Abstract
Babesiosis is a global zoonotic disease acquired by the bite of a Babesia-infected Ixodes tick or through blood transfusion with clinical relevance affecting humans and animals. In this study, we evaluated a series of small molecule compounds that have previously been shown to target specific apicomplexan enzymes in Plasmodium, Toxoplasma and Cryptosporidium. The compounds, bumped kinase inhibitors (BKIs), have strong therapeutic potential targeting apicomplexa-specific calcium dependent protein kinases (CDPKs). We investigated if BKIs also show inhibitory activities against piroplasms such as Babesia. Using a subset of BKIs that have promising inhibitory activities to Plasmodium and Toxoplasma, we determined that their actions ranged from 100% and no inhibition against B. bovis blood stages. One specific BKI, RM-1-152, showed complete inhibition against B. bovis within 48h and was the only BKI that showed noticeable phenotypic changes to the parasites. Focusing our study on this BKI, we further demonstrated that RM-1-152 has Babesia-static activity and involves the prohibition of merozoite egress while replication and re-invasion of host cells are unaffected. The distinct, abnormal phenotype induced by RM-1-152 suggests that this BKI can be used to investigate less studied cellular processes such as egression in piroplasm.
Keywords: Babesia, bumped kinase inhibitors, egression, static, chemotherapy
Graphical abstract
1. Introduction
Emerging and re-emerging infectious diseases cause significant global impact not only to human health (http://www.cdc.gov/ncidod/eid/vol4no3/fauci.htm) but also animal health with consequent economic impact. These diseases frequently emerge or expand their range as a result of changes in ecology, climate, and global trade (Jones et al., 2008). Babesiosis, a tick-borne zoonotic disease, is an imminent threat to the U.S. livestock industry and economy. Although eradicated from the continental U.S. (the Commonwealth of Puerto Rico remains endemic) in the 1940s, re-emergence from Mexico, where the disease is endemic, is a major risk especially for bovine babesiosis. This risk is increasing due to increased trade, expansion of wildlife reservoirs for the tick vectors (Rhipicephalus annulatus and R. microplus), and the development of multi-acaricide resistant ticks in Mexico. Current prophylaxis involves the immunization of naïve cattle with live attenuated vaccine. Due to safety concerns, this preventive care is not registered in the U.S. The administration of imidocarb dipropionate (3.5 mgkg−1 intramuscularly) as a chemotherapy for infected animals is available but residual metabolite in musculature and milk renders the treated animal unmarketable.
Similarly, human babesiosis is a growing public health concern in the United States (Herwaldt et al., 2011). The incidence of this zoonosis, mostly due to B. microti infections has increased significantly over the last decade with eighty percent (80%) of babesiosis cases documented in the U.S. since 1969 were reported between 2000–2009 (Herwaldt et al., 2004). Human babesiosis is especially problematic in the U.S. Northeast corridor (Rodgers and Mather, 2007) although the rise of this zoonotic cases is a global trend (Herwaldt et al., 2004; Kim et al., 2007; Senanayake et al., 2012) attributable to increased blood transfusions, climate changes expanding tick habitats and human encroachment into tick-infested areas (Aguirre and Tabor, 2008). Due to a lack of rapid and sensitive screening protocols for donated blood, transfusion recipients such as children and immunocompromised patients are the primarily affected cohort (Krause et al., 2008; Rodgers and Mather, 2007; Vannier and Krause, 2009). Co-infection of babesiosis and Lyme disease is fairly common, given that both pathogens are spread by the same tick vector, Ixodes scapularis. Some reports document 66% of Lyme patients, may also be co-infected with Babesia (Krause et al., 1996). Although drug regimen, mostly antibiotic combination therapies, inhibiting B. microti exist (Iseki et al., 2008; Salama et al., 2012; Ueno et al., 2013), these chemotherapeutics either require high dosage to be effective or have undesirable side effects (Galay et al., 2011; Munkhjargal et al., 2012; Salama et al., 2012).
Advances in kinase biology have described a class of highly-conserved, apicomplexan-specific serine/threonine enzymes known as calcium dependent protein kinases (CDPKs). CDPKs are involved in multiple cellular processes such as host cell invasion, gliding motility and secretion (Lovett et al., 2002; Lovett and Sibley, 2003). Homologues of apicomplexan CDPKs have not been found in animals, hence, they have the potential to be exploited as selective drug targets for apicomplexan parasites. This revelation led to the development of bumped kinase inhibitors (BKIs) that specifically target ATP-binding domains with characteristically small gatekeeper residues as found in Plasmodium falciparum CDPK4, Toxoplasma gondii CDPK1 and Cryptosporidium parvum CDPK1 (Doerig et al., 2005; Johnson et al., 2012; Murphy et al., 2010; Ojo et al., 2010; Ojo et al., 2012). BKIs have low inhibition of host protein kinases, as ATP-binding domains of mammalian kinases typically have larger gatekeeper residues (e.g. human SRC, ABL) that exclude the binding of BKIs (Doerig et al., 2005; Johnson et al., 2012; Murphy et al., 2010; Ojo et al., 2010; Ojo et al., 2012). Therefore, BKIs offer tremendous selectivity for inhibition of apicomplexan CDPKs over human kinases.
CDPK orthologues have not been characterized in piroplasms such as Babesia and Theileria (Brayton et al., 2007; Cornillot et al., 2013; Cornillot et al., 2012; Kappmeyer et al., 2012), although genome sequencing of these pathogens reveal putative CDPKs with sufficiently small gatekeeper residues in the ATP-binding domains for selective BKI-targeting. In fact, we recently identified such a CDPK, specifically a bona fide CDPK4 in B. microti and its bovine relative, B. bovis (Keyloun et al., 2014). We also determined that additional protein kinases may function as CDPKs or act as BKI-targets from our in silico structural analysis using primary protein sequences of Plasmodium and Toxoplasma CDPKs and screening both B. microti and B. bovis genomes (Table 1) although CDPK1 orthologues were not identified in both genomes. In order to confirm that BKIs are effective in the inhibition of piroplasms such as Babesia, this study (i) evaluates a subset of BKIs that were previously shown to be inhibitory against related apicomplexans and (ii) reports those that are growth inhibitory against Babesia asexual erythrocytic stages using B. bovis as a model system and (iii) suggests one BKI target may be involved in egression and not in erythrocyte invasion.
Table 1.
Putative bumped kinase inhibitor targets in Babesia microti and orthologous proteins in B. bovis based on conserved ATP-binding domain. Gatekeeper residue and putative function for each protein is also shown.
| B. microti | gatekeeper | annotation | B. bovis | gatekeeper | annotation |
|---|---|---|---|---|---|
| CCF74481 | Thr131 | CDPK-like | BBOV_IV003210 | Thr141 | CDPK4 |
| CCF74596 | Val183 | CDPK-like | BBOV_II007640 | Met219 | CDPK, putative |
| CCF75526 | Thr87 | AMP-dependent PK | BBOV_III009120 | Met119 | PK containing domain |
| CCF75730 | Thr133 | cAMP-dependent PK | BBOV_III005470 | Met374 | PK containing domain |
| CCF75355 | Thr623 | cGMP-dependent PK | BBOV_I004690 | Thr654 | cGMP-dependent PK |
| CCF75428 | Ala163 | Ser/Thr kinase | BBOV_III007550 | Thr155 | PK containing domain |
| CCF75969 | Met131 | Ser/Thr kinase | BBOV_II000760 | Ser460 | PK containing domain |
PK, protein kinase; Thr, threonine, Ser, serine; Val, valine; Met, methionine; Ala, alanine
2. Materials and Methods
2.1 Compound Library
Bumped kinase inhibitors were synthesized as previously described (Doerig et al., 2005; Johnson et al., 2012; Murphy et al., 2010; Ojo et al., 2010; Ojo et al., 2012). Purity of all compounds (>98%) was confirmed by reverse-phase HPLC and 1H-NMR before dissolution in 100% DMSO. At a final stock concentration of 20 mM, aliquots were stored at −20°C.
2.2 In vitro cultivation of B. bovis T2Bo virulent strain
Parasites were grown and expanded in long-term microaerophilous stationery phase conditions as previously described (Goff and Yunker, 1986; Levy and Ristic, 1980).
2.3 Evaluation of Babesia growth inhibition of BKIs
Using the B. bovis asexual erythrocytic stage expansion culture, parasites were seeded in flat bottom 96-well tissue culture plates. Each well contained 150 μl of culture at 10% hematocrit with a starting percent parasitized erythrocytes (PPE) of 0.5 as determined by microscopic counting of smears stained with Diff-Quick® (Dade Behring). Drug compound stock solutions were provided at 20 mM in 100% DMSO and aliquots were diluted daily to 30 μM with HL-1 culture media (BioWhittaker®). Controls included no treatment (NT) and imidocarb dipropionate (ID) (Imizol® Schering-Plough Animal Health) which is a well established Babesia-cidal compound. Final DMSO concentration was 0.15% for all conditions. Eighty percent of drug containing media was replaced at 24h intervals. PPE was enumerated over a 72h period by manual counting of 2 μl culture smears stained with Diff-Quick® using a Zeiss Axioskop microscope fitted with a 100x oil-immersion phase contrast objective. Initial drug screening to determine which BKIs were inhibitory against B. bovis was performed in a single assay. Percent parasitemia was measured to evaluate the proportion of morphological abnormal parasites to normal ones in both 24h and 48h treated cultures. Compounds that exhibited significant parasite growth retardation when assayed at 30 μM were further tested at 0.3 μM–12.0 μM in the same manner as described above in triplicates.
2.4 Determination of parasite-static verses-cidal effect of RM-1-152
RM-1-152 at 30 μM was prepared as described above. B. bovis asexual erythrocytic stage culture at 2.5 PPE and 10% hematocrit was used for all conditions. RM-1-152 evaluations were conducted in quadruplicates. ID, clindamycin hydrochloride (CL) (Sigma, St. Louis, MO), both at 20 μM were used as Babesia-cidal and -static controls, respectively (Arai et al., 1998; Brasseur et al., 1998; Rashid et al., 2008) while NT was included as the negative control. Culture media were changed daily and 50 μl was collected from each condition for real time PCR analysis at designated time points described below. Cultures in the presence of drugs were exposed ≤ 48h followed by 3x washing with HL-1 medium to remove all drug traces. The cultures were then incubated in normal medium for the remaining of the study. The experiment was terminated after 144h upon which the cultures were treated with Trizol for RNA (Invitrogen, Grand Island, NY) and cDNA preparation made with the RETRO script Reverse Transcription kit (Ambion, Grand Island, NY). Fifty μl of culture was collected at time 0h and the same volume was collected at the termination of the experiment (i.e. 144h). cDNA templates were amplified in quadruplicate using iSYBR Green Supermix (Bio-Rad, Hercules, CA) according to manufacturer protocol on a Bio-Rad CFX96 instrument. The cycling conditions were as follows, 95°C for 3 min, followed by 40 cycles of 95°C for 10 s, 55°C for 10 s, and 72°C for 30 s. Melt curve analysis was done using 95°C for 10 s, 65°C to 95°C with 0.5°C increment of 5s/step. Bio-Rad CFX Manager 3.1 software was used for data analysis. Two genes were selected to determine if RM-1-152 effect was Babesia-cidal or -static. These were topoisomerase II (BBOV_II004820) and 40S ribosomal protein S3 encoding gene (BBOV_II001650). Both genes are single copy genes within the genome and are highly expressed during the asexual erythrocytic stages as determined previously from our microarray analysis (Pedroni et al., 2013). Gene specific primers were designed (MacVector v.12.0) to amplify product sizes of 100–200 bp. Because topoisomerase II gene has no intron, primers designed for 40S ribosomal protein S3 encoding gene span the intronic region to rule out gDNA contamination (Table S1).
2.5 Determination of RM-1-152 effect influence on re-invasion
B. bovis culture was also used to investigate if RM-1-152 inhibits Babesia reinvasion. Two groups of culture were set up where group one consisted of 30 μM RM-1-152 treatment for ≤ 48h while group two was the NT control. This time point was chosen as the culture begins to die off approximately 48h under continuous RM-1-152 treatment (Figure 1). Infected erythrocytes were lysed, washed 3x with HL-1 media and centrifuged at 1,800 rpm for 5 min at room temperature to pellet the freed merozoites. These merozoites were divided into two aliquots and used to re-invade fresh erythrocytes in the presence or absence of 30 μM RM-1-152 for a total of 72h. NT-free merozoites in the presence and absence of RM-1-152 were included as controls. The experiment was conducted in triplicates in the same culture condition as described in section 2.4. Blood smears were stained with Diff-Quick® and using a Zeiss Axioskop microscope fitted with a 100x oil-immersion phase contrast objective, PPE was enumerated and morphological changes were recorded.
Figure 1.
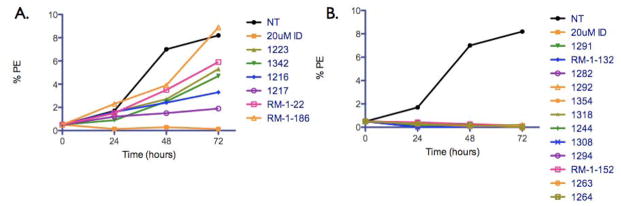
Effect of bumped kinase inhibitors (BKIs) at 30 μM on Babesia asexual erythrocytic stages over 72h. (A) BKIs with partial to no inhibition and (B) BKIs with inhibitory effect as early as 24h post-incubation. NT, no treatment control; ID, imidocarb diproprionate positive control; PE, parasitized erythrocytes.
2.6 Statistical analysis
Mean and standard error means (SEM) of PPE under the influence of the BKIs based on replicates and non-linear regression curve were calculated using PRISM v. 5.0a.
3. Results and Discussion
To determine whether BKIs inhibit asexual erythrocytic Babesia growth, 18 BKIs were chosen based either on successful inhibitory effect against related apicomplexans (Larson et al., 2012) or because they were closely related to the active compounds (Figure S1, Table 2). Our results showed that constant exposure of the parasites to these BKIs at 30 μM demonstrated a diversity of action. Figure 1A illustrates six BKIs that had intermediate or no inhibitory activities on B. bovis growth while the remaining 12 had complete inhibitory action as early as 24h post-incubation (Figure 1B). A plausible explanation as to why only 66% (n=12) of the tested BKIs were effective against Babesia sp. is because these inhibitors were originally designed to bind T. gondii CDPK1, which contains a hydrophobic pocket with a glycine gatekeeper residue. B. bovis does not appear to contain a bona fide CDPK1 orthologue and that all Babesia CDPKs that we identified do not have such an atypically small gatekeeper (Table 1). Presumably, the “bumped” chemical group of BKIs exhibits steric clash with the larger gatekeeper residues, disfavoring binding. The seven putative kinases identified in B. microti and B. bovis genomes with conserved ATP-binding domains do contain “relatively” small gatekeeper residues which suggest that the inhibitory action of the 12 BKIs that was observed may be target specific after all. Furthermore, we cannot rule out that additional regions on the Babesia CDPKs may also influence the effectiveness of the BKIs’ overall action and that these regions differ from those in Plasmodium, Toxoplasma and Cryptosporidium. Studies have shown that some BKIs affect host cell invasion by binding to specific CDPKs such as CDPK1 (Johnson et al., 2012). With that said, although these BKIs may have different B. bovis targets as in other apicomplexans, the final outcome/readout is the same.
Table 2.
Effective concentration (EC)50 values, structures and Src cytotoxicity values of bumped kinase inhibitors (BKIs) that were inhibitory to Babesia bovis blood stage grown over 72 h.
| BKIs | Structure | EC50 (μM) | Src cytotoxicity (μM) |
|---|---|---|---|
| 1244 |
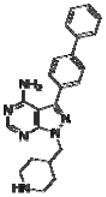
|
8.0 | NA |
| 1263 |
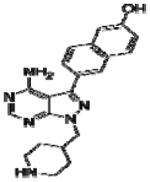
|
3.0 | NA |
| 1264 |
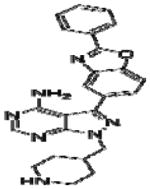
|
9.0 | >10 |
| 1282 |
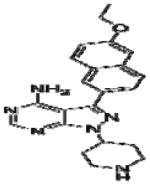
|
5.9 | >5.02 |
| 1291 |
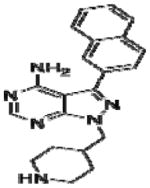
|
26.0 | >10 |
| 1292 |
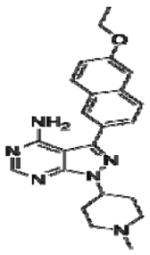
|
9.9 | >5.58 |
| 1294 |
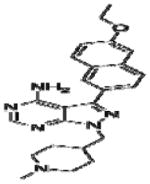
|
7.4 | >10 |
| 1308 |
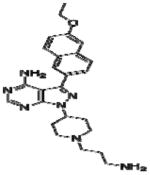
|
5.5 | 0.73 |
| 1318 |
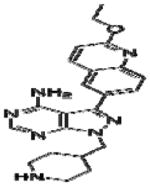
|
11.0 | >10 |
| 1354 |
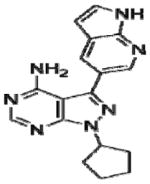
|
2.2 | 0.08 |
| RM-1-132 |
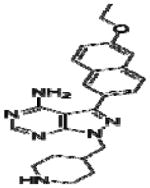
|
5.0 | >20 |
| RM-1-152 |
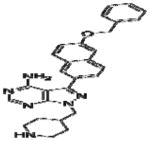
|
11.2 | >10 |
Effective concentration (EC)50 values of all 20 BKIs were evaluated as shown in Table 2 where compound 1291 and 1354 showed the highest and lowest EC50 values at 26 μM and 3 μM, respectively. The remaining 10 BKIs have similar EC50 values that range from 5.0 μM such as RM-1-132 to 11.2 μM as in RM-1-152. Many of the tested BKIs were previously evaluated with favorable results for specificity against human kinases such as SRC whose minimum inhibitory concentrations are also represented in Table 2. Certain BKIs, namely, RM-1-132, 1318, and 1294 are designed to be exquisitely selective which means they are likely to interact with a single kinase in B. bovis.
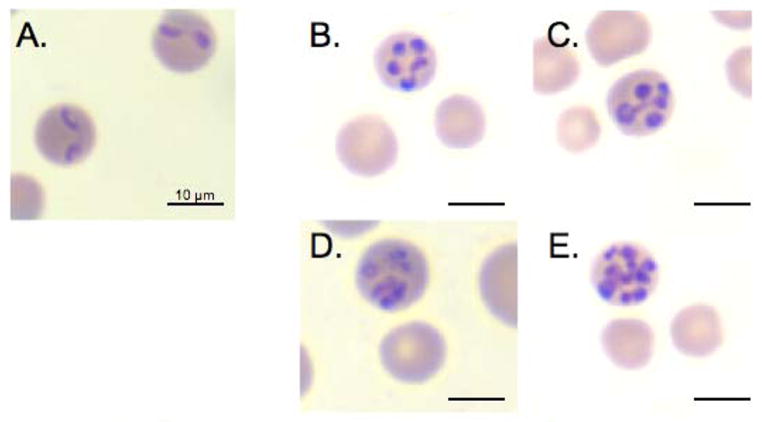
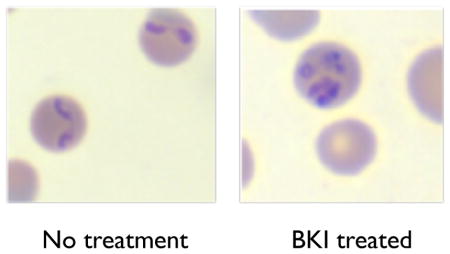
Bumped kinase inhibitors have previously been associated with phenotypic inhibition of invasion and egression of parasites from mammalian host cells (Doerig et al., 2005; Johnson et al., 2012; Murphy et al., 2010; Ojo et al., 2010; Ojo et al., 2012). High resolution microscopic analysis of all drug-treated cultures revealed only one anti-babesial BKI, RM-1-152, that showed phenotypic effects (merozoite entrapment and abnormal morphology) that suggest interference with parasite egression. Under normal conditions, B. bovis merozoites invade erythrocytes, each developing into a single trophozoite per cell. The trophozoite replicates by binary fission and results in two, tear-drop shaped merozoites (Figure 2A) that subsequently egress independently to re-invade uninfected erythrocytes. However, under the constant influence of 30 μM RM-1-152, Babesia replicated within host cells but failed to egress. These trapped merozoites remained within the infected cell and cumulated in numbers over 24h (Figure 2C-F). Trapped merozoites within infected erythrocytes also appeared round/oval instead of the typical tear-dropped shape. The maximum and minimum numbers of RM-1-152 treated-round merozoites within a single host cell were 16 and three, respectively. Percent parasitemia of morphologically abnormal B. bovis under RM-1-152 treatment for 24h and 48h were approximately 56% and 30%, respectively. The decrease of percent abnormal parasites over time was unexpected. One possible explanation is that there was an overall parasitemia decline over 24h due to RM-1-152’s inhibitory effect and the parasites that were already affected by the treatment simply died while those that escaped from the drug treatment continued to replicate asexually. Nonetheless, presence of RM-1-152 remains lethal by 72h.
Figure 2.
Visualization of Babesia bovis under the influence of 30 μM RM-1-152 after 24h. (A) No treatment control, showing twin tear-drop shaped merozoites occupying a single erythrocyte. (B-C) Five circular merozoite-like parasites within a single erythrocyte and (D-E) infected erythrocytes harboring >5 circular merozoites per cell under continuous presence of RM-1-152.
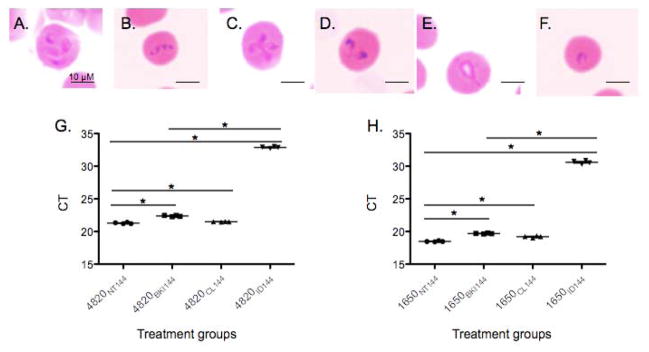
Due to the fact that RM-1-152 was the only BKI where we captured a phenotypic change in the parasite’s appearance, we focused our study by determining the inhibitory action of this BKI. In order to determine if RM-1-152 causes irreversible consequences to B. bovis growth, two assays were carried out. In the first assay, B. bovis cultures were either left untreated (NT control) or exposed to RM-1-152 ≤ 48 h followed by merozoite isolation for re-invasion ability assessment. Using equal quantity of free merozoites in both groups confirmed via light microscopy, merozoites that were previously exposed to RM-1-152 reinvaded uninfected erythrocytes regardless of the presence (abnormal numbers of merozoites) (Figure 3A) or absence (normal number of merozoites) (Figure 3B) of RM-1-152 in the media. However, once these merozoites became intracellular, those that were re-exposed to RM-1-152 in the media developed abnormal morphology (Figure 3C) but the previously drug-exposed merozoites recovered in the absence of RM-1-152 (Figure 3D). Similarly, free merozoites from the NT group (Figure 3B) reinvaded fresh erythrocytes in the presence and absence of RM-1-152. However, these intracellular merozoites when re-exposed to RM-1-152, developed abnormal morphology (Figure 3E) while those that were allowed to propagate free of RM-1-152, showed normal morphology (Figure 3F). These results demonstrate that RM-1-152 does not prohibit host cell invasion by the merozoites that were previously exposed to the compound but that it interferes with egression alone. One plausible explanation for this observation is that different enzymes, such as CDPKs, are responsible for the two independent processes where RM-1-152 is target-specific to the kinase involved in egress. The fact that after NT-merozoites exposure to RM-1-152 became intracellular but failed to egress, further strengthens the suspicion that RM-1-152 targets egress-specific CDPKs.
Figure 3.
Babesia bovis successfully re-invaded uninfected erythrocytes upon the removal of bumped kinase inhibitor’s effect. Merozoites from the RM-1-152-treated (A) and no treatment (NT) (B) groups were freed by erythrocyte lysis. (C) and (D) correspond to merozoites-infected erythrocytes previously exposed to RM-1-152 in the presence (C) and absence (D) of 30 μM RM-1-152 at 72h, respectively while (E) and (F) correspond to NT-merozoites reinvaded erythrocytes in the presence and absence of 30 μM RM-1-152 at 72h, respectively. Comparison of B. bovis-infected erythrocytes using quantitative RT-PCR amplifying (G) BBOV_II004820 and (H) BBOV_II001650 transcripts in four treatment groups at 144h. No treatment, NT control; BKI, RM-1-152 at 30 μM; CL, 20 μM clindamycin hydrochloride and ID, 20 μM imidocarb dipropionate. *, statistically significant with p<.05.
In the second assay, qRT-PCR was employed to confirm that RM-1-152 treated Babesia remains viable because RM-1-152 is a Babesia-static inhibitor. Clindamycin hydrochloride was used as a Babesia-static inhibitor (Arai et al., 1998) where ID, was used as the Babesia-cidal inhibitor controls. The detection of topoisomerase II (Figure 3G) and 40s ribosomal (Figure 3H) protein transcripts confirmed that the parasites were viable upon removal of the compound. Furthermore, B. bovis in the NT, CL and BKI groups were successful in invasion, replication and egression of the erythrocytes, which were in contrast to the ID group. Among the NT, CL and BKI groups, slight statistically significant higher parasitemia was observed in the NT group with lowest cycle titration (CT) values than CL and BKI groups while BKI and CL groups had the same level of parasitemia. It is possible that CL and BKI treatment groups resulted in permanent damage to the parasites or that the parasites required more than 144h to recover from the static effect of both inhibitors. As expected, parasites previously treated with ID failed to recover (Figure 3G, H).
In summary, this study reports anti-Babesia activity of BKIs. Among the 12 BKIs tested to be inhibitory, RM-1-152’s action is correlated with the inhibition of B. bovis egress process but not invasion of the host cell. Under continuous exposure of the BKIs, B. bovis replicated but the culture eventually died. This is most likely attributed to factors such as diminishing supply of host nutrients, as trapped parasites remained replicative prior to 48h. Earlier observations suggest that each CDPK has defined regulatory functions at distinct stages of the apicomplexan life cycle. Expression levels and specific functions of CDPKs in each organism may vary based on the specific life cycle requirements (Keyloun, et al., 2014), and that RM-1-152 inhibits a different BKI than the other BKIs. It is also concluded that the inhibitory action of RM-1-152 is Babesia-static, consistent with previously described effects of BKIs (Doerig et al., 2005; Johnson et al., 2012; Murphy et al., 2010; Ojo et al., 2010; Ojo et al., 2012). With that said, our recent study evaluating the action of these BKIs in Theileria equi suggests that these compounds are broadly inhibitory against members of the apicomplexan phylum which is potentially good news as these may be possible treatment option for human babesiosis caused by B./T. microti (Cornillot et al., 2012; Hines et al., 2015). Future efforts will be directed towards the identification of BKI targets, refinement of BKI structural designs for optimizing target specificity and host toxicity, and the investigation of potential resistance to BKI treatment using B. bovis as the in vitro model system. Future experiments also include in vivo evaluation of these BKIs’ action on B. microti as in vitro culture system is not feasible for this Babesia/Theileria (Cornillot et al., 2012). Last but not least, host cell egression mechanism is a knowledge gap in the piroplasm field. With the help of RM-1-152, characterization of such an essential and potential drug-target pathway can finally be initiated.
Supplementary Material
Highlights.
Bumped kinase inhibitors are effective anti-babesia agents
Morphological changes under the influence of one inhibitor are noted
Mechanism of egression may be the target of these inhibitors
Babesia genome predicts seven putative bumped kinase inhibitor targets
Acknowledgments
This work is supported by the National Institutes of Health grants R01AI089441, R01 AI111341-01 (W.C.V.), R01GM086858 (D.J.M.), the National Science Foundation ADVANCE grant (A.O.T.L.) and USDA # 2014-06183. In addition, K.R.K. was supported by a training scholarship from the University of Washington Plain Endowment for Geriatric Pharmacy Research.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aguirre AA, Tabor GM. Global factors driving emerging infectious diseases. Ann N Y Acad Sci. 2008;1149:1–3. doi: 10.1196/annals.1428.052. [DOI] [PubMed] [Google Scholar]
- Arai S, Tsuji M, Kim SJ, Nakade T, Kanno Y, Ishihara C. Babesia canis infection in canine-red blood cell-substituted SCID mice. Int J Parasitol. 1998;28:1429–1435. doi: 10.1016/s0020-7519(98)00094-0. [DOI] [PubMed] [Google Scholar]
- Brasseur P, Lecoublet S, Kapel N, Favennec L, Ballet JJ. In vitro evaluation of drug susceptibilities of Babesia divergens isolates. Antimicrob Agents Chemother. 1998;42:818–820. doi: 10.1128/aac.42.4.818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brayton KA, Lau AO, Herndon DR, Hannick L, Kappmeyer LS, Berens SJ, Bidwell SL, Brown WC, Crabtree J, Fadrosh D, Feldblum T, Forberger HA, Haas BJ, Howell JM, Khouri H, Koo H, Mann DJ, Norimine J, Paulsen IT, Radune D, Ren Q, Smith RK, Jr, Suarez CE, White O, Wortman JR, Knowles DP, Jr, McElwain TF, Nene VM. Genome sequence of Babesia bovis and comparative analysis of apicomplexan hemoprotozoa. PLoS Pathog. 2007;3:1401–1413. doi: 10.1371/journal.ppat.0030148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornillot E, Dassouli A, Garg A, Pachikara N, Randazzo S, Depoix D, Carcy B, Delbecq S, Frutos R, Silva JC, Sutton R, Krause PJ, Mamoun CB. Whole Genome Mapping and Re-Organization of the Nuclear and Mitochondrial Genomes of Babesia microti Isolates. PLoS ONE. 2013;8:e72657. doi: 10.1371/journal.pone.0072657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cornillot E, Hadj-Kaddour K, Dassouli A, Noel B, Ranwez V, Vacherie B, Augagneur Y, Bres V, Duclos A, Randazzo S, Carcy B, Debierre-Grockiego F, Delbecq S, Moubri-Menage K, Shams-Eldin H, Usmani-Brown S, Bringaud F, Wincker P, Vivares CP, Schwarz RT, Schetters TP, Krause PJ, Gorenflot A, Berry V, Barbe V, Ben Mamoun C. Sequencing of the smallest Apicomplexan genome from the human pathogen Babesia microti{dagger} Nucleic Acids Res. 2012 doi: 10.1093/nar/gks700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doerig C, Billker O, Pratt D, Endicott J. Protein kinases as targets for antimalarial intervention: Kinomics, structure-based design, transmission-blockade, and targeting host cell enzymes. Biochim Biophys Acta. 2005;1754:132–150. doi: 10.1016/j.bbapap.2005.08.027. [DOI] [PubMed] [Google Scholar]
- Galay RL, Maeda H, Aung KM, Umemiya-Shirafuji R, Xuan X, Igarashi I, Tsuji N, Tanaka T, Fujisaki K. Anti-babesial activity of a potent peptide fragment derived from longicin of Haemaphysalis longicornis. Trop Anim Health Prod. 2011;44:343–348. doi: 10.1007/s11250-011-0027-7. [DOI] [PubMed] [Google Scholar]
- Goff WL, Yunker CE. Babesia bovis: increased percentage parasitized erythrocytes in cultures and assessment of growth by incorporation of [3H]hypoxanthine. Exp Parasitol. 1986;62:202–210. doi: 10.1016/0014-4894(86)90024-x. [DOI] [PubMed] [Google Scholar]
- Herwaldt BL, de Bruyn G, Pieniazek NJ, Homer M, Lofy KH, Slemenda SB, Fritsche TR, Persing DH, Limaye AP. Babesia divergens-like infection, Washington State. Emerg Infect Dis. 2004;10:622–629. doi: 10.3201/eid1004.030377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herwaldt BL, Linden JV, Bosserman E, Young C, Olkowska D, Wilson M. Transfusion-Associated Babesiosis in the United States: A Description of Cases. Ann Intern Med. 2011 doi: 10.7326/0003-4819-155-8-201110180-00362. [DOI] [PubMed] [Google Scholar]
- Hines SA, Ramsay JD, Kappmeyer LS, Lau AO, Ojo KK, Van Voorhis WC, Knowles DP, Mealey RH. Theileria equi isolates vary in susceptibility to imidocarb dipropionate but demonstrate uniform in vitro susceptibility to a bumped kinase inhibitor. Parasit Vectors. 2015;8:33. doi: 10.1186/s13071-014-0611-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iseki H, Takabatake N, Ota N, Ishigame T, Yokoyama N, Igarashi I. Babesia: the protective effects of killed Propionibacterium acnes on the infections of two rodent Babesia parasites in mice. Exp Parasitol. 2008;118:543–548. doi: 10.1016/j.exppara.2007.11.006. [DOI] [PubMed] [Google Scholar]
- Johnson SM, Murphy RC, Geiger JA, DeRocher AE, Zhang Z, Ojo KK, Larson ET, Perera BG, Dale EJ, He P, Reid MC, Fox AM, Mueller NR, Merritt EA, Fan E, Parsons M, Van Voorhis WC, Maly DJ. Development of Toxoplasma gondii calcium-dependent protein kinase 1 (TgCDPK1) inhibitors with potent anti-toxoplasma activity. J Med Chem. 2012;55:2416–2426. doi: 10.1021/jm201713h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kappmeyer LS, Thiagarajan M, Herndon DR, Ramsay JD, Caler E, Djikeng A, Gillespie JJ, Lau AO, Roalson EH, Silva JC, Silva MG, Suarez CE, Ueti MW, Nene VM, Mealey RH, Knowles DP, Brayton KA. Comparative genomic analysis and phylogenetic position of Theileria equi. BMC Genomics. 2012;13:603. doi: 10.1186/1471-2164-13-603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keyloun KR, Reid MC, Choi R, Song Y, Fox AM, Hillesland HK, Zhang Z, Vidadala R, Merritt EA, Lau AO, Maly DJ, Fan E, Barrett LK, WCVANV, Ojo KK. The gatekeeper residue and beyond: homologous calcium-dependent protein kinases as drug development targets for veterinarian Apicomplexa parasites. Parasitology. 2014;141:1499–1509. doi: 10.1017/S0031182014000857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim JY, Cho SH, Joo HN, Tsuji M, Cho SR, Park IJ, Chung GT, Ju JW, Cheun HI, Lee HW, Lee YH, Kim TS. First case of human babesiosis in Korea: detection and characterization of a novel type of Babesia sp. (KO1) similar to ovine babesia. J Clin Microbiol. 2007;45:2084–2087. doi: 10.1128/JCM.01334-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause PJ, Gewurz BE, Hill D, Marty FM, Vannier E, Foppa IM, Furman RR, Neuhaus E, Skowron G, Gupta S, McCalla C, Pesanti EL, Young M, Heiman D, Hsue G, Gelfand JA, Wormser GP, Dickason J, Bia FJ, Hartman B, Telford SR, 3rd, Christianson D, Dardick K, Coleman M, Girotto JE, Spielman A. Persistent and relapsing babesiosis in immunocompromised patients. Clin Infect Dis. 2008;46:370–376. doi: 10.1086/525852. [DOI] [PubMed] [Google Scholar]
- Krause PJ, Telford SR, 3rd, Spielman A, Sikand V, Ryan R, Christianson D, Burke G, Brassard P, Pollack R, Peck J, Persing DH. Concurrent Lyme disease and babesiosis. Evidence for increased severity and duration of illness. Jama. 1996;275:1657–1660. [PubMed] [Google Scholar]
- Larson ET, Ojo KK, Murphy RC, Johnson SM, Zhang Z, Kim JE, Leibly DJ, Fox AM, Reid MC, Dale EJ, Perera BG, Kim J, Hewitt SN, Hol WG, Verlinde CL, Fan E, Van Voorhis WC, Maly DJ, Merritt EA. Multiple determinants for selective inhibition of apicomplexan calcium-dependent protein kinase CDPK1. J Med Chem. 2012;55:2803–2810. doi: 10.1021/jm201725v. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy MG, Ristic M. Babesia bovis: continuous cultivation in a microaerophilous stationary phase culture. Science. 1980;207:1218–1220. doi: 10.1126/science.7355284. [DOI] [PubMed] [Google Scholar]
- Lovett JL, Marchesini N, Moreno SN, Sibley LD. Toxoplasma gondii microneme secretion involves intracellular Ca(2+) release from inositol 1,4,5-triphosphate (IP(3))/ryanodine-sensitive stores. J Biol Chem. 2002;277:25870–25876. doi: 10.1074/jbc.M202553200. [DOI] [PubMed] [Google Scholar]
- Lovett JL, Sibley LD. Intracellular calcium stores in Toxoplasma gondii govern invasion of host cells. J Cell Sci. 2003;116:3009–3016. doi: 10.1242/jcs.00596. [DOI] [PubMed] [Google Scholar]
- Munkhjargal T, AbouLaila M, Terkawi MA, Sivakumar T, Ichikawa M, Davaasuren B, Nyamjargal T, Yokoyama N, Igarashi I. Inhibitory effects of pepstatin A and mefloquine on the growth of Babesia parasites. Am J Trop Med Hyg. 2012;87:681–688. doi: 10.4269/ajtmh.2012.12-0218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy RC, Ojo KK, Larson ET, Castellanos-Gonzalez A, Perera BG, Keyloun KR, Kim JE, Bhandari JG, Muller NR, Verlinde CL, White AC, Jr, Merritt EA, Van Voorhis WC, Maly DJ. Discovery of Potent and Selective Inhibitors of Calcium-Dependent Protein Kinase 1 (CDPK1) from C. parvum and T. gondii. ACS Med Chem Lett. 2010;1:331–335. doi: 10.1021/ml100096t. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojo KK, Larson ET, Keyloun KR, Castaneda LJ, Derocher AE, Inampudi KK, Kim JE, Arakaki TL, Murphy RC, Zhang L, Napuli AJ, Maly DJ, Verlinde CL, Buckner FS, Parsons M, Hol WG, Merritt EA, Van Voorhis WC. Toxoplasma gondii calcium-dependent protein kinase 1 is a target for selective kinase inhibitors. Nat Struct Mol Biol. 2010;17:602–607. doi: 10.1038/nsmb.1818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ojo KK, Pfander C, Mueller NR, Burstroem C, Larson ET, Bryan CM, Fox AM, Reid MC, Johnson SM, Murphy RC, Kennedy M, Mann H, Leibly DJ, Hewitt SN, Verlinde CL, Kappe S, Merritt EA, Maly DJ, Billker O, Van Voorhis WC. Transmission of malaria to mosquitoes blocked by bumped kinase inhibitors. J Clin Invest. 2012;122:2301–2305. doi: 10.1172/JCI61822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pedroni MJ, Sondgeroth KS, Gallego-Lopez GM, Echaide I, Lau AO. Comparative transcriptome analysis of geographically distinct virulent and attenuated Babesia bovis strains reveals similar gene expression changes through attenuation. BMC Genomics. 2013;14:763. doi: 10.1186/1471-2164-14-763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rashid HB, Chaudhry M, Rashid H, Pervez K, Khan MA, Mahmood AK. Comparative efficacy of diminazene diaceturate and diminazene aceturate for the treatment of babesiosis in horses. Trop Anim Health Prod. 2008;40:463–467. doi: 10.1007/s11250-007-9121-2. [DOI] [PubMed] [Google Scholar]
- Rodgers SE, Mather TN. Human Babesia microti incidence and Ixodes scapularis distribution, Rhode Island, 1998–2004. Emerg Infect Dis. 2007;13:633–635. doi: 10.3201/eid1304.061035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salama AA, Aboulaila M, Moussa AA, Nayel MA, El-Sify A, Terkawi MA, Hassan HY, Yokoyama N, Igarashi I. Evaluation of in vitro and in vivo inhibitory effects of fusidic acid on Babesia and Theileria parasites. Vet Parasitol. 2012;191:1–10. doi: 10.1016/j.vetpar.2012.08.022. [DOI] [PubMed] [Google Scholar]
- Senanayake SN, Paparini A, Latimer M, Andriolo K, Dasilva AJ, Wilson H, Xayavong MV, Collignon PJ, Jeans P, Irwin PJ. First report of human babesiosis in Australia. Med J Aust. 2012;196:350–352. doi: 10.5694/mja11.11378. [DOI] [PubMed] [Google Scholar]
- Ueno A, Terkawi MA, Yokoyama M, Cao S, Aboge G, Aboulaila M, Nishikawa Y, Xuan X, Yokoyama N, Igarashi I. Farsenyl pyrophosphate synthase is a potential molecular drug target of risedronate in Babesia bovis. Parasitol Int. 2013;62:189–192. doi: 10.1016/j.parint.2012.12.005. [DOI] [PubMed] [Google Scholar]
- Vannier E, Krause PJ. Update on babesiosis. Interdiscip Perspect Infect Dis. 2009;2009:984568. doi: 10.1155/2009/984568. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.