ABSTRACT
Treatment of Staphylococcus aureus infections has become increasingly difficult because of the emergence of multidrug-resistant isolates. Development of a vaccine to prevent staphylococcal infections remains a priority. To determine whether clumping factor A (ClfA) is a good target protein for inclusion in a multivalent vaccine, we evaluated its efficacy in a variety of relevant staphylococcal infection models, challenging with different S. aureus strains. ClfA adsorbed to Alhydrogel and mixed with Sigma Adjuvant System was more immunogenic and stimulated a more robust Th17 response than ClfA administered with alum alone. ClfA immunization induced the production of functional antibodies in rabbits and mice that blocked S. aureus binding to fibrinogen and were opsonic for S. aureus strains that produced little or no capsular polysaccharide. Mice immunized with ClfA showed a modest reduction in the bacterial burden recovered from subcutaneous abscesses provoked by S. aureus USA300 strain LAC. In addition, the ClfA vaccine reduced lethality in a sepsis model following challenge with strain Newman, but not ST80. Vaccination with ClfA did not protect against surgical wound infection, renal abscess formation, or bacteremia. Passive immunization with antibodies to ClfA did not protect against staphylococcal bacteremia in mice or catheter-induced endocarditis in rats. Some enhancement of bacteremia was observed by ClfA immunization or passive administration of ClfA antibodies when mice were challenged by the intraperitoneal route. Although rodent models of staphylococcal infection have their limitations, our data do not support the inclusion of ClfA in an S. aureus multivalent vaccine.
IMPORTANCE
Antibiotics are often ineffective in eradicating Staphylococcus aureus infections, and thus, a preventative vaccine is sorely needed. Two single-component vaccines and two immunoglobulin preparations failed to meet their designated endpoints in phase III clinical trials. Importantly, recipients of an S. aureus surface protein (iron surface determinant B) vaccine who developed a staphylococcal infection experienced a higher rate of multiorgan failure and mortality than placebo controls, raising safety concerns. Multicomponent S. aureus vaccines have now been generated, and several include surface protein clumping factor A (ClfA). We immunized mice with ClfA and generated a robust T cell response and serum antibodies that were functional in vitro. Nonetheless, ClfA was not protective in a number of relevant animal models of S. aureus infection, and high levels of ClfA antibodies enhanced bacteremia when mice were challenged with community-acquired methicillin-resistant S. aureus strains. Evidence supporting ClfA as a vaccine component is lacking.
INTRODUCTION
Staphylococcus aureus is a Gram-positive, extracellular bacterium that causes both superficial and invasive infections, such as abscesses, sepsis, bacteremia, and endocarditis (1). It is among the most frequently isolated bacterial pathogens in hospitals, and during the past decade, community-acquired methicillin-resistant S. aureus (CA-MRSA) strains with high virulence have infected individuals without underlying risk factors (1, 2). Treatment of staphylococcal infections has become increasingly difficult because of the emergence of multidrug-resistant strains (3, 4). As such, development of a vaccine to prevent S. aureus infections remains a priority.
S. aureus expresses a broad range of cell surface proteins that play important roles during the pathogenesis of S. aureus-caused disease (5, 6). Many of these proteins are anchored to the cell wall of S. aureus by the enzyme sortase A (7); they modulate bacterial adherence to host cells by engaging host extracellular matrix molecules, such as fibronectin, collagen, and fibrinogen (Fg). Cell wall-anchored protein A binds to the Fc fragment of IgG and to the Fab portion of VH3-type B cell receptors (8), resulting in bacterial evasion of the host immune response (6, 8).
S. aureus clumping factor A (ClfA) is a major staphylococcal adhesin. ClfA binds to dimeric host Fg through the carboxy-terminal domain of the Fg gamma chain, resulting in bacterial aggregation in plasma or in purified Fg (9). As the major Fg binding protein, ClfA mediates staphylococcal binding to immobilized Fg- or fibrin-coated surfaces, promoting bacterial adherence to blood clots and biomaterials (9, 10). ClfA also binds to complement factor I, resulting in cleavage of the complement opsonin C3b (11, 12). In addition, ClfA has been reported to bind in vitro to serum apolipoprotein E (13) and human platelets in a fibrinogen-independent manner (14).
The full-length ClfA protein comprises an N-terminal Fg binding domain (the A region), followed by a variable number of serine-aspartate dipeptide repeats, a sorting signal, and a C-terminal wall-spanning region (6, 9). The N-terminal A region is composed of three separately folded domains: N1, N2, and N3. The N2 and N3 subdomains of ClfA (ClfAN23; amino acids [aa] 221 to 559) form IgG-like folds that bind Fg (6, 15), whereas the N1 subdomain (aa 40 to 220) is required for export and cell wall localization (16). Recently, the serine-aspartate repeats were shown to be modified by O-linked N-acetylglucosamine residues (17, 18).
ClfA is recognized as an important S. aureus virulence factor, since it has been shown to enhance staphylococcal virulence in experimental models of septic arthritis (19), sepsis (20), endocarditis (10), and skin infection (21). Josefsson et al. (22) constructed a mutant ClfA protein that no longer binds to Fg by mutating P336 and Y338 to serine and alanine, respectively. An S. aureus strain producing the ClfA mutant protein was attenuated in murine models of septic arthritis, lethality, and abscess formation (21, 22). Moreover, Flick et al. (23) showed that mice that produced Fg lacking the ClfA binding motif showed a consistent survival advantage compared to wild-type mice when challenged intravenously (i.v.) with a lethal dose of S. aureus Newman or USA300 FPR3757.
Preclinical studies testing ClfA as a vaccine antigen showed modest protection in rodent models of S. aureus septic arthritis, lethality, endocarditis, and abscess formation, but not osteomyelitis (19, 22, 24–26). Inhibitex first targeted ClfA in an immunotherapeutic approach to prevent S. aureus infection in humans. INH-21 (Veronate) was a pooled human immunoglobulin preparation from donors with high antibody titers against two Fg binding proteins—ClfA from S. aureus and SdrG from Staphylococcus epidermidis. In a phase III clinical trial, neonates received either placebo or 750 mg of INH-A21/kg of body weight, but the product failed to protect against late-onset sepsis by S. aureus or S. epidermidis (27, 28). Tefibazumab (Aurexis), a humanized monoclonal antibody that binds ClfA, was evaluated in a phase II trial in hospitalized patients with documented S. aureus bacteremia who received either a single tefibazumab dose plus standard therapy or standard therapy alone (29). To evaluate efficacy, a composite clinical endpoint was used, consisting of a relapse of S. aureus bacteremia, a complication related to bacteremia, or death. In the tefibazumab group, 2 of 30 patients reached the clinical endpoint compared with 4 of 30 patients in the placebo group (P = 0.455). The most notable vaccine failure was the cell wall-anchored S. aureus protein antigen iron surface determinant B, which was compared to a placebo in patients prior to elective cardiothoracic surgery with median sternotomy. The IsdB vaccine did not reduce the rate of serious postoperative staphylococcal infections, but it was associated with a significantly higher rate of multiorgan failure and mortality among vaccine recipients who developed S. aureus infections (30).
Subsequent to these clinical failures and those of other vaccine antigens (30, 31), the focus toward vaccine development against S. aureus has moved toward development of a multicomponent vaccine, particularly one that elicits not only a Th2 response but also Th1 and Th17 responses in the host. ClfA was included in vaccines that reached early clinical trials sponsored by both Pfizer (32) (ClinicalTrials.gov NCT01018641) and GlaxoSmithKline (33) (ClinicalTrials.gov NCT01160172), and ClfA is included in the ongoing Pfizer phase IIb trial of SA4Ag vaccine in humans having elective lumbar spinal fusion procedures (NCT02388165). However, neither group has published preclinical data showing that ClfA was protective in relevant models of S. aureus infection. Recently, scientists from Pfizer reported that immunization with recombinant ClfA protected mice from lethality induced by 109 CFU of a Lactococcus lactis strain that was engineered to produce abundant ClfA on its surface (34). Their report did not include challenge with S. aureus, a pathogenic microbe with many virulence determinants that allow it to adhere to and damage host tissues and evade the immune system.
To further evaluate the impact of immunization with S. aureus ClfA on staphylococcal disease, we investigated its efficacy as a protective immunogen in a variety of relevant staphylococcal infection models. In our investigations, we challenged the animals with several different S. aureus strains. Our results do not confirm those of other groups who reported that ClfA was protective in preventing staphylococcal disease.
RESULTS
ClfA immunogenicity.
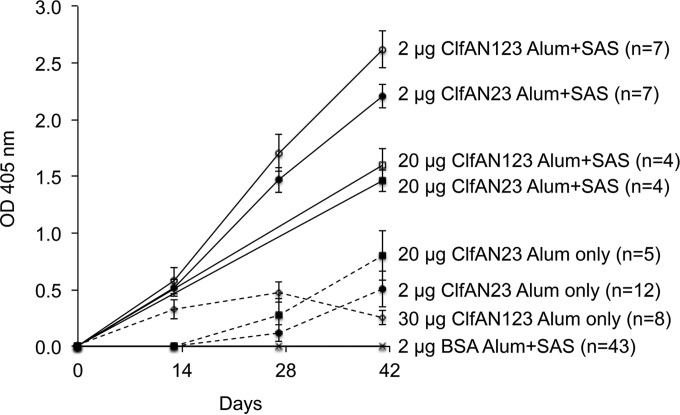
Mice were actively immunized with the N1, N2, and N3 subdomains of ClfA (ClfAN123) (aa 40 to 599) or ClfAN23 (aa 221 to 559) at doses ranging from 2 µg to 30 µg per mouse. Control animals were vaccinated with either bovine serum albumin (BSA) or an irrelevant Shigella flexneri 2a O antigen conjugated to Pseudomonas aeruginosa exoprotein A (2a-Epa) vaccine. When the ClfA protein (2, 20, or 30 µg) was adsorbed to alum, the antibody responses elicited by ClfAN123 and ClfAN23 were fairly comparable (Fig. 1). The antibody levels were enhanced when the ClfA protein (adsorbed to alum) was mixed with Sigma Adjuvant System (SAS) prior to immunization. As shown in Fig. 1, the optimal dose for either ClfAN123 or ClfAN23 was 2 µg/mouse.
FIG 1 .
ClfA-specific antibody levels in sera from mice immunized on days 0, 14, and 28 with various quantities of ClfAN123, ClfAN23, or BSA, as determined by ELISA at a 1:100 serum dilution. Solid lines depict mice immunized with vaccines adsorbed to alum and then mixed with SAS. Dashed lines depict mice immunized with vaccines adsorbed to alum only.
T cell-derived interleukin 17 (IL-17) and interferon gamma (IFN-γ) play important roles in host clearance of experimental infection of mice with S. aureus (35, 36). When we compared the T cell response of mice vaccinated with ClfA adsorbed to alum with that of mice vaccinated with ClfA adsorbed to alum and then mixed with SAS (ClfA plus alum/SAS) prior to subcutaneous (s.c.) injection, only the latter group elicited a demonstrable T cell response in the immunized mice. Lymph node cells recovered from mice given ClfA adsorbed to alum did not produce IFN-γ or IL-17 in response to in vitro stimulation with ClfA (Fig. 2A and B), nor could we detect significant intracellular cytokine staining compared to controls (Fig. 2C and D). However, cells from mice immunized with ClfA plus alum/SAS secreted significantly more IFN-γ and IL-17 than did cells from control mice injected with Tris-buffered saline (TBS) plus alum/SAS (Fig. 2E and F). IL-4 secretion could not be detected in any group of mice (not shown). Intracellular cytokine staining experiments were performed with in vitro-stimulated cells to determine whether the cytokines were derived from CD4+ T cells. As shown in Fig. 2G and H, vaccination of mice with ClfAN23 plus alum/SAS resulted in a significant increase in the numbers of both Th1 (CD4+ IFN-γ-positive [IFN-γ+]) and Th17 (CD4+ IL-17-positive [IL-17+]) cells in the draining lymph nodes compared to the control mice, indicating that immunization of mice with ClfA adsorbed to alum plus SAS elicited both Th1 and Th17 immune responses.
FIG 2 .
T cell response in mice immunized s.c. with ClfA or Tris-buffered saline (TBS) administered with alum (A, B, C, and D) or alum mixed with SAS (E, F, G, and H). Lymph node cells from immunized mice were stimulated with either 10 µg/ml ClfAN23 or medium alone for 6 days. IFN-γ (A and E) and IL-17 (B and F) in the culture supernatants were analyzed by ELISA. Antigen-specific responses by CD4+ lymph node cells were assessed by ex vivo restimulation followed by flow cytometric analyses of IFN-γ- and IL17-producing T cells. The data presented are pooled from six immunized mice in two independent experiments. Panels C, D, G, and H depict the fold change in the percentage of CD4+ T cells producing IFN-γ (C and G) or IL-17 (D and H) in mice vaccinated with TBS versus ClfAN23 and stimulated in vitro with 10 µg/ml ClfA.
Inhibition of S. aureus binding to Fg by anti-ClfA IgG.
S. aureus binds to Fg, and ClfA is the major Fg binding protein (9). To determine whether antibodies elicited by ClfA vaccination were functional, we assessed whether the ClfA antibodies would block S. aureus binding to Fg immobilized on a 96-well plate. As shown in Fig. 3, the binding of S. aureus Newman Δspa mutant strain to human Fg was blocked in a dose-dependent fashion by serial dilutions of either rabbit IgG to ClfA (Fig. 3A) or serum from mice immunized with ClfA (Fig. 3B). Similar results were obtained in blocking experiments performed with the wild-type Newman strain (not shown). In contrast, control antibodies to capsular polysaccharide type 8 (CP8)-Epa (Fig. 3A) or S. aureus Hla (Fig. 3B) showed no Fg binding inhibition. These results indicate that antibodies to ClfA can functionally block the binding of S. aureus surface-associated ClfA to human Fg.
FIG 3 .
Inhibition of S. aureus Newman Δspa mutant strain binding to immobilized Fg by rabbit anti-ClfAN23 IgG (α ClfA) (A) or mouse serum to ClfAN23 (B). Percent binding was calculated by dividing the signal of the antibody-treated S. aureus by the signal of untreated control bacteria.
Opsonic activity of antibodies to ClfA.
Antibodies to S. aureus capsular polysaccharides serotypes 5 and 8 have been shown to mediate opsonophagocytic killing (OPK) of encapsulated S. aureus strains (37–40). Moreover, Nanra et al. reported that antibodies to ClfA were opsonic against encapsulated S. aureus (41). To determine whether the ClfA antibodies utilized in our study were opsonic, we performed OPK assays with HL60 phagocytic cells, serum complement, and antibodies to ClfAN123 or ClfAN23. As shown in Fig. 4A, rabbit IgG raised to either ClfAN23 or ClfAN123 was opsonic for the acapsular CA-MRSA strain LAC (USA300). IgG concentrations of 10 µg/ml resulted in ≥50% killing, and the opsonic activities of ClfAN23 and ClfAN123 IgG were not significantly different at the lower antibody concentrations. In contrast, rabbit IgG to ClfAN23 was not opsonic for strains that were encapsulated, e.g., the serotype 8 strain Reynolds (CP8) and ST80 and the serotype 5 strain Newman (Fig. 4B). Control CP8-Epa antibodies were opsonic for the CP8+ strains Reynolds (CP8) and ST80 (Fig. 4B) but not for the CP5+ strain Newman (not shown). As expected, the irrelevant control IgG to Shigella 2a-Epa was not opsonic.
FIG 4 .

Opsonic activity of antibodies to ClfA against S. aureus strains: acapsular USA300 strain LAC, Reynolds (CP8), CP8+ strain ST80, and CP5+ strain Newman. (A) Rabbit IgG to ClfAN23 or ClfAN123 showed similar opsonic activity against S. aureus LAC. The concentration (in micrograms per milliliter) of anti-ClfAN23 antibody (αClfAN23) or anti-ClfAN123 antibody is shown above the bars. no Ab, no antibody; SEM, standard error of the mean. (B) Rabbit IgG to ClfAN23 was opsonic for USA300 LAC, but it showed no opsonic activity against encapsulated S. aureus Reynolds (CP8), PS80, or Newman. IgG specific for CP8-Epa showed opsonic activity for the serotype 8 strains, while the control 2a-Epa antibodies showed no activity. (C) Threefold serial dilutions of sera from mice immunized with ClfAN23 showed opsonic activity for S. aureus that was inversely proportional to capsule production. The opsonic titers for strains LAC, ST80, Newman, and Reynolds (CP8) were 3,240, 360, 40, and <40, respectively. The data are pooled from at least three independent experiments.
We then tested sera from mice actively immunized with ClfAN23 plus alum/SAS for opsonic activity. As shown in Fig. 4C, strain LAC was susceptible to OPK (>50% killing of the input inoculum) at mouse serum dilutions as high as 1:3,240 (Fig. 4C). The opsonic titers of the mouse ClfAN23 antiserum were substantially lower against the encapsulated isolates—1:360 against the serotype 8 strain ST80 and 1:40 against the serotype 5 strain Newman. The ClfA antiserum showed no opsonic activity against the heavily encapsulated Reynolds (CP8) strain. Serum antibodies to the CP8-Epa conjugate vaccine were opsonic for both CP8+ strains ST80 and Reynolds (CP8), whereas antiserum to the heterologous control vaccine (2a-Epa) was poorly opsonic for all four strains tested. Of note, the acapsular strain LAC showed some nonspecific background killing activity (5 to 25% reduction in the number of CFU/milliliter) with all of the IgG and serum samples tested.
Efficacy of ClfA immunization for the prevention of S. aureus infections.
S. aureus is a major cause of skin and soft tissue infections, and Kwiecinski et al. showed that a ClfA mutant strain of S. aureus that lacked Fg binding showed attenuated virulence in an s.c. abscess infection model (21). These results prompted us to evaluate the ability of immunization with a ClfA vaccine (mutated to abrogate Fg binding) to prevent s.c. abscess formation.
We actively immunized groups of mice with the optimally immunogenic dose of 2 µg ClfAN123, ClfAN23, or BSA, and each vaccine preparation was administered with alum and SAS. Two weeks after the last immunization, S. aureus LAC was mixed with Cytodex beads, and the suspension was injected by the s.c. route into the mouse flanks. Abscesses provoked by strain LAC are not necrotic in this infection model, and we aseptically excised them for quantitative culture 2 days after bacterial challenge. As shown in Fig. 5A, mice immunized with ClfAN23 showed a modest but significant reduction in the abscess bacterial burden compared to the control group immunized with BSA. Immunization with ClfAN123 had no effect, and there were no differences between the groups for abscess weight (Fig. 5B) or mouse weight loss (Fig. 5C).
FIG 5 .
Protective efficacy of ClfA in the mouse model of s.c. abscess formation. Mice were actively immunized with 2 µg ClfAN123, ClfAN23, or BSA adsorbed to alum and mixed with SAS. Two weeks after the third immunization, the animals were challenged s.c. with 4 × 105 CFU of S. aureus LAC. (A to C) Number of CFU/abscess (A), abscess weight (B), and mouse weight change (C) were determined 2 days after challenge. The Kruskal-Wallis test showed a significant (P = 0.0306) difference among the three groups in panel A. The values for the BSA and ClfAN23 groups were significantly different (P = 0.0369) by Dunn’s multiple-comparison posttest and are indicated by a bar and asterisk. Each symbol represents the value for an individual animal, and the median value for each group is indicated by a horizontal line. The lower limit by quantitative culture is indicated by a dotted line. Data are representative of two independent experiments.
Timofeyeva et al. (42) demonstrated that ClfA is produced in vivo in an S. aureus surgical wound infection model, although they observed considerable strain-to-strain variation. In initial experiments, we immunized mice with 30 µg ClfAN23 or ClfAN123 absorbed to alum; on day 42, the animals were subjected to surgical wound infection. Three days after challenge with 90 CFU of S. aureus Newman, the tissue bacterial burden and mouse weight loss were evaluated. Immunization with ClfA provided no protection against staphylococcal wound infection (Fig. 6A) or associated weight loss (Fig. 6B). Additional mice were vaccinated with 2 µg ClfAN23 (mixed with alum and SAS) because this dose resulted in a robust antibody response and elicited a Th1 and Th17 response in the animals (Fig. 2). The immunized mice were challenged with 150 to 300 CFU of S. aureus Newman. As shown in Fig. 6C and D, we observed no additional protection against surgical wound infection or weight loss in mice vaccinated with ClfA plus alum/SAS.
FIG 6 .
Protective efficacy of ClfA immunization in the surgical wound infection model. Mice immunized with 30 µg ClfAN23, ClfAN123, or 2a-Epa (adsorbed with alum) were inoculated with 90 CFU of S. aureus Newman. (A and B) Bacterial burden (A) and weight change (B) were evaluated 3 days after bacterial challenge. Data are representative of three independent experiments. In a separate study, mice actively immunized with 2 µg ClfAN23 or BSA (mixed with alum and SAS) were challenged with 150 to 300 CFU of strain Newman. (C and D) The tissue bacterial burden (C) and weight change (D) were measured on day 3. k. Each symbol represents the value for an individual animal, and the median value for each group is indicated by a horizontal line. The lower limit by quantitative culture is indicated by a dotted line. The data are pooled from two independent experiments.
S. aureus is a major cause of sepsis, a systemic inflammation that arises from bacteremia and affects multiple organs; sepsis often results in a lethal outcome (43). Previous studies indicated that BALB/c mice immunized with 10 µg ClfAN123 administered with alum (24) or 50 µg ClfAN123 mixed with Freund’s adjuvant (20) reduced lethality following challenge with S. aureus 834 or Newman, respectively. Accordingly, we evaluated ClfA as a vaccine candidate in the lethality model. Mice immunized with ClfAN23, 2a-Epa, or BSA were challenged intravenously (i.v.) with ~108 CFU of S. aureus strain Newman or ST80, a serotype 8 CA-MRSA isolate prevalent in Europe (44). Strain Newman appeared more virulent than ST80 in this model, and ClfAN23 immunization increased the survival time in mice challenged with strain Newman (Fig. 7A). However, vaccination with ClfA did not enhance survival in mice challenged with strain ST80 (Fig. 7B), indicating the strain specificity of protection in this model.
FIG 7 .

Protective efficacy of ClfA in mice challenged i.v. with ~108 CFU of S. aureus Newman (8 mice/group) (A) or ST80 (8 to 16 mice/group) (B). Groups of mice were actively immunized with ClfAN23 or BSA mixed with alum plus SAS. Survival was monitored for 14 days, and the results were analyzed by the log rank test. Data in panel B are pooled from two independent experiments. **, P = 0.0048.
Narita et al. also reported that immunization with ClfAN123 and alum significantly reduced the bacterial burden in the kidneys and spleens of mice challenged i.v. with a sublethal dose of the Japanese isolate 834 (24). We immunized mice with ClfAN23 or BSA and challenged them with either S. aureus ST80 or LAC. On day 3, we evaluated the tissue bacterial burdens and weight loss. ClfA vaccination failed to reduce the bacterial burden in the mouse kidneys (Fig. 8A and D) or spleen (Fig. 8B and E). Weight loss for both groups was similar (Fig. 8C and F).
FIG 8 .
Protective efficacy of ClfA in mice challenged i.v. with S. aureus ST80 (2.7 × 107 CFU/mouse) or LAC (1.3 × 108 CFU/mouse). Groups of mice were actively immunized with 2 µg of ClfAN23 or BSA mixed with alum plus SAS. On day 3 after challenge, the bacterial burdens in the kidneys (A and D) and spleens (B and E) and mouse weight loss (C and G) were quantified. Horizontal lines represent group medians, and the lower limit of detection by culture is depicted by dotted lines. The P value for the median values for the two groups in panel A, determined by the Mann-Whitney test, was 0.0648.
Because ClfA is produced in vivo in the murine bacteremia infection model (42), we assessed whether immunization with ClfA might reduce bacteremia. In the initial studies, mice were immunized with either 2 µg ClfA plus alum or BSA plus alum and challenged intraperitoneally (i.p.) with strain Reynolds (CP8) 2 weeks after the third immunization. The results of quantitative blood cultures performed 2 h after bacterial challenge were not significantly different between the two groups (P = 0.1044), although the mice given ClfA showed an overall increase in the number of CFU/milliliter of blood (see Fig. S1A in the supplemental material). None of the mice were still bacteremic on day 5, and kidney cultures were significantly higher (P < 0.05) in the mice given ClfA (Fig. S1B). Changes in mouse weight on day 5 were similar between animals vaccinated with ClfA or BSA (Fig. S1C).
To enhance the possible efficacy of ClfA as a protective immunogen in the bacteremia model, we immunized additional groups of mice with 2 µg BSA or ClfAN23 (with alum plus SAS) and challenged them with S. aureus strains that produce little or no capsular polysaccharide. Quantitative blood culture performed 1.5 h following bacterial challenge with the acapsular strain LAC revealed that mice vaccinated with ClfAN23 showed significantly higher bacteremia levels than control mice given BSA (Fig. 9A). To determine whether the adverse outcome of ClfA immunization was unique to strain LAC, additional mice were immunized and challenged with ST80, which produces trace amounts of CP8 (B. Liu and J. C. Lee, submitted for publication). Mice vaccinated with ClfAN23 showed bacteremia levels similar to those of mice given BSA (Fig. 9B). Likewise, significant differences were not observed in the kidneys, hearts, or spleens of mice immunized with ClfA or BSA and challenged with strain ST80 (data not shown).
FIG 9 .

Protective efficacy of active immunization with 2 µg ClfAN23 (mixed with alum plus SAS) in the murine bacteremia model. Mice were challenged i.p. with 1.5 × 108 CFU of strain LAC (A) or 6 × 107 CFU of strain ST80 (B). Quantitative blood culture were performed 1 to 1.5 h after challenge. Panel A shows data pooled from two independent experiments. The median values for the two groups in panel A were statistically different (P < 0.01) as indicated by the two asterisks.
Efficacy of passive immunization with antibodies to ClfA for the prevention of S. aureus bacteremia.
Because of the elevated LAC bacteremia levels that we observed following active immunization with ClfA, we performed additional bacteremia studies in which naive mice were passively immunized with rabbit IgG. Each mouse received 1 mg of either ClfAN23 IgG or Shigella 2a-Epa IgG by the i.v. route 24 h prior to bacterial challenge by the i.p. route. As shown in Fig. 10, the bacteremia results were dependent upon the S. aureus challenge strain. Mice that received ClfAN23 IgG showed significantly higher bacteremia levels when challenged with either strain LAC (Fig. 10A) (P <0.0001) or ST80 (Fig. 10B) (P <0.05). In contrast, passive immunization had no protective or deleterious effect when the mice were challenged with the serotype 5 Newman strain (Fig. 10C). Moreover, the Newman ΔclfA mutant was not attenuated in the bacteremia model, since mice challenged with the mutant strain showed bacteremia levels similar to those of the parental strain Newman.
FIG 10 .
Protective efficacy of passive immunization with antibodies to ClfA in the mouse bacteremia model. Mice passively immunized i.v. with 1 mg IgG purified by protein A affinity chromatography from rabbits immunized with ClfAN23 or 2a-Epa. (A to C) The following day, the animals were challenged i.p. with S. aureus LAC (A), ST80 (B), or Newman (C). Data presented in panels A and B are pooled from three and two independent experiments, respectively. In panel C, mice were challenged with the wild-type (wt) strain Newman or its isogenic ΔclfA mutant (DU5852). (D to G) Mice passively immunized with 1 mg IgG to ClfA or 2a-Epa were challenged i.v. with S. aureus LAC. Bacteremia was measured 4 h (D) and 3 days (E) after bacterial challenge, and the bacterial burden in the spleen (F) and kidneys (G) was measured 3 days after challenge. Values for the treatment group that are statistically significantly different from the value for the control group administered 2a-Epa IgG are indicated as follows: **, P < 0.01; ****, P < 0.0001.
We considered the possibility that antibodies to ClfA might be preventing Fg-mediated clumping of S. aureus LAC within the peritoneal cavity, allowing S. aureus to reach the bloodstream in higher numbers following i.p. challenge. Therefore, we assessed the bacterial load in both mouse blood and peritoneal washes 1.5 h after S. aureus LAC challenge by the i.p. route. Although mice given antibodies to ClfAN23 had significantly (P = 0.026) higher levels of S. aureus recovered from blood samples (median of 4.51 log10 CFU/ml) than mice given 2a-Epa antibodies (2.49 log10 CFU/ml), similar numbers of staphylococci were recovered from the peritoneal wash fluid samples (medians of 6.18 versus 5.81 log10 CFU/ml for the anti-ClfA and anti-2a-Epa groups, respectively). Plating the peritoneal wash fluid samples following sonication increased the number of CFU/milliliter recovered by a factor of 4- to 5-fold for each mouse group (not shown).
Additional groups of mice were passively immunized before i.v. challenge with 1.2 × 108 CFU S. aureus LAC. Under these conditions, there was no difference in the 4 h postinfection bacteremia levels between mouse groups given antibodies to ClfA or 2a-Epa (Fig. 10D). The mice were euthanized on day 3. At that time point, the bacterial levels in blood samples (Fig. 10E), spleens (Fig. 10F), and kidneys (Fig. 10G) of the mice challenged i.v. were similar for mice given ClfA or 2a-Epa IgG.
Efficacy of passive immunization with antibodies to ClfA for the prevention of S. aureus endocarditis.
Catheterization of rats results in deposition of platelet-fibrin thrombi on the aortic valves, resulting in sterile vegetations (45). When S. aureus is introduced into the bloodstream, it adheres to the damaged valves via the interaction between surface-associated ClfA, platelets, and fibrin (10). Moreillon et al. (10) reported that a clfA mutant of strain Newman was less virulent than the parental strain in the rat model of catheter-induced endocarditis. Moreover, Patti (46) showed that catheterized rabbits treated with ClfA antibodies and vancomycin showed improved bacterial clearance from blood compared to rabbits treated with vancomycin treatment alone.
Accordingly, we passively immunized catheterized rats with 3 mg IgG against ClfAN123, ClfAN23, or 2a-Epa prior to bacterial challenge with S. aureus Newman. Bacteremia was measured daily by quantitative cultures of blood samples, and surviving rats were sacrificed 72 h postinfection. Bacteremia, weight loss, vegetation size, and the bacterial burden in the vegetations and kidneys were assessed. Neither antibodies to ClfAN123 or ClfAN23 protected rats against catheter-induced endocarditis provoked by strain Newman. Compared to the control group of rats that received the irrelevant Shigella 2a-Epa IgG, the rats that received either ClfAN123 or ClfAN23 IgG showed similar results for each parameter of the infection. There were no significant differences between the groups in the vegetation weights (Fig. 11A), and the bacterial burdens in the vegetations (Fig. 11B) and kidneys (Fig. 11C) were comparable. Rats given ClfAN23 IgG showed modestly enhanced survival, but the difference between the groups was not significant (Fig. 11D). Bacteremia levels (Fig. 11E) and weight change (Fig. 11F) over time were similar among the three rat groups.
FIG 11 .
Passive immunization with rabbit IgG specific for ClfAN23 or ClfAN123 failed to protect rats against infective endocarditis induced by S. aureus Newman. (A to D) Vegetation weight (A), number of CFU/vegetation (B), kidney bacterial burden (C), and survival (D) were assessed on day 3. (E and F) Bacteremia (E) and weight change (F) were analyzed at 24, 48, and 72 h after bacterial challenge. Horizontal lines represent group medians. The lower limit of detection by culture is indicated by a dotted line. The depicted data are pooled from three independent experiments.
DISCUSSION
S. aureus ClfA has been regarded as a prime vaccine candidate because it is produced by most clinical isolates of S. aureus, it is surface associated, and it mediates staphylococcal binding to host Fg and fibrin. Moreover, clfA mutants of S. aureus have been shown to be less virulent in preclinical models of lethality, renal abscess formation, and septic arthritis (21, 22), attesting to the importance of ClfA in the pathogenesis of staphylococcal infection.
The majority of ClfA efficacy studies reported in the literature utilized Freund’s adjuvant in the immunization regimen, an adjuvant too reactogenic for use in humans (47). Josefsson et al. reported that mice immunized with 30 µg ClfA and Freund’s adjuvant showed reductions in arthritis and lethality in a septic arthritis model, but protection was dependent on the strain, and the tissue bacterial burdens were not reduced by immunization (19). Likewise, mice immunized with 30 µg of the ClfAPY mutant protein (that lacks Fg binding) mixed with Freund’s adjuvant were protected against lethality induced by strain Newman, but reductions in arthritis were not significant (22). Therapy with ClfA antibodies and vancomycin resulted in better bacterial clearance from the blood of rabbits with catheter-induced S. aureus endocarditis than vancomycin treatment alone (48). However, the bacterial burdens in the tissues of the infected animals were not significantly reduced in these studies. Stranger-Jones et al. (25) immunized mice by the intramuscular route with 100 µg ClfA mixed with Freund’s adjuvant. Compared to mice given phosphate-buffered saline (PBS), ClfA immunization significantly reduced the bacterial burden in the kidneys 4 days after retroorbital challenge with S. aureus Newman (25). Similarly, McAdow et al. showed that immunization with 50 µg ClfA and Freund’s adjuvant increased the survival time of mice challenged with a lethal dose of strain Newman (20). However, the same investigators reported that ClfA antibodies did not protect against lethality induced by S. aureus strain N315 or MW2 (20).
Other vaccine studies have administered ClfA with alum, an adjuvant approved for use in humans. Narita et al. immunized BALB/c mice with 10 µg ClfAN123 adsorbed to alum and demonstrated protection against a lethal challenge with S. aureus 834, a clinical sepsis isolate from Japan (24). The same study reported a reduction in the bacterial burden in the kidneys and spleens of mice immunized with ClfA adsorbed to alum and challenged i.v. with a sublethal dose of strain 834 (24). In a GlaxoSmithKline-supported study, immunization with 20 µg ClfAN123 adsorbed to alum was not efficacious in protecting rats from experimental osteomyelitis when the animals were challenged with two different strains of S. aureus (26). In the most recent study, Scully et al. immunized mice with an undisclosed quantity of ClfAN123 adsorbed to alum and demonstrated that this protected the animals for 24 h from lethality induced by i.v. challenge with 109 CFU of an L. lactis strain expressing plasmid-encoded ClfA (34). Passive immunization with 1.5 mg of ClfA-specific monoclonal antibody 12-9 also protected mice against lethal L. lactis-ClfA challenge in mice. However, the Scully et al. studies do not support the efficacy of ClfA as a vaccine candidate against the pathogen S. aureus.
Our study was designed to evaluate ClfA as a protective immunogen in the absence of Freund’s adjuvant and in mouse models of S. aureus infection that are relevant to human disease. Initially, we compared the immunogenicity of ClfAN123 and ClfAN23. Because ClfAN123 is readily degraded to ClfAN23 when expressed in Escherichia coli (49), ClfAN23 was utilized for the majority of our experiments. Only ClfAN23 has been crystallized for structural analysis (49), and its immunogenicity in mice was similar to that of ClfAN123 in our hands. We report that immunization with ClfA mixed with alum did not induce a Th1 or Th17 response in C57BL6/J mice, but the addition of a monophosphoryl lipid A (MPL)-containing adjuvant (SAS) did induce both a high antibody response and CD4+ lymph node cells that produced IFN-γ and IL-17 upon in vitro stimulation with ClfA. The antibodies elicited by immunization with ClfAN23 showed in vitro functionality because they prevented the binding of S. aureus to immobilized Fg and were opsonic for acapsular S. aureus in an OPK assay.
We immunized mice under what we determined to be optimal conditions (2 µg ClfAN23 adsorbed to alum plus SAS) before evaluating protective efficacy in murine models of S. aureus disease. Our positive results include a modest but significant (0.5-log-unit) reduction in bacterial numbers recovered from subcutaneous abscesses of mice immunized with ClfA. However, protection was observed only in mice given ClfAN23, not in mice vaccinated with ClfAN123. In addition, we were able to achieve a significant increase in survival time in mice immunized with ClfAN23 and challenged with a lethal dose of strain Newman, confirming the results reported by other groups (12, 20, 22). However, ClfA-mediated protection against lethality was not achieved in immunized mice challenged with S. aureus strain ST80, confirming the strain specificity observed previously by another group (20).
Vaccination with ClfA or ClfA IgG was not protective in rodent models of surgical wound infection, catheter-induced endocarditis, or the sublethal i.v. challenge model that results in renal abscess formation. Of note was the enhanced bacteremia that we observed following i.p. challenge with strain USA300 LAC following active immunization with ClfA or passive immunization with rabbit antibodies to ClfA. Passive immunization with ClfA antibodies also enhanced bacteremia in mice challenged with S. aureus ST80, but no effect was seen following passive immunization and challenge with strain Newman. We hypothesized that antibodies to ClfA (but not 2a-Epa) might block the fibrinogen-mediated aggregation of S. aureus within the peritoneal cavity, resulting in enhanced access of the bacteria to the bloodstream. To address this possibility, blood and peritoneal lavage cultures were performed on passively immunized mice challenged i.p. with strain LAC. Although the concentrations of S. aureus were significantly higher in blood samples 1.5 h after bacterial challenge, the peritoneal wash fluid samples from mice given 2a-Epa or ClfA antibodies yielded similar numbers of S. aureus (before or after sample sonication), suggesting that ClfA antibodies were not enhancing bacteremia by preventing bacterial aggregation within the peritoneal cavity. Moreover, we were not able to visualize differences in bacterial aggregation when peritoneal wash fluid samples were concentrated, stained, and visualized by confocal microscopy (data not shown). It is possible that ClfA antibodies prevented Fg-mediated clumping of S. aureus in the bloodstream itself, although the results of plating blood samples from infected mice before and after sonication did not support this hypothesis either (not shown). Nor did ClfA antibodies impact the bacterial burden in the spleens of mice challenged with either strain ST80 or LAC (Fig. 8). If opsonization with ClfA antibodies could promote phagocytosis of the unencapsulated strain LAC in vivo but not promote intracellular bacterial killing by the phagocytes, it is possible that these bacteria persist for longer periods in the bloodstream than nonopsonized S. aureus.
Our results differ from those of other groups who immunized with Freund’s adjuvant and showed that ClfA mediated protection against infection in mice. The protection against lethality (strain Newman) or enhancement of bacteremia (strain LAC) that we observed was strain dependent and affected by the challenge route of bacterial inoculation. We could not reproduce the findings of Narita et al. (24), who immunized BALB/c mice with ClfA adsorbed to alum and reported protection against infection of the kidneys and spleen following i.v. challenge. It is possible that differences in S. aureus challenge strains or inbred mouse strains could account for the discrepancies observed. Murphy et al. described the diversity of the ClfA protein sequence in a collection of clinical isolates (50). Our ClfA vaccine was derived from the strain Newman protein sequence, and Newman ClfAN123 is 91% identical (95% similar) to that produced by ST80 strains (51). However, the enhanced bacteremia that we observed in immunized mice was most striking following challenge with the USA300 strain LAC, which produces a ClfA protein with 99% identity in the N123 region to that of strain Newman (52). Thus, it is unlikely that the lack of efficacy that we observed can be attributed to sequence diversity in the ClfA protein, although Brady et al. demonstrated that the ClfA sequence type could affect ClfA immunogenicity and the quality of the antibody response (52).
By the very nature of rodent studies, they may have limited generalizability to human disease. Moreover, the correlates of protection against staphylococcal infection in humans remain unknown. Nonetheless, current vaccines that target microbial pathogens and are licensed for use in humans do protect mice against infection. Each antigen comprising a multicomponent vaccine should function to enhance protection consistently in at least one S. aureus infection model. The results of our studies do not support the inclusion of a ClfA antigen in a multicomponent S. aureus vaccine.
MATERIALS AND METHODS
Clumping factor A production and purification.
Clumping factor A (ClfA) variants and the control Shigella flexneri 2a O antigen conjugated to Pseudomonas aeruginosa exoprotein A (2a-Epa) were produced and provided by GlycoVaxyn AG, Schlieren, Switzerland. Open reading frames (ORFs) encoding ClfA P336S Y338A (mutations that abrogate ClfA binding to fibrinogen [Fg] [22]) comprising strain Newman N-terminal domains 1, 2, and 3 (ClfAN123) (aa 40 to 559) or N-terminal domains 2 and 3 (ClfN23) (aa 221 to 559) and carrying a C-terminal six-His affinity tag were cloned into a pEC415 expression vector (53) after an OmpA signal sequence. ClfA variants were expressed in Escherichia coli W3110 in shaking flasks with super optimal or terrific broth medium with 100 µg/ml ampicillin and arabinose as inducer. ClfA variants were extracted from the periplasm of E. coli cells using lysozyme. Samples were subjected to immobilized metal ion affinity chromatography by standard procedures, and fractions containing ClfA were pooled. Endotoxin was removed with a high-capacity endotoxin removal kit (Pierce), and ClfA was further purified by size exclusion chromatography in Tris-buffered saline, pH 7.4.
IgG production and purification.
Purified rabbit IgGs were produced and provided by GlycoVaxyn AG, Schlieren, Switzerland. The rabbits were maintained, immunized, and bled at Eurogentec SA (Seraing, Belgium) according to institutional guidelines. New Zealand White rabbits were immunized subcutaneously on days 0, 7, 10, and 18 with 100 µg ClfAN123 or ClfAN23 mixed with Eurogentec’s proprietary adjuvant system according to their speedy protocol. The rabbits were euthanized on day 29, and sera from two rabbits each were pooled. IgG was purified from rabbits immunized with ClfAN123, ClfAN23, 2a-Epa, nontoxic alpha-hemolysin (HlaH35L), or S. aureus capsular polysaccharide type 8 (CP8)-Epa by protein A affinity chromatography as described previously (37).
Bacterial strains and culture conditions.
Bacterial isolates used in this study included S. aureus strains LAC, ST80, Newman, Newman ΔclfA (strain DU5852), Newman Δspa (strain DU5873), and Reynolds (CP8) (9, 38, 44, 54, 55). S. aureus was cultivated overnight in brain heart infusion broth (Difco) for use in fibrinogen binding inhibition studies or in tryptic soy broth (TSB) (Difco) for lethality studies. Opsonophagocytic killing (OPK) assays and bacteremia experiments were performed with staphylococci cultivated for 24 h on Columbia agar (Difco) supplemented with 2% NaCl (38). For all other infection models, S. aureus was harvested from TSB cultures grown to an optical density at 600 nm (OD600) of 0.8.
Active and passive immunization studies.
Animal experiments were carried out in accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals (56), and the protocols were approved by the Harvard Medical School Standing Committee on Animals. Female C57BL/6 mice (4 weeks old; The Jackson Laboratory) were immunized on days 0, 14, and 28 by the s.c. route with ClfA adsorbed to 60 µg Alhydrogel (Brenntag) (alum) or ClfA adsorbed to 60 µg alum and mixed with Sigma Adjuvant System (SAS) (10 µg monophosphoryl lipid A [MPL]). SAS is a stable emulsion of oil in water containing 0.5 mg MPL and 0.5 mg synthetic trehalose dicorynomylate in 44 µl of squalene oil, 0.2% Tween 80, and water. Control animals were immunized similarly with BSA (Sigma) or Shigella 2a-Epa, as described previously (37). Blood was collected from the mice by tail vein puncture before each vaccination and again before challenge. Sera were diluted 1:100 and tested by enzyme-linked immunosorbent assay (ELISA) on 96-well plates coated with 1 µg/ml ClfAN123, as described previously (37). For passive immunization of mice, female Swiss-Webster mice (7 to 8 weeks old; Charles River) were given 1 mg of rabbit IgG in 0.2 ml delivered i.v. 24 h before bacterial challenge.
T cell assays and cytokine analyses.
To define the immune cell populations induced by vaccination, C57BL/6J animals were immunized s.c. as described above with ClfAN23 adsorbed to alum or adsorbed to alum and mixed with SAS. Control mice were given Tris-buffered saline (TBS) mixed with alum or alum plus SAS. One week after the last immunization, the cervical, axillary, and brachial lymph nodes were harvested from each animal, and the cells were passed through 100-µm mesh nylon screens. Cells (2 × 105 cells) were seeded in wells of a 24-well tissue culture plate and stimulated with either 10 µg/ml of ClfAN23 or TBS in a total volume of 1 ml RPMI 1640 supplemented with 100 U/ml penicillin, 100 µg/ml streptomycin, 2 mM l-glutamine, 10% fetal bovine serum, and 50 µM 2-mercaptoethanol. After 6 days in culture, the supernatants were collected and analyzed by ELISA (R&D system) to measure interleukin 17 (IL-17), interferon gamma (IFN-γ), and interleukin 4 (IL-4) production. The lymph node cell cultures were incubated for an additional 6 h with 50 ng/ml phorbol myristate 13-acetate, 1 µg/ml ionomycin, and 1 µl of BD GolgiPlug (BD Biosciences) prior to cell harvest. The cells were washed with staining buffer (0.01 M phosphate buffer, 1% fetal bovine serum, and 0.09% sodium azide) and incubated with fluorescein isothiocyanate (FITC)-labeled anti-CD4 (rat IgG2a; BioLegend) or isotype control antibodies. The cells were then treated with fixation/permeabilization solution (BD Biosciences) and stained with rat phycoerythrin (PE)-labeled anti-mouse IL-17A, allophycocyanin (APC)-labeled anti-IFN-γ, or their isotype control antibodies (BioLegend). Cells gated on CD4 were analyzed by flow cytometry to assess the frequency of Th1 (CD4+ IFN-γ+ IL-17−) or Th17 (CD4+ IL-17+ IFN-γ−) lymphocytes induced by vaccination compared to the frequencies in mice given adjuvant only. The samples were analyzed on a BD FACScan instrument, the data were analyzed using CellQuest software, and the results were expressed as the fold change in percentage of cytokine-producing T cells compared to the cells from control mice.
Fg binding inhibition assay.
The ability of mouse serum or rabbit IgG to inhibit S. aureus binding to immobilized Fg was measured by a previously described assay (57) with slight modifications. Briefly, 5 × 106 CFU of strain Newman Δspa cells in 60 µl was incubated for 1 h at room temperature with an equal volume of serial threefold dilutions of rabbit IgG (3.33 to 0.013 µg/ml), mouse sera, or phosphate-buffered saline (PBS). The treated S. aureus cells were then added in duplicate to wells of a microtiter plate coated with 2 µg/ml human Fg (Calbiochem) and blocked with 5% skim milk. After incubation for 1 h at room temperature, the plates were washed and incubated for 15 min at 37°C with BacTiter-Glo lysis reagent (Promega), which produces a luminescent signal that is proportional to the amount of ATP present in the sample and corresponds to the number of adherent S. aureus. Luminescence was recorded on a SpectraMax L microplate reader (Molecular Devices, LLC), and the percent binding was calculated by dividing the signal of the antibody-treated S. aureus to that of PBS-treated control bacteria.
OPK assay.
The HL60-based OPK assay was performed in 96-well plates as described previously (37). Each well (80 µl) contained 4 × 105 viable and differentiated HL60 cells, 1 × 103 CFU S. aureus, various dilutions of heat-inactivated mouse serum or rabbit IgG, and 0.1% guinea pig serum (Cedarlane) as a complement source. Control samples included S. aureus incubated with complement and HL60 cells (no antibody), S. aureus incubated with antibodies and complement (no HL60 cells), and S. aureus and HL60 cells only. The 96-well plates were incubated at 37°C with shaking at 700 rpm. After 2 h, 20 µl of 1% Triton X-100 was added to each well to lyse the HL60 cells. After a 3-min incubation at 37°C with shaking, 5-µl aliquots of the final reaction mixtures were plated in duplicate on tryptic soy agar plates. The percent change in the number of CFU/milliliter (i.e., killing) was defined as the reduction in the number of CFU/milliliter after 2 h compared with the number of CFU/milliliter at time zero. The opsonic titer was defined as the IgG concentration or serum dilution at which ≥50% of the inoculum was killed.
S. aureus infection models.
Subcutaneous abscess formation was evaluated as described previously (21, 58). Briefly, groups of mice were anesthetized with ketamine and xylazine, and their backs were shaved and disinfected with 70% ethanol. An inoculum of 4 × 105 S. aureus LAC was mixed with an equal volume of sterile dextran beads (Cytodex-1; Sigma), and 0.2 ml of the mixture was injected s.c. into the shaved flanks of immunized mice. After 48 h, the mice were euthanized, and the abscesses were aseptically excised and homogenized in 1 ml TSB. Serial dilutions of the homogenates were cultured quantitatively on tryptic soy agar (TSA) plates, and the results were expressed as the log CFU of S. aureus per abscess.
The mouse model of S. aureus surgical wound infection was previously described (59). Briefly, immunized mice were anesthetized with ketamine and xylazine, and their right thighs were shaved and disinfected. The thigh muscle was exposed, and a 1-cm incision was made in the muscle to the depth of the bone. The incision was closed with one silk (4-0 silk) suture, and 3 µl of an S. aureus suspension (90 to 300 CFU) was introduced into the muscle incision under the suture. The skin was closed with surgical wound clips. After 3 days, the mice were euthanized, and the muscle tissue was excised, weighed, and homogenized. Serial dilutions of the homogenates were plated in duplicate on TSA plates, and the results were expressed as the log CFU of S. aureus per gram of tissue.
For the lethality experiments, groups of 8 to 16 immunized mice were challenged i.v. with ~1 × 108 CFU of S. aureus strain Newman or ST80. Survival was assessed for 14 days. For the sublethal renal abscess infection model (24, 60), mice were injected i.v. with 0.2 ml containing 2.5 × 107 to 1 × 108 CFU S. aureus ST80 or LAC. For certain experiments, the mice were bled by tail vein puncture 4 h after bacterial challenge for quantitative blood cultures. The mice were euthanized on day 3 and bled by cardiac puncture, and the kidneys and spleens were excised, weighed, and homogenized in 1 ml of TSB. Serial dilutions of the samples were plated in duplicate on solid medium, and the results were expressed as the number of CFU of S. aureus per milliliter of blood or per gram of tissue.
The bacteremia model was performed as described previously (37, 38). Briefly, immunized mice were inoculated by the intraperitoneal (i.p.) route with S. aureus in a 0.5-ml volume. Heparinized blood for culture was collected by tail vein puncture 1.5 h after bacterial challenge. Mouse weight loss and the bacterial burdens in the kidneys were quantified on day 4. Alternatively, mice were euthanized 1.5 h after i.p. challenge with S. aureus, and peritoneal lavage fluid samples (3 ml) were collected, serially diluted, and cultured quantitatively.
The endocarditis infection model was performed by methods similar to those we described previously (61). On day 0, male Sprague-Dawley rats (180 to 220 g; Charles River) were anesthetized with ketamine and xylazine. A polyethylene catheter was passed through the right carotid artery and the aortic valve until vigorous pulsation of blood within the catheter was observed. The catheter was sealed and tied in place, and the incision was closed. On day 1, rats with indwelling catheters were passively immunized by i.v. injection of 3 mg of IgG to ClfA or Shigella 2a-Epa. On day 2, a 0.2-ml inoculum containing ~1.5 × 105 CFU S. aureus Newman was administered via the tail vein. On days 3, 4, and 5, heparinized blood was collected from each animal by tail vein puncture, and the number of CFU/milliliter of blood was determined by quantitative plating. Surviving rats were euthanized on day 5, and the aortic valve vegetations and kidneys were excised, weighed, and homogenized in TSB. Serial dilutions of the homogenates were plated in duplicate on TSA plates, and the results were expressed as the number of CFU of S. aureus per vegetation or per gram of tissue.
Statistical analysis.
Statistical analyses were performed using Prism 6 (GraphPad Software, Inc.). P values were calculated using the Mann-Whitney test to compare median values of nonparametric data from two experimental groups. The Kruskal-Wallis test for nonparametric data was used to compare three or more unmatched groups, followed by the Dunn’s multiple-comparison posttest to compare the differences between each pair of groups. Survival rates in lethality experiments were analyzed by the log rank test. P values of <0.05 were considered significant.
SUPPLEMENTAL MATERIAL
Protective efficacy of active immunization with 2 µg ClfAN23 plus alum or 2 µg Epa plus alum in the murine bacteremia model. Mice were challenged i.p. with 4 × 107 CFU of strain Reynolds (CP8). (A) Quantitative blood cultures were performed 2 h after bacterial challenge. (B and C) Five days later, mouse kidneys were excised and cultured quantitatively (B) and percent weight change in the mice was recorded (C). The data are representative of two independent experiments. *, P < 0.05. Download
ACKNOWLEDGMENTS
We thank scientists at GlycoVaxyn AG for providing ClfAN123, ClfAN23, Shigella 2a-Epa, and the rabbit IgG used for these experiments. Timothy Foster generously provided S. aureus Newman ΔclfA mutant (strain DU5852) and Newman Δspa mutant (strain DU5873). Kelly Shields Lapierre and Meghan Dowd provided expert technical assistance.
J.C.L. has served as a consultant on S. aureus vaccine development for Sanofi Pasteur and Crucell.
Funding Statement
The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Wan Beom Park was supported by Seoul National University Hospital, Seoul, Korea
Footnotes
Citation Li X, Wang X, Thompson CD, Park S, Park WB, Lee JC. 2016. Preclinical efficacy of clumping factor A in prevention of Staphylococcus aureus infection. mBio 7(1):e02232-15. doi:10.1128/mBio.02232-15.
REFERENCES
- 1.Lowy FD. 1998. Staphylococcus aureus infections. N Engl J Med 339:520–532. doi: 10.1056/NEJM199808203390806. [DOI] [PubMed] [Google Scholar]
- 2.Dantes R, Mu Y, Belflower R, Aragon D, Dumyati G, Harrison LH, Lessa FC, Lynfield R, Nadle J, Petit S, Ray SM, Schaffner W, Townes J, Fridkin S, Emerging Infections Program−Active Bacterial Core Surveillance MRSA Surveillance Investigators . 2013. National burden of invasive methicillin-resistant Staphylococcus aureus infections, United States, 2011. JAMA Intern Med 173:1970–1978. doi: 10.1001/jamainternmed.2013.10423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lowy FD. 2003. Antimicrobial resistance: the example of Staphylococcus aureus. J Clin Invest 111:1265–1273. doi: 10.1172/JCI18535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chambers HF, Deleo FR. 2009. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat Rev Microbiol 7:629–641. doi: 10.1038/nrmicro2200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Smeltzer MS, Lee CY, Harik N, Hart ME, Crossley K, Jefferson K, Archer G, Fowler V. 2009. Molecular basis of pathogenicity, p 65–108. In Crossley KB, Jefferson GL, Archer GL, Fowler VG (ed), Staphylococci in human disease, 2nd ed. Wiley-Blackwell, Oxford, United Kingdom. [Google Scholar]
- 6.Foster TJ, Geoghegan JA, Ganesh VK, Höök M. 2014. Adhesion, invasion and evasion: the many functions of the surface proteins of Staphylococcus aureus. Nat Rev Microbiol 12:49–62. doi: 10.1038/nrmicro3161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mazmanian SK, Liu G, Ton-That H, Schneewind O. 1999. Staphylococcus aureus sortase, an enzyme that anchors surface proteins to the cell wall. Science 285:760–763. doi: 10.1126/science.285.5428.760. [DOI] [PubMed] [Google Scholar]
- 8.Kim HK, Cheng AG, Kim HY, Missiakas DM, Schneewind O. 2010. Nontoxigenic protein A vaccine for methicillin-resistant Staphylococcus aureus infections in mice. J Exp Med 207:1863–1870. doi: 10.1084/jem.20092514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McDevitt D, Francois P, Vaudaux P, Foster TJ. 1994. Molecular characterization of the clumping factor (fibrinogen receptor) of Staphylococcus aureus. Mol Microbiol 11:237–248. doi: 10.1111/j.1365-2958.1994.tb00304.x. [DOI] [PubMed] [Google Scholar]
- 10.Moreillon P, Entenza JM, Francioli P, McDevitt D, Foster TJ, François P, Vaudaux P. 1995. Role of Staphylococcus aureus coagulase and clumping factor in pathogenesis of experimental endocarditis. Infect Immun 63:4738–4743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hair PS, Ward MD, Semmes OJ, Foster TJ, Cunnion KM. 2008. Staphylococcus aureus clumping factor A binds to complement regulator factor I and increases factor I cleavage of C3b. J Infect Dis 198:125–133. doi: 10.1086/588825. [DOI] [PubMed] [Google Scholar]
- 12.Hair PS, Echague CG, Sholl AM, Watkins JA, Geoghegan JA, Foster TJ, Cunnion KM. 2010. Clumping factor A interaction with complement factor I increases C3b cleavage on the bacterial surface of Staphylococcus aureus and decreases complement-mediated phagocytosis. Infect Immun 78:1717–1727. doi: 10.1128/IAI.01065-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Elkhatib WF, Hair PS, Nyalwidhe JO, Cunnion KM. 2015. New potential role of serum apolipoprotein E mediated by its binding to clumping factor A during Staphylococcus aureus invasive infections to humans. J Med Microbiol 64:335–343. doi: 10.1099/jmm.0.000010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Siboo IR, Cheung AL, Bayer AS, Sullam PM. 2001. Clumping factor A mediates binding of Staphylococcus aureus to human platelets. Infect Immun 69:3120–3127. doi: 10.1128/IAI.69.5.3120-3127.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McDevitt D, Nanavaty T, House-Pompeo K, Bell E, Turner N, McIntire L, Foster T, Höök M. 1997. Characterization of the interaction between the Staphylococcus aureus clumping factor (ClfA) and fibrinogen. Eur J Biochem 247:416–424. doi: 10.1111/j.1432-1033.1997.00416.x. [DOI] [PubMed] [Google Scholar]
- 16.McCormack N, Foster TJ, Geoghegan JA. 2014. A short sequence within subdomain N1 of region A of the Staphylococcus aureus MSCRAMM clumping factor A is required for export and surface display. Microbiology 160:659–670. doi: 10.1099/mic.0.074724-0. [DOI] [PubMed] [Google Scholar]
- 17.Thomer L, Becker S, Emolo C, Quach A, Kim HK, Rauch S, Anderson M, Leblanc JF, Schneewind O, Faull KF, Missiakas D. 2014. N-acetylglucosaminylation of serine-aspartate repeat proteins promotes Staphylococcus aureus bloodstream infection. J Biol Chem 289:3478–3486. doi: 10.1074/jbc.M113.532655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hazenbos WL, Kajihara KK, Vandlen R, Morisaki JH, Lehar SM, Kwakkenbos MJ, Beaumont T, Bakker AQ, Phung Q, Swem LR, Ramakrishnan S, Kim J, Xu M, Shah IM, Diep BA, Sai T, Sebrell A, Khalfin Y, Oh A, Koth C, Lin SJ, Lee BC, Strandh M, Koefoed K, Andersen PS, Spits H, Brown EJ, Tan MW, Mariathasan S. 2013. Novel staphylococcal glycosyltransferases SdgA and SdgB mediate immunogenicity and protection of virulence-associated cell wall proteins. PLoS Pathog 9:e1003653. doi: 10.1371/journal.ppat.1003653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Josefsson E, Hartford O, O’Brien L, Patti JM, Foster T. 2001. Protection against experimental Staphylococcus aureus arthritis by vaccination with clumping factor A, a novel virulence determinant. J Infect Dis 184:1572–1580. doi: 10.1086/324430. [DOI] [PubMed] [Google Scholar]
- 20.McAdow M, Kim HK, Dedent AC, Hendrickx AP, Schneewind O, Missiakas DM. 2011. Preventing Staphylococcus aureus sepsis through the inhibition of its agglutination in blood. PLoS Pathog 7:e1002307. doi: 10.1371/journal.ppat.1002307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kwiecinski J, Jin T, Josefsson E. 2014. Surface proteins of Staphylococcus aureus play an important role in experimental skin infection. APMIS 122:1240–1250. doi: 10.1111/apm.12295. [DOI] [PubMed] [Google Scholar]
- 22.Josefsson E, Higgins J, Foster TJ, Tarkowski A. 2008. Fibrinogen binding sites P336 and Y338 of clumping factor A are crucial for Staphylococcus aureus virulence. PLoS One 3:e2206. doi: 10.1371/journal.pone.0002206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Flick MJ, Du X, Prasad JM, Raghu H, Palumbo JS, Smeds E, Höök M, Degen JL. 2013. Genetic elimination of the binding motif on fibrinogen for the S. aureus virulence factor ClfA improves host survival in septicemia. Blood 121:1783–1794. doi: 10.1182/blood-2012-09-453894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Narita K, Hu DL, Mori F, Wakabayashi K, Iwakura Y, Nakane A. 2010. Role of interleukin-17A in cell-mediated protection against Staphylococcus aureus infection in mice immunized with the fibrinogen-binding domain of clumping factor A. Infect Immun 78:4234–4242. doi: 10.1128/IAI.00447-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stranger-Jones YK, Bae T, Schneewind O. 2006. Vaccine assembly from surface proteins of Staphylococcus aureus. Proc Natl Acad Sci U S A 103:16942–16947. doi: 10.1073/pnas.0606863103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lattar SM, Noto Llana M, Denoël P, Germain S, Buzzola FR, Lee JC, Sordelli DO. 2014. Protein antigens increase the protective efficacy of a capsule-based vaccine against Staphylococcus aureus in a rat model of osteomyelitis. Infect Immun 82:83–91. doi: 10.1128/IAI.01050-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.DeJonge M, Burchfield D, Bloom B, Duenas M, Walker W, Polak M, Jung E, Millard D, Schelonka R, Eyal F, Morris A, Kapik B, Roberson D, Kesler K, Patti J, Hetherington S. 2007. Clinical trial of safety and efficacy of INH-A21 for the prevention of nosocomial staphylococcal bloodstream infection in premature infants. J Pediatr 151:260–265. doi: 10.1016/j.jpeds.2007.04.060. [DOI] [PubMed] [Google Scholar]
- 28.Otto M. 2010. Novel targeted immunotherapy approaches for staphylococcal infection. Expert Opin Biol Ther 10:1049–1059. doi: 10.1517/14712598.2010.495115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Weems JJ, Steinberg JP, Filler S, Baddley JW, Corey GR, Sampathkumar P, Winston L, John JF, Kubin CJ, Talwani R, Moore T, Patti JM, Hetherington S, Texter M, Wenzel E, Kelley VA, Fowler VG. 2006. Phase II, randomized, double-blind, multicenter study comparing the safety and pharmacokinetics of tefibazumab to placebo for treatment of Staphylococcus aureus bacteremia. Antimicrob Agents Chemother 50:2751–2755. doi: 10.1128/AAC.00096-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fowler VG, Allen KB, Moreira ED, Moustafa M, Isgro F, Boucher HW, Corey GR, Carmeli Y, Betts R, Hartzel JS, Chan IS, McNeely TB, Kartsonis NA, Guris D, Onorato MT, Smugar SS, DiNubile MJ, Sobanjo-ter Meulen A. 2013. Effect of an investigational vaccine for preventing Staphylococcus aureus infections after cardiothoracic surgery: a randomized trial. JAMA 309:1368–1378. doi: 10.1001/jama.2013.3010. [DOI] [PubMed] [Google Scholar]
- 31.Shinefield H, Black S, Fattom A, Horwith G, Rasgon S, Ordonez J, Yeoh H, Law D, Robbins JB, Schneerson R, Muenz L, Fuller S, Johnson J, Fireman B, Alcorn H, Naso R. 2002. Use of a Staphylococcus aureus conjugate vaccine in patients receiving hemodialysis. N Engl J Med 346:491–496. doi: 10.1056/NEJMoa011297. [DOI] [PubMed] [Google Scholar]
- 32.Nissen M, Marshall H, Richmond P, Shakib S, Jiang Q, Cooper D, Rill D, Baber J, Eiden J, Gruber W, Jansen KU, Emini EA, Anderson AS, Zito ET, Girgenti D. 2015. A randomized phase I study of the safety and immunogenicity of three ascending dose levels of a 3-antigen Staphylococcus aureus vaccine (SA3Ag) in healthy adults. Vaccine 33:1846–1854. doi: 10.1016/j.vaccine.2015.02.024. [DOI] [PubMed] [Google Scholar]
- 33.Levy J, Licini L, Haelterman E, Moris P, Lestrate P, Damaso S, Van Belle P, Boutriau D. 2015. Safety and immunogenicity of an investigational 4-component Staphylococcus aureus vaccine with or without AS03B adjuvant: results of a randomized phase I trial. Hum Vaccin Immunother 11:620–631. doi: 10.1080/21645515.2015.1011021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Scully IL, Timofeyeva Y, Keeney D, Matsuka YV, Severina E, McNeil LK, Nanra J, Hu G, Liberator PA, Jansen KU, Anderson AS. 2015. Demonstration of the preclinical correlate of protection for Staphylococcus aureus clumping factor A in a murine model of infection. Vaccine 33:5452–5457. doi: 10.1016/j.vaccine.2015.08.029. [DOI] [PubMed] [Google Scholar]
- 35.Lin L, Ibrahim AS, Xu X, Farber JM, Avanesian V, Baquir B, Fu Y, French SW, Edwards JE Jr, Spellberg B. 2009. Th1-Th17 cells mediate protective adaptive immunity against Staphylococcus aureus and Candida albicans infection in mice. PLoS Pathog 5:e1000703. doi: 10.1371/journal.ppat.1000703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cho JS, Pietras EM, Garcia NC, Ramos RI, Farzam DM, Monroe HR, Magorien JE, Blauvelt A, Kolls JK, Cheung AL, Cheng G, Modlin RL, Miller LS. 2010. IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J Clin Invest 120:1762–1773. doi: 10.1172/JCI40891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wacker M, Wang L, Kowarik M, Dowd M, Lipowsky G, Faridmoayer A, Shields K, Park S, Alaimo C, Kelley KA, Braun M, Quebatte J, Gambillara V, Carranza P, Steffen M, Lee JC. 2014. Prevention of Staphylococcus aureus infections by glycoprotein vaccines synthesized in Escherichia coli. J Infect Dis 209:1551–1561. doi: 10.1093/infdis/jit800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Watts A, Ke D, Wang Q, Pillay A, Nicholson-Weller A, Lee JC. 2005. Staphylococcus aureus strains that express serotype 5 or serotype 8 capsular polysaccharides differ in virulence. Infect Immun 73:3502–3511. doi: 10.1128/IAI.73.6.3502-3511.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thakker M, Park J-S, Carey V, Lee JC. 1998. Staphylococcus aureus serotype 5 capsular polysaccharide is antiphagocytic and enhances bacterial virulence in a murine bacteremia model. Infect Immun 66:5183–5189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nanra JS, Buitrago SM, Crawford S, Ng J, Fink PS, Hawkins J, Scully IL, McNeil LK, Aste-Amézaga JM, Cooper D, Jansen KU, Anderson AS. 2013. Capsular polysaccharides are an important immune evasion mechanism for Staphylococcus aureus. Hum Vaccin Immunother 9:480–487. doi: 10.4161/hv.23223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nanra JS, Timofeyeva Y, Buitrago SM, Sellman BR, Dilts DA, Fink P, Nunez L, Hagen M, Matsuka YV, Mininni T, Zhu D, Pavliak V, Green BA, Jansen KU, Anderson AS. 2009. Heterogeneous in vivo expression of clumping factor A and capsular polysaccharide by Staphylococcus aureus: implications for vaccine design. Vaccine 27:3276–3280. doi: 10.1016/j.vaccine.2009.01.062. [DOI] [PubMed] [Google Scholar]
- 42.Timofeyeva Y, Scully IL, Anderson AS. 2014. Immunofluorescence microscopy for the detection of surface antigens in methicillin-resistant Staphylococcus aureus (MRSA). Methods Mol Biol 1085:85–95. doi: 10.1007/978-1-62703-664-1_4. [DOI] [PubMed] [Google Scholar]
- 43.Stearns-Kurosawa DJ, Osuchowski MF, Valentine C, Kurosawa S, Remick DG. 2011. The pathogenesis of sepsis. Annu Rev Pathol 6:19–48. doi: 10.1146/annurev-pathol-011110-130327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Linde H, Wagenlehner F, Strommenger B, Drubel I, Tanzer J, Reischl U, Raab U, Höller C, Naber KG, Witte W, Hanses F, Salzberger B, Lehn N. 2005. Healthcare-associated outbreaks and community-acquired infections due to MRSA carrying the Panton-Valentine leucocidin gene in southeastern Germany. Eur J Clin Microbiol Infect Dis 24:419–422. doi: 10.1007/s10096-005-1341-7. [DOI] [PubMed] [Google Scholar]
- 45.Héraïef E, Glauser MP, Freedman LR. 1982. Natural history of aortic valve endocarditis in rats. Infect Immun 37:127–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Patti JM. 2004. A humanized monoclonal antibody targeting Staphylococcus aureus. Vaccine 22(Suppl 1):S39–S43. doi: 10.1016/j.vaccine.2004.08.015. [DOI] [PubMed] [Google Scholar]
- 47.Lindblad EB. 2000. Freund’s adjuvants, p 49–63. In O’Hagan DT (ed), Vaccine adjuvants: preparation methods and research protocols. Humana Press, Inc, Totowa, NJ. [Google Scholar]
- 48.Vernachio J, Bayer AS, Le T, Chai YL, Prater B, Schneider A, Ames B, Syribeys P, Robbins J, Patti JM. 2003. Anti-clumping factor A immunoglobulin reduces the duration of methicillin-resistant Staphylococcus aureus bacteremia in an experimental model of infective endocarditis. Antimicrob Agents Chemother 47:3400–3406. doi: 10.1128/AAC.47.11.3400-3406.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Deivanayagam CC, Wann ER, Chen W, Carson M, Rajashankar KR, Höök M, Narayana SV. 2002. A novel variant of the immunoglobulin fold in surface adhesins of Staphylococcus aureus: crystal structure of the fibrinogen-binding MSCRAMM, clumping factor A. EMBO J 21:6660–6672. doi: 10.1093/emboj/cdf619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Murphy E, Lin SL, Nunez L, Andrew L, Fink PS, Dilts DA, Hoiseth SK, Jansen KU, Anderson AS. 2011. Challenges for the evaluation of Staphylococcus aureus protein based vaccines: monitoring antigenic diversity. Hum Vaccin 7(Suppl):51–59. doi: 10.4161/hv.7.0.14562. [DOI] [PubMed] [Google Scholar]
- 51.Stegger M, Price LB, Larsen AR, Gillece JD, Waters AE, Skov R, Andersen PS. 2012. Genome sequence of Staphylococcus aureus strain 11819-97, an ST80-IV European community-acquired methicillin-resistant isolate. J Bacteriol 194:1625–1626. doi: 10.1128/JB.06653-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Brady RA, Mocca CP, Burns DL. 2013. Immunogenicity analysis of Staphylococcus aureus clumping factor A genetic variants. Clin Vaccine Immunol 20:1338–1340. doi: 10.1128/CVI.00275-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schulz H, Hennecke H, Thöny-Meyer L. 1998. Prototype of a heme chaperone essential for cytochrome c maturation. Science 281:1197–1200. doi: 10.1126/science.281.5380.1197. [DOI] [PubMed] [Google Scholar]
- 54.Voyich JM, Otto M, Mathema B, Braughton KR, Whitney AR, Welty D, Long RD, Dorward DW, Gardner DJ, Lina G, Kreiswirth BN, DeLeo FR. 2006. Is Panton-Valentine leukocidin the major virulence determinant in community-associated methicillin-resistant Staphylococcus aureus disease? J Infect Dis 194:1761–1770. doi: 10.1086/509506. [DOI] [PubMed] [Google Scholar]
- 55.Duthie ES, Lorenz LL. 1952. Staphylococcal coagulase: mode of action and antigenicity. J Gen Microbiol 6:95–107. doi: 10.1099/00221287-6-1-2-95. [DOI] [PubMed] [Google Scholar]
- 56.National Research Council. 2011. Guide for the care and use of laboratory animals, 8th ed. National Academies Press, Washington, DC. [Google Scholar]
- 57.Hawkins J, Kodali S, Matsuka YV, McNeil LK, Mininni T, Scully IL, Vernachio JH, Severina E, Girgenti D, Jansen KU, Anderson AS, Donald RG. 2012. A recombinant clumping factor A-containing vaccine induces functional antibodies to Staphylococcus aureus that are not observed after natural exposure. Clin Vaccine Immunol 19:1641–1650. doi: 10.1128/CVI.00354-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Portolés M, Kiser KB, Bhasin N, Chan KH, Lee JC. 2001. Staphylococcus aureus Cap5O has UDP-ManNAc dehydrogenase activity and is essential for capsule expression. Infect Immun 69:917–923. doi: 10.1128/IAI.69.2.917-923.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.McLoughlin RM, Solinga RM, Rich J, Zaleski KJ, Cocchiaro JL, Risley A, Tzianabos AO, Lee JC. 2006. CD4+ T cells and CXC chemokines modulate the pathogenesis of Staphylococcus aureus wound infections. Proc Natl Acad Sci U S A 103:10408–10413. doi: 10.1073/pnas.0508961103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lee JC, Betley MJ, Hopkins CA, Perez NE, Pier GB. 1987. Virulence studies, in mice, of transposon-induced mutants of Staphylococcus aureus differing in capsule size. J Infect Dis 156:741–750. doi: 10.1093/infdis/156.5.741. [DOI] [PubMed] [Google Scholar]
- 61.Lee JC, Park JS, Shepherd SE, Carey V, Fattom A. 1997. Protective efficacy of antibodies to the Staphylococcus aureus type 5 capsular polysaccharide in a modified model of endocarditis in rats. Infect Immun 65:4146–4151. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Protective efficacy of active immunization with 2 µg ClfAN23 plus alum or 2 µg Epa plus alum in the murine bacteremia model. Mice were challenged i.p. with 4 × 107 CFU of strain Reynolds (CP8). (A) Quantitative blood cultures were performed 2 h after bacterial challenge. (B and C) Five days later, mouse kidneys were excised and cultured quantitatively (B) and percent weight change in the mice was recorded (C). The data are representative of two independent experiments. *, P < 0.05. Download