Abstract
Objective
To assess the effectiveness of robotically assisted body weight supported treadmill training (RABWSTT) for improving cardiovascular fitness in chronic motor incomplete spinal cord injury (CMISCI).
Design
Pilot prospective randomized, controlled clinical trial.
Setting
Outpatient rehabilitation specialty hospital.
Participants
Eighteen individuals with CMISCI with American Spinal Injury Association (ASIA) level between C4 and L2 and at least one-year post injury.
Interventions
CMISCI participants were randomized to RABWSTT or a home stretching program (HSP) three times per week for three months. Those in the home stretching group were crossed over to three months of RABWSTT following completion of the initial three month phase.
Outcome measures
Peak oxygen consumption (peak VO2) was measured during both robotic treadmill walking and arm cycle ergometry: twice at baseline, once at six weeks (mid-training) and twice at three months (post-training). Peak VO2 values were normalized for body mass.
Results
The RABWSTT group improved peak VO2 by 12.3% during robotic treadmill walking (20.2 ± 7.4 to 22.7 ± 7.5 ml/kg/min, P = 0.018), compared to a non-significant 3.9% within group change observed in HSP controls (P = 0.37). Neither group displayed a significant change in peak VO2 during arm cycle ergometry (RABWSTT, 8.5% (P = 0.25); HSP, 1.76% (P = 0.72)). A repeated measures analysis showed statistically significant differences between treatments for peak VO2 during both robotic treadmill walking (P = 0.002) and arm cycle ergometry (P = 0.001).
Conclusion
RABWSTT is an effective intervention model for improving peak fitness levels assessed during robotic treadmill walking in persons with CMISCI.
Keywords: Spinal cord injury, Robotic exercise, Treadmill training, Peak oxygen consumption
Introduction
Individuals living with spinal cord injury (SCI), even those with motor incomplete injuries, generally have sedentary lifestyles, resulting in an increased risk of developing premature coronary artery disease.1 Autopsy studies have shown that there is a greater atherosclerotic burden in those with SCI.2 Additionally, heart disease represents one of the leading causes of death in SCI.3–6 Aerobic fitness is comparatively poor in SCI populations, with nearly 25% of otherwise healthy young individuals with SCI failing to achieve oxygen consumption levels sufficient to perform many essential activities of daily living (ADLs).7 This loss of aerobic fitness is associated with progression of several cardiovascular risk factors, including dyslipidemia, glucose intolerance, obesity, and a sedentary lifestyle.8,9
Body weight supported treadmill training is an emerging exercise intervention for individuals with chronic motor incomplete spinal cord injury (CMISCI). Initial trials have focused on the feasibility of the technique and its use for functional gait recovery, both in the sub-acute10 and chronic condition.11–13 The peak fitness effects of body weight supported treadmill training (BWSTT) in CMISCI have been addressed in several studies. Nash et al.14 reported a single case of an individual with American Spinal Injury Association (ASIA) Impairment Scale (AIS) B who underwent a single episode of BWSTT in which there was a recorded increase in VO2 and minute ventilation upon onset of locomotion. Additionally, Jack et al.15 reported that two subjects with CMISCI completing 20 weeks of BWSTT increased peak work rate and peak oxygen uptake. This observation differs from Kressler et al.16 in their study of four different approaches to body weight supported locomotor training. In this study, individuals assigned to a three-month, five-days-a-week robotic treatment group failed to display a significant change in peak aerobic capacity over the course of the intervention. In contrast, individuals assigned to overground-electrical stimulation, treadmill with manual assistance, or treadmill with electrical stimulation groups exhibited improved peak VO2 responses post intervention. It should be noted, though, that the robotic training paradigm employed in their study was passive in nature (using 100% guidance force from the robot). Israel et al.,17 in a non-interventional study, examined aerobic capacity during robotic exercise and concluded that VO2 responses were similar to those derived during therapist-assisted treadmill walking. This observation occurred only if participants were instructed to actively engage during the robot-assisted task and were presented with feedback regarding this effort.
The primary purpose of our study was to assess the extent to which RABWSTT would improve cardiovascular fitness as measured by open circuit spirometry measurement of peak VO2 in subjects with CMISCI. We hypothesized that participants with CMISCI would prove capable of training at levels required to significantly impact peak VO2 within group, as well as produce significant between-group effects when compared with a Home Stretching Program (HSP) control group. It should be noted that the subjects enrolled in this current study were also involved in a test-retest reliability study of peak VO2 during robotic treadmill walking and arm cycle ergometry in our laboratory.18 In a study of 21 individuals tested using the same modality on separate days, a correlation of r = 0.96 and 0.95 were found for robotic treadmill walking and arm cycle ergometry respectively. This reproducibility across days suggests that peak VO2 can be reliably used as an outcome measure in a CMICSI exercise intervention trial.
Materials and methods
Participants
The University of Maryland Baltimore Institutional Review Board (IRB), the Baltimore VA Medical Center (VAMC) Research Committee, University of Maryland Rehabilitation and Orthopaedic Institute (UMROI, formerly Kernan Hospital) Medical Executive Committee approved this protocol.
Individuals with CMISCI were recruited from the outpatient SCI clinics at an academically- affiliated 132-bed rehabilitation hospital center (UMROI) and an academically affiliated outpatient Department of Veterans Affairs SCI clinic (VAMC). All facets of the study were performed at UMROI except for the dual-energy X-ray absorptiometry (DXA) studies, which were performed at the VAMC. Eligible participants had sustained traumatic SCI at least 12 months prior to enrollment, were between 18 and 80 years old, had a level of injury between C4 and L2, and an AIS of either C or D. Study participants were required to tolerate an upright posture in a standing frame for at least 30 minutes, but did not necessarily have to be community ambulators. Subjects were excluded if they had a history of uncontrolled hypertension, unstable angina, congestive heart failure, chronic obstructive pulmonary disease, symptomatic peripheral arterial occlusive disease, or recent (within the last 3 months) hospitalization for any medical problem. Individuals with severe contractures or frequent uncontrolled bouts of autonomic dysreflexia were also excluded. All participants underwent baseline whole body DXA scans using a Lunar iDXA System (Analysis Version 13.20, GE HealthCare, Madison, WI, USA), and were excluded if their average bone mineral density T score was −3.5 or below, as this was indicative of risk for long bone fracture during RABWSTT.
Baseline evaluations
Baseline evaluations included a full history and physical, electrocardiogram, and serum laboratory tests to screen for renal disease, liver disease and uncontrolled diabetes. Neurologic assessments were done using the International Standards for Neurological Classification of Spinal Cord Injury (ISNCSCI), and lower extremity spasticity was assessed at the hip and knee using the Modified Ashworth Scale. In addition, demographic information was collected on all individuals. Finally, all individuals were tested by one of the physical therapists to determine their Walking Index for Spinal Cord Injury (WISCI II) score.
Randomization and intervention
Participants were randomized to either the RABWSTT or the HSP control group. Both the treatment and control interventions were performed three times per week for three months. Those randomized to the control group were subsequently crossed over into RABWSTT for an additional three-month period. Randomization was done by the study coordinators, and was unknown to the treating therapists and exercise physiologist until after baseline testing. More individuals with tetraplegia than paraplegia presented for inclusion in the study. Because of chance, the size of the treatment group was larger than that of the control group, and no individuals with paraplegia ended up in the control group.
Lokomat acclimatization
RABWSTT was performed using the Lokomat® robotic treadmill training device. The Lokomat used in the study had a continuously variable weight suspension system installed. The study was designed to include several acclimatization sessions, and several baseline peak VO2 determination sessions, followed by initiation of the exercise intervention. Each participant's first acclimatization session allowed the physical therapist (NP, JM or ME) to measure leg length, determine appropriate strap configurations, and assess the individual's gait pattern at approximately 80% partial weight support at a slow treadmill walking speed. Adjustments were made to body weight support and initial speed based on comfort and the need to avoid safety deactivation of the robotic device due to excessive spasticity. Ultimately, partial body weight support was set between 70 and 80% if the participant was able to support his or her weight. One hundred percent body weight support was used if the participant was unable to support his or her weight. Participants performed two acclimation sessions in the Lokomat prior to the assessment of peak VO2 with additional sessions scheduled if deemed necessary in order to achieve full acclimation in the robotic device.
Peak VO2 testing procedures
Peak oxygen consumption (peak VO2) assessments were done at baseline, six and twelve weeks in all subjects (beginning, middle and end of intervention period). Additionally, those initially assigned to the control group were re-assessed after 18 and 24 weeks to determine the effects of the 12-week RABWSTT crossover period. Measurement of peak VO2 was performed using the SensorMedics VMAX ENCORE 20N Indirect Calorimetry System (SensorMedics Corporation, a subsidiary of VIASYS Healthcare Inc., Yorba Linda, CA, USA), an open circuit spirometry device permiting continuous assessment of oxygen consumption (VO2) during robotic treadmill walking and arm ergometry bouts. A Hans Rudolph mask, a tight-fitting face-piece with a head harness attachment, was placed over the participant's mouth and nose to collect and transport expired air flow and gas concentrations to the SensorMedics cart. A computer software program integrated this information to calculate VO2 data. Subjects were given time to acclimatize to the mask prior to the measurement of peak VO2. All peak tests were performed on separate days.
Robotic treadmill walking peak VO2 testing
Two peak VO2 robotic data collection sessions were performed by all participants at baseline and twelve-week time points (Fig. 1A). One peak VO2 data collection session was performed at the six-week mid-point. Participants performed a brief three-minute warm-up on the robotic system at 100% body weight support with the treadmill speed at 1.5 to 1.8 kilometers per hour (kph). Peak VO2 was induced by periodically modifying treadmill speed, body weight support, or guidance force every 2–3 minutes. Treadmill speed was increased by 0.2 to 0.3 kph during adjustment intervals. Using visual gait assessment, the physical therapist adjusted body weight support during these intervals, ideally minimizing body weight support while maintaining a desired gait pattern. Guidance force was adjusted downward by 10% intervals during this process. Participants were instructed to actively assist the robotic device through the gait-cycle to generate a cardiac ‘workout’, with verbal and visual feedback regarding this effort provided by a research team member (therapist or exercise physiologist) and LCD monitor. Termination of each test (9–15 minutes) occurred with volitional fatigue or if the participant was unable to continue to perform the required work rate using a safe and effective gait pattern.
Figure 1 .
Photographs of a subject engaging in VO2 peak measurements while performing exercise in the Lokomat (A) and with arm cycle ergometry (B). (Photographic permission on file).
Arm cycle ergometer peak VO2 testing
All participants performed two peak VO2 arm cycle ergometer tests using a standard desk mounted device (Fig. 1B). Weights were used to secure the arm cycle ergometer to the desktop. If needed, hand stability positioning straps were utilized during arm cycle ergometry.
Participants performed a 3–5 minute warm-up on the arm cycle ergometer at zero resistance using a self-selected arm rotation cadence. The peak VO2 response was elicited by periodically (every minute) increasing the belt tension on the ergometer flywheel. Participants were instructed to maintain their self-selected rotational cadence at each new work rate. The peak test was terminated at volitional fatigue or if participants were incapable of maintaining the self-selected pedal cadence. The test duration was approximately 9 to 15 minutes.
Determination of peak VO2 values
For both the robotic treadmill exercise and arm cycle ergometer tests, gas exchange data was continuously monitored with open circuit spirometry. A computer software program integrated the data to provide VO2 every 20 seconds. The peak VO2 value was the average of the two highest VO2 consecutive 20-second sampling points.
DXA studies
DXA evaluations were done on all participants at baseline and then at completion of the exercise intervention. Body composition results obtained from the DXA evaluations were considered secondary outcome measures. Data collected were tissue fat percentage (%), lean tissue mass (kg), total body mass (kg), and bone mineral content (kg).
RABWSTT treatment intervention
Once baseline peak VO2 testing was completed, subjects assigned to the treatment group started a three times weekly exercise regimen. Training sessions lasted from 20 to 45 minutes and consisted of a graded coached exercise session with the training intensity established at 80–85% of heart rate reserve as determined using the Karvonen equation.19 Intensity was manipulated by adjustments to treadmill speed, guidance force (resistance), and body weight support. The decision as to which parameter to adjust was made by the research team based on a clinical judgment for that particular individual. The first week intervention sessions lasted 20 minutes. At each subsequent week, the training session duration was increased by five minutes until the maximal exercise duration reached 45 minutes. Initially, the protocol called for gradual decrease in partial weight suspension and then a gradual increase in treadmill speed. This sequencing proved difficult, as the need for partial weight suspension clinically depended more on the need to avoid tripping and collapse than was initially predicted. The variable partial weight suspension system also was dynamic, and therefore the weight suspension was variable throughout the gait cycle. Therefore, in order to provide aerobic challenge, the speed of the treadmill was increased up to 3.2 kph. Additional aerobic challenge was provided by coaching the subjects to exert themselves against the exoskeletal device. Visual feedback of force generation was provided to subjects via a screen display. A Polar® monitor was worn by subjects to assess heart rate responses during exercise and test bouts. The Borg scale (0–10) was administered to participants, every two minutes during the treadmill walking sessions, to subjectively assess exercise intensity.
Subjects were asked to come in for training sessions three times per week for 12 consecutive weeks. Compliance rates were monitored, and additional sessions were added if participants failed to reach an 80% attendance rate at week 12. Any participant who missed two consecutive weeks of training due to health or personal issues was asked to repeat baseline testing and resume training starting from week one.
Home Stretching Program (HSP) control intervention
Individuals randomized to the HSP exercised three days a week for approximately 20–25 minutes. The HSP incorporated traditional rehabilitative strengthening and stretching exercises with an emphasis specific to the needs of each particular individual. A physical therapist met with the participant after the initial randomization to review the exercise program. Participants were provided with instructions and diagrams on how to perform each exercise at home. A training log was provided to each participant to record these activities. A research team member periodically (once every 2 weeks) contacted participants to track progress during the control intervention period.
Statistical analyses
Means and standard deviations were generated for demographic information in both the treatment and control groups using Excel (Microsoft Corp, Redmond, WA, USA). Peak VO2 measures were normalized relative to body mass in units of ml/kg/min. Peak oxygen consumption outcomes were analyzed using two different methods. First, within-group effects before and after the three-month intervention period were assessed with paired t-tests. This method of analysis, in essence, treated the intervention group results as those stemming from a non-controlled pilot study. Furthermore, this type of analysis was more conducive to adding in crossover data from those initially randomized to the HSP control group, thereby enabling a look at within-group results using a larger sample size. Second, a comparative analysis of the treatment versus the control groups was performed by comparing mean values between RABWSTT and HSP groups across all 3 time points. This was done using between groups repeated measures analysis of variance (IBM SPSS Statistics 20, IBM Corp., Armonk, NY, USA). Two statistical methods were used based on the presence of an unbalanced number of subjects in the treatment and control groups as well as inequities with respect to group characteristics, which potentially confounded the intergroup comparisons. Hence, inclusion of the single group comparison is relevant in the context of this initial pilot study. All outcome data was expressed as mean ± standard deviation with significance set at the P < 0.05 level.
Results
Demographic data
A total of 35 individuals who had the diagnosis of CMISCI were screened for this study. Eight individuals failed screening. Three were found to be AIS B, two had bone mineral densities below the IRB established criteria, one had severe pulmonary disease, one had an abnormal electrocardiogram, and one individual failed to complete the screening process. Twenty-seven individuals met the inclusion criteria and were enrolled to participate. Six participants were withdrawn from the study before completion of the baseline testing due to compliance issues (3), psychosocial difficulties (2), and symptomatic hypotension during the Lokomat acclimation phase (1). Three additional subjects dropped out of the study after completing the baseline tests. One participant was overwhelmed with work and school obligations. Another dropped out after deciding the risk of a possible bone fracture outweighed any potential benefits from the intervention. The third failed to attend his initial training sessions and was dropped from the intervention after not responding to numerous attempts to reschedule missed appointments. This left 18 subjects that completed the entire protocol. Because of chance and an unbalanced allocation schema, twelve subjects were randomized to robotic group and six were randomized to the stretching control group. All of the six stretching control group individuals did eventually cross over to robotic treatment.
Table 1 details the demographics of the two groups. The groups were fairly well matched for age, lower extremity motor score, and their baseline WISCI II assessments. Fifty percent of the participants in both groups were community ambulators. By standard ASIA classification methods there were fifteen individuals with tetraplegia and three with paraplegia. Using the motor classification schema alone, fourteen would be classified as having tetraplegia and four would be considered as having paraplegia. By chance, eight of the individuals with tetraplegia were randomized to RABWSTT and six to HSP control. Again by chance all of the motor classified individuals with paraplegia were randomized to RABWSTT. Table 2 displays the body composition data for the two groups both for baseline and post intervention. The HSP control group tended to be heavier than the robotic group, and had a slightly higher percentage of body fat on baseline DXA scan, but these differences were not statistically significant (body mass, P = 0.16 and % body fat, P = 0.88). Baseline peak VO2 levels during both robotic and arm ergometry testing were different between groups (robotic treadmill walking, P < 0.001; ergometer, P < 0.001).
Table 1 .
Baseline demographics for the robotic treatment and control groups (Means ± Standard Deviations)
| Robotic treatment group | Control group | |
|---|---|---|
| Number (n) | 12 | 6 |
| Average age (years.) | 51.5 ± 12.7 | 52 ± 15.4 |
| Age range (years.) | 28–66 | 30–72 |
| Average lower extremity motor score (LEMS) | 34.2 ± 11.5 | 36.2 ± 8.4 |
| Community ambulation (%) | 50 | 50 |
| Paraplegia (n) | 4 | 0 |
| Tetraplegia (n) | 8 | 6 |
| ASIA C (n) | 2 | 0 |
| ASIA D (n) | 10 | 6 |
Table 2 .
Body composition results from the DXA evaluations
| Pre | Post | P-value | |
|---|---|---|---|
| Robotic treatment group | |||
| Body mass (kg) | 80.8 ± 14.6 | 79.8 ± 15.0 | 0.15 |
| Tissue fat (%) | 33.6 ± 7.9 | 32.4 ± 7.7 | 0.05 |
| Lean tissue (kg) | 51.2 ± 8.5 | 51.4 ± 8.0 | 0.76 |
| Bone mineral content (kg) | 3.0 ± 0.5 | 3.0 ± 0.4 | 0.26 |
| Control group | |||
| Body mass (kg) | 94.3 ± 25.0 | 92.3 ± 21.3 | 0.27 |
| Tissue fat (%) | 34.2 ± 6.9 | 33.3 ± 7.4 | 0.26 |
| Lean tissue (kg) | 58.9 ± 11.3 | 58.3 ± 9.6 | 0.66 |
| Bone mineral content (kg) | 3.2 ± 0.5 | 3.2 ± 0.55 | 0.89 |
| Combined (treatment plus control-crossovers) | |||
| Body mass (kg) | 85.0 ± 18.1 | 84.8 ± 18.5 | 0.72 |
| Tissue fat (%) | 33.5 ± 7.4 | 32.8 ± 7.2 | 0.20 |
| Lean tissue (kg) | 53.8 ± 9.4 | 54.2 ± 9.4 | 0.40 |
| Bone mineral content (kg) | 3.1 ± 0.5 | 3.1 ± 0.4 | 0.36 |
Protocol compliance and adverse events
Subjects that were randomized to the robotic group were expected to exercise three days per week for twelve consecutive weeks. Compliance rates with this schedule were approximately 84%. Reasons for cancellation of individual sessions included medical issues (33.9%), weather (8.1%), equipment problems (8.9%) and a myriad of other reasons (49.2%). In addition, nine percent of the completed exercise sessions were truncated by robotic safety shutdowns, most of which were triggered by excessive subject spasticity.
The main training-related adverse event that occurred during the study was that of skin abrasion. Of the 18 subjects completing the study, two developed some type of skin irritation or abrasion. The sites affected were hip, groin, penis, back, wrist, glutei and scapula. Over the course of the study, we reduced the incidence of these issues by providing additional padding within the parachute harness setup.
Within-group effects results
Data points taken at baseline, six weeks and twelve weeks for both the RABSWTT group and the HSP stretching control group were reviewed for validity. Robotic treadmill walking data collected from a single participant assigned to the control group was omitted from data analysis due to problems in the collection of baseline peak VO2. His remaining data was included in the final analysis.
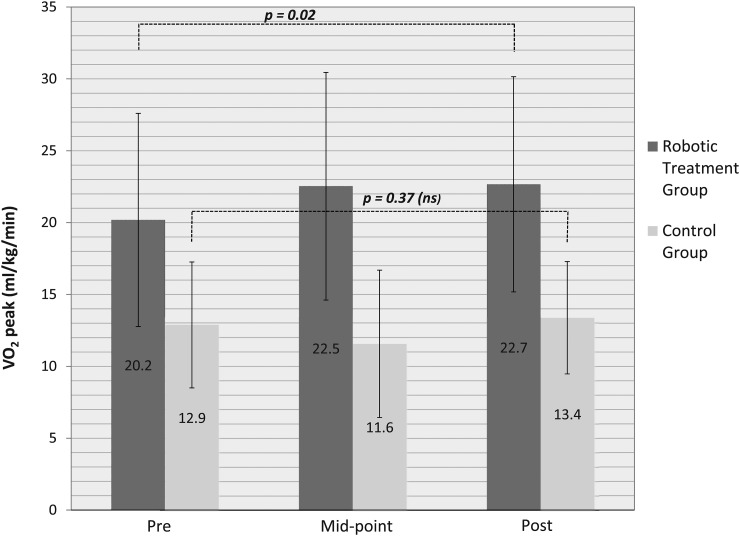
Peak VO2 during the robotic treadmill walking test improved from 20.2 ± 7.4 to 22.7 ± 7.5 ml/kg/min (12.3%, P = 0.018) in the treatment group over the course of the three-month intervention. When the six crossover subjects (control group) completed the robotic intervention and were added, peak VO2 gains were slightly diminished but still statistically significant (19.7 ± 7.6 to 21.4 ± 7.4 ml/kg/min, 8.5%, P = 0.038) (Fig. 2). Analysis of the midpoint six-week data when compared to baseline and without the crossover subjects showed a trend towards improvement in peak VO2, but this was not statistically significant (11.6%, P = 0.064). Inclusion of crossover subjects also failed to reach significance at the mid-way time point, but the trend was stronger (P = 0.051). Peak VO2 during the robotic treadmill walking test did not change in the HSP control group across 3 months (12.9 ± 4.4 vs. 13.4 ± 3.9 ml/kg/min; 3.9% increase, P = 0.37).
Figure 2 .
Peak oxygen consumption measured during robotic treadmill ambulation before, at mid-point (1½ months) and after a three month training intervention in individuals randomized to a robotic treatment or control group. Means and standard deviations are displayed for both groups in this figure. The significance level was set at P < 0.05.
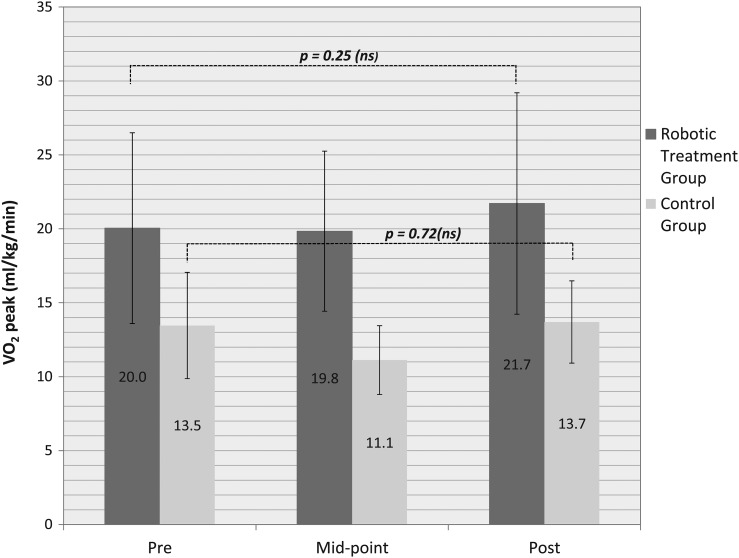
Peak oxygen consumption measured during arm cycle ergometry testing for the twelve subjects randomized to RABWSTT went from 20.0 ± 6.5 to 21.7 ± 7.5 ml/kg/min across training, which was statistically insignificant (8%, P = 0.25). Including the six crossover subjects made little difference to the results (17.8 ± 6.4 to 18.8 ± 7.7 ml/kg/min, 6%, P = 0.29) (Fig. 3). The control group also did not display a significant change in arm ergometry-induced peak VO2 over the course of the three-month observation period (13.5 ± 3.3 to 13.7 ± 2.5, ml/kg/min, P = 0.72).
Figure 3 .
Peak Oxygen Consumption measured during Arm Cycle Ergometry before, at mid-point (1½ months) and after a three month training intervention in individuals randomized to a robotic or control group. Means and standard deviations are displayed in this figure. The significance level was set at P < 0.05.
Lower extremity motor scores (LEMS) as determined from the initial ISNCSCI examination were compared to the baseline peak VO2 and change in peak VO2 data after three months of exercise (Δ peak VO2) using linear regression analysis. As might be expected, individuals with higher LEMS were able to mount higher baseline peak VO2 levels (R = 0.40) with the robotic device. No such correlation existed for the baseline peak VO2 levels recorded using arm cycle ergometry (R = 0.06). LEMS negatively correlated with Δ peak VO2 under both the robotic exercise (R = −0.55) and arm cycle ergometry (R = −0.51) conditions over the course of the three-month intervention. Lower extremity motor scores were not significantly different between groups at baseline (P = 0.97). No significant changes were observed for LEMS in either group following the intervention (robotic group, 34.2 ± 11.5 to 34.5 ± 10.7, P = 0.80; control group 34.0 ± 7.3 to 35.8 ± 7.9, P = 0.27).
Comparative analysis of treatment versus control group
Time by group interactions were assessed for peak VO2 using data from all three time points (baseline, mid-training, post-training). Comparing change in peak VO2 between groups across time indicated that the increase in RABWSTT group was greater than the increase in HSP controls during the robotic assessment (P = 0.002). Similarly, the group comparison of arm ergometry Δ peak VO2 yielded a significant time by group interaction, indicative of greater change in the RABWSTT treatment group compared to HSP controls (P = 0.001), despite the fact that there was no significant change detected by the within group data analysis for arm ergometry in RABWSTT.
Agreement of the two testing methods for VO2 peak adaptation
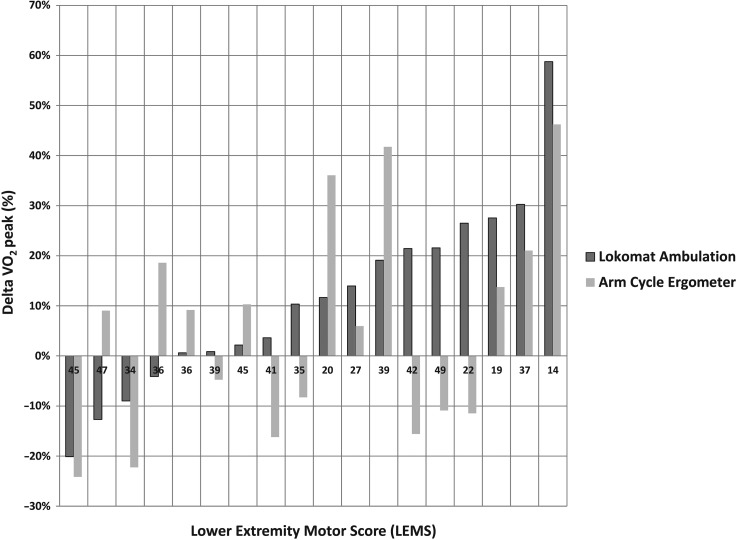
Degree of improvement in peak VO2 for most participants was in fairly good agreement across the 2 testing methods (robotic treadmill walking and arm ergometer). Fig. 4 displays the percent improvement in peak VO2 in individual subjects in the treatment arm of the study as measured with both modalities. Subjects are ranked left to right according to their percent improvement in robotic treadmill walking based peak VO2. Those that improved the most with robotic treadmill walking were generally the ones that improved the most according to arm ergometry testing, although this wasn't universally the case. Interestingly, there was no clear correspondence between LEMS and the extent of improvement (or lack thereof).
Figure 4 .
Change in VO2 peak across the treatment intervention as measured both during Robotic treadmill ambulation and arm-cycle ergometry. Each pair of bars represents an individual subject. Subjects are presented in rank order of their Delta VO2 peak values during Lokomat ambulation (Robotic Treadmill Walking). Individual subject LEMS are noted along the x-axis of the graph.
Heart rate and perceived exertion
Heart rates were collected during the peak robotic treadmill walking and arm cycle ergometer tests. Results showed no significant differences for maximal heart rate responses between tests in this group of individuals (robotic treadmill walking; 140 ± 23 vs. arm cycle ergometry 141 ± 25 bpm, P = 0.9). Perceived exertion values were also obtained at the completion of both testing formats. Participants reported an average exertion score of 8.9 ± 1.9 (range 4–10) and 9.0 ± 1.7 (range 5–10) at the termination of the robotic and arm cycle ergometer tests, respectively. This observation suggests that participants experienced similar perceptual experiences between modalities.
DXA results
The RABWSTT treatment group demonstrated reduced percentages of tissue fat after the three-month intervention. The percent tissue fat decreased from 33.6 ± 7.9 to 32.4 ± 7.7% (P = 0.05) post intervention. Total body mass also trended towards a decrease with RABWSTT, but this result did not reach statistical significance (P = 0.15). Lean tissue mass and bone mineral content were not altered by the robotic exercise intervention. The HSP control group did not show any significant body composition changes according to DXA. When the subjects that started as controls and then crossed over to robotic treatment were included in the analysis, the results were actually diluted and the change in tissue fat was no longer statistically significant. Table 2 provides summary information for the DXA results.
Discussion
This pilot study demonstrates that measurement of peak VO2 during robotic treadmill walking testing is feasible in individuals with CMISCI. Furthermore, the study supports the hypothesis that the RABWSTT intervention model is robust enough to significantly improve robotic treadmill-derived peak VO2 measures in this patient population. The arm ergometry-derived peak VO2 measures did not significantly improve after the RABWSTT training paradigm, but using the comparative analysis to the control group, there was a significant difference. A larger study with greater power might have resulted in a more robust outcome with regard to non-specific (i.e. arm ergometry after leg exercise) adaptations.
The effect size for peak VO2 (12.3%) in our participants with CMISCI was comparable to that observed in survivors of stroke undergoing non-body weight supported treadmill training as reported by Macko.20 In that study of patients with hemiparetic stroke, treadmill exercise training over six months achieved a 17% increase in peak VO2. In a prior study by Jack et al.,15 two subjects with incomplete SCI participating in a three times per week exercise protocol showed exercise training induced peak VO2 increases of 11.4 and 21.5% across the same training time period as the current study. This study reported considerable variability and challenges associated with these types of measurements in subjects with CMISCI with different neurological baselines, both in terms of motor control and autonomic function, and suggested that larger cohorts of subjects would be needed to determine the effectiveness of exercise testing protocols. Our study provides the needed data from a larger number of subjects with CMICSI, showing training effect sizes that are comparable to the few subjects previously reported for this important measure.
Peak VO2 in SCI using other modalities
Our findings show improved cardiovascular fitness in individuals with CMISCI following a three-month robotic training intervention. This is in contrast to the work of Kressler et al.16 who did not find this effect with the use of robotic training. Others have also examined cardiovascular responses in individuals with SCI using different exercise modalities. In most instances, robotic treadmill walking produced lower acute cardiovascular responses compared to values observed during therapist-assisted stepping,17,21 ZeroG® walking,22 and manual treadmill walking.22 A possible explanation for these differences was highlighted in an earlier study by Israel et al.17 In their investigation, the authors used both passive and active approaches during robotic stepping and compared these results with values obtained during therapist assisted treadmill stepping. The passive robotic condition resulted in lower peak VO2 responses compared to values observed during the therapist assisted walking. The differences between these modalities disappeared when participants actively worked with the robotic device and were provided visual feedback on this task.
Methodology differences provide a possible explanation for the discrepancies between our findings and those reported by others. Previous investigations employed a passive approach to the robotic intervention with individuals instructed to work with the device at 100% guidance force with little to no feedback regarding the task. Our training methodology utilized a different approach requiring individuals to actively participate during the gait cycle. We adjusted guidance force downward during exercise so as to increase the intensity of the aerobic challenge. We also provided visual and verbal feedback (coaching) during this effort. Our results suggest that aerobic demand may be manipulated during robotic treadmill walking by adjustments to guidance force and through feedback.
The methodology used to assess peak VO2 provides another possible explanation for the differences between our findings and those reported by other investigators. Kressler et al.16 assessed peak VO2 during over-ground walking. The current study assessed peak VO2 during robotic stepping and arm cycle ergometry. The over-ground (Kressler) and robotic stepping (current study) groups in these interventions displayed improved cardiovascular fitness when peak VO2 was assessed with the device implemented in the training intervention. It is possible that the method of exercise employed during VO2 assessment biases the results towards success of that same method of exercise over others. This specificity is supported in part by our data when comparing changes in VO2 peak as measured during robotic exercise and arm cycle ergometry (see generalizability section below).
Baseline peak VO2 differences
We observed significant differences for peak VO2 at baseline between groups during both robotic treadmill walking and arm cycling ergometry tests. This difference was not due to age, body composition, lower extremity strength, or ability to ambulate. Previously, injury level had been reported as factor in the attainment of peak VO2, with individuals with paraplegia displaying higher values compared to those with tetraplegia.23 This observation was not a factor accounting for the peak VO2 differences between groups of participants in our study. At this time, we can only speculate what may have been the reason(s) for the difference in peak VO2 at baseline between groups. The small sample size and chance are the most likely factors contributing to the discrepancy. By chance, it appears that the smaller control group was comprised of a greater number of individuals with extremely low baseline aerobic capacity (peak VO2 below <15 ml/kg/min). In contrast, the treatment group (with a larger sample size) had ten individuals with peak VO2 values greater than 15 ml/kg/min during both the robotic treadmill walking and arm cycle ergometry baseline testing.
Generalizability of the findings
The exercise of leg musculature three days per week for three months using a treadmill training intervention produced an improvement in peak oxygen consumption when tested with robotic treadmill exercise. There was also a non-significant trend toward improvement seen when arm exercise was used in the assessment of peak VO2. When using the time by group analysis, a significant result with arm cycle ergometry was seen however. Therefore, the results suggest some generalizability of the cardiovascular effect. A larger sample size would likely better define this relationship.
Comparisons of upper vs. lower body exercise in healthy and CMISCI
The present study provides evidence to support an aerobic effect for individuals with CMISCI using a lower extremity exercise paradigm. This finding is in alignment with an upper extremity investigation by Grange et al.24 illustrating significant improvements in peak VO2 in the same population following a six-week arm ergometer intervention. That study also compared training-induced fitness gains in SCI with those examined in an age-matched able-bodied group, reporting that the able-bodied individuals displayed greater peak VO2 values at baseline and larger changes in peak VO2 capacity post-intervention compared to the CMISCI group. This finding and our observations verify the efficacy of different types of exercise interventions for producing positive adaptations in peak fitness in people with motor incomplete spinal cord injury. Nevertheless, it should be emphasized that these changes in peak VO2 are likely to be smaller than those observed in able-bodied individuals across a similar training stimulus. Therefore, it may be worth studying whether more optimal degrees of peak VO2 adaptation are possible in CMISCI by combining robotically assisted treadmill training with other modalities (e.g. functional electrical stimulation (FES) or lower extremity strength training). One might also consider modifying the intensity or duration of the RABWSTT stimulus in order to achieve larger degrees of adaptation in CMISCI.
In the present study, individuals with incomplete spinal cord injuries displayed similar baseline peak VO2 values during robotic (lower extremity) and arm cycle ergometer (upper extremity) tests. This observation differs from information reported in the literature for able-bodied individuals where arm ergometry induced VO2 levels are consistently less than VO2 levels stemming from exercise bouts focusing predominantly on the lower extremities, reflecting a difference in total muscle groups utilized between modalities. For example, Castro et al.25 examined cardiovascular responses in able-bodied individuals performing arm and leg cycle ergometry tests and reported significantly higher peak oxygen consumption during the leg cycle ergometry task. Similar responses were observed in an investigation by McNarry et al.26 In this study, pubertal trained and untrained female swimmers displayed significantly greater peak oxygen consumption values during a leg cycle ergometer test than during arm cycle ergometry. The difference between our CMISCI findings and those from healthier populations is probably accounted for by loss of skeletal muscle mass and activation. Further work is needed before this is definitely pinpointed as the cause in those with CMISCI.
Significance of lower extremity motor score (LEMS)
The positive correlation between baseline LEMS and robotic treadmill walking peak VO2 makes logical sense in that one would expect that those subjects that have more available leg muscle power would have a greater baseline cardiovascular level of fitness. Also, it is known that individuals with high LEMS are more likely to ambulate,27 therefore providing greater opportunities to limit the decline in VO2 typically observed in sedentary individuals with SCI. To our knowledge, this is the first study to document this correlation between LEMS and aerobic capacity in individuals with CMISCI. In the present study, we also observed a negative correlation between baseline LEMS and the change in Δ peak VO2 with a three month exercise intervention. The inverse relationship between lower extremity motor score and ΔVO2 following the intervention indicates that those with the greatest weakness in the lower extremities at baseline are the most likely to show large gains in peak VO2 with this intervention and testing model.
Body composition and body mass adaptations
Body composition changes were minimal in the treatment and control groups following the three-month exercise intervention, with neither group displaying significant modifications in fat-free mass, total body mass, or bone density, although total percent body fat decreased in the treatment group in response to the intervention. The lack of overall change in fat-free mass and bone density likely reflects an inability to provide a sufficient load to our participant's skeletal muscle structure. In this study, it was necessary to provide some participants with greater body weight support to facilitate an appropriate and effective gait pattern. This procedure reduced the ground reaction force and stress placed on the skeletal muscle system during the training intervention and possibly attenuated potential benefits to body composition. Additionally, it may be necessary to lengthen the training intervention to induce positive modifications to muscle mass and bone density in this population (>12 months).
Heart rate response
Subjects with motor incomplete SCI were able to consistently elevate their heart rate during coached aerobic robotic and arm cycle ergometry activities, indicating that the training sessions were of sufficient intensity. Importantly, consistently elevated heart rates also indicate that sympathetic innervations to the peripheral vasculature and the heart remain intact. Our results provide evidence that a true cardiovascular response can be achieved in this patient group using a robotic treadmill training approach. These results are in agreement to those of Taylor et al.28 who examined a subject with AIS A paraplegia who underwent arms-only and hybrid FES exercise (combined voluntary arm and FES leg rowing) and Jung et al.29 who did a similar study of individuals who primarily but not exclusively had AIS A paraplegia.
Withdrawals and dropouts
Twenty-seven individuals fulfilled the inclusion and exclusion criteria outlined for this investigation. Six subjects dropped from the study before randomization but only three of the remaining 21 subjects (14%) did not complete the full protocol after randomization. This dropout rate is consistent with other intensive exercise interventions in this patient population. A review of these individuals’ folders failed to reveal a consistent causative factor for dropout. It would appear that the nature and the potential adverse effects of the intervention were not major factors.
Clinical significance
This investigation demonstrates that it is possible to produce an improvement in peak VO2 through three months of robotic training intervention in those with CMISCI. The translation of this into meaningful improvement in the ability to perform activities of daily living was not specifically demonstrated. It has been reported however by Manns et al.30 in a population of individuals with paraplegia that normalized peak VO2 does positively correlate with level of self reported physical activity. A systemic review nonetheless has yet to conclude that exercise, while able to improve physical capacity (as was seen in our study), translates to improvement in functional capacity31 in CMISCI, although in another neurologically disabled population, namely stroke, this has been clearly demonstrated.20 Therefore, given the mixed information, it can only be cautiously suggested that improvement in peak VO2 by our subjects may translate to greater independence and capacity in the performance of activities in daily life.
Challenges and limitations
The implementation of this trial was met with multiple challenges. First, our treatment and control groups were heterogeneous by age, injury level, and AIS classification. This factor combined with the relatively small sample size may affect how our findings are extrapolated to individuals with specific injury level classifications.
Second, there are no generally established accepted criteria for exclusion of subjects from robotic training based on osteopenia. It is well known, however, that the risk of fracture does increase in subjects with SCI with a decrease in bone mineral density.32 There are no published reports of long bone fracture using robotic treadmill training, although the manufacture of the Lokomat®, Hocoma, does have internal records of a single case possibly confounded by lack of adequate supervision during exercise. Our own Institutional Review Board tasked us with coming up with an empiric cut-off for osteopenia that might put subjects at excessive risk of fracture with participation in this aggressive exercise protocol. Therefore, after consultation with members of the Hocoma staff, we established the −3.5 T value as a cut off for inclusion.
Third, skin irritation was the most common adverse event that occurred during the study, resulting in multiple sessions missed. When the goal of the robotic exercise is aerobic, subjects tend to work harder and stress their support system to a greater degree than when gait training is the only objective. It is therefore important to make sure that the subjects are appropriately padded at areas of contact with the parachute harness.
Fourth, attendance rates for this type of extended study are less than perfect but overall the rate of 84% is adequate and compares favorably with other exercise intervention trials run in both neurologically disabled and healthy populations.
Fifth, ensuring adequate peak metabolic aerobic testing is not a trivial task. The original paradigm designed for this study was based on standard graded treadmill testing. This proved impractical since changing treadmill speed or reducing partial weight suspension did not necessarily translate into increased aerobic challenge. The robotic exoskeleton allows subjects to expend less than maximal effort and still achieve movement through the gait cycle. For this reason, participants were instructed to actively work through the gait cycle and were provided verbal and visual feedback regarding this effort in an attempt to elicit a peak VO2 response.
Limitations of the study included a control comparison design that was not purely randomized. Furthermore, the relative small sample size may have led to the observed pre-intervention differences between groups.
Conclusion
A controlled trial of robotically assisted body weight supported treadmill training in individuals with CMISCI has been accomplished. Three months of three times per week RABWSTT in persons with CMISCI can improve cardiovascular fitness as measured by peak oxygen consumption. The general exercise VO2 effect is similar to that seen in other disease entities such as stroke and peripheral vascular disease, but is not as robust as that observed in able-bodied individuals. Lower extremity motor scores can predict baseline peak VO2 values to some degree, but a high LEMS at baseline may predict a smaller effect of exercise due to a ceiling effect. Body composition measures did not appreciably change with this intervention with the exception of percent body fat, although this outcome did not reach significance when the crossover data was included in the analysis.
This study supports the clinical efficacy of robotic treadmill training as a modality to induce cardiovascular fitness modifications in CMISCI. The implementation of robotic treadmill training in this population remains complex and intensive, and further study is warranted to arrive at the ideal mix of training modalities needed to optimally impact peak VO2 in the CMISCI population. Further clarification as to the significance of these improvements with regards to physical activity is also needed.
Acknowledgments
The authors would like to thank nurse coordinators Gertrude Morrison and Pattie McCrae for their assistance. Kevin Chen, PhD assisted with the statistical analysis. The work is dedicated to Debbie Miller, RN who was the initial coordinator for the study.
Disclaimer statements
Contributors PG: PI, designer of the study, main manuscript writing; WS: exercise physiologist, main analyzer of data; HY: co-investigator, medical monitor, patient screening; MT: co-investigator, patient screening; NP-M, JMQ and ME — all physical therapists doing assessments and setting up all Lokomat therapies; FI: co-designer, main statistical reviewer; RM: co-PI and co-author of the grant.
Funding This work was funded by Department of Veterans Affairs Rehabilitation R&D Service Merit Review Award B4027I. This study was registered at clinicaltrials.gov with the identifier number NCT00385918.
Conflicts of interest None.
Ethics approval We certify that all applicable institutional and governmental regulations concerning the ethical use of human volunteers were followed during the course of this research.
References
- 1.Groah SL, Weitzenkamp D, Sett P, Soni B, Savic G. The relationship between neurological level of injury and symptomatic cardiovascular disease risk in the aging spinal injured. Spinal Cord 2001;39(6):310–7. [DOI] [PubMed] [Google Scholar]
- 2.Orakzai SH, Orakzai RH, Ahmadi N, Agrawal N, Bauman WA, Yee F, et al. . Measurement of coronary artery calcification by electron beam computerized tomography in persons with chronic spinal cord injury: evidence for increased atherosclerotic burden. Spinal Cord 2007;45(12):775–9. [DOI] [PubMed] [Google Scholar]
- 3.DeVivo MJ, Black KJ, Stover SL. Causes of death during the first 12 years after spinal cord injury. Arch Phys Med Rehabil 1993;74(3):248–54. [PubMed] [Google Scholar]
- 4.Frankel HL, Coll JR, Charlifue SW, Whiteneck GG, Gardner BP, Jamous MA, et al. . Long-term survival in spinal cord injury: a fifty year investigation. Spinal Cord 1998;36(4):266–74. [DOI] [PubMed] [Google Scholar]
- 5.Krause JS, Devivo MJ, Jackson AB. Health status, community integration, and economic risk factors for mortality after spinal cord injury. Arch Phys Med Rehabil 2004;85(11):1764–73. [DOI] [PubMed] [Google Scholar]
- 6.Whiteneck GG, Charlifue SW, Frankel HL, Fraser MH, Gardner BP, Gerhart KA, et al. . Mortality, morbidity, and psychosocial outcomes of persons spinal cord injured more than 20 years ago. Paraplegia 1992;30(9):617–30. [DOI] [PubMed] [Google Scholar]
- 7.Noreau L, Shephard RJ, Simard C, Pare G, Pomerleau P. Relationship of impairment and functional ability to habitual activity and fitness following spinal cord injury. Int J Rehabil Res 1993;16(4):265–75. [DOI] [PubMed] [Google Scholar]
- 8.Myers J, Lee M, Kiratli J. Cardiovascular disease in spinal cord injury: an overview of prevalence, risk, evaluation, and management. Am J Phys Med Rehabil 2007;86(2):142–52. [DOI] [PubMed] [Google Scholar]
- 9.Bauman WA, Spungen AM. Metabolic changes in persons after spinal cord injury. Phys Med Rehabil Clin N Am 2000;11(1):109–40. [PubMed] [Google Scholar]
- 10.Dobkin B, Apple D, Barbeau H, Basso M, Behrman A, Deforge D, et al. . Weight-supported treadmill vs over-ground training for walking after acute incomplete SCI. Neurology 2006;66(4):484–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hornby TG, Zemon DH, Campbell D. Robotic-assisted, body-weight-supported treadmill training in individuals following motor incomplete spinal cord injury. Phys Ther 2005;85(1):52–66. [PubMed] [Google Scholar]
- 12.Nooijen CF, Ter Hoeve N, Field-Fote EC. Gait quality is improved by locomotor training in individuals with SCI regardless of training approach. J Neuroeng Rehabil 2009;6:36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hicks AL, Adams MM, Martin Ginis K, Giangregorio L, Latimer A, Phillips SM, et al. . Long-term body-weight-supported treadmill training and subsequent follow-up in persons with chronic SCI: effects on functional walking ability and measures of subjective well-being. Spinal Cord 2005;43(5):291–8. [DOI] [PubMed] [Google Scholar]
- 14.Nash MS, Jacobs PL, Johnson BM, Field-Fote E. Metabolic and cardiac responses to robotic-assisted locomotion in motor-complete tetraplegia: a case report. J Spinal Cord Med 2004;27(1):78–82. [DOI] [PubMed] [Google Scholar]
- 15.Jack LP, Allan DB, Hunt KJ. Cardiopulmonary exercise testing during body weight supported treadmill exercise in incomplete spinal cord injury: a feasibility study. Technol Health Care 2009;17(1):13–23. [DOI] [PubMed] [Google Scholar]
- 16.Kressler J, Nash MS, Burns PA, Field-Fote EC. Metabolic responses to 4 different body weight-supported locomotor training approaches in persons with incomplete spinal cord injury. Arch Phys Med Rehabil 2013;94(8):1436–42. [DOI] [PubMed] [Google Scholar]
- 17.Israel JF, Campbell DD, Kahn JH, Hornby TG. Metabolic costs and muscle activity patterns during robotic- and therapist-assisted treadmill walking in individuals with incomplete spinal cord injury. Phys Ther 2006;86(11):1466–78. [DOI] [PubMed] [Google Scholar]
- 18.Gorman PH, Geigle PR, Chen K, York H, Scott W. Reliability and relatedness of peak VO2 assessments during body weight supported treadmill training and arm cycle ergometry in individuals with chronic motor incomplete spinal cord injury. Spinal Cord 2014;52(4):287–91. [DOI] [PubMed] [Google Scholar]
- 19.Karvonen MJ, Kentala E, Mustala O. The effects of training on heart rate; a longitudinal study. Annales medicinae experimentalis et biologiae Fenniae 1957;35(3):307–15. [PubMed] [Google Scholar]
- 20.Macko RF, Ivey FM, Forrester LW, Hanley D, Sorkin JD, Katzel LI, et al. . Treadmill exercise rehabilitation improves ambulatory function and cardiovascular fitness in patients with chronic stroke: a randomized, controlled trial. Stroke 2005;36(10):2206–11. [DOI] [PubMed] [Google Scholar]
- 21.Hornby TG, Kinnaird CR, Holleran CL, Rafferty MR, Rodriguez KS, Cain JB. Kinematic, muscular, and metabolic responses during exoskeletal-, elliptical-, or therapist-assisted stepping in people with incomplete spinal cord injury. Phys Ther 2012;92(10):1278–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fenuta AM, Hicks AL. Metabolic demand and muscle activation during different forms of bodyweight supported locomotion in men with incomplete SCI. BioMed Res Int 2014;2014:632765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Burkett LN, Chisum J, Stone W, Fernhall B. Exercise capacity of untrained spinal cord injured individuals and the relationship of peak oxygen uptake to level of injury. Paraplegia 1990;28(8):512–21. [DOI] [PubMed] [Google Scholar]
- 24.Grange CC, Bougenot MP, Groslambert A, Tordi N, Rouillon JD. Perceived exertion and rehabilitation with wheelchair ergometer: comparison between patients with spinal cord injury and healthy subjects. Spinal Cord 2002;40(10):513–8. [DOI] [PubMed] [Google Scholar]
- 25.Castro RR, Pedrosa S, Nobrega AC. Different ventilatory responses to progressive maximal exercise test performed with either the arms or legs. Clinics (Sao Paulo) 2011;66(7):1137–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.McNarry MA, Welsman JR, Jones AM. Influence of training and maturity status on the cardiopulmonary responses to ramp incremental cycle and upper body exercise in girls. J Appl Physiol (1985) 2011;110(2):375–81. [DOI] [PubMed] [Google Scholar]
- 27.Calder KB, Estores IM, Krassioukov A. Autonomic dysreflexia and associated acute neurogenic pulmonary edema in a patient with spinal cord injury: a case report and review of the literature. Spinal Cord 2009;47(5):423–5. [DOI] [PubMed] [Google Scholar]
- 28.Taylor JA, Picard G, Widrick JJ. Aerobic capacity with hybrid FES rowing in spinal cord injury: comparison with arms-only exercise and preliminary findings with regular training. PMR 2011;3(9):817–24. [DOI] [PubMed] [Google Scholar]
- 29.Jung DW, Park DS, Lee BS, Kim M. Development of a motor driven rowing machine with automatic functional electrical stimulation controller for individuals with paraplegia; a preliminary study. Ann Rehabil Med 2012;36(3):379–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Manns PJ, McCubbin JA, Williams DP. Fitness, inflammation, and the metabolic syndrome in men with paraplegia. Arch Phys Med Rehabil 2005;86(6):1176–81. [DOI] [PubMed] [Google Scholar]
- 31.Hicks AL, Martin Ginis KA, Pelletier CA, Ditor DS, Foulon B, Wolfe DL. The effects of exercise training on physical capacity, strength, body composition and functional performance among adults with spinal cord injury: a systematic review. Spinal Cord 2011;49(11):1103–27. [DOI] [PubMed] [Google Scholar]
- 32.Lazo MG, Shirazi P, Sam M, Giobbie-Hurder A, Blacconiere MJ, Muppidi M. Osteoporosis and risk of fracture in men with spinal cord injury. Spinal Cord 2001;39(4):208–14. [DOI] [PubMed] [Google Scholar]