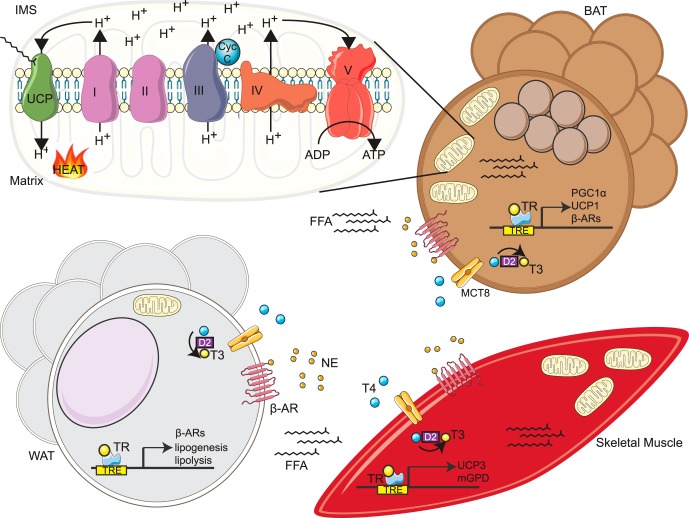
Homoeothermic species engage thermogenic components of cellular metabolism to both maintain and raise core body temperature (Tc) when exposed to cold, feeding, infection, and certain classes of drugs (sympathomimetics). Body heat production stems from two main thermogenic mechanisms: 1) obligatory thermogenesis, or the heat produced as a collective byproduct of the inefficiency of all cellular metabolic reactions, and 2) facultative thermogenesis (also referred to as adaptive or nonshivering thermogenesis [NST]), the additional heat generation required along with obligatory thermogenesis to rapidly increase body temperature. Thyroid hormone (TH) is the well-established principal endocrine regulator of both obligatory and facultative thermogenesis. Here we aim to provide a brief overview of the mechanisms by which TH regulates facultative thermogenesis and to highlight recent studies describing a role for TH in the regulation of thermogenic mitochondrial uncoupling proteins. For a broader overview of the role of the endocrine control of thermoregulation, we would refer readers to the excellent review by Silva (1).
Facultative thermogenesis is controlled by both a temporary shivering (muscle contraction) component and by more sustained nonshivering mechanisms. NST is regulated primarily by both the circulating levels of TH and the acute activity of the sympathetic nervous system (SNS) and mainly occurs in brown adipose tissue (BAT) and skeletal muscle (2). The primary role of TH in NST regulation is a permissive one; it chronically sets the capacity of thermogenic and metabolic responses to SNS stimulation through its transcriptional regulation of a vast repertoire of metabolic genes (3–5). In contrast, the SNS mainly controls the acute activity of thermogenic signaling pathways culminating in the activation of heat-generating proteins downstream of β-adrenergic receptors (6, 7). Owing to the fundamental importance of TH in human thermoregulation, hyper- and hypothyroid patients are heat and cold intolerant, respectively (8). Similarly, hypothyroid animal models demonstrate that the TH is absolutely required for a normal thermogenic response to both cold and sympathomimetic stimulation (9).
TH is synthesized in the thyroid gland and controlled by thyroid peroxidase activity that regulates the iodination, coupling, and ultimately proteolysis of tyrosine residues on thyroglobulin to release the THs, T4 and T3, into the bloodstream (10). The lesser active T4 is released from the thyroid gland at higher concentrations than the more active T3 and is locally converted to T3 in target tissues by the actions of tissue-specific deiodinases (11–13). Two genes, THRA and THRB, are responsible for the expression of distinct thyroid hormone receptors (TRs), each of which are alternatively spliced to produce multiple isoforms, TRα1, TRα2, TRβ1, and TRβ2, respectively (14–16). With the exception of TRα2, which does not bind T3 and functions to repress T3 actions, TR isoforms mediate distinct functions (both stimulatory and repressive) in response to and in the absence of T3 (2). Integral to their functions as transcriptional regulators, TRs bind other nuclear hormone receptors, coactivators, and corepressors, the details of which have been reviewed elsewhere (2). In addition to its transcriptional regulation, recent work has also revealed that TH may regulate cell signaling pathways nongenomically (17, 18). However, it is not yet established whether and how TH may influence body temperature apart from its role as a ligand for thyroid hormone receptor-dependent gene transactivation.
Uncoupling protein (UCP) mediators of thermogenesis
Mitochondrial uncoupling proteins are highly conserved, nuclear-encoded, six-transmembrane-spanning members of the mitochondrial solute carrier family localized to the inner mitochondrial membrane (19). The prototypical uncoupling protein, UCP1 (previously referred to as thermogenin), was first characterized more than 3 decades ago as the first protein established to mediate cold-, SNS-, and TH-stimulated BAT thermogenesis (6, 20, 21). UCP1 functions ostensibly as a mitochondrial inner membrane proton carrier that, when activated, dissipates the electrochemical proton gradient, decreasing the proton motive force used to synthesize ATP. Proton leak regulated by UCP1 thereby uncouples the proton gradient from ATP synthesis, with the free energy released as heat (22, 23). UCP1 is activated by free fatty acids released from SNS-dependent lipolysis in both white adipose tissue (WAT) and BAT (24–27). In turn, UCP1 activation increases fatty acid oxidation to compensate for respiratory inefficiency and decreased mitochondrial membrane potential (28). UCP1 has classically been thought to be the primary, if not only, physiologically relevant thermogenic protein in mammals. However, its expression is largely, if not exclusively, confined to BAT and beige adipocytes (discussed later) (29), and because of its relatively small size in most adult large mammals, the significance of BAT as a general mediator of NST in humans is debated.
In 1997, 2 decades after UCP1 was initially characterized, the UCP1 homologs UCP2 and UCP3 were identified with relatively broader expression relative to UCP1 (30–32). UCP2 appears to have wide expression in the brain and periphery, including in dividing cells, and its levels are induced by TH in heart and skeletal muscle (33–35). In contrast, UCP3 expression is more restricted to skeletal muscle, heart, and BAT and as we have recently observed in murine epidermis (30, 36, 37). Whereas UCP1 has been established as a physiological mediator of cold thermogenesis, the specific thermogenic functions of UCP2 and UCP3 are currently debated (38). Genetic overexpression and knockout studies in both cells and tissues have demonstrated that these novel UCPs are, like UCP1, also associated with markers of proton leak, including decreased mitochondrial membrane potential and mitigation of reactive oxidant generation, and both homologs are activated by free fatty acids (28, 36, 39, 40). Observations that UCP3 protein is particularly enriched in skeletal muscle led to the early assumption that it functions in a similar manner to BAT UCP1 to control NST, and this was supported by reports of strong induction of UCP3 by TH in skeletal muscle (5, 30, 33, 41). However, unlike UCP1 knockout mice (42), cold-exposed UCP3-null mice (under normal dietary conditions) were able to maintain their body temperatures similar to wild-type mice (43). This observation guided the vast majority of researchers in the field to conclude that UCP3 is not a significant thermoregulatory mediator in mice. Whereas this finding confirmed the necessity and sufficiency of UCP1 in cold-induced NST, it did not rule out the possibility that UCP3 mediates NST in response to other stimuli and the regulation by TH. Consistent with this hypothesis, UCP3 knockout animals administered T3 produce less heat than wild-type animals (44).
The role of UCP2 as an effector of thermogenesis and TH actions has not been established. Although there are reports of TH induction of UCP2 mRNA in skeletal muscle, whether UCP2 protein is present in muscle or BAT mitochondria or corresponds to changes in mRNA levels, is unclear. Interestingly, very recent work suggests that UCP2 may function in the export of C4 metabolites from mitochondria and may actually thereby decrease mitochondrial oxidative functions, suggesting that UCP2 actions could actually oppose thermogenesis (45). More work is needed to understand the significance of TH regulation of UCP2 and its pertinence to NST. Two other brain-expressed homologues have been identified (UCP4 and the brain mitochondrial carrier protein 1 [BMCP1/UCP5]), both of which have more distant homology (<40% amino acid) with UCP1–3 (46–48). These isoforms have not been well studied with respect to TH regulation but are thought to act as regulators of mitochondrial biogenesis, calcium flux, and neurotransmission (49). It is entirely conceivable that UCP4 and UCP5, like UCP2, could act as central regulators of body temperature circuitry and in at least some neuronal cell types be regulated by TH.
TH and the regulation of UCP1-dependent thermogenesis in BAT
Regulation of BAT thermogenesis by T3 is initiated by the actions of type II deiodinases (D2) expressed locally that convert T4 to T3 and, importantly, that are inhibited by high T4 levels (12, 50, 51). The expression of D2 in BAT is induced by norepinephrine in response to cold exposure and is mediated by the activation of β1/β2/β3 adrenergic receptors, leading to the elevation of cAMP and protein kinase A activity (9, 11, 13). Consistent with the requirement for T3 in cold-induced BAT thermogenesis, mice lacking D2 exhibit impaired thermogenesis and hypothermia in response to cold exposure despite markers of chronic SNS stimulation (52). Similarly, the treatment of rodents with norepinephrine (NE), the main acute mediator of BAT thermogenesis, failed to produce an increase in BAT heat generation in hypothyroid animals, but NST could be rescued by subchronic T3 or T4 replacement (8, 53). Importantly, the adrenergic receptors as a class are induced by TH, supporting the notion that TH and adrenergic signaling synergize to control the maximal output of heat during BAT-regulated NST (54–56). In support of this, β-less mice lacking all β-adrenergic receptors (β1/β2/β3 [TKO]) have reduced UCP1 expression and D2 activity in BAT as well as lowered Tc and reduced cold tolerance that can be rescued by acute T3 administration in a manner partially dependent on skeletal muscle UCP3 (44, 57). Notably, however, high levels of T4 may suppress BAT function while stimulating thermogenic activity in skeletal muscle in both rodents and humans (58).
The TR receptors have divergent roles in the regulation of BAT NST. Whereas TRα appears to be most important for the regulation of adrenergic sensitivity, TRβ is critical for the induction of UCP1 gene expression (5, 59). TRα−/− mice (lacking both TRα1 and TRα2) exhibit significantly decreased basal body temperatures that are further decreased by acute cold exposure (60, 61). In contrast, TRβ−/− (lacking both TRβ1 and TRβ2) have normal body temperatures and cold responses (60, 62). Both UCP1 and the master mitochondrial biogenesis regulatory transcription factor, peroxisome proliferator-activated receptor-γ coactivator 1-α (PGC1α) mRNA levels are induced in BAT upon adrenergic stimulation, and PGC1α has been demonstrated to bind and activate TRβ/retinoic acid receptor heterodimers (63). Interestingly, compared with the wild type, TRα1−/− mice show no difference in mRNA levels of UCP1 or PGC1α in BAT at 22°C or after acute cold exposure, suggesting that TRα1 is dominant for NST activation but is not required for induction of UCP1 or PGC1α under these conditions (64). In contrast, the TRβ receptor drives the up-regulation of PGC1α and other mitochondrial metabolic machinery components in BAT in response to T3 production (65).
Recent studies have also demonstrated the presence of UCP1-containing cells in sc WAT in mice and humans stimulated by cold, β-AR agonists, or peroxisome proliferator-activated receptor-γ activators (66–69). These brown-in-white (Brite) or beige adipocytes are present in low numbers compared with white adipocytes and, when fully stimulated, express levels of UCP1 and display uncoupled respiration similar to brown adipocytes (69). Although most evidence suggests that SNS stimulation is the primary effector of beige adipocyte activation, recent studies suggests that T3 treatment can also induce UCP1 expression and increase oxygen consumption in human multipotent adipocytes (70). The T3-mediated regulation of metabolism in WAT has been well studied in mouse and human models and have demonstrated the requirement of T3 for lipogenesis and triglyceride storage and ultimately adipocyte differentiation (71). Additionally, T3 is required for the SNS-mediated elevation of cAMP levels, protein kinase A activation, and lipase activity in WAT, leading to free fatty acid release in response to cold exposure or other thermogenic stimuli (56). These studies indicate the necessity for T3 in the metabolic regulation of white adipocytes to maximize the β-AR stimulation and suggest that beige precursors in WAT depots may also respond to T3 as part of the browning process.
TH in skeletal muscle NST
Unlike rodents and hibernators in which BAT has a significant metabolic function across development, many large mammals, including humans, retain only a small amount, if any, BAT into adulthood. Indeed, despite recent observations suggesting that BAT may play a greater role in human thermoregulation than previously thought, its small size relative to total body weight in most adults has challenged the BAT-centric view of human NST (24). The role of TH in BAT function may also differ between humans and rodents. In rats, there is an absolute dependence and linear relationship between TH levels and sympathomimetic thermogenesis (72). However, a recent positron emission tomography-computed tomography study using 18F-fluorodeoxyglucose as a BAT activity tracer in normal and hyperthyroid human patients failed to detect any amounts of BAT activity in the hyperthyroid state, but instead skeletal muscle glucose uptake was significantly augmented in these patients, suggesting a role for skeletal muscle as a significant site of TH-regulated thermogenesis in humans (36, 73, 74). A similar study showed that humans administered the thermogenic sympathomimetic drug ephedrine failed to show an activation of BAT (75).
The contribution of skeletal muscle to NST is most apparent in mice lacking all β-adrenergic receptors (TKO), which have decreased Tc and cold intolerance that can be rescued upon acute T3 stimulation (44, 57). These mice lack BAT activity but have increased UCP3 expression in skeletal muscle (SKM) upon T3 administration, suggesting that TH actions in SKM may be sufficient to rescue cold tolerance in the absence of SNS stimulation and BAT thermogenesis (44, 57). Similar to the regulation of BAT UCP1, TH is essential for a maximal thermogenic response in skeletal muscle (36, 76). Indeed, euthyroid skeletal muscle generates more heat than hypothyroid muscle (77). This effect may result in part from the TH-dependent regulation of free fatty acid (FFA) levels in SKM, leading to the activation of uncoupling proteins. Indeed, SKM mitochondria from hyperthyroid rats display increased proton leak that is abolished when FFAs are removed (78). Additionally, because of its size in humans, skeletal muscle also contributes significantly to NST responses through its tonic control of body temperature via obligatory thermogenesis. A current model for TH-mediated SKM heat production is one in which TH regulates the expression of thermogenic machinery, and SNS stimulation activates the machinery via increased D2 activity and FFA release from WAT, which activates UCP3 and stimulates non-UCP-mediated thermogenesis (discussed below) (5, 36, 40, 52, 79). The role of SNS stimulation in the promotion of NST through UCP3 activity in SKM is supported by phenotypes in UCP3−/− mice. The finding that sympathomimetic drugs (3,4-methylenedioxy-methamphetamine, methamphetamine) fail to elicit a thermogenic response in UCP3−/− mice demonstrates that there are conditions in which UCP1 expression cannot compensate for lack of UCP3. Sympathomimetic agents also fail to elicit thermogenic responses in hypothyroid mice, suggesting that in skeletal muscle, as in BAT, SNS stimulation is insufficient to compensate for a lack of normal TH levels and that TH is necessary for a full thermogenic response in SKM as it is in BAT (72). Finally, unpublished data from our laboratory indicates that SKM-specific overexpression of UCP3 is sufficient to rescue a thermogenic response to methamphetamine normally lost in UCP3−/− mice.
UCP-independent mechanisms of TH mediated thermogenesis
As noted above, the only established physiological NST mechanism involves the activation of brown adipocyte UCP1. However, in addition to evidence that skeletal muscle UCP3 may also participate in NST, UCP-independent NST mechanisms can also play a role in TH-regulated thermoregulation (1, 80). For example, the glycerol 3-phosphate shuttle, dihydroxyacetone phosphate is reduced to glycerol 3-phosphate (G3P) in the cytosol, and G3P is then reoxidized to dihydroxyacetone phosphate by mitochondrial glycerol 3-phosphate dehydrogenase (mGPD), a flavin adenine dinucleotide-linked enzyme that donates electrons directly to complex III of the respiratory chain (81, 82). Because the G3P shuttle's electrons bypass complex I for ATP synthesis, the process is inherently inefficient, and therefore thermogenic (1). The G3P shuttle is a predominant metabolic pathway in skeletal muscle and BAT and is stimulated by TH (1). Knockout of the mitochondrial GPD (mGPD−/−) in mice leads to decreased energy turnover (intake/oxygen consumption) associated with increases in serum T3 and T4 levels as well as chronic SNS stimulation of BAT (83, 84). These data suggest that these animals have a reduced energy turnover due to cold stress from reduction of non-UCP-mediated heat generating mechanisms in thermogenic tissues. Furthermore, these animals display a TH-dependent, increased expression of UCP3 in both BAT and skeletal muscle, indicating a UCP3-dependent compensation mechanism in these tissues during both obligatory and facultative thermogenesis (84). Another example of a UCP-independent NST pathway has been demonstrated recently linking TRα1 with control of vascular function as a mechanism to regulate heat dissipation and conservation. Mice with a heterozygous mutation in TRα1 (R384C) exhibit decreased Tc at night despite increased heat production from BAT. In this model, decreased Tc results from increased heat dissipation from the tail in response to a failure of arterial constriction in the tail after SNS stimulation (85). These data indicate a clear role for TH and TRs in thermoregulation outside BAT and SKM and independent of UCP actions.
Concluding remarks and future directions
An overview of the various mouse models and their phenotypes relevant to TH and UCP actions are summarized in Table 1. In response to cold exposure, NE released from the hypothalamus binds β3-adrenergic receptors in BAT, WAT, and skeletal muscle, increasing FFA release, the known substrates/second messengers for thermogenesis along with an induction/activation of UCP1/3 and other potential thermogenic genes. Simultaneously, SNS stimulation activates D2 deiodinases in BAT and skeletal muscle that increase T3 levels, leading to the transactivation via TRs of thermogenic genes that ultimately govern the thermogenic capacities of BAT and skeletal muscle (Figure 1). The activities of both UCP1 and UCP3 are stimulated by FFAs, and increased TH signaling in white adipose tissue is required for lipolysis and the liberation of FFAs and may be associated with beige adipocyte activation. Moreover, skeletal muscle UCP3 is induced by conditions of increased fatty acid oxidation demand, including exercise, fasting, increased TH signaling, and obesity. In addition, UCP3 expression is increased in BAT and skeletal muscle, in a TH-dependent manner, upon the reduction of additional thermogenic pathways. More work in genetically modified mice harboring tissue-selective deletions in TH receptors and UCPs subjected to thermoregulatory and metabolic challenges will be necessary to more clearly dissect the roles of UCPs as determinants of TH functions.
Table 1.
Mouse Models of TH and UCP Thermogenesis
| Genetic Mouse Models | Obligatory Thermogenic Phenotype | Facultative Thermogenic Phenotype | References |
|---|---|---|---|
| UCP1−/− | Normal baseline temperature | Hypothermia in response to cold | Enerback et al (1997) (42) |
| Gong et al (2000) (36) | |||
| Golozoubova et al (2006) | |||
| UCP3−/− | Normal baseline temperature | Normal thermogenesis in response to cold, hypothermia in response to sympathomimetics | Mills et al (2003) (72) |
| Vidal-Puig et al (2000) (43) | |||
| Flandin et al (2009) (44) | |||
| TRα1−/− | Reduced baseline temperature | Normal thermogenesis in response to cold | Wikström et al (1998) (61) |
| Golozoubova et al (2003) | |||
| TRa 0/0 | Reduced baseline temperature | Hypothermia in response to cold | Marrif et al (2005) (64) |
| Pelletier et al (2008) (86) | |||
| TRβ−/− | Normal baseline temperatures | Normal response to thermogenesis | Johnsson et al (1999) (62) |
| Golozoubova et al (2003) | |||
| TRα1/TRβ−/− | 0.4ºC decrease in core body temperature | Hypothermia in response to cold | Johansson et al (1999) (62) |
| Golozoubova et al (2003) | |||
| D2−/− (D2KO) | Normal baseline temperature | Cold exposure leads to increased SNS stimulation of BAT but mice are hypothermic | Schneider et al (2001) (87) |
| Impaired embryonic BAT differentiation results in defective BAT function in adult mice | de Jesus et al (2001) (50) | ||
| Christoffolete et al (2004) (52) | |||
| Hall (2010) (88) | |||
| Castillo et al (2011) (89) | |||
| β1/β2/β3−/− (TKO) | 1.2ºC decrease in core body temperature | Hypothermia in response to cold, rescued by acute T3 administration | Jimenez et al (2002) (57) |
| Flandin et al (2009) (44) |
Abbreviation: D2KO, mice lacking D2.
Figure 1.
Tissue specific mechanisms of TH-mediated thermogenesis. In response to cold exposure, NE released from SNS nerve terminals binds β3-adrenergic receptors (β-AR) in BAT and WAT, increasing local and systemic FFA release along with an induction/activation of UCP1/3 and other potential thermogenic genes in BAT and skeletal muscle, respectively. Simultaneously, SNS stimulation activates D2 deiodinases in BAT and skeletal muscle that increase T3 levels, leading to the transactivation via TRs of thermogenic genes that ultimately govern the thermogenic capacities of BAT and skeletal muscle including PGC1α and mGPD. In BAT, UCP1 is activated by FFA release to transport protons from the mitochondrial intermembrane space (IMS) to the matrix, dissipating the proton gradient to produce heat. Similarly, we propose that skeletal muscle NST is activated in part by the uptake of FFA released from SNS-stimulated WAT lipolysis and UCP3 activation. MCT8, monocarboxylate transporter 8.
Acknowledgments
This work was funded in part by the National Institutes of Health.
Disclosure Summary: The authors have nothing to disclose.
Footnotes
- BAT
- brown adipose tissue
- D2
- type II deiodinase
- FFA
- free fatty acid
- G3P
- glyceraldehyde 3-phosphate
- mGPD
- mitochondrial glycerol 3-phosphate dehydrogenase
- NE
- norepinephrine
- NST
- nonshivering thermogenesis
- PGC1α
- peroxisome proliferator-activated receptor-γ coactivator 1-α
- SKM
- skeletal muscle
- SNS
- sympathetic nervous system
- Tc
- core body temperature
- TH
- thyroid hormone
- TKO
- mice lacking all β-adrenergic receptors
- TR
- thyroid hormone receptor
- UCP
- uncoupling protein
- WAT
- white adipose tissue.
References
- 1. Silva JE. Thermogenic mechanisms and their hormonal regulation. Physiol Rev. 2006;86(2):435–464. [DOI] [PubMed] [Google Scholar]
- 2. Mullur R, Liu Y-Y, Brent GA. Thyroid hormone regulation of metabolism. Physiol Rev. 2014;94(2):355–382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Harper ME, Brand MD. The quantitative contributions of mitochondrial proton leak and ATP turnover reactions to the changed respiration rates of hepatocytes from rats of different thyroid status. J Biol Chem. 1993;268(20):14850–14860. [PubMed] [Google Scholar]
- 4. Oppenheimer JH, Schwartz HL, Lane JT, Thompson MP. Functional relationship of thyroid hormone-induced lipogenesis, lipolysis, and thermogenesis in the rat. J Clin Invest. 1991;87(1):125–132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Solanes G, Pedraza N, Calvo V, Vidal-Puig A, Lowell BB, Villarroya F. Thyroid hormones directly activate the expression of the human and mouse uncoupling protein-3 genes through a thyroid response element in the proximal promoter region. Biochem J. 2005;386(Pt 3):505–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Cannon B. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84(1):277–359. [DOI] [PubMed] [Google Scholar]
- 7. Astrup A, Lundsgaard C, Madsen J, Christensen NJ. Enhanced thermogenic responsiveness during chronic ephedrine treatment in man. Am J Clin Nutr. 1985;42(1):83–94. [DOI] [PubMed] [Google Scholar]
- 8. Silva JE. The thermogenic effect of thyroid hormone and its clinical implications. Ann Intern Med. 2003;139(3):205–213. [PubMed] [Google Scholar]
- 9. Silva JE, Larsen PR. Adrenergic activation of triiodothyronine production in brown adipose tissue. Nature. 1983;305(5936):712–713. [DOI] [PubMed] [Google Scholar]
- 10. Dumont JE, Willems C, Van Sande J, Nève P. Regulation of the release of thyroid hormones: role of cyclic AMP. Ann NY Acad Sci. 1971;185:291–316. [DOI] [PubMed] [Google Scholar]
- 11. Silva JE, Larsen PR. Hormonal regulation of iodothyronine 5′-deiodinase in rat brown adipose tissue. Am J Physiol. 1986;251(6 Pt 1):E639–E643. [DOI] [PubMed] [Google Scholar]
- 12. Bianco AC, Silva JE. Intracellular conversion of thyroxine to triiodothyronine is required for the optimal thermogenic function of brown adipose tissue. J Clin Invest. 1987;79(1):295–300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Drigo RAE, Fonseca TL, Werneck-de-Castro JPS, Bianco AC. Role of the type 2 iodothyronine deiodinase (D2) in the control of thyroid hormone signaling. Biochim Biophys Acta. 2013;1830(7):3956–3964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Weinberger C, Thompson CC, Ong ES, Lebo R, Gruol DJ, Evans RM. The c-erb-A gene encodes a thyroid hormone receptor. Nature. 1986;324(6098):641–646. [DOI] [PubMed] [Google Scholar]
- 15. Sap J, Muñoz A, Damm K, et al. The c-erb-A protein is a high-affinity receptor for thyroid hormone. Nature. 1986;324(6098):635–640. [DOI] [PubMed] [Google Scholar]
- 16. Benbrook D, Pfahl M. A novel thyroid hormone receptor encoded by a cDNA clone from a human testis library. Science. 1987;238(4828):788–791. [DOI] [PubMed] [Google Scholar]
- 17. Cao X, Kambe F, Moeller LC, Refetoff S, Seo H. Thyroid hormone induces rapid activation of Akt/protein kinase B-mammalian target of rapamycin-p70 S6K cascade through phosphatidylinositol 3-kinase in human fibroblasts. Mol Endocrinol. 2005;19(1):102–112. [DOI] [PubMed] [Google Scholar]
- 18. Bergh JJ, Lin H-Y, Lansing L, et al. Integrin αVβ3 contains a cell surface receptor site for thyroid hormone that is linked to activation of mitogen-activated protein kinase and induction of angiogenesis. Endocrinology. 2005;146(7):2864–2871. [DOI] [PubMed] [Google Scholar]
- 19. Jezek P, Jezek J. Sequence anatomy of mitochondrial anion carriers. FEBS Lett. 2003;534(1–3):15–25. [DOI] [PubMed] [Google Scholar]
- 20. Lin CS, Klingenberg M. Isolation of the uncoupling protein from brown adipose tissue mitochondria. FEBS Lett. 1980;113(2):299–303. [DOI] [PubMed] [Google Scholar]
- 21. Klingenberg M, Lin CS. Isolation and hydrodynamic characterization of the uncoupling protein from brown adipose tissue. Methods Enzymol. 1986;126:490–498. [DOI] [PubMed] [Google Scholar]
- 22. Klingenberg M, Winkler E. Reconstitution of an H+ translocator, the “uncoupling protein” from brown adipose tissue mitochondria, in phospholipid vesicles. Methods Enzymol. 1986;127:772–779. [DOI] [PubMed] [Google Scholar]
- 23. Klingenberg M, Winkler E. The reconstituted isolated uncoupling protein is a membrane potential driven H+ translocator. EMBO J. 1985;4(12):3087–3092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Blondin DP, Labbé SM, Phoenix S, et al. Contributions of white and brown adipose tissues and skeletal muscles to acute cold-induced metabolic responses in healthy men. J Physiol (Lond). 2015;593(3):701–714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Cunningham SA, Wiesinger H, Nicholls DG. Quantification of fatty acid activation of the uncoupling protein in brown adipocytes and mitochondria from the guinea pig. Eur J Biochem. 1986;157(2):415–420. [DOI] [PubMed] [Google Scholar]
- 26. Strieleman PJ, Schalinske KL, Shrago E. Fatty acid activation of the reconstituted brown adipose tissue mitochondria uncoupling protein. J Biol Chem. 1985;260(25):13402–13405. [PubMed] [Google Scholar]
- 27. Deykin D, Vaughan M. Release of free fatty acids by adipose tissue from rats treated with triiodothyronine or propylthiouracil. J Lipid Res. 1963;4(2):200–203. [PubMed] [Google Scholar]
- 28. Wang S, Subramaniam A, Cawthorne MA, Clapham JC. Increased fatty acid oxidation in transgenic mice overexpressing UCP3 in skeletal muscle. Diabetes Obes Metab. 2003;5(5):295–301. [DOI] [PubMed] [Google Scholar]
- 29. Cannon B, Hedin A, Nedergaard J. Exclusive occurrence of thermogenin antigen in brown adipose tissue. FEBS Lett. 1982;150(1):129–132. [DOI] [PubMed] [Google Scholar]
- 30. Vidal-Puig A, Solanes G, Grujic D, Flier JS, Lowell BB. UCP3: an uncoupling protein homologue expressed preferentially and abundantly in skeletal muscle and brown adipose tissue. Biochem Biophys Res Commun. 1997;235(1):79–82. [DOI] [PubMed] [Google Scholar]
- 31. Boss O, Samec S, Paoloni-Giacobino A, et al. Uncoupling protein-3: a new member of the mitochondrial carrier family with tissue-specific expression. FEBS Lett. 1997;408(1):39–42. [DOI] [PubMed] [Google Scholar]
- 32. Fleury C, Neverova M, Collins S, et al. Uncoupling protein-2: a novel gene linked to obesity and hyperinsulinemia. Nat Genet. 1997;15(3):269–272. [DOI] [PubMed] [Google Scholar]
- 33. Lanni A, Moreno M, Lombardi A, Goglia F. Thyroid hormone and uncoupling proteins. FEBS Let. 2003;543(1–3):5–10. [DOI] [PubMed] [Google Scholar]
- 34. Barbe P, Larrouy D, Boulanger C, et al. Triiodothyronine-mediated up-regulation of UCP2 and UCP3 mRNA expression in human skeletal muscle without coordinated induction of mitochondrial respiratory chain genes. FASEB J. 2001;15(1):13–15. [DOI] [PubMed] [Google Scholar]
- 35. Masaki T, Yoshimatsu H, Kakuma T, Hidaka S, Kurokawa M, Sakata T. Enhanced expression of uncoupling protein 2 gene in rat white adipose tissue and skeletal muscle following chronic treatment with thyroid hormone. FEBS Lett. 1997;418(3):323–326. [DOI] [PubMed] [Google Scholar]
- 36. Gong DW, He Y, Karas M, Reitman M. Uncoupling protein-3 is a mediator of thermogenesis regulated by thyroid hormone, β3-adrenergic agonists, and leptin. J Biol Chem. 1997;272(39):24129–24132. [DOI] [PubMed] [Google Scholar]
- 37. Lago CU, Nowinski SM, Rundhaug JE, et al. Mitochondrial respiratory uncoupling promotes keratinocyte differentiation and blocks skin carcinogenesis. Oncogene. 2012;31(44):4725–4731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Matthias A, Ohlson KB, Fredriksson JM, Jacobsson A, Nedergaard J, Cannon B. Thermogenic responses in brown fat cells are fully UCP1-dependent. UCP2 or UCP3 do not substitute for UCP1 in adrenergically or fatty SCID-induced thermogenesis. J Biol Chem. 2000;275(33):25073–25081. [DOI] [PubMed] [Google Scholar]
- 39. Bezaire V, Spriet LL, Campbell S, et al. Constitutive UCP3 overexpression at physiological levels increases mouse skeletal muscle capacity for fatty acid transport and oxidation. FASEB J. 2005;19(8):977–979. [DOI] [PubMed] [Google Scholar]
- 40. MacLellan JD, Gerrits MF, Gowing A, Smith PJS, Wheeler MB, Harper M-E. Physiological increases in uncoupling protein 3 augment fatty acid oxidation and decrease reactive oxygen species production without uncoupling respiration in muscle cells. Diabetes. 2005;54(8):2343–2350. [DOI] [PubMed] [Google Scholar]
- 41. Larkin S, Mull E, Miao W, et al. Regulation of the third member of the uncoupling protein family, UCP3, by cold and thyroid hormone. Biochem Biophys Res Commun. 1997;240(1):222–227. [DOI] [PubMed] [Google Scholar]
- 42. Enerback S, Jacobsson A, Simpson EM, Guerra C. Mice lacking mitochondrial uncoupling protein are cold-sensitive but not obese. Nature. 1997;387(6628):90–94. [DOI] [PubMed] [Google Scholar]
- 43. Vidal-Puig AJ, Grujic D, Zhang CY, et al. Energy metabolism in uncoupling protein 3 gene knockout mice. J Biol Chem. 2000;275(21):16258–16266. [DOI] [PubMed] [Google Scholar]
- 44. Flandin P, Lehr L, Asensio C, et al. Uncoupling protein-3 as a molecular determinant of the action of 3,5,3′-triiodothyronine on energy metabolism. Endocrine. 2009;36(2):246–254. [DOI] [PubMed] [Google Scholar]
- 45. Vozza A, Parisi G, De Leonardis F, et al. UCP2 transports C4 metabolites out of mitochondria, regulating glucose and glutamine oxidation. Proc Natl Acad Sci USA. 2014;111(3):960–965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Mao W, Yu XX, Zhong A, et al. UCP4, a novel brain-specific mitochondrial protein that reduces membrane potential in mammalian cells. FEBS Lett. 1999;443(3):326–330. [DOI] [PubMed] [Google Scholar]
- 47. Sanchis D, Fleury C, Chomiki N, et al. BMCP1, a novel mitochondrial carrier with high expression in the central nervous system of humans and rodents, and respiration uncoupling activity in recombinant yeast. J Biol Chem. 1998;273(51):34611–34615. [DOI] [PubMed] [Google Scholar]
- 48. Yu XX, Mao W, Zhong A, et al. Characterization of novel UCP5/BMCP1 isoforms and differential regulation of UCP4 and UCP5 expression through dietary or temperature manipulation. FASEB J. 2000;14(11):1611–1618. [DOI] [PubMed] [Google Scholar]
- 49. Andrews ZB, Diano S, Horvath TL. Mitochondrial uncoupling proteins in the CNS: in support of function and survival. Nat Rev Neurosci. 2005;6(11):829–840. [DOI] [PubMed] [Google Scholar]
- 50. de Jesus LA, Carvalho SD, Ribeiro MO, et al. The type 2 iodothyronine deiodinase is essential for adaptive thermogenesis in brown adipose tissue. J Clin Invest. 2001;108(9):1379–1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Carvalho SD, Kimura ET, Bianco AC, Silva JE. Central role of brown adipose tissue thyroxine 5′-deiodinase on thyroid hormone-dependent thermogenic response to cold. Endocrinology. 1991;128(4):2149–2159. [DOI] [PubMed] [Google Scholar]
- 52. Christoffolete MA, Linardi CCG, de Jesus L, et al. Mice with targeted disruption of the Dio2 gene have cold-induced overexpression of the uncoupling protein 1 gene but fail to increase brown adipose tissue lipogenesis and adaptive thermogenesis. Diabetes. 2004;53(3):577–584. [DOI] [PubMed] [Google Scholar]
- 53. Silva JE. The multiple contributions of thyroid hormone to heat production. J Clin Invest. 2001;108(1):35–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Bianco AC, Sheng XY, Silva JE. Triiodothyronine amplifies norepinephrine stimulation of uncoupling protein gene transcription by a mechanism not requiring protein synthesis. J Biol Chem. 1988;263(34):18168–18175. [PubMed] [Google Scholar]
- 55. Branco M, Ribeiro M, Negrão N, Bianco AC. 3,5,3′-Triiodothyronine actively stimulates UCP in brown fat under minimal sympathetic activity. Am J Physiol. 1999;276(1 Pt 1):E179–E187. [DOI] [PubMed] [Google Scholar]
- 56. Rubio A, Raasmaja A, Maia AL, Kim KR, Silva JE. Effects of thyroid hormone on norepinephrine signaling in brown adipose tissue. I. β1- and β2-adrenergic receptors and cyclic adenosine 3′,5′-monophosphate generation. Endocrinology. 1995;136(8):3267–3276. [DOI] [PubMed] [Google Scholar]
- 57. Jimenez M, Léger B, Canola K, et al. β1/β2/β3-adrenoceptor knockout mice are obese and cold-sensitive but have normal lipolytic responses to fasting. FEBS Lett. 2002;530(1–3):37–40. [DOI] [PubMed] [Google Scholar]
- 58. Matsukawa T, Mano T, Gotoh E, Minamisawa K, Ishii M. Altered muscle sympathetic nerve activity in hyperthyroidism and hypothyroidism. J Auton Nerv Syst. 1993;42(2):171–175. [DOI] [PubMed] [Google Scholar]
- 59. Rabelo R, Reyes C, Schifman A, Silva JE. Interactions among receptors, thyroid hormone response elements, and ligands in the regulation of the rat uncoupling protein gene expression by thyroid hormone. Endocrinology. 1996;137(8):3478–3487. [DOI] [PubMed] [Google Scholar]
- 60. Golozoubova V, Gullberg H, Matthias A, Cannon B, Vennström B, Nedergaard J. Depressed thermogenesis but competent brown adipose tissue recruitment in mice devoid of all hormone-binding thyroid hormone receptors. Mol Endocrinol. 2004;18(2):384–401. [DOI] [PubMed] [Google Scholar]
- 61. Wikström L, Johansson C, Saltó C, et al. Abnormal heart rate and body temperature in mice lacking thyroid hormone receptor alpha 1. EMBO J. 1998;17(2):455–461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Johansson C, Göthe S, Forrest D. Cardiovascular phenotype and temperature control in mice lacking thyroid hormone receptor-β or both α1 and β. Am J Physiol. 1999;276(6 Pt 2):H2006–H2012. [DOI] [PubMed] [Google Scholar]
- 63. Puigserver P, Wu Z, Park CW, Graves R, Wright M. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell. 1998;92(6):829–839. [DOI] [PubMed] [Google Scholar]
- 64. Marrif H, Schifman A, Stepanyan Z, Gillis M-A, et al. Temperature homeostasis in transgenic mice lacking thyroid hormone receptor-α gene products. Endocrinology. 2005;146(7):2872–2884. [DOI] [PubMed] [Google Scholar]
- 65. Ribeiro MO, Carvalho SD, Schultz JJ, Chiellini G, Scanlan TS, Bianco AC, Brent GA. Thyroid hormone-sympathetic interaction and adaptive thermogenesis are thyroid hormone receptor isoform specific. J Clin Invest. 2001;108(1):97–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Klaus S, Ely M, Encke D, Heldmaier G. Functional assessment of white and brown adipocyte development and energy metabolism in cell culture. Dissociation of terminal differentiation and thermogenesis in brown adipocytes. J Cell Sci. 1995;108(10):3171–3180. [DOI] [PubMed] [Google Scholar]
- 67. Ohno H, Shinoda K, Spiegelman BM, Kajimura S. PPARγ agonists Induce a white-to-brown fat conversion through stabilization of PRDM16 protein. Cell Metab 2012;15(3):395–404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Petrovic N, Walden TB, Shabalina IG, Timmons JA, Cannon B, Nedergaard J. Chronic peroxisome proliferator-activated receptor γ (PPARγ) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J Biol Chem. 2010;285(10):7153–7164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Wu J, Boström P, Sparks LM, et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012;150(2):366–376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Lee J-Y, Takahashi N, Yasubuchi M, et al. Triiodothyronine induces UCP-1 expression and mitochondrial biogenesis in human adipocytes. Am J Physiol Cell Physiol. 2012;302(2):C463–C472. [DOI] [PubMed] [Google Scholar]
- 71. Jiang W, Miyamoto T, Kakizawa T, et al. Expression of thyroid hormone receptor alpha in 3T3-L1 adipocytes; triiodothyronine increases the expression of lipogenic enzyme and triglyceride accumulation. J Endocrinol. 2004;182(2):295–302. [DOI] [PubMed] [Google Scholar]
- 72. Mills EM, Banks ML, Sprague JE, Finkel T. Pharmacology: uncoupling the agony from ecstasy. Nature 2003;426(6965):403–404. [DOI] [PubMed] [Google Scholar]
- 73. Zhang Q, Miao Q, Ye H, et al. The effects of thyroid hormones on brown adipose tissue in humans: a PET-CT study. Weng J, Pozzilli P, eds. Diabetes Metab Res Rev. 2014;30(6):513–520. [DOI] [PubMed] [Google Scholar]
- 74. Astrup A, Simonsen L, Bülow J, Madsen J, Christensen NJ. Epinephrine mediates facultative carbohydrate-induced thermogenesis in human skeletal muscle. Am J Physiol. 1989;257(3 Pt 1):E340–E345. [DOI] [PubMed] [Google Scholar]
- 75. Cypess AM, Chen Y-C, Sze C, et al. Cold but not sympathomimetics activates human brown adipose tissue in vivo. Proc Natl Acad Sci USA. 2012;109(25):10001–10005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Sprague JE, Mallett NM, Rusyniak DE, Mills E. UCP3 and thyroid hormone involvement in methamphetamine-induced hyperthermia. Biochem Pharmacol. 2004;68(7):1339–1343. [DOI] [PubMed] [Google Scholar]
- 77. Leijendekker WJ, van Hardeveld C, Elzinga G. Heat production during contraction in skeletal muscle of hypothyroid mice. Am J Physiol. 1987;253(2 Pt 1):E214–E220. [DOI] [PubMed] [Google Scholar]
- 78. Brand MD, Steverding D, Kadenbach B, Stevenson PM, Hafner RP. The mechanism of the increase in mitochondrial proton permeability induced by thyroid hormones. Eur J Biochem. 1992;206(3):775–781. [DOI] [PubMed] [Google Scholar]
- 79. Sprague JE, Yang X, Sommers J, Gilman TL, Mills EM. Roles of norepinephrine, free fatty acids, thyroid status, and skeletal muscle uncoupling protein 3 expression in sympathomimetic-induced thermogenesis. J Pharmacol Exp Ther. 2006;320(1):274–280. [DOI] [PubMed] [Google Scholar]
- 80. Simonides WS, van Hardeveld C. Thyroid hormone as a determinant of metabolic and contractile phenotype of skeletal muscle. Thyroid. 2008;18(2):205–216. [DOI] [PubMed] [Google Scholar]
- 81. Dawson AG. Oxidation of cytosolic NADH formed during aerobic metabolism in mammalian cells. Trends Biochem Sci. 1979;4(8):171–176. [Google Scholar]
- 82. Klingenberg M. Localization of the glycerol-phosphate dehydrogenase in the outer phase of the mitochondrial inner membrane. Eur J Biochem. 1970;13(2):247–252. [DOI] [PubMed] [Google Scholar]
- 83. Alfadda A, DosSantos RA, Stepanyan Z, Marrif H, Silva JE. Mice with deletion of the mitochondrial glycerol-3-phosphate dehydrogenase gene exhibit a thrifty phenotype: effect of gender. Am J Physiol Regul Integr Comp Physiol. 2004;287(1):R147–R156. [DOI] [PubMed] [Google Scholar]
- 84. DosSantos RA, Alfadda A, Eto K, Kadowaki T, Silva JE. Evidence for a compensated thermogenic defect in transgenic mice lacking the mitochondrial glycerol-3-phosphate dehydrogenase gene. Endocrinology. 2003;144(12):5469–5479. [DOI] [PubMed] [Google Scholar]
- 85. Warner A, Rahman A, Solsjö P, et al. Inappropriate heat dissipation ignites brown fat thermogenesis in mice with a mutant thyroid hormone receptor α1. Proc Natl Acad Sci USA. 2013;110(40):16241–16246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Pelletier P, Gauthier K, Sideleva O, Samarut J, Silva JE. Mice lacking the thyroid hormone receptor-α gene spend more energy in thermogenesis, burn more fat, and are less sensitive to high-fat diet-induced obesity. Endocrinology. 2008;149:6471–6486. [DOI] [PubMed] [Google Scholar]
- 87. Schneider MJ, Fiering SN, Pallud SE, Parlow AF, St Germain DL, Galton VA. Targeted disruption of the type 2 selenodeiodinase gene (DIO2) results in a phenotype of pituitary resistance to T4. Mol Endocrinol. 2001;15:2137–2148. [DOI] [PubMed] [Google Scholar]
- 88. Hall JA, Ribich S, Christoffolete MA, Simovic G, Correa-Medina M, Patti ME, Bianco AC. Absence of thyroid hormone activation during development underlies a permanent defect in adaptive thermogenesis. Endocrinology. 2010;151:4573–4582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Castillo M, Hall JA, Correa-Medina M, Ueta C, Kang HW, Cohen DE, Bianco AC. Disruption of thyroid hormone activation in type 2 deiodinase knockout mice causes obesity with glucose intolerance and liver steatosis only at thermoneutrality. Diabetes. 2011;60:1082–1089. [DOI] [PMC free article] [PubMed] [Google Scholar]