Abstract
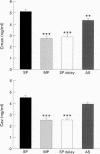
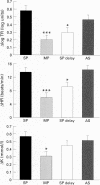
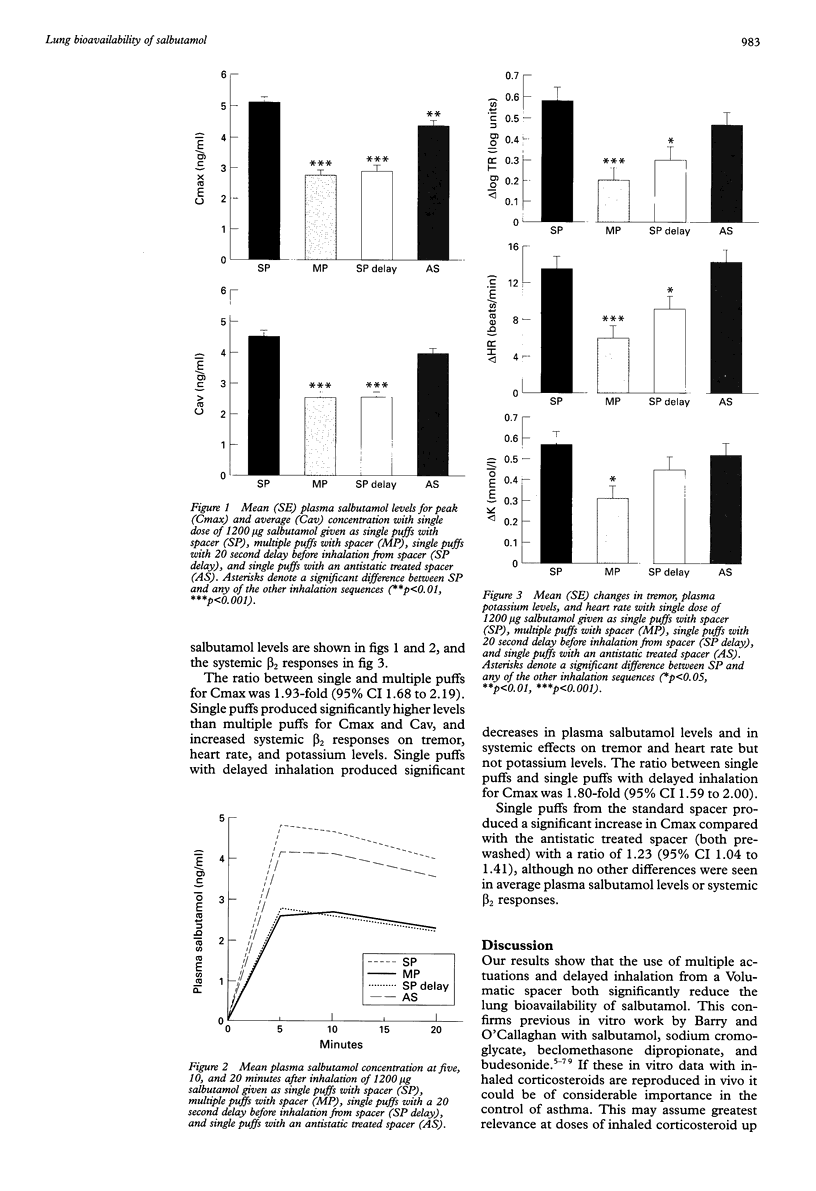
BACKGROUND: The aim of this study was to extend previous in vitro observations regarding the effects of multiple actuations of aerosols into spacer devices, delayed inhalation, and antistatic treatment of spacer devices on the amount of drug delivered for inhalation. An in vivo study of lung bioavailability of salbutamol from a large volume (Volumatic) spacer was conducted. METHODS: Ten healthy volunteers of mean age 20.5 years with a mean forced expiratory volume in one second of 112.1% predicted were studied in a randomised single blind (investigator blind) crossover study. 1200 micrograms of salbutamol was given with mouth rinsing (100 micrograms/puff) on four study days: single puffs via spacer, multiple puffs via spacer (3 x 4 puffs), single puffs with 20 second delay before inhalation via spacer, and single puffs via an antistatic treated spacer. All spacers, including those treated with antistatic, were prewashed prior to each study day. Measurements of lung bioavailability were made at five, 10, and 20 minutes after inhalation to determine peak (Cmax) and average (Cav) plasma salbutamol levels. Systemic beta 2 responses including finger tremor, heart rate, and plasma potassium levels were also evaluated. RESULTS: Single puffs from the spacer produced higher plasma salbutamol levels and greater systemic beta 2 responses than either multiple puffs or single puffs with delayed inhalation for a 1200 micrograms dose. For Cmax this amounted to a 1.93-fold (95% CI 1.68 to 2.19) greater lung bioavailability for single puffs than for multiple puffs and a 1.80-fold (95% CI 1.59 to 2.00) greater lung bioavailability for single puffs than for single puffs with a 20 second delay. Comparison of the normal and antistatic treated spacers (both prewashed) revealed differences for Cmax with levels 1.23-fold (95% CI 1.04 to 1.41) greater for the normal spacer. CONCLUSIONS: Delayed inhalation from a Volumatic spacer and the use of multiple puffs results in a considerable decrease in the delivery of salbutamol to the lungs with an approximate twofold reduction in lung bioavailability. Washing a Volumatic spacer is as effective as an antistatic lining in reducing the effects of static charge on salbutamol delivery in vivo.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barry P. W., O'Callaghan C. Multiple actuations of salbutamol MDI into a spacer device reduce the amount of drug recovered in the respirable range. Eur Respir J. 1994 Sep;7(9):1707–1709. doi: 10.1183/09031936.94.07091707. [DOI] [PubMed] [Google Scholar]
- Barry P. W., O'Callaghan C. The effect of delay, multiple actuations and spacer static charge on the in vitro delivery of budesonide from the Nebuhaler. Br J Clin Pharmacol. 1995 Jul;40(1):76–78. doi: 10.1111/j.1365-2125.1995.tb04538.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown P. H., Blundell G., Greening A. P., Crompton G. K. Do large volume spacer devices reduce the systemic effects of high dose inhaled corticosteroids? Thorax. 1990 Oct;45(10):736–739. doi: 10.1136/thx.45.10.736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keeley D. Large volume plastic spacers in asthma. BMJ. 1992 Sep 12;305(6854):598–599. doi: 10.1136/bmj.305.6854.598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lipworth B. J. Airway and systemic effects of inhaled corticosteroids in asthma: dose response relationship. Pulm Pharmacol. 1996 Feb;9(1):19–27. doi: 10.1006/pulp.1996.0002. [DOI] [PubMed] [Google Scholar]
- Lipworth B. J., Clark R. A., Dhillon D. P., Moreland T. A., Struthers A. D., Clark G. A., McDevitt D. G. Pharmacokinetics, efficacy and adverse effects of sublingual salbutamol in patients with asthma. Eur J Clin Pharmacol. 1989;37(6):567–571. doi: 10.1007/BF00562546. [DOI] [PubMed] [Google Scholar]
- Lipworth B. J., McDevitt D. G. Beta-adrenoceptor responses to inhaled salbutamol in normal subjects. Eur J Clin Pharmacol. 1989;36(3):239–245. doi: 10.1007/BF00558154. [DOI] [PubMed] [Google Scholar]
- O'Callaghan C., Cant M., Robertson C. Delivery of beclomethasone dipropionate from a spacer device: what dose is available for inhalation? Thorax. 1994 Oct;49(10):961–964. doi: 10.1136/thx.49.10.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Callaghan C., Lynch J., Cant M., Robertson C. Improvement in sodium cromoglycate delivery from a spacer device by use of an antistatic lining, immediate inhalation, and avoiding multiple actuations of drug. Thorax. 1993 Jun;48(6):603–606. doi: 10.1136/thx.48.6.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Callaghan C., Milner A. D., Swarbrick A. Spacer device with face mask attachment for giving bronchodilators to infants with asthma. BMJ. 1989 Jan 21;298(6667):160–161. doi: 10.1136/bmj.298.6667.160. [DOI] [PMC free article] [PubMed] [Google Scholar]