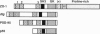Abstract
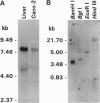
Tight junctions form an intercellular barrier between epithelial cells, serve to separate tissue compartments, and maintain cellular polarity. Paracellular sealing properties vary among cell types and are regulated by undefined mechanisms. Sequence of the full-length cDNA for human ZO-1, the first identified tight junction component, predicts a protein of 1736 aa. The N-terminal 793 aa are homologous to the product of the lethal(1)discs-large-1 (dlg) tumor suppressor gene of Drosophila, located in septate junctions, and to a 95-kDa protein located in the postsynaptic densities of rat brain, PSD-95. All three proteins contain both a src homology region 3 (SH3 domain), previously identified in membrane proteins involved in signal transduction, and a region homologous to guanylate kinase. ZO-1 contains an additional 943-aa C-terminal domain that is proline-rich (14.1%) and contains an alternatively spliced domain, whose expression was previously shown to correlate with variable properties of tight junctions. dlg mutations result in loss of apical-basolateral epithelial cell polarity and in neoplastic growth. These results suggest a protein family specialized for signal transduction on the cytoplasmic surface of intercellular junctions. These results also provide biochemical evidence for similarity between invertebrate septate and vertebrate tight junctions. The C-terminal domain of ZO-1, and its alternatively spliced region, appears to confer variable properties unique to tight junctions.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson J. M., Van Itallie C. M., Peterson M. D., Stevenson B. R., Carew E. A., Mooseker M. S. ZO-1 mRNA and protein expression during tight junction assembly in Caco-2 cells. J Cell Biol. 1989 Sep;109(3):1047–1056. doi: 10.1083/jcb.109.3.1047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balda M. S., Anderson J. M. Two classes of tight junctions are revealed by ZO-1 isoforms. Am J Physiol. 1993 Apr;264(4 Pt 1):C918–C924. doi: 10.1152/ajpcell.1993.264.4.C918. [DOI] [PubMed] [Google Scholar]
- Balda M. S., González-Mariscal L., Contreras R. G., Macias-Silva M., Torres-Marquez M. E., García-Sáinz J. A., Cereijido M. Assembly and sealing of tight junctions: possible participation of G-proteins, phospholipase C, protein kinase C and calmodulin. J Membr Biol. 1991 Jun;122(3):193–202. doi: 10.1007/BF01871420. [DOI] [PubMed] [Google Scholar]
- Berger A., Schiltz E., Schulz G. E. Guanylate kinase from Saccharomyces cerevisiae. Isolation and characterization, crystallization and preliminary X-ray analysis, amino acid sequence and comparison with adenylate kinases. Eur J Biochem. 1989 Sep 15;184(2):433–443. doi: 10.1111/j.1432-1033.1989.tb15035.x. [DOI] [PubMed] [Google Scholar]
- Bryant P. J., Woods D. F. A major palmitoylated membrane protein of human erythrocytes shows homology to yeast guanylate kinase and to the product of a Drosophila tumor suppressor gene. Cell. 1992 Feb 21;68(4):621–622. doi: 10.1016/0092-8674(92)90136-z. [DOI] [PubMed] [Google Scholar]
- Cho K. O., Hunt C. A., Kennedy M. B. The rat brain postsynaptic density fraction contains a homolog of the Drosophila discs-large tumor suppressor protein. Neuron. 1992 Nov;9(5):929–942. doi: 10.1016/0896-6273(92)90245-9. [DOI] [PubMed] [Google Scholar]
- Cicchetti P., Mayer B. J., Thiel G., Baltimore D. Identification of a protein that binds to the SH3 region of Abl and is similar to Bcr and GAP-rho. Science. 1992 Aug 7;257(5071):803–806. doi: 10.1126/science.1379745. [DOI] [PubMed] [Google Scholar]
- Citi S. Protein kinase inhibitors prevent junction dissociation induced by low extracellular calcium in MDCK epithelial cells. J Cell Biol. 1992 Apr;117(1):169–178. doi: 10.1083/jcb.117.1.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Citi S., Sabanay H., Jakes R., Geiger B., Kendrick-Jones J. Cingulin, a new peripheral component of tight junctions. Nature. 1988 May 19;333(6170):272–276. doi: 10.1038/333272a0. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FARQUHAR M. G., PALADE G. E. Junctional complexes in various epithelia. J Cell Biol. 1963 May;17:375–412. doi: 10.1083/jcb.17.2.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gumbiner B., Lowenkopf T., Apatira D. Identification of a 160-kDa polypeptide that binds to the tight junction protein ZO-1. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3460–3464. doi: 10.1073/pnas.88.8.3460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howarth A. G., Hughes M. R., Stevenson B. R. Detection of the tight junction-associated protein ZO-1 in astrocytes and other nonepithelial cell types. Am J Physiol. 1992 Feb;262(2 Pt 1):C461–C469. doi: 10.1152/ajpcell.1992.262.2.C461. [DOI] [PubMed] [Google Scholar]
- Itoh M., Nagafuchi A., Yonemura S., Kitani-Yasuda T., Tsukita S., Tsukita S. The 220-kD protein colocalizing with cadherins in non-epithelial cells is identical to ZO-1, a tight junction-associated protein in epithelial cells: cDNA cloning and immunoelectron microscopy. J Cell Biol. 1993 May;121(3):491–502. doi: 10.1083/jcb.121.3.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. An analysis of vertebrate mRNA sequences: intimations of translational control. J Cell Biol. 1991 Nov;115(4):887–903. doi: 10.1083/jcb.115.4.887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. The scanning model for translation: an update. J Cell Biol. 1989 Feb;108(2):229–241. doi: 10.1083/jcb.108.2.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurihara H., Anderson J. M., Farquhar M. G. Diversity among tight junctions in rat kidney: glomerular slit diaphragms and endothelial junctions express only one isoform of the tight junction protein ZO-1. Proc Natl Acad Sci U S A. 1992 Aug 1;89(15):7075–7079. doi: 10.1073/pnas.89.15.7075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madara J. L., Parkos C., Colgan S., Nusrat A., Atisook K., Kaoutzani P. The movement of solutes and cells across tight junctions. Ann N Y Acad Sci. 1992;664:47–60. doi: 10.1111/j.1749-6632.1992.tb39748.x. [DOI] [PubMed] [Google Scholar]
- Mayer B. J., Baltimore D. Signalling through SH2 and SH3 domains. Trends Cell Biol. 1993 Jan;3(1):8–13. doi: 10.1016/0962-8924(93)90194-6. [DOI] [PubMed] [Google Scholar]
- Musacchio A., Gibson T., Lehto V. P., Saraste M. SH3--an abundant protein domain in search of a function. FEBS Lett. 1992 Jul 27;307(1):55–61. doi: 10.1016/0014-5793(92)80901-r. [DOI] [PubMed] [Google Scholar]
- Quinonez G., Simon G. T. Cellular junctions in a spectrum of human malignant tumors. Ultrastruct Pathol. 1988;12(4):389–405. doi: 10.3109/01913128809064209. [DOI] [PubMed] [Google Scholar]
- Ruff P., Speicher D. W., Husain-Chishti A. Molecular identification of a major palmitoylated erythrocyte membrane protein containing the src homology 3 motif. Proc Natl Acad Sci U S A. 1991 Aug 1;88(15):6595–6599. doi: 10.1073/pnas.88.15.6595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sadler I., Crawford A. W., Michelsen J. W., Beckerle M. C. Zyxin and cCRP: two interactive LIM domain proteins associated with the cytoskeleton. J Cell Biol. 1992 Dec;119(6):1573–1587. doi: 10.1083/jcb.119.6.1573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schnabel E., Anderson J. M., Farquhar M. G. The tight junction protein ZO-1 is concentrated along slit diaphragms of the glomerular epithelium. J Cell Biol. 1990 Sep;111(3):1255–1263. doi: 10.1083/jcb.111.3.1255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneeberger E. E., Lynch R. D. Structure, function, and regulation of cellular tight junctions. Am J Physiol. 1992 Jun;262(6 Pt 1):L647–L661. doi: 10.1152/ajplung.1992.262.6.L647. [DOI] [PubMed] [Google Scholar]
- Stehle T., Schulz G. E. Three-dimensional structure of the complex of guanylate kinase from yeast with its substrate GMP. J Mol Biol. 1990 Jan 5;211(1):249–254. doi: 10.1016/0022-2836(90)90024-G. [DOI] [PubMed] [Google Scholar]
- Stevenson B. R., Siliciano J. D., Mooseker M. S., Goodenough D. A. Identification of ZO-1: a high molecular weight polypeptide associated with the tight junction (zonula occludens) in a variety of epithelia. J Cell Biol. 1986 Sep;103(3):755–766. doi: 10.1083/jcb.103.3.755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willott E., Balda M. S., Heintzelman M., Jameson B., Anderson J. M. Localization and differential expression of two isoforms of the tight junction protein ZO-1. Am J Physiol. 1992 May;262(5 Pt 1):C1119–C1124. doi: 10.1152/ajpcell.1992.262.5.C1119. [DOI] [PubMed] [Google Scholar]
- Woods D. F., Bryant P. J. Molecular cloning of the lethal(1)discs large-1 oncogene of Drosophila. Dev Biol. 1989 Jul;134(1):222–235. doi: 10.1016/0012-1606(89)90092-4. [DOI] [PubMed] [Google Scholar]
- Woods D. F., Bryant P. J. The discs-large tumor suppressor gene of Drosophila encodes a guanylate kinase homolog localized at septate junctions. Cell. 1991 Aug 9;66(3):451–464. doi: 10.1016/0092-8674(81)90009-x. [DOI] [PubMed] [Google Scholar]
- Zhong Y., Saitoh T., Minase T., Sawada N., Enomoto K., Mori M. Monoclonal antibody 7H6 reacts with a novel tight junction-associated protein distinct from ZO-1, cingulin and ZO-2. J Cell Biol. 1993 Jan;120(2):477–483. doi: 10.1083/jcb.120.2.477. [DOI] [PMC free article] [PubMed] [Google Scholar]