Abstract
Aim of the study
The prognostic value of the detection of circulating tumour cells (CTCs) in gastric cancer has been studied intensely in recent years. However, the application of different technologies led to inconsistent results between the studies. Here, we performed a meta-analysis of published studies to summarise the evidence.
Material and methods
Medline and ISI Web of Knowledge were searched up to March 2013 using “circulating tumor cells” and “gastric cancer” as search terms. Hazard ratio (HR) with 95% confidence intervals (CIs) for prognostic outcomes and clinical characteristics were extracted from each study. Pooled hazard ratios (HR) and odds ratios (OR) were calculated using random or fixed-effects models.
Results
Twelve studies enrolling 774 patients were included. The combined HR estimate for overall survival (OS), disease-free survival (DFS), and progression-free survival (PFS) were 1.41 (95% CI: 1.28–1.62), 2.99 (95% CI: 2.01–4.45) and 1.64 (95% CI: 1.02–2.62), respectively. Subgroup analysis concerning detection methods and sampling time showed that results of RT-PCR for the OS group and RT-PCR for the DFS group suggest a prognostic significance of CTC detection (pooled HR [95% CI]: 1.45 [1.28–1.65], I2 = 38%, p = 0.13; 2.99 [2.01–4.45], I2 = 0%, p = 0.32). In addition, results of the baseline CTC detection group also indicated a significant prognostic value to predict OS and DFS (pooled HR [95% CI]: 1.47 [1.19–1.82], I2 = 38%, p = 0.14; 2.99 [2.01–4.45], I2 = 0%, p = 0.32). We simultaneously found that the detection of CTCs correlated with pathological stage (pooled OR [95% CI]: 2.95 [1.65–5.28], I2 = 56%, p = 0.03), lymph node status (pooled OR [95% CI]: 2.26 [1.50–3.41], I2 = 37%, p = 0.09), the depth of invasion (pooled OR [95% CI]: 3.21 [1.38–7.43], I2 = 72%, p = 0.002), and distant metastasis (pooled OR [95% CI]: 2.68 [1.25–5.73], I2 = 43%, p = 0.15).
Conclusions
Detection of CTCs is associated with poorer prognosis in gastric cancer patients.
Keywords: circulating tumour cells, gastric cancer, prognosis, meta-analysis
Introduction
Gastric cancer (GC) is the fourth most common cancer and the second most common cause of cancer death globally [1]. To date, pathological stage, Lauren's histological type, invasion in lymphatic and vascular system, and residual tumour presence are widely used factors to predict survival outcomes of GC patients [2–6]. However, prediction of the aforementioned factors is clinically insufficient. Though increasing prognostic markers are being discovered, more powerful factors are still needed [7, 8].
The presence of tumour cells in the blood stream was first reported by Ashworth [9] in 1869. The low concentration in peripheral blood makes it difficult to detect circulating tumour cells (CTCs). The CELLSEARCH system, Immunocytochemistry (ICC), and reverse transcriptase polymerase chain reaction (RT-PCR) are widely used methods to detect CTCs currently, and the CELLSEARCH system has been ratified by the FDA (Food and Drug Administration) for the application of prognosis prediction in breast cancer patients. Recently, meta-analyses pooling studies using the above-mentioned approaches have documented the prognostic value of CTC detection in patients with lung cancer [10], breast cancer [11], and colorectal cancer [12]. Pooled HRs of these studies show that the presence of CTCs indicate a poorer prognostic outcome. However, the prognostic relevance of CTC detection in gastric cancer patients remains controversial. Varied CTC detection methods and contrasting survival outcomes can be found in studies focusing on the prognostic value of CTCs [13–24] in gastric cancer.
Here, we conducted the first comprehensive meta-analysis of published literature on this topic to summarise the evidence of the prognostic value of CTC detection in gastric cancer patients.
Material and methods
Search strategy
Medline and the ISI Web of Knowledge database were searched in March 2013. The following keywords were variably combined: “circulating tumor cells”, “CTCs” and “gastric cancer”. No language or time restrictions were made.
Data extraction
Three reviewers (HY Wang, J Wei, and ZY Zou) independently extracted the primary data and baseline characteristics of the included studies. The primary data were hazard ratio (HR) and its 95% confidence interval (CI) of OS, PFS, and DFS. In nine included articles, only the p-value and/or the Kaplan-Meier survival curves, but not HR and its 95% CI, were given. As for these articles [14–22], methods according to the work of Parmar, Williamson, and Tierney were used to calculate the HR [25–27]. The baseline characteristics included first author, publication year, study size, patients’ age, pathological stage, sampling time, methods of detection, CTCs markers and positive definition, detection rate, observed survival outcomes, and HR estimation methods. No included studies reported histological subtype data. Therefore, this characteristic is lacking in our analysis. All disagreements were resolved by discussion.
Inclusion criteria
In order to be eligible, studies had to: (i) discuss the relevance of CTC detection in peripheral blood and survival outcomes such as OS, PFS, and DFS; and (ii) provide sufficient data for extracting or estimating HR and its 95% CI. If more than one marker was used in a certain study, the results of each marker were recorded as an independent set.
Exclusion criteria
Studies were excluded from the analysis if: (i) the articles were not written in English, (ii) the articles were reviews or letters, (iii) studies had a sample size < 20 patients, or (iv) studies lacked requisite information to extract or calculate primary data for meta-analysis.
Statistical methods
We calculated the logHR and standard error (SE) by using software designed by Matthew Sydes and Jayne Tierney (Medical Research Council Clinical Trials Unit, London, UK) [27]. The pooled HR was gained using fixed or random-effects models according to the heterogeneity between studies. Heterogeneity was evaluated with the Cochran's Q test as well as the I2 index and was defined as p < 0.10 or I2 > 50% [28]. Subgroup research for detection methods and sampling time were performed if the included studies were ≥ 2. The association of detection of CTCs with clinical variables (sexuality, pathological stage, lymph node metastasis, depth of invasion, and distant metastasis) were also investigated. Four additional articles [29–32] were included in this section because clinical characteristics and CTC conditions were provided. The odds ratio was used as a measure index of the correlation.
We used Forrest plots to show the pooled HR, and HR > 1 indicated worse survival outcome. All calculations were conducted by using Review Manager Version 5.2 (The Cochrane Collaboration, Software Update, Oxford, UK).
Results
Characteristics of eligible studies
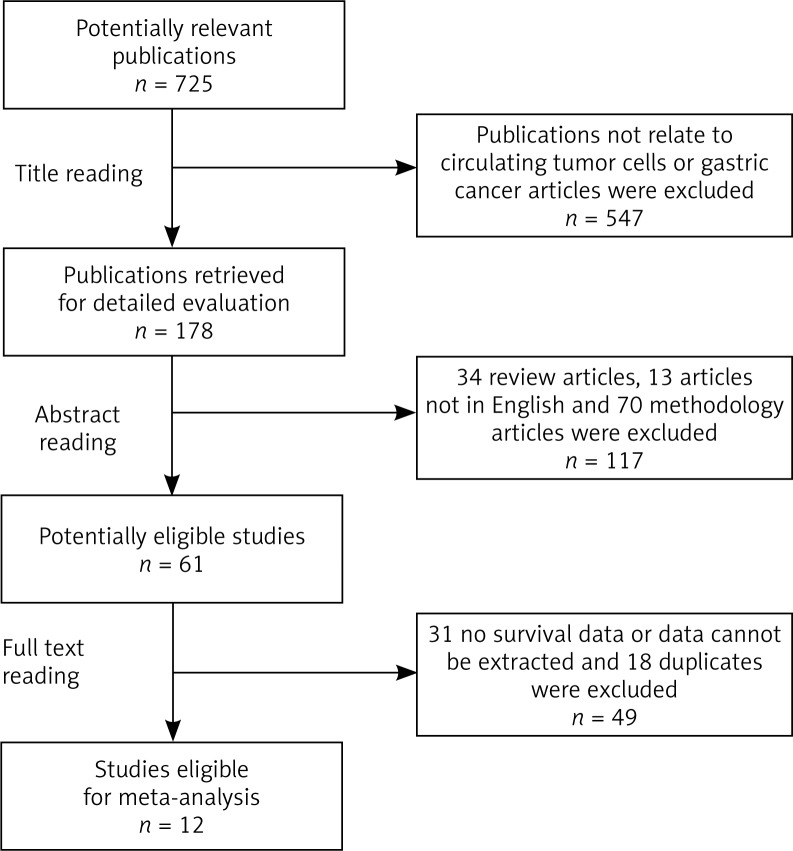
The literature search yielded 725 articles. After title reading, abstract reading, and full-text reviewing, a total of 12 articles were included (Fig. 1). Eligible studies encompassed 772 gastric cancer patients and the sample size ranged from 26 to 123 patients. The included studies were conducted between 2005 and 2011. The main features of these studies are listed in Table 1. Uen's study [18] and Koga's study [19] used different markers to detect CTCs, and Matsusaka's study [15] recorded both PFS and OS. Therefore, each result of these studies was analysed independently. Furthermore, four of excluded studies containing investigable data were additionally used in the investigation of the correlation between detection of CTCs and clinical characteristics.
Fig. 1.
Flowchart of the selection
Table 1.
Baseline characteristics of included studies
| Author [ref.], year | No. of patients | Age | Stage | Sampling time | Methods | Markers and positive definition | Detection rate | Outcome | HR estimation |
|---|---|---|---|---|---|---|---|---|---|
| Qiu [13], 2010 | 123 | median, 59 | M0, M1 | prior to surgery | RT-PCR | CEA mRNA (+) | 36.6 | DFS | Reported in text |
| Arigami [14], 2011 | 95 | average, 68 | M0, M1 | prior to surgery | RT-PCR | B7-H3 mRNA (+) | 50.5 | OS | data extrapolated |
| Matsusaka [15], 2010 | 52 | median, 62 | – | before treatment | CELLSEARCH | ≥ 4CTCs/7.5 ml blood | 32.7 | OS, PFS | data extrapolated |
| Pituch-Noworolska [16], 2007 | 57 | mean, 57.0 | M0, M1 | prior to surgery | flow cytometry | ≥ 3 cells CK+ per slide | 54.4 | OS | data extrapolated |
| Hiraiwa [17], 2008 | 27 | mean, 68.9 | M1 | before treatment | CELLSEARCH | ≥ 2 CTCs/7.5 ml blood | 55.6 | OS | data extrapolated |
| Uen [18], 2006 | 52 | mean, 60.0 | M0, M1 | during surgery | RT-PCR | C-MET, MUC-1 mRNA (+) | 61.5 (C-MET) 71.2 (MUC-1) 74.3 (both) |
OS | data extrapolated |
| Koga [19], 2008 | 69 | mean, 65.9 | M0, M1 | prior to surgery | RT-PCR | CK19, CK20 mRNA (+) | 11.6 (CK19 + ) 15.5 (CK20 +) |
OS | data extrapolated |
| Yie [20], 2008 | 26 | median, 58 | M0, M1 | – | RT-PCR | survivin mRNA (+) | 45.4 | PFS | data extrapolated |
| Wu [21], 2006 | 64 | mean, 60.5 | M0, M1 | during surgery | high-throughput colorimetric membrane-array | hTERT, CK-19, CEA, MUC1 mRNA (all +) |
60.9 | OS | data extrapolated |
| Illert [22], 2005 | 41 | median, 69 | M0, M1 | prior to surgery | RT-PCR | CK20 mRNA (+) | 36.6 | OS | data extrapolated |
| Bertazza [23], 2009 | 70 | median, 68 | M0, M1 | after surgery | RT-PCR | survivin mRNA (+) | 98.6 | OS | Reported in text |
| Cao [24], 2011 | 98 | – | M0, M1 | prior to surgery | RT-PCR | survivin mRNA (+) | 45.9 | DFS | Reported in text |
OS – overall survival; PFS – progression-free survival; DFS – disease-free survival; RT-PCR – reverse transcriptase polymerase chain reaction
Overall analyses of circulating tumour cells and survival
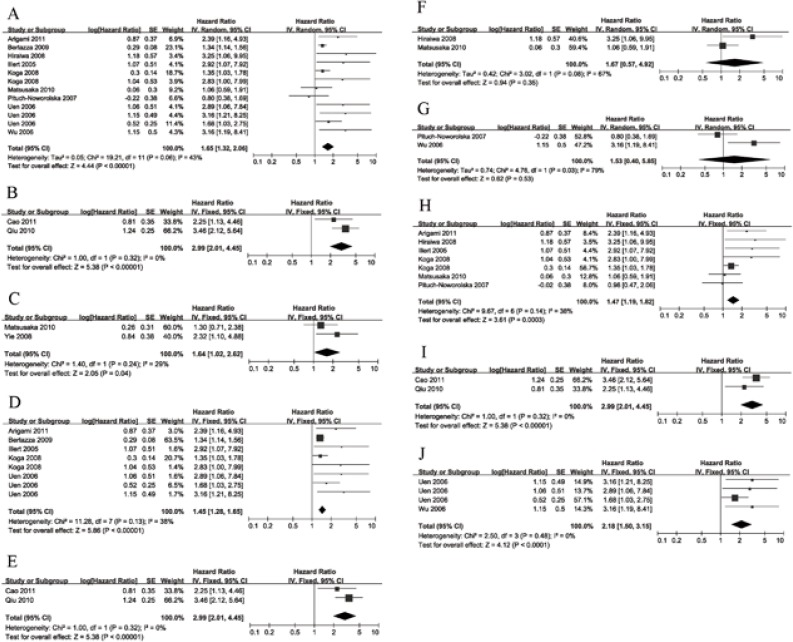
Twelve HRs for OS extracted from 9 studies accounting for 527 patients were pooled [14–19, 21–23]. The pooled HR was 1.65 (95% CI: 1.32–2.06) (I2 = 43%, p = 0.06). The result showed an increased mortality in patients with positive CTCs (Fig. 2, Table 2).
Fig. 2.
Forrest plots of estimated hazard ratios (HRs) for (A) CTC detection and OS, B) CTC detection and PFS, C) CTC detection and DFS, D) CTC detection using RT-PCR and OS, E) CTC detection using RT-PCR and DFS, F) CTC detection using CELLSEARCH and OS, G) CTC detection using other methods and OS, H) baseline CTC detection and OS, I) baseline CTC detection and DFS, and (J) CTC detection during surgery and OS
CTCs – circulating tumour cells; OS – overall survival; PFS – progression-free survival; DFS – disease-free survival; RT-PCR – reverse transcriptase polymerase chain reaction
Table 2.
Results of overall and subgroup meta-analyses
| n | OS | PFS | DFS | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HR [95% CI] | I2 | p | n | HR [95% CI] | I2 | p | n | HR [95% CI] | I2 | p | ||
| Total | 12 | 1.65 [1.32–2.06] | 43% | 0.06 | 2 | 1.64 [1.02–2.62] | 29% | 0.24 | 2 | 2.99 [2.01–4.45] | 0% | 0.32 |
| Method RT-PCR CELLSEARCH other methods |
8 2 2 |
1.45 [1.28–1.65] 1.67 [0.57–4.92] 1.53 [0.40–5.85] |
38% 67% 79% |
0.13 0.08 0.03 |
1 1 0 |
2.32 [1.10–4.88] 1.30 [0.71–2.38] – |
– – – |
2 0 0 |
2.99 [2.01–4.45] – – |
0% – – |
0.32 |
|
| Sampling time baseline During surgery after treatment |
7 4 1 |
1.47 [1.19–1.82] 2.18 [1.50–3.15] 1.34 [1.14–1.56] |
38% 0% – |
0.14 0.48 |
1 0 0 |
1.30 [0.71–2.38] – – |
– – – |
2 0 0 |
2.99 [2.01–4.45] – – |
0% – – |
0.32 |
|
OS – overall survival; PFS – progression-free survival; DFS – disease-free survival; n – study numbers; p – p value; RT-PCR – reverse transcriptase polymerase chain reaction
Two HRs for PFS and two HRs for DFS were extracted from 4 studies accounting for 299 patients [13, 15, 20, 24]. The pooled HRs for PFS and DFS were 1.64 (95% CI: 1.02–2.62) (I2 = 29%, p = 0.24) and 2.99 (95% CI: 2.01–4.45) (I2 = 0%, p = 0.32), respectively. It revealed that patients with CTCs detected had an increased risk of disease progression or recurrence (Fig. 2, Table 2).
Subgroup analyses of detection methods and sampling times
Subgroups were stratified by different detection methods and sampling times (Table 2). Meta-analysis was conducted if the subgroup encompassed more than one study.
We implemented meta-analysis in the subgroups that had sufficient studies. Among these subgroups, the method-stratified ones included RT-PCR for the OS group [14, 18, 19, 22], RT-PCR for the DFS group [13, 24], CELLSEARCH for the OS group [15, 17], and other methods for the OS group [16, 21]. Sampling-time stratified ones included baseline for the OS group [14–17, 19, 22] and during surgery for the OS group [18, 21]. Two subgroups (RT-PCR for the OS group and RT-PCR for the DFS group) suggested the prognostic significance of CTC detection (pooled HR [95% CI]: 1.45 [1.28–1.65], I2 = 38%, p = 0.13; 2.99 [2.01–4.45], I2 = 0%, p = 0.32). The baseline CTC group also indicated a significant prognostic value to predict OS and DFS (pooled HR [95% CI]: 1.47 [1.19–1.82], I2 = 38%, p = 0.14; 2.99 [2.01–4.45], I2 = 0%, p = 0.32). However, the results of the CELLSEARCH group and the other-methods group were not significant (pooled HR [95% CI]: 1.67 [0.57–4.92], I2 = 67%, p = 0.08; 1.53 [0.40–5.85], I2 = 79%, p = 0.03).
Correlation between detection of circulating tumour cells and clinical characteristics
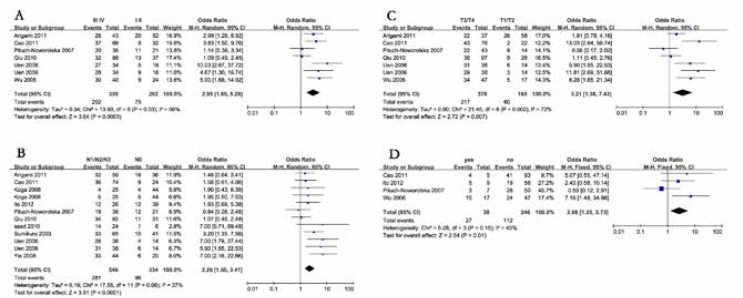
We extracted clinical characteristics from the included studies and four additional articles [29–32]. The potential correlation between detection of CTCs and clinical variables was investigated. The pooled odds ratio demonstrated that CTCs were more likely to be detected in patients with stage III/IV [13, 14, 16, 18, 21, 24], pT3/T4 [13, 14, 16, 18, 21, 24], positive lymph node metastasis [13, 14, 16, 18–20, 24, 29, 30, 32], or positive distant metastasis [16, 21, 24, 29] (Table 3, Fig. 3). Significant correlation was not observed when we stratified the studies by sexuality [13, 14, 16, 18–20, 24, 30–32]. The odds ratio was 1.02 (95% CI: 0.75–1.37) (I2 = 0%, p = 0.46).
Table 3.
Detection of CTCs and clinical characteristics
| Clinical characteristics | Study (n) | Patient (n) | OR [95% CI] | I2 | I value |
|---|---|---|---|---|---|
| Sexuality (male vs. female) | 13 | 875 | 1.02 [0.75–1.37] | 0% | 0.46 |
| pathological stage(III/IV vs. I/II) | 7 | 541 | 2.95 [1.65–5.28] | 56% | 0.03 |
| Lymph node (N1/N2/N3 vs. N0) | 12 | 880 | 2.26 [1.50–3.41] | 37% | 0.09 |
| The depth of invasion (pT3/pT4 vs. pT1/pT2) | 7 | 541 | 3.21 [1.38–7.43] | 72% | 0.002 |
| Distant metastasis (yes vs. no) | 4 | 284 | 2.68 [1.25–5.73] | 43% | 0.15 |
Fig. 3.
Forrest plots of estimated odds ratios for correlation of circulating tumour cells appearance and (A) pathological stage; B) lymph node metastasis; C) depth of invasion; and (D) distant metastasis
Discussion
Recently, a series of meta-analysis articles documented that CTC detection had prognostic value in patients with colorectal cancer [12], lung cancer [10], breast cancer [33, 34], melanoma [35], and prostate cancer [36]. It was the first time that a meta-analysis to confirm the prognostic value of CTCs in gastric cancer patients had been conducted. The pooled HRs for OS, PFS, and DFS were all above 1 and no overlap with 1 was observed. Our results indicated that the appearance of CTCs in peripheral blood betokened a poorer survival outcome.
Subgroup analyses were carried out according to different detection methods and sampling times. The CELLSEARCH system, RT-PCR, and immunocytochemistry were the most commonly used methods to detect CTCs. Among them, the CELLSEARCH system is the only one that is certificated by the FDA. The studies were divided into a RT-PCR group, a CELLSEARCH system group, and an other-methods group. The results of the RT-PCR group was in agreement with overall analyses, while those of the CELLSEARCH and the other-methods groups were not. Matsusaka et al. [15] and Hiraiwa et al. [17] used the CELLSEARCH system to detect CTCs. However, our analysis showed that the prognostic value was not significant (Table 2). We pooled the HRs by use of random effects model as I2 = 67% and p = 0.08. Significant heterogeneity may be caused by different cutoff of CTC detection and relatively small sample size. Similarly, non-significant prognostic value and high heterogeneity were observed in the subgroups of other methods. Ring's research indicated that RT-PCR was more sensitive than other CTC detection methods [37]. The most commonly applied method of included studies was RT-PCR. Hence, we believe that more studies using the CELLSEARCH system and other methods to evaluate the prognostic value of CTCs in gastric cancer patients could help to solve the puzzle. Besides, novel detection methods emerge continuously [38] and should be taken into consideration in future.
According to our results, detection of CTCs at baseline showed the ability to predict OS and DFS. Patients before any treatment with CTC presence in blood had shorter survival time and relapsed earlier if they underwent radical surgery. Interestingly, if CTCs were detected in a blood sample taken during surgery, it also indicated a poorer OS. This might be explained by Hou JM's point of view that CTCs have the ability to promote metastasis [39]. However, heterogeneity was the greatest problem in these subgroup analyses because the therapeutic regimens differed from each other. Therefore, more studies with sufficient key information like surgery type and chemotherapy regimens are needed to obtain further understanding of the CTC detection's prognostic value at different time points in gastric cancer patients.
We found that sexuality was not related to detection of CTCs in gastric cancer patients. Correlations were found between detection of CTCs and clinical characteristics including pathological stage, lymph node metastasis, depth of invasion, and distant metastasis. To avoid the heterogeneity caused by variant pathological staging version, in the analysis focusing on pathological stage and the depth of invasion, we only enrolled studies adopting UICC version 5 or 6. According to the same reasoning, patients were divided into “III/IV vs. I/II” and “pT3/pT4 vs. pT1/pT2” groups to keep the analysis powerful. The pooled odds ratio were all above 2 and indicated a higher detection rate of CTCs in patients with advanced stage, deeper tumour invasion, and lymph node/distant metastasis. Therefore, researchers suggested that CTCs could provide useful information for tumour staging and even cancer diagnosis [40]. Paterlini-Brechot's article indicated that CTCs were tumour cells from local or metastasis niduses that invaded blood vessels and contaminated peripheral blood [41]. This point of view may explain the correlation of detection of CTCs and clinical characteristics.
The limitations of the present meta-analysis need to be discussed. Firstly, HRs and 95% CI of some included studies were extracted. Log(HR) and se(log(HR)) were then calculated by the software provided by Matthew Sydes and Jayne Tierney. Potential biases may relate to this process. Secondly, heterogeneity existed between studies because of diverse detection methods, different cut off of CTCs, etc. We tried to solve this problem by extracting more information from the articles and performing subgroup analyses. However, significant heterogeneity still existed in some subgroups and a random-effects model was used for more conservative estimates. Hence, to validate the prognostic value of CTC detection, large multicentre prospective studies enrolling homogeneous populations are required in future. Thirdly, our meta-analysis only used published data. Updated individual patient data were not obtained. If those data were added to our analyses, the accuracy and determinacy could be better.
Our meta-analysis suggests that detection of CTCs in peripheral blood is a prognostic factor to predict survival outcomes, including OS, PFS, and DFS, in gastric cancer patients. We found that CTCs were inclined to be positive in patients with more advanced disease. This may explain why detection of CTCs is associated with poorer prognosis. To confirm this conclusion and discover further significance of CTCs, like guiding treatment, more multicentre, well-designed prospective studies are needed in future.
The authors declare no conflict of interest.
References
- 1.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61:69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 2.Kim JP, Lee JH, Kim SJ, Yu HJ, Yang HK. Clinicopathologic characteristics and prognostic factors in 10 783 patients with gastric cancer. Gastric Cancer. 1998;1:125–33. doi: 10.1007/s101200050006. [DOI] [PubMed] [Google Scholar]
- 3.Setälä LP, Kosma VM, Marin S, Lipponen PK, Eskelinen MJ, Syrjänen KJ, Alhava EM. Prognostic factors in gastric cancer: the value of vascular invasion, mitotic rate and lymphoplasmacytic infiltration. Br J Cancer. 1996;74:766–72. doi: 10.1038/bjc.1996.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Seeliger H, Spatz H, Jauch KW. Minimal residual disease in gastric cancer. Recent Results Cancer Res. 2003;162:79–87. doi: 10.1007/978-3-642-59349-9_7. [DOI] [PubMed] [Google Scholar]
- 5.Sobin LH, Wittekind C. TNM classification of malignant tumours. 5th ed. New York: John Wiley; 1997. International Union against Cancer. [Google Scholar]
- 6.Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31–49. doi: 10.1111/apm.1965.64.1.31. [DOI] [PubMed] [Google Scholar]
- 7.Huszno J, Mrochen-Domin I, Zembala-Nożyńska E, Tukiendorf A, Lange D, Nowara E. Mucin secretion activity of gastric cancer as a prognostic factor: a clinicopathological analysis. Contemp Oncol (Pozn) 2012;16:159–64. doi: 10.5114/wo.2012.28796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brzozowa M, Mielańczyk L, Michalski M, Malinowski L, Kowalczyk-Ziomek G, Helewski K, Harabin-Słowińska M, Wojnicz R. Role of Notch signaling pathway in gastric cancer pathogenesis. Contemp Oncol (Pozn) 2013;17:1–5. doi: 10.5114/wo.2013.33765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ashworth TR. A case of cancer in which cells similar to those in the tumours were seen in the blood after death. Aus Med J. 1869;14:146–9. [Google Scholar]
- 10.Ma XL, Xiao ZL, Liu L, Liu XX, Nie W, Li P, Chen NY, Wei YQ. Meta-analysis of circulating tumor cells as a prognostic marker in lung cancer. Asian Pac J Cancer Prev. 2012;13:1137–44. doi: 10.7314/apjcp.2012.13.4.1137. [DOI] [PubMed] [Google Scholar]
- 11.Zhang L, Riethdorf S, Wu G, Wang T, Yang K, Peng G, Liu J, Pantel K. Meta-analysis of the prognostic value of circulating tumor cells in breast cancer. Clin Cancer Res. 2012;18:5701–10. doi: 10.1158/1078-0432.CCR-12-1587. [DOI] [PubMed] [Google Scholar]
- 12.Rahbari NN, Aigner M, Thorlund K, et al. Meta-analysis shows that detection of circulating tumor cells indicates poor prognosis in patients with colorectal cancer. Gastroenterology. 2010;138:1714–26. doi: 10.1053/j.gastro.2010.01.008. [DOI] [PubMed] [Google Scholar]
- 13.Qiu MZ, Li ZH, Zhou ZW, et al. Detection of carcinoembryonic antigen messenger RNA in blood using quantitative real-time reverse transcriptase-polymerase chain reaction to predict recurrence of gastric adenocarcinoma. J Transl Med. 2010;8:107. doi: 10.1186/1479-5876-8-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Arigami T, Uenosono Y, Hirata M, Yanagita S, Ishigami S, Natsugoe S. B7-H3 expression in gastric cancer: a novel molecular blood marker for detecting circulating tumor cells. Cancer Sci. 2011;102:1019–24. doi: 10.1111/j.1349-7006.2011.01877.x. [DOI] [PubMed] [Google Scholar]
- 15.Matsusaka S, Chìn K, Ogura M, et al. Circulating tumor cells as a surrogate marker for determining response to chemotherapy in patients with advanced gastric cancer. Cancer Sci. 2010;101:1067–71. doi: 10.1111/j.1349-7006.2010.01492.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pituch-Noworolska A, Kolodziejczyk P, Kulig J, Drabik G, Szczepanik A, Czupryna A, Popiela T, Zembala M. Circulating tumour cells and survival of patients with gastric cancer. Anticancer Res. 2007;27:635–40. [PubMed] [Google Scholar]
- 17.Hiraiwa K, Takeuchi H, Hasegawa H, et al. Clinical significance of circulating tumor cells in blood from patients with gastrointestinal cancers. Ann Surg Oncol. 2008;15:3092–100. doi: 10.1245/s10434-008-0122-9. [DOI] [PubMed] [Google Scholar]
- 18.Uen YH, Lin SR, Wu CH, Hsieh JS, Lu CY, Yu FJ, Huang TJ, Wang JY. Clinical significance of MUC1 and c-Met RT-PCR detection of circulating tumor cells in patients with gastric carcinoma. Clin Chim Acta. 2006;367:55–61. doi: 10.1016/j.cca.2005.11.013. [DOI] [PubMed] [Google Scholar]
- 19.Koga T, Tokunaga E, Sumiyoshi Y, et al. Detection of circulating gastric cancer cells in peripheral blood using real time quantitative RT-PCR. Hepatogastroenterology. 2008;55:1131–5. [PubMed] [Google Scholar]
- 20.Yie SM, Lou B, Ye SR, et al. Detection of survivin-expressing circulating cancer cells (CCCs) in peripheral blood of patients with gastric and colorectal cancer reveals high risks of relapse. Ann Surg Oncol. 2008;15:3073–82. doi: 10.1245/s10434-008-0069-x. [DOI] [PubMed] [Google Scholar]
- 21.Wu CH, Lin SR, Yu FJ, Wu DC, Pan YS, Hsieh JS, Huang SY, Wang JY. Development of a high-throughput membrane-array method for molecular diagnosis of circulating tumor cells in patients with gastric cancers. Int J Cancer. 2006;119:373–9. doi: 10.1002/ijc.21856. [DOI] [PubMed] [Google Scholar]
- 22.Illert B, Fein M, Otto C, Cording F, Stehle D, Thiede A, Timmermann W. Disseminated tumor cells in the blood of patients with gastric cancer are an independent predictive marker of poor prognosis. Scand J Gastroenterol. 2005;40:843–9. doi: 10.1080/00365520510015557. [DOI] [PubMed] [Google Scholar]
- 23.Bertazza L, Mocellin S, Marchet A, Pilati P, Gabrieli J, Scalerta R, Nitti D. Survivin gene levels in the peripheral blood of patients with gastric cancer independently predict survival. J Transl Med. 2009;7:111. doi: 10.1186/1479-5876-7-111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cao W, Yang W, Li H, et al. Using detection of survivin-expressing circulating tumor cells in peripheral blood to predict tumor recurrence following curative resection of gastric cancer. J Surg Oncol. 2011;103:110–5. doi: 10.1002/jso.21777. [DOI] [PubMed] [Google Scholar]
- 25.Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med. 1998;17:2815–34. doi: 10.1002/(sici)1097-0258(19981230)17:24<2815::aid-sim110>3.0.co;2-8. [DOI] [PubMed] [Google Scholar]
- 26.Williamson PR, Smith CT, Hutton JL, Marson AG. Aggregate data meta-analysis with time-to-event outcomes. Stat Med. 2002;21:3337–51. doi: 10.1002/sim.1303. [DOI] [PubMed] [Google Scholar]
- 27.Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials. 2007;8:16. doi: 10.1186/1745-6215-8-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–60. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ito H, Inoue H, Sando N, et al. Prognostic impact of detecting viable circulating tumour cells in gastric cancer patients using a telomerase-specific viral agent: a prospective study. BMC Cancer. 2012;12:346. doi: 10.1186/1471-2407-12-346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Saad AA, Awed NM, Abd Elkerim NN, et al. Prognostic significance of E-cadherin expression and peripheral blood micrometastasis in gastric carcinoma patients. Ann Surg Oncol. 2010;17:3059–67. doi: 10.1245/s10434-010-1151-8. [DOI] [PubMed] [Google Scholar]
- 31.Yeh KH, Chen YC, Yeh SH, Chen CP, Lin JT, Cheng AL. Detection of circulating cancer cells by nested reverse transcription-polymerase chain reaction of cytokeratin-19 (K19) – possible clinical significance in advanced gastric cancer. Anticancer Res. 1998;18:1283–6. [PubMed] [Google Scholar]
- 32.Sumikura S, Ishigami S, Natsugoe S, et al. Disseminated cancer cells in the blood and expression of sialylated antigen in gastric cancer. Cancer Lett. 2003;200:77–83. doi: 10.1016/s0304-3835(03)00388-4. [DOI] [PubMed] [Google Scholar]
- 33.Zhao S, Liu Y, Zhang Q, et al. The prognostic role of circulating tumor cells (CTCs) detected by RT-PCR in breast cancer: a meta-analysis of published literature. Breast Cancer Res Treat. 2011;130:809–16. doi: 10.1007/s10549-011-1379-4. [DOI] [PubMed] [Google Scholar]
- 34.Zhang L, Wu G, Pantel K. Detection of circulating tumor cells by RT-PCR significantly associated with poor prognosis in breast cancer. Breast Cancer Res Treat. 2011;130:359–64. doi: 10.1007/s10549-011-1636-6. [DOI] [PubMed] [Google Scholar]
- 35.Mocellin S, Hoon D, Ambrosi A, Nitti D, Rossi CR. The prognostic value of circulating tumor cells in patients with melanoma: a systematic review and meta-analysis. Clin Cancer Res. 2006;12:4605–13. doi: 10.1158/1078-0432.CCR-06-0823. [DOI] [PubMed] [Google Scholar]
- 36.Wang FB, Yang XQ, Yang S, Wang BC, Feng MH, Tu JC. A higher number of circulating tumor cells (CTC) in peripheral blood indicates poor prognosis in prostate cancer patients – a meta-analysis. Asian Pac J Cancer Prev. 2011;12:2629–35. [PubMed] [Google Scholar]
- 37.Ring AE, Zabaglo L, Ormerod MG, Smith IE, Dowsett M. Detection of circulating epithelial cells in the blood of patients with breast cancer: comparison of three techniques. Br J Cancer. 2005;92:906–12. doi: 10.1038/sj.bjc.6602418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Saucedo-Zeni N, Mewes S, Niestroj R, et al. A novel method for the in vivo isolation of circulating tumor cells from peripheral blood of cancer patients using a functionalized and structured medical wire. Int J Oncol. 2012;41:1241–50. doi: 10.3892/ijo.2012.1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hou JM, Krebs M, Ward T, et al. Circulating tumor cells as a window on metastasis biology in lung cancer. Am J Pathol. 2011;178:989–96. doi: 10.1016/j.ajpath.2010.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Takeuchi H, Kitagawa Y. Circulating tumor cells in gastrointestinal cancer. J Hepatobiliary Pancreat Sci. 2010;17:577–82. doi: 10.1007/s00534-009-0193-4. [DOI] [PubMed] [Google Scholar]
- 41.Paterlini-Brechot P, Benali NL. Circulating tumor cells (CTC) detection: clinical impact and future directions. Cancer Lett. 2007;253:180–204. doi: 10.1016/j.canlet.2006.12.014. [DOI] [PubMed] [Google Scholar]