INTRODUCTION
It has been reported that renin-angiotensin system exists in tissue[1] and aldosterone can be synthesized in extra-adrenal tissue including heart, blood vessels[2] and brain[3]. Recent studies have brought rich evidences in favour of aldosterone as a strong stimulator of fibrogenesis and mitogenesis[4-9]. Recently Wu PS reported that aldosterone synthase gene CYP11B2 can be expressed in hepatic stellate cells (HSCs) of liver[10]. As the activation of HSCs is the central event in fibrogenesis of liver[11-26], we undertook the present study to investigate the relationship between hepatic fibrogenesis and locally produced aldosterone in liver.
MATERIALS AND METHODS
The establishment of animal model
Male Wistar rats (250 g-280 g, n = 48, purchased from Animal Center of the First Military Medical University) were divided into 2 groups randomly. Model group (n = 24): the rats were injected with 40% CCl4 (the mixture of CCl4 and olive oil) 0.25 mL/100 g subcutaneously three times a week. Control group (n = 24): the rats were injected with olive oil only.
Histology
At the end of the week 4, 6, 8, and 10, 6 rats in each group were sacrificed. The rat liver was regularly fixed, embedded, sliced and stained with VG and HE. Cryosections of liver tissue were rinsed in 0.2 g/L gold chloride solution for 6 h at room temperature, then the sections were placed in 50 g/L sodium thiosulphate for 5 min and mounted with resin.
Extraction of total RNA
The tissue of liver was promptly frozen in liquid nitrogen and stored at -70 °C prior to use. Total RNA was extracted with GTC solution (6 M guanidinium thiocyanate, 5 mM sodium citrate, 5 g/L sodium sarcosyl, 0.1 M beta. mercaptoethanol). The amount of total RNA was measured at 260 nm using an ultraviolet spectrophotometer.
RT-PCR for CYP11B2 mRNA
One microgram of total RNA was incubated at 25 °C for 30 min in 20 μL reverse-transcriptase buffer containing 20 U AMV reverse transcriptase and 2 μL random primerp (dN)6 (Boehringer Mannheim, Germany). Reverse transcription was terminated by heating at 99 °C for 5 min. The polymerase chain reactions were performed in 50 μL PCR buffer containing 0.32 μg of each primer and 2.5 U Taq DNA polymerase (Promega). Samples were subjected to 30 cycles of PCR amplification. Each cycle includes denaturation at 94 °C for 1 min, annealing at 56 °C for 1 min, and primer extension at 72 °C for 2 min. The rat CYP11B2 PCR primers were 5’-ACCATGGATGTCCAGCAA-3’ and 5’-GAGAGCTGCCGAGTCTGA-3’, synthesized by Shanghai Cell Institute according to the published sequences of Oaks and Raff[27], corresponding to positions 657-954 of the gene that does not cross-react with the CYP11B1 gene. As control, β-actin primers were used and had the following sequences: 5’-TTTCTGGCAAGTTAGGTTTTGTCAA-3’ and 5’-CCTAGCACCATGAAGATCAA-3’ [28]. Each 6 μL amplification mixture was subjected to electrophoresis on 15 g/L agarose gel, and DNA was visualized by ethidium bromide staining. The signal intensity was quantified by a computerized medical image-processing system (GDS-7500, UVP, England). Six separate gels were run for the enzyme, and the data were then averaged. The ratio of CYP11B2 to β-actin was used to express relative mRNA levels.
In situ hybridization
The Wistar rats (model group, 6th week, n = 6; control group, n = 6) were anesthetized by intraperitoneal injection of pentobarbital, and perfused with 9 g/L NaCl and 40 g/L paraformaldehyde solution buffer at pH7.4 in 0.1 M phosphate buffered saline (PBS). Each specimen was divided into halves, immersed in the same fixative solution for 4 h at 4 °C. After the tissue samples were rinsed in PBS containing 300 g/L sucrose, cryosections 10 μm thick were cut and amounted on polylysine-coated slides. In situ hybridization[29], slides were washed in 0.1 M PBS, treated with 3 g/L triton X-100 for 20 min, washed in 0.1 M glycine for 5 min; and incubated in 2 μg/mL proteinase K at 37 °C for 30 min, fixed in buffered 40 g/L paraformaldehyde at RT for 10 min, washed in PBS for 20 min; immersed in 2.5 g/L acetic anhydride in 0.1 M triethanolamine solution at RT for 10 min, washed in 2 × SSC for 15 min; incubated in a prehybridization mixture at RT for 8 h. After blotting the solution, the slides were incubated in a hybridization solution consisting of 500 g/L formamide, 5 × SSC, 20 g/L SDS, 1 × Denhardt’s solution, 100 mg/L salmon sperm DNA, 100 g/L dextran sulfate, 2 mg/L Digoxigenin labeling probe (Boehringer Mannheim, Germany) at 42 °C for 24 h. After hybridization, the slides were washed in 4 × SSC, 2 × SSC, 1 × SSC, 0.5 × SSC and 0.05 M PBS at 37 °C for 20 min respectively. The detection procedure was performed according to the maneuver of DIG DNA labeling and detection kit (Boehringer Mannheim, Germany). The slide exposure duration was 12 h at 4 °C. The slides were dehydrated in ethanol and mounted with resin. A negative control was prepared for each sample using a hybridization solution without CYP11B2 probe. We selected 7 high power fields randomly and counted the positive cells per high power field.
Statistics
Analysis of data was performed with one way ANOVA (SPSS 7.5) and rank sum test. Results were expressed as mean ± SD, a value of P < 0.05 was regarded as statistical significance.
RESULTS
Morphological changes
Observed with naked eyes, at the end of week 4, the liver surface of model group presented tiny particle-like changes. After that, the tissue became hard and shrank progressively.
Microscopically, at the end of week 4, fibroblasts proliferated obviously in the portal tracts of model group, and collagen invaded into the hepatic lobules along with the injured limiting laminae, but the collagen was not completely connected with each other. At the end of week 6, most rats developed cirrhosis in model group.
RT-PCR
The expressions of CYP11B2 mRNA in model group were significantly up-regulated compared to those in control group (P < 0.01). The levels of CYP11B2 mRNA in model group (week 8) were higher than those in model group (week 4) (P < 0.05), without significant difference between the two groups (P > 0.05) (Figure 1, Figure 2).
Figure 1.
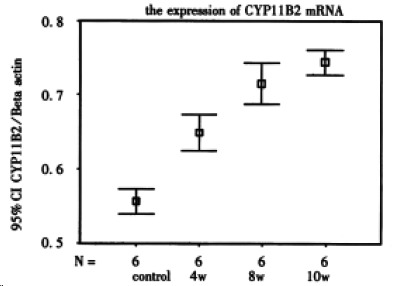
The ratio of CYP11B2/β-action presents the relative level of CYPP11B2.
Figure 2.
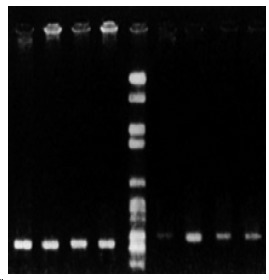
Lanes 1: model group (week 4) β-actin; Lanes 9: model group (week 4) CYP11B2; Lanes 2: model group (week 8) β-actin; Lanes 8: model group (week 8) CYP11B2; Lanes 7: model group (week 10) CYP11B2; Lanes 4: control group β actin; Lanes 6: control group CYP11B2; Lanes 5: Makers
In situ hybridization
Duck purple precipitate was localized in the endoplasm of HSCs corresponding to the gold chloride stained section. The number of positive cells per high power field in model group (4.5) was higher than that in control group (0.55) (P < 0.05).
DISCUSSION
It has been reported that renin-angiotensin system exists in tissues. Aldosterone can be synthesized in extra-adrenal tissue including blood vessels and brain. Aldosterone is a strong stimulator of fibrogenesis and mitogenesis which exerts a marked effect on stimulating the proliferation of myofibroblasts and collagen production.
There are two genes encoding the enzymes of aldosterone biosynthesis[30,31]. One is P450 11β (CYP11B1) that is responsible for the early steps of aldosterone biosynthesis. The other is P450 aldo (CYP11B2) that encodes the key enzyme for the final steps of its biosynthesis.
HSCs are mesenchymal cells located in the space of Disse. They demonstrated synthetic activity of collagen and other extracellular matrix proteins involved in hepatic fibrosis. As a source of cytokines, prostaglandins and other bioactive substances, they play a crucial role in the mechanisms of liver injury, regeneration and fibrosis. Now, fat-storing cell function is expanding from a retinol (fat)-storing site to a center of extracellular matrix metabolism and mediator production in the liver. Transition of the stellate cells from the vitamin A-storing phenotype to “activated” or “myofibroblastic” cells is the central pathobiochemical event for liver fibrosis[32-36].
Using RT-PCR, we clearly provided direct evidence for the expression of CYP11B2 mRNA in liver. The expression was up-regulated when fibrogenesis occurred. In situ hybridization further revealed that the expression of CYP11B2 mRNA located in the endoplasm of HSCs increased in fibrotic liver.
In conclusion, based on present study, we first proposed that there is a positive acting relationship between the expression of CYP11B2 in HSCs and fibrogenesis of liver. Locally produced aldosterone in liver is likely to promote the process of fibrogenesis. The present study provides a new way to investigate the mechanism and treatment of hepatic fibrosis.
ACKNOWLEDGEMENTS
The author thanks Dr. MichiyoI from Keio University, Japan for kindly supplying the rat CYP11B2 cDNA.
Footnotes
Edited by You DY and Ma JY
Supported by the National Natural Science Foundation of China, No.39870331.
References
- 1.Lindpaintner K, Ganten D. The cardiac renin-angiotensin system. An appraisal of present experimental and clinical evidence. Circ Res. 1991;68:905–921. doi: 10.1161/01.res.68.4.905. [DOI] [PubMed] [Google Scholar]
- 2.Hatakeyama H, Miyamori I, Fujita T, Takeda Y, Takeda R, Yamamoto H. Vascular aldosterone. Biosynthesis and a link to angiotensin II-induced hypertrophy of vascular smooth muscle cells. J Biol Chem. 1994;269:24316–24320. [PubMed] [Google Scholar]
- 3.Gomez-Sanchez CE, Zhou MY, Cozza EN, Morita H, Foecking MF, Gomez-Sanchez EP. Aldosterone biosynthesis in the rat brain. Endocrinology. 1997;138:3369–3373. doi: 10.1210/endo.138.8.5326. [DOI] [PubMed] [Google Scholar]
- 4.Young M, Fullerton M, Dilley R, Funder J. Mineralocorticoids, hypertension, and cardiac fibrosis. J Clin Invest. 1994;93:2578–2583. doi: 10.1172/JCI117269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fullerton MJ, Funder JW. Aldosterone and cardiac fibrosis: in vitro studies. Cardiovasc Res. 1994;28:1863–1867. doi: 10.1093/cvr/28.12.1863. [DOI] [PubMed] [Google Scholar]
- 6.Robert V, Van Thiem N, Cheav SL, Mouas C, Swynghedauw B, Delcayre C. Increased cardiac types I and III collagen mRNAs in aldosterone-salt hypertension. Hypertension. 1994;24:30–36. doi: 10.1161/01.hyp.24.1.30. [DOI] [PubMed] [Google Scholar]
- 7.Weber KT, Brilla CG. Pathological hypertrophy and cardiac interstitium. Fibrosis and renin-angiotensin-aldosterone system. Circulation. 1991;83:1849–1865. doi: 10.1161/01.cir.83.6.1849. [DOI] [PubMed] [Google Scholar]
- 8.Morgan HE, Baker KM. Cardiac hypertrophy. Mechanical, neural, and endocrine dependence. Circulation. 1991;83:13–25. doi: 10.1161/01.cir.83.1.13. [DOI] [PubMed] [Google Scholar]
- 9.Rossi GP, Sacchetto A, Pavan E, Palatini P, Graniero GR, Canali C, Pessina AC. Remodeling of the left ventricle in primary aldosteronism due to Conn's adenoma. Circulation. 1997;95:1471–1478. doi: 10.1161/01.cir.95.6.1471. [DOI] [PubMed] [Google Scholar]
- 10.Wu PS, Liang XW, Dai Y, Liu H, Zhang Y, Guo ZG, Zhang RH, Lai WY, Zhang YH, Liu YL. Aldosterone biosythesis in extradrenal tissues. Zhonghua Xinxueguan Zazhi. 1998;26:139–141. [Google Scholar]
- 11.Minato Y, Hasumura Y, Takeuchi J. The role of fat-storing cells in Disse space fibrogenesis in alcoholic liver disease. Hepatology. 1983;3:559–566. doi: 10.1002/hep.1840030414. [DOI] [PubMed] [Google Scholar]
- 12.Hirata K, Ogata I, Ohta Y, Fujiwara K. Hepatic sinusoidal cell destruction in the development of intravascular coagulation in acute liver failure of rats. J Pathol. 1989;158:157–165. doi: 10.1002/path.1711580211. [DOI] [PubMed] [Google Scholar]
- 13.Burt AD. C. L. Oakley Lecture (1993). Cellular and molecular aspects of hepatic fibrosis. J Pathol. 1993;170:105–114. doi: 10.1002/path.1711700203. [DOI] [PubMed] [Google Scholar]
- 14.Geerts A, Vrijsen R, Rauterberg J, Burt A, Schellinck P, Wisse E. In vitro differentiation of fat-storing cells parallels marked increase of collagen synthesis and secretion. J Hepatol. 1989;9:59–68. doi: 10.1016/0168-8278(89)90076-7. [DOI] [PubMed] [Google Scholar]
- 15.Weiner FR, Giambrone MA, Czaja MJ, Shah A, Annoni G, Takahashi S, Eghbali M, Zern MA. Ito-cell gene expression and collagen regulation. Hepatology. 1990;11:111–117. doi: 10.1002/hep.1840110119. [DOI] [PubMed] [Google Scholar]
- 16.Bhunchet E, Fujieda K. Capillarization and venularization of hepatic sinusoids in porcine serum-induced rat liver fibrosis: a mechanism to maintain liver blood flow. Hepatology. 1993;18:1450–1458. [PubMed] [Google Scholar]
- 17.Tsutsumi M, Takada A, Takase S. Characterization of desmin-positive rat liver sinusoidal cells. Hepatology. 1987;7:277–284. doi: 10.1002/hep.1840070212. [DOI] [PubMed] [Google Scholar]
- 18.Kent G, Gay S, Inouye T, Bahu R, Minick OT, Popper H. Vitamin A-containing lipocytes and formation of type III collagen in liver injury. Proc Natl Acad Sci USA. 1976;73:3719–3722. doi: 10.1073/pnas.73.10.3719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Al Adnani MS. Differential immunohistochemical localization of cytokeratins and collagen types I and III in experimentally-induced cirrhosis. J Pathol. 1989;159:151–158. doi: 10.1002/path.1711590210. [DOI] [PubMed] [Google Scholar]
- 20.Takahara T, Nakayama Y, Itoh H, Miyabayashi C, Watanabe A, Sasaki H, Inoue K, Muragaki Y, Ooshima A. Extracellular matrix formation in piecemeal necrosis: immunoelectron microscopic study. Liver. 1992;12:368–380. doi: 10.1111/j.1600-0676.1992.tb00591.x. [DOI] [PubMed] [Google Scholar]
- 21.Knittel T, Schuppan D, Meyer zum Büschenfelde KH, Ramadori G. Differential expression of collagen types I, III, and IV by fat-storing (Ito) cells in vitro. Gastroenterology. 1992;102:1724–1735. doi: 10.1016/0016-5085(92)91736-n. [DOI] [PubMed] [Google Scholar]
- 22.Rojkind M, Novikoff PM, Greenwel P, Rubin J, Rojas-Valencia L, de Carvalho AC, Stockert R, Spray D, Hertzberg EL, Wolkoff AW. Characterization and functional studies on rat liver fat-storing cell line and freshly isolated hepatocyte coculture system. Am J Pathol. 1995;146:1508–1520. [PMC free article] [PubMed] [Google Scholar]
- 23.Wang YJ, Sun ZQ, Quan QZ, Yu JJ. Fat storing cells and liver fibrosis. China Natl J New Gastroenterol. 1996;2:58–60. [Google Scholar]
- 24.Huang ZG, Zhai WR, Zhang YE, Zhang XR. Study of heteroserum-induced rat liver fibrosis model and its mechanism. World J Gastroenterol. 1998;4:206–209. doi: 10.3748/wjg.v4.i3.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Du WD, Zhang YE, Zhai WR, Zhou XM. Dynamic changes of type I, III and IV collagen synthesis and distribution of collagen-producing cells in carbon tetrachloride-induced rat liver fibrosis. World J Gastroenterol. 1999;5:397–403. doi: 10.3748/wjg.v5.i5.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wu CH. Fibrodynamics-elucidation of the mechanisms and sites of liver fibrogenesis. World J Gastroenterol. 1999;5:388–390. doi: 10.3748/wjg.v5.i5.388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Oaks MK, Raff H. Differentiation of the expression of aldosterone synthase and 11 beta-hydroxylase mRNA in the rat adrenal cortex by reverse transcriptase-polymerase chain reaction. J Steroid Biochem Mol Biol. 1995;54:193–199. doi: 10.1016/0960-0760(95)00143-n. [DOI] [PubMed] [Google Scholar]
- 28.Nishimura J, Chen X, Jahan H, Shikasho T, Kobayashi S, Kanaide H. cAMP induces up-regulation of ETA receptor mRNA and increases responsiveness to endothelin-1 of rat aortic smooth muscle cells in primary culture. Biochem Biophys Res Commun. 1992;188:719–726. doi: 10.1016/0006-291x(92)91115-7. [DOI] [PubMed] [Google Scholar]
- 29.Yabu M, Senda T, Nonaka Y, Matsukawa N, Okamoto M, Fujita H. Localization of the gene transcripts of 11 beta-hydroxylase and aldosterone synthase in the rat adrenal cortex by in situ hybridization. Histochemistry. 1991;96:391–394. doi: 10.1007/BF00315995. [DOI] [PubMed] [Google Scholar]
- 30.Mellon SH, Bair SR, Monis H. P450c11B3 mRNA, transcribed from a third P450c11 gene, is expressed in a tissue-specific, developmentally, and hormonally regulated fashion in the rodent adrenal and encodes a protein with both 11-hydroxylase and 18-hydroxylase activities. J Biol Chem. 1995;270:1643–1649. doi: 10.1074/jbc.270.4.1643. [DOI] [PubMed] [Google Scholar]
- 31.Tremblay A, Parker KL, Lehoux JG. Dietary potassium supplementation and sodium restriction stimulate aldosterone synthase but not 11 beta-hydroxylase P-450 messenger ribonucleic acid accumulation in rat adrenals and require angiotensin II production. Endocrinology. 1992;130:3152–3158. doi: 10.1210/endo.130.6.1597135. [DOI] [PubMed] [Google Scholar]
- 32.Okanoue T, Burbige EJ, French SW. The role of the Ito cell in perivenular and intralobular fibrosis in alcoholic hepatitis. Arch Pathol Lab Med. 1983;107:459–463. [PubMed] [Google Scholar]
- 33.Mak KM, Leo MA, Lieber CS. Alcoholic liver injury in baboons: transformation of lipocytes to transitional cells. Gastroenterology. 1984;87:188–200. [PubMed] [Google Scholar]
- 34.Maher JJ. Fat-storing cells and myofibroblasts: one cell or two. Hepatology. 1989;9:903–904. doi: 10.1002/hep.1840090621. [DOI] [PubMed] [Google Scholar]
- 35.Gao ZL, Li DG, Lu HM, Gu XH. The effect of retinoic acid on Ito cell proliferation and content of DNA and RNA. World J Gastroenterol. 1999;5:443–444. doi: 10.3748/wjg.v5.i5.443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu CH, Liu C, Liu P, Xu LM. Seropharmalogical effects of Fuzheng Huayu decoction on rat Ito cell morphology and functions in culture. China Natl J New Gastroenterol. 1997;3:263–265. doi: 10.3748/wjg.v3.i4.263. [DOI] [PMC free article] [PubMed] [Google Scholar]


