Abstract
Directed neuroblast and neuronal migration is important in the proper development of nervous systems. In C. elegans the bilateral Q neuroblasts QR (on the right) and QL (on the left) undergo an identical pattern of cell division and differentiation but migrate in opposite directions (QR and descendants anteriorly and QL and descendants posteriorly). EGL-20/Wnt, via canonical Wnt signaling, drives the expression of MAB-5/Hox in QL but not QR. MAB-5 acts as a determinant of posterior migration, and mab-5 and egl-20 mutants display anterior QL descendant migrations. Here we analyze the behaviors of QR and QL descendants as they begin their anterior and posterior migrations, and the effects of EGL-20 and MAB-5 on these behaviors. The anterior and posterior daughters of QR (QR.a/p) after the first division immediately polarize and begin anterior migration, whereas QL.a/p remain rounded and non-migratory. After ~1 hour, QL.a migrates posteriorly over QL.p. We find that in egl-20/Wnt, bar-1/β-catenin, and mab-5/Hox mutants, QL.a/p polarize and migrate anteriorly, indicating that these molecules normally inhibit anterior migration of QL.a/p. In egl-20/Wnt mutants, QL.a/p immediately polarize and begin migration, whereas in bar-1/β-catenin and mab-5/Hox, the cells transiently retain a rounded, non-migratory morphology before anterior migration. Thus, EGL-20/Wnt mediates an acute inhibition of anterior migration independently of BAR-1/β-catenin and MAB-5/Hox, and a later, possible transcriptional response mediated by BAR-1/β-catenin and MAB-5/Hox. In addition to inhibiting anterior migration, MAB-5/Hox also cell-autonomously promotes posterior migration of QL.a (and QR.a in a mab-5 gain-of-function).
Introduction
The directed migration of neurons and neuroblasts is important in nervous system development to establish proper connectivity and circuits. Wnt signaling has been broadly implicated in mammalian cortical and hippocampal neurogenesis [1–3]. The Q neuroblasts in C. elegans have been a useful system in which to dissect the molecular mechanisms of directed neuroblast and neuronal migration [4]. The Q cells are bilateral neuroblasts in the posterior region of C. elegans. The left and right Q cells produce three neurons through an identical pattern of division and programmed cell death [4–6]. Despite these similarities, QR and descendants migrate to the anterior, and QL and descendants migrate to the posterior. The Q neuroblasts are born in embryogenesis as the sister cells of the V5 hypodermal seam cell and reside between the V4 and V5 seam cells in the posterior-lateral region [5, 6]. At hatching in the L1 larva, the Q neuroblasts undergo an initial protrusion and migration such that by 4h post hatching in the L1, QL has migrated posteriorly atop the V5 seam cell, and QR has migrated anteriorly atop the V4 seam cell [4, 7, 8]. After migrating, the Q neuroblasts undergo their first cell division producing anterior and posterior daughters (QR.a/p and QL.a/p). A posterior EGL-20/Wnt signal induces the expression of the MAB-5 Hox transcription factor in QL.a/p but not QR.a/p via canonical Wnt signaling and BAR-1/β-catenin [9–17]. MAB-5 is required for the further posterior migration of QL descendants. After this initial directional decision, Wnts redundantly control both QL and QR descendant migrations [18].
The QR descendant AQR and QL descendant PQR migrate the longest distances to the anterior and posterior of any of the three Q descendant neurons, with AQR residing near the anterior deirid and posterior pharyngeal bulb and PQR residing among the phasmid ganglia posterior to the anus [7]. In egl-20 and mab-5 loss-of-function mutants, the initial anterior and posterior migrations of QR and QL are normal [7], but QL descendants fail to migrate posteriorly and instead migrate anteriorly [19]. In a mab-5 gain-of-function mutant, both QR and QL descendants migrate posteriorly despite normal initial migration of QR to the anterior and QL to the posterior [7, 15, 19]. Thus, MAB-5 is a determinant of posterior migration and can act in both QL and QR descendants, but normally only acts in QL due to EGL-20/Wnt-induced expression of MAB-5 in QL but not QR. The patterns of cell division and cell death, and the gross differentiation of the three neurons produced from each cell are unaffected by mab-5 mutation, suggesting that mab-5 is involved in terminal differentiation to specify direction of migration, and not cell fate or differentiation generally.
While it is clear that EGL-20/Wnt and MAB-5 promote posterior migration, the mechanism by which these molecules alter QL.a/p behavior relative to QR.a/p during posterior versus anterior migration remains unclear. In this study, we analyzed the behavior of the QR.a/p and QL.a/p cells as they migrate in wild-type, egl-20, and mab-5 mutants. We found that in wild-type, QR.a/p began anterior migration shortly after division, whereas QL.a/p retained a rounded, non-migratory morphology. Approximately one hour after QR.a/p migration, QL.a began to migrate posteriorly over QL.p. In mab-5 and egl-20 loss-of-function, QL.a/p polarized and migrated anteriorly, similar to QR.a/p in wild-type, suggesting that both normally inhibit anterior QL.a/p migration. However, in mab-5, QL.a/p transiently retained a rounded, non-migratory morphology, whereas in egl-20, QL.a/p began migration shortly after division, similar to QR.a/p in wild-type. bar-1/β-catenin mutants resembled mab-5 and displayed the transient non-migratory morphology not seen in egl-20. These results suggest that EGL-20 has both an acute MAB-5-independent role and a later MAB-5-dependent role in inhibiting anterior QL.a/p migration, and that the acute role of egl-20 does not involve BAR-1/β-catenin. The mab-5 gain-of-function mutant caused QR.a/p posterior migration similar to QL.a/p, including posterior migration of QR.a. Thus, in addition to inhibiting anterior migration, MAB-5 also promoted posterior migration.
Results
Wnt Signaling Redundantly Controls AQR and PQR Migration
A genome-wide screen for new mutations affecting AQR and PQR migration identified two new mutations, lq42 and lq74. Both caused anterior PQR migration, and minor or no defects in AQR migration (Fig 1). Genes affected by the mutations were mapped by whole genome single nucleotide polymorphism resequencing using the Cloudmap protocol [20] (data not shown). Both mapped to a region in the center of LG IV, and each harbored a mutation in the egl-20 gene, which encodes a C. elegans Wnt molecule. egl-20 has been shown previously to affect PQR but not AQR migration [7], consistent with the phenotypes of lq42 and lq74. lq42 introduced a premature stop codon at arginine 296 (C to T), and lq74 was a missense mutation changing cysteine 248 to tyrosine (G to A) (see Methods). Both failed to complement egl-20(n585) for PQR migration (data not shown). The egl-20(gk453010) allele was generated by the Million Mutation Project (present in strain VC40076) [21]. gk453010 is a G to A transition resulting in an arginine 245 to opal stop codon mutation. The egl-20(hu105) allele also introduces a premature stop codon [22, 23]. lq42, lq74, hu105, and gk453010 each displayed nearly completely penetrant anterior PQR migration (Fig 1), suggesting that they are all putative null mutations, with the exception of the lq74 missense mutation which may retain some function (1% PQR did not migrate completely to head).
Fig 1. AQR and PQR migration.
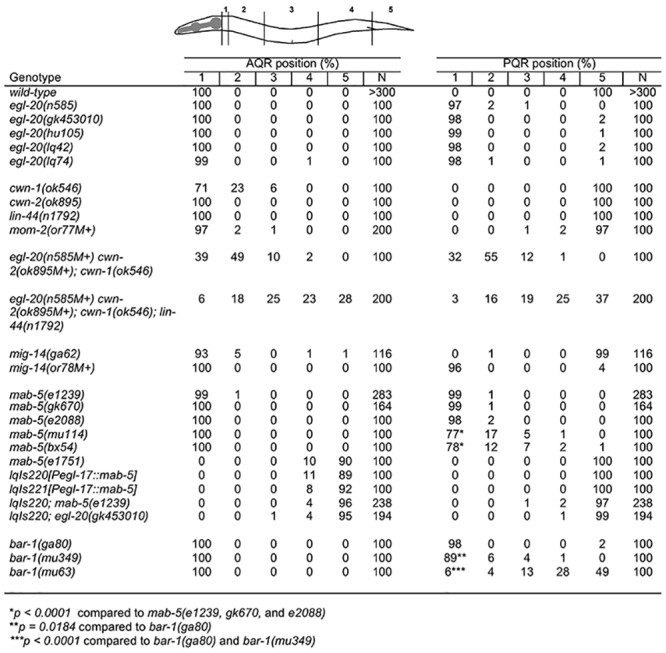
The percentages of AQR and PQR in mutants is shown, with respect to postions 1–5 as described in Methods and shown on the diagram. Statistical significance was calculated using Fisher’s Exact test.
AQR and PQR migration was analyzed in mutants of the four other C. elegans Wnt genes cwn-1, cwn-2, lin-44, and mom-2 (Fig 1). Putative null alleles of cwn-2 and lin-44 displayed no defects. A null cwn-1 allele displayed 29% failure of AQR to complete anterior migration. mom-2, which has not been previously reported to affect Q descendant migrations, displayed 3% AQR and PQR migration defects in the partial loss of function or77M+ allele (M+ is used to designate homozygous animals from a heterozygous mother and therefore with a wild-type maternal contribution of gene function). Thus, egl-20, cwn-1, and, mom-2 each affect AQR and PQR migration in distinct ways.
Redundancy among Wnts in Q descendant migrations has been reported previously [18]. The Wnt triple mutant egl-20(n585) cwn-2(ok895); cwn-1(ok546) displayed highly penetrant PQR anterior migration and a failure of both AQR and PQR migration along the anterior route (Fig 1). No AQR or PQR migrated posteriorly in this genotype. Mutation of lin-44 in the egl-20(n585M+) cwn-2(ok895M+); cwn-1(ok546); lin-44(n1792) quadruple mutant resulted in posterior migration of both AQR and PQR (28% and 37%) (Fig 1). This indicates that lin-44 activity inhibits posterior AQR and PQR migration in the absence of egl-20, cwn-1, and cwn-2, and suggests that lin-44 can act as a posterior repellant. Previous studies indicated that lin-44 caused posterior displacement of QL descendants PVM and SDQL [18], and HSN [24], in certain multiple Wnt mutant combinations, consistent with a posterior repellant activity of lin-44. The triple and quadruple Wnt mutants were scored with maternal contribution of both egl-20 and cwn-2, so we cannot be certain that maternal Wnt function remains in these mutants. MIG-14/Wntless is required for Wnt processing and function and affects QL descendant migrations [25]. Two mig-14 mutants had less severe defects than the Wnt triple and quadruple mutants (Fig 1). The likely hypomorph ga62 had weak effects on both AQR and PQR, and the lethal in-frame deletion mutation or78, with wild-type maternal contribution, resembled egl-20 and only affected PQR migration. These data suggest either that these mutations did not completely eliminate mig-14 function, or that mig-14 is not involved in all Wnt-related signaling events.
Wnt Signaling Might Not Affect Early Q Neuroblast Migration
The above results, along with previous studies [18, 26], indicate that the five C. elegans Wnt genes and four Frizzled genes mig-1, lin-17, cfz-2, and mom-5 act redundantly to control Q descendant migration. However, egl-20 alone and the egl-20(n585) cwn-2; cwn-1 triple mutant displayed no defects in early Q anterior-posterior protrusion and migration, despite severe AQR and PQR descendant migration defects [7, 26] (Fig 1). To further investigate the role of Wnts in early Q protrusion and migration, we analyzed early QL and QR protrusion and migration with the ayIs9[Pegl-17::gfp] transgene, expressed in the early Q cells [27–29]. Two additional mutant combinations were tested: the Wnt triple mutant egl-20(lq42M+); mom-2(or77M+); lin-44(n1792) and the Frizzled double mutant mom-5(ne12M+) lin-17(n761M+). Neither displayed defects in direction of initial QL and QR migration (QL divided atop V5 and QR atop V4 (n ≥ 10)) (data not shown). We have not scored early Q migrations in the absence of all Wnt signaling due to maternal perdurance and/or incomplete knockdown of all Wnt genes at once. However, we have not observed early Q directional migration defects in any Wnt signaling mutant combination tested, despite strong AQR and PQR migration defects. This suggests Wnt signaling might not be involved in early Q anterior-posterior protrusion and migration, with function restricted to later migrations of the Q descendants.
QR Daughters Begin Migration Immediately after QR Division
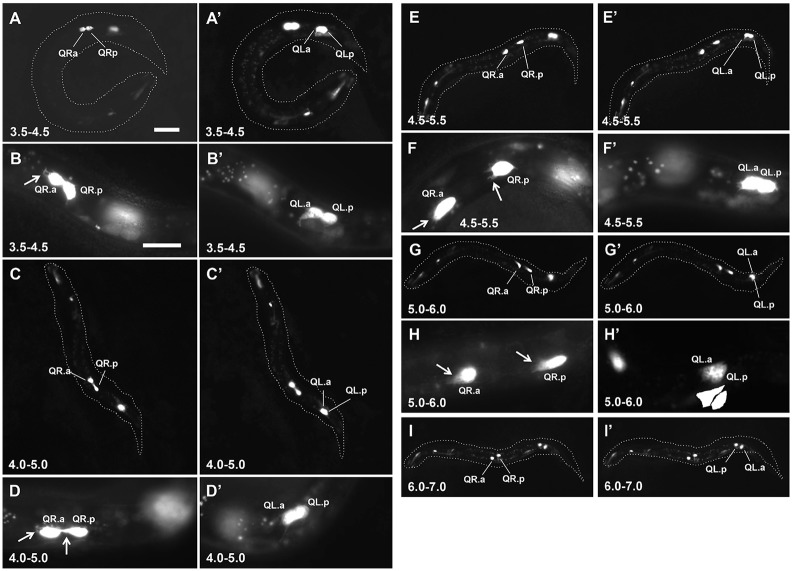
In order to understand how EGL-20/Wnt signaling drives posterior QL descendant migration, the behaviors of QR.a/p and QL.a/p just after division were analyzed. We monitored the position of the Q cell daughters using a Pegl-17::gfp transgene ayIs9 expressed in the Q cells (Fig 2). Newly-hatched L1 larvae were synchronized within the same hour-long age range at half-hour timepoints after hatching (e.g. 3.5–4.5h, 4.0–5.0h, 4.5–5.5h, 5.0–6.0h, 5.5–6.5h, 6.0–7.0h) (see Methods for synchronization of L1 larvae). The positions of QR, QL, and their daughters were determined in 20 animals at each timepoint. At 3.5–4.5h, 19/20 of the QR examined had divided, whereas only 13/20 of the QL had divided. This trend held true in all genotypes analyzed, suggesting that QL divides slightly later than QR. Indeed, at 4.0–5.0h, 4/20 QL had not yet divided, but 20/20 of the QRs had divided.
Fig 2. Q descendant migrations in wild-type.
Fluorescent micrographs of ayIs9[Pegl-17::gfp] expression in Q descendants are shown in the same animals (QR, A-I; QL, A’-I’). Anterior is to the left. The dashed lines indicate the body of the whole animal in the micrograph. Arrows indicate anterior protrusions characteristic of the QR migratory morphology. The scale bar in A represents 5μm for A, C, E, G, and I; and the scale bar in B represents 5μm for B, D, F, and H.
After division, the QR and QL daughters had a rounded morphology with little or no protrusion (Figs 2A, 2B and 3). Shortly after division in the 4.0–5.0h timepoint, the QR daughters polarized and begun migrating to the anterior (Fig 2C and 2D). Migrating QR.a/p cells first elongated in the anterior-posterior axis and then extended F-actin-rich lamellipodia-like protrusions in the direction of migration (to the anterior), similar to what has been previously reported (Fig 3A and S1 Movie) [30–32]. The QR.a/p cells maintained this polarized, migratory morphology as they migrated anteriorly until their second round of cell division at approximately 7h post-hatching (Figs 2D–2I and 3A). At 6.0–7.0, QR.a/p cells began to lose the polarized morphology and assumed a rounded morphology in preparation for the second round of cell division (Figs 2I and 3A). Time-lapse imaging with the casIs330 transgene (S1 Movie, S2 Movie, and Fig 3) [30] resulted in an approximately 2-fold delay in migration and division relative to the ayIs9 transgene.
Fig 3. Time-lapse imaging of QR and QL descendant migrations.
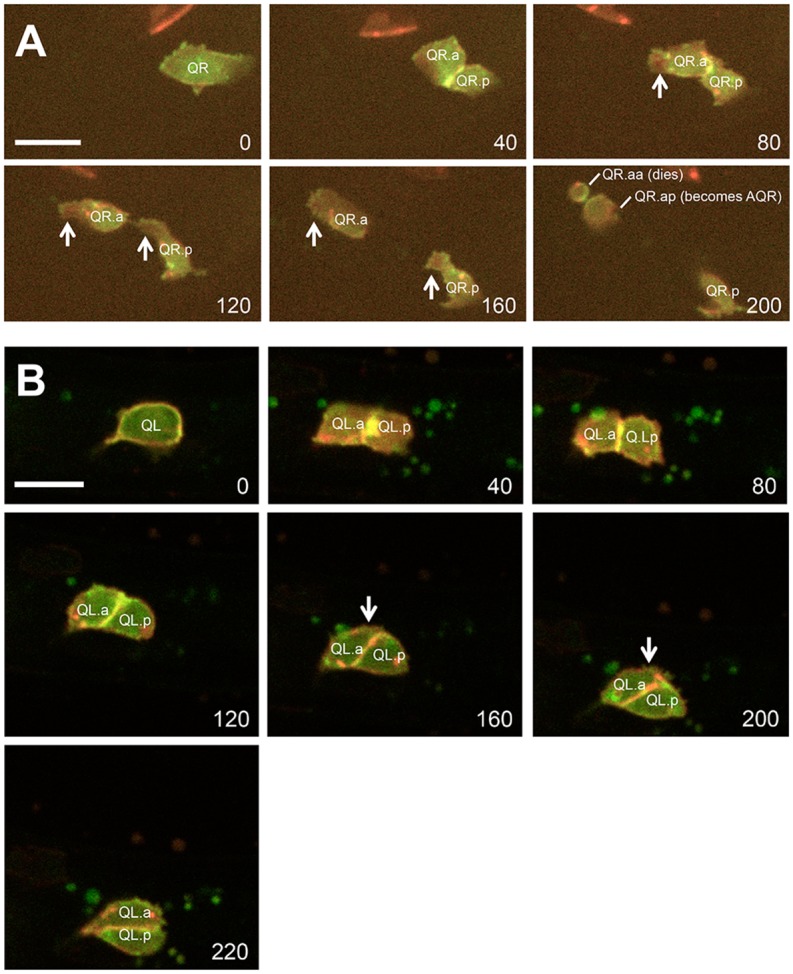
Micrographs from time lapse imaging of QR (A) and QL (B) are shown, using the casIs330 transgene [31] (see Methods). Individual images were taken from S1 and S2 Movies at the timepoints in minutes indicated in the lower right of each micrograph. Red indicates MYR::mCherry at membranes and in chromatin (HIS-24::mCherry), and green represents F-actin (MOEabd::GFP). (A) QR and descendants are indicated. Arrows point to the F-actin-rich lamellipodial protrusions that accompany polarization and anterior migration. F-actin also accumulated at the furrows between dividing cells (e.g. between QR.a and QR.p at 40min, and between QR.aa and QR.ap at 200min). (B) QL and descendant migration. Arrows point to the extensions from QL.a posteriorly over QL.p. In these time-lapse imaging experiments with the casIs330 transgene, migrations and cell divisions were delayed by ~2-fold compared to ayIs9 still images, but the same pattern of QL.a/p and QR.a/p migration as observed with ayIs9 still images was conserved.
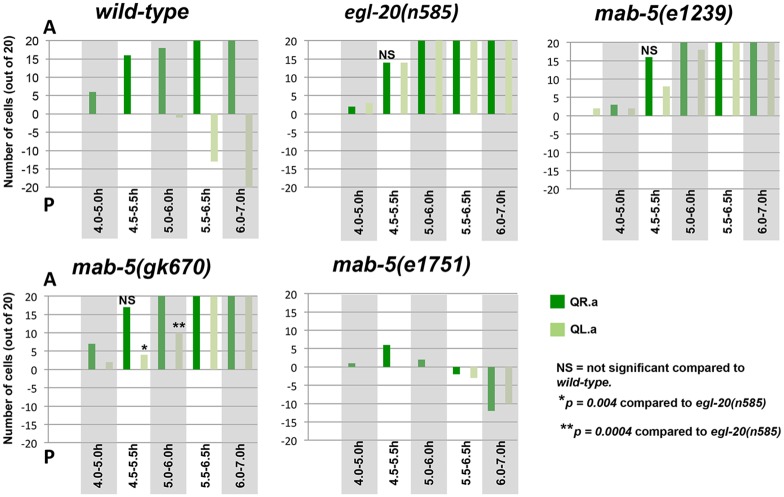
To account for variability in the timing of migration, QR.a/p migration was quantified in 20 animals at each time point using ayIs9 (Fig 4). Cells were scored as migratory if they extended protrusions to the anterior, often accompanied by migration from their birthplace and also separation from one another (Figs 2E, 2F and 3A). At 4.0–5.0h, 6/20 of the QR daughters had polarized and started migrating to the anterior (Fig 4). At 4.5–5.5h, 16/20 were migrating, and at 5.0–6.0, 18/20. By 5.5–6.5h, all (20/20) of the QR.a/p were polarized and migrating to the anterior (Fig 4).
Fig 4. Quantitative representation of Q descendant migrations.
Graphs represent Q migrations in indicated genotypes. The number (out of 20) of QR.a (dark green) and QL.a (light green) that had migrated (Y axis) at each timepoint (X axis) are indicated. Positive numbers on the Y axis represent anterior migration, and negative numbers represent posterior migration. A cell was scored a migratory if it extended an anterior lamellipodial like protrusion and separated from its sister (for anterior migration) or if it extended a posterior protrusion over its sister or migrated behind its sister (for posterior migration).
QL Daughters Remain Rounded and Non-Migratory
In contrast to QR.a/p, the QL.a/p daughters did not polarize and migrate anteriorly. From 3.5–5.5h, at a time when QR.a/p had polarized and begun migration, the QL.a/p cells retained a rounded, non-migratory morphology and did not migrate (Figs 2A’–2F’ and 3B). Occasionally, protrusions were observed on non-migratory QL.a/p (e.g. Fig 2F’), but they did not assume the elongated morphology and robust anterior protrusion observed in QR.a/p. At 5.0–6.0h, the anterior QL.a daughters began migrating posteriorly over the anterior QL.p daughters, which retained a rounded morphology (Figs 2G’–2H’ and 3B) (S1 Movie). QL.p showed no significant migration over this time period, consistent with previous observations [33]. Often, QL.a apparently migrated over the top of QL.p (Figs 2H’ and 3B). QL.a was scored as migrating posteriorly if it was over QL.p, or if it was indistinguishable from QL.p suggesting it was migrating in front of or behind QL.p (data not shown).
Quantification of migration in 20 animals showed that only 1/20 QL.a began posterior migration at the 5.0–6.0h timepoint (Fig 4). At 5.5–6.5h, 13/20 of the anterior QL.a cells had migrated. At 6.0–7.0h, all QL.a had migrated posterior to QL.p and began to assume a rounded morphology preceding the second round of cell division (Fig 2I’). Posterior QL.p showed little or no polarization or migration in the entire time before the second round of cell division (7.0–8.0h after hatching).
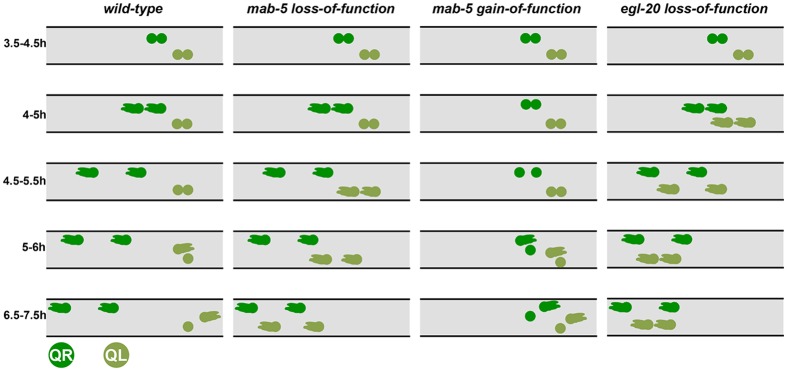
In sum, QR.a/p polarized and began to migrate anteriorly shortly after division. The division of QL was delayed compared to QR, and the QL.a/p daughters remained rounded and non-migratory in the 3.5–5.0h time window while the QR.a/p daughters migrated to the anterior. At 5.0–6.0h, QL.a began migrating posteriorly over QL.p and continued posterior migration. QL.p did not migrate in this time period. A schematic summary of QR.a/p and QL.a/p migratory behavior is shown in Fig 5.
Fig 5. Schematic summary QR.a/p and QL.a/p migration behavior.
QR.a/p are dark green and QL.a/p are light green. Genotypes and time points using ayIs9[Pegl-17::gfp] are indicated. QL.a/p begin anterior migration soon after division in the 4.0–5.0h timepoint, whereas QL.a/p in mab-5 delay anterior migration until the 4.5–5.5h timepoint. Anterior is to the left.
EGL-20/Wnt and MAB-5/Hox Inhibit QL.a/p Anterior Polarization and Migration
egl-20 and mab-5 loss-of-function mutations caused anterior migration of PQR and did not affect AQR migration (Fig 1). mab-5(e1239, gk670, and e2088) all showed nearly completely penetrant PQR anterior migration, suggesting that they are all strong loss-of-function alleles. mab-5(e2088) was a complex rearrangement affecting the second exon of mab-5 (see Methods). mab-5(bx54), a missense mutation in the homeodomain of mab-5 [34], and mab-5(mu114), a premature stop codon early in the first exon (see Methods), displayed weaker defects, indicating that they are hypomorphic alleles.
As expected, direction and extent of initial QR and QL migration prior to the first Q cell division was unaffected by egl-20 and mab-5 (Figs 4, 6A and 7A). Furthermore, the QR.a/p anterior and posterior daughters polarized and migrated to the anterior in all cases (Figs 4, 6 and 7). Previous work showed that in egl-20 mutants, the posterior QR.p daughter displayed variable posterior polarization during migration [35]. We did not follow this phenotype in these studies.
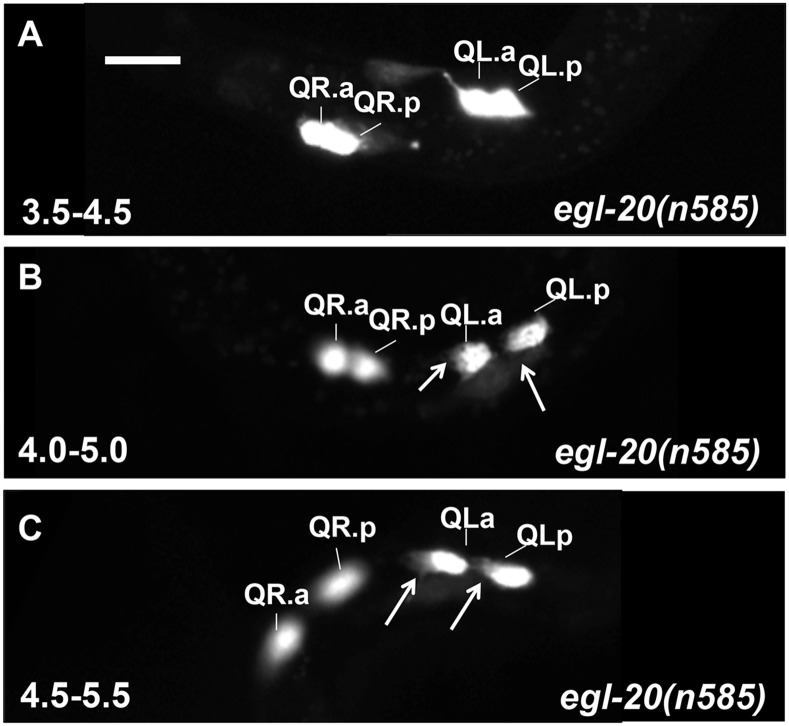
Fig 6. Q descendant migrations in egl-20(n585) mutants.
Fluorescent micrographs of ayIs9[Pegl-17::gfp] expression in Q descendants are shown. Anterior is to the left. Arrows indicate anterior protrusions characteristic of migratory morphology. QL.a has begun anterior migration before QR.a in this animal at the 4.0–5.0h timepoint. The scale bar in A represents 5μm.
Fig 7. Q descendant migrations in mab-5 loss-of-function.
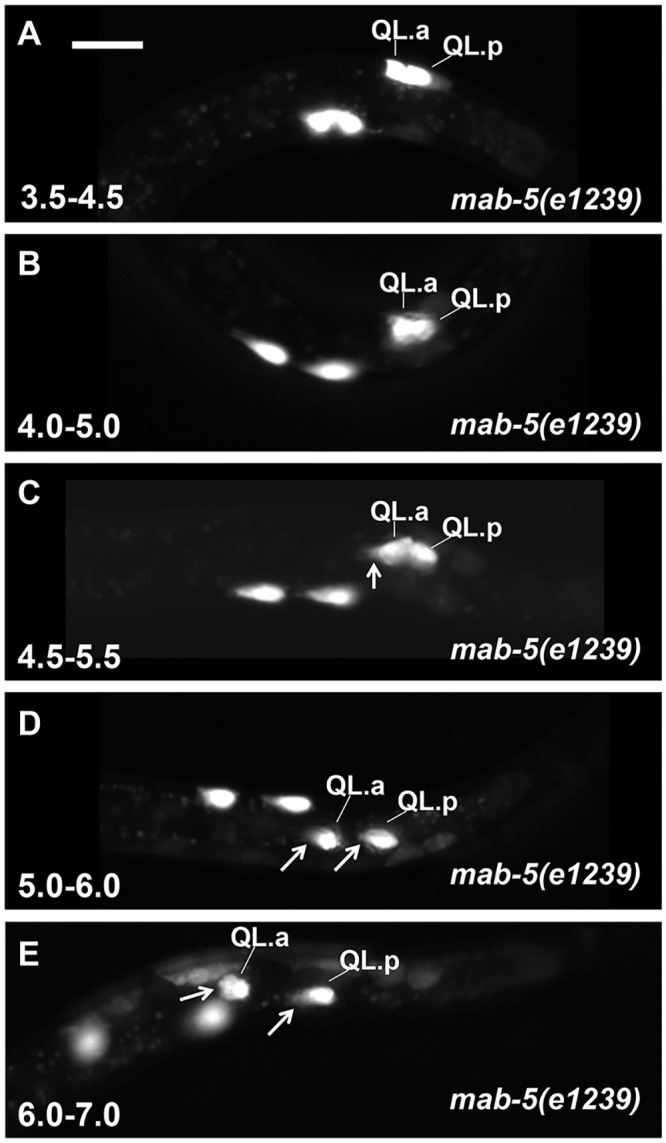
Fluorescent micrographs of ayIs9[Pegl-17::gfp] expression in the Q cells of mab-5(e1239) loss-of-function mutants are shown. At 4.0–5.0h and 4.5–5.5h, QL.a/p have not migrated, but QL.a is beginning to extend an anterior protrusion (arrow in C). Beginning at the 5.5–6.5h timepoint, both QL.a and QR.a begin posterior migration. Anterior is to the left, and the scale bar in A represents 5μm.
In egl-20(n585), QL.a/p polarized and migrated anteriorly shortly after division at the 4.0–5.0h timepoint, similar to QR.a/p (Fig 4). In some cases, QL.a/p polarized and began to migrate before QR.a/p (Fig 6B). Upon quantification, 3/20 QL.a/p at the 4.0–5.0 timepoint and 14/20 at the 4.5–5.5h timepoint had polarized and begun anterior migration, similar to QR.a/p (Fig 4). QL.a/p resembled QR.a/p in their anterior migrations through the remaining timepoints (Fig 6C). Thus, in the absence of EGL-20/Wnt, QL.a/p polarized and migrated anteriorly, indicating that EGL-20 inhibits polarization and anterior migration of QL.a/p.
In mab-5 mutants, QL.a/p also polarized and migrated to the anterior similar to egl-20 (Fig 7). Upon quantification of two mab-5 mutants, 2/20 QL.a/p polarized and began migrating to the anterior at the 4.0–5.0h timepoint in both mutants, and 7/20 and 4/20 at the 4.5–5.5h timepoint (Fig 4). By 5.5–6.5h, all QL.a/p had migrated anteriorly in both mab-5 mutants (Fig 4). These results suggest that MAB-5/Hox also inhibits QL.a/p anterior polarization and migration.
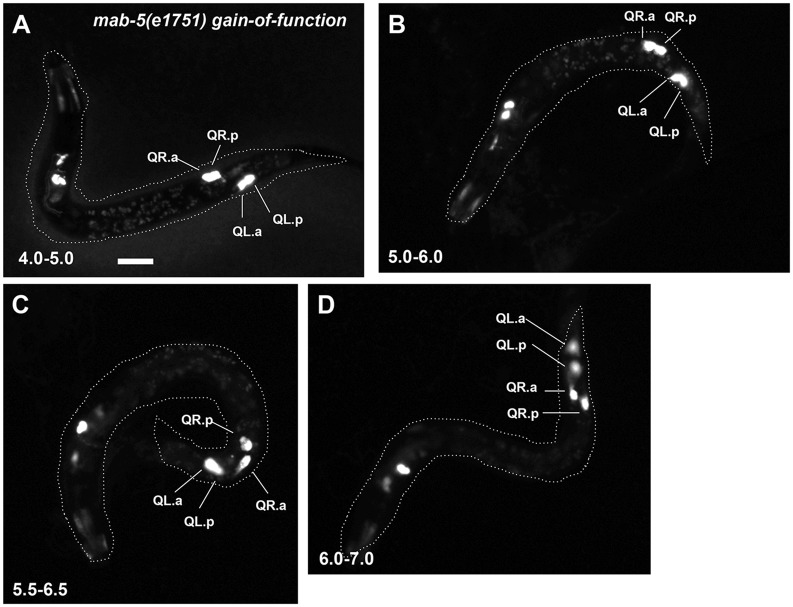
mab-5 Gain-Of-Function Causes Posterior QR.a/p Migration
mab-5(e1751) is a gain-of-function variant that results in ectopic expression of mab-5 in cells, including in QRs which normally do not express it [36]. mab-5(e1751) mutants display posterior migration of both QL and QR descendant neurons AQR and PQR, the opposite of mab-5 loss-of-function [19] (Fig 1). We analyzed Q descendant behaviors in mab-5(e1751). Initial Q migrations were normal, as QR migrated anteriorly and QL posteriorly, indicating that mab-5(e1751) does not affect initial Q migration (Fig 8A). QL.a/p behavior generally resembled what is seen in wild type (persistent rounded and non-migratory morphology until 5.0–6.0h, with QL.a beginning to migrate posteriorly over QL.p at this time) (Fig 8).
Fig 8. Q descendant migrations in mab-5(e1751) gain-of-function.
Fluorescent micrographs of ayIs9[Pegl-17::gfp] expression in Q descendants are shown. Anterior is to the left. The dashed lines indicate the body of the whole animal in the micrograph. Both QL.a/p and QR.a/p retain a rounded, non-migratory morphology until 5.5–6.5h, when QL.a and QR.a begin posterior migration. At 6.0–7.0h, both anterior daughters have migrated posteriorly to the posterior daughters. The scale bar in A represents 5μm.
Strikingly, QR.a/p behaved similarly to QL.a/p in mab-5(e1751) (Fig 8). Some QR.a/p slightly separated from one another, but did not show the polarized and anterior migratory behavior seen in wild-type QR.a/p from 4h onward. Eventually, QR.a migrated posteriorly over QR.p just as QL.a did, beginning at the 5.5–6.5h timepoint (Figs 4 and 8). mab-5(e1751) resulted in a delay in QR.a/p and QL.a/p posterior migration relative to wild-type QL.a/p (Fig 4). At the 6.0–7.0h timepoint, all of QL.a had migrated posteriorly in wild-type where only 12/20 QR.a and 10/20 QL.a had migrated posteriorly in mab-5(e1751). Together with mab-5 loss-of-function, these data suggest that MAB-5 normally inhibits anterior migration of QL.a/p and can also inhibit QR.a/p anterior migration in the gain-of-function mutant (Fig 5). In addition to inhibiting anterior migration, mab-5 activity can also induce posterior migration of the QR.a cell, similar to wild-type QL.a.
mab-5 is expressed in QL as well as in other cells surrounding QL in the posterior of the animal. We constructed a transgene driving mab-5 expression from the egl-17 promoter expressed in the early Q cells. Q descendant neurons AQR and PQR both migrated posteriorly in animals harboring this transgene, similar to mab-5(e1751) (Fig 1). Initial QL and QR migration were normal in Pegl-17::mab-5 animals, but both QL.a/p and QR.a/p resembled mab-5(e1751) (they remained rounded and non-migratory) (Fig 9). These data indicate that MAB-5 expression in the Q cells themselves is sufficient to prevent anterior migration of QL.a/p and QR.a/p and drive their posterior migration, consistent with previous results showing autonomy of mab-5 function in neuronal descendant migrations [15].
Fig 9. Q descendant migration in animals with transgenic mab-5 expression in QL and QR.
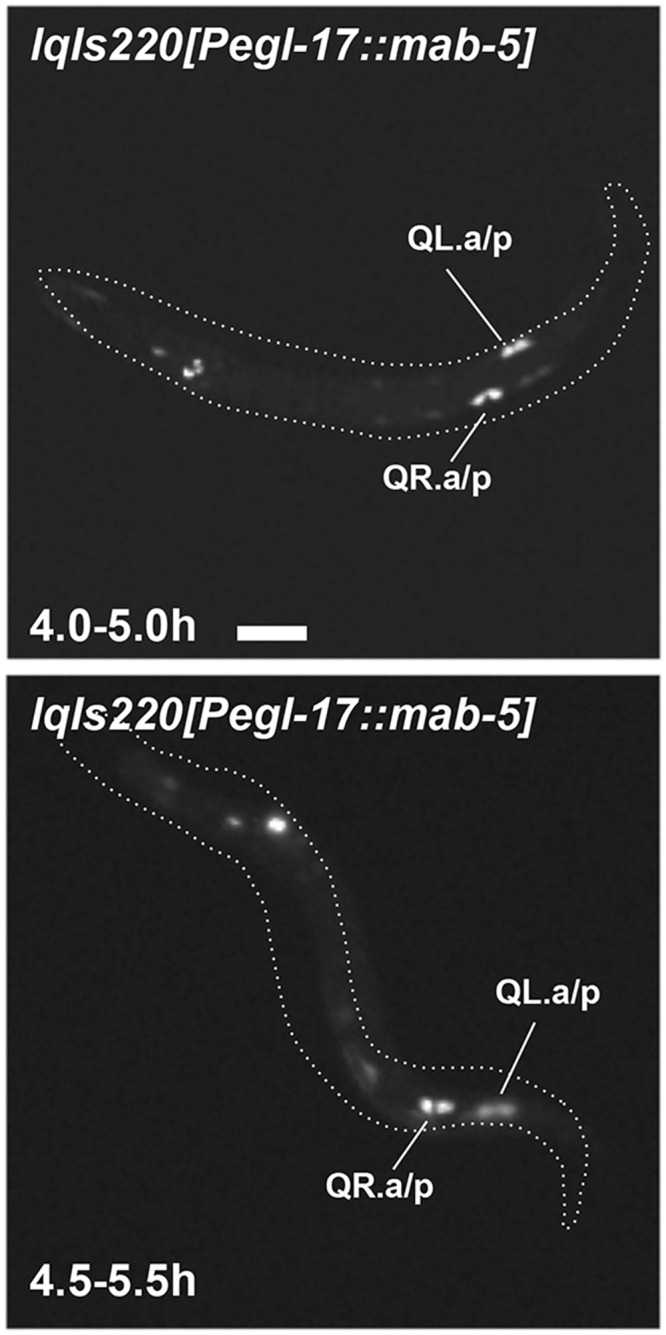
Fluorescent micrographs of Pegl-17::gfp expression in Q descendants are shown. Anterior is to the left. The dashed lines indicate the body of the whole animal in the micrograph. Both QR.a/p and QL.a/p are non migratory at the 4.5–5.5h timepoint. The scale bar represents 5μm for both micrographs.
In egl-20(n585); mab-5(e1751) and egl-20(n585); Pegl-17::mab-5 animals, AQR and PQR both migrated posteriorly similar to mab-5 gain-of-function alone, consistent with mab-5 acting downstream of egl-20. At the 4.5–5.5h timepoint, QL.a/p and QR.a/p resembled mab-5(e1751) alone and had not begun migration to the anterior as in egl-20(n585) (data not shown). However, both QL.a/p and QR.a/p showed the transient separation before posterior migration as observed for QR.a/p in mab-5(e1751) (2/21 QL.a/p and 4/21 QR.a/p for egl-20(n585); mab-5(e1751), and 1/22 QL.a/p and 7/22 QR.a/p for egl-20(n585); Pegl-17::mab-5). In sum, these results indicate that the effects of mab-5 gain-of-function are independent of egl-20, and are consistent with MAB-5 acting downstream of EGL-20.
QL.a/p Anterior Migration Is Delayed in mab-5 Mutants Compared to egl-20
Loss of function of egl-20 or mab-5 resulted in anterior migration of QL.a/p. While similar to egl-20, in mab-5 mutants we noted a delay in QL.a/p polarization relative to QR.a/p not seen in egl-20 (Fig 4). QL.a/p often remained unpolarized while QR.a/p had begun migration (Fig 7B). In mab-5(e1239) at 4.5–5.5h, 7/20 QL.a/p had begun migration while 16/20 QR.a/p had (Fig 4). The difference was more extreme in mab-5(gk670) (4/20 versus 17/20). Meanwhile, 14/20 QL.a/p began migration in egl-20(n585) at the same timepoint (significantly more than mab-5(gk670) (p = 0.004)). This delay is also observed in the 5.0–6.0h timepoint (Fig 4), where 20/20 QL.a/p began anterior migration in egl-20(n585), compared to 17/20 and 10/20 (p = 0.0004) in two mab-5 mutants, despite all QR.a/p having begun migration in mab-5 mutants.
To explore this difference between mab-5 and egl-20 further, we determined, at the 4.5–5.5h timepoint, the proportion of QL.a/p that had polarized and begun to migrate anteriorly in animals with migrating QR.a/p (Fig 10). In wild type, 4% of QL.a had begun migration (to the posterior), while in egl-20(n585) and egl-20(hu105), 78% and 80% of QL.a/p had begun anterior migration (p < 0.05). Furthermore, we noted that 8% of QL.a/p polarized and migrated before QR.a/p in egl-20(n585) (Fig 6B).
Fig 10. QL.a/p migrate anteriorly sooner in egl-20 than in mab-5.
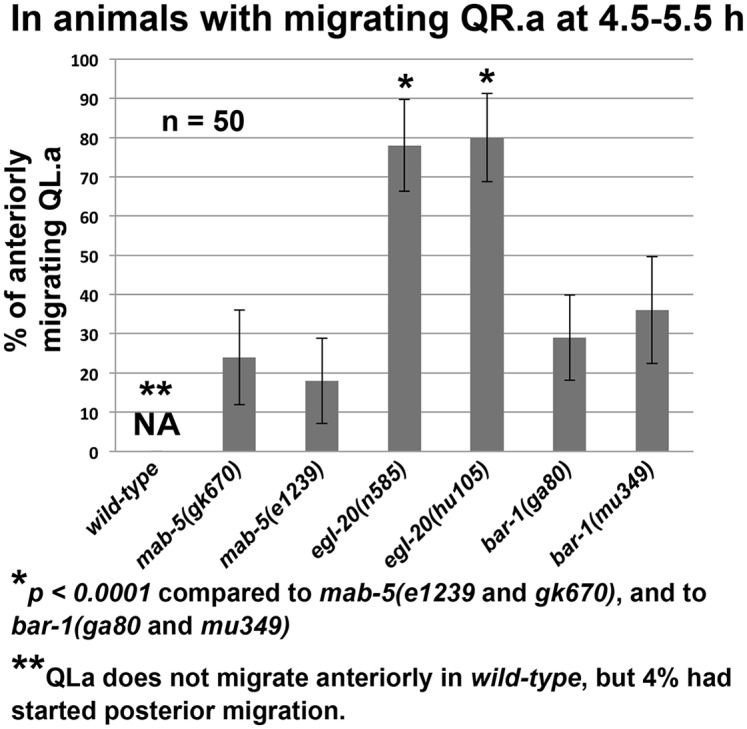
Animals from the 4.5–5.5h timepoint with migrating QR.a were analyzed for QL.a anterior migration. The percentage of animals with anteriorly-migrating QL.a (Y axis) is indicated for each genotype (X axis). In wild-type, QL.a migrated posteriorly (4%). Error bars represent 2x standard error of the proportion, and p values were determined using Fisher’s exact test.
In this assay, two mab-5 mutants displayed significantly less QL.a/p migration compared egl-20 (24% and 18% compared to 78% and 80%, p < 0.05) (Fig 10). These data demonstrate that QL.a/p anterior polarization and migration in mab-5 mutants is delayed relative to egl-20, with QL.a/p in mab-5 transiently retaining a rounded, non-migratory morphology (Fig 7B), a delay not observed in egl-20 mutants.
bar-1/β-catenin QL.a/p Migration Resembles mab-5
MAB-5 expression in QL.a/p is induced by canonical Wnt signaling involving EGL-20/Wnt and BAR-1/β-catenin [9–16]. bar-1 loss-of-function mutations displayed anterior PQR migration similar to egl-20 and mab-5 (Fig 1). bar-1(ga80 and mu349) displayed nearly completely-penetrant anterior PQR migration, suggesting they cause strong loss of function, although mu349 showed a significantly weaker effect (Fig 1). bar-1(mu63) showed much weaker PQR defects and is likely a hypomorphic allele (Fig 1). We analyzed QL.a/p behavior in the strong bar-1 alleles ga80 and mu349. As in egl-20 and mab-5, QL.a/p polarized and migrated anteriorly (Fig 10). To determine if bar-1 resembled egl-20 or displayed the delay of mab-5, we scored the percentage of migrating QL.a/p in bar-1 mutants in which QR.a/p had begun migration at the 4.5–5.5h timepoint (Fig 10): 29% of bar-1(ga80) and 36% of bar-1(mu349) showed QL.a/p migration. This was significantly less than egl-20 mutants (p < 0.05) and was not significantly different from mab-5 (Fig 10). Thus, bar-1 mutants also displayed a delay in QL.a/p anterior migration similar to mab-5 and not seen in egl-20.
Discussion
MAB-5/Hox and EGL-20/Wnt Inhibit Anterior Migration
Here we describe the distinct behaviors of QL and QR descendants in response to Wnt signaling. After the first Q cell division, QR.a/p and QL.a/p had a rounded morphology with little or no protrusion. Shortly after division, QR.a/p began to elongate in the anterior-posterior axis by extending F-actin-rich lamellipodial protrusions to the anterior. They then began anterior migration with polarized, migratory morphology for approximately 2h, at which time they stopped migrating, become rounded, and underwent their second round of cell division. In contrast, QL.a/p remain rounded with little or no protrusive morphology and migration. Our results show that EGL-20/Wnt and MAB-5/Hox are required to prevent QL.a/p from polarizing and migrating to the anterior. When MAB-5/Hox was ectopically expressed in QR.a/p via the mab-5(e1751) mutation or transgenic expression of mab-5 in both Q cells, QR.a/p polarization and migration was similarly inhibited.
EGL-20/Wnt has MAB-5-Independent and MAB-5-Dependent Roles
Both egl-20 and mab-5 loss-of-function mutants displayed anterior QL.a/p migration. However, we noted a delay in anterior migration in mab-5 not observed in egl-20. QL.a/p in mab-5 remain rounded and non-migratory for a short time (~0.5-1h) before they polarized and migrated anteriorly. In contrast, QL.a/p in egl-20 mutants resembled QR.a/p: they immediately polarized and began anterior migration. mab-5 expression is induced in QL by canonical Wnt signaling via EGL-20/Wnt and BAR-1/ β-catenin. The delay in QL.a/p anterior migration was also observed in bar-1/β-catenin mutants, similar to mab-5. This suggests that EGL-20/Wnt has at least two distinct roles in inhibiting migration (Fig 11). EGL-20/Wnt might acutely inhibit migration in a MAB-5 and BAR-1-independent pathway, and later, via canonical Wnt signaling and MAB-5 expression, consolidate this inhibition. Consistent with this idea of step-wise regulation of Q descendant migration by EGL-20, previous studies showed that QR descendants display cell-intrinsic temporal responses to Wnt signals that determines their final positions in the anterior-posterior axis [35].
Fig 11. A model of EGL-20/Wnt and MAB-5/Hox function in QL migration.
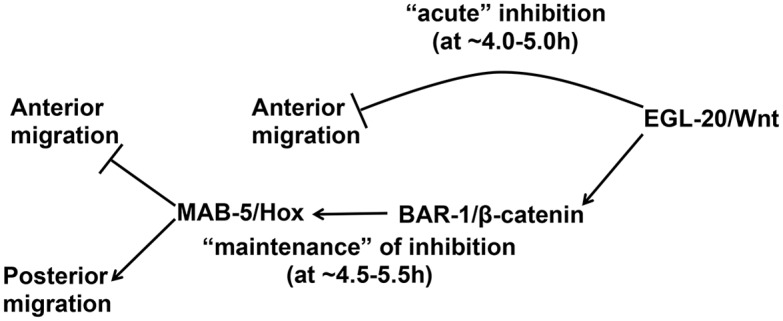
Data presented here indicate that both EGL-20/Wnt and MAB-5/Hox are required to inhibit anterior migration of QL.a/p. EGL-20 might immediately inhibit QL.a/p anterior migration via an “acute” mechanisms independent of BAR-1/β-catenin and MAB-5/Hox, as well as through a later “maintenance” phase that requires BAR-1 and MAB-5 and might require changes in gene expression. MAB-5 might also promote posterior migration.
How might EGL-20 and MAB-5 inhibit QL.a/p anterior migration? EGL-20 could activate an acute response by acting directly on the ability of the cell to polarize and migrate (e.g. e.g. inhibiting cytoskeletal rearrangements and protrusive events necessary for migration). This response does not require BAR-1 or MAB-5, suggesting that it might be a non-canonical Wnt signaling pathway that acts directly on protrusive ability (e.g. the cytoskeleton). There is precedence for BAR-1-independent Wnt signaling in later QR descendant migrations, which are shortened in Wnt mutants independent of BAR-1/β-catenin [18]. In addition to the acute role, which is transient, EGL-20 can also activate canonical Wnt signaling via BAR-1/β-catenin resulting in MAB-5 expression. The acute EGL-20 signal might inhibit anterior migration until MAB-5 is expressed, which begins in QL during its initial migration and before division [17]. In this scenario, the delay in anterior migration in mab-5 mutants might indicate the lag time in mab-5 expression and regulation of target gene expression to inhibit anterior migration. Wnt signaling and mab-5 expression remain tied in a feedback loop that ensures consistent and continuous levels of mab-5 expression [17].
MAB-5 might consolidate inhibition of anterior migration by regulating genes involved in polarization and migration and/or genes involved in responding to an A-P guidance cue. Indeed, expression of the Hox factor LIN-39 is inhibited in QL.a/p by MAB-5 [30]. LIN-39 normally promotes anterior migration in QR.a/p by driving the expression of the transmembrane MIG-13 molecule [30]. In parallel to SDN-1/Syndecan [37], MIG-13 responds to an A-P guidance cue by mediating cytoskeletal rearrangements underlying anterior protrusion and migration [30]. Thus, MAB-5 might inhibit anterior QL.a/p migration in part by inhibiting LIN-39 expression in QL.a/p.
MAB-5 Promotes Posterior Migration
While QL.a/p normally remain rounded and non-migratory while QR.a/p migrate anteriorly, QL.a eventually migrates posteriorly over QL.p. QL.a posterior migration is dependent on MAB-5, as we found that ectopic expression of MAB-5 in both QL and QR resulted in posterior migration of both anterior daughters. Thus, MAB-5 is not simply inhibiting migration. One possibility is that by inhibiting anterior migration, MAB-5 allows QL.a/p to respond to a later de novo guidance signal that directs posterior migration. By not migrating anteriorly, QL.a/p are in a position to respond to this new signal. Alternatively, MAB-5 might regulate gene expression in QL.a/p that alters response to a posterior guidance cue, possibly the same A-P cue used by QR.a/p to migrate anteriorly. As Wnts regulate Q descendant migrations, MAB-5 might alter the response of QL.a/p to Wnt signals directing anterior-posterior migrations.
The Wnt quadruple mutant cwn-1; egl-20 cwn-2; lin-44 showed some posterior migration of both AQR and PQR. EGL-20/Wnt is required to activate mab-5 expression, so it is possible that in the Wnt quadruple, posterior migration occurs in the absence of mab-5. This indicates that Wnts constitute the anterior-posterior guidance system to which mab-5 modifies responses. In the absence of Wnts, some cells migrate posteriorly despite not having mab-5 expression due to a disrupted anterior-posterior guidance system. It is also possible that mab-5 is activated in an egl-20-independent manner in both AQR and PQR in this quadruple mutant and is responsible for posterior migration.
In sum, our results indicate that both QR.a/p and QL.a/p can both respond to an anterior migration signal. Normally, EGL-20 prevents QL.a/p from responding by first acutely inhibiting migration, likely due to the inherent sensitivity of QL to the EGL-20 signal [16]. EGL-20 also activates mab-5 expression in QL.a/p via BAR-1/ β-catenin and canonical Wnt signaling, which likely results in changes in gene expression that maintains inhibition of anterior migration. MAB-5-induced gene expression might also then promote posterior migration. However, it is also possible that by inhibiting anterior migration, mab-5 allows the QL.a/p cells to respond to a later posterior migration signal that is not present early in QL.a/p migration. These results indicate that egl-20 and mab-5 mutations do not transform QL into a QR-like fate, but rather discretely modify how these cells differentially respond to anterior-posterior guidance information.
These studies and others are beginning to paint a picture of the early migration events of the Q neuroblasts and descendants. Initial anterior QR and posterior QL migration are regulated by inherent differences in the functions of the transmembrane molecules UNC-40/DCC and PTP-3/LAR in QR versus QL [29], and does not appear to involve Wnt signaling. While the signal specifying initial anterior versus posterior migration is unknown, the Fat-like Cadherin CDH-4 non-autonomously controls UNC-40/DCC and PTP-3/LAR function in QR versus QL [26]. Due to posterior migration of QL and inherent differences in sensitivity, an EGL-20/Wnt signal acutely inhibits QL.a/p anterior migrations, possibly via a non-canonical mechanism. Via canonical Wnt signaling and BAR-1/β-catenin, EGL-20/Wnt also induces MAB-5 expression in QL.a/p, which inhibits LIN-39/Hox and thus MIG-13 expression and consolidates inhibition of anterior migration. Additionally, MAB-5 might regulate other genes that inhibit anterior migration and direct posterior migration. It will be important to identify the potential non-canonical mechanism of EGL-20/Wnt inhibition of anterior migration, and to define other genes regulated by MAB-5 to autonomously inhibit anterior migration and promote posterior migration.
Methods
Genetics
C. elegans were cultured by standard techniques and all experiments performed at 20°C. The following mutations and transgenes were used: LGI: lin-44(n1792), mom-5(ne12), lin-17(n761); LGII cwn-1(ok546), ayIs9[Pegl-17::gfp], mig-14(ga62 and or78); LGIII mab-5(gk670, e1239, e2088, mu114, bx54, and e1751), lin-39(e1760); LGIV cwn-2(ok895), egl-20(n585, ju105, gk453010, lq42, and lq74); LGV mom-2(or77), lqIs58[Pgcy-32::cfp]; LGX: bar-1(ga80, mu63, and mu349); unmapped lqIs220[Pegl-17::mab-5::gfp], lqIs221[Pegl-17::mab-5::gfp], casIs330[Pegl-17::gfp, Pegl-17::myr::mCherry, Pegl-17::his-24::mCherry, Pegl-17::MOEabd::gfp] [31]. Genotypes containing M+ indicate that homozygous animals from a heterozygous mother were scored (i.e. had wild-type maternal gene function). In the case of the Wnt triple and quadruple mutant, linkage group IV containing egl-20 and cwn-2 was balanced by the nT1 balancer chromosome marked with pharyngeal GFP.
The Pegl-17::mab-5::gfp transgene was produced by placing a full-length mab-5 cDNA downstream of the egl-17 promoter and fused in frame to the gfp coding region at the 3’ end (C-terminal tag). The sequence of this plasmid, pEL862, is available upon request. Six independent extrachromosomal arrays and two independent integrants (lqIs220 and lqIs221) had a similar effect of causing posterior AQR migration (Fig 1).
egl-20 and mab-5 Allele Sequencing
The lesions associated with egl-20(lq42 and lq74) and mab-5(mu114) were determined by whole genome sequencing using Cloudmap [20] and confirmed with Sanger sequencing of the region. egl-20(lq42) was a C to T transition at chromosome IV position 9,813,864 (Wormbase release WS249) resulting in an arginine to stop; egl-20(lq74) was a G to A transition at 9,814,127 resulting in a cysteine to tyrosine missense; egl-20(gk453010) was a C to T transition at 9,814,137 resulting in an arginine to stop; and mab-5(mu114) was a G to A transition at chromosome III position 7,783,349 in the first exon of mab-5 resulting in a tryptophan to stop. mab-5(e2088) was a complex rearrangement involving the second exon of mab-5 at position 7,782,932. The exact molecular nature of e2088 could not be determined.
L1 Synchronization and Q Cell Imaging
L1 larvae were synchronized by hatching as previously described. Gravid adults and larvae were washed from plates on which many eggs had been laid. Eggs were allowed to hatch for one hour, and newly hatched larvae, all within an hour’s age of one another, were washed off and allowed to develop for specific times: 3.5h for the 3.5–4.5h timepoint; 4h for the 4-5h timepoint; 4.5h for the 4.5–5.5h timepoint; 5h for the 5-6h timepoint; 5.5h for the 5.5–6.5h timepoint; 6h for the 6-7h timepoint; 6.5h for the 6.5–7.5h timepoint; and 7h for the 7-8h timepoint. At the specified timepoint, L1 larvae were mounted for microscopic inspection and imaging of Q cell position and morphology using the ayIs9[Pegl-17::gfp] transgene.
Scoring Q Cell Position
From micrographs of L1 larvae expressing ayIs9[Pegl-17::gfp], the morphology and position of QL.a/p and QR.a/p were determined. QR.a/p cells were scored as migrating if they had assumed an elongated migratory morphology with anterior protrusions and/or if QL.a had separated from QL.p (Figs 2 and 3). Cells that remained rounded and adjacent to one another were scored as non-migratory. Posterior migration of QL.a in wild type and QL.a and QR.a in mab-5 gain-of-function was defined by QX.a extending protrusions on top of QX.p, or if QX.a could not be distinguished from QX.p, indicating that QX.a was migrating behind or in front of QX.p (Figs 2 and 3). Significance of differences in Fig 4 were determined using Fisher’s exact test.
For data in Fig 10, L1 larvae were synchronized and imaged at the 4.5–5.5h timepoint, when most QR.a/p had migrated anteriorly and QL.a/p remained rounded and non-migratory. The migrations of QL.a/p were scored in those animals with migratory QR.a/p (n = 50 for each genotype). Significances of difference were determined by Fisher’s exact test.
Time-Lapse Imaging of QL and QR
For time-lapse imaging, the casIs330 transgene was used to visualize QL, QR and descendants. casIs330 contains the Q-cell promoter egl-17 driving the expression of myr::mCherry (to mark membranes), his-24::mCherry (to mark chromatin), and MOEabd::gfp, the actin-binding domain of human moesin fused to GFP, to mark F-actin [31]. Images were acquired every 2 minutes using a spinning disk confocal microscope (Zeiss Axio Observer Z1, with Yokogawa CSU-X1 Spinning Disk Unit) using previously-described methods to anesthetize and immobilize animals. Images were analyzed and assembled using ImageJ and Adobe Photoshop. Time-lapse imaging with the casIs330 transgene delayed migrations and cell divisions by ~2-fold compared to ayIs9 still images, but the same pattern of QL.a/p and QR.a/p migration as observed with ayIs9 still images was conserved.
Scoring AQR and PQR Migration
AQR and PQR are neuronal descendants of QR and QL, respectively, and migrate the longest distances of Q descendants into the head and tail of the animal. AQR normally migrates anteriorly to a region near the anterior deirid and the posterior pharyngeal bulb, and PQR normally migrates posteriorly near the phasmid ganglia posterior to the anus in the tail. AQR and PQR position was scored as described previously. The animal is divided into five regions in the anterior-posterior axis: Position 1 is the normal position of AQR; position 2 is posterior to position 1 but anterior to the vulva; position 3 is adjacent to the vulva; position 4 is the position of Q cell birth; and position 5 is the normal position of PQR posterior to the anus. AQR and PQR position was scored in 100 animals of each genotype indicated in Fig 1.
Supporting Information
casIs330[Pegl-17::myr::mCherry, Pegl-17::HIS-24::mCherry, Pegl-17:: MOEabd::GFP] was used to image QR division and QR.a/p migrations. mCherry labeled cell membranes and chromatin (red), and GFP labeled F-actin (green). Images were acquired every 2 minutes using a spinning disk confocal microscope (Zeiss Axio Observer Z1, with Yokogawa CSU-X1 Spinning Disk Unit). Total imaging time was 200 minutes. Time-lapse imaging with casIs330 resulted in an approximately 2-fold delay in events relative to ayIs9 in timepoint analysis. Anterior is left, and dorsal is up.
(AVI)
casIs330[Pegl-17::myr::mCherry, Pegl-17::HIS-24::mCherry, Pegl-17:: MOEabd::GFP] was used to image QL division and QL.a/p migrations. mCherry labels cell membranes and chromatin (red) and GFP labels F-actin (green). Images were acquired every 2 minutes using a spinning disk confocal microscope (Zeiss Axio Observer Z1, with Yokogawa CSU-X1 Spinning Disk Unit). Total imaging time was 250 minutes. Time-lapse imaging with casIs330 resulted in an approximately 2-fold delay in events relative to ayIs9 in timepoint analysis. Anterior is left, and dorsal is up.
(AVI)
Acknowledgments
We thank members of the Lundquist and Ackley labs for discussion, and E. Struckhoff for technical assistance. This work was supported by NIH grants R01 NS040945 and R21 NS070417 to E.A.L., the Kansas Infrastructure Network of Biomedical Research Excellence (NIH P20 GM103418), the National Basic Research Program of China to G.O. (973 Program, 2013CB945600 and 2012CB945002), and the National Natural Science Foundation of China to G.O. and Y.C. (31201009, 31222035, 31171295 and 31190063). Some strains were provided by the CGC, which is funded by NIH Office of Research Infrastructure Programs (P40 OD010440). Some next-generation sequencing was provided by the University of Kansas Genome Sequencing Core Laboratory of the Center for Molecular Analysis of Disease Pathways (NIH P20 GM103638).
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
Funded by National Institutes of Health grants R01 NS040945, R21 NS070417, P20 GM103638, and P20 GM103418 to E.A.L. (http://www.nih.gov/), The National Basic Research Program of China to G.O. (973 Program, 2013CB945600 and 2012CB945002) (http://rd.zju.edu.cn/en/index.php?c=main&a=detail&id=93), and The National Natural Science Foundation of China to G.O. and Y.C. (31201009, 31222035, 31171295 and 31190063) (http://www.nsfc.gov.cn/publish/portal1/). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Freese JL, Pino D, Pleasure SJ. Wnt signaling in development and disease. Neurobiol Dis. 2010;38(2):148–53. 10.1016/j.nbd.2009.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boitard M, Bocchi R, Egervari K, Petrenko V, Viale B, Gremaud S, et al. Wnt signaling regulates multipolar-to-bipolar transition of migrating neurons in the cerebral cortex. Cell reports. 2015;10(8):1349–61. 10.1016/j.celrep.2015.01.061 . [DOI] [PubMed] [Google Scholar]
- 3.Schafer ST, Han J, Pena M, von Bohlen Und Halbach O, Peters J, Gage FH. The Wnt adaptor protein ATP6AP2 regulates multiple stages of adult hippocampal neurogenesis. J Neurosci. 2015;35(12):4983–98. 10.1523/JNEUROSCI.4130-14.2015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Middelkoop TC, Korswagen HC. Development and migration of the C. elegans Q neuroblasts and their descendants. WormBook. 2014:1–23. Epub 2014/10/16. 10.1895/wormbook.1.173.1 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chalfie M, Sulston J. Developmental genetics of the mechanosensory neurons of Caenorhabditis elegans. Dev Biol. 1981;82(2):358–70. [DOI] [PubMed] [Google Scholar]
- 6.Sulston JE, Horvitz HR. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev Biol. 1977;56(1):110–56. . [DOI] [PubMed] [Google Scholar]
- 7.Chapman JO, Li H, Lundquist EA. The MIG-15 NIK kinase acts cell-autonomously in neuroblast polarization and migration in C. elegans. Dev Biol. 2008;324(2):245–57. Epub 2008/10/09. S0012-1606(08)01206-2 [pii] 10.1016/j.ydbio.2008.09.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Honigberg L, Kenyon C. Establishment of left/right asymmetry in neuroblast migration by UNC-40/DCC, UNC-73/Trio and DPY-19 proteins in C. elegans. Development. 2000;127(21):4655–68. . [DOI] [PubMed] [Google Scholar]
- 9.Chalfie M, Thomson JN, Sulston JE. Induction of neuronal branching in Caenorhabditis elegans. Science. 1983;221(4605):61–3. Epub 1983/07/01. . [DOI] [PubMed] [Google Scholar]
- 10.Eisenmann DM. Wnt signaling. WormBook. 2005:1–17. Epub 2007/12/01. 10.1895/wormbook.1.7.1 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Harris J, Honigberg L, Robinson N, Kenyon C. Neuronal cell migration in C. elegans: regulation of Hox gene expression and cell position. Development (Cambridge, England). 1996;122(10):3117–31. Epub 1996/10/01. . [DOI] [PubMed] [Google Scholar]
- 12.Herman MA. Wnt signaling in C. elegans In Wnt signaling in Development (ed. Kühl M.), pp. 187–212. Georgetown, TX: Landes Biosciences; 2003. [Google Scholar]
- 13.Kenyon C. A gene involved in the development of the posterior body region of C. elegans. Cell. 1986;46(3):477–87. [DOI] [PubMed] [Google Scholar]
- 14.Korswagen HC, Herman MA, Clevers HC. Distinct beta-catenins mediate adhesion and signalling functions in C. elegans. Nature. 2000;406(6795):527–32. Epub 2000/08/22. 10.1038/35020099 . [DOI] [PubMed] [Google Scholar]
- 15.Salser SJ, Kenyon C. Activation of a C. elegans Antennapedia homologue in migrating cells controls their direction of migration. Nature. 1992;355(6357):255–8. . [DOI] [PubMed] [Google Scholar]
- 16.Whangbo J, Kenyon C. A Wnt signaling system that specifies two patterns of cell migration in C. elegans. Mol Cell. 1999;4(5):851–8. . [DOI] [PubMed] [Google Scholar]
- 17.Ji N, Middelkoop TC, Mentink RA, Betist MC, Tonegawa S, Mooijman D, et al. Feedback control of gene expression variability in the Caenorhabditis elegans Wnt pathway. Cell. 2013;155(4):869–80. Epub 2013/11/12. 10.1016/j.cell.2013.09.060 . [DOI] [PubMed] [Google Scholar]
- 18.Zinovyeva AY, Yamamoto Y, Sawa H, Forrester WC. Complex Network of Wnt Signaling Regulates Neuronal Migrations During Caenorhabditis elegans Development. Genetics. 2008;179(3):1357–71. Epub 2008/07/16. genetics.108.090290 [pii] 10.1534/genetics.108.090290 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tamayo JV, Gujar M, Macdonald SJ, Lundquist EA. Functional transcriptomic analysis of the role of MAB-5/Hox in Q neuroblast migration in Caenorhabditis elegans. BMC genomics. 2013;14:304 Epub 2013/05/07. 10.1186/1471-2164-14-304 ; PubMed Central PMCID: PMCPmc3651406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Minevich G, Park DS, Blankenberg D, Poole RJ, Hobert O. CloudMap: a cloud-based pipeline for analysis of mutant genome sequences. Genetics. 2012;192(4):1249–69. Epub 2012/10/12. 10.1534/genetics.112.144204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Thompson O, Edgley M, Strasbourger P, Flibotte S, Ewing B, Adair R, et al. The million mutation project: a new approach to genetics in Caenorhabditis elegans. Genome Res. 2013;23(10):1749–62. Epub 2013/06/27. 10.1101/gr.157651.113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Penigault JB, Felix MA. High sensitivity of C. elegans vulval precursor cells to the dose of posterior Wnts. Dev Biol. 2011;357(2):428–38. Epub 2011/06/29. 10.1016/j.ydbio.2011.06.006 . [DOI] [PubMed] [Google Scholar]
- 23.Coudreuse DY, Roel G, Betist MC, Destree O, Korswagen HC. Wnt gradient formation requires retromer function in Wnt-producing cells. Science. 2006;312(5775):921–4. Epub 2006/04/29. 10.1126/science.1124856 . [DOI] [PubMed] [Google Scholar]
- 24.Pan CL, Howell JE, Clark SG, Hilliard M, Cordes S, Bargmann CI, et al. Multiple Wnts and frizzled receptors regulate anteriorly directed cell and growth cone migrations in Caenorhabditis elegans. Dev Cell. 2006;10(3):367–77. Epub 2006/03/07. 10.1016/j.devcel.2006.02.010 . [DOI] [PubMed] [Google Scholar]
- 25.Yang PT, Lorenowicz MJ, Silhankova M, Coudreuse DY, Betist MC, Korswagen HC. Wnt signaling requires retromer-dependent recycling of MIG-14/Wntless in Wnt-producing cells. Dev Cell. 2008;14(1):140–7. Epub 2007/12/28. 10.1016/j.devcel.2007.12.004 . [DOI] [PubMed] [Google Scholar]
- 26.Sundararajan L, Norris ML, Schoneich S, Ackley BD, Lundquist EA. The fat-like cadherin CDH-4 acts cell-non-autonomously in anterior-posterior neuroblast migration. Dev Biol. 2014;392(2):141–52. Epub 2014/06/24. 10.1016/j.ydbio.2014.06.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Branda CS, Stern MJ. Mechanisms controlling sex myoblast migration in Caenorhabditis elegans hermaphrodites. Dev Biol. 2000;226(1):137–51. Epub 2000/09/20. 10.1006/dbio.2000.9853 . [DOI] [PubMed] [Google Scholar]
- 28.Cordes S, Frank CA, Garriga G. The C. elegans MELK ortholog PIG-1 regulates cell size asymmetry and daughter cell fate in asymmetric neuroblast divisions. Development. 2006;133(14):2747–56. Epub 2006/06/16. 10.1242/dev.02447 . [DOI] [PubMed] [Google Scholar]
- 29.Sundararajan L, Lundquist EA. Transmembrane proteins UNC-40/DCC, PTP-3/LAR, and MIG-21 control anterior-posterior neuroblast migration with left-right functional asymmetry in Caenorhabditis elegans. Genetics. 2012;192(4):1373–88. Epub 2012/10/12. 10.1534/genetics.112.145706 ; PubMed Central PMCID: PMCPmc3512145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang X, Zhou F, Lv S, Yi P, Zhu Z, Yang Y, et al. Transmembrane protein MIG-13 links the Wnt signaling and Hox genes to the cell polarity in neuronal migration. Proc Natl Acad Sci U S A. 2013;110(27):11175–80. Epub 2013/06/21. 10.1073/pnas.1301849110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shen Z, Zhang X, Chai Y, Zhu Z, Yi P, Feng G, et al. Conditional knockouts generated by engineered CRISPR-Cas9 endonuclease reveal the roles of coronin in C. elegans neural development. Developmental cell. 2014;30(5):625–36. Epub 2014/08/27. 10.1016/j.devcel.2014.07.017 . [DOI] [PubMed] [Google Scholar]
- 32.Tian D, Diao M, Jiang Y, Sun L, Zhang Y, Chen Z, et al. Anillin Regulates Neuronal Migration and Neurite Growth by Linking RhoG to the Actin Cytoskeleton. Curr Biol. 2015;25(9):1135–45. 10.1016/j.cub.2015.02.072 . [DOI] [PubMed] [Google Scholar]
- 33.Ou G, Vale RD. Molecular signatures of cell migration in C. elegans Q neuroblasts. The Journal of cell biology. 2009;185(1):77–85. Epub 2009/04/08. 10.1083/jcb.200812077 ; PubMed Central PMCID: PMCPmc2700521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chow KL, Emmons SW. HOM-C/Hox genes and four interacting loci determine the morphogenetic properties of single cells in the nematode male tail. Development. 1994;120(9):2579–92. . [DOI] [PubMed] [Google Scholar]
- 35.Mentink RA, Middelkoop TC, Rella L, Ji N, Tang CY, Betist MC, et al. Cell intrinsic modulation of Wnt signaling controls neuroblast migration in C. elegans. Developmental cell. 2014;31(2):188–201. 10.1016/j.devcel.2014.08.008 . [DOI] [PubMed] [Google Scholar]
- 36.Salser SJ, Loer CM, Kenyon C. Multiple HOM-C gene interactions specify cell fates in the nematode central nervous system. Genes & development. 1993;7(9):1714–24. Epub 1993/09/01. . [DOI] [PubMed] [Google Scholar]
- 37.Sundararajan L, Norris ML, Lundquist EA. SDN-1/Syndecan Acts in Parallel to the Transmembrane Molecule MIG-13 to Promote Anterior Neuroblast Migration. G3. 2015;5(8):1567–74. 10.1534/g3.115.018770 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
casIs330[Pegl-17::myr::mCherry, Pegl-17::HIS-24::mCherry, Pegl-17:: MOEabd::GFP] was used to image QR division and QR.a/p migrations. mCherry labeled cell membranes and chromatin (red), and GFP labeled F-actin (green). Images were acquired every 2 minutes using a spinning disk confocal microscope (Zeiss Axio Observer Z1, with Yokogawa CSU-X1 Spinning Disk Unit). Total imaging time was 200 minutes. Time-lapse imaging with casIs330 resulted in an approximately 2-fold delay in events relative to ayIs9 in timepoint analysis. Anterior is left, and dorsal is up.
(AVI)
casIs330[Pegl-17::myr::mCherry, Pegl-17::HIS-24::mCherry, Pegl-17:: MOEabd::GFP] was used to image QL division and QL.a/p migrations. mCherry labels cell membranes and chromatin (red) and GFP labels F-actin (green). Images were acquired every 2 minutes using a spinning disk confocal microscope (Zeiss Axio Observer Z1, with Yokogawa CSU-X1 Spinning Disk Unit). Total imaging time was 250 minutes. Time-lapse imaging with casIs330 resulted in an approximately 2-fold delay in events relative to ayIs9 in timepoint analysis. Anterior is left, and dorsal is up.
(AVI)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.