Abstract
The purpose of this study was to determine the relative importance of blood vessels (hemangiogenesis) versus lymphatic vessels (lymphangiogenesis) in mediating immunological responses after transplantation. Using the murine model of corneal transplantation, graft survival was compared in differentially prevascularized and avascular recipient beds. Donor corneas (C57BL/6) were transplanted into uninflamed or inflamed avascular, prehemvascularized only or prehemvascularized and prelymphvascularized recipient murine eyes (BALB/C). Selective inhibition of lymphangiogenesis was achieved using antivascular endothelial growth factor receptor 3 Abs and anti-integrin α5 small molecules. Grafts placed into only prehemvascularized recipient beds had a similarly good graft survival compared with grafts placed into completely avascular, normal recipients, whereas the pre-existence of lymphatic vessels significantly deteriorated corneal graft survival (p < 0.05). Lymphatic vessels seem to contribute significantly to graft rejection after (corneal) transplantation. That may allow for selective, temporary, perioperative antilymphangiogenic treatment to promote graft survival without affecting blood vessels, even after solid organ transplantation.
Immune-mediated graft rejections remain the most common cause for graft failure after organ and tissue transplantation. A great medical need exists for pharmacologic strategies to promote graft survival without unduly compromising the health of the recipient (for review see Ref. 1).
The three structural components of the immune system allowing for immune responses against foreign tissue after transplantation are afferent lymphatic vessels (“afferent arm of the immune reflex arc”), regional lymph nodes (“central processing unit”), and efferent blood vessels (“efferent arm of the immune reflex arc”) (2).
Lymphatic vessels allow the transport of APCs with foreign tissue Ags and soluble antigenic material to the regional lymph node and, thereby, constitute one of the earliest events in the immune-cascade leading to rejection. The precise relative importance of lymphatic vessels (“afferent arm”) versus blood vessels (“efferent arm”) for immune reactions after transplantation is unclear. However, every solid organ or vascularized tissue transplantation is accompanied by hemangiogenesis and lymphangiogenesis across the wound edges. In fact, lymphatic vessels have been identified in allogenic grafts after heart and kidney transplantation, where their presence seems to be related to graft rejection (3–7).
In this study, we try to unravel the relative importance of pre-existing hemangiogenesis versus lymphangiogenesis for immune responses after transplantation. To do that, we used the murine model of corneal transplantation. Corneal transplantation (also called keratoplasty) is the most frequently performed tissue transplantation, with >40,000 surgeries per year in the United States. In addition, corneal transplantation can experimentally serve as a model for allogenic transplantation, which allows for the analysis of the impact of lymphatic and blood vessels on the graft outcome, because of the cornea’s normal avascularity. Corneal hemangiogenesis and lymphangiogenesis occurring before as well as after corneal transplantation significantly increase the risk for immune rejection (8). The rate of immune rejections in patient eyes with avascular graft beds is ~10%, whereas the rate in prevascularized, so-called “high-risk” patient eyes increases to 50–100% (9). Lymphatic vessels and blood vessels override the so-called “immune privilege” of the normally avascular cornea.
It was shown that a combined modulation of hemangiogenesis and lymphangiogenesis by vascular endothelial growth factor (VEGF)-TrapR1R2 after normal-risk corneal transplantation improved graft survival in the murine model of corneal transplantation (8). Blocking lymphangiogenesis preferentially over hemangiogenesis may lead to inhibition of the induction of an immune response and, at the same time, blood vessels could still support the graft with nutrients and enable wound healing (essential in solid organ transplants).
Therefore, it is important to identify ways to preferentially block lymphangiogenesis to promote graft survival. Until very recently, specific inhibition of lymphangiogenesis was not possible. We and other investigators identified ways to preferentially inhibit lymphangiogenesis over hemangiogenesis by a dose-dependent, systemic integrin α5β1 blockade with small molecule inhibitors (JSM6427) (10, 11). We found that preferential inhibition of lymphangiogenesis is possible by integrin α5β1 inhibition using an intermediate dose (10). In addition, we recently found a preferential inhibitory effect of the anti-VEGF receptor (VEGFR)3 Ab mF4-31C1 on corneal inflammatory lymphangiogenesis in the murine model of suture-induced neovascularization (12).
We used these two novel pharmacologic functional assays to create new models of differentially vascularized allogenic transplantation in corneal host beds. These contained only blood vessels (“alymphatic”), blood and lymphatic vessels (“high-risk”), or no vessels (“normal-risk”) prior to transplantation. The purpose of this study was to determine whether the high-risk status of corneal allografts in vascularized host beds is defined by the lymphatic or blood vessels and whether preferential inhibition of lymphangiogenesis prior to transplantation is able to improve graft survival by interfering with sensitization and immune rejection.
Materials and Methods
Mice and anesthesia
Six- to 8-wk-old female C57BL/6 mice were used as graft donors; aged-matched female BALB/C mice (Charles River Germany, Sulzfeld, Germany) were used as recipients. All animals were treated in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research. For surgical procedures, mice were anesthetized using a mixture of KetanestS (8 mg/kg) and Rompun (0.1 ml/kg).
Suture-induced corneal neovascularization assay
We used the mouse model of suture-induced inflammatory corneal neovascularization as previously described (13). Briefly, mice were put under general anesthesia, and three intrastromal 11-0 nylon sutures (70-μm-diameter needles; Serag-Wiesner, Naila, Germany) were placed in the corneal stroma, with two incursions extending over 120° of corneal circumference each. Corneal sutures were removed after 14 d. This creates an inflamed, hemvascularized, and lymphvascularized recipient bed (“high-risk”).
Different treatments during the vascularization phase prior to transplantation
To generate differently vascularized and avascular recipient beds, the following treatments were performed during the suture-induced corneal neovascularization assay (Fig. 1). VEGF-TrapR1R2 (25 mg/kg; n = 10; developed and provided by Regeneron Pharmaceuticals), VEGFR3 Ab mF4-31C (25 mg/kg; n = 10; developed and provided by ImClone Systems, Tarrytown, NY), or saline solution as control (n = 10) was applied by i.p. injections on the day of surgery and 3 and 7 d later. Integrin inhibitor JSM6427: mice in this treatment group (n = 23) received integrin α5β1 inhibiting molecules [JSM6427; developed and provided by Jerini AG (14)] or carrier solution of JSM6427 as control (n = 10) systemically via s.c. osmotic pumps (Alzet pumps, Cupertino, CA), as described previously (10), for 14 d after suture placement prior to corneal transplantation.
FIGURE 1.
Generation of different transplantation models. Schematic diagram showing the generation of normal-risk (avascular), high-risk (inflamed and hemvascularized and lymphvascularized), avascular high-risk (inflamed, but avascular, recipients), and alymphatic high-risk recipient beds (inflamed and hemvascularized, but no lymphatic vessels) as transplantation models.
Corneal allogenic transplantation in mice
After generating different corneal recipient beds, corneal transplantation was performed as previously described (15). Briefly, a central 2-mm circular button of the donor cornea of C57BL/6 mice was excised with curved Vannas scissors. The graft beds were prepared in the recipient eye (right eye) of BALB/C mice by cutting out a circular 1.8-mm area in the central cornea and discarding it. The donor cornea was placed in this central cavity of the recipient corneal tissue. The transplant was secured in place with eight equidistant single sutures (11-0 nylon) along the graft–host interface. Eyelids were sutured with a 7-0 suture to avoid graft injury. Eyelid sutures were removed 3 d later, and corneal sutures were removed after 7 d. The opacity of the grafts was evaluated and scored as described previously (16) from 0 (no opacity; pupil margin and iris vessels clearly visible through the cornea) to 5 (maximum [corneal] stromal opacity; anterior chamber not visible). Grafts with a score worse than 2 (minimal deep stromal opacity, pupil margin and iris vessels visible) were considered rejected.
Immunohistochemistry in corneal wholemounts for prevascularization status
The corneas were excised after 14 d, rinsed in PBS, and stained as described previously (17). Briefly, after acetone fixation, we used a rabbit anti-mouse LYVE-1 (1:500; a kind gift of D.G. Jackson, Oxford University, Oxford, U.K.) (8, 17) and a goat anti-rabbit Cy3-conjugated secondary Ab for the lymphatic vessels. Blood vessels were stained with an FITC-conjugated rat anti-CD-31 Ab (Acris Antibodies, Hiddenhausen, Germany). Isotype control was assured with an FITC-conjugated normal rat IgG2A for CD31-FITC and a normal rabbit IgG for LYVE-1 (both from Santa Cruz Biotechnology, Santa Cruz, CA).
Functional and statistical analysis
Postoperative survival of the corneal allografts was analyzed using Kaplan–Meier survival curves. Subsequent statistical analysis was done using InStat 3, version 3.06 (GraphPad, San Diego, CA). Graphs were drawn using Prism4, version 4.03 (GraphPad).
Results
Presence of pathologic blood and lymphatic vessels in corneal recipient beds prior to transplantation significantly increases the rate of subsequent graft rejections
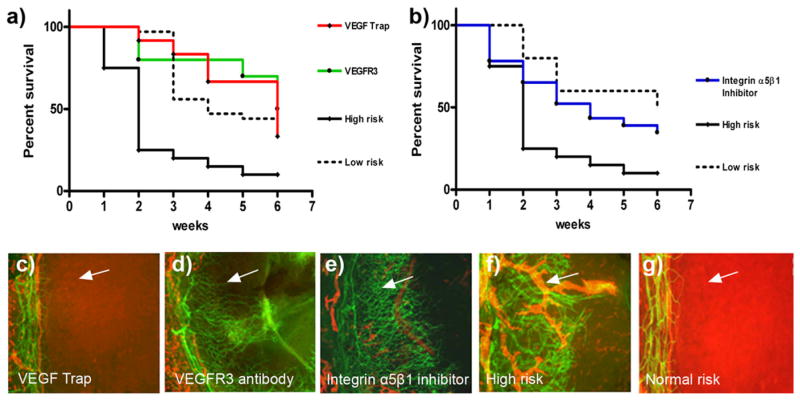
To determine the effect of the blood and lymphatic vessels themselves versus the effect of the associated inflammation on graft survival, we compared graft survival between so-called “high-risk” hemvascularized and lymphvascularized corneas and avascular, but inflamed, ones (Figs. 1 and 2). To achieve this reduced inflammatory, but avascular, status in the suture-induced corneal neovascularization model, hemangiogenesis and lymphangiogenesis were inhibited by a VEGF cytokine Trap (VEGF-TrapR1R2; Fig. 2) during the vascularization phase prior to corneal transplantation. The results showed that grafts placed into avascular, but low-inflamed, recipient beds (after VEGF-TrapR1R2 treatment) exhibited survival rates comparable to the standard survival of “so-called” normal-risk grafting (without previous inflammatory stimuli; Fig. 2), although the treated corneas undergo inflammatory stimuli, while, at the same time, hemangiogenesis and lymphangiogenesis are inhibited. In contrast, the placement of grafts into pathologically hemvascularized and lymphvascularized and inflamed hosts significantly worsens graft survival (p<0.0001; n = 10; Fig. 2a).
FIGURE 2.
Lymphatic vessels in the recipient bed prior to transplantation determine graft survival. In the 2 wk prior to transplantation (when corneal suture placement was used to induce pathologic corneal neovascularization in the recipient bed), mice were treated with VEGF-TrapR1R2 (a [red line] and c; resulting in no blood or lymphatic vessels, but reduced inflammation in the recipient bed at the time of transplantation), the VEGFR3 Ab mF4-31C1 (a [green line] and d; resulting in no lymphatic vessels, but only blood vessels present in the recipient bed at the time of transplantation), or the JSM6427 integrin α5β1 inhibitor (b [blue line] and e; resulting in no lymphatic vessels, but only blood vessels, present in the recipient bed at the time of transplantation). Graft survival was compared with prehemvascularized and prelymphvascularized controls (a and b [black line], f: “high-risk” recipient bed) and avascular recipient controls (a and b [dotted line], g: “low-risk” recipient bed). The graft survival was significantly better when transplants were placed into recipient beds lacking lymphatic vessels compared with beds with lymphatic vessels present at the time of transplantation (VEGF-Trap versus high-risk: p < 0.0001; VEGFR3 versus high-risk: p < 0.0002; n = 10; JSM6427 versus high-risk: p < 0.032, n = 23; Kaplan–Meyer survival curve). (c–g) Representative images of recipient corneal beds at the time of transplantation after corneas were treated with VEGF-TrapR1R2 (c), mF4-31C1 (VEGFR3 Ab) (d ), JSM6427 (e), or untreated high-risk (f ) and normal-risk (g) recipient beds (original magnification ×100). Green, blood vessels; red, lymphatic vessels; arrow, prevascularized cornea.
Absence of lymphatic vessels in the recipient bed prior to transplantation significantly promotes subsequent graft survival: the VEGFR3 Ab mF4-31C1
To assess the role of lymphatic vessels in the survival prognosis after corneal grafting, we used the anti-VEGFR3 Ab mF4-31C1 in the mouse model of inflammatory, suture-induced corneal neovascularization. The outgrowth of lymphatic vessels was preferentially inhibited by the VEGFR3 Ab mF4-31C1 treatment (12). The survival of grafts placed into these alymphatic, but hemvascularized, recipient beds was significantly better compared with the simultaneously hemvascularized and lymphvascularized recipient beds (high-risk eyes) by specific inhibition of lymphangiogenesis during the inflammatory vascularization phase prior to corneal transplantation (p < 0.0002; n = 10; Fig. 2a).
Absence of lymphatic vessels in the recipient bed prior to transplantation significantly promotes subsequent graft survival: integrin inhibitor JSM6427 assay
We analyzed the impact of pre-existing lymphatic versus blood vessels as mediators of immune rejection after corneal transplantation by using a second possibility of selective inhibition of lymphangiogenesis in the vascularization period prior to high-risk corneal transplantation. We inhibited integrin α5β1 function by small molecule antagonists JSM6427. Using an intermediate dose, this treatment led to a selective and specific inhibition of lymphangiogenesis, whereas hemangiogenesis was not affected significantly, as shown before (10). Systemic integrin α5β1 inhibition and, thereby, the absence of lymphatic vessels in the recipient bed, significantly improved corneal graft survival after high-risk corneal transplantation (p = 0.031; n = 23; Fig. 2b) compared with control and reduced the risk for rejection to the level of low-risk corneal transplantation.
Discussion
Lymphatic vessels are thought to be important mediators of immune processes after organ and tissue transplantation. They act as a conduit for foreign antigenic material and APCs, such as dendritic cells. The relative importance of lymphatic vessels as the “afferent arm” of an immune reflex arc versus blood vessels as the “efferent arc” of an immune reflex arc has not been studied, primarily because, until very recently, it was not possible to molecularly dissect and separate these two closely intermingled vascular pathways. We and other investigators recently described novel approaches to differentially inhibit lymphangiogenesis during the process of inflammatory neovascularization: on the one hand by blocking the main lymphatic VEGF receptor (VEGFR3) (12) with a specific VEGFR3 Ab (12, 18, 19) and, on the other hand, by using small molecules against a lymphatic endothelial integrin (integrin α5β1) (10) to specifically inhibit lymphangiogenesis. This allowed us, using the murine model of corneal transplantation into a normally avascular recipient bed, to compare graft survival rates among grafts placed into avascular, prehemvascularized and lymphvascularized, only prehemvascularized, and low-inflamed, but avascular, recipient beds.
Our findings, obtained by specific and selective preoperative inhibition of lymphangiogenesis, suggest that corneal lymphatic vessels promoting sensitization are the most important mediator of immune rejections after corneal transplantation. In two different approaches of vascularized high-risk recipient beds without lymphatic vessels, graft survival rates equaled those of avascular recipient beds. In contrast, the survival was significantly worse when lymphatic vessels and blood vessels were present.
Pre-existing corneal blood and lymphatic vessels are an established risk factor for the immune rejection after corneal transplantation (16, 20). Corneal allograft survival in the avascular normal-risk mouse model (C57BL/6 to BALB/C) was reduced from ~50% after 6 wk to nearly 0% after 2 wk, if the recipient bed was prevascularized (13, 15) with blood and lymphatic vessels. This enables immediate access of donor tissue to draining host lymphatic vessels after high-risk grafting and its exposure to efferent host blood vessels. Although grafts placed into high-risk eyes induce donor-specific sensitization promptly (within 7 d) (15), allografts placed into avascular low-risk eyes do not generate sensitization until 2–4 wk after grafting (21). Once the drainage system is established, graft-derived Ags reach the local lymph node and activate donor-specific alloreactive T cells, which can cause rejection. Blood vessels reaching the graft are essential for delivery of APCs and alloreactive T lymphocytes to the graft. If sensitized T cells do not occur until 14–21 d, they must compete with the regulatory T cells of anterior chamber-associated immune deviation, which begin to emerge at that time (22). Retarding lymphangiogenesis in the graft bed might narrow the window of opportunity during which recipient sensitization takes place and, therefore, may reflect a shift in the balance of the recipient alloimmune response toward immunological tolerance. This is compatible with the observation that a temporary depletion of local macrophages by subconjunctival injection of clodronate liposomes at the time of corneal transplantation in low-risk eyes results in permanent survival of most of the grafts (23, 24). In addition, the removal of regional lymph nodes can promote 90%or complete survival of corneal allografts placed in normal- and high-risk settings, respectively (25, 26).
In contrast to the avascular cornea, solid organ grafting requires a blood supply to provide wound healing and nutrition. The role of lymphatic vessels in transplanted solid organs has yet to be determined; they seem to be involved in inflammatory processes and immune rejections, but they also display important functions for the homeostasis of the graft (3–7). It was shown that lymphatic endothelial cells undergo significant alterations of their molecular markers after heart transplantation; this seems to be influenced by immune rejection episodes (8). In contrast, in renal grafting, it was shown that the graft function after 1 y was better in patients whose biopsies showed lymphatic vessels in their infiltrates compared with patients whose infiltrates were free of lymphatic vessels.
Our finding that a temporary selective inhibition of lymphangiogenesis prior to grafting potentially causes an early postoperative lack of access to the lymphatic vascular system and leads to significantly better (intermediate-term) graft survival reveals the potential of a selective and specific antilymphangiogenic therapy after solid organ transplantation. In solid organ transplantation, a blood supply is essential for wound healing, nutrition, and homeostasis of the graft, whereas lymphangiogenesis was recently implicated to play an important role in mediating immune rejections after kidney and heart transplantation (3, 7). Selective temporary anti-lymphangiogenic therapies might be a promising strategy for transplantation in the future.
Acknowledgments
This work was supported by the Interdisciplinary Centre for Clinical Research Erlangen (A9), the German Research Foundation (Sonderforschungsbereich SFB 643: B10), and the National Institutes of Health.
We thank Prof. Dr. D.G. Jackson, Oxford, U.K., for the kind gift of the LYVE-1 Ab.
Abbreviations used in this paper
- VEGF
vascular endothelial growth factor
- VEGFR
vascular endothelial growth factor receptor
Footnotes
Disclosures
G.Z. is an employee of Jerini AG (Berlin, Germany). S.W. is an employee of Regeneron Pharmaceuticals (Tarrytown, NY).
References
- 1.Lechler RI, Sykes M, Thomson AW, Turka LA. Organ transplantation—how much of the promise has been realized? Nat Med. 2005;11:605–613. doi: 10.1038/nm1251. [DOI] [PubMed] [Google Scholar]
- 2.Kaplan HJ, Streilein JW, Stevens TR. Transplantation immunology of the anterior chamber of the eye. II. Immune response to allogeneic cells. J Immunol. 1975;115:805–810. [PubMed] [Google Scholar]
- 3.Kerjaschki D, Huttary N, Raab I, Regele H, Bojarski-Nagy K, Bartel G, Kröber SM, Greinix H, Rosenmaier A, Karlhofer F, et al. Lymphatic endothelial progenitor cells contribute to de novo lymphangiogenesis in human renal transplants. Nat Med. 2006;12:230–234. doi: 10.1038/nm1340. [DOI] [PubMed] [Google Scholar]
- 4.Yamamoto I, Yamaguchi Y, Yamamoto H, Hosoya T, Horita S, Tanabe K, Fuchinoue S, Teraoka S, Toma H. A pathological analysis of lymphatic vessels in early renal allograft. Transplant Proc. 2006;38:3300–3303. doi: 10.1016/j.transproceed.2006.10.095. [DOI] [PubMed] [Google Scholar]
- 5.Stuht S, Gwinner W, Franz I, Schwarz A, Jonigk D, Kreipe H, Kerjaschki D, Haller H, Mengel M. Lymphatic neoangiogenesis in human renal allografts: results from sequential protocol biopsies. Am J Transplant. 2007;7:377–384. doi: 10.1111/j.1600-6143.2006.01638.x. [DOI] [PubMed] [Google Scholar]
- 6.Jonigk D, Lehmann U, Stuht S, Wilhelmi M, Haverich A, Kreipe H, Mengel M. Recipient-derived neoangiogenesis of arterioles and lymphatics in quilty lesions of cardiac allografts. Transplantation. 2007;84:1335–1342. doi: 10.1097/01.tp.0000287458.72440.75. [DOI] [PubMed] [Google Scholar]
- 7.Geissler HJ, Dashkevich A, Fischer UM, Fries JW, Kuhn-Régnier F, Addicks K, Mehlhorn U, Bloch W. First year changes of myocardial lymphatic endothelial markers in heart transplant recipients. Eur J Cardiothorac Surg. 2006;29:767–771. doi: 10.1016/j.ejcts.2005.12.024. [DOI] [PubMed] [Google Scholar]
- 8.Cursiefen C, Cao J, Chen L, Liu Y, Maruyama K, Jackson D, Kruse FE, Wiegand SJ, Dana MR, Streilein JW. Inhibition of hemangiogenesis and lymphangiogenesis after normal-risk corneal transplantation by neutralizing VEGF promotes graft survival. Invest Ophthalmol Vis Sci. 2004;45:2666–2673. doi: 10.1167/iovs.03-1380. [DOI] [PubMed] [Google Scholar]
- 9.Küchle M, Cursiefen C, Nguyen NX, Langenbucher A, Seitz B, Wenkel H, Martus P, Naumann GO. Risk factors for corneal allograft rejection: intermediate results of a prospective normal-risk keratoplasty study. Graefes Arch Clin Exp Ophthalmol. 2002;240:580–584. doi: 10.1007/s00417-002-0496-5. [DOI] [PubMed] [Google Scholar]
- 10.Dietrich T, Onderka J, Bock F, Kruse FE, Vossmeyer D, Stragies R, Zahn G, Cursiefen C. Inhibition of inflammatory lymphangiogenesis by integrin alpha5 blockade. Am J Pathol. 2007;171:361–372. doi: 10.2353/ajpath.2007.060896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Okazaki T, Ni A, Ayeni OA, Baluk P, Yao LC, Vossmeyer D, Zischinsky G, Zahn G, Knolle J, Christner C, McDonald DM. alpha5beta1 Integrin blockade inhibits lymphangiogenesis in airway inflammation. Am J Pathol. 2009;174:2378–2387. doi: 10.2353/ajpath.2009.080942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bock F, Onderka J, Dietrich T, Bachmann B, Pytowski B, Cursiefen C. Blockade of VEGFR3-signalling specifically inhibits lymphangiogenesis in inflammatory corneal neovascularisation. Graefes Arch Clin Exp Ophthalmol. 2008;246:115–119. doi: 10.1007/s00417-007-0683-5. [DOI] [PubMed] [Google Scholar]
- 13.Streilein JW, Bradley D, Sano Y, Sonoda Y. Immunosuppressive properties of tissues obtained from eyes with experimentally manipulated corneas. Invest Ophthalmol Vis Sci. 1996;37:413–424. [PubMed] [Google Scholar]
- 14.Stragies R, Osterkamp F, Zischinsky G, Vossmeyer D, Kalkhof H, Reimer U, Zahn G. Design and synthesis of a new class of selective integrin alpha5beta1 antagonists. J Med Chem. 2007;50:3786–3794. doi: 10.1021/jm070002v. [DOI] [PubMed] [Google Scholar]
- 15.Sano Y, Ksander BR, Streilein JW. Fate of orthotopic corneal allografts in eyes that cannot support anterior chamber-associated immune deviation induction. Invest Ophthalmol Vis Sci. 1995;36:2176–2185. [PubMed] [Google Scholar]
- 16.Sonoda Y, Streilein JW. Orthotopic corneal transplantation in mice—evidence that the immunogenetic rules of rejection do not apply. Transplantation. 1992;54:694–704. doi: 10.1097/00007890-199210000-00026. [DOI] [PubMed] [Google Scholar]
- 17.Cursiefen C, Chen L, Borges LP, Jackson D, Cao J, Radziejewski C, D’Amore PA, Dana MR, Wiegand SJ, Streilein JW. VEGF-A stimulates lymphangiogenesis and hemangiogenesis in inflammatory neovascularization via macrophage recruitment. J Clin Invest. 2004;113:1040–1050. doi: 10.1172/JCI20465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cursiefen C, Ikeda S, Nishina PM, Smith RS, Ikeda A, Jackson D, Mo JS, Chen L, Dana MR, Pytowski B, et al. Spontaneous corneal hem- and lymphangiogenesis in mice with destrin-mutation depend on VEGFR3 signaling. Am J Pathol. 2005;166:1367–1377. doi: 10.1016/S0002-9440(10)62355-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tammela T, Zarkada G, Wallgard E, Murtomäki A, Suchting S, Wirzenius M, Waltari M, Hellström M, Schomber T, Peltonen R, et al. Blocking VEGFR-3 suppresses angiogenic sprouting and vascular network formation. Nature. 2008;454:656–660. doi: 10.1038/nature07083. [DOI] [PubMed] [Google Scholar]
- 20.Maguire MG, Stark WJ, Gottsch JD, Stulting RD, Sugar A, Fink NE, Schwartz A Collaborative Corneal Transplantation Studies Research Group. Risk factors for corneal graft failure and rejection in the collaborative corneal transplantation studies. Ophthalmology. 1994;101:1536–1547. doi: 10.1016/s0161-6420(94)31138-9. [DOI] [PubMed] [Google Scholar]
- 21.Sonoda Y, Sano Y, Ksander B, Streilein JW. Characterization of cell-mediated immune responses elicited by orthotopic corneal allografts in mice. Invest Ophthalmol Vis Sci. 1995;36:427–434. [PubMed] [Google Scholar]
- 22.Sonoda Y, Streilein JW. Impaired cell-mediated immunity in mice bearing healthy orthotopic corneal allografts. J Immunol. 1993;150:1727–1734. [PubMed] [Google Scholar]
- 23.Slegers TP, van der Gaag R, van Rooijen N, van Rij G, Streilein JW. Effect of local macrophage depletion on cellular immunity and tolerance evoked by corneal allografts. Curr Eye Res. 2003;26:73–79. doi: 10.1076/ceyr.26.2.73.14510. [DOI] [PubMed] [Google Scholar]
- 24.Slegers TP, van Rooijen N, van Rij G, van der Gaag R. Delayed graft rejection in pre-vascularised corneas after subconjunctival injection of clodronate liposomes. Curr Eye Res. 2000;20:322–324. [PubMed] [Google Scholar]
- 25.Yamagami S, Dana MR. The critical role of lymph nodes in corneal alloimmunization and graft rejection. Invest Ophthalmol Vis Sci. 2001;42:1293–1298. [PubMed] [Google Scholar]
- 26.Yamagami S, Dana MR, Tsuru T. Draining lymph nodes play an essential role in alloimmunity generated in response to high-risk corneal transplantation. Cornea. 2002;21:405–409. doi: 10.1097/00003226-200205000-00014. [DOI] [PubMed] [Google Scholar]