Abstract
Histone deacetylase 1 (HDAC1) has been reported to be important for multiple aspects of normal embryonic development, but little is known about its function in the development of mechanosensory organs. Here, we first confirmed that HDAC1 is expressed in the developing otic vesicles of zebrafish by whole-mount in situ hybridization. Knockdown of HDAC1 using antisense morpholino oligonucleotides in zebrafish embryos induced smaller otic vesicles, abnormal otoliths, malformed or absent semicircular canals, and fewer sensory hair cells. HDAC1 loss of function also caused attenuated expression of a subset of key genes required for otic vesicle formation during development. Morpholino-mediated knockdown of HDAC1 resulted in decreased expression of members of the Fgf family in the otic vesicles, suggesting that HDAC1 is involved in the development of the inner ear through regulation of Fgf signaling pathways. Taken together, our results indicate that HDAC1 plays an important role in otic vesicle formation.
The vertebrate inner ear is a conserved and complex sensory organ that develops from the otic vesicle and consists of two sensory organs, the auditory apparatus (the cochlea in mammals) and the vestibular apparatus1. The incomplete or aberrant development of the auditory organs or damage to the fully developed organs can cause hearing loss. The zebrafish (Danio rerio) has become an attractive model organism for the study of the developmental and regulatory mechanisms of hearing and vestibular disorders because it has features that are closely related to the auditory systems of higher vertebrates2. During development, the initial steps in otic placode induction are highly complex, and combinations of factors appear to be involved in this process. The family members of the fibroblast growth factors (Fgfs) have been reported to be involved in otic induction. In particular, two ligands of the Fgf signaling pathway, fgf3 and fgf8, are expressed in the hindbrain and cooperate to induce otic development in zebrafish3,4,5. The acerebellar mutation in fgf8 results in smaller otic vesicles, which have abnormal otoliths and semicircular canals3. Knockdown of fgf3 by injection of morpholino oligomers disrupts otic induction in a similar manner as acerebellar mutation of fgf8. Moreover, loss of both fgf8 and fgf3 causes severe reduction or total loss of otic tissues3,4,5. A number of Fgf-signaling downstream target genes have been identified as important in otic formation and early ear fate decision. For example, Pax genes are the main effectors downstream of Fgf signaling and are involved in otic specification in zebrafish5,6.
Covalent chromatin modifications play essential roles in transcriptional regulation in eukaryotes7,8. Acetylation and deacetylation are the most widespread modifications of histones and serve as the key modulators of gene transcription and chromatin structure9. Histone acetylation and deacetylation are dynamic processes controlled by the activities of two histone-modifying enzymes, the histone acetyltransferases (HATs) and the histone deacetylases (HDACs). HATs are generally associated with transcriptional activation, while HDACs oppose the activity of HATs by removing acetyl groups from histone tails, which results in chromatin compaction and transcriptional repression10. The balance between the activities of HATs and HDACs is a critical regulator of cell differentiation, proliferation, and apoptosis and plays an important role in numerous developmental processes and disease states.
The HDAC family consists of highly conserved enzymes, and these can be divided into four classes based on their involvement in different cellular and developmental processes10. The vast majority of studies in HDACs have focused on the knockdown or overexpression of HDACs. Aberrant expression of HDACs has been reported in various cancer types11, and HDAC inhibitors (HDACis) are currently attracting enormous attention as anticancer drugs because of their ability to inhibit cancer cell proliferation, induce cell-cycle arrest, and cause cell death12,13. As epigenetic regulators, HDACs also play important roles in embryonic development14,15. Class I HDACs, for instance, are present in most cell types and have been shown to be required for proper formation of the eye, central nervous system (CNS), craniofacial cartilage, pectoral fin, liver, and exocrine pancreas16,17,18,19. Recently, HDACs have been reported to play essential roles in vertebrate heart tube formation by activating Wnt/β-catenin signaling20. However the requirements for individual HDACs have not been fully determined.
HDAC1 has a wide expression pattern and is important for multiple aspects of normal embryonic development10. Deletion of murine HDAC1 leads to lethality before embryonic day 10.5, and these mice display severe proliferation defects and growth retardation21. In zebrafish, HDAC1 activity is required to promote specification of neural progenitors in the developing brain by antagonizing the gene expression of Notch targets17,22. HDAC1 has also been shown to exert its function in early dorsal–ventral and brain patterning by repressing the expression of canonical Wnt genes and activating the expression of non-canonical Wnt signaling23,24. Additionally, HDAC1 has a region-specific effect during embryonic development. In the hindbrain, HDAC1 mutations cause a significant reduction in cell proliferation in zebrafish17. In contrast, HDAC1 promotes cell cycle exit in the retina and regulates retinal neurogenesis by blocking Wnt and Notch signaling pathways19,22.
We have previously reported important roles of HDACs in hair cell regeneration in zebrafish lateral line neuromasts and have investigated the expression of Class I and Class II HDACs after HDACi treatment25. In the current work, we have used HDACis in zebrafish embryos to show the requirement of HDAC in ear development. We show that HDACi treatment leads to defects in ear development, including abnormal otoliths and fewer sensory hair cells. We also show that HDAC1 is expressed in the developing inner ear and that knockdown of HDAC1 results in severe malformation of the otolith and semicircular canal during ear morphogenesis. Reduction of HDAC1 activity also affected hair cell differentiation in the inner ear. Finally, we report the association of HDAC1 activity with cell proliferation, cell survival, and the expression of hearing-associated genes. Taken together, these results indicate that the chromatin modification factor HDAC1 might be an important regulator of the formation and maintenance of auditory organs in zebrafish.
Results
HDAC inhibition by Trichostatin A (TSA) or valproic acid (VPA) induces otic malformations during embryogenesis
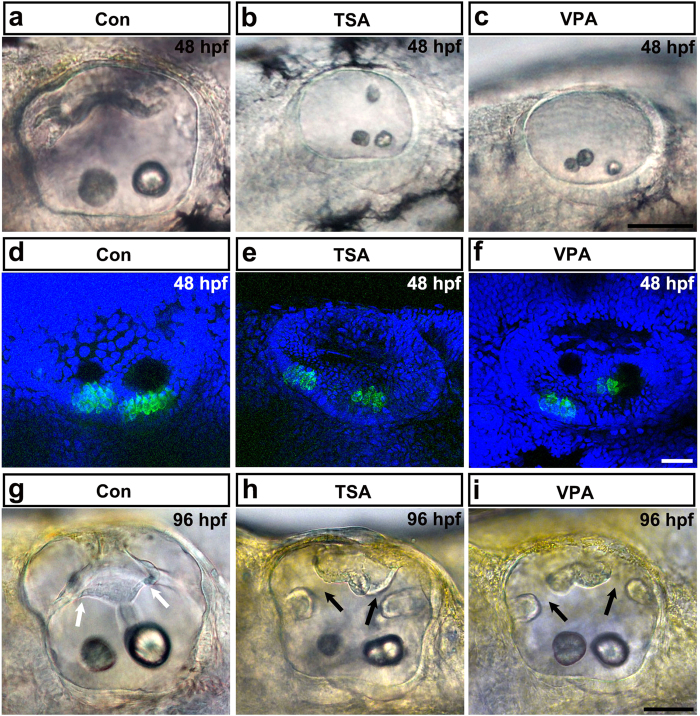
To investigate the function of HDAC activity during inner ear development, zebrafish embryos were continuously treated from 12 hours post fertilization (hpf) onwards with 0.1 μM TSA or 100 μM VPA. At 48 hpf, we found that about 58% (98/169) of the TSA-treated and 42% (43/103) of the VPA-treated embryos exhibited a variety of abnormal otolith morphologies, including multiplication, aggregation, or ectopic location (Fig. 1a–c). In addition, we observed that hair cell numbers in the maculae of HDACi-treated embryos were reduced compared with control embryos at 48 hpf (Fig. 1d–f and Supplemental Figure 1a and e, p < 0.05, ANOVA). In order to determine if the timing of TSA or VPA action corresponds to that of hair cell differentiation in zebrafish, two series of experiments were added. First, TSA or VPA pulse treatments were started at 12 hpf and continued until 19 hpf, a time frame covering the main steps of otic placode development. The number of hair cells was examined at 48 hpf. In a second series of experiments, treatment started from 19 hpf onwards until 48 hpf. When TSA or VPA was present from 19 to 48 hpf, embryos had fewer hair cells compared to the controls (Supplemental Figure 1b,d,e, p < 0.05, ANOVA). However, transient HDACi treatment from 12 hpf to 19 hpf did not lead to hair cell defects (Supplemental Figure 1c, ANOVA). The embryos that were continuously treated with 0.1 μM TSA or 100 μM VPA also exhibited semicircular canal defects at 96 hpf. As shown in Fig. 1g–i, about 63% (68/108) of the TSA-treated and 37% (42/114) of the VPA-treated embryos showed malformed protrusions of the semicircular canals. These results provided further evidence that HDAC activity is important for proper inner ear development. Because TSA and VPA can efficiently inhibit many HDACs, especially HDAC1, our attention was focused on HDAC1.
Figure 1. HDAC inhibitor treatment induces otic abnormalities and reduces the number of hair cells in the inner ear.
(a–c) The morphology of the otoliths in control embryos and in embryos treated with TSA or VPA from 12 hpf onwards. HDAC inhibitor treatment caused morphant otolith defects at 48 hpf of development. (d–f) Images of hair cells expressing GFP in control, TSA-treated, and VPA-treated transgenic zebrafish at 48 hpf. Hair cells were detected by brn3c:gfp expression. HDAC inhibitor treatment reduced hair cell numbers in the inner ear. (g–i) The morphology of the semicircular canals in control (g), TSA-treated (h), and VPA-treated (i) zebrafish. HDAC inhibitor treatment induced aberrant semicircular canal formation. White arrows mark fused pillars, and black arrows mark unfused projections. All images are lateral views with the anterior to the left and the dorsal side up. Scale bars are 50 μm (a–c,g–i) and 20 μm (d–f).
Expression pattern of hdac1 during zebrafish organogenesis
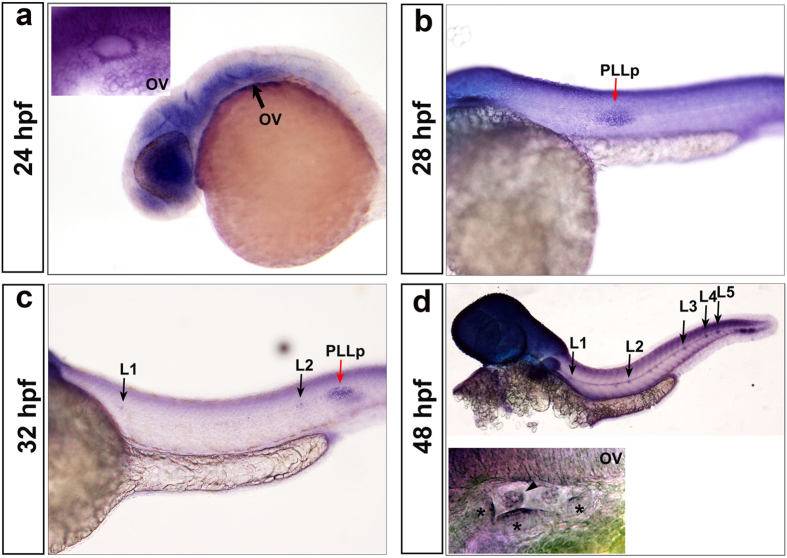
Previous studies reported that hdac1 is ubiquitously expressed in early zebrafish embryos10 and is required for the differentiation of the retina19,22, melanophores26, and motor neurons in the CNS17 as well as in heart development27. Here, we focused on the role of HDAC1 during zebrafish sensory organ development. Whole-mount in situ hybridization data showed that hdac1 expression was mainly concentrated in the encephalic region, eye, and otic vesicle at 24 hpf (Fig. 2a). Hdac1 was also expressed in the migrating posterior lateral line primordium and the deposited neuromasts, and HDAC1 levels were maintained throughout their development (Fig. 2b,c). At 48 hpf, hdac1 was detected in the posterior lateral line neuromasts and remained highly expressed in the brain, including the inner ear (Fig. 2d). Our results are consistent with observations by Cunliffe (2004)17 and suggest that HDAC1 might be correlated with the development of the inner ear.
Figure 2. Zebrafish HDAC1 expression pattern revealed by in situ hybridization.
(a–d) Zebrafish hdac1 is broadly expressed in the otic vesicle ((a,d) OV; indicated by black arrows at 24 hpf and 48 hpf), the posterior lateral line primordium (PLLp) ((b,c); indicated by red arrows at 28 hpf and 32 hpf), and the deposited neuromasts ((c,d) indicated by black arrows at 32 hpf and 48 hpf). The arrowhead shows protrusions of the semicircular canals, and the asterisks show the three cristae.
Suppression of HDAC1 results in severe otic vesicle defects
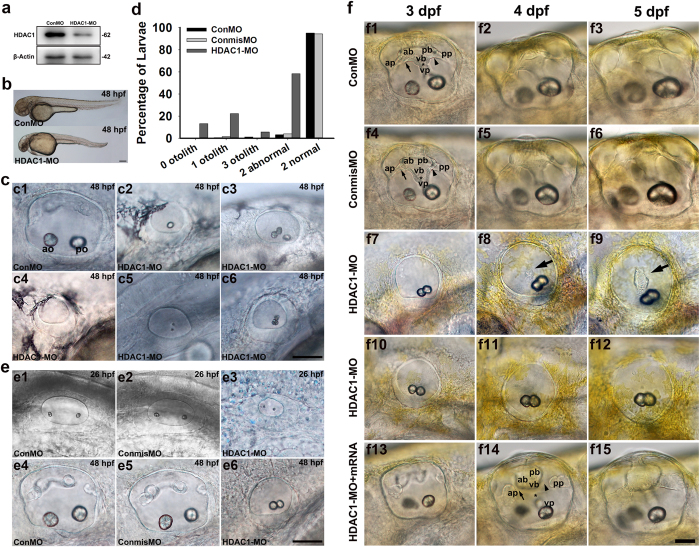
The HDAC1 gene is expressed in the otic vesicle during zebrafish development. To investigate the role of HDAC1, we used an antisense morpholino oligonucleotide (MO) injected into 1–2-cell stage embryos to suppress translation of HDAC1 protein. The efficiency of the morpholino was confirmed by immunoblot detection (Fig. 3a). In contrast to embryos injected with a control morpholino (ConMO), injection of HDAC1 MO caused obvious defects in the gross morphology of 48-hpf zebrafish such as reduced total body length and cardiac edema (Fig. 3b). The reduction in HDAC1 function also resulted in apparent otic defects. Compared to the embryos injected with ConMO, the HDAC1 morphant embryos displayed significant malformations of otoliths (Fig. 3c1–c6). Otolith defects were observed in almost all of the HDAC1 morphants (>99%) but only in a minority (<5%) of control embryos. We categorized morphants into the following five groups according to otolith number: the absent otolith group; the one otolith group; the two normal otolith group, which had otoliths of comparable size to the wild-type embryos; the two abnormal otolith group; and the multiple otolith group (Fig. 3c–d). For zebrafish with two abnormal otoliths, the otoliths were often visibly smaller and/or in the form of ectopic or fused otoliths.
Figure 3. Phenotypes of HDAC1 knockdown by antisense morpholino oligonucleotides.
(a) The HDAC1 protein level in HDAC1-MO-injected embryos was significantly lower than control embryos at 24 hpf. (b) Gross phenotypic morphology of control and HDAC1 morphant embryos. The HDAC1 morphant at 48 hpf shows morphological abnormalities and abnormal otic vesicles. (c) Morphological development of the inner ear requires HDAC1. (c1) Control larvae (48 hpf) have two normal otoliths. The positions of the anterior otolith (ao) and posterior otolith (po) are indicated. Based on the otolith phenotypes, zebrafish HDAC1 morphants were classified into the following five categories: (c2) the one otolith group; (c3) the multiple otolith group; (c4) the absent otolith group; (c5) the two abnormal otolith group; and (c6) the two normal otolith group. (d) Percentages of embryos in each category (n = 321 for ConMO n = 157 for ConmisMO, and n = 211 for HDAC1 morphants). (e) The overall morphology of otic vesicles in control embryos (ConMO and ConmisMO) and HDAC1 morphants at 26 hpf (e1–e3) and 48 hpf (e4–e6). (f) Zebrafish semicircular canal phenotypes in controls (ConMO and ConmisMO), HDAC1 morphants, and HDAC1-MO + HDAC1mRNA co-injected embryos at 3 dpf, 4 dpf, and 5 dpf. Arrows show the junction of the anterior protrusion (ap) and the anterior bulge (ab). Arrowheads show the junction of the posterior bulge (pb) and posterior protrusion (pp). Asterisks show the junction between the ventral bulge (vb) and ventral protrusion (vp). Arrows in f8 and f9 mark ventral projections. The arrow, arrowhead, and asterisk in f14 mark unfused projections. All images are lateral views with the anterior to the left and the dorsal side up. Scale bars are 200 μm (b) and 50 μm (c,e,f).
The absent or one otolith phenotype might be mainly due to the failure of otolith seeding. For HDAC1 morphants with fused otoliths, otoliths were formed but the two otolith precursors might have been seeded too close to each other such that the two otoliths would eventually connect and fuse to become a single otolith. To confirm our hypothesis that HDAC1 is involved in otolith formation, we compared the morphologies of the otic vesicles and the numbers of otoliths between control and HDAC1 morphants early in development. At 26 hpf, the overall morphology of the otic vesicles appeared to be normal in the morphants, but the overall size was reduced (59% compared to controls, n = 40, Fig. 3e1,e3). However, in HDAC1 morphants, fused otoliths located at the position of the saccular maculae were frequently observed, and the size of the otic vesicles was significantly smaller at 48 hpf (68% compared to controls, n = 76) (Fig. 3e4,e6). In addition to the otolith phenotypes, the development of semicircular canals was also abnormal throughout embryonic development. By 3 dpf, HDAC1 morphants showed aberrant semicircular canal formation with absent protrusions of semicircular canals (Fig. 3f) that is consistent with the absence of semicircular canal projections reported by Cunliffe in HDAC1 mutants, and this suggests that HDAC1 function is required for the formation of semicircular canals17. This is not likely due to developmental delay because fusion failure was found in HDAC1 morphants at much later stages (4 and 5 dpf) and because the defects were seen in all three canals. Additionally, our analysis showed that the embryos injected with a 5-bp mismatch MO (ConmisMO) at the same concentration did not produce any of the otic defects observed in HDAC1 morphants (Fig. 3d–f). In order to verify that the semicircular canal defect observed in embryos injected with HDAC1 MO was specifically due to disruption of HDAC1 function, we injected zebrafish HDAC1 mRNA in combination with HDAC1 MO. In this assay, we observed that the majority of the co-injected embryos showed outgrowth of semicircular canals (Fig. 3f15) indicating a partial rescue of the HDAC1 MO phenotype. Taken together, these results suggested that reduction of HDAC1 activity leads to malformations of the otoliths and semicircular canals in the inner ear.
Suppression of HDAC1 results in fewer hair cells
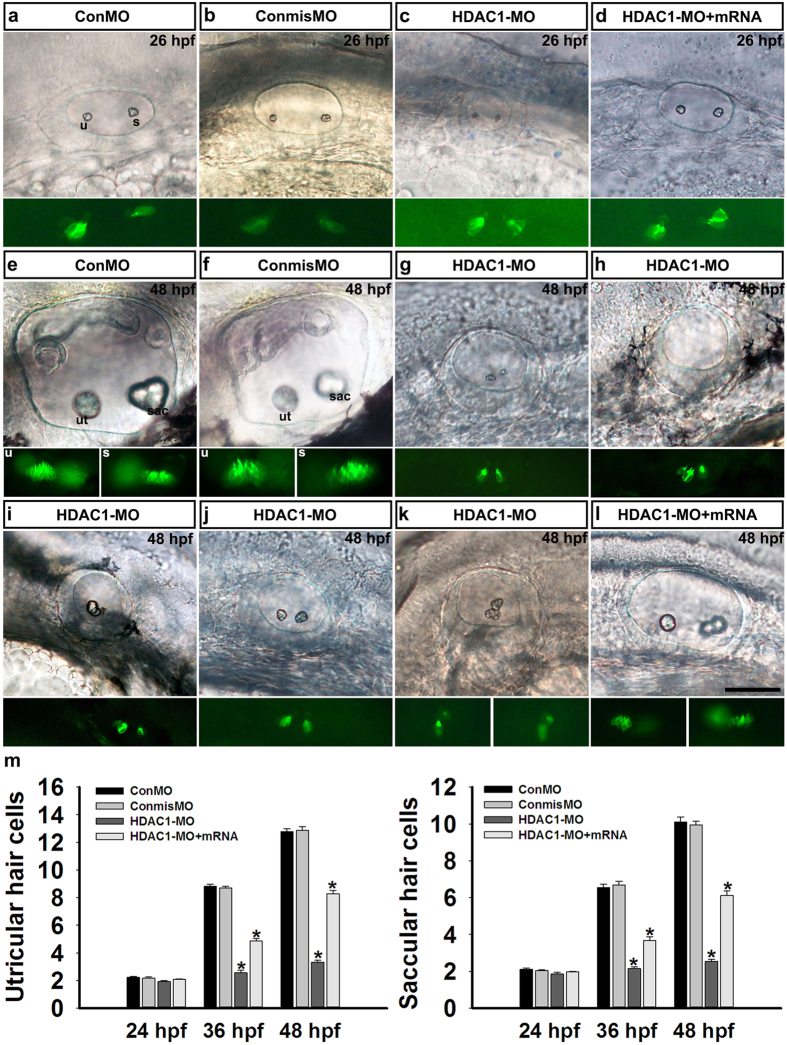
To determine whether knockdown of HDAC1 perturbs hair cell development in the inner ear, we injected HDAC1 MO into transgenic Tg(brn3c:gfp) zebrafish that express GFP in differentiated hair cells28. We first examined the formation of early hair cells (named tether cells) that initially form in pairs and are required for otolith localization29. At 26 hpf, HDAC1 morphants were similar to control embryos, which had a pair of tether cells at both the anterior and posterior ends of otic vesicle (Fig. 4a,c), but occasionally tether cells in HDAC1 morphants appeared to be closer to each other and located on the inside of the otic placode rather than in their correct location. These will eventually fuse at the location of the saccular macula to form a single sensory patch, and this was accompanied by fusion of the two otoliths at 48 hpf (Fig. 4k). Compared to controls, there were significantly fewer later-forming hair cells in all HDAC1 morphants (p < 0.05, ANOVA) (Fig. 4m). Additionally, our analysis showed that the embryos injected with ConmisMO had no defects in hair cell development (Fig. 4b,f, and m, ANOVA). Because otic expression of atoh1a began at 14 hpf in two domains in the otic placode, which marked the primordia of the utricular and saccular sensory epithelia, we next examined expression of atoh1a in embryos injected with HDAC1 MO. In HDAC1 morphants, atoh1a was expressed normally at 14 hpf (Supplemental Figure 2). Together, these results indicated that the absence of HDAC1 has no apparent effect on otolith seeding. However, HDAC1 might be an important regulator in later-forming hair cell formation, which was also confirmed by the effect of HDAC inhibition on hair cell development in the inner ear of zebrafish (Supplemental Figure 1). To confirm whether the effects of HDAC1 MO on hair cell development could be rescued, we co-injected HDAC1 MO with HDAC1 mRNA and observed that co-injected embryos produced more GFP-positive later-forming hair cells than HDAC1 morphants (Fig. 4l,m).
Figure 4. Hair cells in the inner ear.
HDAC1 morphants exhibited a normal number of hair cells at 26 hpf but showed significantly fewer hair cells at later time points. (a–d) Brn3c:gfp expression in the inner ear of a control MO-injected embryo (ConMO) (a), a mismatch MO-injected control embryo (ConmisMO) (b), an HDAC1 morphant (c), and an HDAC1-MO + HDAC1mRNA co-injected embryo (d) at 26 hpf. (e–l) Brn3c:gfp expression in the utricle and saccule of a control MO-injected embryo (ConMO) (e), a mismatch MO-injected control embryo (ConmisMO) (f), HDAC1 morphants (g–k), and an HDAC1-MO + HDAC1mRNA co-injected embryo (l) at 48 hpf. All images show lateral views with the anterior to the left and the dorsal side to the top. Positions of the utricular (ut) and saccular (sac) maculae are indicated. (m) Quantification of the numbers of hair cells in the inner ear for each experimental condition at the indicated stages. Data are shown as mean ± SEM (n = 25–45). *p < 0.05. Scale bars, 50 μm.
Cell division and cell death are affected by suppression of HDAC1
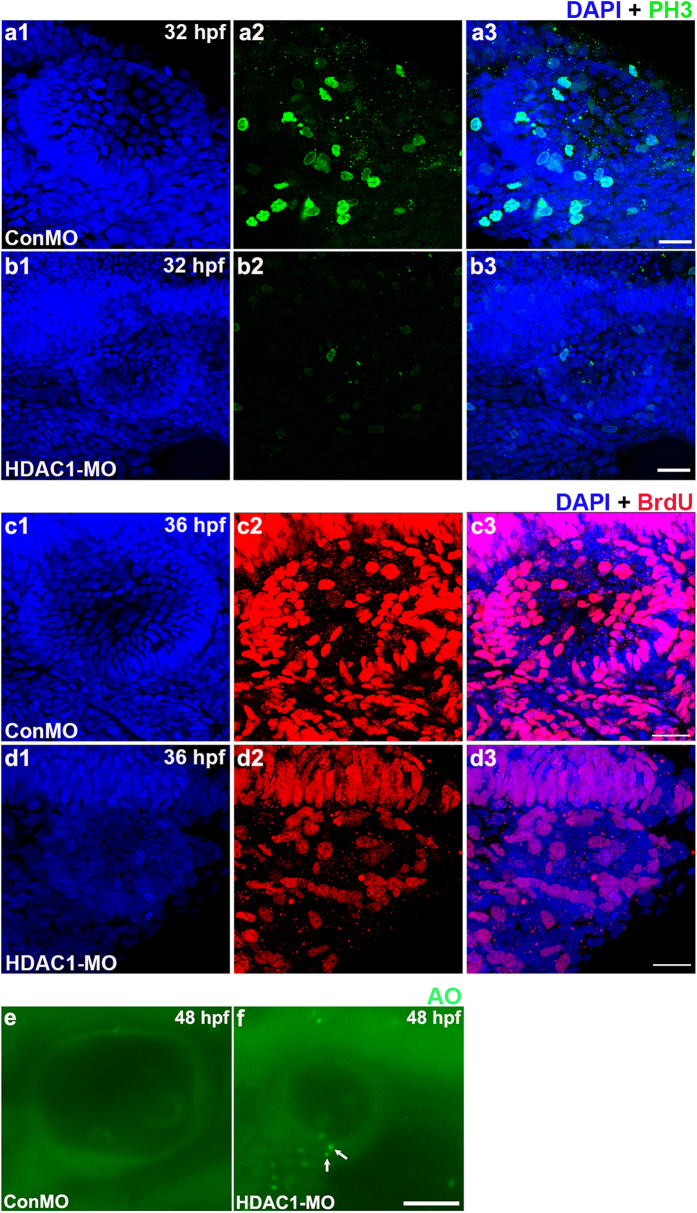
It has been previously observed that HDAC1 plays a role in regulating cell proliferation during mouse and zebrafish development17,19,21, and we have shown that disruption of HDAC1 perturbs otic development and reduces the number of hair cells. To test whether the reduced hair cell number was due to changes in cell proliferation, we used the phospho-histone H3 (PH3) antibody, which labels the cells in M phase, to mark mitotic cells, and observed that the number of PH3-positive cells was significantly reduced in HDAC1 morphant otic vesicles at 32 hpf compared with controls (22 ± 10.5 and 4.4 ± 2.3 PH3-positive cells in ConMO (n = 12) and HDAC1 MO (n = 16) otic vesicles, respectively; p < 0.001, Student t-test) (Fig. 5a,b). We used BrdU uptake to determine the numbers of proliferating cells in the otic vesicles at 36 hpf and found an apparent decrease in BrdU-positive cells in HDAC1 morphant otic vesicle compared to controls (Fig. 5c,d). Interestingly, these proliferating cells seem to be mainly localized to the anterior and posterior otic epithelium in the inner ear of control embryos, whereas there were very few proliferating cells in HDAC1 morphants, which is in agreement with the smaller inner ear and reduced number of hair cells observed in HDAC1 morphants.
Figure 5. Analysis of cell proliferation and death in the otic vesicle of HDAC1 morphants.
(a,b) Mitotic cells in the 32 hpf otic vesicle of a control embryo (a) and an HDAC1 morphant (b) were identified by phospho-histone H3 expression, a mitotic marker. (c,d) Dividing cells in the otic vesicle of a control embryo (c) and an HDAC1 morphant (d) at 36 hpf were identified by BrdU staining. HDAC1 morphants often contained decreased numbers of proliferating cells within the sensory epithelia. (e,f) AO labeling of dying cells in the otic vesicle of a control embryo (e) and an HDAC1 morphant (f) at 48 hpf. White arrows indicate AO-positive cells. HDAC1 morphants often contained multiple dying cells within the sensory epithelia. All images show lateral views with the anterior to the left and the dorsal side up. Scale bars, 20 μm (a–d), Scale bars, 50 μm (e,f).
To test whether increased cell death of placodal cells also contributes to the reduction of otic vesicle size in HDAC1 morphants, we used acridine orange (AO) vital dye to examine the dying cells. At 48 hpf, we found significantly more AO-positive cells in HDAC1 morphants than in control otic vesicles (1.6 and 4.7 AO-positive cells per ear in controls (n = 37) and HDAC1 morphants (n = 40), respectively; p < 0.001, Student t-test) (Fig. 5e–f). The majority of AO-positive cells (56%) were seen within the developing maculae of HDAC1 morphants. These results indicated that cell death is nearly undetectable in sensory epithelial cells of controls, but is common in HDAC1 morphants, and this confirmed that HDAC1 directly or indirectly influences hair cell survival. Taken together, these data show HDAC1 is required in regulating cell proliferation and survival within the inner ear.
Effect of HDAC1 knockdown on the expression of otic markers
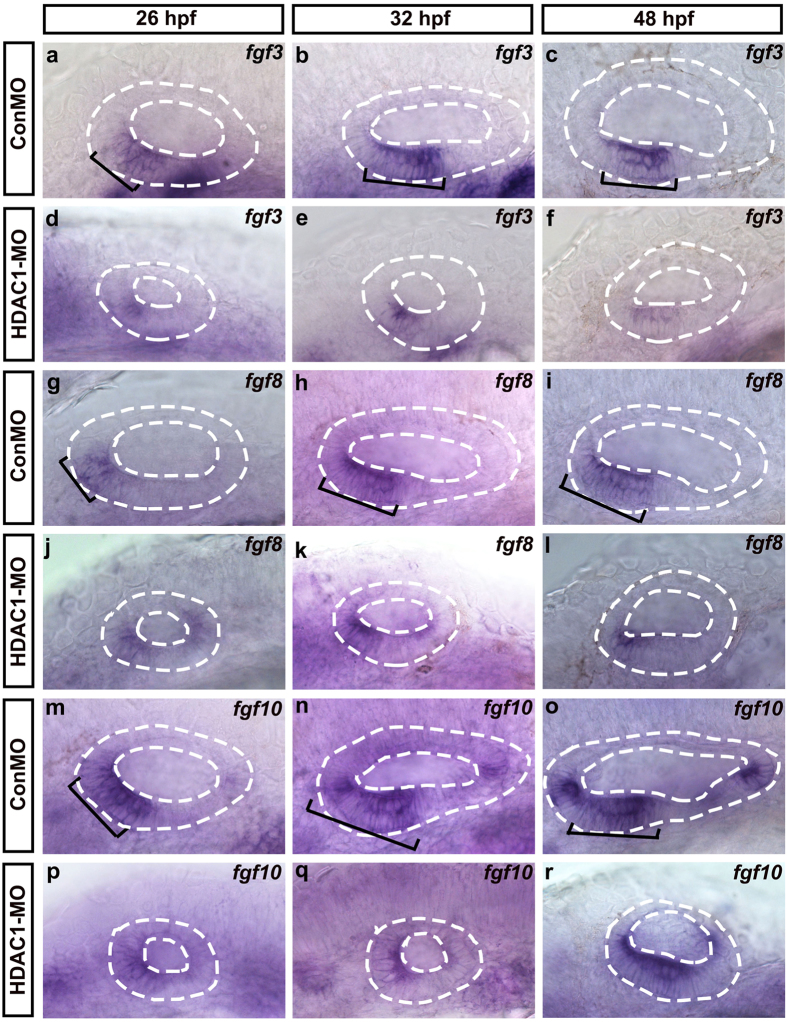
To determine whether or not HDAC1 is required at the earliest stages of otic placode development, we examined expression of three early placode markers—pax2a, cldna, and eya1—in embryos injected with HDAC1 MO at 14 hpf. We found that HDAC1 MO injection did not affect the expression of otic pax2a, cldna, or eya1 (Supplemental Figure 3), and the overall size of the otic placode was normal in the morphants. This suggests that the absence or reduced size of otic vesicles in HDAC1 morphants might not be due to defects in the earliest stages of otic placode induction. To better understand the role of HDAC1 in otic development, we performed an in situ hybridization to examine the expression of several genes known to regulate otic vesicle specification and patterning. Mutational studies in zebrafish have suggested an important role for Fgf signaling in otic induction and vesicle patterning5,30, and in the current work we first tested whether reduction of HDAC1 activity affects the expression of Fgf genes. We found that the zebrafish fgf3, fgf8, and fgf10 genes were expressed normally in the ventroanterior maculae of the otic vesicles in control embryos at 26 hpf, 32 hpf, and 48 hpf, but in morphants the reduction of HDAC1 activity appears to have reduced the expression of these genes during otic development (Fig. 6). These results suggested that HDAC1 might be required to maintain the expression of Fgf ligands in the developing anterior otic vesicle.
Figure 6. The effect of HDAC1 knockdown on the expression of fgf3/8/10 in the developing inner ear.
Whole-mount in situ hybridizations with fgf3 (a–f), fgf8 (g–l), and fgf10 (m–r) probes in control embryos (a–c,g–i,m–o) and HDAC1 MO-injected embryos (d–f,j–l,p–r) at different developmental stages (as indicated). Black brackets indicate the localization of fgf3-expressing (a–c), fgf8-expressing (g–i), and fgf10-expressing (m–o) cells. Embryos injected with HDAC1 morpholino show reduction in levels of fgf3 (d–f), fgf8 (j–l), and fgf10 (p–r) expression. The otic vesicles are outlined by dashed lines. All images show lateral views with the anterior to the left and the dorsal side up.
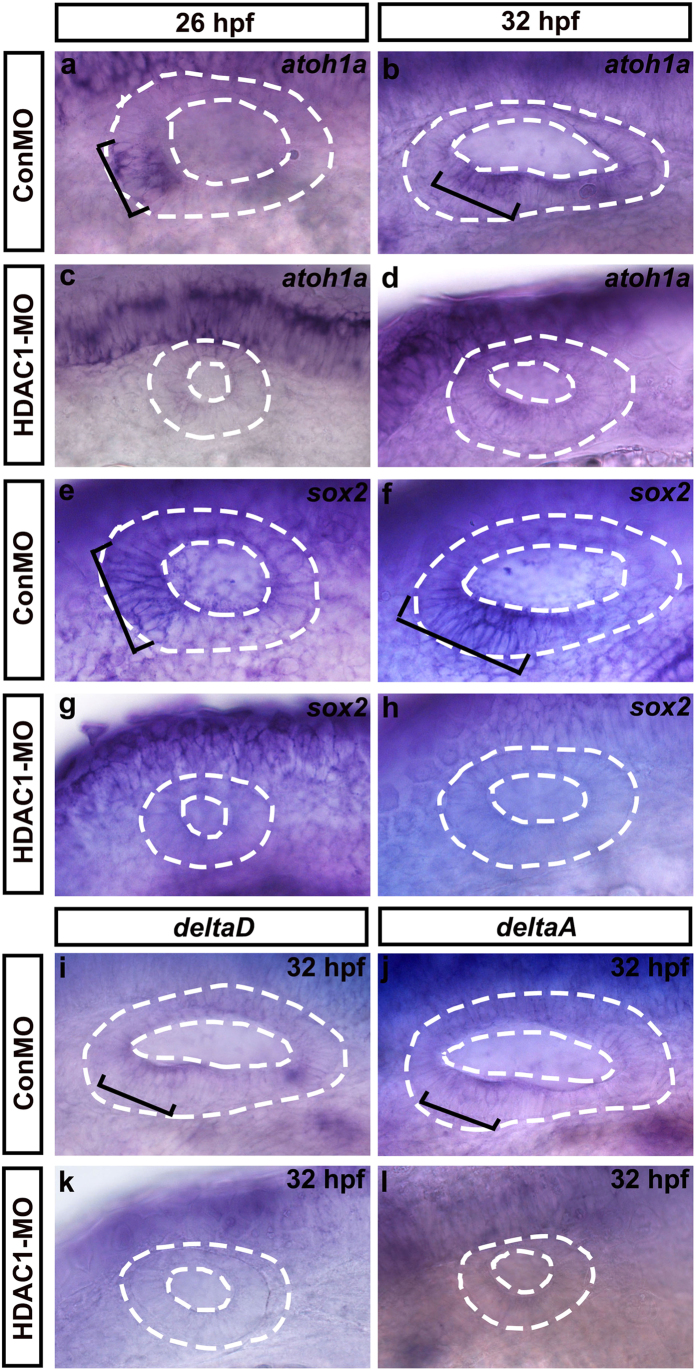
Math1 (mouse atonal homolog 1), a bHLH proneural gene, has been shown to be required for hair-cell specification in mice31. At 26 hpf, atoh1a (the zebrafish homolog of math1) was expressed in the anterior ventral otic domain that contains precursors of both hair cells and supporting cells (Fig. 7a). When HDAC1 was knocked down, anterior expression of atoh1a was significantly reduced compared to control otic vesicles (Fig. 7c). At 32 hpf, atoh1a expression was still visible in the sensory maculae in controls (Fig. 7b), but in HDAC1 morphants atoh1a expression was diminished and marked only a few cells in the anterior patch (Fig. 7d). We next examined the expression of sox2, which is normally co-expressed with atoh1a and marks both hair cells and supporting cells in the developing sensory epithelia. At 26 hpf and 32 hpf, the expression of sox2 in controls was intense along the ventral otic vesicle floor with elevated expression in the macular domains, from which the utricular and saccular maculae will develop (Fig. 7e,f), and there was a significant reduction in the expression of sox2 in HDAC1 morphants at these stages (Fig. 7g,h).
Figure 7. Whole-mount in situ hybridizations to atoh1a, sox2, deltaD, and deltaA.
Lateral views of control (ConMO) and HDAC1 morphant otic vesicles at 26 hpf and 32 hpf. Black brackets indicate the regions of atoh1a-expressing (a,b) and sox2-expressing (e,f) cells as appropriate. Embryos injected with HDAC1 morpholino show a reduction in levels of atoh1a (c,d) and sox2 (g,h) expression. (i–l) Whole-mount in situ hybridizations to deltaD and deltaA. Lateral views of control (ConMO) and HDAC1 morphant otic vesicles at 32 hpf. Black brackets indicate the localization of deltaD-expressing (i) and deltaA-expressing (j) cells. Loss of function of HDAC1 reduced the expression of deltaD (k) and deltaA (l). Otic vesicles are outlined by dashed lines. All images show lateral views with the anterior to the left and the dorsal side up.
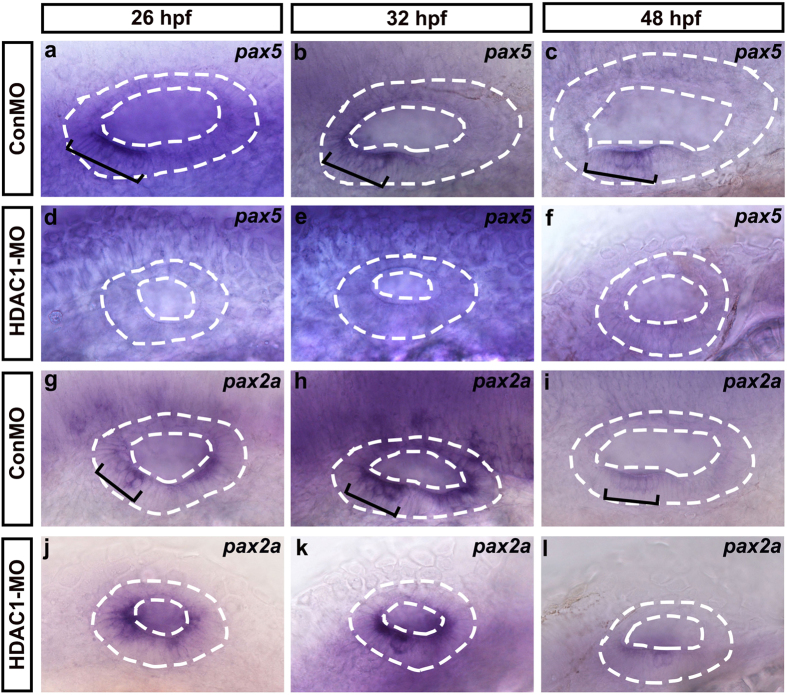
Notch pathway genes deltaD and deltaA are the earliest targets of atoh1 in the zebrafish inner ear, and they eventually become restricted to emerging hair cells32. Therefore, the defects in the otic maculae in HDAC1 morphants prompted us to examine the expression patterns of deltaD and deltaA. We found that both deltaD and deltaA expression in the ventromedial wall of the otic vesicle was severely reduced or ablated at 32 hpf in HDAC1 morphants (Fig. 7i–l). We next examined the expression of Pax-family genes, which are required for development and maintenance of hair cells within the sensory patches in the otic vesicle33. Knocking down HDAC1 diminished pax5 expression, which is normally expressed in the anterior domain of the otic vesicle at 26 hpf and is restricted to the anterior-medial domain at 48 hpf (Fig. 8a–f). Down-regulation of HDAC1 did not obviously diminish pax2a expression (which is a ventromedial marker of the otic vesicle) at 26 hpf (Fig. 8g,j), but at 32 hpf and 48 hpf pax2a expression in the anterior macula of the HDAC1 morphants was significantly reduced compared to controls (Fig. 8h–i,k–l). These data showed that full expression of pax5 and pax2a requires HDAC1 function.
Figure 8. Whole-mount in situ hybridizations to pax5 and pax2a.
(a–l) Lateral views of control (ConMO) and HDAC1 morphant otic vesicles at 26 hpf, 32 hpf, and 48 hpf. Black brackets indicate the localization of pax5-expressing (a–c) and pax2a-expressing (g–i) cells as appropriate. Embryos injected with HDAC1 morpholino show reductions in the levels of pax5 (d–f) and pax2a (j–l) expression. The otic vesicles are outlined by dashed lines. All images show lateral views with the anterior to the left and the dorsal side up.
Discussion
Epigenetic chromatin remodeling events are essential for a wide range of cellular processes and disease states34. By altering chromatin structure, HDACs play important roles in cell growth arrest and subsequent differentiation. HDAC1 is a class I enzyme that has been shown to be ubiquitously expressed in early zebrafish embryos. HDAC1 has been shown to be required in the differentiation of the heart, eyes, neurons, oligodendrocytes, pectoral fin, and ear in zebrafish17,18,19,22. A recent study using antisense morpholino-mediated knockdown of HDAC1 in zebrafish also suggested that HDAC1 is involved in oligodendrocyte development by regulating Wnt signaling35. Class I HDACs are highly expressed in the developing brain and otic vesicles in both chicken and mouse embryos36, but the function of HDAC1 in inner ear organogenesis is less well characterized. In this study, consistent with previous observations, the whole-mount in situ hybridization results clearly demonstrated the expression of HDAC1 in the developing inner ear of the zebrafish, suggesting the potential role of HDAC1 in the development of auditory organs.
We investigated the function of HDAC1 during inner ear development by examining morpholino antisense oligonucleotide knockdown in zebrafish embryos. Placode induction and vesicle cavitation appeared to occur normally, but morphogenesis of the otic vesicle was affected by HDAC1 deficiency. We observed obvious differences in the phenotypes of the ear between HDAC1 morphants and controls. The significant morphological defects of HDAC1 morphants included impairment of otolith formation, reduced otic vesicle size, and reduced numbers of hair cells in the maculae of the inner ear. Therefore, we proposed that HDAC1 deficiency likely disrupts the biological processes that are critical for the formation of the otic vesicle in the zebrafish.
HDAC1 plays a crucial role in regulating cell proliferation and cell death during development. HDAC1 mutant mouse embryos died at very early stages of development, mainly due to an overall reduction of proliferation that was correlated with a decrease in cyclin-dependent kinase activity and upregulation of the cyclin-dependent-kinase inhibitors p21 and p2721. In zebrafish, HDAC1-deficient embryos pass through gastrulation and survive longer and develop to later embryonic stages than mouse HDAC1 mutants, but HDAC1-deficient embryos also exhibit a severe defect of cell proliferation in the hindbrain17,23,24. Here we found that HDAC1 deficiency reduced cell proliferation and induced cell death in the zebrafish ear, which might contribute to the reduced number of later-forming hair cells. Moreover, the role of HDAC1 activity in cell cycle regulation is not uniform but is tissue specific. HDAC1 stimulates proliferation in many contexts during development, but in the zebrafish retina HDAC1 promotes cell cycle exit, and cyclin D and E transcripts are up-regulated in the retina when HDAC1 activity is lost19. This suggests that HDAC1 has both positive and negative roles in cell cycle regulation depending on the location and the cell types. We found that HDAC1 deficiency led to reduced proliferation over a broader region in the zebrafish inner ear, and this is consistent with previous reports that there is an overall reduction of cell proliferation in zebrafish HDAC1 morphant embryos. Decreases in cell proliferation in the otic vesicles of HDAC1 morphants could also be due to induction of cell death, so we performed a cell death analysis by AO staining. We observed significant differences in the number of AO-positive cells in the otic epithelium of HDAC1 morphants compared to controls, indicating the important role of HDAC1 in cell survival in the otic vesicle of zebrafish. Additionally, we found that the otic defects in HDAC1 morphants can be partially rescued by co-injection of mRNA indicating that it has a redundant function. It is possible that HDAC1 is more globally required for embryogenesis, consistent with the global expression of this gene in zebrafish embryos. Thus, HDAC1 morphants showed severe developmental defects similar to hdac1 mutants reported earlier16,17. Although we cannot exclude the possibility that the relationship between otic defects and inherent variability of the antisense MO, the 5-bp mismatch HDAC1 MO (ConmisMO) was injected at the same concentration to better address this problem.
Our results indicate that HDAC1 is essential for otic morphogenesis in zebrafish, but very little is known about the mechanisms of HDAC1 in ear development. Previous zebrafish studies have identified many developmental signaling molecules that are responsible for the formation of the otic vesicle37. As an important epigenetic (transcriptional) regulator, HDAC1 deficiency could alter the normal expression of many genes involved in developmental processes. HDACs have been implicated in various developmental signaling events, including homeobox gene repression38, Fgf signaling17,39, Notch signaling17, and Wnt signaling22,23,24. Studies in zebrafish reported that hindbrain-derived Fgf family members are involved in the initial induction of the otic placode and that otic-derived Fgf signals are later involved in the formation and patterning of the otic vesicle, and inhibition of Fgf can prevent inner ear development3,5,40,41. For example, fgf3 and fgf8 are expressed in the hindbrain and have redundant roles in zebrafish otic patterning3,4,5. The zebrafish fgf3 or fgf8 mutants have fused otoliths and fewer hair cells in the utricular maculae at 48 hpf42,43. These results were similar to HDAC1 morphants, which have smaller otic placodes and vesicles as well as fused otoliths, and this prompted us to investigate the temporal and spatial expression patterns of Fgf genes in HDAC1 morphants. At 26 hpf, the expression of fgf3, fgf8, and fgf10 was observed in the ventroanterior quadrant of the ear in HDAC1 morphants, which was similar to controls, but the expression level was greatly reduced compared to controls. At 48 hpf, the expression of fgf3, fgf8, and fgf10 was shifted to the ventral side of the inner ear in control embryos, while in many morphants the expression was significantly decreased in the anterior side of the ear, suggesting that the expression domains were affected by the reduction in HDAC1 activity. However these expression domains were not entirely eliminated, indicating that the differentiation of otic epithelium still occurred to some extent without HDAC1. The requirement for HDAC activity in regulating Fgf signaling has been shown in Xenopus mesoderm induction and in the developing zebrafish39,44. HDAC1/2 can be recruited to a transcriptional corepressor, RERE, which is implicated in multiple embryonic tissues by modulating the Fgf signaling pathway during embryogenesis, and this imparts tissue specificity to histone deacetylation44. Thus it will be interesting to investigate the potential interactions between the Fgf signaling pathway and HDAC1 during zebrafish inner ear development. Interestingly, we found that HDAC1 morphants failed to form semicircular canal pillars at 96 hpf; however, fgf3 knockdown embryos have normal semicircular canal pillar and crista development, suggesting that other key developmental regulators might cooperate with HDAC1 activity to induce the development of the semicircular canal. Therefore, it will be important to identify the gene targets of HDAC1 regulation and to determine how HDAC1 mediates the different signaling events during otic development and semicircular canal morphogenesis.
In this study, we showed that the formation of the tether cells in early development is normal in HDAC1 morphants. However, HDAC1 deficiency leads to the reduction of later-forming utricular and saccular hair cells in a manner similar to the phenotypes observed in the atoh1a morphant32. Atoh1a is necessary for hair cell development, and it is primarily responsible for specifying later-forming hair cells in the zebrafish inner ear. We examined the expression pattern of atoh1a in the HDAC1 morphants by whole-mount in situ hybridization and found that HDAC1 deficiency strongly diminished atoh1a expression in the developing otic maculae, suggesting that HDAC1 might be required for the normal activation of atoh1a gene expression. It has been shown that zebrafish atoh1a is regulated by Fgf signaling, and fgf3 and fgf8 are upstream activators of atoh1a gene expression during the development of the ear32. Thus, in the current study, the reduced expression of atoh1a in the otic vesicles of HDAC1 morphant embryos is probably due to the loss of Fgf signals. This suggests that appropriate levels of HDAC1 are necessary for regulation of the Fgf signaling pathway and subsequent activation of genes required for hair cell development in the zebrafish inner ear.
A variety of genes are induced and maintained by Fgfs at the otic placode and otic vesicle stages of ear development. For example, the transcription factors pax2 and pax5, which are expressed at specific times, are required for specification and patterning of otic vesicles and play key roles in otic development33,37. Pax2 is abundantly expressed at the midbrain–hindbrain boundary and in the hindbrain, the optic stalk, and the otic vesicles41,45. In zebrafish, loss of pax2 affects hair cell development but does not hinder otic induction46. A recent study showed that HDAC1 and HDAC2 play an important role in the regulation of pax2. HDAC inhibition stimulates histone hyperacetylation of pax2 and significantly reduces its expression during renal development47. Pax5 is initially detected in the anterior of the nascent otic vesicle at a uniformly high level at 24 hpf, but later it is restricted to the utricular macula6,33. Knockdown of pax5 during larval development causes vestibular defects and results in the failure to produce the appropriate number of utricular hair cells due to excess apoptosis6. In our experiments, pax2a and pax5 were strongly expressed at the anterior end of the otic vesicle in control embryos, but both were severely reduced in HDAC1 morphant embryos. Previous studies reported that the expression pattern of pax5 in the anterior quarter of the otic vesicle is dependent on Fgf signaling and that fgf3 serves as an upstream regulator of pax56,33. In zebrafish, fgf3 morphants and fgf3-null mutants have very similar defects in the ear as the HDAC1 morphants described here, with strong reduction in pax5 expression in the anterior otic vesicle. These observations indicate that Pax gene expression might be regulated by HDAC1 and that this expression requires Fgf signaling during otic vesicle development. Taken together, our analysis showed that the expression of inner ear markers (fgf3, fgf8, fgf10, atoh1a, sox2 deltaD, deltaA, pax2a, and pax5) are all significantly down-regulated in the otic vesicle of HDAC1 morphants. Although these transcript analysis results support the idea that the events of inner ear development, such as organogenesis and specification, were affected by HDAC1 inhibition, we cannot completely rule out the possibility that the reduced size of the otic vesicle is responsible for the down-regulation of inner ear markers. Microarray analysis will provide important directions for future studies regarding how HDAC1 effects specific genes and developmental processes.
In summary, we found strong HDAC1 expression in the developing otic vesicles of the zebrafish by whole-mount in situ hybridization. We found that knockdown of HDAC1 affects the development of the otic vesicle and reduces the number of hair cells in the inner ear. Decreased cell proliferation and increased apoptosis were observed in the HDAC1 morphants, which might lead to the observed reduction in size of the otic vesicles. Finally, we demonstrated the role of HDAC1 in regulating the Fgf pathway in the zebrafish inner ear. The influence of HDAC1 on Fgf signaling might directly or indirectly influence the development of the inner ear in zebrafish. However, the molecular details of this interaction require further investigation. Our findings provide insight into the role of HDAC1 in otic vesicle formation and suggest that maintenance of histone deacetylation at an appropriate level is critical for normal organogenesis.
Materials and Methods
Zebrafish strains and maintenance
All zebrafish animal experiments were performed following the institutional guidelines approved by the Institutional Animal Care and Use Committee of Fudan University, Shanghai. The experimental protocols were also approved through the Institutional Animal Care and Use Committee of Fudan University, Shanghai. The methods were performed in accordance with the approved guidelines. Zebrafish were maintained in our facility according to standard procedures. The transgenic line brn3c:mGFP was obtained from Professor Zhengyi Chen, our collaborator at Harvard University, while all other embryos were obtained from natural spawning of wild-type adults. The age of embryos and larvae are described as hours post-fertilization (hpf). To prevent pigment formation, embryos older than 48 hpf were treated with 0.003% 1-phenyl-2-thiourea (PTU; Sigma-Aldrich, St Louis, MO, USA) in fish water from 14 hpf onwards.
Pharmacological treatments
Valproic acid (VPA; Sigma-Aldrich) and Trichostatin A (TSA; Sigma-Aldrich) were dissolved either in water (VPA) or DMSO (TSA) at stock concentrations of 200 mM and 500 μM, respectively, and then diluted to the indicated concentrations in fish water. Embryos were raised up to 12 hpf and then HDAC inhibitors were added to the fish water. Embryos were soaked in VPA- or TSA-containing fish water until an appropriate stage. The fish water was replaced every day with fresh chemicals.
Morpholino injection and mRNA rescue
A translation-blocking morpholino oligomer (MO) targeting HDAC1 was purchased from Gene Tools, Inc. (Philomatch, Oregon, USA), and was diluted in sterile water at a stock concentration of 1 mM. The sequences were as follows: HDAC1-MO (5 ng)—TTG TTC CTT GAG AAC TCA GCG CCA T, 5-bp mismatch control (ConmisMO)—TTc TTC CTT cAG AAg TCA cCG CgA T16, and standard control MO (ConMO) (6 ng)—CCT CTT ACC TCA GTT ACA ATT TAT A. To rescue the HDAC1 MO-injected embryos, HDAC1 5′-capped mRNA was synthesized using the mMACHINE in-vitro transcription kit (SP6; Ambion, Austin, TX, USA) according to the manufacturer’s instructions.
Whole-mount in situ hybridization and immunolabeling
Whole-mount in situ hybridization was performed according to standard procedures48. Digoxigenin-labelled probes were prepared as recommended by the manufacturer (Roche, Mannheim, Germany). Details of the probes used are available on request. PH3 (Abcam, Cambridge, UK) immunostaining was performed using standard staining procedures. Briefly, embryos were fixed for 2 h at room temperature (RT) in 4% paraformaldehyde (PFA). Fixed embryos were blocked using 10% newborn donkey serum and incubated with antibodies overnight at 4 °C. PH3 was used at a 1:1000 dilution. Fluorescently labeled embryos were captured with a Leica confocal fluorescence microscope (TCS SP5; Leica, Wetzlar, Germany). Bright-field images were acquired using a Leica fluorescent microscope (Heerbrugg, Switzerland). Images were processed using Photoshop software (Adobe).
BrdU incorporation and analysis
BrdU incorporation was performed as described15. Briefly, zebrafish embryos were incubated in 10 mM 5-bromo-2-deoxyuridine (BrdU; Sigma-Aldrich) between 35 hpf and 36 hpf at 28.5 °C. BrdU incorporation was detected by immunocytochemistry. Larvae were anesthetized in MS-222 (Sigma-Aldrich) and fixed with 4% PFA for 2 h at RT. The fixed larvae were washed three times in PBS with 0.5% Triton X-100 (PBT-2) and placed into 2N HCl for 0.5 h at 37 °C. Larvae were washed again in PBT-2, and nonspecific binding was blocked with blocking solution for 1 h at RT followed by incubation with mouse monoclonal BrdU antibody (1:200 dilution; Santa Cruz, Dallas, TX, USA. Cat. no. sc-32323) overnight at 4 °C. The next day, the larvae were washed and incubated with secondary antibody for 1 h at 37 °C. Fluorescently labeled embryos were captured with a Leica confocal fluorescence microscope (TCS SP5; Leica). Images were processed using Photoshop software (Adobe).
Acridine orange staining
Apoptosis in whole zebrafish larvae was observed with the vital dye acridine orange (acridinium chloride hemi-[zinc chloride], AO; Sigma-Aldrich). AO staining was performed as described previously49. Briefly, larvae were placed in 2 μg/mL AO in fish water in the dark at 28 °C for 30 minutes. Larvae were then washed with fish water 3 times for 10 minutes each. Images were obtained with confocal microscopy (TCS SP5, Leica) and were processed using Photoshop software (Adobe).
Western blot analysis
Total protein was isolated with the AllPrep DNA/RNA/Protein Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. Protein concentrations were measured using a BCA protein kit (Thermo Fisher Scientific, Rockford, IL), and proteins were separated on SDS-polyacrylamide gels and transferred onto PVDF membranes (Immobilon-P; Millipore, Bedford, MA, USA). The membranes were blocked with 5% nonfat dried milk in TBST (50 mM Tris-HCl (pH 7.4), 150 mM NaCl, and 0.1% Tween-20) for 1 h at RT and then blotted overnight with HDAC1 antibody (1:1000 dilution, Cell Signaling Technology Inc., Danvers, MA, USA) or anti-β-actin antibody (1:1000 dilution, Sigma) at 4 °C.
Statistics
Prior to analysis, all data were first examined for normality and homogeneity of variances by the Shapiro–Wilk test and Levene’s test, respectively. Data were analyzed by either Student’s t-test or analysis of variance (ANOVA) with multiple comparisons using SigmaPlot (version 12.0 for Windows; Systat Software Inc., CA, USA). All data are presented as the mean ± SD. A p-value < 0.05 was considered statistically significant, and p < 0.001 was considered highly significant.
Additional Information
How to cite this article: He, Y. et al. Histone deacetylase 1 is required for the development of the zebrafish inner ear. Sci. Rep. 6, 16535; doi: 10.1038/srep16535 (2016).
Supplementary Material
Acknowledgments
This work was supported by grants from the Major State Basic Research Development Program of China (973 Program) (2011CB504506, 2015CB965000), the National Natural Science Foundation of China (Nos.81230019, 81470687, 81470692, 81500784), the Program for Changjiang Scholars and Innovative Research Team in Universities (IRT1010), the Jiangsu Province Natural Science Foundation, (BK20140620), the Specialized Research Fund for the Doctor Program of Higher Education (20120071110077), the Fundamental Research Funds for the Central Universities (2242014R30022, NO2013WSN085), the Construction Program of Shanghai Committee of Science and Technology (12DZ2251700), the Major Program of Shanghai Committee of Science and Technology (11441901000), and the China Postdoctoral Science Foundation Funded Project (2014M551328). The authors would like to thank Dong Liu, Min Yu, and Shaoyang Sun for their technical assistance and Yalin Huang for help with the confocal microscope.
Footnotes
Author Contributions Y.Z.H. and H.W.L. conceived and designed the work. Y.Z.H. and R.J.C. wrote the manuscript. Y.Z.H. and D.M.T. performed the zebrafish experiments. Y.Z.H., W.Y.L., and H.W.L. performed data analyses. All authors discussed the data, and all authors reviewed the manuscript.
References
- Fekete D. M. Cell fate specification in the inner ear. Curr Opin Neurobiol. 6, 533–541 (1996). [DOI] [PubMed] [Google Scholar]
- Haddon C. & Lewis J. Early ear development in the embryo of the zebrafish, Danio rerio. J Comp Neurol. 365, 113–128 (1996). [DOI] [PubMed] [Google Scholar]
- Leger S. & Brand M. Fgf8 and Fgf3 are required for zebrafish ear placode induction, maintenance and inner ear patterning. Mech Dev. 119, 91–108 (2002). [DOI] [PubMed] [Google Scholar]
- Phillips B. T., Bolding K. & Riley B. B. Zebrafish fgf3 and fgf8 encode redundant functions required for otic placode induction. Dev Biol. 235, 351–365 (2001). [DOI] [PubMed] [Google Scholar]
- Maroon H. et al. Fgf3 and Fgf8 are required together for formation of the otic placode and vesicle. Development. 129, 2099–2108 (2002). [DOI] [PubMed] [Google Scholar]
- Kwak S. J. et al. Zebrafish pax5 regulates development of the utricular macula and vestibular function. Dev Dynam. 235, 3026–3038 (2006). [DOI] [PubMed] [Google Scholar]
- Jenuwein T. & Allis C. D. Translating the histone code. Science. 293, 1074–1080 (2001). [DOI] [PubMed] [Google Scholar]
- Strahl B. D. & Allis C. D. The language of covalent histone modifications. Nature. 403, 41–45 (2000). [DOI] [PubMed] [Google Scholar]
- Grunstein M. Histone acetylation in chromatin structure and transcription. Nature. 389, 349–352 (1997). [DOI] [PubMed] [Google Scholar]
- de Ruijter A. J., van Gennip A. H., Caron H. N., Kemp S. & van Kuilenburg A. B. Histone deacetylases (HDACs): characterization of the classical HDAC family. Biochem J. 370, 737–749 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weichert W. HDAC expression and clinical prognosis in human malignancies. Cancer Lett. 280, 168–176 (2009). [DOI] [PubMed] [Google Scholar]
- Bi G. & Jiang G. The molecular mechanism of HDAC inhibitors in anticancer effects. Cell Mol Immunol. 3, 285–290 (2006). [PubMed] [Google Scholar]
- Kim D. H., Kim M. & Kwon H. J. Histone deacetylase in carcinogenesis and its inhibitors as anti-cancer agents. J Biochem Mol Biol. 36, 110–119 (2003). [DOI] [PubMed] [Google Scholar]
- He Y. et al. Role of histone deacetylase activity in the developing lateral line neuromast of zebrafish larvae. Exp Mol Med. 46, e94 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Y. et al. Histone deacetylase activity is required for embryonic posterior lateral line development. Cell Proliferat. 47, 91–104 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farooq M. et al. Histone deacetylase 3 (hdac3) is specifically required for liver development in zebrafish. Dev Biol. 317, 336–353 (2008). [DOI] [PubMed] [Google Scholar]
- Cunliffe V. T. Histone deacetylase 1 is required to repress Notch target gene expression during zebrafish neurogenesis and to maintain the production of motoneurones in response to hedgehog signalling. Development. 131, 2983–2995 (2004). [DOI] [PubMed] [Google Scholar]
- Pillai R., Coverdale L. E., Dubey G. & Martin C. C. Histone deacetylase 1 (HDAC-1) required for the normal formation of craniofacial cartilage and pectoral fins of the zebrafish. Dev Dynam. 231, 647–654 (2004). [DOI] [PubMed] [Google Scholar]
- Stadler J. A. et al. Histone deacetylase 1 is required for cell cycle exit and differentiation in the zebrafish retina. Dev Dynam. 233, 883–889 (2005). [DOI] [PubMed] [Google Scholar]
- Kim Y. S. et al. Histone deacetylase is required for the activation of Wnt/beta-catenin signaling crucial for heart valve formation in zebrafish embryos. Biochem Bioph Res Co. 423, 140–146 (2012). [DOI] [PubMed] [Google Scholar]
- Lagger G. et al. Essential function of histone deacetylase 1 in proliferation control and CDK inhibitor repression. Embo J. 21, 2672–2681 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi M. et al. Histone deacetylase 1 regulates retinal neurogenesis in zebrafish by suppressing Wnt and Notch signaling pathways. Development. 132, 3027–3043 (2005). [DOI] [PubMed] [Google Scholar]
- Nambiar R. M. & Henion P. D. Sequential antagonism of early and late Wnt-signaling by zebrafish colgate promotes dorsal and anterior fates. Dev Biol. 267, 165–180 (2004). [DOI] [PubMed] [Google Scholar]
- Nambiar R. M., Ignatius M. S. & Henion P. D. Zebrafish colgatelhdacl functions in the non-canonical Wnt pathway during axial extension and in Wnt-independent branchiomotor neuron migration. Mech Develop. 124, 682–698 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- He Y. Z., Cai C. F., Tang D. M., Sun S. & Li H. W. Effect of histone deacetylase inhibitors trichostatin A and valproic acid on hair cell regeneration in zebrafish lateral line neuromasts. Front Cell Neurosci. 8, 382 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ignatius M. S., Moose H. E., El-Hodiri H. M. & Henion P. D. colgate/hdac1 repression of foxd3 expression is required to permit mitfa-dependent melanogenesis. Dev Biol. 313, 568–583 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montgomery R. L. et al. Histone deacetylases 1 and 2 redundantly regulate cardiac morphogenesis, growth, and contractility. Gene Dev. 21, 1790–1802 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiao T., Roeser T., Staub W. & Baier H. A GFP-based genetic screen reveals mutations that disrupt the architecture of the zebrafish retinotectal projection. Development. 132, 2955–2967 (2005). [DOI] [PubMed] [Google Scholar]
- Riley B. B., Zhu C. W., Janetopoulos C. & Aufderheide K. J. A critical period of ear development controlled by distinct populations of ciliated cells in the zebrafish. Dev Biol. 191, 191–201 (1997). [DOI] [PubMed] [Google Scholar]
- Whitfield T. T. et al. Mutations affecting development of the zebrafish inner ear and lateral line. Development. 123, 241–254 (1996). [DOI] [PubMed] [Google Scholar]
- Bermingham N. A. et al. Math1: An essential gene for the generation of inner ear hair cells. Science. 284, 1837–1841 (1999). [DOI] [PubMed] [Google Scholar]
- Millimaki B. B., Sweet E. M., Dhason M. S. & Riley B. B. Zebrafish atoh1 genes: classic proneural activity in the inner ear and regulation by Fgf and Notch. Development. 134, 295–305 (2007). [DOI] [PubMed] [Google Scholar]
- Pfeffer P. L., Gerster T., Lun K., Brand M. & Busslinger M. Characterization of three novel members of the zebrafish Pax2/5/8 family: dependency of Pax5 and Pax8 expression on the Pax2.1 (noi) function. Development. 125, 3063–3074 (1998). [DOI] [PubMed] [Google Scholar]
- Surani M. A., Hayashi K. & Hajkova P. Genetic and epigenetic regulators of pluripotency. Cell. 128, 747–762 (2007). [DOI] [PubMed] [Google Scholar]
- Cunliffe V. T. & Casaccia-Bonnefil P. Histone deacetylase 1 is essential for oligodendrocyte specification in the zebrafish CNS. Mech Develop. 123, 24–30 (2006). [DOI] [PubMed] [Google Scholar]
- Murko C. et al. Expression of class I histone deacetylases during chick and mouse development. Int J Dev Biol. 54, 1527–1537 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mackereth M. D., Kwak S. J., Fritz A. & Riley B. B. Zebrafish pax8 is required for otic placode induction and plays a redundant role with Pax2 genes in the maintenance of the otic placode. Development. 132, 371–382 (2005). [DOI] [PubMed] [Google Scholar]
- Chang Y. L. et al. Essential role of Drosophila Hdac1 in homeotic gene silencing. P Natl Acad Sci USA. 98, 9730–9735 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu R. H. et al. Histone acetylation is a checkpoint in FGF-stimulated mesoderm induction. Dev Dynam. 218, 628–635 (2000). [DOI] [PubMed] [Google Scholar]
- Zelarayan L. C. et al. Differential requirements for FGF3, FGF8 and FGF10 during inner ear development. Dev Biol. 308, 379–391 (2007). [DOI] [PubMed] [Google Scholar]
- Liu D. et al. Fgf3 and Fgf8 dependent and independent transcription factors are required for otic placode specification. Development. 130, 2213–2224 (2003). [DOI] [PubMed] [Google Scholar]
- Reifers F. et al. Fgf8 is mutated in zebrafish acerebellar (ace) mutants and is required for maintenance of midbrain-hindbrain boundary development and somitogenesis. Development. 125, 2381–2395 (1998). [DOI] [PubMed] [Google Scholar]
- Herzog W. et al. Fgf3 signaling from the ventral diencephalon is required for early specification and subsequent survival of the zebrafish adenohypophysis. Development. 131, 3681–3692 (2004). [DOI] [PubMed] [Google Scholar]
- Plaster N., Sonntag C., Schilling T. F. & Hammerschmidt M. REREa/atrophin-2 interacts with histone deacetylase and Fgf8 signaling to regulate multiple processes of zebrafish development. Dev Dynam. 236, 1891–1904 (2007). [DOI] [PubMed] [Google Scholar]
- Hans S., Liu D. & Westerfield M. Pax8 and Pax2a function synergistically in otic specification, downstream of the Foxi1 and Dlx3b transcription factors. Development. 131, 5091–5102 (2004). [DOI] [PubMed] [Google Scholar]
- Riley B. B., Chiang M. Y., Farmer L. & Heck R. The deltaA gene of zebrafish mediates lateral inhibition of hair cells in the inner ear and is regulated by pax2.1. Development. 126, 5669–5678 (1999). [DOI] [PubMed] [Google Scholar]
- Chen S. W. et al. Histone Deacetylase (HDAC) Activity Is Critical for Embryonic Kidney Gene Expression, Growth, and Differentiation. J Biol Chem. 286, 32775–32789 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thisse C. & Thisse B. High-resolution in situ hybridization to whole-mount zebrafish embryos. Nat Protoc. 3, 59–69 (2008). [DOI] [PubMed] [Google Scholar]
- He Y. Z. et al. Trans-2-phenylcyclopropylamine regulates zebrafish lateral line neuromast development mediated by depression of LSD1 activity. Int J Dev Biol. 57, 365–373 (2013). [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.