Abstract
Hashimoto thyroiditis (HT) is an autoimmune disease, known to be the most common cause of hypothyroidism in nonendemic goitrous areas. It is usually characterized by symmetric, painless, and diffused but sometimes localized swelling of the thyroid gland with features of hypothyroidism. Papillary thyroid carcinoma (PTC), on the other hand, is the most common yet less aggressive form of thyroid cancer, especially in iodine-deficient areas. The coexistence of the two diseases is possible but not common. This case study reports a 50-year-old female with a 10-year history of a huge goiter, which was essentially symptom-free until about 3 months prior to presentation when the patient started complaining of neck pain, dysphagia, productive cough, and cold intolerance. Physical examination revealed focal cystic and tender area in the multinodular swelling and associated cervical lymphadenopathy on the left side of the neck. The serum thyroid stimulating hormone was high, sub-normal T3, and the T4 was low. The fine needle aspiration cytology yielded 10 ml of aspirate of pus admixed with altered blood which on microscopy showed a few suspicious follicular epithelial cells with open nuclei admixed with mainly neutrophil polymorphs, siderophages, and foam cells in a hemorrhagic background. The patient had an incision biopsy that showed areas displaying PTC and HT.
Keywords: Hashimoto thyroiditis, histopathology, Hurthle cell, papillary thyroid carcinoma
INTRODUCTION
The name Hashimoto thyroiditis (HT) was derived from the 1912 report by Hashimoto describing patients with goiter and intense lymphocytic infiltration of the thyroid (struma lymphomatosa).1 HT is known to be the most common cause of hypothyroidism in iodine-deficient areas of the world.2 Papillary carcinomas are also the most common form of thyroid cancer, and majority of them are associated with previous exposure to ionizing radiation. The association of papillary thyroid carcinoma (PTC) and HT has been a subject of debate, since it was first described by Dailey et al. in 1955.3,4,5 Lee et al., Cipolla et al., Liu et al., Pino Rivero et al., and Singh et al. in their various studies concluded that there was an association between HT and PTC.6,7,8,9 We found on a microscopic examination of a thyroid biopsy from 50-year-old female, foci showing HT, PTC, and Hurthle cell hyperplasia; and it is hereby being reported.
PATIENTS AND OBSERVATION
The patient was a 50-year-old woman who initially presented to the Surgery Department of Benue State University Makurdi with a 10-year history of goiter. The swelling increased gradually in size and appeared multinodular in contour, approximating 10 cm × 8 cm [Figure 1] and was all the while painless until about 3 months prior to presentation to the Surgical Outpatient Unit of the Hospital with pain and tenderness, and mild difficulty in swallowing. The pain was described as dull which gradually increased in intensity necessitation her decision to seek medical intervention. She also complained of intolerance to cold and easy fatigability. The serum thyroid-stimulating hormone (TSH) was high −40.5 µIU (normal reference range of 0.4–6.2 µIU), and the T4 was low- < 50 nmol/L while T3 was subnormal. The full blood count revealed mild microcytic hypochromic anemia with normal differential counts. Urea and creatinine were within the normal reference range. The erythrocyte sedimentation rate was mildly elevated–35 mm/h (Westergren method). The fine needle aspiration cytology done yielded about 10 ml of aspirate of pus admixed with altered blood, and the microscopic examination of the smears showed a few suspicious follicular epithelial cells with open nuclei, and pseudocytoplasmic nuclear inclusions in a background containing mainly neutrophil polymorphs, lymphocytes, foam cells, and hemosiderin-laden macrophages. The patient had an incision biopsy which on microscopic examination showed areas with PTC, HT, and Hurthle cell metaplastic changes [Figures 2–4].
Figure 1.

Multinodular goiter in a 50-year-old woman diagnosed with concurrent Hashimoto thyroiditis, papillary thyroid carcinoma, and Hurthle cell adenoma. (a) Anterior view of goiter with focal cystic areas filled with pus. (b) The lateral view of the goiter showing extension of the swelling into the posterior aspect of the neck
Figure 2.

Hashimoto thyroiditis. Residual thyroid follicle being destroyed by a dense lymphocytic infiltrates
Figure 4.

Papillary carcinoma of the thyroid. The microscopic appearance of a papillary carcinoma showing cells with characteristic empty-appearing nuclei called “Orphan Annie eye” nuclei
Figure 3.
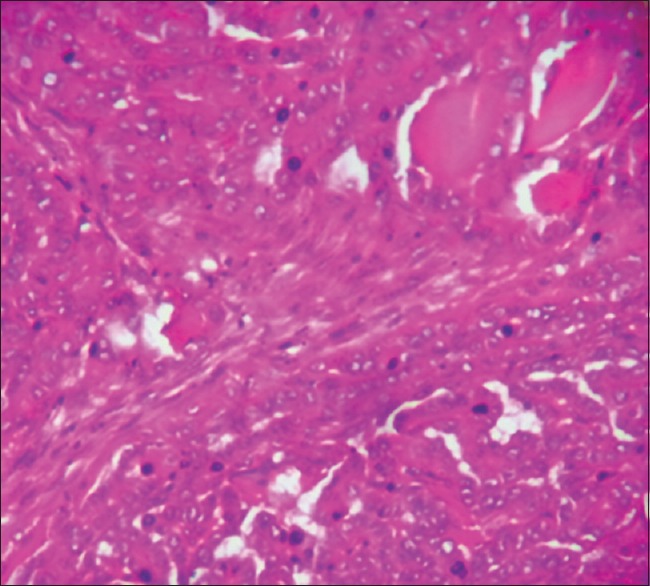
The microscopic appearance of a papillary carcinoma showing well-formed papillae and cells with ground glass nuclei
DISCUSSION
In a research by Repplinger et al. that sought to answer a question, “Is Hashimoto's thyroiditis a risk factor for papillary thyroid cancer?” They discovered that a subgroup of patients with HT had strong associations with PTC.2 They also documented that there was a significantly greater percentage of PTC occurrence in females with HT as compared with females without HT.2 Gasbarri et al. in a study showed that the diagnosis of HT was a representation of a variety of disease mechanisms which influenced the clinical presentation of the disease including PTC.10 It is possible to extrapolate that certain factors influence varying thyroid growth including goiters or carcinomas. This index case certainly substantiates the causative relationship between HT and PTC. Some authors have proposed mechanisms and associations between the two lesions. Expression of the RET/PTC1 and RET/PTC3 oncogenes in HT patients was documented by Wirtschafter et al.11 as a major association, and this hypothesis was supported by Arif et al. who demonstrated that both diseases have a similar immunophenotypic expression, histological features, and the RET/PTC gene rearrangement on molecular studies.12 Moreover, Unger et al. documented p63 expression in a patient with HT, who also had concurrent papillary thyroid cancer.13 This hypothesis was further substantiated by Burstein et al. in their work where they made some propositions that the initiator of the two diseases was pluripotent p63-positive stem cell remnants.14 The intimate relationship between HT and PTC in this report goes further to substantiate the proposals made by these researchers that the two diseases may most probably have similar or common triggering factors. Another angle to this discuss is whether serum thyroid TSH has a causal role in the subsequent development of PTC in preexisting HT. Boelaert et al. documented that serum TSH is an independent predictor of malignancy in thyroid nodules,15 and Haymart et al. further showed that higher serum TSH in thyroid correlated with extrathyroidal extension of disease.16 Jonklaas et al. in their work concluded that higher TSH concentrations, even within the normal range, are associated with a subsequent diagnosis of thyroid cancer in individuals with thyroid abnormalities.17 The hypothesis that TSH stimulates the growth or development of thyroid malignancy during its early or preclinical phase was further supported by their work. Jonklaas et al. also demonstrated that patients with thyroid cancer also have lower T3 levels than patients with benign disease.17 This finding is in keeping with the clinical profile of this index case. It is interesting to conclude by joining Boelaert with the consensus opinion that serum TSH concentration can be used as a diagnostic adjunct in the identification of high-risk patients, who may require further investigation and/or surgical interventions.18
CONCLUSION
A high index of suspicion is required for establishing the coexistence of Hashimoto's thyroiditis with PTC; therefore, clinicians must thoroughly evaluate patients with Hashimoto's thyroiditis while pathologists have to always do a thorough job by painstakingly doing a macroscopic and microscopic examination of thyroid specimens more especially, ones with suspected or confirmed Hashimoto's thyroiditis.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Hashimoto H. Zur Kenntnis der lymphomatösen Veränderung der Schilddrüse (Struma lymphomatosa) Archiv für klinische Chirurgie, Berlin. 1912;97:S219–48. [Google Scholar]
- 2.Repplinger D, Bargren A, Zhang YW, Adler JT, Haymart M, Chen H. Is Hashimoto's thyroiditis a risk factor for papillary thyroid cancer? J Surg Res. 2008;150:49–52. doi: 10.1016/j.jss.2007.09.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dailey ME, Lindsay S, Skahen R. Relation of thyroid neoplasms to Hashimoto disease of the thyroid gland. AMA Arch Surg. 1955;70:291–7. doi: 10.1001/archsurg.1955.01270080137023. [DOI] [PubMed] [Google Scholar]
- 4.Jankovic B, Le KT, Hershman JM. Clinical review: Hashimoto's thyroiditis and papillary thyroid carcinoma: Is there a correlation? J Clin Endocrinol Metab. 2013;98:474–82. doi: 10.1210/jc.2012-2978. [DOI] [PubMed] [Google Scholar]
- 5.Lee JH, Kim Y, Choi JW, Kim YS. High prevalence of suspicious cytology in thyroid nodules associated with positive thyroid autoantibodies. Eur J Endocrinol. 2005;153:637. doi: 10.1530/eje.1.02020. [DOI] [PubMed] [Google Scholar]
- 6.Cipolla C, Sandonato L, Graceffa G, Fricano S, Torcivia A, Vieni S, et al. Hashimoto thyroiditis coexistent with papillary thyroid carcinoma. Am Surg. 2005;71:874–8. [PubMed] [Google Scholar]
- 7.Liu LH, Bakhos R, Wojcik EM. Concomitant papillary thyroid carcinoma and Hashimoto's thyroiditis. Semin Diagn Pathol. 2001;18:99–103. [PubMed] [Google Scholar]
- 8.Pino Rivero V, Guerra Camacho M, Marcos García M, Trinidad Ruiz G, Pardo Romero G, González Palomino A, et al. The incidence of thyroid carcinoma in Hashimoto's thyroiditis. Our experience and literature review. An Otorrinolaringol Ibero Am. 2004;31:223–30. [PubMed] [Google Scholar]
- 9.Singh B, Shaha AR, Trivedi H, Carew JF, Poluri A, Shah JP. Coexistent Hashimoto's thyroiditis with papillary thyroid carcinoma: Impact on presentation, management, and outcome. Surgery. 1999;126:1070–6. doi: 10.1067/msy.2099.101431. [DOI] [PubMed] [Google Scholar]
- 10.Gasbarri A, Sciacchitano S, Marasco A, Papotti M, Di Napoli A, Marzullo A, et al. Detection and molecular characterisation of thyroid cancer precursor lesions in a specific subset of Hashimoto's thyroiditis. Br J Cancer. 2004;91:1096–104. doi: 10.1038/sj.bjc.6602097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wirtschafter A, Schmidt R, Rosen D, Kundu N, Santoro M, Fusco A, et al. Expression of the RET/PTC fusion gene as a marker for papillary carcinoma in Hashimoto's thyroiditis. Laryngoscope. 1997;107:95–100. doi: 10.1097/00005537-199701000-00019. [DOI] [PubMed] [Google Scholar]
- 12.Arif S, Blanes A, Diaz-Cano SJ. Hashimoto's thyroiditis shares features with early papillary thyroid carcinoma. Histopathology. 2002;41:357–62. doi: 10.1046/j.1365-2559.2002.01467.x. [DOI] [PubMed] [Google Scholar]
- 13.Unger P, Ewart M, Wang BY, Gan L, Kohtz DS, Burstein DE. Expression of p63 in papillary thyroid carcinoma and in Hashimoto's thyroiditis: A pathobiologic link? Hum Pathol. 2003;34:764–9. doi: 10.1016/s0046-8177(03)00239-9. [DOI] [PubMed] [Google Scholar]
- 14.Burstein DE, Nagi C, Wang BY, Unger P. Immunohistochemical detection of p53 homolog p63 in solid cell nests, papillary thyroid carcinoma, and hashimoto's thyroiditis: A stem cell hypothesis of papillary carcinoma oncogenesis. Hum Pathol. 2004;35:465–73. doi: 10.1016/j.humpath.2003.10.027. [DOI] [PubMed] [Google Scholar]
- 15.Boelaert K, Horacek J, Holder RL, Watkinson JC, Sheppard MC, Franklyn JA. Serum thyrotropin concentration as a novel predictor of malignancy in thyroid nodules investigated by fine-needle aspiration. J Clin Endocrinol Metab. 2006;91:4295–301. doi: 10.1210/jc.2006-0527. [DOI] [PubMed] [Google Scholar]
- 16.Haymart MR, Glinberg SL, Liu J, Sippel RS, Jaume JC, Chen H. Higher serum TSH in thyroid cancer patients occurs independent of age and correlates with extrathyroidal extension. Clin Endocrinol (Oxf) 2009;71:434–9. doi: 10.1111/j.1365-2265.2008.03489.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jonklaas J, Nsouli-Maktabi H, Soldin SJ. Endogenous thyrotropin and triiodothyronine concentrations in individuals with thyroid cancer. Thyroid. 2008;18:943–52. doi: 10.1089/thy.2008.0061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Boelaert K. The association between serum TSH concentration and thyroid cancer. Endocr Relat Cancer. 2009;16:1065–72. doi: 10.1677/ERC-09-0150. [DOI] [PubMed] [Google Scholar]


