Abstract
Na+/K+-ATPase (NKA) is abundantly expressed in the basolateral membrane of epithelial cells, which is necessary for tight junction formation. The tight junction is an urothelial barrier between urine and the underlying bladder. Impairment of tight junctions allows migration of urinary solutes in patients with interstitial cystitis/painful bladder syndrome (IC/PBS). We evaluated NKA expression and activity in bladder samples from patients with IC/PBS. The study group consisted of 85 patients with IC/PBS, and the control group consisted of 20 volunteers. Bladder biopsies were taken from both groups. We determined the expression and distribution of NKA using NKA activity assays, immunoblotting, immunohistochemical staining, and immunofluorescent staining. The protein levels and activity of NKA in the study group were significantly lower than the control group (1.08 ± 0.06 vs. 2.39 ± 0.29 and 0.60 ± 0.04 vs. 1.81 ± 0.18 µmol ADP/mg protein/hour, respectively; P < 0.05). Additionally, immunofluorescent staining for detection of CK7, a marker of the bladder urothelium, predominantly colocalized with NKA in patients in the study group. Our results demonstrated the expression and activity of NKA were decreased in bladder biopsies of patients with IC/PBS. These findings suggest that NKA function is impaired in the bladders from patients with IC/PBS.
Keywords: Na+/K+-ATPase, Bladder Urothelium, Interstitial Cystitis, Painful Bladder Syndrome
INTRODUCTION
Na+/K+-ATPase (NKA; sodium-potassium pump) is abundantly expressed in the basolateral membrane of epithelial cells and maintains cell homeostasis by transporting three Na+ molecules out and two K+ molecules into a cell, using the energy from the hydrolysis of adenosine triphosphate (ATP) (1,2). NKA is the primary active transporter that produces the driving force for the activation of other secondary ion transporters on cell membranes (1,2). The function of NKA controls a variety of ion and metabolite transport systems. Moreover, this pump alone accounts for approximately 30% of ATP consumption in animal cells for the purposes of ion regulation to maintain intracellular homeostasis (3,4).
NKA is also a potential regulator of tight junction formation and function (5,6,7). Tight junctions function as urothelial barriers between urine and the underlying bladder (8,9). They also play a critical role in the biogenesis of polarized epithelial cells during early vertebrate development and during recovery from tissue damage after ischemic or toxic injury (2,10). Previous studies have reported that tight junction protein expression is decreased in the bladder urothelium (11,12,13) and that increased potassium leakage causes clinical symptoms in patients with interstitial cystitis/painful bladder syndrome (IC/PBS) (14). Therefore, studies of changes in NKA function in bladder tissues may be important for development of appropriate therapeutic strategies in patients with bladder disease.
Here, we aimed to investigate changes in NKA expression and activity in bladder biopsies from patients with IC/PBS. To the best of our knowledge, this is the first study to investigate changes in NKA function in patients with this bladder disease.
MATERIALS AND METHODS
Patients and tissue samples
Bladder specimens were obtained from 85 patients (78 women and 7 men) with IC/PBS who were undergoing cystoscopy under anesthesia for diagnosis of IC/PBS or for therapeutic bladder hydrodistention. All IC/PBS patients were defined with the cystoscopic criteria of National Institute of Diabetes, Digestive, and Kidney Diseases (15) and had experienced clinical symptoms for more than 6 months. The average age of patients in the cohort was 41.2 ± 9.4 years. Patient backgrounds, including clinical symptoms and past therapeutic history, are shown in Table 1. All patients underwent medical history collection, physical examination, urinalysis, and culture for exclusion screening and were required to stop the use of intravesical instillation drugs for at least 1 month before biopsy. IC/PBS symptoms were evaluated by questionnaires assessing O'Leary-Sant symptoms, problem indexes, and pelvic pain and urgency/frequency (PUF) (16). In the study group, a cold-cup biopsy was taken at sites demonstrating glomerulation after hydrodistension. Control specimens were obtained from 20 patients (median age: 42.7 years, range: 38–58 years; 18 women and 2 men who had undergone transurethral resection of the prostate more than 1 year prior) undergoing bladder neck suspension for stress urinary incontinence who had no symptoms of IC/PBS and showed no evidence of bladder mucosal disease after hydrodistension under cystoscopy. Hydrodistension was performed using normal saline for infusion fluid under gravity (100 cm pressure) until complete. The distension time was started, and metal compression was applied to prevent leakage for 2 minutes (17). All tissues were stored at -80°C for immunoblotting and NKA activity assays or fixed in 10% formalin for immunostaining. All specimens were collected after obtaining informed written consent from patients.
Table 1. Demographic and clinicopathological data for patients with interstitial cystitis/painful bladder syndrome.
| Parameters | Values |
|---|---|
| Age (yr) | 41.22 ± 9.37 |
| Age of first symptom presentation (yr) | 32.82 ± 9.14 |
| Frequency of voiding/day | 14.15 ± 10.7 |
| Voiding amount, mL/time | 120.86 ± 69.06 |
| Number of nocturnal frequency | 3.39 ± 2.65 |
| PUF (Pain and urgency/frequency) score | 20.78 ± 6.72 |
| Pain index | 4.67 ± 3.48 |
| O'Leary-Sant symptom index | 12.33 ± 2.43 |
| O'Leary-Sant problem index | 9.94 ± 3.59 |
| History of prior therapy | |
| Oral medication | 100% |
| Intravesical instillation | 34.5% |
| Botulinum toxin A injection | Excluded |
Antibodies
Four primary antibodies were used in this study as follows. Mouse monoclonal anti-NKA antibodies (α5; Developmental Studies Hybridoma Bank, Iowa City, IA, USA) were used for immunoblotting (dilution 1:200) and immunohistochemical (IHC) staining (dilution 1:200). Rabbit polyclonal anti-NKA antibodies (EP1845Y; Abcam, Cambridge, MA, USA) were used for immunofluorescence (IF) staining (dilution 1:50) to detect all α-isoforms of NKA. Additionally, mouse monoclonal anti-cytokeratin 7 (CK7) antibodies (NCL-L-CK7-OVTL; Novocastra, Milton Keynes, UK) was used for IF staining (dilution 1:50) to identify bladder uroepithelium (18,19). Rabbit polyclonal anti-actin antibodies (sc-1616-R; Santa Cruz Biotechnology, Santa Cruz, CA, USA) raised against the C-terminus of human ACTB were used as a loading control for immunoblotting (dilution 1:5,000).
The secondary antibodies for immunoblotting were horseradish peroxidase (HRP)-conjugated goat anti-mouse IgG or goat anti-rabbit IgG (#0031430 or #0031460, respectively; Pierce, Hercules, CA, USA). The secondary antibodies used for immunostaining included a commercial kit (PicTure; Zymed, South San Francisco, CA, USA) for IHC staining and Alexa Fluor 488-conjugated goat anti-mouse or Alexa Fluor 546-conjugated goat anti-rabbit antibodies (Molecular Probes, Eugene, OR, USA; dilution 1:50 and 1:200, respectively) for IF staining.
Preparation of tissue homogenates
Each sample was mixed with 200 µL SEID buffer (300 mM sucrose, 20 mM EDTA, 100 mM imidazole, 0.1% sodium deoxycholate, pH 7.5) containing proteinase inhibitor (#11836145001; Roche, Indianapolis, IN, USA; v/v: 25/1) and then homogenized on ice. The homogenate was centrifuged at 5,000 × g at 4°C for 5 minutes. The supernatants were collected and used for immunoblotting, NKA activity assays, or protein concentration analysis. Protein concentrations were determined using a BCA protein assay kit (#23225; Pierce) with bovine serum albumin (#23209; Pierce) as a standard.
Immunoblotting
The method for immunoblotting was modified from our previous studies (18,20). In brief, aliquots containing 50 µg of homogenates prepared as stated above were added to the sample buffer and heated to 60°C for 15 minutes. After electrophoresis and transformation, the blots were incubated sequentially in blocking buffer, primary antibodies (α5 or actin), and secondary antibodies. Finally, protein bands were detected using a SuperSignal West Pico Detection Kit (#34082; Pierce) and imaged using a cooling-CCD image sensor (ChemiDoc XRS+; Bio-Rad, Hercules, CA, USA) with specialized software (Quantity One version 4.6.8; Bio-Rad). Immunoreactive bands were analyzed using Image Lab software version 3.0 (Bio-Rad). The results were converted to numerical values in order to compare the relative protein abundance of the immunoreactive bands.
NKA activity assay
Enzyme activity was measured using the NADH-linked method. ADP derived from the hydrolysis of ATP by ATPase was enzymatically coupled to the oxidation of reduced NADH using lactate dehydrogenase (LDH) and pyruvate kinase (PK). In this study, the method for assaying NKA activity was modified from previous studies (21,22). The assay reaction was started by adding 10 µL of sample supernatant and 200 µL of reaction mixture (0.38 mM ATP, 1.50 mM phosphoenolpyruvate, 0.24 mM NADH, 2.48 U/mL LDH, 2.70 U/mL: PK, 47.3 mM NaCl, 2.63 mM MgCl2·6H2O, 10.5 mM KCl, 50 mM imidazole, pH 7.5) with or without 0.75 mM of ouabain (NKA inhibitor) to each well. The samples were examined every 15 seconds for up to 20 minutes using a VERSAmax microplate reader (Molecular Devices, Sunnyvale, CA, USA) at 340 nm and 37°C. The linear rate from 5 to 20 minutes for each pair of triplicate wells was determined. NKA activity was calculated as the difference in the slope of ATP hydrolysis (NADH reduction) in the presence or absence of ouabain, with activity expressed as µmol ADP per µg protein per hour.
IHC staining
The staining and microscopy methods were modified from our previous studies (19,23). The formalin-fixed, paraffin-embedded sections (4 µm) of biopsy samples were dewaxed and then rinsed with phosphate-buffered saline. Endogenous peroxidase was inactivated by incubating the sections with 3% hydrogen peroxide. The sections were stained with anti-NKA (α5) or phosphate-buffered saline (as a negative control) before analysis using a commercial kit. Sections were observed by optic microscopy (BX50; Olympus, Tokyo, Japan) with cooling CCD (DP72; Olympus) using CellSens software (CellSens standard version 1.4; Olympus).
Multiple IF staining
The method for IF staining was modified from our previous studies (18,20). In brief, paraffin-embedded sections were incubated at 4°C overnight with diluted primary antibodies (α5 or CK7) and then exposed to the appropriate secondary antibodies for 1 h. Finally, the sections were covered with a coverslip using mounting solution (DAPI Fluoromount G, #0100-20; SouthernBiotech, Birmingham, AL, USA). To identify and compare the localization of specific proteins, IF-stained samples were observed with a fluorescent microscope (BX50; Olympus) with cooling CCD (DP72; Olympus) using CellSens software (CellSens standard version 1.4; Olympus). Micrographs were subsequently merged to visualize differently colored labels simultaneously.
Statistical analysis
Differences between the groups were analyzed using Mann-Whitney U tests, and all values are expressed as the mean ± standard error (SE). Differences with P values of less than 0.05 were considered significant. The analyses were performed with SPSS 12.0 software (SPSS, Chicago, IL, USA).
Ethics statement
This study was approved by the institutional review board of Tri-Service General Hospital (approval No.TC100-11). Informed consent was exempted by the board.
RESULTS
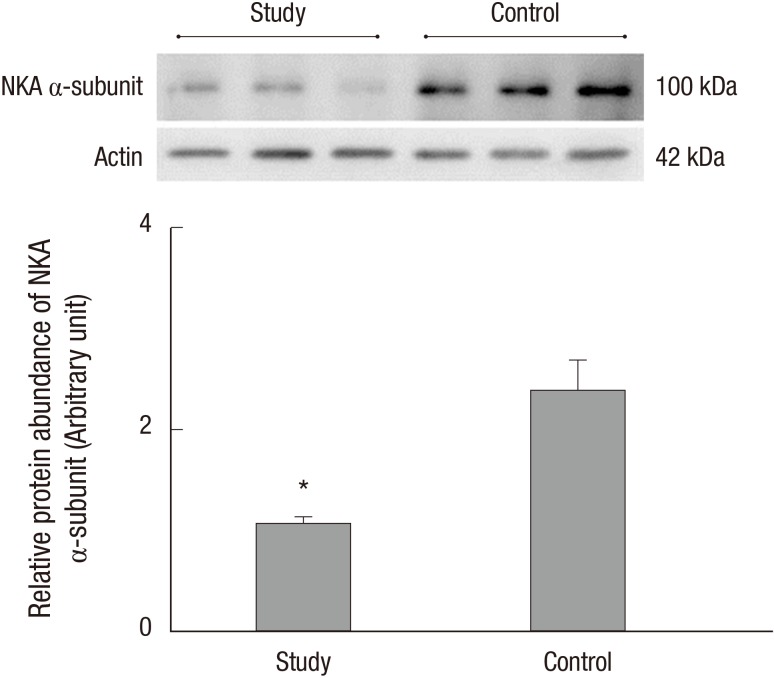
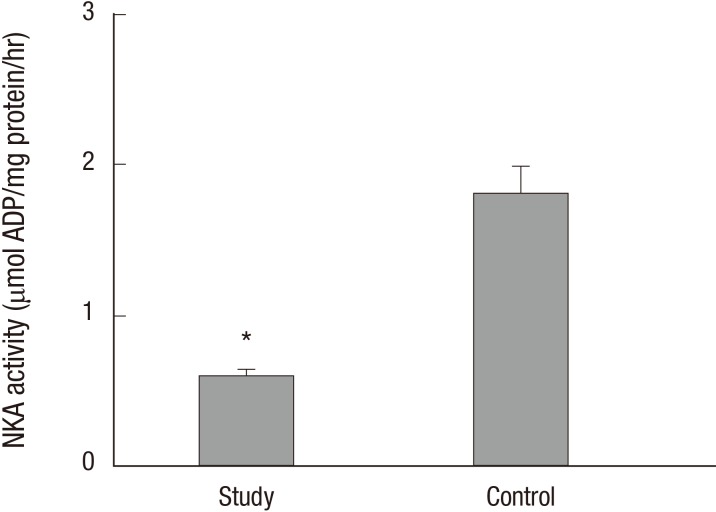
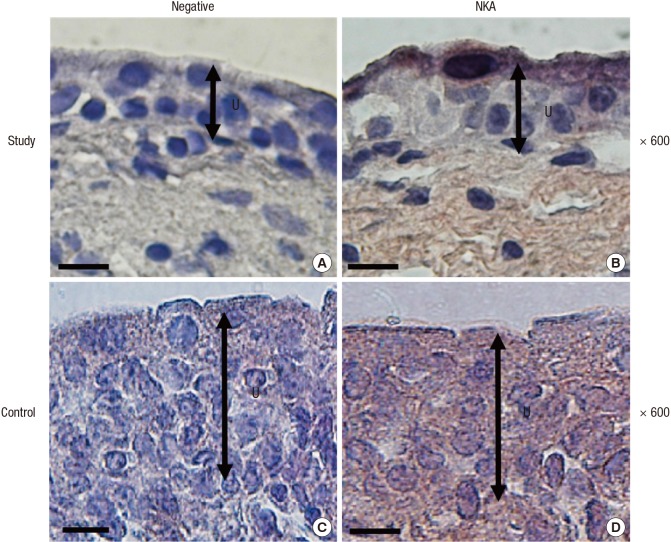
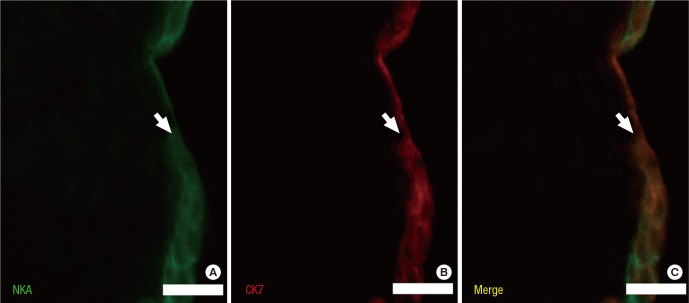
The protein levels (1.08 ± 0.06 vs. 2.39 ± 0.29, respectively) and activity (0.60 ± 0.04 vs. 1.81 ± 0.18 µmol ADP/mg protein/hour, respectively) of NKA were significantly lower in the study group (about 45% and 1/3-fold, respectively) than in the control group (P < 0.05; Fig. 1 and 2). Additionally, the bladder urothelium was thinner in patients with IC/PBS (Fig. 3A) compared with patients in the control group (Fig. 3C), and IHC staining for NKA (red) showed weaker immunoreactivity in the study group (Fig. 3B) than in the control group (Fig. 3D). Furthermore, multiple IF staining for NKA (green) and CK7 (uroepithelial marker, red) demonstrated that these two targets colocalized in the bladder urothelium of patients with IC/PBS (Fig. 4). These staining results revealed that NKA was predominantly expressed in the bladder urothelium (Fig. 3 and 4). Taken together, these data showed that the expression and activity of NKA were lower in bladders of patients with IC/PBS than in control individuals.
Fig. 1. Representative immunoblot and relative intensities of the Na+/K+-ATPase (NKA) α-subunit in patients in the study and control groups. Actin was used as the loading control. Values are the mean ± SE. The asterisk indicates a significant difference compared with the control group.
Fig. 2. Na+/K+-ATPase (NKA) activity in the study and control groups. Values are the mean ± SE. The asterisk indicates a significant difference compared with the control group.
Fig. 3. Representative tissue sections from patients in the study (A, B) and control (C, D) groups after negative control (A, C) or Na+/K+-ATPase (NKA) immunostaining (B, D). Magnification: 600 ×. The black double-headed arrow indicates the urothelium. Scale bar = 20 µm.
Fig. 4. Representative tissue sections from patients in the study group after multiple IF staining for Na+/K+-ATPase (NKA, green; A), CK7 (uroepithelial marker, red; B) and merged image (C). The arrow indicates the urothelium. Scale bar = 20 µm.
DISCUSSION
NKA is expressed in the basolateral plasma membrane of most epithelial cells (1,2) and plays a key role in maintaining cell homeostasis in the epithelium of organs and tissues, including the bladder (2,5). In the present study, our results showed that NKA was predominantly expressed in the bladder urothelium of both the study and control groups, similar to the results of other mammalian studies (24,25,26). Moreover, the expression and enzyme activity of NKA were decreased in patients with IC/PBS compared with those in the control group. We found no evidence that this pump compensated for potassium leakage in the study group. Thus, these results implied that bladder urothelial NKA was dysfunctional in patients with IC/PBS.
Rajasekaran et al. reported that NKA activity is necessary for tight junction formation in epithelial cells (5,7). The downregulation of tight junction proteins, such as zonula occludens-1, occludin, and E-cadherin, in the bladder urothelium in patients with IC/PBS has been reported previously (19,27). Moreover, inhibition of NKA ion transport function increases the permeability of tight junctions to ionic and nonionic solutes (28,29). The increased permeability and decreased tight junction formation in bladder urothelial cells was confirmed in patients with IC/PBS (11) and was shown to permit the migration of urinary solutes (8,12,14). In particular, high potassium levels in urine (i.e., 24–133 mEq/L) (8,14) could easily lead to leakage into interstitial tissue through dysfunctional bladder urothelium, causing depolarization of nerves and detrusor contractions and resulting in bladder pain, urgency, and increased frequency of urination in patient with IC/PBS (30,31,32).
Previous studies have reported that NKA is a signaling molecule involved in multiple cell signaling pathways. Inhibition of NKA function activates certain signaling, including Ras, phosphoinositol 3 kinase, mitogen-activated protein kinase (MAPK) and apoptotic pathway (7,33). Higher levels of bladder urothelial apoptosis have been observed in patients with IC/PBS (18,27). Therefore, dysfunction of bladder urothelial NKA may contribute to the observed increase in apoptosis.
In addition, NKA requires direct coupling with metabolic energy (i.e., ATP) (1,2). Our previous study showed that hypoxia-inducible factor-1α expression is increased in the bladder tissues of patients with IC/PBS (20), indicating that hypoxia occurring in these tissues may cause lower energy (ATP) production and impair NKA function (34,35,36). Taken together, these data suggested that decreased NKA expression and activity may be associated with tissues hypoxia (20,23), which could decrease tight junction formation (19) and increase apoptosis (18). These events would result in the formation of thinner, dysfunctional bladder urothelium in patients with IC/PBS.
There were several potential limitations to this study. First, patients' clinical symptoms and disease duration may have been subjected to selection bias. Second, there are still no uniform standard criteria and definitions with which to diagnose this disease. Third, the proposed relationships among hypoxia, apoptosis, tight junctions, and NKA are based on the results of our previous studies (18,19,20,23).
In conclusion, we found that the expression and activity of NKA were decreased in bladder biopsies from patients with IC/PBS and that NKA was predominantly localized on the bladder urothelium. These findings suggest that impaired NKA function in the bladder urothelium may contribute to the diseased state in patients with IC/PBS.
ACKNOWLEDGMENT
The monoclonal antibody against the Na+/K+-ATPase α-subunit (α5) was purchased from the Developmental Studies Hybridoma Bank maintained by the Department of Pharmacology and Molecular Sciences, Johns Hopkins University School of Medicine, Baltimore, MD, USA and the Department of Biological Sciences, University of Iowa, Iowa City, IA, USA under contract N01-HD-6-2915 (NICHD, USA).
Footnotes
Funding: This study was supported by a grant (grant No. 101-10) from Taichung Armed Forces General Hospital, Taiwan.
DISCLOSURE: None of the contributing authors has any conflicts of interest, including specific financial interests or relationships and affiliations relevant to the subject matter or materials discussed in the manuscript.
AUTHOR CONTRIBUTION: Conception and coordination of the study: Lee JD, Lee MH, Yang WK. Consultant: Lee MH.Data acquisition and analysis: Yang WK. Writing: Lee JD. Review and revision: Lee JD, Yang WK. Manuscript approval: all authors.
References
- 1.Brinson BE. Lactate dehydrogenase and Na+/K+ ATPase activity in Leiostomus xanthurus (spot) in response to hypoxia. Explorations. 2011;4:22–29. [Google Scholar]
- 2.Rajasekaran AK, Rajasekaran SA. Role of Na-K-ATPase in the assembly of tight junctions. Am J Physiol Renal Physiol. 2003;285:F388–F396. doi: 10.1152/ajprenal.00439.2002. [DOI] [PubMed] [Google Scholar]
- 3.Comellas AP, Dada LA, Lecuona E, Pesce LM, Chandel NS, Quesada N, Budinger GR, Strous GJ, Ciechanover A, Sznajder JI. Hypoxia-mediated degradation of Na,K-ATPase via mitochondrial reactive oxygen species and the ubiquitin-conjugating system. Circ Res. 2006;98:1314–1322. doi: 10.1161/01.RES.0000222418.99976.1d. [DOI] [PubMed] [Google Scholar]
- 4.Dada LA, Chandel NS, Ridge KM, Pedemonte C, Bertorello AM, Sznajder JI. Hypoxia-induced endocytosis of Na,K-ATPase in alveolar epithelial cells is mediated by mitochondrial reactive oxygen species and PKC-zeta. J Clin Invest. 2003;111:1057–1064. doi: 10.1172/JCI16826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rajasekaran SA, Palmer LG, Moon SY, Peralta Soler A, Apodaca GL, Harper JF, Zheng Y, Rajasekaran AK. Na,K-ATPase activity is required for formation of tight junctions, desmosomes, and induction of polarity in epithelial cells. Mol Biol Cell. 2001;12:3717–3732. doi: 10.1091/mbc.12.12.3717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Violette MI, Madan P, Watson AJ. Na+/K+ -ATPase regulates tight junction formation and function during mouse preimplantation development. Dev Biol. 2006;289:406–419. doi: 10.1016/j.ydbio.2005.11.004. [DOI] [PubMed] [Google Scholar]
- 7.Rajasekaran SA, Rajasekaran AK. Na,K-ATPase and epithelial tight junctions. Front Biosci (Landmark Ed) 2009;14:2130–2148. doi: 10.2741/3367. [DOI] [PubMed] [Google Scholar]
- 8.Parsons CL. The role of the urinary epithelium in the pathogenesis of interstitial cystitis/prostatitis/urethritis. Urology. 2007;69(Suppl):9–16. doi: 10.1016/j.urology.2006.03.084. [DOI] [PubMed] [Google Scholar]
- 9.Dell JR, Mokrzycki ML, Jayne CJ. Differentiating interstitial cystitis from similar conditions commonly seen in gynecologic practice. Eur J Obstet Gynecol Reprod Biol. 2009;144:105–109. doi: 10.1016/j.ejogrb.2009.02.050. [DOI] [PubMed] [Google Scholar]
- 10.Fleming TP, Ghassemifar MR, Sheth B. Junctional complexes in the early mammalian embryo. Semin Reprod Med. 2000;18:185–194. doi: 10.1055/s-2000-12557. [DOI] [PubMed] [Google Scholar]
- 11.Zhang CO, Wang JY, Koch KR, Keay S. Regulation of tight junction proteins and bladder epithelial paracellular permeability by an antiproliferative factor from patients with interstitial cystitis. J Urol. 2005;174:2382–2387. doi: 10.1097/01.ju.0000180417.11976.99. [DOI] [PubMed] [Google Scholar]
- 12.Hurst RE, Moldwin RM, Mulholland SG. Bladder defense molecules, urothelial differentiation, urinary biomarkers, and interstitial cystitis. Urology. 2007;69:17–23. doi: 10.1016/j.urology.2006.03.083. [DOI] [PubMed] [Google Scholar]
- 13.Lavelle JP, Meyers SA, Ruiz WG, Buffington CA, Zeidel ML, Apodaca G. Urothelial pathophysiological changes in feline interstitial cystitis: a human model. Am J Physiol Renal Physiol. 2000;278:F540–F553. doi: 10.1152/ajprenal.2000.278.4.F540. [DOI] [PubMed] [Google Scholar]
- 14.Parsons CL. The role of a leaky epithelium and potassium in the generation of bladder symptoms in interstitial cystitis/overactive bladder, urethral syndrome, prostatitis and gynaecological chronic pelvic pain. BJU Int. 2011;107:370–375. doi: 10.1111/j.1464-410X.2010.09843.x. [DOI] [PubMed] [Google Scholar]
- 15.Gillenwater JY, Wein AJ. Summary of the National Institute of Arthritis, Diabetes, Digestive and Kidney Diseases Workshop on Interstitial Cystitis, National Institutes of Health, Bethesda, Maryland, August 28-29, 1987. J Urol. 1988;140:203–206. doi: 10.1016/s0022-5347(17)41529-1. [DOI] [PubMed] [Google Scholar]
- 16.Evans RJ, Sant GR. Current diagnosis of interstitial cystitis: an evolving paradigm. Urology. 2007;69:64–72. doi: 10.1016/j.urology.2006.05.048. [DOI] [PubMed] [Google Scholar]
- 17.Ottem DP, Teichman JM. What is the value of cystoscopy with hydrodistension for interstitial cystitis? Urology. 2005;66:494–499. doi: 10.1016/j.urology.2005.04.011. [DOI] [PubMed] [Google Scholar]
- 18.Lee JD, Lee MH. Activation of extrinsic apoptotic pathway from bladder biopsy in patients with interstitial cystitis/painful bladder syndrome. Urology. 2013;82:1451.e7–1451.11. doi: 10.1016/j.urology.2013.08.042. [DOI] [PubMed] [Google Scholar]
- 19.Lee JD, Lee MH. Decreased expression of zonula occludens-1 and occludin in the bladder urothelium of patients with interstitial cystitis/painful bladder syndrome. J Formos Med Assoc. 2014;113:17–22. doi: 10.1016/j.jfma.2012.03.010. [DOI] [PubMed] [Google Scholar]
- 20.Lee JD, Lee MH. Increased expression of hypoxia-inducible factor-1α and vascular endothelial growth factor associated with glomerulation formation in patients with interstitial cystitis. Urology. 2011;78:971.e11–971.e15. doi: 10.1016/j.urology.2011.05.050. [DOI] [PubMed] [Google Scholar]
- 21.Yang WK, Kang CK, Chen TY, Chang WB, Lee TH. Salinity-dependent expression of the branchial Na+/K +/2Cl- cotransporter and Na+/K+-ATPase in the sailfin molly correlates with hypoosmoregulatory endurance. J Comp Physiol B. 2011;181:953–964. doi: 10.1007/s00360-011-0568-0. [DOI] [PubMed] [Google Scholar]
- 22.Haghighi K, Pritchard T, Bossuyt J, Waggoner JR, Yuan Q, Fan GC, Osinska H, Anjak A, Rubinstein J, Robbins J, et al. The human phospholamban Arg14-deletion mutant localizes to plasma membrane and interacts with the Na/K-ATPase. J Mol Cell Cardiol. 2012;52:773–782. doi: 10.1016/j.yjmcc.2011.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee JD, Lee MH. Metallothionein overexpression of bladder biopsies associated with tissue hypoxia in patients with interstitial cystitis/painful bladder syndrome. Int J Urol. 2014;21:719–723. doi: 10.1111/iju.12402. [DOI] [PubMed] [Google Scholar]
- 24.Smith PR, Mackler SA, Weiser PC, Brooker DR, Ahn YJ, Harte BJ, McNulty KA, Kleyman TR. Expression and localization of epithelial sodium channel in mammalian urinary bladder. Am J Physiol. 1998;274:F91–F96. doi: 10.1152/ajprenal.1998.274.1.F91. [DOI] [PubMed] [Google Scholar]
- 25.Espineda C, Seligson DB, James Ball W, Jr, Rao J, Palotie A, Horvath S, Huang Y, Shi T, Rajasekaran AK. Analysis of the Na,K-ATPase alpha- and beta-subunit expression profiles of bladder cancer using tissue microarrays. Cancer. 2003;97:1859–1868. doi: 10.1002/cncr.11267. [DOI] [PubMed] [Google Scholar]
- 26.Spector DA, Deng J, Coleman R, Wade JB. The urothelium of a hibernator: the American black bear. Physiol Rep. 2015;3:e12429. doi: 10.14814/phy2.12429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Shie JH, Kuo HC. Higher levels of cell apoptosis and abnormal E-cadherin expression in the urothelium are associated with inflammation in patients with interstitial cystitis/painful bladder syndrome. BJU Int. 2011;108:E136–E141. doi: 10.1111/j.1464-410X.2010.09911.x. [DOI] [PubMed] [Google Scholar]
- 28.Rajasekaran SA, Barwe SP, Gopal J, Ryazantsev S, Schneeberger EE, Rajasekaran AK. Na-K-ATPase regulates tight junction permeability through occludin phosphorylation in pancreatic epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2007;292:G124–G133. doi: 10.1152/ajpgi.00297.2006. [DOI] [PubMed] [Google Scholar]
- 29.Rajasekaran SA, Hu J, Gopal J, Gallemore R, Ryazantsev S, Bok D, Rajasekaran AK. Na,K-ATPase inhibition alters tight junction structure and permeability in human retinal pigment epithelial cells. Am J Physiol Cell Physiol. 2003;284:C1497–C1507. doi: 10.1152/ajpcell.00355.2002. [DOI] [PubMed] [Google Scholar]
- 30.Hauser PJ, Dozmorov MG, Bane BL, Slobodov G, Culkin DJ, Hurst RE. Abnormal expression of differentiation related proteins and proteoglycan core proteins in the urothelium of patients with interstitial cystitis. J Urol. 2008;179:764–769. doi: 10.1016/j.juro.2007.09.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erickson DR, Schwarze SR, Dixon JK, Clark CJ, Hersh MA. Differentiation associated changes in gene expression profiles of interstitial cystitis and control urothelial cells. J Urol. 2008;180:2681–2687. doi: 10.1016/j.juro.2008.08.007. [DOI] [PubMed] [Google Scholar]
- 32.Keay S. Cell signaling in interstitial cystitis/painful bladder syndrome. Cell Signal. 2008;20:2174–2179. doi: 10.1016/j.cellsig.2008.06.004. [DOI] [PubMed] [Google Scholar]
- 33.Panayiotidis MI, Bortner CD, Cidlowski JA. On the mechanism of ionic regulation of apoptosis: would the Na+/K+-ATPase please stand up? Acta Physiol (Oxf) 2006;187:205–215. doi: 10.1111/j.1748-1716.2006.01562.x. [DOI] [PubMed] [Google Scholar]
- 34.Zhou G, Dada LA, Sznajder JI. Regulation of alveolar epithelial function by hypoxia. Eur Respir J. 2008;31:1107–1113. doi: 10.1183/09031936.00155507. [DOI] [PubMed] [Google Scholar]
- 35.Gusarova GA, Trejo HE, Dada LA, Briva A, Welch LC, Hamanaka RB, Mutlu GM, Chandel NS, Prakriya M, Sznajder JI. Hypoxia leads to Na,K-ATPase downregulation via Ca2+ release-activated Ca2+ channels and AMPK activation. Mol Cell Biol. 2011;31:3546–3556. doi: 10.1128/MCB.05114-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Wheaton WW, Chandel NS. Hypoxia. 2. Hypoxia regulates cellular metabolism. Am J Physiol Cell Physiol. 2011;300:C385–93. doi: 10.1152/ajpcell.00485.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]