Abstract
p16 and Ki-67 immunohistochemistry can be used as an ancillary method for the diagnosis of squamous intraepithelial lesion (SIL) versus atrophic change and atypical squamous metaplasia. The aim of the present study was to evaluate the efficacy of these two immunohistochemical markers in the accurate interpretation of cervical biopsies and correlate this data with human papilloma virus (HPV) infection status. The study included 103 formalin-fixed cervical punch and cone biopsy samples, with corresponding HPV DNA test data. Histopathological staining with hematoxylin and eosin, and immunohistochemical staining for p16 and Ki-67 were reviewed by two pathologists. The positivity of p16 and Ki-67 increased significantly with the severity of the cervical lesion in patients with a high-risk-HPV (HR-HPV) infection status (P<0.001). However, there was discordance in the HPV-negative group. Furthermore, concomitant diffuse, strong and block positive staining of p16, and a high Ki-67 index were implicated in high-grade SIL in the HR-HPV group. Thus, the two markers were efficient in advancing the diagnostic accuracy of cervical biopsies in the HR-HPV group; however, application of immunohistochemical results should be carefully considered in the HPV-negative group.
Keywords: uterine cervix, squamous intraepithelial lesion, human papilloma virus, p16, Ki-67
Introduction
Uterine cervical cancer represents the second most common type of cancer, in terms of incidence and mortality rates (15.2 and 7.8 cases per 100,000 individuals, respectively), in women worldwide, with an increased incidence (17.8 cases per 100,000 individuals) in low resource countries (1). Almost all cases of cervical cancer are caused by persistent infections with high-risk-human papilloma virus (HR-HPV), which may cause cervical intraepithelial neoplasia (CIN) and squamous cell carcinoma (SCC) (2).
Cervical biopsy, which is used in conjunction with Papanicolaou cytology testing, HPV DNA testing and colposcopy, has an important role in the evaluation and management of women with cervical dysplastic lesions. Thus, cervical biopsy is crucial for the prevention and early detection of cervical cancer (3). However, misinterpretation of histological changes may occur for various reasons, such as the presence of atrophy, immature metaplasia, transitional metaplasia, reparative or inflammatory atypia, inadequate sample size and tissue artifacts. These factors may cause inter-observer variation and poor intra-observer reproducibility (4–6).
A number of biomarkers have been evaluated for their potential to improve the diagnostic consistency and accuracy of cervical biopsy interpretation (4–6). Many of these studies (7–9) and other such studies (10,11) endorse the use of p16 and Ki-67 immunostains, and more recently the ProExC immunostain (11–13), as useful adjunct techniques to confirm a diagnosis of high-grade squamous intraepithelial lesion (HSIL) and to distinguish it from its mimics. p16 is a cyclin-dependent kinase inhibitor and a surrogate marker for HPV E7-mediated degradation of retinoblastoma protein (pRb) (14,15). There is evidence to suggest that a negative feedback mechanism controlling p16 levels in normal cells is disrupted by a reduction of pRb activity in proliferating squamous epithelial cells expressing HR-HPV E7 (16). With regard to p16, it has been demonstrated that almost 100% of cases of HSIL and SCC associated with HR-HPV express high levels of p16, whereas non-dysplastic cervical epithelium or low-grade SIL (LSIL) associated with low-risk (LR) or negative HPV types do not express p16 (17). Ki-67 is a nuclear protein and is detected by mindbomb E3 ubiquitin protein ligase-1 (MIB-1), a monoclonal antibody that is associated with RNA transcription and cell cycle progression (18). To improve diagnostic accuracy, other markers, including Ki-67, have been used in conjunction with p16 in the histological assessment of SIL and SCC of the uterine cervix (5,19). Similar to p16, Ki-67 is overexpressed in HSIL and SCC (20).
Considering the aforementioned knowledge, the present study aimed to investigate the efficacy of p16 and Ki-67 immunohistochemical stains in the pathological assessment of uterine cervical biopsy samples, and identify any viral and histopathological correlations.
Materials and methods
Materials
The following terms were used to search in the pathology data program of Chosun University Hospital, (Gwangju, Republic of Korea) for the final reports of patients who had undergone HPV DNA testing of cervical biopsy specimens obtained between January 2012 and March 2013: ‘Squamous metaplasia’, ‘LSIL’ and ‘HSIL’. According to the 2014 WHO classification of tumors of the female genital tract (21), a total of 103 specimens were identified with the following pathological diagnoses: Squamous metaplasia, 1 case; LSIL with HPV, 30 cases; LSIL with CIN 1, 22 cases; HSIL with CIN 2, 21 cases; and HSIL with CIN 3, 29 cases. By reviewing the medical records and pathological reports of the selected patients, details of the patients' age, underlying disease, other tumor history, cervico-vaginal cytological diagnosis and HPV infection status were identified. The present study was approved by the Institutional Review Board (IRB) of the Chosun University Hospital (Gwangju, Korea) (CHOSUN 2014-03-002). IRB approval included a waiver of informed consent.
HPV DNA testing and genotyping of HPV DNA for HR-HPV were achieved by polymerase chain reaction-based HPV genotyping assay using a HPV 9G DNA chip (Fammed Co., Ltd., Seongnam, Republic of Korea), according to the manufacturer's instructions. The DNA chip was able to detect 28 HPV genotypes, including 14 HR-HPVs (16, 18, 31, 33, 39, 45, 51, 52, 54, 56, 58, 59, 66 and 68) and 7 LR-HPVs (6, 11, 34, 40, 42, 43 and 44). The HPV genotypes were classified as HR-HPV or LR-HPV according to the scheme proposed by Dunne et al (22).
Immunohistochemical analysis
Prior to immunohistochemical analysis, 3-µm thick serial sections were prepared from representative formalin-fixed, paraffin-embedded tissue blocks and mounted on positively charged glass slides. Immunostaining was performed using a BenchMark XT autostainer (Ventana Medical Systems, Tucson, AZ, USA). The primary antibodies were anti-human p16 (mouse monoclonal antibody, clone E6H4; cat no. 725–4713; 1:100 dilution; Ventana Medical Systems) and Ki-67 (mouse monoclonal antibody, clone MM1; cat no. ACK02; 1:50 dilution; Leica Biosystems, Ltd., Newcastle, UK). Briefly, antigen retrieval was performed using peroxidase and alkaline phosphatase blocking reagent (Dako Korea LLC, Seoul, Republic of Korea) for 10 min at 95–99°C in a water bath. After inactivation of endogenous peroxidases, the slides were incubated for 30 min at room temperature with primary antibodies or negative control antibody. The slides were then incubated with polyclonal anti-mouse secondary antibody (cat. no. E0433; 1:250 dilution) for 30 min at room temperature. Primary antibodies and alkaline phosphatase enzyme complexes (cat. no. D0651; Dako Korea LLC) were incubated for 30 min at room temperature, then visualized following incubation with 3,3′-diaminobenzidine (Dako Korea LLC) for 20 min at room temperature. Next, the slides were counterstained with Mayer's hematoxylin (Merck Millipore, Darmstadt, Germany) for 5 min at room temperature. The slides were visualized under a microscope (BX50; Olympus Corporation, Tokyo, Japan).
Immunohistochemical interpretation
p16 staining was interpreted as positive when nuclear or nuclear and cytoplasmic, strong and diffuse block staining beginning from the basal cell layer of the epithelium was observed. By contrast, non-specific focal or patch nuclear staining was considered to indicate negative p16 staining, with cytoplasmic only, wispy, blob-like, puddled, scattered, single cells and a complete lack of staining also defined as negative p16 expression (23). The expression of Ki-67 was categorized into four groups based on the distribution and proportions of cells with positive nuclear staining, as follows: Score 0, <10% of the cells, restricted to the parabasal cell layers; score 1, 10–29% of the cells, restricted to the lower third of the epithelium; score 2, 30–69% of the cells, reaching the upper third of the epithelium; score 3, ≥70% epithelial cells, including full thickness expression of Ki-67 (24).
Histological evaluation
Hematoxylin and eosin staining was performed according to standard protocols (25). Two independent observers evaluated the hematoxylin and eosin (H&E)-stained slides, and then determined initial diagnoses based on the histological features according to the 2014 WHO Classification (21). The whole slides were reviewed independently by two observers who were blinded to all clinicopathological information. These diagnoses included benign lesions, such as squamous metaplasia or LSIL, and precancerous lesions, such as HSIL. The observers used a two-tier system of terminology, according to The Lower Anogenital Squamous Terminology Standardization Project for HPV-Associated Lesions (23). Then, each observer evaluated the H&E slides a second time, and analyzed p16 and Ki-67 staining. The final diagnosis was determined by revising all diagnoses against each other and by a final comparison of all diagnoses by a third observer. The two pathologists' diagnoses were concordant with the diagnosis of the third pathologist in all 103 cases. Finally, consensus diagnoses were reached.
Statistical analysis
SPSS software (version 22.0; SPSS, Inc., Chicago, IL, USA) was used for statistical analysis. The χ2 test was performed between the final diagnoses of p16 and Ki-67 status. These values were then compared with the following HPV infection statuses: HR-HPV, LR-HPV and HPV-negative. P<0.05 was considered to indicate a statistically significant difference.
Results
Expression of p16 and Ki-67
HR-HPV group
The HPV genotyping assay detected HR-HPV genotypes in 68.93% (71/103) of cases. Of the 28 cases of benign lesion (NILM and LSIL), 27 cases were p16 negative, with only 1 case of LSIL exhibiting p16 positivity. Excluding 1 case of negative p16 expression, almost all of the 43 cases of HSIL exhibited positive expression for p16 (Table I).
Table I.
Comparison of p16 result and final diagnosis according to HPV infection status.
| Final diagnosis, n (%) | |||||
|---|---|---|---|---|---|
| HPV status | p16 | HSIL | LSIL | NILM | P-value |
| HR (n=71) | + | 42 (59.1) | 1 (1.4) | 0 (0.0) | <0.001 |
| − | 1 (1.4) | 26 (36.6) | 1 (1.4) | ||
| LR (n=6) | + | 1 (16.6) | 0 (0.0) | 0 (0.0) | 0.014 |
| − | 0 (0.0) | 5 (83.3) | 0 (0.0) | ||
| N (n=26) | + | 5 (19.2) | 0 (0.0) | 0 (0.0) | 0.001 |
| − | 3 (11.5) | 16 (61.5) | 2 (7.7) | ||
| Total (n=103) | + | 48 (46.6) | 1 (0.9) | 0 (0.0) | |
| − | 4 (3.9) | 47 (45.6) | 3 (2.9) | ||
HPV, human papilloma virus; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; NILM, negative for intraepithelial lesion or malignancy; HR, high-risk; +, positive; -, negative; LR, low-risk; N, negative.
There were 17 cases HR-HPV with Ki-67 negativity, including 16 cases of LSIL and 1 case of NILM. All Ki-67 staining scores of 3 in the HR-HPV group were HSIL. Twelve cases showed score of 1 in Ki-67 staining that of 3 cases of HSIL and 9 cases of LSIL. A Ki-67 score of 2 was observed in 16 cases, including 14 cases of HSIL and 2 cases of LSIL (Table II). In HR-HPV patients, p16 positivity (Table I) and Ki-67 score (Table II) were found to correlate with the severity of dysplasia (both P<0.001).
Table II.
Comparison of Ki-67 result and final diagnosis according to HPV infection status.
| Final diagnosis, n (%) | |||||
|---|---|---|---|---|---|
| HPV status | Ki-67 score | HSIL | LSIL | NILM | P-value |
| HR (n=71) | 0 | 0 (0.0) | 16 (22.5) | 1 (1.4) | <0.001 |
| 1 | 3 (4.2) | 9 (12.6) | 0 (0.0) | ||
| 2 | 14 (19.7) | 2 (2.8) | 0 (0.0) | ||
| 3 | 26 (36.6) | 0 (0.0) | 0 (0.0) | ||
| LR (n=6) | 0 | 0 (0.0) | 2 (33.3) | 0 (0.0) | <0.001 |
| 1 | 1 (16.6) | 1 (16.6) | 0 (0.0) | ||
| 2 | 0 (0.0) | 2 (33.3) | 0 (0.0) | ||
| 3 | 0 (0.0) | 0 (0.0) | 0 (0.0) | ||
| N (n=26) | 0 | 0 (0.0) | 10 (38.4) | 2 (7.7) | <0.001 |
| 1 | 0 (0.0) | 2 (7.7) | 0 (0.0) | ||
| 2 | 7 (26.9) | 1 (3.8) | 0 (0.0) | ||
| 3 | 1 (3.8) | 1 (3.8) | 0 (0.0) | ||
| Total (n=103) | 0 | 0 (0.0) | 28 (27.2) | 3 (2.9) | |
| 1 | 4 (3.9) | 14 (13.6) | 0 (0.0) | ||
| 2 | 21 (20.3) | 5 (4.8) | 0 (0.0) | ||
| 3 | 27 (26.2) | 1 (0.9) | 0 (0.0) | ||
HPV, human papilloma virus; HSIL, high-grade squamous intraepithelial lesion; LSIL, low-grade squamous intraepithelial lesion; NILM, negative for intraepithelial lesion or malignancy; HR, high-risk; LR, low-risk; N, negative.
LR-HPV group
The HPV genotyping assay detected 5.82% (6/103) cases with LR-HPV genotypes. In the 5 cases of LSIL, all were p16 negative. Furthermore, there was only 1 case of HSIL with p16 positivity (Table I) and 2/5 cases (33.3%) of LSIL exhibited a Ki-67 score of 0. In addition, two and one cases of LSIL had Ki-67 scores of 2 and 1, respectively. There was 1 case of HSIL with a Ki-67 score of 1 (Table II). Furthermore, in the LR-HPV group, p16 positivity (Table I) and Ki-67 score (Table II) were significantly associated with malignant potential (P=0.014 and P<0.001, respectively).
HPV-negative group
The HPV genotyping assay detected 25.24% (26/103) cases with negative HPV genotypes. All 18 cases of benign lesion (NILM and LSIL) were p16 negative. Excluding 3 cases, 5/8 cases (19.2%) of HSIL showed positive expression for p16 (Table I). A Ki-67 score of 0 was exhibited by 12/18 cases of benign lesion, including of 2 cases of NILM and 10 cases of LSIL. Furthermore, 4/16 cases of LSIL showed a Ki-67 expression score of 1, while only 1/16 cases of LSIL exhibited a Ki-67 expression score of 2 and 3. There were 8 cases of HSIL, with a score of 3 in 1 case and a score of 2 in 7 cases (Table II). In the HPV-negative group, p16 positivity (Table I) and Ki-67 score (Table II) were significantly associated with the degree of dysplasia (P=0.001 and P<0.001, respectively).
Discordance between p16 and Ki-67 expression, and the HPV genotype
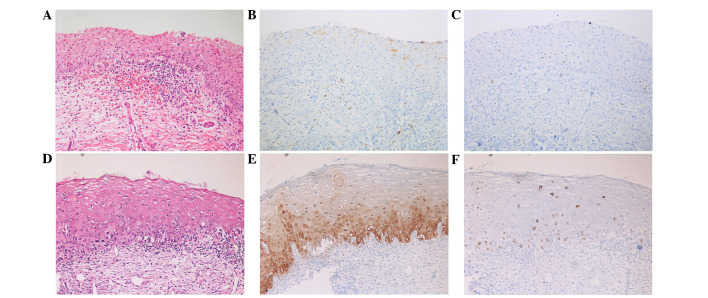
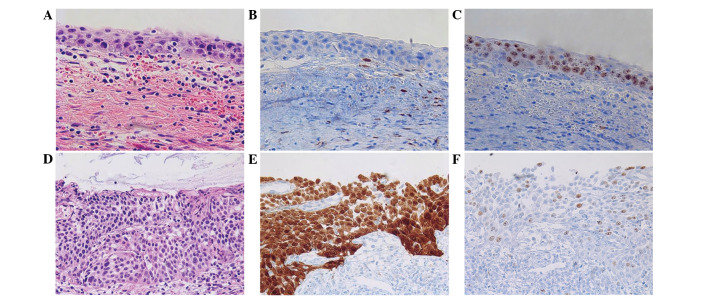
Analysis of the H&E slides only revealed 2 cases of immature squamous metaplasia with an initial diagnosis of LSIL (n=1) and HSIL (n=1). These cases exhibited immunohistochemical negativity for p16 and a low Ki-67 index (Fig. 1). However, discordance was identified in the HPV-negative group (Table I). In the HPV-negative group, 3/26 cases exhibited completely negative expression for p16; thus, the discordance rate was 11.54%. However, these cases were consistent with a pathological diagnosis of HSIL, according to the H&E slides and the high Ki-67 index (score, 2) (Fig. 2A–C). Therefore, these cases were finally diagnosed based on histological findings and the Ki-67 result (Table III). In the LR-HPV and HR-HPV groups, expression of p16 and Ki-67 exhibited a strong correlation with malignant potential of the lesion (Fig. 2D–F). Only one case of HSIL diagnosis was negative in p16 result and one case of LSIL was positive for p16 positive in the total HR-HPV group (n=71), thus, the discordant rate was 2.82% (Table I).
Figure 1.
Upper row shows immature squamous metaplasia. (A) Hematoxylin and eosin staining. Immunohistochemical staining, revealing (B) negative p16 expression and (C) a low Ki-67 (score 0). Lower row shows low-grade squamous intraepithelial lesion. (D) Hematoxylin and eosin staining. Immunohistochemical staining, revealing (E) a patch stain for p16 in the parabasal layer that was interpretated as negative and (F) increased expression of Ki-67 (score 1) in the lower third area. Magnification, ×200.
Figure 2.
Upper row shows a high-grade squamous intraepithelial lesion (HSIL) in the human papilloma virus (HPV)-negative group. (A) Hematoxylin and eosin staining. Immunohistochemical staining, revealing (B) negative staining for p16 and (C) increased expression of Ki-67 (score 3) in the full thickness of the upper third area. Lower row shows HSIL in the high-risk-HPV group. (D) Hematoxylin and eosin staining. Immunohistochemical staining, revealing (E) strong block positivity of p16 and (F) an increase in Ki-67 expression in the full thickness of the sample. Magnification, ×200.
Table III.
Cases of discordance between HPV N group and result of HPV genotyping.
| Case | Initial diagnosis | Final diagnosis | p16 | Ki-67 score | HPV viral Pap | Genotype |
|---|---|---|---|---|---|---|
| 1 | HSIL | HSIL | − | 2 | N | N |
| 2 | HSIL | HSIL | − | 2 | N | N |
| 3 | HSIL | HSIL | − | 2 | N | N |
HPV, human papilloma virus; Pap, Papanicolaou; HSIL, high-grade squamous intraepithelial lesion; −/N, negative.
Discussion
Diagnostic interpretation of dysplasia in the uterine cervix typically includes analysis of hypercellularity, significant atypia, mitotic figures and disorientation from the parabasal to upper layers (21). However, unusual histological features, including mildly increased cellularity, the absence of mitotic figures and questionable atypia, may be observed in the lesion (9). In daily practice, the accurate diagnosis of cervical dysplastic lesions is subject to inter-observer and intra-observer variability by these factors (6).
Immunostaining may improve the diagnostic reproducibility and accuracy of the CIN lesion. Previous studies have demonstrated that p16 and Ki-67 are co-expressed in almost 100% of cases of high-grade squamous and glandular lesions, and these markers are rarely co-expressed in normal or benign lesions of cervical epithelial lesion (14,26–29). Immunohistochemical staining for p16 has been investigated in cervical pathology as a marker for HPV-transformed lesions, and various studies have demonstrated diffuse and continuous staining of p16, beginning in the basal and parabasal cell layers, which is defined as block positive staining (7,23,30). Based on the concept that HPV-mediated transformation is triggered by dysregulated expression of the viral oncogenes E6 and E7 in basal and parabasal cells, p16 immunohistochemistry was hypothesized to distinguish between transforming and nontransforming HPV infections, and only the block positive p16 expression pattern was defined as a hallmark of HPV-dependent transformation and thus considered as p16 positive (27,31,32).
Ki-67 is detected by the MIB-1 monoclonal antibody and is a nuclear protein that is associated with RNA transcription and cell cycle progression (18). Similar to p16, Ki-67 is overexpressed in CIN 2/3, SCC, adenocarcinoma in situ and adenocarcinoma (20). However, in contrast to p16, Ki-67 is also overexpressed in the basal cells of normal squamous mucosa and in benign proliferative lesions, including basal cell hyperplasia of the squamous mucosa (26). Therefore, a combination of p16 and Ki-67 immunostaining is recommended for specificity in distinguishing LSIL versus HSIL from its mimickers, as opposed to using each immunostaining marker alone (11).
By conducting a review of previous studies, it was found that these immunostaining markers could be applied in selective HR-HPV groups and high-grade dysplastic lesions (29,30). Thus, the present study investigated the efficacy of p16 and Ki-67 immunostaining markers in the accurate interpretation of cervical biopsies, and comparing the results of HR-HPV, LR-HPV and HPV-negative groups. Diffuse and strong staining for p16 and Ki-67 revealed HSIL in the HR-HPV and LR-HPV groups. Almost all HSIL cases in the HR-HPV group were p16 positive. Obvious expression of p16 has been associated HR-HPV infection (5); this was also found in the present study. In addition, 2 cases of immature squamous metaplasia with previous LSIL (n=1)/HSIL (n=1) were identified by H&E staining alone; furthermore, these cases demonstrated negativity for p16 and a low Ki-67 index in the HR-HPV and HPV-negative viral test groups. However, numerous cases in the HPV-negative group exhibited discordancy between the H&E and immunohistochemical findings. Three unusual cases that showed an unexpected p16 immunostaining result were identified out of the total 26 HPV-negative patients (Table III). Representatively, 2 cases were implicated pathological significance. Case 3 (Table III) demonstrated p16 negativity and a Ki-67 score of 3, and exhibited histopathological features consistent with HSIL. Thus, a final diagnosis of HSIL was determined. However, the patient's clinical stage was advanced; therefore, the therapeutic options were limited. The patient underwent radiation therapy and chemotherapeutic regimen two times but succumbed due to pancytopenia and multiple lymph node metastasis 9 months later. Case 2 (Table III) was initially diagnosed with HSIL, although the patient's immunohistochemical profile of p16 was not compatible with HSIL. However, H&E staining and Ki-67 index results indicated HSIL and thus, the final diagnosis was confirmed by conization specimens resected with a clear resection margin 1 month later. Case 1 (Table III) had an initial diagnosis of HSIL with negative p16 expression and a Ki-67 score of 2; in this case the first diagnosis was not revised. The diagnosis was finally confirmed by analysis of a hysterectomy specimen 3 weeks later. Three months after hysterectomy, case 1 exhibited no evidence of local recurrence or metastasis. The aforementioned 3 cases of the negative-HPV group were diagnosed via different diagnostic methods, due to the unusual p16 pathology results. Therefore further treatments, including conization and hysterectomy were performed to allow for additional pathological examinations, that did not rely on the atypical p16 scores. In HPV negative patients, we hypothesize that HSIL with a negative p16 score must be considered when interpreting cervical biopsies to prevent underdiagnosis to LSIL or NILM. For case 1, the negative p16 result did not correspond with the clinicopathological setting, and thus H&E staining and Ki-67 index were applicable. It is currently unknown what causes true-negative p16 immunostaining in HSIL or SCC in the HPV-negative group. The positivity of p16 expression and the Ki-67 score increased with the severity of the cervical lesion in the HR-HPV and LR-HPV groups. However, correlation between p16 score and final diagnosis was stronger in the HR-HPV group compared with the HPV-negative group. Thus, Ki-67 score is a more useful marker than p16 score in HPV-negative cases.
In conclusion, simultaneously positive p16 expression and high Ki-67 index are implicated in diagnosis of HSIL for HR-HPV and LR-HPV precancerous lesions. The two markers are efficient in advancing the diagnostic accuracy of cervical biopsies in cases of HR-HPV and LR-HPV, however, application in discrete daily sign-out processing of immunohistochemical findings should be considered in the HPV-negative group. This is a pilot study with a small number of cases, however, pathologists should be aware that unusual immunostaining results in HPV-negative patients, such as negative p16 staining in HSIL, may imply factors other than HR-HPV and LR-HPV infection. In the future, novel immunostaining markers or other methods that may be applicable for HPV-negative patients should be assessed for the reproducible diagnosis of cervical disease.
Acknowledgements
The current study was supported by a National Research Foundation of Korea Grant funded by the Ministry of Education, Science and Technology (MEST) through the Research Center for Resistant Cells (grant no. R13-2003-009).
References
- 1.Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 2.Schiffman M, Castle PE, Jeronimo J, Rodriguez AC, Wacholder S. Human papillomavirus and cervical cancer. Lancet. 2007;370:890–907. doi: 10.1016/S0140-6736(07)61416-0. [DOI] [PubMed] [Google Scholar]
- 3.Wright TC, Jr, Massad LS, Dunton CJ, Spitzer M, Wilkinson EJ, Solomon D. 2006 American Society for Colposcopy and Cervical Pathology-sponsored Consensus Conference: 2006 consensus guidelines for the management of women with cervical intraepithelial neoplasia or adenocarcinoma in situ. J Low Genit Tract Dis. 2007;11:223–239. doi: 10.1097/LGT.0b013e318159408b. [DOI] [PubMed] [Google Scholar]
- 4.Wright TC, Jr, Cox JT, Massad LS, Twiggs LB, Wilkinson EJ. ASCCP-Sponsored Consensus Conference: 2001 Consensus guidelines for the management of women with cervical cytological abnormalities. JAMA. 2002;287:2120–2129. doi: 10.1001/jama.287.16.2120. [DOI] [PubMed] [Google Scholar]
- 5.Kalof AN, Cooper K. p16INK4a immunoexpression: surrogate marker of high-risk HPV and high-grade cervical intraepithelial neoplasia. Adv Anat Pathol. 2006;13:190–194. doi: 10.1097/00125480-200607000-00006. [DOI] [PubMed] [Google Scholar]
- 6.Klaes R, Benner A, Friedrich T, Ridder R, Herrington S, Jenkins D, et al. p16INK4a immunohistochemistry improves interobserver agreement in the diagnosis of cervical intraepithelial neoplasia. Am J Surg Pathol. 2002;26:1389–1399. doi: 10.1097/00000478-200211000-00001. [DOI] [PubMed] [Google Scholar]
- 7.Horn LC, Reichert A, Oster A, Arndal SF, Trunk MJ, Ridder R, et al. Immunostaining for p16INK4a used as a conjunctive tool improves interobserver agreement of the histologic diagnosis of cervical intraepithelial neoplasia. Am J Surg Pathol. 2008;32:502–512. doi: 10.1097/PAS.0b013e31815ac420. [DOI] [PubMed] [Google Scholar]
- 8.Ma L, Fisk JM, Zhang RR, Ulukus EC, Crum CP, Zheng W. Eosinophilic dysplasia of the cervix: a newly recognized variant of cervical squamous intraepithelial neoplasia. Am J Surg Pathol. 2004;28:1474–1484. doi: 10.1097/01.pas.0000141407.10204.c5. [DOI] [PubMed] [Google Scholar]
- 9.Aoyama C, Liu P, Ostrzega N, Holschneider CH. Histologic and immunohistochemical characteristic of neoplastic and nonneoplastic subgroups of atypical squamous lesions of the uterine cervix. Am J Clin Pathol. 2005;123:699–706. doi: 10.1309/FB900YXTF7D2HE99. [DOI] [PubMed] [Google Scholar]
- 10.Benevolo M, Mottolese M, Marandino F, Vocaturo G, Sindico R, Piperno G, et al. Immunohistochemical expression of p16INK4A is predictive of HR-HPV infection in cervical low-grade lesions. Mod Pathol. 2006;19:384–391. doi: 10.1038/modpathol.3800551. [DOI] [PubMed] [Google Scholar]
- 11.Pinto AP, Schlecht NF, Woo TY, Crum CP, Cibas ES. Biomarker (ProExC, p16INK4A, and MiB-1) distinction of high-grade squamous intraepithelial lesion from its mimics. Mod Pathol. 2008;21:1067–1074. doi: 10.1038/modpathol.2008.101. [DOI] [PubMed] [Google Scholar]
- 12.Sanati S, Huettner P, Ylagan LR. Role of ProExC: a novel immunoperoxidase marker in the evaluation of dysplastic squamous and glandular lesions in cervical specimens. Int J Gynecol Pathol. 2010;29:79–87. doi: 10.1097/PGP.0b013e3181ae81a0. [DOI] [PubMed] [Google Scholar]
- 13.Walts AE, Bose S. p16, Ki-67, and BD ProExC immunostaining: a practical approach for diagnosis of cervical intraepithelial neoplasia. Hum Pathol. 2009;40:957–964. doi: 10.1016/j.humpath.2008.12.005. [DOI] [PubMed] [Google Scholar]
- 14.Cameron RI, Maxwell P, Jenkins D, McCluggage WG. Immunohistochemical staining with MIB1, bcl2 and p16 assists in the distinction of cervical glandular intraepithelial neoplasia from tubo-endometrial metaplasia, endometriosis and microglandular hyperplasia. Histopathology. 2002;41:313–321. doi: 10.1046/j.1365-2559.2002.01465.x. [DOI] [PubMed] [Google Scholar]
- 15.Doorbar J, Quint W, Banks L, Bravo IG, Stoler M, Broker TR, et al. The biology and life-cycle of human papillomaviruses. Vaccine. 2012;30(Suppl 5):F55–F70. doi: 10.1016/j.vaccine.2012.06.083. [DOI] [PubMed] [Google Scholar]
- 16.Khleif SN, DeGregori J, Yee CL, Otterson GA, Kaye FJ, Nevins JR, Howley PM. Inhibition of cyclin D-CDK4/CDK6 activity is associated with an E2F-mediated induction of cyclin kinase inhibitor activity. Proc Natl Acad Sci USA. 1996;93:4350–4354. doi: 10.1073/pnas.93.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Guo M, Baruch AC, Silva EG, Jan YJ, Lin E, Sneige N, Deavers MT. Efficacy of p16 and ProExC immunostaining in the detection of high-grade cervical intraepithelial neoplasia and cervical carcinoma. Am J Clin Pathol. 2011;135:212–220. doi: 10.1309/AJCP1LLX8QMDXHHO. [DOI] [PubMed] [Google Scholar]
- 18.Hitchcock CL. Ki-67 staining as a means to simplify analysis of tumor cell proliferation. Am J Clin Pathol. 1991;96:444–446. doi: 10.1093/ajcp/96.4.444. [DOI] [PubMed] [Google Scholar]
- 19.McCluggage WG. Immunohistochemistry as a diagnostic aid in cervical pathology. Pathology. 2007;39:97–111. doi: 10.1080/00313020601123961. [DOI] [PubMed] [Google Scholar]
- 20.Cina SJ, Richardson MS, Austin RM, Kurman RJ. Immunohistochemical staining for Ki-67 antigen, carcinoembryonic antigen, and p53 in the differential diagnosis of glandular lesions of the cervix. Mod Pathol. 1997;10:176–180. [PubMed] [Google Scholar]
- 21.Reich O, Regauer S, Marth C, Schmidt D, Horn LC, Dannecker C, Menton M, Beckmann MW. Precancerous lesions of the cervix, vulva and vagina according to the 2014 WHO classification of tumors of the female genital tract. Geburtshilfe Frauenheilkd. 2015;75:1018–1020. doi: 10.1055/s-0035-1558052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dunne EF, Unger ER, Sternberg M, McQuillan G, Swan DC, Patel SS, Markowitz LE. Prevalence of HPV infection among females in the United States. JAMA. 2007;297:813–819. doi: 10.1001/jama.297.8.813. [DOI] [PubMed] [Google Scholar]
- 23.Darragh TM, Colgan TJ, Cox JT, Heller DS, Henry MR, Luff RD, et al. The Lower Anogenital Squamous Terminology Standardization Project for HPV-Associated Lesions: Background and consensus recommendations from the College of American Pathologists and the American Society for Colposcopy and Cervical Pathology. J Low Genit Tract Dis. 2012;16:205–242. doi: 10.1097/LGT.0b013e31825c31dd. [DOI] [PubMed] [Google Scholar]
- 24.Reuschenbach M, Seiz M, von Knebel Doeberitz C, Vinokurova S, Duwe A, Ridder R, et al. Evaluation of cervical cone biopsies for coexpression of p16INK4a and Ki-67 in epithelial cells. Int J Cancer. 2012;130:388–394. doi: 10.1002/ijc.26017. [DOI] [PubMed] [Google Scholar]
- 25.Kiernan JA, editor. Histological and Histochemical Methods: Theory and Practice. 3rd. Vol. 12. Oxford, UK: Butterworth-Heinemann; 1999. [Google Scholar]
- 26.Agoff SN, Lin P, Morihara J, Mao C, Kiviat NB, Koutsky LA. p16INK4a expression correlates with degree of cervical neoplasia: a comparison with Ki-67 expression and detection of high-risk HPV types. Mod Pathol. 2003;16:665–673. doi: 10.1097/01.MP.0000077518.78046.0C. [DOI] [PubMed] [Google Scholar]
- 27.Liang J, Mittal KR, Wei JJ, Yee H, Chiriboga L, Shukla P. Utility of p16INK4a, CEA, Ki67, P53 and ER/PR in the differential diagnosis of benign, premalignant, and malignant glandular lesions of the uterine cervix and their relationship with Silverberg scoring system for endocervical glandular lesions. Int J Gynecol Pathol. 2007;26:71–75. doi: 10.1097/01.pgp.0000225851.97739.9f. [DOI] [PubMed] [Google Scholar]
- 28.Pavlakis K, Messini I, Athanassiadou S, Kyrodimou E, Pandazopoulou A, Vrekoussis T, Stathopoulos EN. Endocervical glandular lesions: a diagnostic approach combining a semiquantitative scoring method to the expression of CEA, MIB-1 and p16. Gynecol Oncol. 2006;103:971–976. doi: 10.1016/j.ygyno.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 29.Qiao X, Bhuiya TA, Spitzer M. Differentiating high-grade cervical intraepithelial lesion from atrophy in postmenopausal women using Ki-67, cyclin E, and p16 immunohistochemical analysis. Low Genit Tract Dis. 2005;9:100–107. doi: 10.1097/00128360-200504000-00006. [DOI] [PubMed] [Google Scholar]
- 30.Bergeron C, Ordi J, Schmidt D, Trunk MJ, Keller T, Ridder R. European CINtec Histology Study Group: Conjunctive p16INK4a testing significantly increases accuracy in diagnosing high-grade cervical intraepithelial neoplasia. Am J Clin Pathol. 2010;133:395–406. doi: 10.1309/AJCPXSVCDZ3D5MZM. [DOI] [PubMed] [Google Scholar]
- 31.Doeberitz MV, Vinokurova S. Host factors in HPV-related carcinogenesis: cellular mechanisms controlling HPV infections. Arch Med Res. 2009;40:435–442. doi: 10.1016/j.arcmed.2009.06.002. [DOI] [PubMed] [Google Scholar]
- 32.von Knebel Doeberitz M. New markers for cervical dysplasia to visualise the genomic created by aberrant oncogenic papillomavirus infections. Eur J Cancer. 2002;38:2229–2242. doi: 10.1016/S0959-8049(02)00462-8. [DOI] [PubMed] [Google Scholar]