Abstract
AIM: To investigate the effects of gastric pacing on gastric emptying and plasma motilin level in a canine model of gastric motility disorders and the correlation between gastric emptying and plasma motilin level.
METHODS: Ten healthy Mongrel dogs were divided into: experimental group of six dogs and control group of four dogs. A model of gastric motility disorders was established in the experimental group undergone truncal vagotomy combined with injection of glucagon. Gastric half-emptying time (GEt1/2) was monitored with single photon emission computerized tomography (SPECT), and the half-solid test meal was labeled with an isotope 99mTc sulfur colloid. Plasma motilin concentration was measured with radioimmunoassay (RIA) kit. Surface gastric pacing at 1.1-1.2 times the intrinsic slow-wave frequency and a superimposed series of high frequency pulses (10-30 Hz) was performed for 45 min daily for a month in conscious dogs.
RESULTS: After surgery, GEt1/2 in dogs undergone truncal vagotomy was increased significantly from 56.35 ± 2.99 min to 79.42 ± 1.91 min (P < 0.001), but surface gastric pacing markedly accelerated gastric emptying and significantly decreased GEt1/2 to 64.94 ± 1.75 min (P < 0.001) in animals undergone vagotomy. There was a significant increase of plasma level of motilin at the phase of IMCIII (interdigestive myoelectrical complex, IMCIII) in the dogs undergone bilateral truncal vagotomy (baseline vs vagotomy, 184.29 ± 9.81 pg/ml vs 242.09 ± 17.22 pg/ml; P < 0.01). But plasma motilin concentration (212.55 ± 11.20 pg/ml; P < 0.02) was decreased significantly after a long-term treatment with gastric pacing. Before gastric pacing, GEt1/2 and plasma motilin concentration of the dogs undergone vagotomy showed a positive correlation (r = 0.867, P < 0.01), but after a long-term gastric pacing, GEt1/2 and motilin level showed a negative correlation (r = -0.733, P < 0.04).
CONCLUSION: Surface gastric pacing with optimal pacing parameters can improve gastric emptying parameters and significantly accelerate gastric emptying and can resume or alter motor function in a canine model of motility disorders. Gastric emptying is correlated well with plasma motilin level before and after pacing, which suggests that motilin can modulate the mechanism of gastric pacing by altering gastric motility.
INTRODUCTION
The incidence of gastric motility disorder has been increasing, but the mechanisms of the development of this disease remain obscure, and specific and effective therapy are lacking. The current treatment mainly relies on conventional prokinetic agents. However, some patients with motility disorder can not undergo a chronic treatment with prokinetic drugs because of their side effects[1-3]. In addition, tachyphylaxis may occur sooner or later and refractoriness to prokinetic agents is observed in a large number of patients. The limited efficacy of drugs in gastric motility disorder has led investigators to examine the effect of gastric pacing on gastric emptying. In the wake of cardiac pacing ,some investigators hope that gastric pacing would be a new way to treat refractory gastric motility disorder. The role of gastric pacing with application of implanted gastric serosa electrodes, suction intraluminal electrodes or gastric mucosal electrodes in gastrointestinal (GI) tract has been greatly discussed in western countries[4-23]. Most studies were restricted to animal experiments because of the invasiveness, substantial risk, and complications associated with the surgical procedure. Howerer, the study of surface gastric pacing was rarely reported in medical literature. To determine the therapeutic efficacy of surface gastric pacing, we combined bilateral truncal vagotomy with administration of glucagon to establish a canine model of gastric motility disorder. Then, we observed the effects of surface gastric pacing on gastric emptying and plasma motilin level before and after pacing, and also explored the possible mechanism of gastric pacing.
MATERIALS AND METHODS
Animals and surgical preparation
Ten healthy Mongrel dogs were divided into: experimental group of six dogs (four males and two females) 12.5-17.5 (average 14.5) kg and control group of four dogs (two males and two females) 13.0-17.0 (average 15.5) kg. Bilateral truncal vagotomy was performed in the experimental group under aseptic conditions with intravenous anaesthesia of pentobarbital sodium after an overnight fasting. The initial dosage of the anesthetics was 30 mg/kg,which was supplemented with 3 mg/kg if necessary on the basis of corneal reflex. Artificial ventilation was given during surgery. After surgery, the animal was first transferred to an intensive care cage for a few hours and then to the regular cage after a complete recovery from anesthesia. The protocal was approved by the Institution of Animal Care of the Third Military Medical University.
Experimental procedure
The dogs were allowed to recover completely from surgery for 2 to 3 wk before any study. The study was conducted in three sessions for each dog and experiments were performed on all the dogs in awake state. The dogs stood quietly on a table in a canvas support sling when gastric pacing was administered and gastric emptying was assessed. In the first session, we established the basic characteristics for gastric emptying in both control dogs and experimental dogs. In the second session, an intravenous injection of glucagon (500 μg/kg)[4,24] was administered. Gastric emptying of semi-solid food was monitored in all 10 dogs in the first 5 min after glucagon injection. Three days later, a second experiment with glucagon was performed, and we utilized gastic pacing to observe if the effects on drug could be altered, and gastric emptying was monitored for 2 h with gastric pacing. In the third session, gastric pacing was performed for 45 min after meal daily for a month in dogs of the experimental group to observe the chronic efficacy of a long-term treatment with gastric pacing, and gastric emptying was assessed for 2 h after cessation of gastric pacing.
Gastric pacing
In order to simulate the basic electrical rhythm (BER) and induce release of acetylcholine from intramural cholinergic fibers, gastric pacing stimulus consisted of two signals: a continuous similar-sine basic wave with a low-frequency (1.1 - 1.2 times the intrinsic slow-wave frequency) and a superimposed series of high frequency pulses (10-30 Hz), the number of a series of pluse was 15-40/cycle. The pacing signals from a pacemaker were delivered with a pair of bipolar skin electrodes. Stimulating electrodes were placed on commonly used positions of the abdominal region (localization through X ray). A long-term surface gastric pacing was performed for 45 min after meal daily for a month.
Measurement of gastric emptying
Gastric emptying was monitored with single photon emission computerized tomography (SPECT) (GE Inc.,USA). The semi-solid test meal consisted of 90 mg of commercial black sesame powder labeled with 74 MBq of 99mTc-sulfur colloid and 120 ml water. The study of gastric emptying consisted of two procedures, with or without gastric pacing, in a randomized order. On the day of gastric emptying test, the animal was fasted over 16 h. Immediately after the intake of isotope-labeled semi-solid meal, the maximum count rates were determined. Afer a first image was taken, the other sets were taken at an interval of 15 min for 2 h. The gastric region of interest was outlined by hand on the initial image by a nuclear medicine technologist who had no interest in the outcome of the study. To reduce the error resulted from the physical radioactive decay of isotope 99mTc, we corrected and normalized the percentage of semi-solid food retained in the stomach over time and chose the linear-fitting decay program to extrapolate GEt1/2.
Measurement of plasma concentration of motilin
Plasma level of motilin was determined with commercially available RIA (radioimmunoassay) kits. Blood was collected before and after vagotomy and after a long-term treatment of gastric pacing at the start of phase III of interdigestive myoelectric complex (IMCIII) respectively. Samples of peripheral venous blood were collected in lyophilized tubes containing 40 μl of 0.3 mol/L Na2-EDTA and 2000 u trasylol. Immediately, plasma was separated with centrifugation at 4000 r/min at 4 °C. The plasma should be frozen within 2 hours and then stored at -70 °C for batch assay with a well-established motilin immunoassay[20] (The motilin kit was supplied by the Radioimmunologic Center of PLA General Hospital, China).
Statistical analysis
Analysis of variance and paired Student’s t test and linear correlation analysis (SPSS 10.0 software) were performed to investigate the effects of gastric pacing on gastric emptying and plasma motilin level in dogs of the two groups. Statistical significance was assigned for P < 0.05. All data were presented as means ± SD.
RESULTS
Basic condition
All dogs generally tolerated well the surgical procedure and surface gastric pacing. Large doses of glucagon injected intravenously triggered hyperglycemia in the diabetic setting and plasma glucose levels were increased sharply from 81-114 mg/dl to 200-233 mg/dl. Only two of the ten dogs were sensitively induced by glucagon to show nausea and vomiting, but the adverse symptoms rapidly disappeared and did not lead to any complication. Long-term gastric pacing resulted in a significant weight gain. The average weight (11.3 ± 2.4 kg) after surgery was significantly lower than that before surgery (15.5 ± 3.2 kg, P < 0.01), but after a long-term treatment with gastric pacing weight gain was found to be 14.7 ± 2.7 kg (P < 0.05) in dogs of the experimental group.
Effects of vagotomy and glucagon on gastric emptying
Gastric emptying data were collected from each dog in all conditions.Results are summarized in Table 1 and Figure 1. The data before and after vagotomy were compared, and GEt1/2 of the dogs undergone truncal vagotomy was significantly increased from 56.35 ± 2.99 min to 79.42 ± 1.91 min (P < 0.001). To observe the effects of glucagon alone on gastric emptying, we compared the GEt1/2 of control dogs and the dogs undergone vagotomy before and after glucagon was administered (no gastric pacing was applied). In comparison with that before injection of glucagon, the GEt1/2 of the dogs undergone vagotomy was significantly longer after glucagon was administered (108.24 ± 10.75 min, P < 0.01). However, the delay of gastric emptying in control dogs after injection of glucagon was not statistically significant (57.73 ± 1.65 min vs 56.15 ± 2.34 min, P > 0.05).
Table 1.
Effects of gastric pacing on gastric emptying parameters
| Groups | n |
GEt1/2 (min) |
||
| No pacing | Acute pacing | Long-term pacing | ||
| Control | 4 | 56.15 ± 2.34 | 58.52 ± 4.77 | |
| Control + glucagon | 4 | 57.73 ± 1.65 | 56.90 ± 2.53 | |
| Vagotomy | 6 | 79.42 ± 1.91a | 74.41 ± 6.10 | 64.94 ± 1.75d |
| Vagotomy + glucagon | 6 | 108.24 ± 10.75b | 76.93 ± 8.55c | |
All the data were expressed as means ± SD.
P < 0.001 vs control,
P < 0.01 vs before injection of glucagon,
P < 0.05 vs before acute pacing,
P < 0.001 vs before a long-term pacing.
Figure 1.
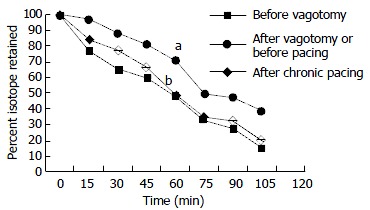
Effects of vagotomy and gastric pacing on gastric isotope retention during monitoring for 120 min. Data were expressed as means ± SD, n = 6. aP < 0.005 vs before vagotomy, bP < 0.005 vs before pacing (by analysis of variance at 120 min).
Effects of acute gastric pacing on gastric emptying
To investigate the acute effects of gastric pacing on gastric emptying in dogs undergone truncal vagotomy in combination with injection of glucagon and in control dogs with injection of glucagon, we observed gastric emptying for 2 h during gastric pacing. The GEt1/2 of the dogs undergone vagotomy and injection of glucagon was significantly shorter with pacing than that without pacing (108.24 ± 10.75 min vs 76.93 ± 8.55 min, P < 0.01). However, such an acute pacing had no significant effect on the GEt1/2 of control dogs with injection of glucagon (57.73 ± 1.65 min vs 56.90 ± 2.53 min, P = 0.4) or when only vagotomy was performed (79.42 ± 1.91 min vs 74.41 ± 6.10 min, P = 0.3).
Effects of chronic gastric pacing on gastric emptying
To investigate the effects of chronic gastric pacing on gastric emptying, we compared the GEt1/2 before pacing with that after a long-term gastric pacing of 30 d in the dogs undergone vagotomy. Gastric pacing significantly accelerated gastric emptying in animals undergone vagotomy (79.42 ± 1.91 min vs 64.94 ± 1.75 min, P < 0.001) and significantly decreased retention of gastric isotope (Figure 1).
Effects of gastric pacing on plasma gastric motilin
There was a significant increase of plasma motilin concentration in the phase of IMCIII after bilateral truncal vagotomy (baseline vs vagotomy, 242.09 ± 17.22 pg/ml vs 184.29 ± 9.81 pg/ml; P < 0.01). But plasma motilin concentration was significantly lower after a long-term surface gastric pacing than that before gastric pacing (212.55 ± 11.20 pg/ml; P < 0.02).
Correlation of plasma gastric motilin and gastric emptying
Statistic analysis indicated that GEt1/2 (79.42 ± 1.91 min) and plasma motilin concentration (242.09 ± 17.22 pg/ml) in the phase of IMCIII of the dogs undergone vagotomy had a positive correlation (r = 0.867, P < 0.01) before surface gastric pacing, that is, vagotomy evoked the increase of plasma motilin level which clearly disrupted gastric emptying. But after a long-term gastric pacing, GEt1/2 (64.94 ± 1.75 min) and plasma motilin level (212.55 ± 11.20 pg/ml) had a negative correlation (r = -0.733, P < 0.04), that is, plasma motilin release was inclined to restore its normal level, which might be helpful to improve gastric emptying.
DISCUSSION
We used bilateral truncal vagotomy in combination administration of glucagon to establish a canine model of gastric motility disorders, which can simulate some aspects of diabetic gastroparesis. Our study demonstrated that suface gastric pacing at 1.1-1.2 times the intrinsic slow-wave frequency superimposed series of high frequency pulses (10-30 Hz) was able to entrain propagated slow waves to replace the spontaneous ones and to improve gastric emptying in dogs after vagotomy and injection of glucagon. It is considered that gastric pacing could be proven to be an effective therapeutic approach for the treatment of gastric motility disorders[13,20-22,25-27].
Complete abdominal vagotomy has been shown to induce gastric stasis and delay emptying of solid food in both humans and animals[4,11]. It could change the contractile pattern during interdigestive migrating motor complex III (IMMCIII)[28].
Extrinsic denervation of the antrum could diminish the frequency of antral action potentials and retard gastric emptying of solids. Our study indicated that gastric emptying of semi-solid food was obviously impaired after vagotomy in six dogs and confirmed the finding of previous researchers[4,11] despite difference in their techniques of measuring gastric emptying. Previous studies have demonstrated that glucagon could delay gastric emptying of intact dogs[29], but our study did not find that glucagon was able to delay gastric emptying in the healthy dogs, suggesting that the dogs undergone vagotomy were more susceptible to gastric motility disorder. The mechanisms by which glucagon delayed gastric emptying in both humans and dogs have not been fully understood. This delay might associate with acute hyperglycemia-evoked gastric slow wave dysrhythmias by endogenous prostaglandins[24,30], such as tachygastria, tachyarrhythmia, bradyarrhythmia, asystolia (electrical silence), and gastric fibrillation which is a complete disorganization of gastric electrical activity due to impairment of coupling and propagation of gastric slow waves causing unpropagated antral contractions and exacerbating the hyperglycemia-evoked antral hypomotility[31-33]. In addition, hyperglycemia might have inhibitory effects on the spike potentials that could induce antral contractions or possible indirect effects of glucagon on gastric emptying through the release of hormones or neurotransmitters[4].
At present, controversial findings were reported about the effects of gastric pacing on gastric emptying. McCallum et al[13,20,22] showed that there was an increase in gastric emptying. On the other hand, Eagon et al[6] observed that neither the motility index nor gastric emptying rate was consistently changed by stimulation at any frequency. In our study, we employed a 1.1-1.2 times the intrinsic physiologic slow-wave frequency superimposed series of high frequency pulses (10-30 Hz) in surface gastric pacing which resulted in a significant acceleration of gastric emptying of semi-solid food in dogs undergone vagotomy but not in intact dogs. Our findings were consistent with those of Bellahsene et al[4] who employed a frequency similar to or slightly higher than the intrinsic slow-wave frequency.
Our study also evaluated the effects of total abdominal vagotomy and surface gastric pacing on plasma concentration of motilin in the phase of IMCIII. Motilin is a straight chain peptide containing 22 amino acids and has a molecular weight of approximately 2700. It has been found that plasma motilin levels varied in a cyclic fashion during the fasting state in accordance with the interdigestive migrating motor complex (IMMC), and IMMC III was concurrent with the peak of plasma motilin level[34]. Motilin has been known to induce IMMC III contractions through cholinergic neural pathways, 5-hydroxytryptamine (HT) receptors, and alpha receptors[35,36]. IMMC can clean large indigestible food residues, enhance antroduodenal and gallbladder coordination, stimulate pancreatic and bile secretion, and keep the small intestine free from stasis[37]. Our results showed that there was a significant difference in plasma motilin concentration in the phase of IMCIII before and after the operation and after gastric pacing. The significant increase of plasma motilin level in the dogs after truncal vagotomy might be explained that total abdominal vagotomy or a complete extrinsic vagal gastric denervation could markedly delay gastric empty and induce gastric stasis, and reduce the sensitivity of motilin receptors, which might feedbackly lead to the release of plasma motilin. After a long-term surface gastric pacing, the delay of gastric emptying was improved and plasma motilin level gradually restored normal. At prsent, its mechanism is not clearly determined. In addition, our study showed gastric emptying was correlated well with plasma motilin level in the phase of IMCIII before and after gastric pacing, which implies that there is a possibility for motilin to mediate the process of gastric pacing by altering gastric motility.
It has been found that gastrointestinal motility is regulated by a spatio-temporally-coordinated electrical pattern called gastrointestinal myoelectrical activity (GMA) that is paced by the GI pacemaker[38]. Recently, it has been recognized that interstitial cells of Cajal known as pacemaker cells could generate rhythmically electrical pacemaker activities (or electrical slow waves) which regulate the frequency of contractions of the tunica muscularis for GI motility[39-44]. The normal patterns of GMA present temporal evolution from endogenous rhythmic oscillation to bursting of spikes associated with contractions, and also order spatial propagation of the oscillating waves. Spatio-temporal modeling of the GMA has been established, thanks to the advances in electronic technology. Therefore, we employed a continuous similar-sine basic wave with a low-frequency (10% - 20% high than intrinsic gastric frequency) superimposed a series of high frequency pulses (10-30 Hz) as signals of surface gastric electrical pacing. In contrast to gastric pacing through implanted serosal electrodes and the suction intraluminal electrodes in gastrointestinal tract, surface gastric pacing is more simple and noninvasive. In addition, surface gastric pacing does not require any surgical procedures such as laparotomy or laparoscopy. So this technique is a very attractive, reliable and acceptable candidate method to treat gastric motility disorders.
Although gastric pacing could restore a normal pattern of slow wave and an efficient gastric motor function and improve gastric emptying, its mechanism remains unclear[45]. Recently, Qian et al[15] reported gastric pacing at a frequency similar to or just above that of the native slow wave to induce gastric slow wave was not via the cholinergic nerves, because although atropine was used to block the vagal activity, gastric pacing could still entrain and normalize the irregular rhythm of gastric slow wave. Moreover, spectral analysis of heart rate indicated that gastric pacing had no effect on the extrinsic autonomic functions. Accordingly, we presume that the mechanism of gastric pacing at a frequency slightly higher than the intrinsic frequency to induce gastric slow wave may be via some other neurohumoral pathways or directly leads to local activation of interstitial cells of Cajal and/or smooth muscle cells. Gastric electrical stimulation with a frequency much higher (about 10-1200 cycles/min) than that of the intrinsic slow wave probably could act on sensory fibers directed to the central nervous system[46]. Gastric electrical stimulation with a frequency 10-100 Hz might be possibly associated with autonomic and enteric nervous system changes, likely induce the release of acetylcholine from the intramural cholinergic fibers, which in turn stimulated the muscular cell contraction, because its effect was prevented by a previous administration of atropine[47-49]. In addition, gastrointestinal hormones such as motilin or NANC neurotransmitter VIP might also mediate the process of gastric pacing[47,50].
Footnotes
Edited by Wang XL
References
- 1.Wang SH, Lin CY, Huang TY, Wu WS, Chen CC, Tsai SH. QT interval effects of cisapride in the clinical setting. Int J Cardiol. 2001;80:179–183. doi: 10.1016/s0167-5273(01)00485-5. [DOI] [PubMed] [Google Scholar]
- 2.Kyrmizakis DE, Chimona TS, Kanoupakis EM, Papadakis CE, Velegrakis GA, Helidonis ES. QT prolongation and torsades de pointes associated with concurrent use of cisapride and erythromycin. Am J Otolaryngol. 2002;23:303–307. doi: 10.1053/ajot.2002.124543. [DOI] [PubMed] [Google Scholar]
- 3.Smalley W, Shatin D, Wysowski DK, Gurwitz J, Andrade SE, Goodman M, Chan KA, Platt R, Schech SD, Ray WA. Contraindicated use of cisapride: impact of food and drug administration regulatory action. JAMA. 2000;284:3036–3039. doi: 10.1001/jama.284.23.3036. [DOI] [PubMed] [Google Scholar]
- 4.Bellahsène BE, Lind CD, Schirmer BD, Updike OL, McCallum RW. Acceleration of gastric emptying with electrical stimulation in a canine model of gastroparesis. Am J Physiol. 1992;262:G826–G834. doi: 10.1152/ajpgi.1992.262.5.G826. [DOI] [PubMed] [Google Scholar]
- 5.Eagon JC, Kelly KA. Effects of gastric pacing on canine gastric motility and emptying. Am J Physiol. 1993;265:G767–G774. doi: 10.1152/ajpgi.1993.265.4.G767. [DOI] [PubMed] [Google Scholar]
- 6.Eagon JC, Kelly KA. Effect of electrical stimulation on gastric electrical activity, motility and emptying. Neurogastroenterol Motil. 1995;7:39–45. doi: 10.1111/j.1365-2982.1995.tb00207.x. [DOI] [PubMed] [Google Scholar]
- 7.Familoni BO, Abell TL, Voeller G, Salem A, Gaber O. Electrical stimulation at a frequency higher than basal rate in human stomach. Dig Dis Sci. 1997;42:885–891. doi: 10.1023/a:1018852011857. [DOI] [PubMed] [Google Scholar]
- 8.Familoni BO, Abell TL, Nemoto D, Voeller G, Johnson B. Efficacy of electrical stimulation at frequencies higher than basal rate in canine stomach. Dig Dis Sci. 1997;42:892–897. doi: 10.1023/a:1018804128695. [DOI] [PubMed] [Google Scholar]
- 9.Sobocki J, Thor PJ, Popiela T, Wasowicz P, Herman RM. [Stomach electrostimulation--new possibility for treating gastroparesis] Folia Med Cracov. 1999;40:63–75. [PubMed] [Google Scholar]
- 10.Mintchev M, Bowes K. Computer model of gastric electrical stimulation. Ann Biomed Eng. 1997;25:726–730. doi: 10.1007/BF02684849. [DOI] [PubMed] [Google Scholar]
- 11.Yokota M, Ando N, Ozawa S, Imazu Y, Kitajima M. Enhanced motility of the vagotomized canine stomach by electrical stimulation. J Gastroenterol Hepatol. 1997;12:338–346. doi: 10.1111/j.1440-1746.1997.tb00432.x. [DOI] [PubMed] [Google Scholar]
- 12.Lin ZY, McCallum RW, Schirmer BD, Chen JD. Effects of pacing parameters on entrainment of gastric slow waves in patients with gastroparesis. Am J Physiol. 1998;274:G186–G191. doi: 10.1152/ajpgi.1998.274.1.G186. [DOI] [PubMed] [Google Scholar]
- 13.McCallum RW, Chen JD, Lin Z, Schirmer BD, Williams RD, Ross RA. Gastric pacing improves emptying and symptoms in patients with gastroparesis. Gastroenterology. 1998;114:456–461. doi: 10.1016/s0016-5085(98)70528-1. [DOI] [PubMed] [Google Scholar]
- 14.Mintchev MP, Sanmiguel CP, Otto SJ, Bowes KL. Microprocessor controlled movement of liquid gastric content using sequential neural electrical stimulation. Gut. 1998;43:607–611. doi: 10.1136/gut.43.5.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Qian L, Lin X, Chen JD. Normalization of atropine-induced postprandial dysrhythmias with gastric pacing. Am J Physiol. 1999;276:G387–G392. doi: 10.1152/ajpgi.1999.276.2.G387. [DOI] [PubMed] [Google Scholar]
- 16.Xing J, Brody F, Rosen M, Chen JD, Soffer E. The effect of gastric electrical stimulation on canine gastric slow waves. Am J Physiol Gastrointest Liver Physiol. 2003;284:G956–G962. doi: 10.1152/ajpgi.00477.2002. [DOI] [PubMed] [Google Scholar]
- 17.Mintchev MP, Sanmiguel CP, Amaris M, Bowes KL. Microprocessor-controlled movement of solid gastric content using sequential neural electrical stimulation. Gastroenterology. 2000;118:258–263. doi: 10.1016/s0016-5085(00)70207-1. [DOI] [PubMed] [Google Scholar]
- 18.Lin Z, Forster J, Sarosiek I, McCallum RW. Treatment of gastroparesis with electrical stimulation. Dig Dis Sci. 2003;48:837–848. doi: 10.1023/a:1023099206939. [DOI] [PubMed] [Google Scholar]
- 19.Lin X, Peters LJ, Hayes J, Chen JD. Entrainment of segmental small intestinal slow waves with electrical stimulation in dogs. Dig Dis Sci. 2000;45:652–656. doi: 10.1023/a:1005466904380. [DOI] [PubMed] [Google Scholar]
- 20.Forster J, Sarosiek I, Delcore R, Lin Z, Raju GS, McCallum RW. Gastric pacing is a new surgical treatment for gastroparesis. Am J Surg. 2001;182:676–681. doi: 10.1016/s0002-9610(01)00802-9. [DOI] [PubMed] [Google Scholar]
- 21.Chang CS, Chou JW, Ko CW, Wu CY, Chen GH. Cutaneous electrical stimulation of acupuncture points may enhance gastric myoelectrical regularity. Digestion. 2002;66:106–111. doi: 10.1159/000065596. [DOI] [PubMed] [Google Scholar]
- 22.Abell TL, Van Cutsem E, Abrahamsson H, Huizinga JD, Konturek JW, Galmiche JP, VoelIer G, Filez L, Everts B, Waterfall WE, et al. Gastric electrical stimulation in intractable symptomatic gastroparesis. Digestion. 2002;66:204–212. doi: 10.1159/000068359. [DOI] [PubMed] [Google Scholar]
- 23.Chen JD, Qian L, Ouyang H, Yin J. Gastric electrical stimulation with short pulses reduces vomiting but not dysrhythmias in dogs. Gastroenterology. 2003;124:401–409. doi: 10.1053/gast.2003.50048. [DOI] [PubMed] [Google Scholar]
- 24.Hasler WL, Soudah HC, Dulai G, Owyang C. Mediation of hyperglycemia-evoked gastric slow-wave dysrhythmias by endogenous prostaglandins. Gastroenterology. 1995;108:727–736. doi: 10.1016/0016-5085(95)90445-x. [DOI] [PubMed] [Google Scholar]
- 25.Abell TL, Minocha A. Gastroparesis and the gastric pacemaker: a revolutionary treatment for an old disease. J Miss State Med Assoc. 2002;43:369–375. [PubMed] [Google Scholar]
- 26.Shafik A, El-Sibai O, Shafik AA, Ahmed I. The motor efficacy of the artificial colonic pacemaker in colonic inertia patients. Front Biosci. 2002;7:b6–13. doi: 10.2741/a754. [DOI] [PubMed] [Google Scholar]
- 27.Chen JD, Lin HC. Electrical pacing accelerates intestinal transit slowed by fat-induced ileal brake. Dig Dis Sci. 2003;48:251–256. doi: 10.1023/a:1021911023155. [DOI] [PubMed] [Google Scholar]
- 28.Tanaka T, Kendrick ML, Zyromski NJ, Meile T, Sarr MG. Vagal innervation modulates motor pattern but not initiation of canine gastric migrating motor complex. Am J Physiol Gastrointest Liver Physiol. 2001;281:G283–G292. doi: 10.1152/ajpgi.2001.281.1.G283. [DOI] [PubMed] [Google Scholar]
- 29.Chernish SM, Brunelle RR, Rosenak BD, Ahmadzai S. Comparison of the effects of glucagon and atropine sulfate on gastric emptying. Am J Gastroenterol. 1978;70:581–586. [PubMed] [Google Scholar]
- 30.Kim TW, Beckett EA, Hanna R, Koh SD, Ordög T, Ward SM, Sanders KM. Regulation of pacemaker frequency in the murine gastric antrum. J Physiol. 2002;538:145–157. doi: 10.1113/jphysiol.2001.012765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Petrakis IE, Kogerakis N, Vrachassotakis N, Stiakakis I, Zacharioudakis G, Chalkiadakis G. Hyperglycemia attenuates erythromycin-induced acceleration of solid-phase gastric emptying in healthy subjects. Abdom Imaging. 2002;27:309–314. doi: 10.1007/s00261-001-0058-z. [DOI] [PubMed] [Google Scholar]
- 32.Schvarcz E, Palmér M, Aman J, Horowitz M, Stridsberg M, Berne C. Physiological hyperglycemia slows gastric emptying in normal subjects and patients with insulin-dependent diabetes mellitus. Gastroenterology. 1997;113:60–66. doi: 10.1016/s0016-5085(97)70080-5. [DOI] [PubMed] [Google Scholar]
- 33.Petrakis IE, Kogerakis N, Prokopakis G, Zacharioudakis G, Antonakakis S, Vrachassotakis N, Chalkiadakis G. Hyperglycemia attenuates erythromycin-induced acceleration of liquid-phase gastric emptying of hypertonic liquids in healthy subjects. Dig Dis Sci. 2002;47:67–72. doi: 10.1023/a:1013211419605. [DOI] [PubMed] [Google Scholar]
- 34.Tanaka T, VanKlompenberg LH, Sarr MG. Selective role of vagal and nonvagal innervation in initiation and coordination of gastric and small bowel patterns of interdigestive and postprandial motility. J Gastrointest Surg. 2001;5:418–433. doi: 10.1016/s1091-255x(01)80072-x. [DOI] [PubMed] [Google Scholar]
- 35.Itoh Z, Mizumoto A, Iwanaga Y, Yoshida N, Torii K, Wakabayashi K. Involvement of 5-hydroxytryptamine 3 receptors in regulation of interdigestive gastric contractions by motilin in the dog. Gastroenterology. 1991;100:901–908. doi: 10.1016/0016-5085(91)90262-j. [DOI] [PubMed] [Google Scholar]
- 36.Haga N, Mizumoto A, Satoh M, Mochiki E, Mizusawa F, Ohshima K, Itoh Z. Role of endogenous 5-hydroxytryptamine in the regulation of gastric contractions by motilin in dogs. Am J Physiol. 1996;270:G20–G28. doi: 10.1152/ajpgi.1996.270.1.G20. [DOI] [PubMed] [Google Scholar]
- 37.Testoni PA, Bagnolo F, Masci E, Colombo E, Tittobello A. Different interdigestive antroduodenal motility patterns in chronic antral gastritis with and without Helicobacter pylori infection. Dig Dis Sci. 1993;38:2255–2261. doi: 10.1007/BF01299905. [DOI] [PubMed] [Google Scholar]
- 38.Wang ZS, Cheung JY, Gao SK, Chen JD. Spatio-temporal nonlinear modeling of gastric myoelectrical activity. Methods Inf Med. 2000;39:186–190. [PubMed] [Google Scholar]
- 39.Sanders KM. A case for interstitial cells of Cajal as pacemakers and mediators of neurotransmission in the gastrointestinal tract. Gastroenterology. 1996;111:492–515. doi: 10.1053/gast.1996.v111.pm8690216. [DOI] [PubMed] [Google Scholar]
- 40.Der-Silaphet T, Malysz J, Hagel S, Larry Arsenault A, Huizinga JD. Interstitial cells of cajal direct normal propulsive contractile activity in the mouse small intestine. Gastroenterology. 1998;114:724–736. doi: 10.1016/s0016-5085(98)70586-4. [DOI] [PubMed] [Google Scholar]
- 41.Hanani M, Freund HR. Interstitial cells of Cajal--their role in pacing and signal transmission in the digestive system. Acta Physiol Scand. 2000;170:177–190. doi: 10.1046/j.1365-201x.2000.00769.x. [DOI] [PubMed] [Google Scholar]
- 42.Horiguchi K, Semple GS, Sanders KM, Ward SM. Distribution of pacemaker function through the tunica muscularis of the canine gastric antrum. J Physiol. 2001;537:237–250. doi: 10.1111/j.1469-7793.2001.0237k.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ward SM, Sanders KM. Physiology and pathophysiology of the interstitial cell of Cajal: from bench to bedside. I. Functional development and plasticity of interstitial cells of Cajal networks. Am J Physiol Gastrointest Liver Physiol. 2001;281:G602–G611. doi: 10.1152/ajpgi.2001.281.3.G602. [DOI] [PubMed] [Google Scholar]
- 44.Takayama I, Horiguchi K, Daigo Y, Mine T, Fujino MA, Ohno S. The interstitial cells of Cajal and a gastroenteric pacemaker system. Arch Histol Cytol. 2002;65:1–26. doi: 10.1679/aohc.65.1. [DOI] [PubMed] [Google Scholar]
- 45.Bortolotti M. The "electrical way" to cure gastroparesis. Am J Gastroenterol. 2002;97:1874–1883. doi: 10.1111/j.1572-0241.2002.05898.x. [DOI] [PubMed] [Google Scholar]
- 46.Chou JW, Chang YH, Chang CS, Chen GH. The effect of different frequency electrical acu-stimulation on gastric myoelectrical activity in healthy subjects. Hepatogastroenterology. 2003;50:582–586. [PubMed] [Google Scholar]
- 47.Furgała A, Thor PJ, Kolasińska-Kloch W, Krygowska-Wajs A, Kopp B, Laskiewicz J. The effect of transcutaneous nerve stimulation (TENS) on gastric electrical activity. J Physiol Pharmacol. 2001;52:603–610. [PubMed] [Google Scholar]
- 48.Amaris MA, Rashev PZ, Mintchev MP, Bowes KL. Microprocessor controlled movement of solid colonic content using sequential neural electrical stimulation. Gut. 2002;50:475–479. doi: 10.1136/gut.50.4.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lin J, Cai G, Xu JY. A comparison between Zhishi Xiaopiwan and cisapride in treatment of functional dyspepsia. World J Gastroenterol. 1998;4:544–547. doi: 10.3748/wjg.v4.i6.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Li W, Zheng TZ, Qu SY. Effect of cholecystokinin and secretin on contractile activity of isolated gastric muscle strips in guinea pigs. World J Gastroenterol. 2000;6:93–95. doi: 10.3748/wjg.v6.i1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]


