Abstract
The tarnished plant bug (TPB), Lygus lineolaris (Palisot de Beauvois) is a polyphagous, phytophagous insect that has emerged as a major pest of cotton, alfalfa, fruits, and vegetable crops in the eastern United States and Canada. Using its piercing-sucking mouthparts, TPB employs a “lacerate and flush” feeding strategy in which saliva injected into plant tissue degrades cell wall components and lyses cells whose contents are subsequently imbibed by the TPB. It is known that a major component of TPB saliva is the polygalacturonase enzymes that degrade the pectin in the cell walls. However, not much is known about the other components of the saliva of this important pest. In this study, we explored the salivary gland transcriptome of TPB using Illumina sequencing. After in silico conversion of RNA sequences into corresponding polypeptides, 25,767 putative proteins were discovered. Of these, 19,540 (78.83%) showed significant similarity to known proteins in the either the NCBI nr or Uniprot databases. Gene ontology (GO) terms were assigned to 7,512 proteins, and 791 proteins in the sialotranscriptome of TPB were found to collectively map to 107 Kyoto Encyclopedia of Genes and Genomes (KEGG) database pathways. A total of 3,653 Pfam domains were identified in 10,421 sialotranscriptome predicted proteins resulting in 12,814 Pfam annotations; some proteins had more than one Pfam domain. Functional annotation revealed a number of salivary gland proteins that potentially facilitate degradation of host plant tissues and mitigation of the host plant defense response. These transcripts/proteins and their potential roles in TPB establishment are described.
Introduction
Insect saliva can perform numerous functions, though, one of its main roles is to initiate digestion [1, 2]. The salivary gland proteins of phytophagous insects include a complement of digestive proteins that can differ depending on feeding strategy. In phytophagous insects with chewing mouthparts, the saliva is used primarily to initiate digestion of the plant during the mastication process. On the other hand, in insects with piercing, sucking mouthparts, the saliva is injected into plant tissues where it solubilizes cellular and extracellular materials in preparation for eventual ingestion, through the insect’s stylet [3]. This mode of feeding has been described as “lacerate and flush”. Additionally, saliva of phytophagous insects can contain proteins and other molecules that can act as “effectors” to facilitate stylet penetration while suppressing host defense responses [4–9]. Thus, the insect saliva is the first metabolically active chemical substance that comes into direct contact with the plant and has a very important role in both food ingestion as well as insect-plant interactions [7, 10, 11].
The mirids (Order: Hemiptera, Sub-Order: Heteroptera, Family: Miridae) typically employ the “lacerate and flush” feeding strategy. The most well-known mirids are agricultural pests with a wide host range. Within the subfamily Mirinae are the lygus bugs (Lygus spp.), the species that cause the most economic damage to agronomically important plants. The tarnished plant bug (TPB) Lygus lineolaris (Palisot de Beauvois), is a highly polyphagous, phytophagous insect, with documented feeding on more than 300 plant species, collectively representing 36 plant families [12, 13]. Plant symptoms resulting from TPB feeding include organ abscission, deformation of developing fruits, necrosis at the feeding site, seeds with aborted embryos, and reduced vegetative growth [14].
The TPB is notorious for causing numerous physiological effects in cotton including: shortened internode length, apical termination, loss of apical dominance leading to the development of many secondary terminals (crazy cotton), anther necrosis, square abscission, yellowish staining of the apex developing floral bud (dirty square), as well as the yellowish staining of the anthers (dirty flower), and complete cellular dissolution of the floral bud [15–18]. Several of these effects are due to TPB’s preferential feeding on the cotton floral structure as it develops [16–18]. With the broad-scale adoption of genetically modified Bacillus thuringiensis (Bt) cotton and the eradication of the boll weevil, insecticide applications to control TPB have increased [16]. Moreover, TPB populations have been shown to be increasingly resistant to some insecticide classes commonly used for their control including cyclodienes, organophosphates, pyrethroids, and neonicotinoids [19–23]. Insecticide resistance and the ability of TPB to enter diapause, which permits insects to overwinter have made TPB a difficult pest to control [24]. Economic thresholds based on TPB counts from sweeps and drop cloths as well as counts of dirty squares have been established to assist growers in determining whether TPB countermeasures are economically advisable [25–27]. Of interest, the dirty square symptom that develops into the dirty flower, appears to be the manifestation of molecular catabolism caused by TPB feeding. This symptom is also used for resistance screening in cotton cultivar and germplasm breeding programs since phenotypic evaluations of cotton plants have suggested that resistance to dirty square (i.e. resistance to TPB) is under genetic control [28–32].
The role of pectin degrading enzymes (polygalacturonases) in the saliva of lygus bugs was recognized almost four decades ago [14], however conclusive evidence for the specific role of polygalacturonases in causing lygus-like damage to plants came much later from studies of the western tarnished plant bug (WTPB) L. hesperus [33, 34]. The study of de la Paz Celorio-Mancera et al. 2008 [35] suggested the presence of four polygalacturonases (Lhpg1, Lhpg2, Lhpg3 and Lhpg4) in salivary gland extracts of the WTPB. Whereas in TPB three polygalacturonase encoding genes (Llpg1, Llpg2 and Llpg3) were described [36]. RNAi mediated knockdown of Llpg1 in TPB displayed no obvious phenotype, with no detrimental effects on longevity [37].
Many of the earlier salivary gland transcriptome analysis conducted in the last decade mainly focused on blood feeding insects such as Ixodes scapularis [38, 39], Anopheles gambiae [40–43], Dermacentor andersoni [44], Triatome brasiliensis [45] and Amblyomma variegatum [46] among others. However, some of the more recent studies on salivary gland transcriptomes have focused on phytophagous insects such as the potato leaf hopper, Empoasca fabae [5], the green rice leaf hopper, Nephotettix cincticeps [47] and the western flower thrips, Frankliniella occidentalis [48]. For the mirid bugs, a study utilizing whole body transcriptome assembly of Lygus hesperus [49] and an EST library of Apolygus lucorum [50] was recently conducted. However, to our knowledge there is a lack of detailed information on the salivary gland transcriptome of TPB and neither does a complete genome sequence exist despite its economic importance.
A detailed proteomic analysis of the salivary proteins of the WTPB was recently conducted, which led to the identification of laccase, glucose dehydrogenase and xanthine dehydrogenase, in addition to the normal complement of PGs, α- amylases, and proteases [51]. The authors proposed that these newly identified enzymes could target plant-defense compounds.
Previous genetic studies of the TPB have used microsatellite markers to explore the genetic diversity of TPB populations from different geographical regions [52, 53]. To date, studies of transcripts have focused on RNAs isolated from the whole body or the gut [54–57]. Recently, a transcriptome was assembled for WTPB as a means to investigate the mechanisms of thermal tolerance [49].
In order to devise suitable genetic strategies for the effective control of TPB, it is important to have an in-depth knowledge of gene expression in salivary glands. While analysis of saliva has yielded considerable information on saliva contents, whole salivary gland transcriptomics should help reveal gene pathways involved in controlling saliva composition, production, and secretion. Such information, in turn, may help in the development of strategies that target specific TPB gene pathways involved in TPB-plant interactions. In this investigation, we present the salivary gland transcriptome (sialotranscriptome) of TPB. Additionally, we discuss gene expression patterns and metabolic pathways that could be targeted in reducing TPB-based damage.
Results and Discussion
RNA extraction, Illumina library preparation and sequencing
RNA extractions from the 3 pools of 25 gland pairs (Fig 1) yielded an average of 6 μg of total RNA. Non-denatured agarose gel electrophoresis and Agilent Bioanalyzer 2100 analysis (with Agilent RNA 6000 Nano kit) showed one predominant rRNA peak near 0.9 kb, characteristic of 28S rRNA cleavage and co-migration of the alpha 28 and beta 28 subunits with the 18S rRNA, a phenomena commonly observed in insect RNA isolations (S1 Fig) [58]. Illumina double-stranded library preparation yielded 1.4, 1.32, and 1.34 μg of library products for the respective pools with fragment sizes of 213, 207, and 212 bp, respectively. After sequencing, a total of 18,389,715 read pairs (paired ends) passed minimum quality requirements. Collectively, these paired end reads represent a total of 7,355,886,000 base pairs of sequence data.
Fig 1. Scanning electron micrograph of excised salivary glands of the tarnished plant bug Lygus lineolaris.
Short read transcriptome assembly
Read trimming with Trimmomatic [59] resulted in 24,744,346 (67.3%) reads meeting quality requirements. Of these, 19,368,620 (52.7%) reads remained paired and 5,375,726 (14.7%) were placed into unpaired single ends. Trinity [60, 61] transcriptome assembly of trimmed paired and unpaired reads resulted in the assembly of 77,171 transcripts. Curation of the assembled transcriptome by the methods of Yang and Smith (2013) [62], yielded 19,884 transcripts with BLASTX alignments to 403,496 different proteins in the Pterygota (winged insect) database downloaded from the UniProtKB [63]. Alignments allowed detection and subsequent trimming of 61 chimeric transcripts with the python scripts described by Yang and Smith 2013 [62]. Post assembly curation resulted in the final transcriptome assembly described in this study. This transcriptome called TPB_SG1, is composed of 62,559 transcripts with an N50 of 817 bp and a total size of 36.12 Mb (Table 1).
Table 1. Summary statistics of the sialotranscriptome of Lygus lineolaris.
| Total number of raw reads | 36,779,430 |
| Total number of trimmed reads (clean) | 24,744,346 |
| Total base pairs in assembly | 36.12 x 106 |
| Read length | 200 bp (paired end) |
| Total number of contigs | 62,559 |
| Mean length of contigs | 577 |
| N50 contig length | 817 |
| Minimum length | 201 |
| Maximum length | 13,408 |
Protein prediction and annotation
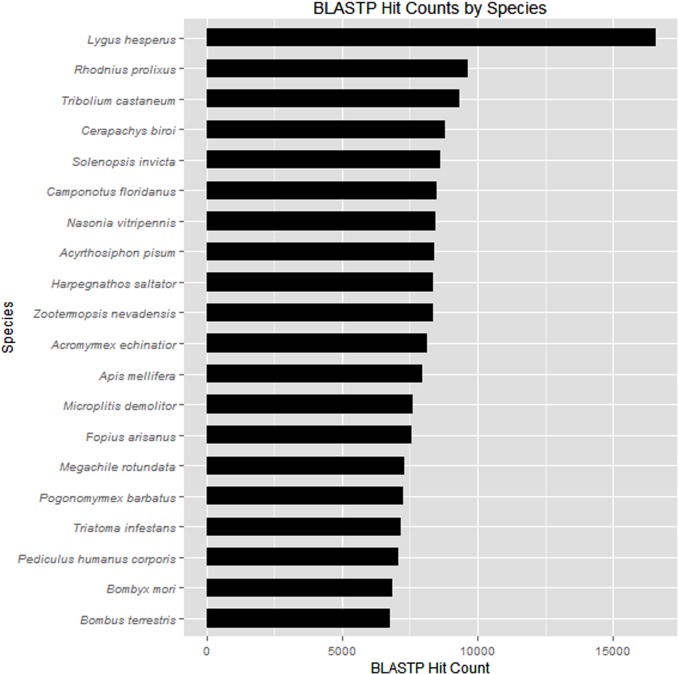
A total of 25,767 proteins were predicted (Table 2) on the sense strand of the transcripts with Transdecoder [61]. The predicted proteins had a total of 883,061 and 858,136 qualifying alignments to the NCBI nr and Uniprot-KB databases, respectively. The species showing the most BLASTP alignments to the TPB transcriptome were Lygus hesperus (n = 16,559), Rhodnius prolixus (n = 9,632), Tribolium castaneum (n = 9,335), and Cerapachys biroi (n = 8,824) (Fig 2) (S1 Table). GO association mapping from the BLASTP alignments yielded 11,090 GO terms to 1,097 predicted proteins. Interproscan identified 5,998 unique InterPro IDs for 11,694 predicted proteins and 20,332 GO terms to 7,512 predicted proteins. Of the 803 predicted proteins with a predicted signal peptide, 663 proteins were classified as being secreted since they did not contain transmembrane helices.
Table 2. Lygus lineolaris TPB_SG1 assembly predicted protein annotation summary.
| Predicted Proteins | 25,767 |
| Protein Completeness | |
| Complete | 7,069 |
| 5' partial | 5,512 |
| 3' partial | 3,591 |
| Internal | 9,595 |
| Proteins with BlastP Alignments | |
| NR | 15,600 |
| Uniprot | 19,526 |
| Either NR or Uniprot | 19,540 |
| Neither NR or Uniprot | 6,227 |
| Pfam Annotations | 12,851 |
| KEGG Annotations | 791 |
| GO Annotations | 31,240 |
| GO terms assigned by BlastP ID mapping | 11,090 |
| GO terms assigned by InterProScan | 20,150 |
| GO Biological Process terms | 13,405 |
| GO Molecular Function terms | 12,019 |
| GO Cellular Component terms | 5,816 |
| Proteins with SignalP | 803 |
| Proteins with SignalP w/o TMHMM | 663 |
Fig 2. Top 20 species exhibiting alignments (E-value ≤1.00E-05) to the L. lineolaris sialotranscriptome.
Comparative analysis
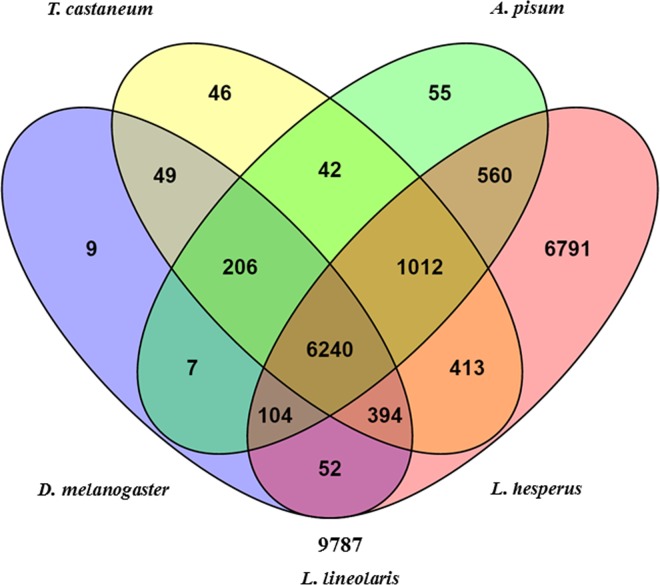
The predicted protein sequences derived from the salivary gland transcriptome of TPB were compared to protein sequences of WTPB as well as model insect species such as the fruit fly Drosophila melanogaster (Diptera), the confused flour beetle Tribolium castaneum (Coleoptera) and the pea aphid Acyrthosiphon pisum (Hemiptera) (Fig 3) [64–66]. Approximately 31.9% (8,226 BLASTP hits) of the 25,767 protein sequences of TPB exhibited sequence similarity with proteins from A. pisum, 32.6% (8, 402 BLAST hits) showed sequence similarity to T. castaneum, and 27.4% (7,061 BLASTP hits) shared significant similarity to proteins from D. melanogaster (Fig 3). Interestingly, within the blast results, D. melanogaster core RNA interference processing proteins Dicer2 Argonaute2 and loquacious were identified. Also, while TPB exhibited 6,446 sequences with high similarity to sequences in all three non-Hemipteran insects, it shared 15,566 BLASTP hits with WTPB (60.4%). The TPB_SG1 transcriptome had a CEGMA completeness of 90.32% for complete proteins and 94.35% when including partial proteins, which is comparable to other insects (Table 3).
Fig 3. Venn diagram showing distribution of 15,980 L. lineolaris salivary gland proteins with BLASTP alignments to Acyrthosiphon pisum, Drosophila melanogaster, Tribolium castaneum, and Lygus hesperus.
Table 3. CEGMA completeness report summary.
| Number of complete proteins | % Complete | Number of partial proteins | % partial | |
|---|---|---|---|---|
| TSA assemblies | ||||
| Lygus hesperus | 239 | 96.37 | 242 | 97.58 |
| Lygus lineolaris | 224 | 90.32 | 234 | 94.35 |
| Genome assemblies | ||||
| Acyrthosiphon pisum | 230 | 92.74 | 243 | 97.98 |
| Drosophila melanogaster | 241 | 97.18 | 245 | 98.79 |
| Tribolium castaneum | 220 | 88.71 | 293 | 96.37 |
Gene ontology
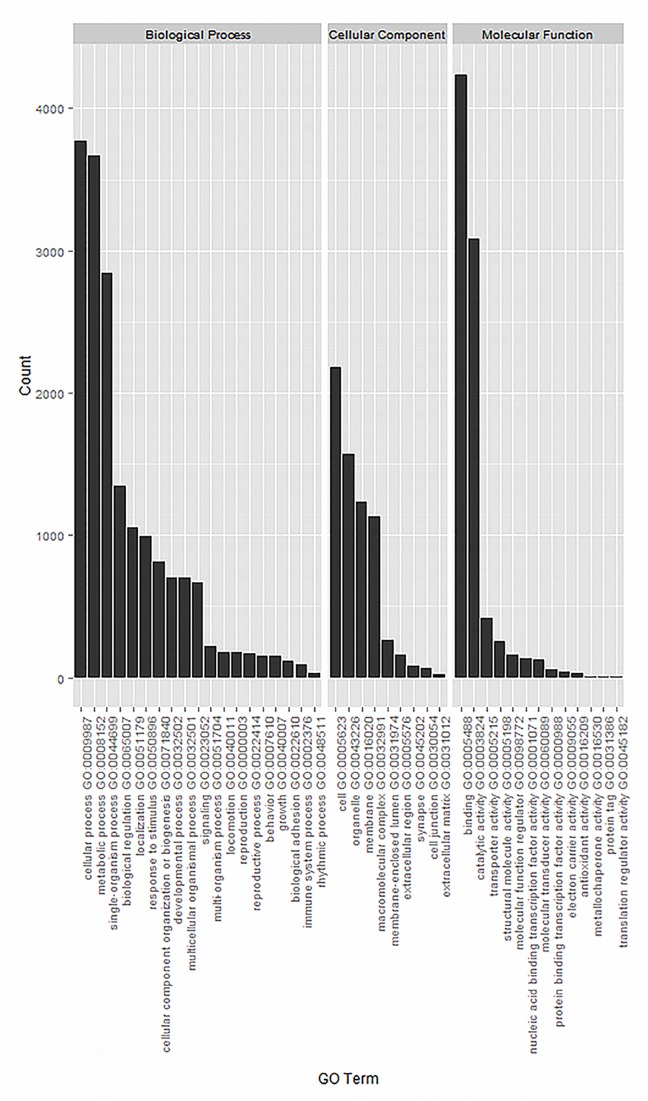
GO terms arising from both the BLASTP and InterProScan (ISA and IEA evidence codes, respectively) were combined for further analysis (S2 Table). The set of combined GO terms were subsequently summarized to GO term level 2 (Fig 4). In the category of biological processes there were 17,855 terms with the three most common categories assigned being cellular processes (3,773), metabolic processes (3,668) and single organism processes (2,842) (Fig 4). In the molecular function category (8,543 terms), the top three categories assigned were binding (4,237), catalytic activity (3,087) and transporter activity (415) (Fig 4). Finally, in the cellular component category (6,703 terms), the three predominant categories assigned are cell (2,183), organelle (1,566) and membrane (1,234). These most common assigned categories are consistent with other sialotranscriptomes among a wide range of phytophagous insects including; Bemisia tabaci, Empoasca fabae, Frankliniella occidentalis, Helicoverpa armigera, and Nilaparvata lugens [5, 48, 67, 68]. GO term enrichment for the predicted secreted proteins identified molecular function terms such as hydrolase activity (GO:0004553), polygalacturonase activity (GO:0004650), peptidase activity (GO:0008233), serine-type endopeptidase activity (GO:0004252) and catalytic activity (GO:0003824) to be overrepresented in the proteins predicted to be secreted (S3 Table). This high representation of GO molecular functions terms relating to proteolysis and degradation emphasizes the role of saliva in digestion, while providing further evidence of the physiological role of the saliva secreted by the SGs in the TPB herbivory.
Fig 4. Classification of L. lineolaris salivary gland transcriptome based on predicted Gene ontology (GO) terms.
(A) Biological Processes (B) Molecular Function (C) Cellular Component.
KEGG pathway
The Kyoto Encyclopedia of Genes and Genomes database was used to identify potential pathways represented in the transcriptome [69, 70]. A total of 791 proteins in the salivary gland transcriptome of TPB were mapped to a total of 107 KEGG pathways (S4 Table), the top 20 of which are depicted in Table 4. The majority of the salivary gland transcript sequences mapped to metabolic pathways including purine metabolism (239 predicted protein sequences with 34 enzymes), pyrimidine metabolism (229 predicted protein sequences with 26 enzymes) and aminoacyl-tRNA biosynthesis (77 predicted protein sequences with 22 enzymes). The presence of the latter indicates a high level of protein synthesis activity occurring in the SGs of the TPB. Consistent with the lacerate and flush feeding strategy, several of the enzymes found in predicted KEGG pathways are involved in sugar metabolism including: amino sugar and nucleotide sugar metabolism (17 enzymes with 41 predicted protein sequences) and glycolysis/gluconeogenesis (18 enzymes with 36 predicted protein sequences). Interestingly, there were also 12 enzymes with 48 predicted protein sequences which were involved with glutathione metabolism. This is consistent with the antioxidant Molecular Function roles assigned to 31 transcripts (Fig 4). It is expected that this high level of antioxidant activity would be necessary to quell the herbivory induced plant defenses which often involve reactive oxygen species [71–73].
Table 4. Top 20 predicted KEGG pathways in the L. lineolaris sialotranscriptome.
| KEGG pathway | Number of enzymes | Number of predicted proteins |
|---|---|---|
| Purine metabolism | 34 | 239 |
| Pyrimidine metabolism | 26 | 229 |
| Aminoacyl-tRNA biosynthesis | 22 | 77 |
| Glycolysis / Gluconeogenesis | 18 | 36 |
| Amino sugar and nucleotide sugar metabolism | 17 | 41 |
| Citrate cycle (TCA cycle) | 15 | 28 |
| Pentose phosphate pathway | 15 | 27 |
| Pyruvate metabolism | 15 | 22 |
| Carbon fixation pathways in prokaryotes | 15 | 23 |
| Glycerophospholipid metabolism | 14 | 24 |
| Cysteine and methionine metabolism | 13 | 23 |
| Methane metabolism | 13 | 21 |
| Alanine, aspartate and glutamate metabolism | 12 | 15 |
| Arginine and proline metabolism | 12 | 17 |
| Glutathione metabolism | 12 | 48 |
| Terpenoid backbone biosynthesis | 11 | 20 |
| Drug metabolism—other enzymes | 10 | 18 |
| Glycine, serine and threonine metabolism | 10 | 18 |
| Porphyrin and chlorophyll metabolism | 10 | 12 |
| Carbon fixation in photosynthetic organisms | 10 | 21 |
Protein domains
We identified 3,653 Pfam domains in 10,421 salivary gland transcriptome-predicted proteins resulting in 12,814 Pfam annotations (S5 Table). Among the top 20 Pfam domains were reverse transcriptases (RNA-dependent DNA polymerases) (281 proteins) indicative of retrotransposons of plant (Ty-1 copia or Ty-3 gypsy) or insect origin and/or retrovirus integrations (e.g. LyLV-1) in the TPB genome [74]. Reverse transcriptases allow retrotransposons to copy themselves to RNA and back to DNA that may integrate back to the genome. Zinc-finger double domains which help in protein dimerization (194 proteins) and are also involved in various cellular processes were noted in the salivary gland transcriptome (Table 5). Protein domains associated with sugar and other transporters were also identified (74 proteins). Fifty-seven TPB_SG1 transcripts associated with protein domains related to cytochrome P450 and 31 transcripts associated with protein domains of carboxylesterase were identified (S5 Table); unlike the sialotranscriptome of the potato leaf hopper [5], cytochrome P450 and carboxylesterase domains were not in the top 20 domain classes recognized by TPB_SG1. One-hundred-and-fifty TPB_SG1 transcripts are associated with the large and rapidly expanding protein family known as the WD-domain G-beta repeats [75]. The WD-repeat proteins participate in biological processes such as signal transduction, transcription regulation, and apoptosis, although the specific function of the WD-repeat domain is unclear [75]. More knowledge of WD-repeat proteins is critical to understanding many biological processes including those that take place in the sialotranscriptome of TPB.
Table 5. Top Pfam domains identified in L. lineolaris sialotranscriptome predicted proteins.
| PFAM ID | PFAM ID Description | Number of proteins |
|---|---|---|
| PF00078 | Reverse transcriptase (RNA-dependent DNA polymerase) | 281 |
| PF13465 | Zinc-finger double domain | 194 |
| PF00665 | Integrase core domain | 187 |
| PF00096 | Zinc finger, C2H2 type | 166 |
| PF00069 | Protein kinase domain | 163 |
| PF00400 | WD domain, G-beta repeat | 150 |
| PF13894 | C2H2-type zinc finger | 93 |
| PF07727 | Reverse transcriptase (RNA-dependent DNA polymerase) | 91 |
| PF00076 | RNA recognition motif. (a.k.a. RRM, RBD, or RNP domain) | 87 |
| PF12796 | Ankyrin repeats (3 copies) | 79 |
| PF00083 | Sugar (and other) transporter | 74 |
| PF00651 | BTB/POZ domain | 72 |
| PF00271 | Helicase conserved C-terminal domain | 71 |
| PF07679 | Immunoglobulin I-set domain | 68 |
| PF00089 | Trypsin | 62 |
Proteins of interest
The TPB sialotranscriptome was screened for several known salivary proteins found in phytophagous insects (S6 Table). Sequence comparisons revealed 36 serine protease snake-like proteins, 31 ATP-dependent zinc metalloproteases, 28 polygalacturonases, 16 cathepsins, and 16 esterase FE-4-like proteins (Table 6). The salivary protein encoding transcripts/ proteins were categorized as those involved in general digestion, sugar metabolism, cell wall digestion, immunity-related responses, detoxification, suppression of plant defense responses, and other phytophagy processes (specific functions unknown). Interestingly, pectin lyases and endoglucanases were not present in the sialotranscriptome of TPB; such proteins were observed in the sialotranscriptome of E. fabae, the potato leaf hopper [5]. However, we did record other glycan degrading enzymes that would possibly target the hemicellulose though they were not represented in the top 20 salivary proteins / enzymes (S4 and S5 Tables). The high representation of serine protease snake-like proteins (36 proteins) in the TPB_SG1 dataset was a bit surprising as these putative immune-response proteins were reported earlier from an immunogenomics study in the squash bug, Anasa tristis (De Geer) [76]. However, serine protease snake-like proteins were represented in the sialotranscriptome of the rice brown plant hopper, N. lugens [68]. In addition to transcripts/proteins involved in general digestive processes and sugar metabolism, genes involved in degradation of plant cell wall components and those involved in detoxification and inhibition of plant defense responses were well represented in the TPB sialotranscriptome (Table 6).
Table 6. Genes of interest identified in the L. lineolaris sialotranscriptome.
| Functional category of genes | Candidate genes | Proteins | Number of TPB SG1 BlastP Hits |
|---|---|---|---|
| General digestion | cathepsin-L, partial | ABF18889.1 | 16 |
| esterase FE4-like | XP_001943214.2 | 16 | |
| cathepsin-L-like cysteine proteinase 2 | ABF18890.1 | 12 | |
| probable chitinase 3 isoform X1 | XP_001943038.2 | 12 | |
| short/branched chain specific acyl-CoA dehydrogenase, mitochondrial | XP_001947176.2 | 8 | |
| lipase 3-like isoform X2 | XP_003246825.1 | 4 | |
| granzyme-like protein 1 | XP_003247717.1 | 3 | |
| carboxypeptidase D-like | XP_001952348.1 | 2 | |
| Sugar metabolism | similar to Lactase-phlorizin hydrolase precursor (Lactase-glycosylceramidase) | XP_001945606.1 | 12 |
| sucrase | ABB55878.1 | 7 | |
| lysosomal alpha-glucosidase-like | XP_001952631.1 | 3 | |
| Alpha-amylase_C | BAH72207.1 | 1 | |
| Extra-oral digestion of cell wall components | polygalacturonase | ACC44799.1 ACC44798.1 ABD63920.1 ABD63922.1 ACZ28127.1 | 28 |
| uncharacterized family 31 glucosidase KIAA1161-like | XP_001952674.2 | 5 | |
| Immune related | serine protease snake-like | XP_003247331.1 | 36 |
| Detoxification and inhibition of plant defenses | ATP-dependent zinc metalloprotease YME1 homolog isoform X1 | XP_001946697.2 | 31 |
| glucose oxidase | NP_001011574.1 | 11 | |
| Xanthine dehydrogenase | P08793.1 | 4 | |
| regucalcin-like | NP_001155519.1 | 3 | |
| laccase-7-like & laccase 2 | XP_001946224.1 BAJ83488.1 | 3 | |
| Unknown function | unknown [Lygus lineolaris] | ABQ18257.1 | 4 |
| unknown [Lygus lineolaris] | ABQ18254.1 | 3 | |
| apyrase-like, partial | XP_001947326.3 | 1 |
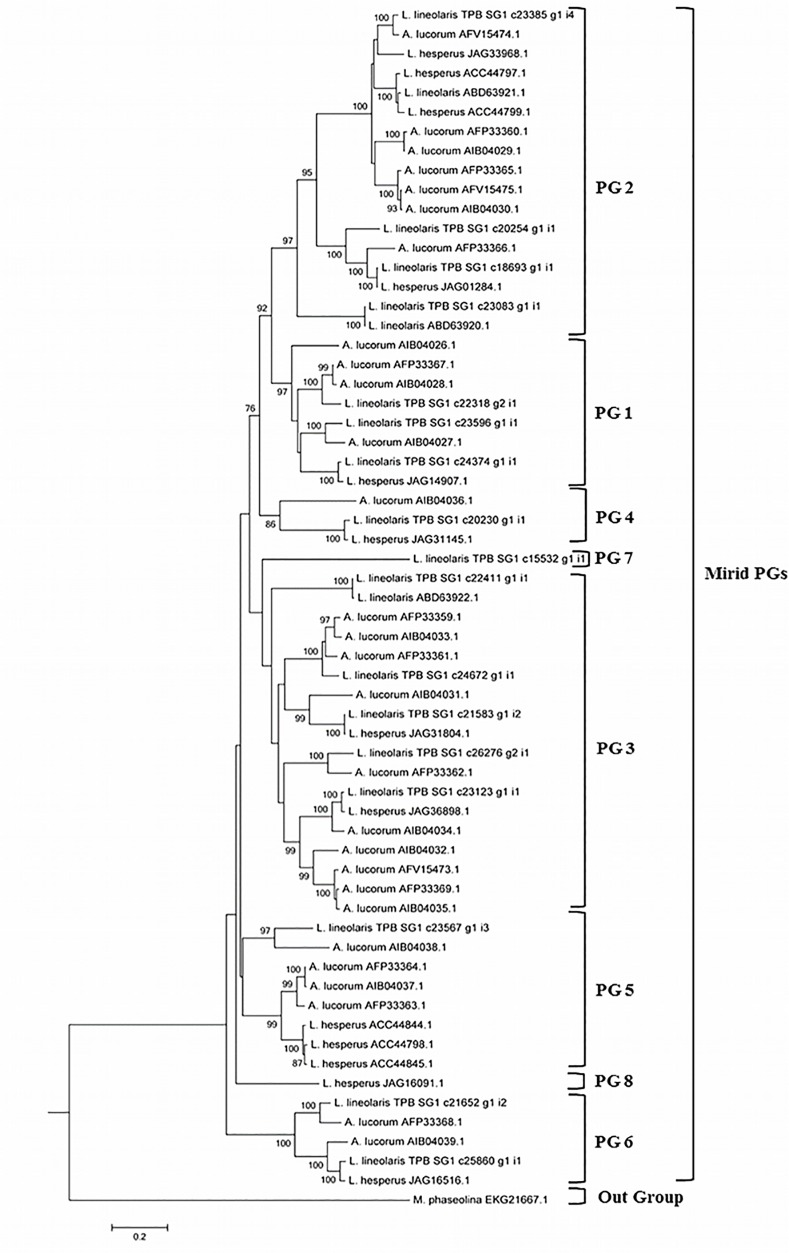
Twenty eight polygalacturonase proteins were identified in the TPB sialotranscriptome, of which 17 were predicted full-length protein sequences. A neighbor-joining tree using Poisson-correction distances was constructed from a multiple sequence alignment using the full length polygalacturonase proteins from mirid bugs including TPB, WTPB, and Apolygus lucorum, where the Macrophomina phaseolina glycoside hydrolase (GH) domain was used as an out group. The mirid bug polygalacturonases in this study were divided into 8 clusters (polygalacturonase 1–8) of which 7 clusters contained representatives from the TPB sialotranscriptome (Fig 5). The previously described TPB polygalacturonase proteins ABD63920 and ACZ28127 clustered into the polygalacturonase 2 cluster that contained four TPB salivary gland transcriptome polygalacturonase protein sequences. The other previously described TPB polygalacturonase protein ABD63922, clustered into the polygalacturonase 3 cluster, where it paired with a TPB_SG1 protein. Polygalacturonase 8 from the WTPB formed a cluster by itself.
Fig 5. Phylogenetic relationships of polygalacturonase transcripts of L. lineolaris with polygalacturonases from other mirid bugs.
The plant cell wall is a resilient and structurally heterogeneous barrier composed of complex polysaccharides and diverse proteins [77]. The main components of the primary cell walls are members of two polysaccharide networks, consisting of cellulose and hemicellulose embedded in a pectin polysaccharide matrix [78]. To breach this complex barrier, herbivorous insects have evolved a remarkable array of polysaccharide degrading enzymes, including exo- and endo-polygalacturonases, pectin methyl-esterases, pectin lyases and pectate lyases, acetyl esterases, xylanases, and a variety of endoglucanases that cleave cellulose, xyloglucan, and other glucans [79–81]. The term cellulases is used to describe cellulolytic enzymes such as endogluconases, exogluconases, and β-glucosidases. In addition to cellulases, degradation of plant cell walls requires pectinases and hemicellulases. These are all grouped into the glycoside hydrolase (GH) family according to their amino acid sequence similarities and their folding patterns based on the Carbohydrate-Active enZymes Database [82]. Cellulases in the GH9 family are found in most insect orders while polygalacturonases of the GH28 family have a much more restricted distribution in insects [83]. Homogalacturonan polymers are the main components of pectin in primary cell walls, and the polygalacturonases identified in this study presumably cleave the 1,4-linkages of the homogalacturonan α-D-galacturonic acid [83]. A phylogenetic analysis has revealed that the polygalacturonases of the mirid bugs cluster with the ascomycete Pezizomycotina + Saccharomycotina clade with yeasts (Saccharomycetacease) as an outgroup [79]. The GH28 family of glycosyl hydrolases includes proteins with different enzymatic specificities including polygalacturonases, rhamnogalacturonases, and xylogalacturonases, but based on phylogenetic analyses and conservation of amino acid residues it appears that this entire gene family had a single origin in the tree of life [83, 84]. It has been demonstrated however that some conserved amino acid sequences enable a distinction between polygalacturonasess and other pectolytic enzymes of the GH28 family [85].
As previously suggested [79], it is possible that polygalacturonase genes in the mirids were obtained by horizontal gene transfer (HGT) as all mirid polygalacturonases have characteristic amino acids conserved in microbial and plant polygalacturonases. The large repertoire of polygalacturonase transcripts in the TPB sialotranscriptome raises the interesting possibility that some of these may actually be translated into catalytically inactive proteins that can act as “decoy” targets for plant derived polygalacturonase inhibitory proteinases suggested earlier [86]. However, this hypotheses remains to be tested. Current work in our laboratory is underway to test if all the PG transcripts eventually translate to functional proteins or if some of them serve as “decoys”.
The second most common sequences in the TPB sialotranscriptome were ATP-dependent zinc metalloproteases. These enzymes have been implicated in mitochondrial protein metabolism and protection of mitochondria from the accumulation of oxidatively damaged membrane proteins [87]. The presence of ATP-dependent zinc metalloproteases is consistent with the fact that herbivory would induce the formation of reactive oxygen species (ROS) at localized areas in the plant tissue, and such ROS could cause damage to the insect if not neutralized [72].
Eleven protein sequences of glucose oxidase (GOX EC 1.1.3.4) were found in the TPB sialotranscriptome. GOX is the first insect salivary enzyme that has been shown to suppress wound-inducible herbivore defenses of plants [88]. Salivary GOX secreted by Helicoverpa zea was demonstrated to inhibit wound induced nicotine production in Nicotiana tabacum as well as delayed–induced defenses in the tomato plant Solanum lycopersicum [88–90]. Salivary GOX is found in many caterpillar species and also in aphids [6, 91, 92]
Signature Pfam domains (PF00199, PF10417) for ROS scavenger enzymes such as catalase (1.11.1.6) and peroxiredoxin (1.11.1.15) were identified in the salivary gland transcriptome. Insect feeding has been shown to increase peroxidase activity in the cotton leaves and sap [93]. The cotton transcript encoding catalase has previously been shown to be down regulated in response to feeding by both a sap-feeder, Aphis gossypii and a chewing insect, Bemisia tabacci [94]. Thus, we hypothesize that through salivary GOX and ROS scavengers, TPB may be limiting ROS production and suppressing ROS-triggered defensive responses in the host plants. Such a strategy would help the TPB successfully establish itself by suppressing host-plant induced defenses during feeding.
Xanthine dehydrogenase (XDH) has been reported in insects and has been most well characterized in D. melanogaster and the silkworm Bombyx mori [95–99]. In contrast to XDH in vertebrates, insect XDH does not appear to be converted into xanthine oxidase. Insect XDH functions in the catabolism of purines and detoxification of dietary purines which can be converted into the powerful antioxidant uric acid in the presence of NAD+. Thus, XDH and uric acid are secreted on the mucosal surface of the salivary gland thereby expanding their function extracellularly and into the saliva [100]. Interestingly, three regucalcin-like transcripts were also found in the sialotranscriptome of the TPB. Regucalcin is a Ca2+ binding protein that has been also reported in the saliva of A. pisum, B. tabaci and F. occidentalis [48, 67, 101, 102].It is hypothesized that regucalcin might have a role in regulation of calcium mediated signaling or in inhibiting calcium-mediated defenses in plant cells in response to herbivory [103]. Several genes with unknown functions were also found such as apyrase-like (1 protein). It is unclear what function the apyrase-like gene would perform in the saliva of a phytophagous insect. In blood sucking insects, salivary apyrases have been reported to play a role in inhibiting ADP and inhibition of ADP-induced platelet aggregation [2, 104]. When the TPB apyrase-like protein was aligned to the NCBI nr database it showed the highest similarity to an apyrase-like protein present in the bean bug, Riptortus pedestris, suggesting that the function of this protein is likely shared among other phytophagous insects. Apyrases are also been known to be ATP degrading enzymes and they can modulate the level of extracellular ATP (eATP) which can impact pathogen defense response in plants [105–107]. For example, apyrase in TPB saliva may degrade eATP in host plants that may impact cell viability and death as well as compromise basal resistance to viral and bacterial infection.
Interestingly some endonucleases were detected but again these were not represented to the extent that they figured in the top 20 proteins of interest. It has been demonstrated earlier that the saliva of TPB digests double stranded ribonucleic acids [108]. We presume that the endonucleases we detected (showing homology to dicer2 and argonaute2 which are known nucleases) could possibly function in degrading the double stranded ribonucleic acids. This would pose a problem for delivery of dsRNA targeted against any major gene in TPB since they would be rapidly degraded by saliva and have very limited persistency in the digestive tract.
Conclusions
Our transcriptomic analysis of the salivary glands of TPB provides biological insights into the means by which this mirid bug establishes a feeding site on a host plant. Catalytic enzymes associated with digestion were highly represented in the TPB sialotranscriptome, which is consistent with the extra-oral digestion and solubilization of nutrients from the plant tissue. In addition to salivary proteins and enzymes involved in general metabolism, we also show the presence of several immune related transcripts with possible involvement in counteracting plant defense responses. Phylogenetic evidence revealed the complement of TPB polygalacturonase enzymes is similar to that of other mirid bugs such as Apolygus lucorum and the WTPB, and constitutes a large expanded family of polygalacturonases. While we have validated some of the transcripts given in this assembly (mainly the polygalacturonases), our future work is focused on elucidating the role of GOX and regucalcin in this important pest insect (data not shown) since these likely target host-plant defenses. The TPB sialotranscriptome is thus a valuable resource for further research on TPB and its interaction with host plants.
Materials and Methods
Insect collection and rearing
Lygus lineolaris (tarnished plant bugs or TPBs) used in this research were obtained from a long term colony housed at the Mississippi State University Insect Rearing Center. TPBs were reared in 8.3 L plastic containers (Rubbermaid Servin' Saver®) filled with shredded paper and equipped with self-sealing lids modified by removing the center of the lid. A fine mesh screen was placed on the containers and held down with the remaining portion of the self-sealing lid. Screen covers and shredded paper were replaced weekly. Insect colonies were kept in a rearing chamber maintained at 26.7 ± 2°C at an approximate relative humidity of 65 ± 5% with a 16:8 light: dark cycle. The insects were fed an oligidic artificial diet similar to that of Cohen (2000) presented on a 5 x 5 cm Parafilm (Beemis Company Inc., Neenah, WI), and placed on top of the screens [109]. The insects fed by puncturing the Parafilm. Food was replaced three times per week. Mixed sex adults were separated from the colony as soon as they eclosed and utilized for the experiments within 2–3 days
RNA extraction, Illumina library preparation and sequencing
Three different Illumina libraries were generated. For each library, salivary gland pairs from 25 individuals were dissected on dry ice, total RNA was extracted using an RNeasy Mini Kit (CAT# 74104, Qiagen, Valencia, CA, USA), and RNA was treated with 20 units of Takara Recombinant DNase I (CAT# 2270A, Clontech Laboratories Inc., Mountain View, CA, USA). RNA integrity was assessed using agarose gel electrophoresis and the Agilent Bioanalyzer 2100 using Agilent RNA 6000 Nano kit (CAT# 5067–1511, Agilent Technologies, Palo Alto, CA, USA). Illumina strand specific libraries were prepared with the Illumina Stranded mRNA library construction kit (CAT# RS-122-2101, Illumina, San Diego, CA, USA) according to the manufacture’s protocol using 3 μg of total RNA. Molar concentrations of resulting DNA libraries were determined using fragment sizes determined via an Agilent Bioanalyzer 2100 with an Agilent DNA 1000 kit (CAT# 5067–1504, Agilent Technologies, Palo Alto, CA, USA). DNA concentrations were determined using Qubit fluorometer with the Qubit dsDNA HS assay kit (CAT# Q32851, Life Technologies, Grand Island, NY, USA). The three libraries were pooled at equal concentration and volume for 15 pM cluster generation followed by paired end (PE) 200 bp (2 X 200) sequencing on the Illumina MiSeq with the Illumina MiSeq Reagent kit v3 (CAT# MS-102-3003, Illumina, San Diego, CA, USA). All raw read data was deposited in the National Center for Biotechnology Information (NCBI) under BioProject PRJNA280549 and BioSample SAMN03464333 with Sequence Read Archive (SRA) [110] accession numbers; SRR1956749, SRR1956751, and SRR1956752.
Short read transcriptome assembly
Raw demultiplexed reads were trimmed with Trimmomatic-PE software using parameters “ILLUMINACLIP: TruSeq3-PE.fa:2:30:10 LEADING: 28 TRAILING: 28 SLIDINGWINDOW: 4: 28 MINLEN: 140” [59]. All trimmed paired and unpaired reads were assembled together in a single assembly with Trinity release r20140717 using the strand specific flag and minimum contig length of 200 bp option [60]. Subsequent assembly curation was performed to remove redundant transcripts and trim miss-assembled chimeras as described by Yang and Smith (2013) [62]. Briefly, RNA-Seq by Expectation Maximization (RSEM, version 1.2.15) values calculated for Bowtie2 (v 2.2.5) mapping of trimmed paired reads were determined for each transcript using the perl script align_and_estimate_abundance.pl provided in the Trinity release [61, 111, 112]. Subsequently, the highest expressed isoform for each Trinity component (gene) was chosen as a representative of all the isoforms within the subcomponent assignment. The resulting highest RSEM isoform transcripts were aligned with BLASTX (NCBI BLAST version 2.2.29) to all the Insecta sub-class Pterygota (Lang 1888) (winged insects) 1,097,562 protein sequences available in UniProtKB (downloaded 11/14/14) using parameters previously described in Yang and Smith (2013) [62]. The BLASTX alignments with the Pterygota database was used in chimera identification and trimming using the python scripts described in Yang and Smith (2013) [62]. Transcripts containing a BLASTN hit to Illumina adapter sequences in the NCBI Univec database using VecScreen specific parameters were removed from the assembly. The resulting transcripts that met repository requirements were deposited as a Transcriptome Shotgun Assembly (TSA) in DDBJ/EMBL/GenBank under the accession GDAW00000000. The version described in this paper is the first version, GDAW01000000 (http://www.ncbi.nlm.nih.gov/Traces/wgs/wgsviewer.cgi?download=GDAW01.1.fsa_nt.gz).
Protein prediction and annotation
Transcripts were translated in silico from the sense strand with TransDecoder (version 2.0.1) while retaining open reading frames with hmm Pfam-A domains (release 27.0) and BLASTP alignments to (UniProt release 2015_04) [61, 63, 113]. The resulting predicted proteins were searched for previously identified protein signatures with Gene3D (version 3.5.0), PANTHER (version 9.0), Pfam (version 27.0), PIRSF (3.01), PRINTS (42.0), Prosite patterns and profiles (20.105), SMART (version 6.2), SUPERFAMILY (1.75), and TIGRFAMs (15.0) applets within InterProScan v51.0. (PRINTS, PANTHER, TIGRFAM, SUPERFAMILY, PIRSF, Gene3D, PrositeProfiles, PrositePatterns, SMART, InterProScan) [113–126]. The resulting Enzyme Commission Numbers (EC), Gene Ontology (GO) terms, and KEGG (Kyoto Encyclopedia of Genes and Genomes) pathway mapping were parsed from the InterProScan signatures. Experimental evidence based GO terms (i.e. GO evidence codes: EXP, IDA,IPO.IMP,IGI and IEP) were associated to the predicted proteins that had a BLASTP alignment to the NCBI nr (downloaded 04/02/2015) and UniProtKB (version 2015_04) databases, using the NCBI blast 2.2.29 with parameters “-evalue 1.00E-05 -max_target_seqs 100 -word_size 3 -gapopen 11 -gapextend 1 -matrix BLOSUM62 and the additional criteria of 70% percent identity and at least 50% query coverage. Taxonomic IDs of protein blast hits were counted by species. All GO term annotations were subject to GO consortium quality control taxon checks; invalid GO term associations were removed following GO consortium recommendations [127]. Signal peptides and transmembrane helixes were identified with SignalP 4.1 and TMHMM, respectively [128, 129]. GO term enrichment for the potential secreted proteins i.e. proteins that contain a predicted signal peptide and lack transmembrane helices, was conducted using GOStats [130].
Comparative analysis
Genomes and predicted proteins were downloaded for D.melanogaster (Diptera), the confused flour beetle T. castaneum (Coleoptera), and the pea aphid A. pisum (Hemiptera) from the NCBI Genomes database. Additionally, the WTPB TSA assembly (version GBHO00000000.1) and the available 31,830 protein sequences in Uniprot-KB (accessed 5/13/15) were obtained for analysis. Completeness of the TPB and WTBP transcriptomes as well as the reference genomes from the genome projects (above) were assessed using Core Eukaryotic Genes Mapping Approach (CEGMA) version 2.5 [131]. TPB predicted proteins were aligned with BLASTP to the predicted proteomes and WTPB protein sequences. Only alignments with a bitscore of ≥100 were considered further.
Proteins of interest
Proteins of interest involved in functions such as digestion, detoxification, inhibition of plant defense, digestion of cell wall components and sugar metabolism (previously described by Cooper et al., 2013 [51] and Stafford-Banks et al., 2014 [48]) were aligned with BLASTP to the TPB_SG1 predicted proteins only alignments with a bitscore ≥ 60 were accepted [48, 51].
Polygalacturonases
Full length polygalacturonase protein sequences previously reported for mirid bugs A. lucorum, WTPB, and TPB as well as 17 full-length protein polygalacturonase TPB_SG1 sequences were subjected to a phylogenetic analysis using procedures described by Zhang et al. (2015) for the characterization of polygalacturonase genes in A. lucorum [50]. The presence of the polygalacturonase domain was confirmed in all the Mirid bug polygalacturonase sequences with the NCBI Conserved Domain (CD) search of the NCBI Conserved Domain Database (CDD) v3.13, default parameters [132]. The Macrophomina phaseolina MS6 glycoside hydrolase protein sequence EKG21667.1 served as the outgroup [50]. MEGA 6.0 software was used to align the complete polygalacturonase protein sequences with ClustalW and construct a neighbor-joining phylogenetic tree, using default parameters, with the single exception of bootstrapping the tree with 1000 replicates [133–135]. The phylogenetic tree was visualized with MEGA 6.0 software [135].
Supporting Information
(TIF)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
Acknowledgments
The authors would like to acknowledge the AgBase for assistance with data analysis, including functional analysis and GO annotations.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This research was supported by the start-up funds from Mississippi State University No. 269110-151250 from National Science Foundation, Experimental Program to Stimulate Competitive Research, and the Mississippi State University Schillig special teaching grant 365082 (N.K.), and in part through funding from United States Department of Agriculture, Agricultural Research Service awards 6402-21310-003-24S and 6402-21310-003-18S (D.G.P), Grant No. 038/2014/P from the Grant Agency of the University of South Bohemia (A.B.). The stay of A.B. in the lab of N.K. was partially covered from the American Science Information Center, Czech-American Science and Technology Cooperative grant KONTAKT No. LH 14047. The funding authorities had no role in the design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Ribeiro JM (1987) Role of saliva in blood-feeding by arthropods. Annu Rev Entomol l 32: 463–478. [DOI] [PubMed] [Google Scholar]
- 2.Ribeiro JMC (1995) Insect Saliva: Function, Biochemistry, and Physiology In: Chapman RF, de Boer G, editors. Regulatory mechanisms in insect feeding: Springer US; pp. 74–97. [Google Scholar]
- 3.Wheeler AG (2001) Biology of the plant bugs (Hemiptera: Miridae): pests, predators, opportunists Ithaca, New York: Cornell University Press. [Google Scholar]
- 4.Alfano JR (2009) Roadmap for future research on plant pathogen effectors. Mol Plant Pathol. 10: 805–813. 10.1111/j.1364-3703.2009.00588.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.DeLay B, Mamidala P, Wijeratne A, Wijeratne S, Mittapalli O, Wang J, et al. (2012) Transcriptome analysis of the salivary glands of potato leafhopper, Empoasca fabae. J Insect Physiol 58: 1626–1634. 10.1016/j.jinsphys.2012.10.002 [DOI] [PubMed] [Google Scholar]
- 6.Eichenseer H, Mathews MC, Bi JL, Murphy JB, Felton GW (1999) Salivary glucose oxidase: multifunctional roles for Helicoverpa zea? Arch Insect Biochem Physiol 42: 99–109. [DOI] [PubMed] [Google Scholar]
- 7.Hogenhout SA, Bos JIB (2011) Effector proteins that modulate plant–insect interactions. Curr Opin Plant Biol 14: 422–428. 10.1016/j.pbi.2011.05.003 [DOI] [PubMed] [Google Scholar]
- 8.Hogenhout SA, Van der Hoorn RAL, Terauchi R, Kamoun S (2009) Emerging concepts in effector biology of plant-associated organisms. Mol Plant Microbe Interac 22: 115–122. [DOI] [PubMed] [Google Scholar]
- 9.Hori K (1992) Insects secretion and their effect on plant growth, with special reference to hemipterans Biology of Insect-Induced Galls. New York, NY: Oxford University Press; pp. 157–170. [Google Scholar]
- 10.De Vos M, Jander G (2009) Myzus persicae (green peach aphid) salivary components induce defence responses in Arabidopsis thaliana. Plant Cell Environ 32: 1548–1560. 10.1111/j.1365-3040.2009.02019.x [DOI] [PubMed] [Google Scholar]
- 11.Miles PW (1999) Aphid saliva. Biol Rev 74: 41–85. [Google Scholar]
- 12.Young OP (1986) Host plants of the tarnished plant bug, Lygus lineolaris (Heteroptera: Miridae). Ann Entomol Soc Am 79: 747–762. [Google Scholar]
- 13.Snodgrass GL, Scott WP, Smith JW (1984) Host plants and seasonal distribution of the tarnished plant bug (Hemiptera: Miridae) in the delta of Arkansas, Louisiana, and Mississippi Environ Entomol 13: 110–116. [Google Scholar]
- 14.Strong FE (1970) Physiology of injury caused by Lygus hesperus. J Econ Entomol63: 808–814. [Google Scholar]
- 15.Hanny BW, Cleveland TC, Meredith WR (1977) Effects of tarnished plant bug, (Lygus lineolaris), infestation on presquaring cotton (Gossypium hirsutum) Environ Entomol 6: 460–462. [Google Scholar]
- 16.Layton MB (2000) Biology and damage of the tarnished plant bug, Lygus lineolaris, in Cotton. Southwest Entomol 23: 7–20. [Google Scholar]
- 17.Williams L, Phillips JR, Tugwell NP (1987) Field technique for identifying causes of pinhead square shed in cotton. J Econ Entomol 80: 527–531. [Google Scholar]
- 18.Williams L 3rd, Tugwell NP (2000) Histological description of tranished plant bug (Heteroptera: Miridae) feeding on small cotton floral buds. J Entomol Sci 35: 187–195. [Google Scholar]
- 19.Snodgrass GL (1996) Insecticide resistance in field populations of the tarnished plant bug (Heteroptera: Miridae) in cotton in the Mississippi delta. J Econ Entomol 83: 783–790. [DOI] [PubMed] [Google Scholar]
- 20.Snodgrass GL, Abel C, Jackson R, Gore J (2008) Bioassay for determining resistance levels in tarnished plant bug populations to neonicotinoid insecticides. Southwest Entomol 33: 173–180. [Google Scholar]
- 21.Snodgrass GL, Gore J, Abel CA, Jackson R (2009) Acephate resistance in populations of the tarnished plant bug (Heteroptera: Miridae) from the Mississippi river delta. J Econ Entomol 102: 699–707. [DOI] [PubMed] [Google Scholar]
- 22.Snodgrass GL, Scott WP (2000) Seasonal changes in pyrethroid resistance in tarnished plant bug (Heteroptera: Miridae) populations during a three-year period in the delta area of Arkansas, Louisiana, and Mississippi. J Econ Entomol 93: 441–446. [DOI] [PubMed] [Google Scholar]
- 23.Snodgrass GL, Scott WP (2003) Effect of ULV Malathion use in boll weevil (Coleoptera: Curculionidae) eradication on resistance in the tarnished plant bug (Heteroptera: Miridae). J Econ Entomol 96: 902–908. [DOI] [PubMed] [Google Scholar]
- 24.Snodgrass GL (2003) Role of reproductive diapause in the adaption of the tarnished plant bug (Heteroptera: Miridae) to its winter habitat in the Mississippi river delta. Environ Entomol 32: 945–952. [Google Scholar]
- 25.Gore J, Catchot A, Musser F, Greene J, Leonard BR, Cook DR, et al. (2012) Development of a plant-based threshold for tarnished plant bug (Hemiptera: Miridae) in cotton. J Econ Entomol 105: 2007–2014. [DOI] [PubMed] [Google Scholar]
- 26.Musser FR, Catchot AL, Stewart SD, Bagwell RD, Lorenz GM, Tindall KV, et al. (2009) Tarnished plant bug (Hemiptera: Miridae) thresholds and sampling comparisons for flowering cotton in the midsouthern United States. J Econ Entomol 102: 1827–1836. [DOI] [PubMed] [Google Scholar]
- 27.Musser FR, Lorenz GM, Stewart SD, Bagwell RD, Leonard BR, Catchot AL, et al. (2009) Tarnished plant bug (Hemiptera: Miridae) thresholds for cotton before bloom in the midsouth of the United States. J Econ Entomol 102: 2109–2115. [DOI] [PubMed] [Google Scholar]
- 28.Bourland F, Myers GO (2015) Conventional Cotton Breeding. In: Fang DD, Percy RG, editors, Cotton. 2nd Edition, Madison, WI, USA pp. 205.
- 29.Bourland FM, Jones DC (2013) Registration of ‘UA103’ Cotton Cultivar. J Plant Registr 7: 135–139. [Google Scholar]
- 30.Bourland FM, Jones DC (2015) Registration of Arkot 0305, Arkot 0306, Arkot 0309, and Arkot 0316 germplasm lines of cotton. J Plant Registr 9:94–98. [Google Scholar]
- 31.Bourland FM, Tugwell NP (1999) Evaluation of injury by tarnished plant bugs (Lygus lineolaris Palisot de Beauvois) to blended cotton genotypes. J Cotton Sci 3: 171–176. [Google Scholar]
- 32.Maredia KM, Tugwell NP, Waddle BA, Bourland FM (1994) Technique for screening cotton germplasm for resistance to tarnished plant bug, Lygus lineolaris (Palisot de Beauvois). Southwest Entomol 19: 63–70. [Google Scholar]
- 33.de la Paz Celorio-Mancera M, Carl Greve L, Teuber LR, Labavitch JM (2009) Identification of endo- and exo-polygalacturonase activity in Lygus hesperus (Knight) salivary glands. Arch Insect Biochem Physiol 70:122–135. 10.1002/arch.20282 [DOI] [PubMed] [Google Scholar]
- 34.Shackel KA, de la Paz Celorio-Mancera M, Ahmadi H, Greve LC, Teuber LR, Backus EA, et al. (2005) Micro-injection of Lygus salivary gland proteins to simulate feeding damage in alfalfa and cotton flowers. Arch Insect Biochem Physiol 58: 69–83. [DOI] [PubMed] [Google Scholar]
- 35.de la Paz Celorio-Mancera M, Allen M, Powell A, Ahmadi H, Salemi M, Phinney B, et al. (2008) Polygalacturonase causes lygus-like damage on plants: cloning and identification of western tarnished plant bug (Lygus hesperus) polygalacturonases secreted during feeding. Arthropod Plant Interact 2: 215–225. [Google Scholar]
- 36.Allen ML, Mertens JA (2008) Molecular cloning and expression of three polygalacturonase cDNAs from the tarnished plant bug, Lygus lineolaris. J Insect Sci 8: 1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Walker WB, Allen ML (2010) Expression and RNA interference of salivary polygalacturonase genes in the tarnished plant bug, Lygus lineolaris. J Insect Sci 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Francischetti IMB, Pham VM, Mans BJ, Andersen JF, Mather TN, Lane RS, et al. (2005) The transcriptome of the salivary glands of the female western black-legged tick Ixodes pacificus (Acari: Ixodidae). Insect Bochem Mol Biol 35: 1142–1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Valenzuela JG, Francischetti IMB, Pham VM, Garfield MK, Mather TN, Ribeiro JMC (2002) Exploring the sialome of the tick Ixodes scapularis. J Exp Biol 205: 2843–2864. [DOI] [PubMed] [Google Scholar]
- 40.Arca B, Lombardo F, Valenzuela JG, Francischetti IM, Marinotti O, Coluzzi M, et al. (2005) An updated catalogue of salivary gland transcripts in the adult female mosquito, Anopheles gambiae. J Exp Biol 208: 3971–3986. [DOI] [PubMed] [Google Scholar]
- 41.Calvo E, Pham VM, Lombardo F, Arcà B, Ribeiro JMC (2006) The sialotranscriptome of adult male Anopheles gambiae mosquitoes. Insect Biochem Mol Biol 36:570–575. [DOI] [PubMed] [Google Scholar]
- 42.Neira Oviedo M, Ribeiro JMC, Heyland A, VanEkeris L, Moroz T, Linser PJ (2009) The salivary transcriptome of Anopheles gambiae (Diptera: Culicidae) larvae: A microarray-based analysis. Insect Biochem Mol Biol. 39: 382–394. 10.1016/j.ibmb.2009.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ribeiro JMC, Alarcon-Chaidez F, Francischetti IM, Mans BJ, Mather TN, Valenzuela JG, et al. (2006) An annotated catalog of salivary gland transcripts from Ixodes scapularis ticks. Insect Biochem Mol Biol 36: 111–129. [DOI] [PubMed] [Google Scholar]
- 44.Das S, Radtke A, Choi Y-J, Mendes A, Valenzuela J, Dimopoulos G (2010) Transcriptomic and functional analysis of the Anopheles gambiae salivary gland in relation to blood feeding. BMC Genomics 11: 566 10.1186/1471-2164-11-566 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alarcon-Chaidez FJ, Sun J, Wikel SK (2007) Transcriptome analysis of the salivary glands of Dermacentor andersoni Stiles (Acari: Ixodidae). Insect Biochem Mol Biol 37: 48–71. [DOI] [PubMed] [Google Scholar]
- 46.Alves-Silva J, Ribeiro J, Abbeele J, Attardo G, Hao Z, Haines L, et al. (2010) An insight into the sialome of Glossina morsitans morsitans. BMC Genomics. 11: 213 10.1186/1471-2164-11-213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Matsumoto Y, Suetsugu Y, Nakamura M, Hattori M (2014) Transcriptome analysis of the salivary glands of Nephotettix cincticeps (Uhler). J Insect Physiol 71: 170–176. 10.1016/j.jinsphys.2014.10.010 [DOI] [PubMed] [Google Scholar]
- 48.Stafford-Banks CA, Rotenberg D, Johnson BR, Whitfield AE, Ullman DE (2014) Analysis of the salivary gland transcriptome of Frankliniella occidentalis. PLoS One. 9: e94447 10.1371/journal.pone.0094447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hull JJ, Geib SM, Fabrick JA, Brent CS (2013) Sequencing and De Novo assembly of the western tarnished plant bug (Lygus hesperus) transcriptome. PLoS One. 8: e55105 10.1371/journal.pone.0055105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang L, Xu P, Xiao H, Lu Y, Liang G, Zhang Y, et al. (2015) Molecular characterization and expression profiles of polygalacturonase genes in Apolygus lucorum (Hemiptera: Miridae). PLoS ONE. 10: e0126391 10.1371/journal.pone.0126391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Cooper WR, Nicholson SJ, Puterka GJ (2013) Salivary proteins of Lygus hesperus (Hemiptera: Miridae). Ann Entomol Soc Am 106: 86–92. [Google Scholar]
- 52.Perera OP, Gore J, Snodgrass GL, Jackson RE, Allen KC, Abel CA, et al. Temporal and spatial genetic variability among tarnished plant bug (Hemiptera: Miridae) populations in a small geographic area. Ann Entomol Soc Am 108: 181–192. [Google Scholar]
- 53.Perera OP, Snodgrass GL, Scheffler BE, Gore J, Abel CA (2007) Characterization of eight polymorphic microsatellite markers in the tarnished plant bug, Lygus lineolaris (Palisot de Beauvois). Mol Ecol Notes 7: 987–989. [Google Scholar]
- 54.Allen ML (2007) Expressed sequenced tags from Lygus lineolaris (Hemiptera: Miridae), the tarnished plant bug. Genet Mol Res 6: 206–213. [PubMed] [Google Scholar]
- 55.Magalhaes LC, van Kretschmar JB, Donohue KV, Roe RM (2013) Pyrosequencing of the adult tarnished plant bug, Lygus lineolaris, and characterization of messages important in metabolism and development. Entomol Exp et Applic 146:364–378. [Google Scholar]
- 56.Zhu YC, Guo Z, He Y, Luttrell R (2012) Microarray analysis of gene regulations and potential association with acephate-resistance and fitness cost in Lygus lineolaris. PLoS One 7: e37586 10.1371/journal.pone.0037586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Zhu YC, Luttrell R (2015) Altered gene regulation and potential association with metabolic resistance development to imidacloprid in the tarnished plant bug, Lygus lineolaris. Pest Manage Sci. 71: 40–57. [DOI] [PubMed] [Google Scholar]
- 58.Winnebeck EC, Millar CD, Warman GR (2010) Why does insect RNA look degraded? J Insect Sci 10:159 10.1673/031.010.14119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinfor 30: 2114–2120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I, et al. (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotech 29: 644–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Haas BJ, Papanicolaou A, Yassour M, Grabherr M, Blood PD, Bowden J, et al. (2013) De novo transcript sequence reconstruction from RNA-seq using the Trinity platform for reference generation and analysis. Nat Protoc 8: 1494–1512. 10.1038/nprot.2013.084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Yang Y, Smith S (2013) Optimizing de novo assembly of short-read RNA-seq data for phylogenomics. BMC Genomics. 14:328 10.1186/1471-2164-14-328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Consortium TU (2015) UniProt: a hub for protein information. Nucleic Acids Res 43: D204–D212. 10.1093/nar/gku989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tribolium Genome Sequencing Consortium, Richards S, Gibbs RA, Weinstock GM, Brown SJ, Denell R, et al. (2008) The genome of the model beetle and pest Tribolium castaneum. Nature 452: 949–955. 10.1038/nature06784 [DOI] [PubMed] [Google Scholar]
- 65.Adams MD, Celniker SE, Holt RA, Evans CA, Gocayne JD, Amanatides PG, et al. (2000) The Genome sequence of Drosophila melanogaster. Science 287: 2185–2195. [DOI] [PubMed] [Google Scholar]
- 66.The International Aphid Genomics C (2010) Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol. 8: e1000313 10.1371/journal.pbio.1000313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Su Y-L, Li J-M, Li M, Luan J-B, Ye X-D, Wang X-W, et al. (2012) Transcriptomic analysis of the salivary glands of an invasive whitefly. PLoS One. 7: e39303 10.1371/journal.pone.0039303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ji R, Yu H, Fu Q, Chen H, Ye W, Li S, et al. (2013) Comparative transcriptome analysis of salivary glands of two populations of rice brown planthopper, Nilaparvata lugens, that differ in virulence. PLoS One 8: e79612 10.1371/journal.pone.0079612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, et al. (2008) KEGG for linking genomes to life and the environment. Nucleic Acids Res 36: D480–D484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kanehisa M, Goto S (2000) KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res 28: 27–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Maffei ME, Mithöfer A, Arimura G-I, Uchtenhagen H, Bossi S, Bertea CM, et al. (2006) Effects of feeding Spodoptera littoralis on lima bean leaves. III. Membrane depolarization and involvement of hydrogen peroxide. Plant Physiol 140: 1022–1035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Maffei ME, Mithöfer A, Boland W (2007) Before gene expression: early events in plant–insect interaction. Trends Plant Sci 12: 310–316. [DOI] [PubMed] [Google Scholar]
- 73.Torres MA (2010) ROS in biotic interactions. Physiol Plantarum 138: 414–429. [DOI] [PubMed] [Google Scholar]
- 74.Perera OP, Snodgrass GL, Allen KC, Jackson RE, Becnel JJ, O’Leary PF, et al. (2012) The complete genome sequence of a single-stranded RNA virus from the tarnished plant bug, Lygus lineolaris (Palisot de Beauvois). J Invertebr Pathol 109: 11–19. 10.1016/j.jip.2011.08.004 [DOI] [PubMed] [Google Scholar]
- 75.Li D, Roberts R (2001) WD-repeat proteins: structure characteristics, biological function, and their involvement in human diseases. Cell Molecular Life Sci 58: 2085–2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Shelby K (2013) Functional immunomics of the squash bug, Anasa tristis (De Geer) (Heteroptera: Coreidae). Insects. 4: 712 10.3390/insects4040712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.O'Neill MA (2003) In: Rose JKC, editor. The Plant Cell Wall: Blackwell Publishing; pp. 1–54. [Google Scholar]
- 78.Willats WG, McCartney L, Mackie W, Knox JP (2001) Pectin: cell biology and prospects for functional analysis. Plant Mol Biol 47: 9–27. [PubMed] [Google Scholar]
- 79.Kirsch R, Gramzow L, Theißen G, Siegfried BD, ffrench-Constant RH, Heckel DG, et al. (2014) Horizontal gene transfer and functional diversification of plant cell wall degrading polygalacturonases: Key events in the evolution of herbivory in beetles. Insect Biochem Mol Biol 52: 33–50. 10.1016/j.ibmb.2014.06.008 [DOI] [PubMed] [Google Scholar]
- 80.Pauchet Y, Kirsch R, Giraud S, Vogel H, Heckel DG (2014) Identification and characterization of plant cell wall degrading enzymes from three glycoside hydrolase families in the cerambycid beetle Apriona japonica. Insect Biochem Mol Biol 49:1–13. 10.1016/j.ibmb.2014.03.004 [DOI] [PubMed] [Google Scholar]
- 81.Pauchet Y, Wilkinson P, Chauhan R, ffrench-Constant RH (2010) Diversity of beetle genes encoding novel plant cell wall degrading enzymes. PLoS One 5: e15635 10.1371/journal.pone.0015635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The Carbohydrate-Active EnZymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37: D233–D238. 10.1093/nar/gkn663 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Markovič O, Janeček Š (2001) Pectin degrading glycoside hydrolases of family 28: sequence-structural features, specificities and evolution. Protein Engineer 14: 615–631. [DOI] [PubMed] [Google Scholar]
- 84.Henrissat B (1991) A classification of glycosyl hydrolases based on amino acid sequence similarities. Biochem J 280: 309–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Choi JK, Lee BH, Chae CH, Shin W (2004) Computer modeling of the rhamnogalacturonase–“hairy” pectin complex. Proteins: Structure, Function, and Bioinformatics 55:22–33. [DOI] [PubMed] [Google Scholar]
- 86.Kirsch R, Wielsch N, Vogel H, Svatos A, Heckel DG, Pauchet Y (2012) Combining proteomics and transcriptome sequencing to identify active plant-cell-wall-degrading enzymes in a leaf beetle. BMC Genomics. 13:587 10.1186/1471-2164-13-587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Stiburek L, Cesnekova J, Kostkova O, Fornuskova D, Vinsova K, Wenchich L, et al. (2012) YME1L controls the accumulation of respiratory chain subunits and is required for apoptotic resistance, cristae morphogenesis, and cell proliferation. Mol Biol Cell. 23: 1010–1023. 10.1091/mbc.E11-08-0674 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Musser RO, Hum-Musser SM, Eichenseer H, Peiffer M, Ervin G, Murphy JB, et al. (2002) Herbivory: Caterpillar saliva beats plant defences. Nature 416: 599–600. [DOI] [PubMed] [Google Scholar]
- 89.Musser RO, Cipollini DF, Hum-Musser SM, Williams SA, Brown JK, Felton GW (2005) Evidence that the caterpillar salivary enzyme glucose oxidase provides herbivore offense in solanaceous plants. Arch Insect Biochem Physiol 58: 128–137. [DOI] [PubMed] [Google Scholar]
- 90.Tian D, Peiffer M, Shoemaker E, Tooker J, Haubruge E, Francis F, et al. (2012) Salivary glucose oxidase from caterpillars mediates the induction of rapid and delayed-induced defenses in the tomato plant. PLoS One 7: e36168 10.1371/journal.pone.0036168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.de la Paz Celorio-Mancera M, Courtiade J, Muck A, Heckel DG, Musser RO, Vogel H (2011) Sialome of a generalist lepidopteran herbivore: Identification of transcripts and proteins from Helicoverpa armigera labial salivary glands. PLoS One 6: e26676 10.1371/journal.pone.0026676 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Harmel N, Létocart E, Cherqui A, Giordanengo P, Mazzucchelli G, Guillonneau F, et al. (2008) Identification of aphid salivary proteins: a proteomic investigation of Myzus persicae. Insect Mol Biol 17:165–174. 10.1111/j.1365-2583.2008.00790.x [DOI] [PubMed] [Google Scholar]
- 93.Singh H, Dixit S, Verma PC, Singh PK (2013) Differential peroxidase activities in three different crops upon insect feeding. Plant Signal Behav 8: e25615 10.4161/psb.25615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Dubey N, Goel R, Ranjan A, Idris A, Singh S, Bag S, et al. (2013) Comparative transcriptome analysis of Gossypium hirsutum L. in response to sap sucking insects: aphid and whitefly. BMC Genomics. 14: 1–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Doyle WA, Burke JF, Chovnick A, Dutton FL, Whittle JRS, Bray RC (1996) Properties of xanthine dehydrogenase variants from rosy mutant strains of Drosophila melanogaster and their relevance to the enzyme's structure and mechanism. European J Biochem 239: 782–795. [DOI] [PubMed] [Google Scholar]
- 96.Gray M, Charpentier A, Walsh K, Wu P, Bender W (1991) Mapping point mutations in the Drosophila rosy locus using denaturing gradient gel blots. Genetics 127: 139–149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hayashi Y (1961) Properties of xanthine dehydrogenase in the silkworm, Bombyx mori L. Nature 192: 756–757. [DOI] [PubMed] [Google Scholar]
- 98.Keith TP, Riley MA, Kreitman M, Lewontin RC, Curtis D, Chambers G (1987) Sequence of the structural gene for xanthine dehydrogenase (rosy Locus) in Drosophila melanogaster. Genetics 116: 67–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kômoto N, Yukuhiro K, Tamura T (1999) Structure and expression of tandemly duplicated xanthine dehydrogenase genes of the silkworm (Bombyx mori). Insect Mol Biol 8: 73–83. [DOI] [PubMed] [Google Scholar]
- 100.Vorbach C, Harrison R, Capecchi MR (2003) Xanthine oxidoreductase is central to the evolution and function of the innate immune system. Trends Immunol 24: 512–517. [DOI] [PubMed] [Google Scholar]
- 101.Carolan JC, Caragea D, Reardon KT, Mutti NS, Dittmer N, Pappan K, et al. (2011) Predicted effector molecules in the salivary secretome of the pea aphid (Acyrthosiphon pisum): A dual transcriptomic/proteomic approach. J Proteome Res 10: 1505–1518. 10.1021/pr100881q [DOI] [PubMed] [Google Scholar]
- 102.Carolan JC, Fitzroy CIJ, Ashton PD, Douglas AE, Wilkinson TL (2009) The secreted salivary proteome of the pea aphid Acyrthosiphon pisum characterised by mass spectrometry. Proteomics 9: 2457–2467. 10.1002/pmic.200800692 [DOI] [PubMed] [Google Scholar]
- 103.Will T, Furch AC, Zimmermann MR (2013) How phloem-feeding insects face the challenge of phloem-located defenses. Front Plant Sci. 4: 336 10.3389/fpls.2013.00336 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Smith TM, Kirley TL (2006) The calcium activated nucleotidases: A diverse family of soluble and membrane associated nucleotide hydrolyzing enzymes. Purinergic Signal 2: 327–333. 10.1007/s11302-005-5300-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Chisava S, Murphy AM, Hamilton JM, Lindsey K, Carr JP, Slabas AR (2009) Extracellular ATP is a regulator of pathogen defence in plants. Plant J 60: 436–448. 10.1111/j.1365-313X.2009.03968.x [DOI] [PubMed] [Google Scholar]
- 106.Chisava A, Tomé DFA, Murphy AM, Hamilton JM, Lindsey K, Carr JP, et al. (2009) Extracellular ATP: A modulator of cell death and pathogen defence in plants. Plat Signal Behav 4: 1078–1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lim MH, Wu J, Yao J, Callardo IF, Dugger JW, Webb LJ, et al. (2014) Apyrase supression raises extracellular ATP levels and induces gene expression and cell wall changes characteristic of stress responses. Plant Physiol 164: 2054–2067. 10.1104/pp.113.233429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Allen ML, Walker WB III (2012) Saliva of Lygus lineolaris digests double stranded ribonucleic acids. J Insect Physiol 58: 391–396. 10.1016/j.jinsphys.2011.12.014 [DOI] [PubMed] [Google Scholar]
- 109.Cohen AC (2000) New oligidic production diet for Lygus hesperus Knight and L. lineolaris (Palisot de Beauvois). J Entomol Sci 25: 301–310. [Google Scholar]
- 110.Kodama Y, Shumway M, Leinonen R (2012) The sequence read archive: explosive growth of sequencing data. Nucleic Acids Res 40:D54–D56. 10.1093/nar/gkr854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9: 357–359. 10.1038/nmeth.1923 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Li B, Dewey CN (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinfor 12: 323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Finn RD, Bateman A, Clements J, Coggill P, Eberhardt RY, Eddy SR, et al. (2014) Pfam: the protein families database. Nucleic Acids Res 42: D222–D230. 10.1093/nar/gkt1223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Attwood TK, Coletta A, Muirhead G, Pavlopoulou A, Philippou PB, Popov I, et al. (2012) The PRINTS database: a fine-grained protein sequence annotation and analysis resource—its status in 2012. Database: The Journal of Biological Databases and Curation.: bas019 10.1093/database/bas019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.de Lima Morais DA, Fang H, Rackham OJL, Wilson D, Pethica R, Chothia C, et al. (2011) SUPERFAMILY 1.75 including a domain-centric gene ontology method. Nucleic Acids Res 39: D427–D434. 10.1093/nar/gkq1130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Haft DH, Selengut JD, Richter RA, Harkins D, Basu MK, Beck E (2013) TIGRFAMs and Genome Properties in 2013. Nucleic Acids Res 41: D387–D395. 10.1093/nar/gks1234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Haft DH, Selengut JD, White O (2003) The TIGRFAMs database of protein families. Nucleic Acids Res 31:371–373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Letunic I, Doerks T, Bork P (2014) SMART: recent updates, new developments and status in 2015. Nucleic Acids Res 43:D257–D260. 10.1093/nar/gku949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Oates ME, Stahlhacke J, Vavoulis DV, Smithers B, Rackham OJL, Sardar AJ, et al. (2015) The SUPERFAMILY 1.75 database in 2014: a doubling of data. Nucleic Acids Res 43: D227–D233. 10.1093/nar/gku1041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Quevillon E, Silventoinen V, Pillai S, Harte N, Mulder N, Apweiler R, et al. (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33: W116–W120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Sigrist CJ, Cerutti L, Hulo N, Gattiker A, Falquet L, Pagni M, et al. (2002) PROSITE: a documented database using patterns and profiles as motif descriptors. Brief Bioinform 3: 265–274. [DOI] [PubMed] [Google Scholar]
- 122.Sigrist CJA, de Castro E, Cerutti L, Cuche BA, Hulo N, Bridge A, et al. (2013) New and continuing developments at PROSITE. Nucleic Acids Res 41: D344–D347. 10.1093/nar/gks1067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Sillitoe I, Lewis TE, Cuff A, Das S, Ashford P, Dawson NL, et al. (2015) CATH: comprehensive structural and functional annotations for genome sequences. Nucleic Acids Res 43: D376–D381. 10.1093/nar/gku947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wu CH, Nikolskaya A, Huang H, Yeh L-SL, Natale DA, Vinayaka CR, et al. (2004) PIRSF: family classification system at the Protein Information Resource. Nucleic Acids Res 32: D112–D114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Mi H, Lazareva-Ulitsky B, Loo R, Kejariwal A, Vandergriff J, Rabkin S, et al. (2005) The PANTHER database of protein families, subfamilies, functions and pathways. Nucleic Acids Res 33: D284–D288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Thomas PD, Campbell MJ, Kejariwal A, Mi H, Karlak B, Daverman R, et al. (2003) PANTHER: A library of protein families and subfamilies indexed by function. Genome Res 13: 2129–2141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Consortium TGO (2015) Gene Ontology Consortium: going forward. Nucleic Acids Res 43: D1049–D1056. 10.1093/nar/gku1179 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Krogh A, Larsson B, von Heijne G, Sonnhammer EL (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305: 567–580. [DOI] [PubMed] [Google Scholar]
- 129.Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. 10.1038/nmeth.1701 [DOI] [PubMed] [Google Scholar]
- 130.Falcon S, Gentleman R (2007) Using GOstats to test gene lists for GO term association. Bioinformatics. 23: 257–258. [DOI] [PubMed] [Google Scholar]
- 131.Parra G, Bradnam K, Korf I (2007) CEGMA: a pipeline to accurately annotate core genes in eukaryotic genomes. Bioinformatics. 23: 1061–1067. [DOI] [PubMed] [Google Scholar]
- 132.Marchler-Bauer A, Derbyshire MK, Gonzales NR, Lu S, Chitsaz F, Geer LY, et al. (2015) CDD: NCBI's conserved domain database. Nucleic Acids Res 43: D222–D226. 10.1093/nar/gku1221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Felsenstein J (1985) Confidence limits on phylogenies: An approach using the bootstrap. Evol 39: 783–791. [DOI] [PubMed] [Google Scholar]
- 134.Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:c406–425. [DOI] [PubMed] [Google Scholar]
- 135.Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol Biol Evol 30: 2725–2729. 10.1093/molbev/mst197 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(TIF)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
(XLSX)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.