Abstract
This study is to investigate the effect and underlying mechanism of Zinc (Zn) on hepatic stellate cell collagen synthesis. The proliferation and collagen synthesis ability of LX-2 cells were detected after adding Zn. The collagen synthesis related proteins of MMP-13 and TIMP1 along with TGF-β signaling pathway related proteins were detected by Western blot. The role of TGF-β signaling pathway in collagen synthesis inhibition was identified by TGF-β RI siRNA silencing. Compared with control group, LX-2 cell proliferation ability was significantly inhibited at all Zn concentrations (50 μM, 100 μM and 200 μM). Zn at 50 μM did not affect the protein content of αSMA and type I collagen while 100 μM and 200 μM Zn could significantly inhibit αSMA expression. Compared with control group, gene expression and protein content of MMP-13 in 200 μM Zn group was significantly increased while no difference in gene expression and protein content of TIMP1 was found. TGF-β RI content in 200 μM Zn group was significantly decreased and the protein content of TGF-β RII was not affected. MMP-13 expression was significantly increased after TGF-β RI siRNA silencing. Further results showed that in LX-2 cells those TGF-β RI expression was inhibited, LX-2 cell proliferation ability and the expression of synthesis collagen related proteins of αSMA and type I collagen were greatly decreased. Zn could significantly inhibit the expression of αSMA and type I collagen by inhibiting TGF-β RI expression and promoting MMP-13 expression.
Keywords: Collagen synthesis, Zn, hepatic fibrosis, hepatic stellate cells
Introduction
Hepatic fibrosis is a common result of a variety of chronic liver injuries. During the pathological process, liver cell necrosis occurs after stimulated by inflammation factors, excessive extracellular matrix proteins deposition appears and abnormal hyperplasia occurs in fibrous connective tissue in the liver, eventually leading to the formation of liver cirrhosis [1,2].
Hepatic stellate cells (HSCs) are the main source of excessive collagen during liver fibrosis [3,4] and once they are activated, collagen expression ability of HSCs is significantly enhanced [5,6]. Type I collagen is the main fibrin in extracellular matrix, its synthesis increases and its degradation reduces in the process of liver fibrosis [7-9]. The degradation of extracellular matrix depends on the activity of matrix metalloproteinase (MMPs) [10]. According to the substrate specificity, matrix MMPs can be divided into five categories, namely interstitial collagenase (MMP-1, -8, -13), gelatinase (MMP-2, -9), matrix degrading enzyme (MMP-3, -7, -10, -11), membrane type matrix metalloproteinase (MMP-14, -15, -16, -17, -24, -25) and metal elastase (MMP-12) [10]. These enzymes are regulated by tissue inhibitor of matrix metalloproteinases (TIMPs) [11-13]. In the progress of liver fibrosis, the imbalance between the activities of MMP s and TIMPs is a key reason that leads to collagen deposition [13,14].
Transforming growth factor β (TGF-β) is a profibrotic cytokine [15], which, can stimulate cell synthesis and deposition of extracellular matrix components such as type I, III, IV collagen, elastin, tenascin, osteonectin, biglycan proteoglycan and core protein polysaccharide [16]. HSCs are the main origin of TGF-β, meanwhile, macrophages, liver cells and platelets also can secrete TGF-β [17]. TGF-β mediated intracellular signal delivery is transmitted by Smad protein [18]. After binding to activated type II receptor, TGF-β phosphorylates type I receptor. The signal is transmitted to Smad2 and Smad3 proteins from type I receptor and phosphorylation takes place in Smad2 and Smad3 proteins [19]. Next, Smad2, Smad3 and Smad4 proteins form an oligomeric complex and transfer to nucleus, then, recognize and regulate transcription on specific sites of target DNA. At the same time, along with the activation of TGF-β signaling pathway, HSCs proliferation ability can be significantly increased [20]. Smad7 protein is an inhibitor of Smad signaling pathway. It can interfere the interaction of type I TGF-β receptor and Smad2 and Smad3 through interaction with type I TGF-β receptor thus effectively inhibits Smad2 and Smad3 phosphorylation and a series of subsequent downstream signals [21].
Zinc (Zn) is an essential trace element for human body with a variety of biological functions such as antioxidant, anti-inflammatory and anti apoptosis [22]. Zn deficiency is generally found in cirrhotic patients and patients with chronic liver disease [23,24]. Zn is the inhibitor of collagen synthase (proline hydroxylase) [25] because it can compete with the iron ion binding site of enzymatic activity center, thus inhibit the enzyme activity, and as a result, inhibit collagen synthesis. In addition, Zn is a co activator of MMPs [26] and is an essential metal ion for MMPs activity maintenance. Zn deficiency can induce decreased MMPs activity, inhibition of collagen degradation and aggregation of collagen in liver tissue, hence, leading to the formation of fibrotic lesions.
In this study, human hepatic stellate cell line LX-2 cells were used as the research model to investigate the molecular mechanism collagen synthesis inhibition mediated by Zn.
Materials and methods
Cell treatment
The cultured LX-2 cells were divided into 4 groups, namely control group, 50 μM group, 100 μM group and 200 μM group. The control group was LX-2 cells cultured with normal medium while corresponding final concentrations of Zn was added to the 50 μM, 100 μM and 200 μM groups. The cells were cultured with H-DMED medium in a cell culture incubator with 5% CO2 and saturated humidity at 37°C.
Atomic absorption spectrometry
After cultured for 24 hours, the LX-2 cells were digestion with 1% TNE and collected by centrifugation. The cells was resuspended with vortex oscillation after adding 100 μl 1% SDS and was lysed with ultrasonic. Cellular debris was removed by 12000 g centrifugation at 4°C for 10 min and clear lysate was reserved. Protein concentration was detected by commercial DAC protein assay Kit (Bio-rad, Hercules, USA). The LX-2 cell protein extraction was digested by adding 5 times volume of concentrated nitric acid. Sample contained Zn contents were detected by using the atomic absorption spectrometry method and the results were standardized with total protein content of LX-2 cells.
Methyl thiazol tetrazolium (MTT) assay
LX-2 cells proliferation was detected after culturing for 1 day, 2 days, 3 days, 4 days, 5 days, 6 days and 7 days. And, 5 wells in each group were measured. Each well was added with 200 μL MCDB131 after the medium was absorbed and another 20 μL MTT was added subsequently. After incubated for 3.5 hours in an incubator, the culture medium was absorbed and 100 μL DMSO was added into each well. Another incubation of 5-10 minutes was carried out and the solution was transferred to another clean 96 wells petri dish. The OD value was measured at wavelength of 490 nm with continuous spectrum microplate reading instrument (Bio-rad, Hercules, USA).
Western blotting
After subject to sodium dodecyl sulfate-polyacrylamide (SDS) gel electrophoresis, the proteins were transferred to polyvinylidene difluoride membranes and blocked with 50 g/L skimmed milk at room temperature for 1 h. The membranes were incubated respectively with rabbit primary antibodies of TGF-β RI, TGF-β RII, MMP13, TIMP-1, αSMA, collagen I (1:5000) and GAPDH (1:2000) (Santa Cruz Biotechnology, Dallas, USA) at 4°C overnight. After washed with phosphate-buffered saline with Tween 20 (PBS-T) for 3 times of 15 min, the membranes were incubated with goat anti-rabbit-horseradish peroxidase conjugate antibody (1:1000; Santa Cruz Biotechnology, Dallas, USA) for 1 h at room temperature before washing with PBS-T for 3 times of 15 min. Then, the membranes were developed with enhanced chemiluminescence detection kit (Sigma-Aldrich, St. Louis, USA) for imaging. Image lab (Bio-Rad, Hercules, USA) software was used to acquire and analyze imaging signals. The relative content of protein was expressed as protein/GAPDH ratio.
siRNA silencing
All three targeting siRNAs of TGF-β RI were purchased from RiboBio (RiboBio Co., Ltd., Guangzhou, China). The siRNA sequences used were as follows: 5’-GAGGGCAUGUAGACGGUUA dTdT-3’; 3’-dTdT CUCCCGUACAUCUGCCAAU-5’, (2) 5’-ACAAUAUGCUACCUCCAAA dTdT-3’; 3’-dTdT UGUUAUACGAUGGAGGUUU-5’ and (3) 5’-GACUGGAUCCAUACAAUAU dTdT-3’; 3’-dTdT CUGACCUAGGUAUGUUAUA-5’. TGF-β RI was silenced with 30 nM siRNAs and mismatched siRNA was used as control. Lipofectamine 2000 (Invitrogen, New York, USA) was used as transfection reagent. After cultured for 48 hours, the transfected cells were collected using trypsin and analyzed by RT-PCR.
RT-PCR analysis
Total RNA was isolated by Trizol method and was quantified by XXX spectrophotometer. Samples those with OD260/OD280 ratio between 1.8-2.00 were taken as good quality RNA. The quality of RNA was further analyzed by Experion Bioanalyzer (BioRad, Hercules, USA) and first-strand cDNA was prepared by using PrimeScript® RT reagent Kit (Takara, Japan) following manufacturer’s instructions. The samples were electrophoresed on 1.5% agarose gel and the bands were analyzed by using band intensity software. Relative expression amounts of miRNA were calculated by 2-ΔΔT ± SEM.
Statistical analysis
All the statistical analyses were performed using SPSS version 13.0 (SPSS Inc, Chicago, IL, USA) for Windows. The test was used to compare between different groups and P value less than 0.05 was considered as statistically significant.
Results
Zn inhibits LX-2 cell proliferation and collagen synthesis
To detect the inhibitory effect of Zn in cell proliferation and collagen synthesis, corresponding analysis were carried out using LX-2 cell model after cultured with Zn for 24 hours. Zn concentration was detected by atomic absorption spectrometry. LX-2 cell proliferation ability was measured using the MTT method. The expressions of α SMA and type I collagen were analyzed by Western blot. As shown in (Figure 1A), as extracellular Zn concentration increased, intracellular Zn concentration significantly increased. Compared with the control group, LX-2 cell proliferation ability was significantly inhibited at all Zn concentrations of 50 μM, 100 μM and 200 μM (Figure 1B). Western blot results showed that Zn with a final concentration of 50 μM did not affect the protein content of αSMA and type I collagen in LX-2 cells while 100 μM and 200 μM Zn could significantly inhibit αSMA expression (P < 0.05). Compared with the control group, 100 μM Zn could inhibit type I collagen expression but the difference was not significant while 200 μM Zn had a significant effect on the expression type I collagen (P < 0.05) (Figure 1C, 1D). As inhibition of 200 μM Zn was apparent, the concentration of 200 μM Zn was adopted in the subsequent experiments. Taken together, the results showed that high concentration of Zn significantly inhibited LX-2 cell proliferation ability and collagen synthesis ability and the effect of a final concentration of 200 μM Zn was most obvious.
Figure 1.
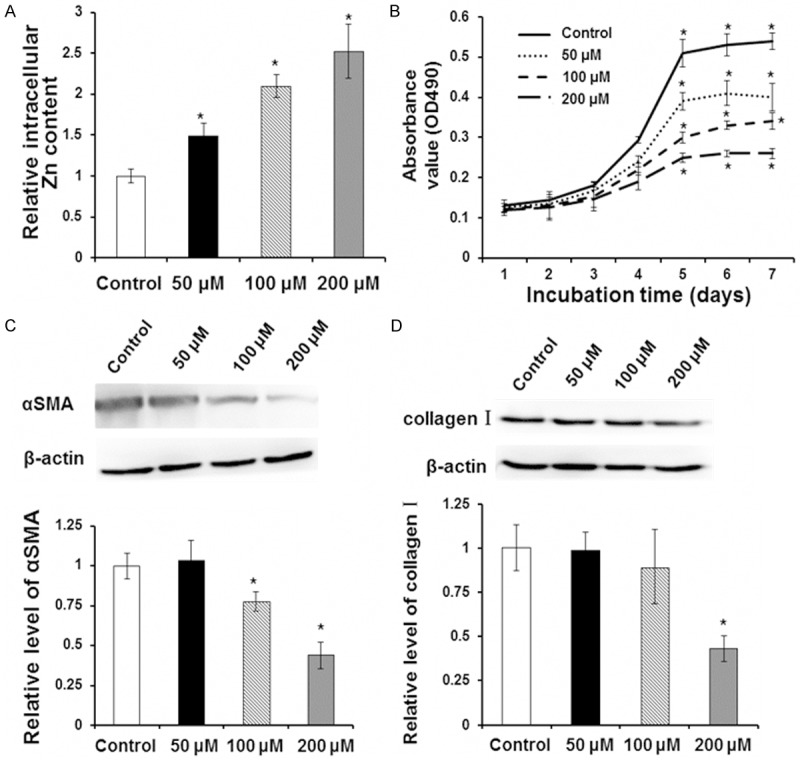
Effect of Zn on LX-2 cell proliferation and collagen synthesis. LX-2 cells were incubated with 0 μM (control group), 50 μM, 100 μM and 200 μM Zn for indicated times. A. After incubation for 24 h, intracellular Zn content was detected with atomic absorption spectrometry. B. After incubation for 1 day, 2 days, 3 days, 4 days, 5 days, 6 days and 7 days, cell proliferation was measured by MTT assay. C. After incubation for 48 h, level of αSMA was detected with Western blot analysis. Representative Western blot results were shown in the upper panel and quantitative Western blot results were shown in the lower panel. D. After incubation for 48 h, level of collagen I was detected with Western blot analysis. Representative Western blot results were shown in the upper panel and quantitative Western blot results were shown in the lower panel. Compared with control group, *P < 0.05.
Zn inhibits LX-2 cell collagen synthesis by increasing MMP-13 expression
To find out how Zn inhibits LX-2 cell collagen synthesis, qRT-PCR and Western blot were adopted to detect levels of MMP-13 and TIMP-1 respectively. LX-2 cells were cultured for 24 h with 0 μM (control group), 50 μM, 100 μM and 200 μM of Zn. Compared with the control group, MMP-13 expression at mRNA level (Figure 2A) and protein level (Figure 2B) at the concentration of 200 μM was significantly increased (P < 0.05). However, there was no significant difference in TIMP-1 expression level (Figure 2C, 2D). To sum up, the results demonstrate that Zn inhibits collagen synthesis ability of LX-2 cells by increasing the expression of collagen degradation associated matrix metalloproteinase MMP-13.
Figure 2.
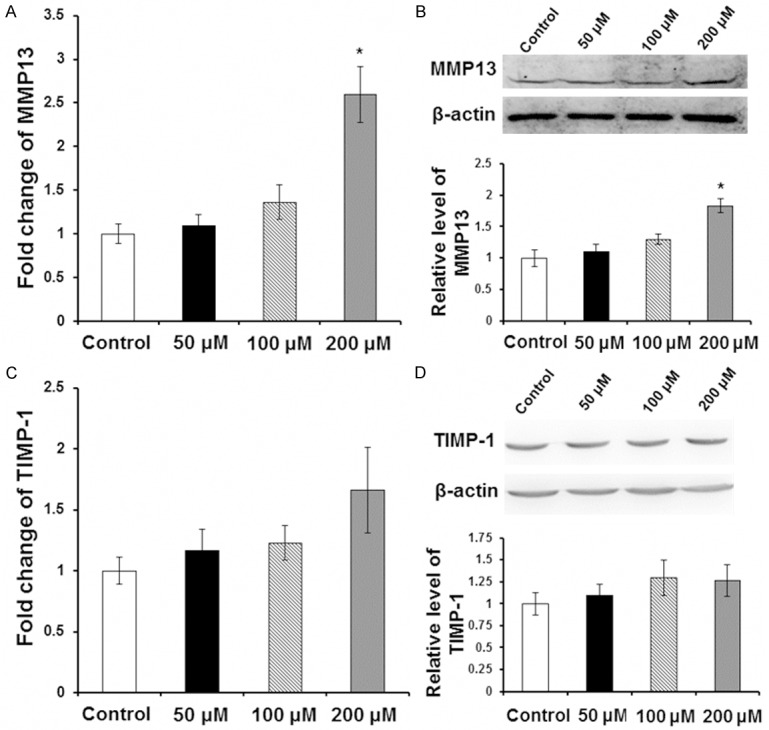
Effect of Zn on expression level of MMP13 and TIMP-1. LX-2 cells were incubated with 0 μM (control group), 50 μM, 100 μM and 200 μM Zn for 24 h. Expression levels of MMP13 and TIMP-1 were detected with qRT-PCR and Western blot analysis. A. Relative expression level of MMP13 by qRT-PCR. B. Representative and quantitative Western blot results of MMP13. C. Relative expression level of TIMP-1 by qRT-PCR. D. Representative and quantitative Western blot results of TIMP-1. Compared with control group, *P < 0.05.
Zn inhibits expression of TGF-β RI
To identify whether Zn can inhibit TGF-β RI and TGF-β RII expression, Western blot was performed. LX-2 cells were incubated with 0 μM (control group) and 200 μM Zn for 24 h. As shown in (Figure 3A), TGF-β RI protein level in 200 μM Zn group was significantly decreased while compared with the control group (P < 0.05). However, Zn with a final concentration of 200 μM did not affect the protein level of TGF-β RII (Figure 3B). In conclusion, the result argues that Zn could inhibit collagen synthesis ability of LX-2 cells by decreasing the content of TGF-β RI.
Figure 3.
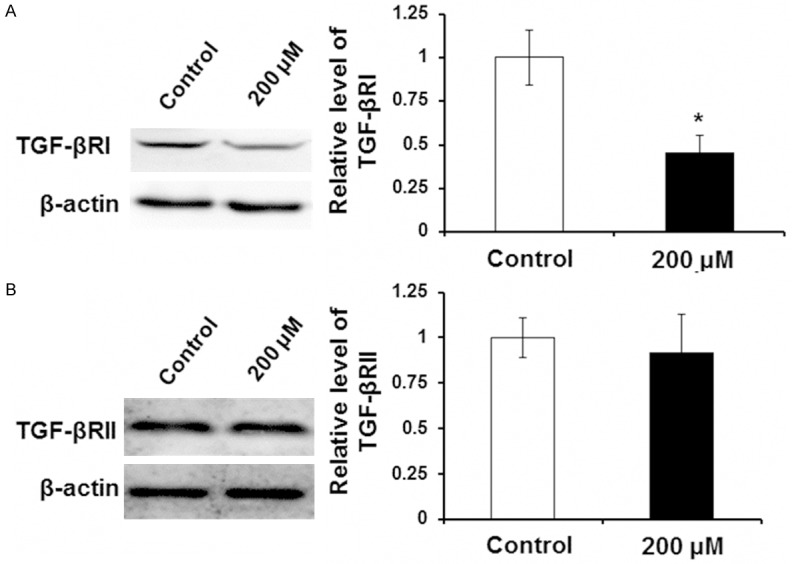
Effect of Zn on expression level of TGF-β RI and TGF-β RII. LX-2 cells were incubated with 0 μM (control group) and 200 μM Zn for 24 h. Expression levels of TGF-β RI and TGF-β RII were detected with Western blot analysis. A. Representative and quantitative Western blot results of TGF-β RI. B. Representative and quantitative Western blot results of TGF-β RII. Compared with control group, *P < 0.05.
TGF-β RI targeted siRNA silencing results
To further identify whether the inhibitory effect of Zn on LX-2 cell proliferation and collagen synthesis was achieved by TGF-β RI inhibition, TGF-β RI targeted siRNA silencing, MTT assay, qRT-PCR and Western blot were carried out. LX-2 cells were divided into two groups. One was the control group, and the other was TGF-β RI siRNA group, of which LX-2 cells were transfected with siRNA targeting silence TGF-β RI. After culturing for 48 hours, proliferation of LX-2 cells was assayed using MTT assay. MMP-13 expression was detected by qRT-PCR and Western blot. Collagen synthesis related proteins of αSMA and type I collagen were analyzed by Western blot. As shown in (Figure 4A), compared with the control group, mismatch siRNA did not affect the expression of TGF-β RI in LX-2 cells while targeting silence siRNA significantly inhibited TGF-β RI expression (P < 0.05). Compared with the control group, once TGF-β RI expression was inhibited, MMP-13 expression was significantly increased (P < 0.05) (Figure 4B, 4C). Further results showed that in LX-2 cells with inhibited TGF-β RI expression, LX-2 cell proliferation ability (Figure 4D) and the expression of synthesis collagen related proteins αSMA (Figure 4E) and type I collagen (Figure 4F) were significantly decreased (P < 0.05). In summary, these results showed that TGF-β RI inhibition had the same effect with 200 μM Zn treatment and further proved that TGF-β RI inhibition played a key role in collagen synthesis ability decrease mediated by Zn in LX-2 cells.
Figure 4.
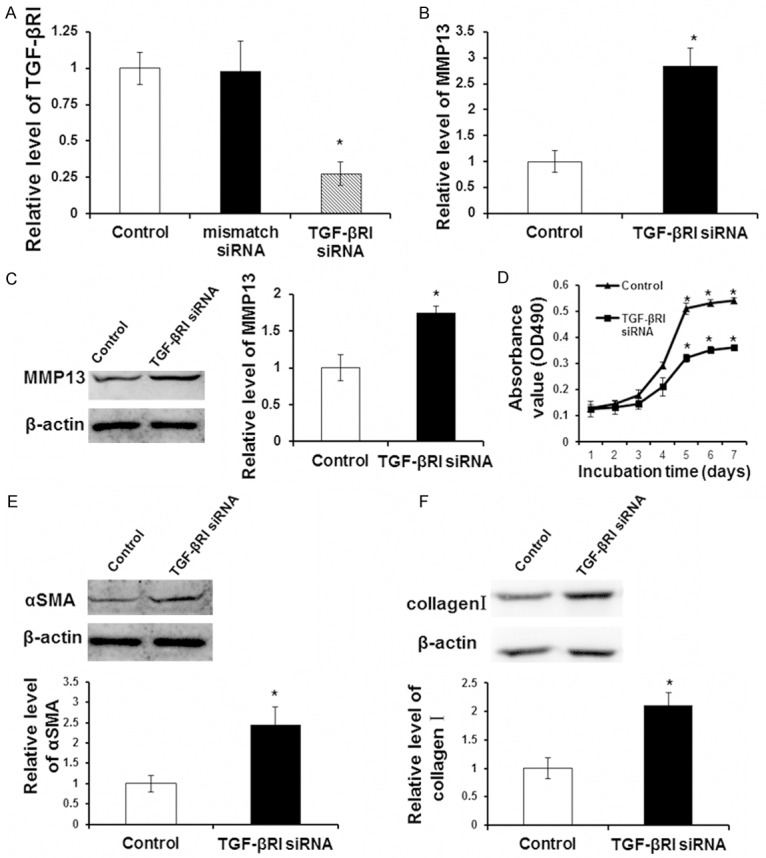
Inhibition of TGF-β RI by siRNA silencing. LX-2 cells were transfected with mismatch siRNA, TGF-β RI siRNA or un-transfected (control group) and then were cultured for 48 h. A. Transfection efficacy was detected by qRT-PCR. Relative level of TGF-β RI was shown. B. MMP13 expression at mRNA level after TGF-β RI siRNA was detected by qRT-PCR. C. MMP13 expression at protein level after TGF-β RI siRNA was detected by Western blot analysis. Representative and quantitative Western blot results were shown. D. Cell proliferation was detected by MTT assay. E. The αSMA expression at protein level after TGF-β RI siRNA was detected by Western blot analysis. Representative and quantitative Western blot results were shown. F. The collagen I expression at protein level after TGF-β RI siRNA was detected by Western blot analysis. Representative and quantitative Western blot results were shown. Compared with control group, *P < 0.05.
Discussion
Hepatic fibrosis is the key factor that leading to cirrhosis and liver failure in the final. Zn is an essential trace element for human body, which plays roles in many aspects such as regulating antioxidant, anti-inflammatory and anti apoptosis [22]. Besides, Zn has an important role on the regulation of liver fibrosis [23]. In this study, it was found that with the concentration of added Zn increased, the activity of LX-2 cells was inhibited. The proliferation ability decreased significantly, meanwhile, αSMA and type I collagen transcription and protein content decreased significantly, which, demonstrated that collagen synthesis was inhibited. This inhibitory effect was most obvious when the added Zn was up to a final concentration of 200 μM.
MMP-13 plays an important role in the degradation of collagen [10]. This study showed that, MMP-13 transcription an protein content were increased by Zn added and this significantly inhibited the expression of αSMA and type I collagen. MMP-13 is a matrix metalloproteinase involved in the regulation of hepatic fibrosis. This study failed to detect the influence of Zn on the expression of other MMPs, however, related experiments will be carried out in follow-up works. TIMP1 plays an inhibitory effect on matrix metalloproteinases, imbalance between them is an important reason that leads to liver fibrosis, in this study, it was shown that Zn did not affect the protein content of TIMP1. These results demonstrated that MMP-13 played a key role in the process of collagen synthesis inhibition by Zn in LX-2 cells.
TGF-β is the most powerful fibrosis promoting cytokine in HSCs [15] and it is demonstrated that the TGF-β signaling pathway can regulate the expression of many proteins. In the present study, it was found that 200 μM Zn could significantly decrease the protein content TGF-β RI, but did not affect protein content of TGF-β RII. Further study found that, TGF-β expression was similarly promoted while TGF-β RI was silenced using siRNA. Meanwhile, proliferative capacity of LX-2 cells and the expression of αSMA and type I collagen were significantly restrained. Zn might functions through inhibiting TGF-β RI gene expression or increasing TGF-β RI protein degradation, unfortunately, the specific molecular mechanism had not been elucidated. Related problems remain to be explored in the future.
In summary, in this study, human hepatic stellate cell line LX-2 was used as study model, Zn with a concentrate of 200 μM could significantly inhibit the activity of LX-2 cells for cell proliferation ability was significantly decreased. TGF-β signaling pathway played a key role in this process, by inhibiting TGF-β RI protein content and promoting the expression of MMP-13, Zn significantly inhibited expression of αSMA and type I collagen in LX-2 cells. Meanwhile, TGF-β RI inhibition by siRNA silencing had the same effect of αSMA and type I collagen expression inhibition.
Acknowledgements
This work was supported by Sichuan Provincial Health Department Project (100282).
Disclosure of conflict of interest
None.
References
- 1.Pietrangelo A. Metals, oxidative stress, and hepatic fibrogenesis. Semin Liver Dis. 1996;16:13–30. doi: 10.1055/s-2007-1007215. [DOI] [PubMed] [Google Scholar]
- 2.Ikejima K, Honda H, Yoshikawa M, Hirose M, Kitamura T, Takei Y, Sato N. Leptin augments inflammatory and profibrogenic responses in the murine liver induced by hepatotoxic chemicals. Hepatology. 2001;34:288–97. doi: 10.1053/jhep.2001.26518. [DOI] [PubMed] [Google Scholar]
- 3.Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterology. 2008;134:1655–69. doi: 10.1053/j.gastro.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Li D, Friedman SL. Liver fibrogenesis and the role of hepatic stellate cells: new insights and prospects for therapy. J Gastroenterol Hepatol. 1999;14:618–33. doi: 10.1046/j.1440-1746.1999.01928.x. [DOI] [PubMed] [Google Scholar]
- 5.Schuppan D. Structure of the extracellular matrix in normal and fibrotic liver: collagens and glycoproteins. Semin Liver Dis. 1990;10:1–10. doi: 10.1055/s-2008-1040452. [DOI] [PubMed] [Google Scholar]
- 6.Milani S, Herbst H, Schuppan D, Kim KY, Riecken EO, Stein H. Procollagen expression by nonparenchymal rat liver cells in experimental biliary fibrosis. Gastroenterology. 1990;98:175–84. doi: 10.1016/0016-5085(90)91307-r. [DOI] [PubMed] [Google Scholar]
- 7.Inagaki Y, Nemoto T, Kushida M, Sheng Y, Higashi K, Ikeda K, Kawada N, Shirasaki F, Takehara K, Sugiyama K, Fujii M, Yamauchi H, Nakao A, de Crombrugghe B, Watanabe T, Okazaki I. Interferon alfa down-regulates collagen gene transcription and suppresses experimental hepatic fibrosis in mice. Hepatology. 2003;38:890–9. doi: 10.1053/jhep.2003.50408. [DOI] [PubMed] [Google Scholar]
- 8.Kim Y, Peyrol S, So CK, Boyd CD, Csiszar K. Coexpression of the lysyl oxidase-like gene (LOXL) and the gene encoding type III procollagen in induced liver fibrosis. J Cell Biochem. 1999;72:181–8. doi: 10.1002/(sici)1097-4644(19990201)72:2<181::aid-jcb3>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 9.Masuda H, Fukumoto M, Hirayoshi K, Nagata K. Coexpression of the collagen-binding stress protein HSP47 gene and the alpha 1(I) and alpha 1(III) collagen genes in carbon tetrachloride-induced rat liver fibrosis. J Clin Invest. 1994;94:2481–8. doi: 10.1172/JCI117617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Okazaki I, Watanabe T, Hozawa S, Arai M, Maruyama K. Molecular mechanism of the reversibility of hepatic fibrosis: with special reference to the role of matrix metalloproteinases. J Gastroenterol Hepatol. 2000;15(Suppl):D26–32. doi: 10.1046/j.1440-1746.2000.02185.x. [DOI] [PubMed] [Google Scholar]
- 11.Knittel T, Mehde M, Grundmann A, Saile B, Scharf JG, Ramadori G. Expression of matrix metalloproteinases and their inhibitors during hepatic tissue repair in the rat. Histochem Cell Biol. 2000;113:443–53. doi: 10.1007/s004180000150. [DOI] [PubMed] [Google Scholar]
- 12.Watanabe T, Niioka M, Ishikawa A, Hozawa S, Arai M, Maruyama K, Okada A, Okazaki I. Dynamic change of cells expressing MMP-2 mRNA and MT1-MMP mRNA in the recovery from liver fibrosis in the rat. J Hepatol. 2001;35:465–73. doi: 10.1016/s0168-8278(01)00177-5. [DOI] [PubMed] [Google Scholar]
- 13.Lee HS, Huang GT, Miau LH, Chiou LL, Chen CH, Sheu JC. Expression of matrix metalloproteinases in spontaneous regression of liver fibrosis. Hepatogastroenterology. 2001;48:1114–7. [PubMed] [Google Scholar]
- 14.Watanabe T, Niioka M, Hozawa S, Kameyama K, Hayashi T, Arai M, Ishikawa A, Maruyama K, Okazaki I. Gene expression of interstitial collagenase in both progressive and recovery phase of rat liver fibrosis induced by carbon tetrachloride. J Hepatol. 2000;33:224–35. doi: 10.1016/s0168-8278(00)80363-3. [DOI] [PubMed] [Google Scholar]
- 15.Rana SV. Simultaneous protective effect of a new chelating agent and zinc, on the carbon tetrachloride induced hepatic injury in squirrels. Res Exp Med (Berl) 1977;170:217–27. doi: 10.1007/BF01851432. [DOI] [PubMed] [Google Scholar]
- 16.Maret W. Zinc coordination environments in proteins determinezinc functions. J Trace Elem Med Biol. 2005;19:7–12. doi: 10.1016/j.jtemb.2005.02.003. [DOI] [PubMed] [Google Scholar]
- 17.Zalewski PD, Truong-Tran AQ, Grosser D, Jayaram L, Murgia C, Ruffin RE. Zinc metabolism in airway epithelium andairway inflammation: basic mechanisms and clinical targets review. Pharmacol Ther. 2005;105:127–149. doi: 10.1016/j.pharmthera.2004.09.004. [DOI] [PubMed] [Google Scholar]
- 18.Tudor R, Zalewski PD, Ratnaike RN. Zinc in health andchronic disease. J Nutr Health Aging. 2005;9:45–51. [PubMed] [Google Scholar]
- 19.Mocchegiani E, Bertoni-Freddari C, Marcellini F, Malavolta M. Brain, aging and neurodegeneration: role of zinc ion availability. Prog Neurobiol. 2005;75:367–390. doi: 10.1016/j.pneurobio.2005.04.005. [DOI] [PubMed] [Google Scholar]
- 20.Pinzani M, Gentilini A, Caligiuri A, De Franco R, Pellegrini G, Milani S, Marra F, Gentilini P. Transforming growth factor-β1 regulates platelet-derived growth factor receptor β subunit in human liver fat-storing cells. Hepatology. 1995;21:232–9. [PubMed] [Google Scholar]
- 21.Hambidge M, Krebs NF. Interrelationships of key variables of human zinc homeostasis: relevance to dietary zinc requirements. Annu Rev Nutr. 2001;21:429–52. doi: 10.1146/annurev.nutr.21.1.429. [DOI] [PubMed] [Google Scholar]
- 22.Powell SR. The antioxidant properties of zinc. J Nutr. 2000;130:1447S–54S. doi: 10.1093/jn/130.5.1447S. [DOI] [PubMed] [Google Scholar]
- 23.Rocchi E, Borella P, Borghi A, Paolillo F, Pradelli M, Farina F, Casalgrandi G. Zinc and magnesium in liver cirrhosis. Eur J Clin Invest. 1994;24:149–55. doi: 10.1111/j.1365-2362.1994.tb00980.x. [DOI] [PubMed] [Google Scholar]
- 24.Bode JC, Hanisch P, Henning H, Koenig W, Richter FW, Bode C. Hepatic zinc content in patients with various stages of alcoholic liver disease and in patients with chronic active and chronic persistent hepatitis. Hepatology. 1988;8:1605–9. doi: 10.1002/hep.1840080622. [DOI] [PubMed] [Google Scholar]
- 25.Anttinen H, Oikarinen A, Puistola U, Pääkkö P, Ryhänen L. Prevention by zinc of rat lung collagen accumulation in carbon tetrachloride injury. Am Rev Respir Dis. 1985;132:536–40. doi: 10.1164/arrd.1985.132.3.536. [DOI] [PubMed] [Google Scholar]
- 26.Seltzer JL, Jeffrey JJ, Eisen AZ. Evidence for mammalian collagenases as zinc ion metalloenzymes. Biochim Biophys Acta. 1977;485:179–87. doi: 10.1016/0005-2744(77)90205-4. [DOI] [PubMed] [Google Scholar]


