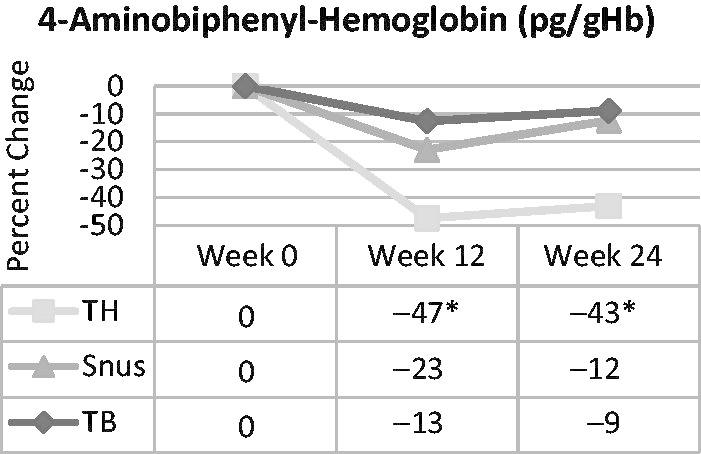
4-Aminobiphenyl hemoglobin adducts over time in smokers switched to ultra-low machine yield tobacco-burning cigarettes (TB), tobacco-heating cigarettes (TH) or snus. *Statistically significant reduction (p < 0.05) from week 0.
Mouth level exposure
Confinement effect
Results of the analysis for potential confinement effects are presented in Table 5. The only evidence of a possible confinement effect was noted at week 24 for nicotine in mg/cigarette, where a statistically significant increase (approximately 0.16 mg/cigarette) was observed for in-clinic yield compared with the day prior to clinical confinement (Table 5). This difference did not result in statistically significant yield changes for total daily nicotine yield, “tar” per cigarette, or total daily “tar” yield.
Table 5. MLE markers, within confinement versus prior to confinement a .
| MLE marker (units) | Time point | N (within-) | N (prior-) | Mean difference b (95% CI) |
|---|---|---|---|---|
| Char line (mm) | Week 0 | 35 | 35 | 0.36 (−1.23, 1.95) |
| Week 12 | 34 | 33 | −0.51 (−2.13, 1.12) | |
| Week 24 | 34 | 32 | −0.32 (−1.97, 1.32) | |
| Nicotine (mg/cigarette) | Week 0 | 31 | 29 | 0.063 (−0.091, 0.216) |
| Week 12 | 30 | 29 | 0.055 (−0.100, 0.211) | |
| Week 24 | 27 | 26 | 0.163 (0.0004, 0.326) c | |
| Nicotine (mg/day) | Week 0 | 31 | 29 | −1.25 (−5.27, 2.78) |
| Week 12 | 30 | 29 | −0.90 (−4.99, 3.18) | |
| Week 24 | 27 | 26 | 2.47 (−1.80, 6.75) | |
| Tar (mg/cigarette) | Week 0 | 31 | 29 | 0.35 (−1.92, 2.62) |
| Week 12 | 30 | 29 | 1.02 (−1.28, 3.32) | |
| Week 24 | 27 | 26 | 2.36 (−0.05, 4.77) | |
| Tar (mg/day) | Week 0 | 31 | 29 | −22.9 (−80.1, 34.3) |
| Week 12 | 30 | 29 | −13.7 (−71.7, 44.3) | |
| Week 24 | 27 | 26 | 28.3 (−32.4, 89.0) |
MLE, mouth level exposure.
Ultra-low machine yield tobacco-burning cigarette group only, per-protocol sample.
Least squares mean from ANOVA; difference calculated as within confinement minus prior to confinement.
p < 0.05.
Compensatory smoking effect
Mean differences in pairwise comparisons of week means of MLE markers prior to confinement and within confinement, as a measure of compensatory smoking, are presented in Tables 6 and 7, respectively. As measured by the char line, subjects smoked statistically significantly less of the cigarette in week 24 compared with usual brand at week 0 prior to clinical confinement (approximately 1.8 mm less) (Table 6). Additionally, the average yield of nicotine (mg/cigarette) was statistically significantly reduced from baseline to weeks 12 and 24 (approximately 0.5–0.6 mg less) both prior to and within clinical confinement (Tables 6 and 7). Statistically significant reductions, prior to and within confinement, were also observed for total nicotine yield per day (approximately 7–11 mg/day less) and “tar” yield per cigarette (approximately 3–5 mg/cigarette less), with non-statistically significant changes from weeks 12 to 24 for these endpoints both prior to and within confinement. The daily “tar” yields were reduced approximately 14–65 mg/day; however, the only statistically significant reduction was the change from baseline to week 24 prior to clinical confinement (approximately 65 mg/day less).
Table 6. Time point mean differences (95% CI) for MLE markers, prior to confinement a .
| MLE marker (units) | Weeks 0–12 | Weeks 0–24 | Weeks 12–24 |
|---|---|---|---|
| Char line (mm) | 0.63 (−0.99, 2.25) | 1.83 (0.20, 3.46) b | 1.20 (−0.45, 2.86) |
| Nicotine (mg/cigarette) | −0.621 (−0.779, −0.464) b | −0.620 (−0.783, −0.457) b | 0.002 (−0.159, 0.162) |
| Nicotine (mg/day) | −9.05 (−13.19, −4.92) b | −10.60 (−14.88, −6.32) b | −1.54 (−5.76, 2.68) |
| Tar (mg/cigarette) | −4.97 (−7.30, −2.64) b | −5.06 (−7.47, −2.65) b | −0.09 (−2.46, 2.29) |
| Tar (mg/day) | −40.6 (−99.3, 18.1) | −65.1 (−125.8, −4.4) b | −24.5 (−84.4, 35.4) |
MLE, mouth level exposure. See Table 5 for sample sizes.
Ultra-low machine yield tobacco-burning cigarette group only.
p<0.05.
Table 7. Time point mean differences (95% CI) in pairwise comparisons for MLE markers, within confinement a .
| MLE marker (units) | Weeks 0–12 | Weeks 0–24 | Weeks 12–24 |
|---|---|---|---|
| Char line (mm) | −0.24 (−1.84, 1.37) | 1.15 (−0.45, 2.75) | 1.38 (−0.23, 3.00) |
| Nicotine (mg/cigarette) | −0.63 (−0.78, −0.48) b | −0.52 (−0.68, −0.36) b | 0.11 (−0.05, 0.27) |
| Nicotine (mg/day) | −8.71 (−12.71, −4.71) b | −6.88 (−11.04, −2.72) b | 1.83 (−2.35, 6.01) |
| Tar (mg/cigarette) | −4.30 (−6.56, −2.05) b | −3.05 (−5.39, −0.71) b | 1.25 (−1.10, 3.60) |
| Tar (mg/day) | −31.4 (−88.1, 25.4) | −13.9 (−72.9, 45.2) | 17.5 (−41.8, 76.8) |
MLE, mouth level exposure. See Table 5 for sample sizes.
Ultra-low machine yield tobacco-burning cigarette group only.
p < 0.05.
Nicotine yield versus uptake
Comparisons of YIU nicotine and NICEq are presented in Table 8. Cigarettes smoked by subjects yielded approximately 9–10 mg less total nicotine per day on average (corresponding to an approximately 27–31% reduction). Uptake by subjects (i.e. NICEq) was approximately 7 mg less total nicotine per day on average (corresponding to an approximately 29% reduction) at weeks 12 and 24 compared with usual brand at baseline (Table 8). A comparison of the YIU nicotine and nicotine biomarker (NICEq) data indicates that approximately 69–74% of the nicotine yielded from cigarettes during smoking was absorbed. This was consistent between usual brand at baseline and the ultra-low machine yield tobacco-burning cigarettes at weeks 12 and 24. These data indicate that YIU is a reliable estimate of relative nicotine yield and uptake when switching cigarettes.
Table 8. Summary and comparison of YIU nicotine and NICEq a .
| Change from baseline |
||||||
|---|---|---|---|---|---|---|
| Marker | Time point | N | Nicotine (mg/day) | Mean (95% CI) | Percent | Percent uptake b |
| YIU nicotine | Week 0 | 29 | 34.0 | – | – | – |
| Week 12 | 29 | 25.0 | −9.0 (−13.2, −4.9) | −26.6 | – | |
| Week 24 | 26 | 23.4 | −10.6 (−14.9, −6.3) | −31.2 | – | |
| NICEq | Week 0 | 33 | 24.4 | – | – | 71.7 |
| Week 12 | 33 | 17.3 | −7.1 (−9.1, −5.1) | −29.2 | 69.1 | |
| Week 24 | 32 | 17.3 | −7.1 (−9.0, −5.2) | −29.1 | 73.9 | |
YIU, yield-in-use; NICEq, nicotine equivalents.
Ultra-low machine yield tobacco-burning cigarette group only.
(NICEq/YIU daily nicotine) × 100.
Discussion
It is not clear that any single compound, or any subset of compounds, in tobacco or tobacco smoke contributes directly to the risk of developing diseases associated with cigarette smoking. However, with substantial reductions in exposure to multiple classes of harmful and potentially harmful compounds, reductions in risk may be possible.
Biomarkers are useful for determining actual exposure to tobacco constituents among consumers of various tobacco products. In this article, changes in biomarkers of exposure among cigarette smokers switched to alternate tobacco products (i.e. tobacco-heating cigarettes, snus and ultra-low machine yield tobacco-burning cigarettes) for 24 weeks have been presented. At baseline (week 0), measured biomarkers in both urine and blood were statistically significantly greater in smokers than in a comparison group of never smokers. This indicated that further evaluation of these biomarkers after randomization to the three product groups was reasonable.
After switching to the alternate tobacco products for 24 weeks, week 12 and 24 reductions in many of the measured biomarkers of exposure were observed in the three product groups, with a few exceptions. A greater number of reductions and a greater magnitude in reductions were observed in the groups switched to tobacco-heating cigarettes and snus, although reductions in some constituents were also observed in subjects switched to ultra-low machine yield tobacco-burning cigarettes (with reductions attributed to product switching, not decreased daily cigarette consumption) (Ogden et al., 2015b). It should be noted that compliance with assigned study product was less than 100% in all three groups, with highest compliance in the tobacco-burning cigarette group, and lowest compliance in the snus group (Ogden et al., 2015b). Thus, biomarker values after 12 and 24 weeks should be interpreted as estimates of the effects due to dual or poly-tobacco product use, rather than as estimates of effects due to complete switching to the assigned alternate tobacco product; further improvements are likely to have occurred with complete product switching.
In the tobacco-heating cigarette group, statistically significant reductions were observed for most measured biomarkers of exposure at both weeks 12 and 24, with the exception of 1-naphthol, 1-OHP, DHBMA, and HPMA in urine, and serum cotinine and percent COHb. As described above, COHb was significantly reduced at week 12 and not different at week 24. Notably, several prior, short-term studies have found significantly increased carbon monoxide exposure among subjects using Eclipse relative to usual brand cigarettes (see, e.g. Lee et al., 2004). The different finding for COHb presented herein might be partly due to the longer-term product use in this study compared with previous studies. However, it is primarily attributed to a redesign of the fuel source (i.e. geometry and composition) for Eclipse in mid-2006, which was specifically targeted to reduce CO yield. This redesigned fuel source was incorporated in the tobacco-heating cigarettes for this study and remains unchanged in current market Eclipse cigarettes to date.
Similarly in the snus group, statistically significant reductions were observed for the majority of the biomarkers of exposure with the exception of NICEq, 1-naphthol, 1-OHP, and DHBMA in urine, and serum cotinine and 4-ABP-Hb in blood. In the group switched to ultra-low machine yield tobacco-burning cigarettes, reductions were observed in approximately one-half of the measured biomarkers, while no significant changes were observed for 1-naphthol, 2-OHF, 1-/9-OHPh, 2-/3-OHPh, 1-OHP, MHBMA, DHBMA, HMPMA, HPMA, and SPMA in urine, and percent COHb and 4-ABP-Hb adducts. Not surprisingly, the biomarkers for tobacco combustion-related, harmful and potentially harmful constituents (e.g. PAHs) were more likely to have been reduced in the tobacco-heating and snus groups than in the ultra-low machine yield tobacco-burning cigarette group. Additionally, changes in exposure were consistent with differences in chemistry among the three products (Bodnar et al., 2012; Borgerding et al., 2012).
In general, both 1-naphthol and 1-OHP may be less tobacco-specific than the four other PAH biomarkers evaluated (Hatsukami et al., 2006; Joseph et al., 2005). In the current study, both 1-naphthol and 1-OHP were statistically significantly elevated in smokers compared with never smokers at baseline. However, the magnitude of differences in both 1-naphthol and 1-OHP between smokers and never smokers was less than those of the other PAH biomarkers, consistent with previous studies (Hecht et al., 2004; IARC, 2004; Yang et al., 1999). Additionally, no differences or inconsistent patterns for 1-naphthol and 1-OHP were observed after 12 and 24 weeks in all three groups switched to alternate tobacco products, supporting the notion of decreased specificity for tobacco-related exposures.
Similarly, although the major urine metabolites of 1,3-butadiene have been reported to be MHBMA and DHBMA (Urban et al., 2003), previous publications have suggested that MHBMA is a more specific marker for 1,3-butadiene exposure from cigarette smoking than DHBMA (Carmella et al., 2009; van Sittert et al., 2000). In this study, statistically significant and greater reductions in MHBMA were observed in the tobacco-heating cigarette and snus groups at weeks 12 and 24, in contrast to DHBMA. DHBMA did not differ statistically significantly in any of the three product groups, and while it was different between smokers and never smokers at baseline, the difference was less than twofold. Coupled with a higher differential for MHBMA levels between smokers and never smokers (i.e. >15-fold), this suggests a strong selectivity for MHBMA in representing 1,3-butadiene exposure changes among consumers of various tobacco products.
Consistent with the current study results, urine concentrations of the aromatic amines 4-ABP, 2-AN, and o-T have been reported to be higher in smokers compared with non-smokers in some studies (Riedel et al., 2006; Seyler & Bernert, 2011), but not all (Hecht, 2002). For 4-ABP-Hb, levels in blood have been consistently higher in smokers compared with non-smokers, and levels are associated with quantity and duration of cigarette smoking, as well as smoking cessation (Hatsukami et al., 2006; Talaska & Al-Zoughool, 2003). The differences in reduction between the two markers for 4-aminobiphenyl (i.e. 4-ABP in urine and 4-ABP-Hb in blood) are likely due to the longer half-life of 4-ABP-Hb adducts compared with the half-life of 4-ABP in urine (Seyler & Bernert, 2011). Accordingly, hemoglobin adducts may be more reliable markers of chronic aromatic amine exposure from tobacco smoking than urine markers (Hecht, 2002), and the determination of the free aromatic amine in the urine is likely a better short-term measure of exposure reduction. In the current study, for both urine 4-ABP and 4-ABP-Hb adducts in blood, the largest reductions from baseline (i.e. at either week 12 or week 24) were observed in the tobacco-heating cigarette group, followed by the snus group, and finally in the ultra-low machine yield tobacco-burning cigarette group. The reductions in urine 4-ABP in all three product groups at both time points were statistically significant; however, for 4-ABP-Hb adducts, only the reductions in the tobacco-heating group were statistically significant. This is possibly because subjects in the snus group tended to be less compliant with use of the assigned product (Ogden et al., 2015b), and continued to smoke some tobacco-burning cigarettes per day.
In this study, urinary HPMA, a marker for acrolein exposure, was higher in the tobacco-heating cigarette group at 12 and 24 weeks compared with baseline, as well as relative to the other two product groups. This was the only biomarker of exposure that presented this pattern, and was specific to the tobacco-heating cigarette group. This increase in urinary HPMA was consistent with differences in the measured machine-yields of acrolein from various cigarette products (i.e. increase in Eclipse compared with tobacco-burning cigarettes) (Bodnar et al., 2012). It is notable that preclinical and observational studies of acrolein-only exposure have documented an association with inflammation, oxidative stress and irritation (ATSDR, 2007; Uchida et al., 1998). However, biomarkers of inflammation and oxidative stress evaluated in the current study were not observed to be increased in the tobacco-heating cigarette group subjects relative to baseline or relative to the other two product groups (Ogden et al., 2015a). Similarly, previous evaluations have indicated reduced biological activity and toxicological risk of the Eclipse cigarette compared with traditional combustible cigarettes (Marano et al., 2012; Wagner & Eclipse Expert Panel, 2000).
MLE analyses were used in this study to evaluate whether subjects altered their smoking topography and behaviour, during confinement and/or in response to switching from usual brand to an ultra-low machine yield tobacco-burning cigarette, as well as to compare nicotine MLE with relative nicotine uptake (i.e. NICEq). The data showed minimal to no confinement effects and/or compensatory smoking in the subjects switched to ultra-low machine yield tobacco-burning cigarettes. Additionally, study findings indicated that YIU is a reliable estimate of relative nicotine yield and smoker uptake when switching cigarettes.
Results from this randomized, multi-center study indicated that adult cigarette smokers switched from their usual brand of cigarettes to alternate tobacco products, including tobacco-heating cigarettes, snus and ultra-low machine yield tobacco-burning cigarettes, had significantly reduced exposure to many potentially harmful constituents found in cigarette smoke. Notably, the average “tar” yields of usual brand cigarettes in all three groups were approximately the same at baseline (Ogden et al., 2015b). In comparison with subjects switched to ultra-low machine yield tobacco-burning cigarettes, subjects switched to tobacco-heating cigarettes or snus had greater reductions in these biomarkers of tobacco exposure, both in number of constituents and magnitude of reductions. These reductions were likely associated with the elimination and near elimination of tobacco combustion in use of snus and tobacco-heating cigarettes, respectively, although reductions in biomarkers of exposure were also observed in the ultra-low machine yield tobacco-burning cigarette group. In general, the results from the current study are consistent with previous studies of smokers switched to alternate tobacco products, including an electrically heated cigarette, snus and lower machine yield tobacco-burning cigarettes (Benowitz et al., 2009; Kotlyar et al., 2011; Leroy et al., 2012; Mendes et al., 2008; Roethig et al., 2008; Sarkar et al., 2010; Smith et al., 1996). Limitations of the study included the predominantly White subject sample in general and the predominantly male sample in the per-protocol sample of smokers switched to snus, limiting generalizability of the findings.
Advantages of the current study included the relatively long duration, the extensive number of biomarkers evaluated, and the inclusion of the ultra-low machine yield tobacco-burning cigarette group as a control and for comparison. These results further the understanding of tobacco and tobacco smoke constituent exposure among combustible and non-combustible tobacco products. Results similarly provide a relevant foundation for initial placement of alternate tobacco products along a continuum of risk.
Supplementary Material
Acknowledgements
The authors gratefully acknowledge Betsy Bombick, Gary Byrd, Summer Hanna, David Heavner, Tao Jin, George Krautter, Paul Nelson, Elaine Round, Thomas Steichen, and Weiying Yan.
SAS and all other SAS Institute Inc. product or service names are registered trademarks or trademarks of SAS Institute Inc., Cary, NC, USA. ® indicates USA registration.
Declaration of interest
All authors are current employees of RAI Services Company or R.J. Reynolds Tobacco Company.
Supplementary material available online
Supplementary Tables 1-4
References
- Adhikari B, Kahende J, Malarcher A, et al. Smoking-attributable mortality, years of potential life lost, and productivity losses – United States, 2000–2004. MMWR. 2008;57:1226–8. [PubMed] [Google Scholar]
- Agency for Toxic Substances and Disease Registry (ATSDR). 2007. http://www.atsdr.cdc.gov/toxprofiles/tp124.pdf [Google Scholar]
- Benowitz NL, Dains KM, Hall SM, et al. Progressive commercial cigarette yield reduction: biochemical exposure and behavioral assessment. Cancer Epidemiol Biomarkers Prev. 2009;18:876–83. doi: 10.1158/1055-9965.EPI-08-0731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodnar JA, Morgan WT, Murphy PA, Ogden MW. Mainstream smoke chemistry analysis of samples from the 2009 US cigarette market. Regul Toxicol Pharmacol. 2012;64:35–42. doi: 10.1016/j.yrtph.2012.05.011. [DOI] [PubMed] [Google Scholar]
- Borgerding MF, Bodnar JA, Curtin GM, Swauger JE. The chemical composition of smokeless tobacco: a survey of products sold in the United States in 2006 and 2007. Regul Toxicol Pharmacol. 2012;64:367–87. doi: 10.1016/j.yrtph.2012.09.003. [DOI] [PubMed] [Google Scholar]
- Borgerding M, Klus H. Analysis of complex mixtures – cigarette smoke. Exp Toxicol Pathol. 2005;57:43–73. doi: 10.1016/j.etp.2005.05.010. [DOI] [PubMed] [Google Scholar]
- Byrd GD, Davis RA, Ogden MW. A rapid LC–MS–MS method for the determination of nicotine and cotinine in serum and saliva samples from smokers: validation and comparison with a radioimmunoassay method. J Chromatogr Sci. 2005;43:133–40. doi: 10.1093/chromsci/43.3.133. [DOI] [PubMed] [Google Scholar]
- Byrd GD, Ogden MW. Liquid chromatographic/tandem mass spectrometric method for the determination of the tobacco-specific nitrosamine metabolite NNAL in smokers' urine. J Mass Spectrom. 2003;38:98–107. doi: 10.1002/jms.406. [DOI] [PubMed] [Google Scholar]
- Carmella SG, Chen M, Han S, et al. Effects of smoking cessation on eight urinary tobacco carcinogen and toxicant biomarkers. Chem Res Toxicol. 2009;22:734–41. doi: 10.1021/tx800479s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatsukami DK, Benowitz NL, Rennard SI, et al. Biomarkers to assess the utility of potential reduced exposure tobacco products. Nicotine Tob Res. 2006;8:600–22. doi: 10.1080/14622200600858166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hecht SS. Human urinary carcinogen metabolites: biomarkers for investigating tobacco and cancer. Carcinogenesis. 2002;23:907–22. doi: 10.1093/carcin/23.6.907. [DOI] [PubMed] [Google Scholar]
- Hecht SS, Carmella SG, Le KA, et al. Effects of reduced cigarette smoking on levels of 1-hydroxypyrene in urine. Cancer Epidemiol Biomarkers Prev. 2004;13:834–42. [PubMed] [Google Scholar]
- International Agency for Research on Cancer (IARC). Tobacco smoke and involuntary smoking. IARC Monogr Eval Carcinog Risks Hum. 2004;83:1–1452. [PMC free article] [PubMed] [Google Scholar]
- International Conference on Harmonization (ICH). 1996. http://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM073122.pdf [Google Scholar]
- Joseph AM, Hecht SS, Murphy SE, et al. Relationships between cigarette consumption and biomarkers of tobacco toxin exposure. Cancer Epidemiol Biomarkers Prev. 2005;14:2963–8. doi: 10.1158/1055-9965.EPI-04-0768. [DOI] [PubMed] [Google Scholar]
- Kado NY, Langley D, Eisenstadt E. A simple modification of the Salmonella liquid-incubation assay. Increased sensitivity for detecting mutagens in human urine. Mutat Res. 1983;121:25–32. doi: 10.1016/0165-7992(83)90082-9. [DOI] [PubMed] [Google Scholar]
- Kotlyar M, Hertsgaard LA, Lindgren BR, et al. Effect of oral snus and medicinal nicotine in smokers on toxicant exposure and withdrawal symptoms: a feasibility study. Cancer Epidemiol Biomarkers Prev. 2011;20:91–100. doi: 10.1158/1055-9965.EPI-10-0349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee PN. The effect of reducing the number of cigarettes smoked on risk of lung cancer, COPD, cardiovascular disease and FEV(1) – a review. Regul Toxicol Pharmacol. 2013;67:372–81. doi: 10.1016/j.yrtph.2013.08.016. [DOI] [PubMed] [Google Scholar]
- Lee EM, Malson JL, Moolchan ET, Pickworth WB. Quantitative comparisons between a nicotine delivery device (Eclipse) and conventional cigarette smoking. Nicotine Tob Res. 2004;6:95–102. doi: 10.1080/14622200310001656911. [DOI] [PubMed] [Google Scholar]
- Lewalter J, Gries W. Hemoglobin adducts for aromatic amines: aniline, o-, m-, p-toluidine, o-anisidine, p-chloroaniline, alpha- and beta-naphthylamine, 4-aminodiphenyl, benzidine, 4,4′-diaminodiphenylmethane, 3,3′-dichlorobenzidine. In: Angerer J, Schaller KH, editors. Analyses of hazardous substances in biological materials. Weinheim: Wiley-VCH; 2000. pp. 191–218. [Google Scholar]
- Leroy CM, Jarus-Dziedzic K, Ancerewica D, et al. Reduced exposure evaluation of an electrically heated cigarette system. Part 7: a one-month, randomized, ambulatory, controlled clinical study in Poland. Reg Tox Pharm. 2012;64:S74–84. doi: 10.1016/j.yrtph.2012.08.006. [DOI] [PubMed] [Google Scholar]
- Marano KM, Naufal ZS, Borgerding MB, Potts RJ. Quantitative risk assessment of tobacco-burning and tobacco-heating cigarettes. Rec Adv Tobacco Sci. 2012;38:3–20. [Google Scholar]
- Mascher DG, Mascher HJ, Scherer G, Schmid ER. High-performance liquid chromatographic-tandem mass spectrometric determination of 3-hydroxypropylmercapturic acid in human urine. J Chromatogr B Biomed Sci Appl. 2001;750:163–9. doi: 10.1016/s0378-4347(00)00385-6. [DOI] [PubMed] [Google Scholar]
- Meger M, Meger-Kossien I, Schuler-Metz A, et al. Simultaneous determination of nicotine and eight nicotine metabolites in urine of smokers using liquid chromatography–tandem mass spectrometry. J Chromatogr B. 2002;778:251–61. doi: 10.1016/s0378-4347(01)00451-0. [DOI] [PubMed] [Google Scholar]
- Mendes P, Kapur S, Wang J, et al. A randomized, controlled exposure study in adult smokers of full flavor Marlboro cigarettes switching to Marlboro Lights or Marlboro Ultra Lights cigarettes. Regul Toxicol Pharmacol. 2008;51:295–305. doi: 10.1016/j.yrtph.2008.04.014. [DOI] [PubMed] [Google Scholar]
- Nelson PR, Chen P, Dixon M, Steichen T. A survey of mouth level exposure to cigarette smoke in the United States. Regul Toxicol Pharmacol. 2011;61:S25–38. doi: 10.1016/j.yrtph.2010.10.001. [DOI] [PubMed] [Google Scholar]
- Nutt DJ, Phillips LD, Balfour D, et al. Estimating the harms of nicotine-containing products using the MCDA approach. Eur Addict Res. 2014;20:218–25. doi: 10.1159/000360220. [DOI] [PubMed] [Google Scholar]
- Ogden MW, Marano KM, Jones BA, et al. Switching from usual brand cigarettes to a tobacco-heating cigarette or snus: Part 3. Biomarkers of biological effect; 2015a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogden MW, Marano KM, Jones BA, Stiles MF. Switching from usual brand cigarettes to a tobacco-heating cigarette or snus: Part 1. Study design and methodology; 2015b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richter E, Rösler S, Scherer G, et al. Haemoglobin adducts from aromatic amines in children in relation to area of residence and exposure to environmental tobacco smoke. Int Arch Occup Environ Health. 2001;74:421–8. doi: 10.1007/s004200100243. [DOI] [PubMed] [Google Scholar]
- Riedel K, Scherer G, Engl J, et al. Determination of three carcinogenic aromatic amines in urine of smokers and nonsmokers. J Anal Toxicol. 2006;30:187–95. doi: 10.1093/jat/30.3.187. [DOI] [PubMed] [Google Scholar]
- Roethig HJ, Feng S, Liang Q, et al. A 12-month, randomized, controlled study to evaluate exposure and cardiovascular risk factors in adult smokers switching from conventional cigarettes to a second-generation electrically heated cigarette smoking system. J Clin Pharmacol. 2008;48:580–91. doi: 10.1177/0091270008315316. [DOI] [PubMed] [Google Scholar]
- Sarkar M, Liu J, Koval T, et al. Evaluation of biomarkers of exposure in adult cigarette smokers using Marlboro snus. Nicotine Tob Res. 2010;12:105–16. doi: 10.1093/ntr/ntp183. [DOI] [PubMed] [Google Scholar]
- Scherer G, Frank S, Riedel K, et al. Biomonitoring of exposure to polycyclic aromatic hydrocarbons of nonoccupationally exposed persons. Cancer Epidemiol Biomarkers Prev. 2000a;9:373–80. [PubMed] [Google Scholar]
- Scherer G, Meger M, Meger-Kossien I, Pachinger A. Biological monitoring of the tobacco-smoke related exposure to acrolein. Proc Am Assoc Cancer Res. 2000b;42:150. [Google Scholar]
- Scherer G, Meger M, Meger-Kossien I, Pachinger A. Biological monitoring of the tobacco-smoke related exposure to benzene. Proc Am Assoc Cancer Res. 2001;41:146. [Google Scholar]
- Scherer G, Urban M, Hagedorn HW, et al. Determination of methyl-, 2-hydroxyethyl- and 2-cyanoethylmercapturic acids as biomarkers of exposure to alkylating agents in cigarette smoke. J Chromatogr B Analyt Technol Biomed Life Sci. 2010;878:2520–8. doi: 10.1016/j.jchromb.2010.02.023. [DOI] [PubMed] [Google Scholar]
- Sexton K. Biomarkers of toxicant exposure. In: DeCaprio AP, editor. Toxicologic biomarkers. New York: Taylor & Francis; 2006. pp. 39–62. [Google Scholar]
- Seyler TH, Bernert JT. Analysis of 4-aminobiphenyl in smoker's and nonsmoker's urine by tandem mass spectrometry. Biomarkers. 2011;16:212–21. doi: 10.3109/1354750X.2010.544755. [DOI] [PubMed] [Google Scholar]
- Smith CJ, McKarns SC, Davis RA, et al. Human urine mutagenicity study comparing cigarettes which burn or primarily heat tobacco. Mutat Res. 1996;361:1–9. doi: 10.1016/s0165-1161(96)90222-8. [DOI] [PubMed] [Google Scholar]
- Stratton K, Shetty P, Wallace R, Bondurant S. Clearing the smoke: the science base for tobacco harm reduction-executive summary. Tob Control. 2001;10:189–95. doi: 10.1136/tc.10.2.189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talaska G, Al-Zoughool M. Aromatic amines and biomarkers of human exposure. J Environ Sci Health C Environ Carcinog Ecotoxicol Rev. 2003;21:133–64. doi: 10.1081/GNC-120026234. [DOI] [PubMed] [Google Scholar]
- Uchida K, Kanematsu M, Sakai K, et al. Protein-bound acrolein: potential markers for oxidative stress. Proc Natl Acad Sci USA. 1998;95:4882–7. doi: 10.1073/pnas.95.9.4882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- US Department of Health and Human Services (USDHHS). Reducing the health consequences of smoking: 25 years of progress: a report of the surgeon general. Atlanta (GA): US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 1989. [Google Scholar]
- US Department of Health and Human Services (USDHHS). The health consequences of smoking: a report of the surgeon general. Atlanta (GA): US Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2004. [Google Scholar]
- Urban M, Gilch G, Schepers G, et al. Determination of the major mercapturic acids of 1,3-butadiene in human and rat urine using liquid chromatography with tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2003;796:131–40. doi: 10.1016/j.jchromb.2003.08.009. [DOI] [PubMed] [Google Scholar]
- Urban M, Kavvadias D, Riedel K, et al. Urinary mercapturic acids and a hemoglobin adduct for the dosimetry of acrylamide exposure in smokers and nonsmokers. Inhal Toxicol. 2006;18:831–9. doi: 10.1080/08958370600748430. [DOI] [PubMed] [Google Scholar]
- US Congress . 2009. http://www.gpo.gov/fdsys/pkg/PLAW-111publ31/html/PLAW-111publ31.htm [Google Scholar]
- van Sittert NJ, Megens HJ, Watson WP, Boogaard PJ. Biomarkers of exposure to 1,3-butadiene as a basis for cancer risk assessment. Toxicol Sci. 2000;56:189–202. doi: 10.1093/toxsci/56.1.189. [DOI] [PubMed] [Google Scholar]
- Wagner BM. Eclipse Expert Panel. A safer cigarette? A comparative study. A consensus report. Inhal Tox. 2000;12:1–48. doi: 10.1080/08958378.2000.11720735. [DOI] [PubMed] [Google Scholar]
- Yang M, Koga M, Katoh T, Kawamoto T. A study for the proper application of urinary naphthols, new biomarkers for airborne polycyclic aromatic hydrocarbons. Arch Environ Contam Toxicol. 1999;36:99–108. doi: 10.1007/s002449900447. [DOI] [PubMed] [Google Scholar]
- Zeller M, Hatsukami D. Strategic Dialogue on Tobacco Harm Reduction Group. The strategic dialogue on tobacco harm reduction: a vision and blueprint for action in the US. Tob Control. 2009;18:324–32. doi: 10.1136/tc.2008.027318. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


