Abstract
The levels of kynurenic acid (KYNA), an endogenous negative modulator of alpha 7 nicotinic acetylcholine receptors (α7nAChRs), are elevated in the brains of patients with schizophrenia (SZ). We reported that increases of brain KYNA in rats, through dietary exposure to its precursor kynurenine from embryonic day (ED)15 to postnatal day (PD) 21, result in neurochemical and cognitive deficits in adulthood. The present experiments focused on the effects of prenatal exposure to elevated kynurenine on measures of prefrontal excitability known to be impaired in SZ. Pregnant dams were fed a mash containing kynurenine (100 mg/day; progeny = EKYNs) from ED15 until ED22. Controls were fed an unadulterated mash (progeny = ECONs). The dietary loading procedure elevated maternal and fetal plasma kynurenine (2223% and 693% above controls, respectively) and increased fetal KYNA (forebrain; 500% above controls) on ED21. Elevations in forebrain KYNA disappeared after termination of the loading (PD2), but KYNA levels in the prefrontal cortex (PFC) were unexpectedly increased again when measured in adults (PD56-80; 75% above controls). We also observed changes in several markers of prefrontal excitability, including expression of the α7nAChR (22% and 17% reductions at PD2 and PD56-80), expression of mGluR2 (31% and 24% reductions at ED21 and PD56-80), dendritic spine density (11–14% decrease at PD56-80), subsensitive mesolimbic stimulation of glutamate release in PFC, and reversal/extra-dimensional shift deficits in the prefrontally-mediated set-shifting task. These results highlight the deleterious impact of elevated KYNA levels during sensitive periods of early development, which model the pathophysiological and cognitive deficits seen in SZ.
Keywords: alpha7 nicotinic receptors, glutamate, kynurenic acid, prefrontal cortex, schizophrenia, set-shifting
1. Introduction
Prevailing hypotheses regarding the etiology of schizophrenia (SZ) emphasize an interaction among genetic polymorphisms in a relatively large number of genes that mediate key neurodevelopmental processes (for reviews see Fromer et al., 2014; Harrison and Weinberger, 2005; Purcell et al., 2014) and early environmental risk factors including malnutrition, infection, trauma and/or social stress (Allardyce and Boydell, 2006; Brown and Patterson, 2011). Such interactions between biological vulnerabilities and environmental risk factors alter the functional maturation of the brain. Indeed, postmortem analyses of the brains of patients with SZ reveal developmental abnormalities in multiple brain regions, including cellular disorganization/altered migration in hippocampus (Kuroki and Mastushita, 1998) and cortex (Akbarian et al., 1993; Benes et al., 1991); reduced spine densities in cortex (Glantz and Lewis, 2000); and altered expression of several genes, including CHRNA7, GAD1, and GRIN2A and GRIN2B, related to the balance of excitatory/inhibitory transmission in key forebrain regions (Freedman et al., 2001; Hashimoto et al., 2003; Schwartz et al., 2012). Collectively, these early brain abnormalities eventually give rise to a complex syndrome of behavioral symptoms that do not fully emerge until after puberty (Castle et al., 1998; DeLisi, 2008).
Further understanding of this complex disorder and the development of more efficacious medications may be advanced using validated animal models that recreate defining pathophysiologies characteristic of those seen in patients with SZ. This may be particularly true in studying the neurobiology of cognitive deficits, which remain the most difficult symptom cluster to treat in SZ yet hold the most promise for enhancing functional outcome (Green et al., 2000; Ibrahim and Tamminga, 2012). Indeed, the Cognitive Neuroscience Treatment Research to Improve Cognition in Schizophrenia (CNTRICS) initiative (Barch et al., 2008; Carter et al., 2008) has placed an emphasis on cognition as a core symptom of SZ, with the goal of assessing specific cognitive processes and their associated neural/psychological mechanisms in both animal models and humans in an effort to develop novel intervention strategies.
We recently introduced chronic elevations of kynurenic acid (KYNA) as a naturalistic and translationally valid approach to experimentally study cognitive impairments seen in SZ. KYNA, an astrocyte-derived product of the kynurenine pathway (KP) of tryptophan metabolism, functions as an endogenous negative modulator of alpha 7 nicotinic acetylcholine (α7nACh) receptors at physiological levels and, at higher concentrations, inhibits N-methyl-D-aspartate (NMDA) receptors (Hilmas et al., 2001; Stone, 1993). Notably, even relatively modest increases in brain KYNA negatively modulate the release of several neurotransmitters critical for effective cognitive processing, including ACh (Zmarowski et al., 2009), glutamate (Konradsson-Geuken et al., 2010; Wu et al., 2010), dopamine (Rassoulpour et al., 2005), and GABA (Beggiato et al., 2014). Patients with SZ have increased CSF and postmortem brain KYNA levels (Erhardt et al., 2001; Schwarcz et al., 2001), possibly due to alterations in the expression/activity of the KP enzymes tryptophan 2,3-dioxygenase (TDO) and kynurenine 3-monooxygenase (KMO). By leading to impaired neurotransmitter functions during critical phases of brain development, these genetic modifications may disrupt the maturation of the excitatory/inhibitory balance in cortical transmission and, in turn, cause cognitive control impairments (i.e., planning, working memory, and verbal processing) in patients with SZ (Holtze et al., 2011, 2012; Miller et al., 2004; Wonodi et al., 2011). Of possible further relevance for SZ pathophysiology, KP abnormalities resulting in increased KYNA production may be exacerbated by environmental factors such as stress, infection, and inflammation (Müller, 2014; Schwarcz et al., 2012).
Administration of kynurenine, the bioprecursor of KYNA, from embryonic day (ED) 15 through postnatal day (PD) 21 results in cognitive inflexibility in an attentional set-shifting task that is dependent upon the prefrontal cortex (PFC; Alexander et al., 2013), and impairs hippocampus-dependent spatial working memory (Pocivavsek et al., 2012). In an effort to further delineate the extent of the developmental sensitive period, in the present study exposure to kynurenine was limited to the last prenatal week (ED15-22), which is analogous to the second trimester of pregnancy in humans (Clancy, 2001). Following this prenatal KYNA elevation, we determined cellular/molecular markers related to cortical excitability in adulthood. In particular, we examined brain KYNA levels and the expression of metabotropic glutamate receptor 2 (mGluR2) and α7nAChR from ED21 through PD56-80, and evaluated dendritic spine density and stimulated glutamate release in the PFC, as well as the integrity of cognitive flexibility, in young adulthood.
2. Materials and methods
2.1. Animals
Animals (Wistar rats) used for these experiments were dams and their progeny from The Ohio State breeding colony and were maintained on a 12:12h light/dark cycle (lights on at 0600 h) in temperature- and humidity-controlled, AAALAC-approved animal facilities with ad libitum access to water. Animals used in behavioral experiments were food-deprived to approximately 85–90% of their basal weight; all other animals received food ad libitum. All procedures were approved by the Institutional Animal Care and Use Committees of The Ohio State University in accordance with the NIH Guide for Care and Use of Laboratory Animals. As such, all efforts were made to minimize animal suffering, to reduce the number of animals used, and to consider alternatives to in vivo techniques.
2.2. Kynurenine supplementation and treatment groups
Breeder females were habituated to wet rodent mash (Teklad Diets, Madison, WI, USA; 30 g per day) beginning on embryonic day (ED) 0. Females received 100 mg/day of L-kynurenine sulfate (99.4% purity; Sai Advantium, Hyderabad, India) in the wet mash each day from ED15 through ED22 (EKYN treatment group); control animals were fed unadulterated mash (ECON treatment group). Standard rodent chow was provided to all animals thereafter. The day on which dams gave birth was denoted postnatal day (PD) 0. On PD2, litters were culled to 9 to 11 pups to standardize growth rates across all litters and to maximize the number of males. One cohort of rats (dams and their fetuses/pups) was selected for analysis of plasma kynurenine, brain KYNA levels, and qPCR at ED21, PD2, and as adults (PD56-80). Subjects for these biochemical assessments were selected from 3–4 litters (2–4 embryos or pups/litter); male and female progeny were used for prepubertal assessments (ED21 and PD2), while male adults were used for PD56-80 assessments. Another cohort of male rats (adult ECON and EKYN offspring) was selected for neurochemical, behavioral, and morphometric analyses at PD56-80. Subjects for adult offspring assessments (dendritic spine analyses, glutamate release, attentional set shifting) were individual male rats selected from the same 4–5 litters (1–3 pups/litter selected for each assessment).
2.3. Biochemical analyses
2.3.1. Kynurenine in plasma of dam and offspring
Plasma was obtained from blood of dams and their fetuses/offspring on ED21 and PD2. Blood was collected into tubes containing disodium EDTA by cardiac puncture from anesthetized (CO2) dams at ED21 (n = 4 ECON and n = 3 EKYN) and PD2 (n = 4 ECON and n = 4 EKYN) while blood from ED21 fetuses (n = 4 ECON litters and n = 3 EKYN litters) and PD2 pups (n = 4 ECON and 4 EKYN litters) was collected by rapid decapitation. The supernatant plasma was isolated by centrifugation (12 000 x g) and transferred to new Eppendorf tubes, frozen on dry ice and stored at −80°C. On the day of the assays, kynurenine was isocratically eluted and fluorimetrically detected using high performance liquid chromatography (HPLC) according to our published method (Pocivavsek et al., 2014).
2.3.2. Kynurenic acid (KYNA) in tissue homogenates
At the time of blood collection (ED21, PD2, and PD56-80), brains from fetuses/offspring were rapidly removed and sectioned through the sagittal midline (n = 8 rats/group/age). Counterbalanced hemi-sections from offspring were used for KYNA determination or quantitative polymerase chain reaction (qPCR) analysis. On ED21, the forebrain was analyzed (cerebellum and olfactory bulbs removed); on PD2 and PD56-80, the PFC was isolated. All tissues were stored at −80°C until analysis. For analysis of KYNA, tissues were thawed and assayed according to previously described HPLC methodology (Pocivavsek et al., 2014).
2.4. Quantitative polymerase chain reaction (qPCR)
Total RNA was extracted from forebrain/frontal cortices of ED21, PD2 and PD56-80 rats (n = 7–8 rats/group/age) using PureZol reagent (Bio-Rad, Hercules, CA, USA) followed by NucleoSpin RNA II kit (Macherey-Nagel, Düren, Germany) according to the manufacturers’ instructions. Genomic DNA contamination was eliminated by performing DNase digestion using RNase-free DNase (Macherey-Nagel, Düren, Germany). cDNA was obtained from the extracted RNA (up to 1 μg/reaction) using iScript reverse transcription Supermix for RT-qPCR (Bio-Rad, Hercules, CA, USA) under the following conditions: priming at 25°C for 5 min, reverse transcription for 30 min at 42°C, and inactivation of transcriptase by heating at 85°C for 5 min. The first-strand cDNA solution was diluted 5-fold with 0.5X Tris-EDTA buffer (Sigma-Aldrich, MO, USA) prior to qPCR.
qPCR was performed using the CFX96, C1000 Thermal Cycler (Bio-Rad, Hercules, CA, USA) in a reaction mixture containing 10 μL Sso Advanced SYBR Green Supermix (Bio-Rad, Hercules, CA, USA), 5 μL of each cDNA sample solution, 15 pmol of forward and reverse gene-specific primer-pairs (Integrated DNA Technologies, Coralville, IA, USA), and DNase-free water to a total volume of 20 μL. The thermal profile for PCR was as follows: initial denaturation at 95°C for 30 sec followed by 39 temperature cycles of denaturation at 95°C for 5 sec, annealing/extension (55°C for 30 sec for mGluR2; 62°C for 30 sec for α7nAChR), and melting curves. Fluorescence was acquired at the end of each extension phase. Fold changes in mRNA expression levels were calculated according to the comparative cycle threshold (CT) method and normalized using the level of GAPDH mRNA (Schmittgen and Livak, 2008). The primers used for each of the three genes were: GAPDH FW: 5′-CAT CAA GAA GGT GGT GAA GCA-3′, RV: 5′-CTG TTG AAG TCA CAG GAG ACA-3′(Rao et al., 2006); mGluR2 FW: 5′-GTG GTG ACA TTG CGC TGT AA-3′, RV: 3′-GCG ATG AGG AGC ACA TTG TA-3′ (Zhang et al., 2009); Alpha7 FW: 5′-TGC ACG TGT CCC TGC AAG GC-3, RV: 5′-GTA CAC GGT GAG CGG CTG CG-3′. Primer quality was evaluated by standard curve, melting curve and the presence of a single PCR product after gel electrophoresis.
2.5. Morphological analyses
2.5.1. Golgi staining and spine density analysis
Rats (n = 7 ECON and n = 6 EKYN) were deeply anesthetized with Euthasol (Virbac Animal Health, Ft. Worth, TX, USA). PFC tissue blocks were incubated in rapid Golgi solutions (GolgiStain Kit™, FD Neurotechnologies, Columbia, MD, USA), based on previously described methods (Leuner and Gould, 2010). Serial coronal sections (150 μm) were cut on a vibratome and mounted on gelatin-coated slides.
Golgi-stained tissue was analyzed with a Nikon 90i microscope (Nikon, Tokyo, Japan). Four to five Golgi-impregnated pyramidal neurons from the prelimbic/infralimbic cortex were selected from each animal and analyzed blind to experimental condition. Golgi-impregnated neurons had to possess the following characteristics for inclusion: (1) The neuron had to be within layer 2/3 and within the boundaries of the prelimbic/infralimbic areas; (2) Cell bodies had to be located in the middle third of the tissue section; (3) Golgi-impregnation had to be dark and consistent throughout the extent of all dendrites; and (4) Cells had to be relatively isolated from neighboring impregnated cells.
Measurement of apical and basal dendrites was conducted on secondary and tertiary branches; apical dendrites were measured at 25–75 μm, 75–150 μm, and 150–225 μm from the cell body. Basal dendrites were measured at least 50 μm from the cell body. Spine density was expressed as the number of spines per 10 μm of dendrite. Densities of spines on three to five segments of a cell were averaged for a cell mean, and the cells from each animal were averaged for an animal mean. Spine density values using this method are underestimates, because spines protruding either above or beneath the dendritic shaft are not accounted for (Leuner and Gould, 2010).
2.6. Basal and evoked glutamate in PFC
Basal and evoked glutamate levels in PFC were quantified using the glutamate sensitive microelectrode array (MEA) according to previously established methods (Bortz et al., 2013; Konradsson-Geuken et al., 2009). Briefly, the MEA is a ceramic microelectrode (Quanteon, LLC, Nicholasville, KY, USA) with four platinum recording sites, with each pair designated to be either glutamate-sensitive or not (sentinel). The glutamate channels were coated with glutamate oxidase (Gluox, 2%, 1 unit/1 μL, 100 nL), bovine serum albumin (BSA; 1%), and glutaraldehyde (0.125%), whereas the sentinel channels were coated with only BSA and glutaraldehyde. MEAs were electroplated with m-phenylene diamine (m-PD) to block the oxidation of larger endogenous molecules (i.e., ascorbic acid, dopamine, norepinephrine, serotonin), which would also oxidize at the applied potential (Mitchell, 2004).
Glutamate that reaches the surface of the MEA is oxidized by Gluox, generating H2O2 (the reporting molecule). H2O2 is then oxidized at a constant potential of +0.7 V vs. an Ag/AgCl reference electrode. The differential coating scheme allows for the isolation of current generated solely by the oxidation of glutamate by self-referencing activity of the sentinel channel from the Gluox channel (Burmeister and Gerhardt, 2001; Konradsson-Geuken et al., 2009; Rutherford et al., 2007).
2.6.1. In vitro calibration of microelectrodes
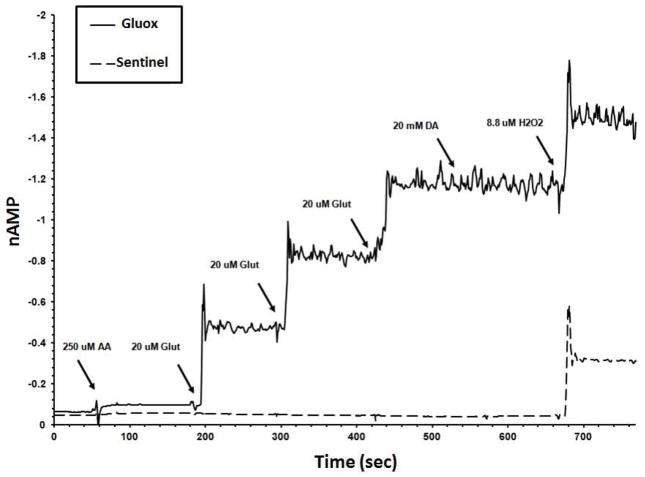
Microelectrodes were calibrated in vitro just prior to implantation (see Figure 1 for details and a representative calibration). The tracings represent change in current detected at each of the channels following the addition of the chemicals listed above (noted by the arrows). The top tracing is from the glutamate sensitive channel (Gluox), whereas the bottom tracing represents the background channel (sentinel).
Figure 1.
Representative Microelectrode Array (MEA) Calibration Curve. The MEA is calibrated in vitro immediately prior to implantation into the mPFC. Calibrations were performed in a stirred solution of phosphate-buffered saline (PBS) (0.05 M, 40 ml; pH 7.4; 37°C; +0.7 V). After a stable baseline was established, ascorbic acid (AA, 250 μM), glutamate (3x 20 μM), dopamine (DA, 2 mM), and H2O2 (8.8 μM), were sequentially added (marked by arrows) to the calibration beaker in 40 μL aliquots, and amperometric signals were acquired at a rate of 1.0 Hz. To simplify the Figure, only two of the four channels are illustrated. The response of glutamate-sensitive (top tracing) and sentinel (bottom tracing) channels in a representative calibration are shown. Current (nAmp) is depicted along the vertical axis and time (s) along the horizontal axis. Successive aliquots of glutamate produced a linear increase in current on the Gluox channel, but no glutamate-related current on the sentinel channel. Important for self-referencing, the calibration also reveals equivalently high sensitivities on both channels to the reporting molecule H2O2. The m-PD barrier prevents the recording of in vivo interferents, AA and DA.
2.6.2. Implantation of microelectrodes and infusion cannulae
Animals were anesthetized using isoflurane (2%, 0.6 L/min) and implanted with the MEAs unilaterally in the PFC (in mm from bregma: AP +2.7, ML ± 0.65, DV −4.0; hemispheres counterbalanced). The Ag/AgCl reference electrode was implanted in the contralateral side at a site distant from the recording area. Stainless steel guide cannulae (Plastics One, Roanoke, VA, USA), were implanted ipsilaterally in the shell of the nucleus accumbens (NACSh; in mm from bregma: AP + 0.4, ML ± 0.70, DV −6.4). The tip of the infusion cannula extended 1 mm beyond the end of the guide cannula. All coordinates were determined from the atlas of Paxinos and Watson (1998).
2.6.3. In vivo recordings of basal and evoked glutamate
Unrestrained ECON (n = 7) and EKYN (n = 5) rats were tested as adults in three recording sessions according to previously reported methods (Bortz et al., 2013). Recordings began on the second day after implantation. After a stable baseline was established (~ 3 hr), compounds were infused into the NACSh over ~2 sec (pH 7.1–7.4, 0.5 μL solutions) using a Hamilton PB600-1 manual dispenser (Hamilton Company, Reno, NV, USA). First, a vehicle solution (artificial cerebrospinal fluid [aCSF]) was infused to serve as a control for the infusion per se. Ten min later, one of three doses of NMDA (0.05, 0.15 or 0.30 μg in 0.5 μL) was infused. Doses of NMDA were counterbalanced across recording sessions.
2.6.4. Histology
At the conclusion of the MEA experiments, animals were given an overdose of Euthasol and were transcardially perfused. Brains were removed and stored in 10% neutral-buffered formalin (≥ 2 days) followed by a 30% sucrose solution (≥ 2 days). Brains were sectioned using a cryostat; coronal sections (50 μm) were mounted on gelatin-coated slides, stained using cresyl violet, and examined under a light microscope for verification of microelectrode and cannula placement.
2.7. Cognitive testing: attentional set shifting task
Rats (n = 7 ECON and n = 8 EKYN) were trained in an attentional set shifting task as described previously (Alexander et al., 2013; Birrell and Brown; 2000; Brooks et al., 2012). Briefly, rats were trained to discriminate between terra cotta pots that varied in texture, medium, or odor. On PD56, rats were placed on food restriction and habituated to the testing box (L 91 cm x W 40.5 cm x H 25.4 cm), which contained two compartments for the pots and a removable barrier used to control the rats’ access to the pots. Rats were food restricted until they reached 85–90% of pre-habituation weights and were then trained to dig in plain pots to retrieve cereal rewards (Day 1) or to discriminate between pots that varied on one of three dimensions (odor, texture, or medium; Day 2). Day 3 testing consisted of seven separate discriminations in which the rats were required to choose between two exemplars, only one of which was rewarded. Each task stage had four exploration trials whereby the rat could dig in either pot for up to 90 sec. Thereafter, rats had 60 sec per trial to make a choice. The trial was terminated if the rat dug in the incorrect.pot. Testing continued until the rat reached criterion of six consecutive correct choices (excluding exploration trials). In the simple discrimination (SD), the pots differed on one of the three stimulus dimensions. In the compound discrimination (CD), a second dimension was introduced such that one dimension was relevant, and one was irrelevant, but the correct and incorrect exemplars were the same as those in the SD. Reversals (REV1, REV2, REV3) consisted of reversing the exemplar rewarded in the previous discrimination. All new exemplars were introduced at the intra-dimensional (ID) and extra-dimensional (ED) shift stages. Further, at the ED the previously irrelevant dimension was now relevant. The order of stages was the same for all animals, but the stimulus dimensions and exemplars used were counterbalanced across animals such that each dimension was represented at each stage of the task.
2.8. Statistical analyses
Due to sample volume limitations, the litter was used as the experimental unit for all analyses of plasma. For all other endpoints (brain KYNA, qPCR, dendritic spines, glutamate release, and attentional set shifting), the rat was the experimental unit. For qPCR, biochemical, and spine density experiments: Group comparisons were made using unpaired t-tests. Glutamate release experiment: An initial, two-way ANOVA was performed for each dependent measure using NMDA dose as a within-subjects factor and kynurenine loading as a between-subjects factor. If significance was found, one-way ANOVAs comparing dose levels within each factor were performed. A minimum number of post-hoc comparisons were conducted using paired and unpaired t-tests. Set shifting experiment: An overall two-way ANOVA (kynurenine loading as a between-subject factor; stage of task as a within-subject factor) was conducted on the number of trials necessary to reach criterion. Following a significant interaction, one-way ANOVAs were performed to determine between-drug group effects on a particular stage of the task. In addition, paired t-tests were performed to compare CD vs. REV1 and ID vs. ED within treatment conditions. In all ANOVAs, the Huynh-Feldt correction was utilized to reduce Type II errors associated with repeated measures ANOVAs (Vasey, 1987). Statistics were performed using the SPSS statistics program (v19, IBM corporation, Armonk, NY, USA), and significance was defined as P < 0.05.
3. Results
3.1. Dietary kynurenine raises kynurenine levels in maternal and offspring plasma, and kynurenic acid in the offspring brain
Kynurenine loading of the dam’s diet during the last week of gestation resulted in a significant elevation of plasma kynurenine in dams (+2223%; t5 = −3.051, P = 0.028) and fetuses (+693%; t5 = −3.820, P = 0.012) on ED21 (Table 1). There was a corresponding increase (+500%) in KYNA in the brains of EKYNs relative to ECONs on ED21 (Figure 2; t14 = −8.957; P < 0.001). In contrast, on PD2, plasma kynurenine levels and brain KYNA levels in the EKYN group were similar to control levels (all P values > 0.05), presumably due to returning the dams to normal rodent chow. Interestingly, by PD56 brain KYNA levels in EKYN rats were 75% higher than in ECONs (t14 = −2.307; P = 0.037), suggesting long-term changes within the KP. Finally, no obvious differences in maternal care (i.e., nest-building, nursing posture, pup retrieval) were observed between the EKYN and ECON groups or in cross-fostered litters.
Table 1.
Kynurenine levels (μM) in plasma of dams and their fetuses/pups
Daily exposure to kynurenine from ED15 (100 mg/day; EKYN) raised kynurenine levels in the plasma of dams and fetuses on ED21. Data are the mean ± SEM (ECON: n = 4 litters; EKYN: n = 3 litters). By PD2, kynurenine levels were similar in dams (ECON: n = 4; and EKYN: n = 4) and pups (ECON: n = 4 litters; EKYN: n = 4 litters).
| Condition | ED21 | PD2 | ||
|---|---|---|---|---|
|
| ||||
| FETUS | DAM | PUP | DAM | |
|
|
||||
| ECON | 4.68 ± 1.9 | 1.3 ± 0.1 | 0.71 ± 0.17 | 1.54 ± 0.03 |
| EKYN | 37.1 ± 9.8 | 30.2 ± 11.3 | 0.99 ± 0.34 | 1.81 ± 0.31 |
| t-test | t5= −3.820; P=0.012 | t5=−3.051; P=0.028 | t6=−0.729; P=0.493 | t6 = −0.838; P=0.4383 |
Figure 2.
Effects of prenatal exposure to kynurenine on levels of KYNA in the PFC throughout development. KYNA levels (mean ± SEM) are presented for ED21 fetuses on the left Y-axis and for PD2 and PD56-80 offspring on the right Y-axis. Exposure to kynurenine (100 mg/day; EKYN) raised brain KYNA near the end of treatment on ED21 (ECON: n = 8; EKYN: n = 8). There was no difference at PD2 (ECON: n = 8; EKYN: n = 8) yet increased KYNA levels re-appeared as EKYNs reached PD56 (ECON: n = 8; EKYN: n = 8). * P <0.05, *** P < 0.001 vs. ECON.
3.2. Prenatal kynurenine exposure reduces mGluR2 and a7nAChR mRNA levels
There were marked age-related changes in mRNA expression of mGlu2Rs and α7nAChRs following prenatal kynurenine exposure. Brain mGluR2 mRNA levels were inversely related to brain KYNA levels. At ED21, expression was 31% lower in EKYN fetuses than in ECON fetuses (t13 = 3.642, P= 0.003; Fig. 3). While similar reductions were not observed at PD2 (P = 0.561), mGluR2 expression in adult offspring at PD56-80 was 24% lower than in controls (t14 = 2.434, P = 0.029). The expression of brain α7nAChR mRNA levels was similar between EKYN and ECON fetuses near the end of treatment, i.e., on ED21 (P = 0.609), but was 22% lower in EKYNs than ECONs on PD2 (t14 = 2.430, P = 0.029). α7nAChR mRNA levels were 17% lower in adult EKYNs than in ECONs, although the difference was not statistically significant (P = 0.111).
Figure 3.
Relative expression of mGluR2 and α7nAChR mRNA levels normalized to GAPDH mRNA at ED21, PD2, and PD56-80. Levels (mean ± SEM) for EKYNs at each age (n = 7, 8, and 8, respectively) are normalized to ECONs (n = 8, 8, and 8, respectively), and were set to a value of 1. mGluR2 expression was inversely related to KYNA levels, with decreased expression on ED21 and PD56-80. In contrast, α7nAChR mRNA was decreased at PD2 and trended toward a decrease at PD56-80. * P < 0.05, ** P <0.01.
3.3. Dendritic spine density
Prenatal kynurenine exposure was associated with decreased dendritic spine density on Golgi-impregnated pyramidal neurons located in layer 2/3 of the mPFC in adults (PD56-80; Fig. 4). The spine density reduction noted on apical dendrites occurred at a similar magnitude at proximal, intermediate, and distal segments evaluated (e.g., 25–75, 75–150, and 150–225 μm from the soma; data not shown); therefore, group total densities were compared. Compared to ECONs, dendritic spine density in EKYN rats was decreased 11% on apical dendrites (t11 = 5.731, P < 0.001) and 14% on basal dendrites (t11 = 2.713, P = 0.020).
Figure 4.
Effects of elevated KYNA on the density of dendritic spines in PFC. ECON (n=7) and EKYN (n=6) progeny were reared to PD56-80, when brains were collected for dendritic spine analysis. A. Golgi-impregnated pyramidal neuron from layer 2/3 of the mPFC (top left), with segments of apical (top middle) and basal (top right) dendrites from representative ECON and EKYN rats. B. Quantification of apical (bottom left) and basal (bottom right) dendritic spine density revealed a significant reduction of layer 2/3 spines of EKYN rats compared to ECON rats. * P < 0.05, *** P < 0.001. Bars represent mean ± SEM; scale bars: 20 μm, cell and 2 μm, segments.
3.4. Mesolimbic activation of glutamate release in PFC
3.4.1. Intracortical placement of microelectrode and infusion cannula
The mesolimbic activation of cortical glutamate release in ECONs and EKYNs was assessed by infusing three doses of NMDA (0.05, 0.15, and 0.30 μg in 0.4 μL), in counterbalanced order, into the NACSh. Figure 5A displays representative tracings of the MEA’s electrochemical signal following infusion of the mid-dose of NMDA (0.15 μg) in ECON and EKYN rats. The recording session revealed a typical phasic release of glutamate in response to the mid-dose of NMDA in an ECON rat, rapidly reaching a maximum increase of 4.65 μM in peak amplitude, which was quickly cleared to basal values within ~20 sec. In contrast, the resulting glutamate levels, evoked by the same stimulus, were markedly attenuated (0.66 μM) in the EKYN rat.
Figure 5.
Stimulated prefrontal glutamate release is attenuated in adult EKYN rats. A. Representative MEA tracings taken from the mPFC following the infusion of the middle-dose (0.15 μg) of NMDA in ECON (left panel) and EKYN (right panel) rats. The top tracings represent the signal from the Gluox (glutamate-sensitive) channel, whereas the middle tracings represent the sentinel (background) channel. Subtraction of the two yields the self-referenced signal and represents the signal obtained exclusively from the oxidation of extracellular glutamate (bottom tracings). The Figure represents passage of time (sec) on the x-axis and molar changes from basal extracellular glutamate, calculated using the regression lines from the calibration which converts H2O2-derived current (nAmp) to glutamate concentration (μM) on the y-axis. NMDA infusion (arrows) results in a robust (4.65 μM) increase in prefrontal glutamate release in the ECON rat, which persisted. In contrast, glutamate release is markedly attenuated in the EKYN rat (0.66 μM).
B. Group data representing the maximum peak amplitude (mean ± SEM) increase in prefrontal glutamate release (μM), relative to basal values, in adult ECON (n = 7) and EKYN (n = 5) rats following the infusion of three different doses of NMDA (0.05, 0.15, and 0.30 μg in 0.5 μL) into the shell of the nucleus accumbens. There was a marked shift in the dose-response curve following prenatal kynurenine treatment, as EKYN rats were less sensitive to infusion of 0.05 and 0.15 μg of NMDA compared to ECONs (** P <0.01, *** P <0.001). In contrast, glutamate release following 0.30 μg NMDA was similar between ECON and EKYN rats.
C. Representative photomicrographs of Nissl-stained tissue sections highlight the minimal tissue disruption and placements of the MEA in the prelimbic/infralimbic PFC, and the termination of the infusion cannula in the shell of the nucleus accumbens.
Comparison of the group data (Figure 5B) revealed a significant shift in the dose-response curve as a result of prenatal kynurenine exposure (F1,10 = 27.36, P < 0.001). EKYN rats were markedly less sensitive (86–99%), relative to ECONs, following infusion of 0.05 μg (F1,11 =11.796, P=0.006) and 0.15 μg (F1,11=72.481, P < 0.001) NMDA. In contrast, glutamate release following 0.30 μg NMDA was not significantly different between the EKYN and ECON groups (P = 0.362). This shift in the dose-response curve was observed despite the fact that the doses were presented in a counterbalanced order. Furthermore, basal glutamate levels were not significantly different between the ECON (0.52 ± 0.10 μM) and EKYN (0.35 ± 0.08 μM) groups (P > 0.05). Likewise, peak clearance, as measured by T80 (time in sec to clear 80% of the maximum glutamate peak) was similar, at all doses of NMDA, between the two groups of rats (P = 0.741).
Following completion of the recording sessions, the position of the MEA in the prelimbic/infralimbic region of the PFC and the termination of the infusion cannula in the NACSh was verified (Figure 5C). Only rats with properly positioned MEA and cannulae were included in analyses.
3.5. Performance in an attentional set-shifting task
As illustrated in Figure 6, exposure to kynurenine during the last prenatal week significantly impaired performance of adult offspring in the attentional set-shifting task (F1,13 = 18.598, P = 0.001), but the deficits were isolated to certain stages of the task (F3,45 = 15.446, P < 0.001). EKYN and ECON rats performed similarly (all P values >0.05) in rule acquisition (SD and CD), indicating that there were no deficits relating to animals’ abilities to acquire or maintain the rules of the task. However, the EKYN rats were impaired when the rules of the CD were reversed, requiring approximately twice as many trials as ECON rats (27 ± 3.7 versus 13 ± 1.8) to complete the REV1 (F1,14 = 11.100, P = 0.005). While EKYN rats were not impaired in the second reversal (REV2: F1,14 = 0.854, P = 0.372), or during the intra-dimensional shift (F1,14 = 3.675, P = 0.077), they required significantly more trials to complete the extra-dimensional shift (F1,14 = 11.084, P = 0.005) stage of the task than did ECON rats. A comparison of ID and ED shift stages indicated that impairments in the ED stage were not due to failure to form an attentional set (t7 = −3.757; P = 0.007).
Figure 6.
Mean trials to criterion (± SEM) for adult ECON (n = 7) and EKYN (n = 8) rats (PD56-80) in various stages of the attentional set-shifting task. All rats readily acquired the single (SD) and compound (CD) stages of training. The EKYN group required more trials than the ECON group to acquire REV1. Each group demonstrated comparable abilities to form an attentional set, as evidenced by the rapid acquisition of an intra-dimensional shift (ID) to a novel stimulus. EKYN rats exhibited a marked deficit in the ability to make an extra-dimensional shift (ED). a = significantly different, within treatment group, from the trials to criterion for the CD stage; b = significantly different from ECON within the REV1 stage; c = significantly different, within treatment group, from the trials to criterion for the ID stage; d = significantly different from ECON within the ED stage (all P values < 0.05).
4. Discussion
Increases in brain kynurenine from ED15-ED22 resulted in several significant changes in prefrontal chemistry, structure and function, including: 1) elevations in forebrain KYNA levels in adulthood; 2) reductions in the expression of mRNA for α7nAChRs that appeared to persist into adulthood; 3) long-lasting deficits in several markers of excitatory transmission in PFC (density of dendritic spines, expression of mRNA for mGluR2 receptors, evoked glutamate release); and 4) impairments in prefrontally-mediated cognition as indicated by characteristic reversal/extra-dimensional shift deficits in an attentional set shifting task. The following discussion will address the implications of these findings for factors that govern the maturation of PFC as well as the validity of the kynurenine/KYNA model for studying the etiology/treatment of cognitive disorders in SZ.
4.1. Dietary elevations of kynurenine and KYNA levels
The KP is present during embryonic development in several mammalian species (Cannazza et al., 2001; Walker et al., 1999), and changes in the metabolism of the pathway therefore have the potential to affect early brain development. Notably, brain KYNA levels are highest during the final days of gestation and drop dramatically immediately after birth (Ceresoli-Borroni and Schwarcz, 2000).
Dietary supplementation with the KYNA bioprecursor kynurenine markedly elevated plasma kynurenine and brain KYNA levels in EKYN dams and fetuses near the end of the treatment period (ED21). Two days after the termination of kynurenine treatment (PD2), levels of kynurenine in the plasma and KYNA in the brain were no longer increased in the EKYN group – presumably as a result of the termination of kynurenine supplementation. Curiously, however, KYNA levels in the PFC were again elevated (75%) when EKYN offspring were sampled as young adults (PD56-80). Notably, plasma kynurenine levels were not elevated in these animals (data not shown), suggesting an increase in KP metabolism specifically in the brain. Importantly, within the brain, the long-term increase in KYNA levels is not limited to the PFC. That is, we recently showed both acute and delayed elevations in hippocampal KYNA levels following prenatal kynurenine supplementation, and these increases were associated with impairments in passive avoidance and spatial working memory in adult EKYN rats (Pocivavsek et al., 2014). The mechanism(s) underlying these KYNA increases in the adult brain in the absence of continued precursor loading is not known, but could be related to changes in the cerebral expression and activity of key KP enzymes such as TDO or KMO, which have been documented post-mortem in the brain of patients with SZ (Miller et al., 2004; Sathyasaikumar et al., 2011; Wonodi et al., 2011).
A series of recent studies by Stone and colleagues examined the consequences of elevating brain KYNA levels during gestation by administering the KMO inhibitor Ro 61-8048 to dams. KMO inhibition resulted in a transient increase in fetal brain KYNA levels. Weanlings subsequently showed increased hippocampal excitability, as evidenced by increased long-term potentiation (LTP) and changes in NMDA subunit expression, as well as in the expression of several neurodevelopmental proteins (Forrest et al., 2013a). Conversely, adult offspring exhibited reduced excitability (reduced LTP and delayed recovery from long-term depression) and reduced dendritic spine density (Forrest et al., 2013b; Khalil et al., 2014). The authors interpreted their results almost exclusively in terms of a KYNA-induced antagonism of NMDA receptor function during critical periods of perinatal development. This restricted focus may not be warranted given the significant role played by α7AChRs in the sculpting of the developing PFC (see below). Resolution of this interpretation awaits further studies.
4.2. Receptor targets for elevated KYNA during early development and subsequent reductions in prefrontal excitation
Increased levels of forebrain KYNA could antagonize activity at both the α7nAChR (nM range; Albuquerque & Schwarcz, 2013; Hilmas et al., 2001) and the glycineB co-agoinst site of the NMDA receptor (μM range; Stone, 1993). During the third week of gestation, a critical period in rodent brain development (reviewed in Semple et al., 2013), reduced activity of these two receptors could significantly alter the maturation of regions with cholinergic or glutamatergic projections to the PFC (e.g., hippocampus, amygdala, basal forebrain, medial dorsal thalamus). Moreover, sustained increases in KYNA could continue to affect the maturation of the PFC, which is not completed until young adulthood (Andersen, 2003).
EKYN animals showed a significant reduction in α7nAChR expression at PD2, a trend which continued into adulthood. This initial decrease, and its persistence, may have interfered with the development of excitatory/inhibitory balance in the PFC. While the expression of GAD67 mRNA has not yet been evaluated in this model, we have recently discovered clear deficits in the maturation of GABAergic tone regulating the hippocampal control of excitatory transmission in prefrontal circuits (Thomases et al., 2014). This impairment in adult EKYN rats resembles a pattern that is seen normally during the juvenile period. The effect seen in adult EKYNs therefore suggests a regression to an earlier ontogenetic profile.
The reduction of α7nAChR expression may also have contributed to effects seen in the development and stabilization of excitatory synaptic contacts. Lozada et al. (2012), using α7nAChR knock-out (KO) mice, showed that KOs develop significantly fewer glutamatergic synapses and show overall decreased excitatory transmission. Enhanced antagonism of α7nAChRs by elevated KYNA levels, and subsequent decreases in excitatory transmission, may also account for the reduction in mGluR2 mRNA in EKYN rats (Figure 3). Type II mGlu receptors are located predominantly presynaptically where they function as either release-inhibiting autoreceptors on glutamatergic terminals or heteroreceptors on GABAergic terminals (Sheffler and Conn, 2010). The enduring decrease in mGluR2 mRNA may be a consequence of the sustained antagonism of α7nAChR function by KYNA (coupled with a reduced expression of α7nAChR mRNA) and a failure of cortical glutamatergic synapses to stabilize.
The relationship between α7nAChR function and excitatory transmission is also consistent with our current results of reduced spine density in layers II/III of PFC in the EKYN group (Figure 4). Chronic antagonism of α7nAChR function by sustained elevation of KYNA may contribute to the marked reduction in glutamate release under conditions of lower intensity stimulation (i.e., subsensitivity to the lower doses of intra-accumbens NMDA; Figure 5) and to the previously reported complete loss of mesolimbic regulation of prefrontal glutamate release following more prolonged, pre- and postnatal exposure to kynurenine during development (Alexander et al., 2011). Adult EKYN animals were, in fact, barely responsive to infusions of 0.05 or 0.15 μg, but exhibited a normal reaction to the highest NMDA dose (0.30 μg). These findings may reflect a decrease in the number of prefrontal glutamatergic synapses and indicate a need for more salient stimulation (consistent with reduced expression of mGluR2 and decreased dendritic spine density). This excitatory system does, however, exhibit a capacity to respond appropriately if the stimulus is sufficiently salient.
The present study does not rule out that KYNA’s antagonism of the glycineB site may have contributed to the changes seen in EKYN rats. Notably, NMDA receptor activity is centrally involved in early phases of brain development, including neuronal migration (Behar et al., 1999), the formation and guidance of axonal branches (Rajan and Cline, 1998), the induction of dendritic spines (Ultanir et al., 2007), and the formation and maintenance of excitatory synaptic contacts (Colonnese et al., 2005; Sala and Segal, 2014). Thus, KYNA-induced reductions in cortical NMDA receptor activity may contribute to the observed decrease in dendritic spines and associated compromises in cortical plasticity (Kasai et al., 2010; McKinney, 2010).
Deficits in performance of specific stages of the attentional set-shifting task are a convincing indicator of functional impairment in the PFC of EKYN animals. Both ECON and EKYN adult offspring were capable of learning the discriminations and acquiring an attentional set. However, like other experimental protocols that disrupt PFC maturation, as well as patients with SZ (Murray et al., 2008; Owen et al., 1991), EKYN rats exhibited marked deficits in the initial reversal and the ability to make an extra-dimensional shift - both functions dependent upon the integrity of the PFC (Birrell and Brown, 2000). The relative contributions of the two most prominent KYNA targets, α7nACh and glycineB receptors, to the set-shifting deficits remain to be determined. Notably, however, we have previously reported alleviation from these impairments with either the α7nAChR partial agonist SSR180711 or the positive α7nACh modulator galantamine in rats treated acutely with kynurenine (Alexander et al., 2012; Pershing et al., 2011) or following more prolonged exposure (ED15-PD21) to kynurenine (Alexander et al., 2013).
4.3. Sustained prenatal KYNA elevations in rats: relevance for the pathophysiology of SZ
The EKYN animals studied in the present experiments have significant face and construct validity for SZ. Elevated levels of KYNA in the PFC are seen in SZ, and the underlying causes (genetic polymorphisms, maternal infection, stress) constitute significant developmental risk factors for the disease (cf: Introduction). Moreover, decreased dendritic spine density has been repeatedly described in the brains of individuals of SZ (Glausier and Lewis, 2013). Reduced mRNA expression of α7nAChR and mGluR2 are also consistent with clinical reports (Bullock et al., 2008; Freedman, 1995; Gonzalez-Maeso et al., 2008), though exceptions have been noted (DeLuca et al., 2006; Ghose et al., 2009). Finally, the observed REV/ED deficits in an attentional set-shifting task are characteristic of impaired cognitive flexibility in patients with SZ (Leeson et al., 2009; Pantelis et al., 2009). Ongoing studies using EKYN rats are designed to investigate possible contributions of KP metabolites other than KYNA and to evaluate peripheral KP metabolites as possible biomarkers of disease. We are currently also attempting to attenuate/reverse the neurochemical, behavioral and structural abnormalities in EKYN animals pharmacologically, using agents that mitigate the elevations in brain KYNA and/or their consequences (Wu et al., 2014). Together, these experiments should provide an improved appraisal of EKYN animals for the study of SZ pathophysiology and treatment.
HIGHLIGHTS.
-
1
Pregnant rats (ED15-22) ate a diet with kynurenine (EKYNs) or not (ECONs).
-
1
KYNA increased 500% and 75% above controls on ED21 and PD56, respectively.
-
2
mGluR2 mRNA declined by 31% and 24% at ED21 and PD56, respectively in EKYNs
-
3
α7nAChR mRNA was reduced by 22% and 17% at PD2 and PD56, respectively in EKYNs
-
4
dendritic spine density and glutamate release in PFC were reduced in EKYNs at PD56
-
5
Adult EKYNs show reversal/extra-dimensional shift deficits in the set-shifting task.
Acknowledgments
This research was supported by NIMH grant MH083729 (to JPB and RS).
Footnotes
Financial Disclosures
The authors report no financial interests or other potential conflicts of interest.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Akbarian S, Bunney WE, Potkin SG, Wigal SB, Hagman JO, Sandman CA, Jones EG. Altered distribution of nicotinamide-adenine dinucleotide phosphate diaphorase cells in frontal-lobe of schizophrenics implies disturbances of cortical development. Archives of General Psychiatry. 1993;50:169–177. doi: 10.1001/archpsyc.1993.01820150007001. [DOI] [PubMed] [Google Scholar]
- Albuquerque EX, Schwarcz R. Kynurenic acid as an antagonist of alpha 7 nicotinic acetylcholine receptors in the brain: Facts and challenges. Biochemical Pharmacology. 2013;85:1027–1032. doi: 10.1016/j.bcp.2012.12.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander KS, Bortz DM, Wu HQ, Brooks JM, Schwarcz R, Bruno JP. Perinatal elevations of kynurenic acid dysregulate prefrontal glutamate release and produce set-shifting deficits in adults: A new model of schizophrenia. Society for Neuroscience Meeting Planner 163.23. 2011 http://www.abstractsonline.com/plan/start.aspx?mkey=%7B8334BE29-8911-4991-8C31-32B32DD5E6C8%7D.
- Alexander KS, Wu HQ, Schwarcz R, Bruno JP. Acute elevations of brain kynurenic acid impair cognitive flexibility: normalization by the alpha7 positive modulator galantamine. Psychopharmacology. 2012;220:627–637. doi: 10.1007/s00213-011-2539-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alexander KS, Pocivavsek A, Wu HQ, Pershing ML, Schwarcz R, Bruno JP. Early developmental elevations of brain kynurenic acid impair cognitive flexibility in adults: reversal with galantamine. Neuroscience. 2013;238:19–28. doi: 10.1016/j.neuroscience.2013.01.063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allardyce J, Boydell J. Review: The wider social environment and schizophrenia. Schizophrenia Bulletin. 2006;32:592–598. doi: 10.1093/schbul/sbl008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen SL. Trajectories of brain development: point of vulnerability or window of opportunity? Neuroscience and Biobehavioral Reviews. 2003;27:3–18. doi: 10.1016/s0149-7634(03)00005-8. [DOI] [PubMed] [Google Scholar]
- Barch DM, Carter CS, Comm CE. Measurement issues in the use of cognitive neuroscience tasks in drug development for impaired cognition in schizophrenia: A report of the second consensus building conference of the CNTRICS initiative. Schizophrenia Bulletin. 2008;34:613–618. doi: 10.1093/schbul/sbn037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beggiato S, Tanganelli S, Fuxe K, Antonelli T, Schwarcz R, Ferraro L. Endogenous kynurenic acid regulates extracellular GABA levels in the rat prefrontal cortex. Neuropharmacology. 2014;82:11–18. doi: 10.1016/j.neuropharm.2014.02.019. [DOI] [PubMed] [Google Scholar]
- Behar TN, Scott CA, Greene CL, Wen XL, Smith SV, Maric D. Glutamate acting at NMDA receptors stimulates embryonic cortical neuronal migration. Journal of Neuroscience. 1999;19:4449–4461. doi: 10.1523/JNEUROSCI.19-11-04449.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benes FM, Mcsparren J, Bird ED, Sangiovanni JP, Vincent SL. Deficits in small interneurons in prefrontal and cingulate cortices of schizophrenic and schizoaffective patients. Archives of General Psychiatry. 1991;48:996–1001. doi: 10.1001/archpsyc.1991.01810350036005. [DOI] [PubMed] [Google Scholar]
- Birrell JM, Brown VJ. Medial frontal cortex mediates perceptual attentional set shifting in the rat. Journal of Neuroscience. 2000;20:4320–4324. doi: 10.1523/JNEUROSCI.20-11-04320.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortz DM, Mikkelsen JD, Bruno JP. Localized infusions of the partial alpha 7 nicotinic receptor agonist SSR180711 evoke rapid and transient increases in prefrontal glutamate release. Neuroscience. 2013;255:55–67. doi: 10.1016/j.neuroscience.2013.09.047. [DOI] [PubMed] [Google Scholar]
- Brooks JM, Pershing ML, Thomsen MS, Mikkelsen JD, Sarter M, Bruno JP. Transient inactivation of the neonatal ventral hippocampus impairs attentional set-shifting behavior: reversal with an alpha7 nicotinic agonist. Neuropsychopharmacology. 2012;37:2476–2486. doi: 10.1038/npp.2012.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown AS, Patterson PH. Maternal infection and schizophrenia: implications for prevention. Schizophrenia Bulletin. 2011;37:284–290. doi: 10.1093/schbul/sbq146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullock WM, Cardon K, Bustillo J, Roberts RC, Perrone-Bizzozero NI. Altered expression of genes involved in GABAergic transmission and neuromodulation of granule cell activity in the cerebellum of schizophrenia patients. American Journal of Psychiatry. 2008;165:1594–1603. doi: 10.1176/appi.ajp.2008.07121845. [DOI] [PubMed] [Google Scholar]
- Burmeister JJ, Gerhardt GA. Self referencing ceramic based multisite microelectrodes for the detection and elimination of interferences from the measurement of L-glutamate and other analytes. Analytical Chemistry. 2001;73:1037–1042. doi: 10.1021/ac0010429. [DOI] [PubMed] [Google Scholar]
- Cannazza G, Chiarugi A, Parenti C, Zanoli P, Baraldi M. Changes in kynurenic, anthranilic, and quinolinic acid concentrations in rat brain tissue during development. Neurochemical Research. 2001;26:511–514. doi: 10.1023/a:1010960812204. [DOI] [PubMed] [Google Scholar]
- Carter CS, Barch DM, Buchanan RW, Bullmore E, Krystal JH, Cohen J. Identifying cognitive mechanisms targeted for treatment development in schizophrenia: An overview of the first meeting of the cognitive neuroscience treatment research to improve cognition in schizophrenia initiative. Biological Psychiatry. 2008;64:4–10. doi: 10.1016/j.biopsych.2008.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castle D, Sham B, Murray R. Differences in distribution of ages of onset in males and females with schizophrenia. Schizophrenia Research. 1998;33:179–183. doi: 10.1016/s0920-9964(98)00070-x. [DOI] [PubMed] [Google Scholar]
- Ceresoli-Borroni G, Schwarcz R. Perinatal kynurenine pathway metabolism in the normal and asphyctic rat brain. Amino Acids. 2000;19:311–323. doi: 10.1007/s007260070062. [DOI] [PubMed] [Google Scholar]
- Clancy B, Darlington RB, Finlay BL. Translating developmental time across mammalian species. Neuroscience. 2001;105:7–17. doi: 10.1016/s0306-4522(01)00171-3. [DOI] [PubMed] [Google Scholar]
- Colonnese MT, Zhao JP, Constantine-Paton M. NMDA receptor currents suppress synapse formation on sprouting axons in vivo. Journal of Neuroscience. 2005;25:1291–1303. doi: 10.1523/JNEUROSCI.4063-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DeLisi LE. The concept of progressive brain change in schizophrenia: Implications for understanding schizophrenia. Schizophrenia Bulletin. 2008;34:312–321. doi: 10.1093/schbul/sbm164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Erhardt S, Blennow K, Nordin C, Skogh E, Lindstrom LH, Engberg G. Kynurenic acid levels are elevated in the cerebrospinal fluid of patients with schizophrenia. Neuroscience Letters. 2001;313:96–98. doi: 10.1016/s0304-3940(01)02242-x. [DOI] [PubMed] [Google Scholar]
- De Luca V, Likhodi O, Van Tol HH, Kennedy JL, Wong AH. Regulation of alpha7-nicotinic receptor subunit and alpha7-like gene expression in the prefrontal cortex of patients with bipolar disorder and schizophrenia. Acta Psychiatrica Scandinavica. 2006;114:211–215. doi: 10.1111/j.1600-0447.2006.00785.x. [DOI] [PubMed] [Google Scholar]
- Forrest CM, Khalil OS, Pisar M, Darlington LG, Stone TW. Prenatal inhibition of the tryptophan-kynurenine pathway alters synaptic plasticity and protein expression in the rat hippocampus. Brain Research. 2013a;1504:1–15. doi: 10.1016/j.brainres.2013.01.031. [DOI] [PubMed] [Google Scholar]
- Forrest CM, Khalil OS, Pisar M, McNair K, Kornisiuk E, Snitcofsky M. Changes in synaptic transmission and protein expression in the brains of adult offspring after prenatal inhibition of the kynurenine pathway. Neuroscience. 2013b;254:241–259. doi: 10.1016/j.neuroscience.2013.09.034. [DOI] [PubMed] [Google Scholar]
- Freedman R, Coon H, Myles-Worsley M, Orr-Urtreger A, Olincy A, Davis A. Linkage of a neurophysiological deficit in schizophrenia to a chromosome 15 locus. Proceedings of the National Academy of Sciences of the United States of America. 1997;94:587–592. doi: 10.1073/pnas.94.2.587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedman R, Hall M, Adler LE, Leonard S. Evidence in postmortem brain tissue for decreased numbers of hippocampal nicotinic receptors in schizophrenia. Biological Psychiatry. 1995;38:22–33. doi: 10.1016/0006-3223(94)00252-X. [DOI] [PubMed] [Google Scholar]
- Freedman R, Leonard S, Olincy A, Kaufmann CA, Malaspina D, Cloninger CR. Evidence for the multigenic inheritance of schizophrenia. American Journal of Medical Genetics. 2001;105:794–800. doi: 10.1002/ajmg.10100. [DOI] [PubMed] [Google Scholar]
- Fromer M, Pocklington AJ, Kavanagh DH, Williams HJ, Dywer S, Owen MJ. De novo mutations in schizophrenia implicate synaptic networks. Nature. 2014;506:179–184. doi: 10.1038/nature12929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghose S, Crook JM, Bartus CL, Sherman TG, Herman MM, Hyde TM. Metabotropic glutamate receptor 2 and 3 gene expression in the human prefrontal cortex and mesencephalon in schizophrenia. International Journal of Neuroscience. 2009;118:1609–1627. doi: 10.1080/00207450802330702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glantz LA, Lewis DA. Decreased dendritic spine density on prefrontal cortical pyramidal neurons in schizophrenia. Archives of General Psychiatry. 2000;57:65–73. doi: 10.1001/archpsyc.57.1.65. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Maeso J, Ang RL, Yuen T, Chan P, Weisstaub NV, Lopez-Gimenez JF. Identification of a serotonin/glutamate receptor complex implicated in psychosis. Nature. 2008;452:93–97. doi: 10.1038/nature06612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green MF, Kern RS, Braff DL, Mintz J. Neurocognitive deficits and functional outcome in schizophrenia: Are we measuring the “right stuff”? Schizophrenia Bulletin. 2000;26:119–136. doi: 10.1093/oxfordjournals.schbul.a033430. [DOI] [PubMed] [Google Scholar]
- Harrison PJ, Weinberger DR. Schizophrenia genes, gene expression, and neuropathology: on the matter of their convergence. Molecular Psychiatry. 2005;10:40–68. doi: 10.1038/sj.mp.4001558. [DOI] [PubMed] [Google Scholar]
- Hashimoto T, Volk DW, Eggan SM, Mirnics K, Pierri JN, Sun ZX. Gene expression deficits in a subclass of GABA neurons in the prefrontal cortex of subjects with schizophrenia. Journal of Neuroscience. 2003;23:6315–6326. doi: 10.1523/JNEUROSCI.23-15-06315.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hilmas C, Pereira EFR, Alkondon M, Rassoulpour A, Schwarcz R, Albuquerque EX. The brain metabolite kynurenic acid inhibits alpha 7 nicotinic receptor activity and increases non-alpha 7 nicotinic receptor expression: Physiopathological implications. Journal of Neuroscience. 2001;21:7463–7473. doi: 10.1523/JNEUROSCI.21-19-07463.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holtze M, Saetre P, Erhardt S, Schwieler L, Werge T, Hansen T. Kynurenine 3-monooxygenase (KMO) polymorphisms in schizophrenia: An association study. Schizophrenia Research. 2011;127:270–272. doi: 10.1016/j.schres.2010.10.002. [DOI] [PubMed] [Google Scholar]
- Holtze M, Saetre P, Engberg G, Schwieler L, Werge T, Andreassen OA. Kynurenine 3-monooxygenase polymorphisms: relevance for kynurenic acid synthesis in patients with schizophrenia and healthy controls. Journal of Psychiatry and Neuroscience. 2012;37:53–57. doi: 10.1503/jpn.100175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibrahim HM, Tamminga CA. Treating impaired cognition in schizophrenia. Current Pharmaceutical Biotechnology. 2012;13:1587–1594. doi: 10.2174/138920112800784772. [DOI] [PubMed] [Google Scholar]
- Kasai H, Fukuda M, Watanabe S, Hayashi-Takagi A, Noguchi J. Structural dynamics of dendritic spines in memory and cognition. Trends in Neurosciences. 2010;33:121–129. doi: 10.1016/j.tins.2010.01.001. [DOI] [PubMed] [Google Scholar]
- Kerns JG, Nuechterlein KH, Braver TS, Barch DM. Executive functioning component mechanisms and schizophrenia. Biological Psychiatry. 2008;64:26–33. doi: 10.1016/j.biopsych.2008.04.027. [DOI] [PubMed] [Google Scholar]
- Khalil OS, Pisar M, Forrest CM, Vincenten MCJ, Darlington LG, Stone TW. Prenatal inhibition of the kynurenine pathway leads to structural changes in the hippocampus of adult rat offspring. European Journal of Neuroscience. 2014;39:1558–1571. doi: 10.1111/ejn.12535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konradsson-Geuken A, Gash CR, Alexander K, Pomerleau F, Huettl P, Gerhardt GA. Second-by-second analysis of alpha 7 nicotine receptor regulation of glutamate release in the prefrontal cortex of awake rats. Synapse. 2009;63:1069–1082. doi: 10.1002/syn.20693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konradsson-Geuken Å, Wu HQ, Gash CR, Alexander KS, Cambell A, Sozeri Y. Cortical kynurenic acid bi-directionally modulates prefrontal glutamate levels as assessed by microdialysis and rapid electrochemistry. Neuroscience. 2010;169:1848–1859. doi: 10.1016/j.neuroscience.2010.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuroki N, Matsushita M. Pyramidal cell orientation and density in the hippocampus in schizophrenia. Neuropathology. 1998;18:235–241. [Google Scholar]
- Leeson VC, Robbins TW, Matheson E, Hutton SB, Ron MA, Barnes TRE, Joyce EM. Discrimination learning, reversal, and set-shifting in first-episode schizophrenia: stability over six years and specific associations with medication type and disorganization syndrome. Biological Psychiatry. 2009;66:586–593. doi: 10.1016/j.biopsych.2009.05.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leuner B, Gould E. Dendritic growth in medial prefrontal cortex and cognitive flexibility are enhanced during the postpartum period. Journal of Neuroscience. 2010;30:13499–13503. doi: 10.1523/JNEUROSCI.3388-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lozada AF, Wang XL, Gounko NV, Massey KA, Duan JJ, Liu ZP. Glutamatergic synapse formation is promoted by alpha 7-containing nicotinic acetylcholine receptors. Journal of Neuroscience. 2012;32:7651–7661. doi: 10.1523/JNEUROSCI.6246-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McKinney RA. Excitatory amino acid involvement in dendritic spine formation, maintenance and remodelling. Journal of Physiology-London. 2010;588:107–116. doi: 10.1113/jphysiol.2009.178905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller CL, Llenos IC, Dulay JR, Barillo MM, Yolken RH, Weis S. Expression of the kynurenine pathway enzyme tryptophan 2,3-dioxygenase is increased in the frontal cortex of individuals with schizophrenia. Neurobiology of Disease. 2004;15:618–629. doi: 10.1016/j.nbd.2003.12.015. [DOI] [PubMed] [Google Scholar]
- Mitchell KM. Acetylcholine and choline amperometric enzyme sensors characterized in vitro and in vivo. Analytical Chemistry. 2004;76:1098–1106. doi: 10.1021/ac034757v. [DOI] [PubMed] [Google Scholar]
- Müller N. Immunology of schizophrenia. Neuroimmunomodulation. 2014;21:109–116. doi: 10.1159/000356538. [DOI] [PubMed] [Google Scholar]
- Murray GK, Cheng F, Clark L, Barnett JH, Blackwell AD, Fletcher PC. Reinforcement and reversal learning in first-episode psychosis. Schizophrenia Bulletin. 2008;34:848–855. doi: 10.1093/schbul/sbn078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owen AM, Roberts AC, Polkey CE, Sahakian BJ, Robbins TW. Extra-dimensional versus intra-dimensional set shifting performance following frontal-lobe excisions, temporal-lobe excisions or amygdalo-hippocampectomy in man. Neuropsychologia. 1991;29:993–1006. doi: 10.1016/0028-3932(91)90063-e. [DOI] [PubMed] [Google Scholar]
- Pantelis C, Yucel M, Bora E, Fornito A, Testa R, Brewer WJ. Neurobiological markers of illness onset in psychosis and schizophrenia: the search for a moving target. Neuropsychology Review. 2009;19:385–398. doi: 10.1007/s11065-009-9114-1. [DOI] [PubMed] [Google Scholar]
- Paxinos G, Watson C. The rat brain in stereotaxic coordinates. 4. Academic Press; San Diego: 1998. [DOI] [PubMed] [Google Scholar]
- Pershing ML, Bortz DM, Alexander KS, Brooks JM, Mikkelsen JD, Thomsen MS. Prefrontal alpha7 nicotinic receptors modulate local glutamate release and set-shifting behavior. Society for Neuroscience Meeting Planner 163.17. 2011 http://www.abstractsonline.com/plan/start.aspx?mkey=%7B8334BE29-8911-4991-8C31-32B32DD5E6C8%7D.
- Pocivavsek A, Wu HQ, Elmer GI, Bruno JP, Schwarcz R. Pre- and postnatal exposure to kynurenine causes cognitive deficits in adulthood. European Journal of Neuroscience. 2012;35:1605–1612. doi: 10.1111/j.1460-9568.2012.08064.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pocivavsek A, Thomas MA, Elmer GI, Bruno JP, Schwarcz R. Continuous kynurenine administration during the prenatal period, but not during adolescence, causes learning and memory deficits in adult rats. Psychopharmacology. 2014;231:2799–2809. doi: 10.1007/s00213-014-3452-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Purcell SM, Moran JL, Fromer M, Ruderfer D, Solovieff N, Roussous P. A polygenic burden of rare disruptive mutations in schizophrenia. Nature. 2014;506:185–190. doi: 10.1038/nature12975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajan I, Cline HT. Glutamate receptor activity is required for normal development of tectal cells dendrites in vivo. Journal of Neuroscience. 1998;18:7836–7846. doi: 10.1523/JNEUROSCI.18-19-07836.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranjan A, Mallick BN. A modified method for consistent and reliable Golgi-cox staining in significantly reduced time. Frontiers of Neurology. 2010;1:157. doi: 10.3389/fneur.2010.00157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao PK, Kumar RM, Farkhondeh M, Baskerville S, Lodish HF. Myogenic factors that regulate expression of muscle-specific microRNAs. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:8721–8726. doi: 10.1073/pnas.0602831103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rassoulpour A, Wu HQ, Ferré S, Schwarcz R. Nanomolar concentrations of kynurenic acid reduce extracellular dopamine levels in the striatum. Journal of Neurochemistry. 2005;93:762–765. doi: 10.1111/j.1471-4159.2005.03134.x. [DOI] [PubMed] [Google Scholar]
- Rutherford EC, Pomerleau F, Huettl P, Stromberg I, Gerhardt GA. Chronic second-by-second measures of L-glutamate in the central nervous system of freely moving rats. Journal of Neurochemistry. 2007;102:712–722. doi: 10.1111/j.1471-4159.2007.04596.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sala C, Segal M. Dendritic spines: the locus of structural and functional plasticity. Physiological Reviews. 2014;94:141–188. doi: 10.1152/physrev.00012.2013. [DOI] [PubMed] [Google Scholar]
- Sathyasaikumar KV, Stachowski EK, Wonodi I, Roberts RC, Rassoulpour A, McMahon RP, Schwarcz R. Impaired kynurenine pathway metabolism in the prefrontal cortex of individuals with schizophrenia. Schizophrenia Bulletin. 2011;37:1147–1156. doi: 10.1093/schbul/sbq112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C-T method. Nature Protocols. 2008;3:1101–1108. doi: 10.1038/nprot.2008.73. [DOI] [PubMed] [Google Scholar]
- Schwarcz R, Bruno JP, Muchowski PJ, Wu HQ. Kynurenines in the mammalian brain: when physiology meets pathology. Nature Reviews Neuroscience. 2012;13:465–477. doi: 10.1038/nrn3257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarcz R, Rassoulpour A, Wu HQ, Medoff D, Tamminga CA, Roberts RC. Increased cortical kynurenate content in schizophrenia. Biological Psychiatry. 2001;50:521–530. doi: 10.1016/s0006-3223(01)01078-2. [DOI] [PubMed] [Google Scholar]
- Schwartz TL, Sachdeva S, Stahl SM. Glutamate neurocircuitry: theoretical underpinnings in schizophrenia. Frontiers in Pharmacology. 2012;3:1–11. doi: 10.3389/fphar.2012.00195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semple BD, Blomgren K, Gimlin K, Ferriero DM, Noble-Haeusslein LJ. Brain development in rodents and humans: Identifying benchmarks of maturation and vulnerability to injury across species. Progress in Neurobiology. 2013;106:1–16. doi: 10.1016/j.pneurobio.2013.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheffler DJ, Conn PJ. Activation of group II metabotropic glutamate receptors (mGluR2 and mGluR3) as a novel approach for treatment of schizophrenia. In: Skolnick P, editor. Glutamate-Based Therapies for Psychiatric Disorders. Birkhäuser; Basel, Switzerland: 2010. pp. 101–116. [Google Scholar]
- Stefani MR, Moghaddam B. Systemic and prefrontal cortical NMDA receptor blockade differentially affect discrimination learning and set-shift ability in rats. Behavioral Neuroscience. 2005;119:420–428. doi: 10.1037/0735-7044.119.2.420. [DOI] [PubMed] [Google Scholar]
- Stone TW. Neuropharmacology of quinolinic and kynurenic acids. Pharmacological Reviews. 1993;45:309–379. [PubMed] [Google Scholar]
- Thomases DT, Flores-Barrera E, Bruno JP, Schwarcz R, Tseng KY. Contribution of alpha-7 nAChR tone in sustaining the gain of GABAergic transmission in the adult prefrontal cortex. Society for Neuroscience Annual Meeting; Washington DC. November 15–19; 2014. Abstract 33.02/B18. [Google Scholar]
- Ultanir SK, Kim JE, Hall BJ, Deerinck T, Ellisman M, Ghosh A. Regulation of spine morphology and spine density by NMDA receptor signaling in vivo. Proceedings of the National Academy of Sciences of the United States of America. 2007;104:19553–19558. doi: 10.1073/pnas.0704031104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vasey MW, Thayer JF. The continuing problem of false positives in repeated measures Anova in psychophysiology - A multivariate solution. Psychophysiology. 1987;24:479–486. doi: 10.1111/j.1469-8986.1987.tb00324.x. [DOI] [PubMed] [Google Scholar]
- Walker DW, Curtis B, Lacey B, Nitsos I. Kynurenic acid in brain and cerebrospinal fluid of fetal, newborn, and adult sheep and effects of placental embolization. Pediatric Research. 1999;45:820–826. doi: 10.1203/00006450-199906000-00007. [DOI] [PubMed] [Google Scholar]
- Wonodi I, Stine OC, Sathyasaikumar KV, Roberts RC, Mitchell BD, Hong LE. Downregulated kynurenine 3-monooxygenase gene expression and enzyme activity in schizophrenia and genetic association with schizophrenia endophenotypes. Archives of General Psychiatry. 2011;68:665–674. doi: 10.1001/archgenpsychiatry.2011.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu HQ, Pereira EFR, Bruno JP, Pellicciari R, Albuquerque EX, Schwarcz R. The astrocyte-derived alpha7 nicotinic receptor antagonist kynurenic acid controls extracellular glutamate levels in the prefrontal cortex. Journal of Molecular Neuroscience. 2010;40:204–210. doi: 10.1007/s12031-009-9235-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T, Zhang J, Shi J, Feng Y, Sun ZS, Li H. Antinociceptive synergistic effect of spinal mGluR2/3 antagonist and glial cells inhibitor on peripheral inflammation-induced mechanical hypersensitivity. Brain Research Bulletin. 2009;79:219–223. doi: 10.1016/j.brainresbull.2009.01.009. [DOI] [PubMed] [Google Scholar]
- Zmarowski A, Wu HQ, Brooks JM, Potter MC, Pellicciari R, Schwarcz R, Bruno JP. Astrocyte-derived kynurenic acid modulates basal and evoked cortical acetylcholine release. European Journal of Neuroscience. 2009;29:529–538. doi: 10.1111/j.1460-9568.2008.06594.x. [DOI] [PubMed] [Google Scholar]