Abstract
Some long non-coding RNAs (lncRNAs) participate in physiological processes that maintain cellular and tissue homeostasis, and thus, the dysregulated expression of lncRNAs is involved in the onset and progression of many pathological conditions. Research has indicated that the genetic knockout of some lncRNAs in mice resulted in peri- or postnatal lethality or developmental defects. Diabetes mellitus (DM) is a major cause of peripheral neuropathy. Our studies showed that the expression levels of lncRNA uc.48+ in the diabetic rat dorsal root ganglia (DRG) and the DM patients’ serum samples were increased. It suggested that lncRNA uc.48+ was involved in the pathophysiological process of DM. The aim of this study was to investigate the effects of lncRNA uc.48+ small interfering RNA (siRNA) on diabetic neuropathic pain (DNP) mediated by the P2X3 receptor in the DRG. The values of the mechanical withdrawal threshold (MWT) and thermal withdrawal latency (TWL) were measured by the von Frey test and Hargreaves’ test, respectively. The levels of P2X3 protein and messenger RNA (mRNA) in the DRG were detected by reverse transcription-polymerase chain reaction (RT-PCR), immunohistochemistry, and western blotting. The experiments showed that the MWT and TWL values in DM rats were lower than those in the control rats. The MWT and TWL values in DM rats treated with lncRNA uc.48+ siRNA were increased compared to those in DM rats, but there was no significant difference between the DM rat group and the DM + scramble siRNA group. The levels of P2X3 protein and mRNA in the DM DRG were higher than those in the control, while the levels of P2X3 protein and mRNA in the DG of DM rats treated with uc.48+ siRNA were significantly decreased compared to those in DM rats. The expression levels of TNF-α in the DRG of DM rats treated with uc.48+ siRNA were significantly decreased compared to those in the DM group. The phosphorylation and activation of ERK1/2 in the DM DRG were decreased by uc.48+ siRNA treatment. Therefore, uc.48+ siRNA treatment may alleviate the DNP by inhibiting the excitatory transmission mediated by the P2X3 receptor in DRG.
Keywords: P2X3 receptor, Long non-coding RNA (lncRNA), Diabetic neuropathic pain, Dorsal root ganglia
Introduction
Peripheral neuropathic pain is initiated by a primary injury or dysfunction in the peripheral nervous system [1–3]. Peripheral sensitization is a mechanism of causing neuropathic pain. The activation thresholds in nociceptors are downregulated following exposure to inflammatory mediators that induce peripheral sensitization. The global prevalence of diabetes mellitus (DM) in 2011 was approximately 366 million (8.3 %), and the numbers are expected to increase to 552 million (9.9 %) by 2030 [4]. DM is a major cause of peripheral neuropathy. Diabetic neuropathic pain (DNP) is a direct consequence of abnormalities in the dorsal root ganglia (DRG) [3, 5, 6]. Hyperglycemia destroys the homeostasis of peripheral tissues by damaging nerve fibers [7]. DNP is a result of glucose toxicity associated with local metabolic changes in DM [5, 8]. DNP can be displayed as spontaneous pain, allodynia (pain to normally innocuous stimuli), and hyperalgesia (increased pain perception to noxious stimuli) [5, 6, 9, 10]. One third of patients with DNP have painful symptoms such as burning, tingling (“pins and needles” or paresthesia), shooting (like an electric shock), or lancing (stabbing) [5, 6, 9, 10]. DNP is a chronic peripheral neuropathy and exerts a substantial impact on the quality of life of diabetic patients.
Human protein-coding genes represent only approximately 1.5 % of the DNA or approximately 20,000 genes [11, 12]. This indicates that up to 90 % of eukaryotic genomes are transcribed, generating a large population of non-coding RNAs (ncRNAs) [13]. Based on transcript size, the non-coding RNAs can be divided into small non-coding RNAs (<200 bp) and long non-coding RNAs (lncRNAs; ≥200 nt). The majority of small RNAs, such as microRNAs (miRNAs), are extensively studied. Small ncRNAs are highly conserved and are involved in transcriptional and posttranscriptional gene silencing via specific base pairing with target genes [14, 15]. In contrast, lncRNAs are often poorly conserved at the sequence level and are considered to be transcriptional “noise” [16, 17]. Although the vast majority of lncRNA functions are unclear, there is evidence that some lncRNAs participate in physiological processes that maintain cellular and tissue homeostasis. Thus, the dysregulated expression of lncRNAs is involved in the onset and progression of many pathological conditions [18–21]. Moreover, one study indicated that the genetic knockout of some lncRNAs in mice resulted in peri- or postnatal lethality or developmental defects [22]. It is possible that the lncRNAs that were previously considered “junk DNA” may be essential for life. LncRNAs are being revealed as important components of gene regulatory networks, working in concert with transcription factors and epigenetic regulators of gene expression [23–28].
Adenosine-5′-triphosphate (ATP) triggers pain under normal conditions and increases inflammatory-mediated pain [29–35]. ATP activates P2X cation-permeable ion channel receptors and P2Y G protein-coupled receptors [30, 31, 36]. The P2X receptor family (P2X1–7) has been cloned [30, 31, 36]. The P2X3 receptor is most highly expressed in a subpopulation of small-diameter primary afferent neurons. The P2X3 receptor plays a crucial role in pain transmission at peripheral sensory neurons [30, 31, 33–40]. The expression of P2X3 protein and messenger RNA (mRNA) is upregulated following chronic constriction injury (CCI) of the sciatic nerve [30, 33–37, 39, 40]. The P2X3 receptor plays an important role in chronic neuropathic pain [30, 33, 34, 36–40]. Therefore, the P2X3 receptor is defined as a suitable target for analgesic drugs. Purinergic signaling is involved in DM [41, 42]. The P2X3 receptor mediated mechanical allodynia in diabetic neuropathic rats [42]. The uc.48+ is an lncRNA (http://genome.ucsc.edu/cgi-bin/hgc?hgsid=427967671_gIIKDUiyguaFXCbRBiWSM7gFOgqn&c=chr2&o=20462844&t=20463142&g=ct_Ultra_7128&i=%28null%29+uc.48). The aim of this study was to investigate the effects of lncRNA uc.48+ small interfering RNA (siRNA) on P2X3 receptor-mediated DNP.
Materials and methods
Animals and animal groups
Male Sprague Dawley rats (180–230 g) were provided by the Center of Laboratory Animal Science of Nanchang University. The procedures were approved by the Animal Care and Use Committee of Nanchang University Medical School. The animals were housed in plastic boxes in groups of three at 21–25 °C. The IASP’s ethical guidelines for pain research in animals were followed. All animals were treated in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research in China. Diabetic rat models were induced by an intraperitoneal (i.p.) injection of streptozotocin (STZ) (high-calorie food and a single STZ 65-mg/kg injection for rats) [43, 44]. STZ was dissolved in citrate buffer (pH 4.5). The control animals received buffer. After 7 days, blood samples were obtained from the tail vein, and glycemia was determined using a glucometer. Non-fasting blood glucose >200 mg/dl (11.1 mM) was considered to be DM [43, 44]. At day 14, the animals were killed by CO2 inhalation, and the L4,5 DRG were removed.
The rats were assigned in a random blinded manner to one of the four following groups: control group (control group), diabetic model group (DM group), uc.48+ siRNA vector-treated diabetic rat group (DM + uc.48+ si group), and DM rats treated with scramble siRNA vector group (DM + scramble si group or DM + negative control siRNA: DM + NC si group). Each group contained 8–10 animals. The uc.48+ siRNA or scrambled control siRNA (100 mg/kg/day) was intraperitoneally (i.p.) injected into rats before the experiment, and then spontaneous pain behaviors were measured at 7 days after STZ injection.
uc.48+ siRNA treatment
The siRNA specific for uc.48+ was purchased from Invitrogen (Carlsbad, CA), with the target sequence of 5′-GGCACTACTACTTGCAGAA-3′. The siRNA oligonucleotides that specifically targeted uc.48+ were used in this experiment [45]. The uc.48+ was knocked down by RNA interference (RNAi) using an Entranster™–in vivo Transfection Reagent [46]. According to the procedure of the transfection reagent, we prepared three different volumes of uc.48+ siRNA (80, 160, and 320 μl) with the same ratio (1:2) of siRNA (μg)/transfection reagent (μl). We found that the interference effect of uc.48+ siRNA at the concentration of 320 μl was the best. Therefore, we used the uc.48+ siRNA injection at the concentration of 320 μl for subsequent experiments. The prepared solution was injected into the sublingual vein of rats.
Behavioral studies
Thermal hyperalgesia
The latency to hind paw withdrawal from a thermal stimulus was determined by exposing the plantar surface of the hind paw to radiant heat using the Thermal Paw Stimulation System (BME-410C, Tianjin) [44, 47, 48]. Rats were placed in a transparent, square, bottomless acrylic box (22 cm × 12 cm × 22 cm) on a glass plate under which a light was located. After a 30-min habituation period, the plantar surface of the paw was exposed to a beam of radiant heat applied through the glass floor. Activation of the bulb simultaneously activated a timer, and both were immediately turned off by paw withdrawal or at the 25-s cutoff time. A blinded observer tested the hind paw withdrawal in triplicate at 5-min intervals.
Mechanical hyperalgesia
The mechanical nociceptive threshold was evaluated by observing the withdrawal responses to mechanical stimulation using von Frey filaments (Stoelting, Wood Dale, IL, USA) [44, 47, 48]. The von Frey filament was used to apply a linearly increasing pressure once, starting with 0.13 g and continuing until a withdrawal response occurred or the force reached 20.1 g (the cutoff value). The pressure was applied through a cone-shaped plastic tip with a diameter of 1 mm onto the dorsal surface of the hind paws. The tip was positioned between the third and fourth metatarsus, and force was applied until the rat attempted to withdraw its paw (paw withdrawal threshold to pressure). The hind paws were alternated at 2-min intervals. The pain threshold was calculated as the mean of three consecutive stable values, expressed in grams, and was determined by a blinded observer.
Antibodies and reagents
P2X3 antibody was purchased from Alomone Labs, Jerusalem, Israel. The array slides were obtained from Qiagen (Valencia, CA, USA). β-Actin was from Advanced ImmunoChemical, Long Beach, CA. Other antibodies and reagents are described below.
Reverse transcription-polymerase chain reaction
Total RNA was isolated using TRIzol (Invitrogen, Carlsbad, CA, USA), according to the manufacturer’s protocol. One thousand nanograms of total RNA was used as a template for reverse transcription using the Applied Biosystems Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA). The PCR amplification of the P2X3 receptor and β-actin (control) was performed according to our previously described method [34]. The oligonucleotides used to amplify the P2X3 receptor and β-actin were as follows: for P2X3, sense 5′-CAACTTCAGGTTTGCCAAA-3′ and antisense 5′-TGAACAGTGAGGGCCTAGAT-3′, and for β-actin, sense 5′-TAAAGACCTCTATGC CAACACAGT-3′ and antisense 5′-CACGATGGAGGGGCCGGACTCATC-3′. The PCR products were amplified using the following cycling parameters: 94 °C for 5 min; followed by 35 cycles of 94 °C for 45 s, 53 °C for 30 s, and 72 °C for 40 s; and finally a single cycle at 72 °C for 5 min.
Real-time RT-PCR
Total RNA was isolated from DRG using the TRIzol Total RNA Reagent. Complementary DNA (cDNA) synthesis was performed with 2 μg total RNA using the RevertAid™ H Minus First Strand cDNA Synthesis Kit. The primers were designed with Primer Express 3.0 Software (Applied Biosystems), and the sequences were as follows: uc.48+, forward 5′-GCAAACTGGATGAGGAT-3′ and reverse 5′-GTAGTGCCACAAGGAGA-3′. Quantitative PCR was performed using the SYBR® Green Master Mix in an ABI PRISM® 7500 Sequence Detection System (Applied Biosystems Inc., Foster City, CA). The quantification of gene expression was performed using the ΔΔCT calculation with CT as the threshold cycle. The relative levels of target genes, normalized to the sample with the lowest CT, were given as 2−ΔΔCT [45].
Immunohistochemistry
Immunohistochemical staining was performed using an SP-9001 kit (Beijing Zhongshan Biotech Co.) according to the manufacturer’s instructions. Briefly, the animals were anesthetized with sodium pentobarbital, and the DRG were removed [34]. The isolated rat DRG were washed with phosphate-buffered saline (PBS). After being fixed with 4 % paraformaldehyde (PFA) for 24 h, the ganglia were dehydrated by 20 % sucrose overnight at 4 °C, and then the ganglia were cut into 20-μm-thick sections on a cryostat. After washing with PBS three times, the preparations were incubated in 3 % H2O2 for 10 min to block the endogenous peroxidase activity and then with 10 % goat serum for 30 min at room temperature to block non-specific antigen binding. After rinsing and washing in PBS, the blocked sections were incubated with rabbit anti-P2X3 (1:2500 diluted in PBS; Alomone Labs, Jerusalem, Israel) overnight at 4 °C. After three rinses in PBS, the sections were incubated with a biotinylated goat anti-rabbit secondary antibody (Beijing Zhongshan Biotech Co.) for 1 h at room temperature. The preparations were washed in PBS, and then streptavidin-horseradish peroxidase (Beijing Zhongshan Biotech Co.) was added for 30 min. After development in the diaminobenzidine chromogen for 2 min, the slides were washed with distilled water and cover slipped. After immunohistochemistry, an image scanning analysis system (HMIV-2000, Wuhan) was used to analyze the changes in the levels integrated optical density (IOD) of P2X3 in the ganglia. The background was determined by averaging the optical density of 10 random areas.
Western blotting
The animals were anesthetized with sodium pentobarbital, the DRG were dissected, and approximately 6–10 samples were harvested from each rat. The DRG were isolated immediately and rinsed in ice-cold phosphate-buffered saline (PBS) [48]. After dilution with the sample buffer (250 mmol/l Tris–Cl, 200 mmol/l dithiothreitol, 10 % sodium dodecyl sulfate (SDS), 0.5 % bromophenol blue, 50 % glycerol) and heating to 95 °C for 10 min, 20-μg samples of total protein were separated using 10 % (for P2X3 receptor analysis) SDS–polyacrylamide gel electrophoresis and transferred onto a polyvinylidene difluoride membrane. After incubation with a primary antibody against the P2X3 receptor (1:1000, Chemicon International, Inc., USA), the membrane was incubated with peroxidase-conjugated secondary antibodies (Cell Signaling Technology, Danvers, MA, USA). Immunodetection was performed using the Pierce-enhanced chemiluminescence substrate (Thermo Scientific, Waltham, MA, USA). β-Actin antibody (1:10,000 Santa Cruz Biotechnology, Santa Cruz, CA, USA) was used as a loading control. The band densities were normalized to each β-actin internal control.
Clinical samples, serum RNA isolation, and RT-PCR
Eight healthy subjects were enrolled from the Blood Center of Jiangxi Province. Eight patients fulfilling the American Diabetes Association (ADA) diagnostic criteria for T2DM were recruited from the First Affiliated Hospital of Nanchang University. Following local ethics committee approval, written informed consent was obtained from each individual. Subjects showing DNP complications were asked to participate in the study. Blood samples obtained from all patients were collected and allowed to coagulate for 30 min at room temperature and centrifuged for 10 min at 1300×g. The collected sera were centrifuged for another 10 min at 3000×g to remove any remaining cellular components, divided into 500-μl aliquots, and stored immediately at −80 °C.
Total RNA was isolated from serum using the RNApure Circulating Reagent (CWBIO, Beijing, cat. no. CW2281), with some modifications. Briefly, 300 μl of serum was added to three volumes of RNApure Circulating Reagent, mixed thoroughly by vortexing, and left to stand at room temperature for 5 min. Next, one-fifth volume of chloroform was added, shaken vigorously for 30 s, incubated for 5 min at room temperature, and centrifuged for 20 min at 12,000×g at 4 °C. The upper aqueous phase was transferred to a new collection tube, and one volume of isopropanol was added, mixed thoroughly for 30 min at room temperature, and centrifuged for 20 min at 12,000×g at 4 °C. The precipitate was washed twice with 1 ml of 75 % ethanol (dilute with DEPC water). RNA was eluted by 20 μl of RNase-free water. The quality of the RNA was profiled using NanoDrop 2000 (Thermo Scientific, USA).
Next, 1000 ng of total RNA was used as a template for reverse transcription using the RevertAid First Strand cDNA Synthesis Kit (Thermo, USA). PCR amplification of uc.48+ and GAPDH (control) was performed according to our previous method using oligonucleotides as described in [34]. The following primers were used: uc.48+ sense 5′-GCAAACTGGATGAGGAT-3′ and antisense 5′-GTAGTGCCACAAGGAGA-3′, and GAPDH sense 5′-CAGGGCTGCTTTTAACTCTGGT-3′ and antisense 5′-GATTTTGGAGGGATCTCGCT-3′. The length of the PCR product was 231 bp for uc.48+ and 199 bp for GAPDH. The PCR conditions of uc.48+ and GAPDH included a hot start at 94 °C for 3 min, 45 s denaturation (94 °C), 45 s annealing (57 °C), and 45 s extension (72 °C) for 36 or 32 amplification cycles as well as a 5-min final extension at 72 °C. The PCR products were run on 1.5 % agarose gels with EB using standard protocols. These results were expressed as the ratio of the uc.48+ band intensity/GAPDH band intensity. Data were analyzed using Image-Pro Plus software.
Statistical analysis
The data were statistically analyzed on a computer (SPSS 11.5). All results are expressed as the mean ± SE. Statistical significance was determined by one-way analysis of variance (ANOVA) followed by Fisher’s post hoc test for multiple comparisons. p < 0.05 was considered significant.
Results
Changes in uc.48+ expression in the DM rat DRG and the DM patients’ serum samples
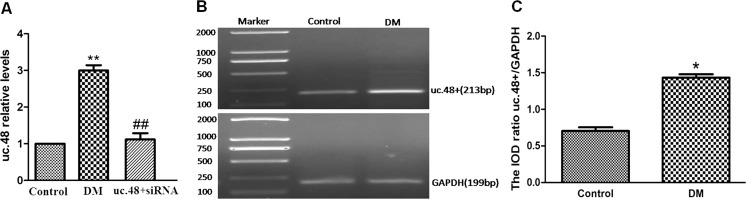
The expression of the lncRNA uc.48+ in the rat DRG was tested by real-time PCR. The results showed that the expression levels of lncRNA uc.48+ in the DM DRG were increased by threefold in comparison with those in the control (p < 0.01; Fig. 1a), while the expression of uc.48+ in rats treated with uc.48+ siRNA (320 μl) was decreased by 60 % compared to that in the DM group (p < 0.01). The expression of the lncRNA uc.48+ in DM patients’ serum samples was tested by reverse transcription-polymerase chain reaction (RT-PCR). The levels of uc.48+ in the serum from DM patients with neuropathic pain complications were increased by threefold than those in the healthy control subjects (p < 0.05, n = 8 for each group; Fig. 1b). These results indicated that uc.48+ was involved in the pathophysiological process of DM.
Fig. 1.
Changes in uc.48+ expression in DM. a Real-time PCR experiments showed the expression of the lncRNA uc.48+ in the rat DRG. The expression of lncRNA uc.48+ in the DM DRG was threefold higher than that in the control group, while the expression of uc.48+ in rats treated with uc.48+ siRNA (320 μl) was decreased by 60 % compared to that in the DM group (a; **p < 0.01 compared with the control group, ##p < 0.01 compared with the DM group). b, c The expression of lncRNA uc.48+ in DM patients’ serum samples was tested by RT-PCR. The levels of uc.48+ in the serum from DM patients with neuropathic pain complications were threefold higher than those in the healthy control subjects (b, c; *p < 0.05 compared with the control group, n = 8 for each group)
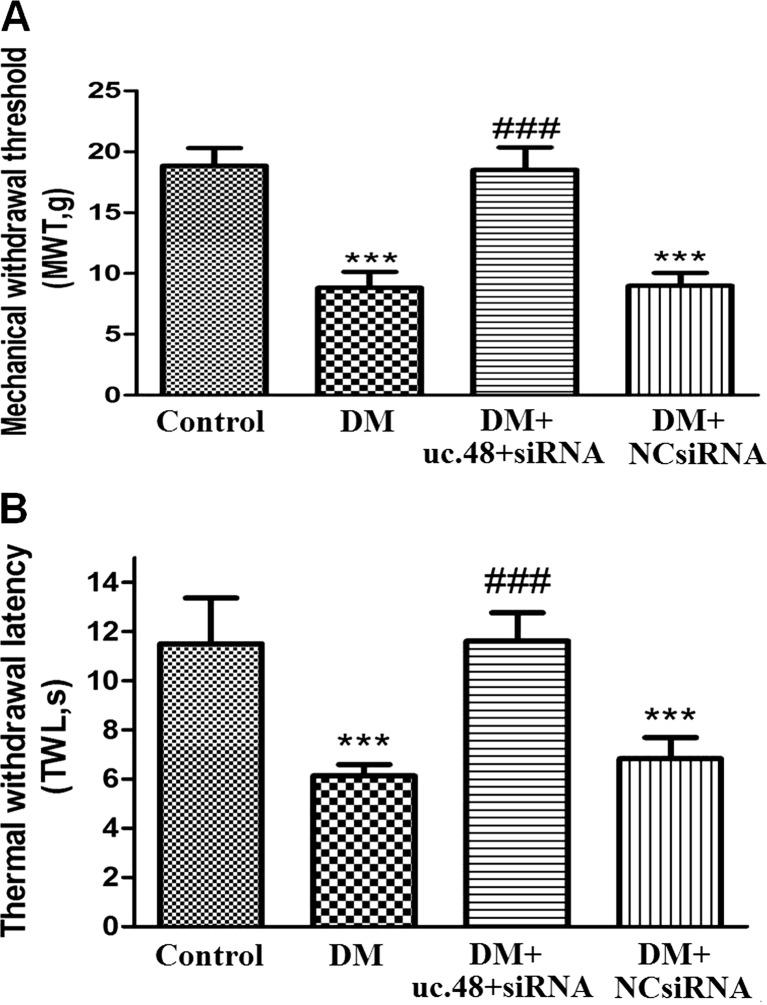
Effects of uc.48+ siRNA on mechanical or thermal hyperalgesia in DM rats
The mechanical withdrawal threshold (MWT) was measured. The MWT in the DM group was decreased by 50 % compared with that in the control group (p < 0.001). The MWT in DM rats treated with uc.48+ siRNA was twofold higher than that in the DM rats (p < 0.001) (Fig. 2a). There was no difference between the DM group and DM + NC siRNA group (p > 0.05). The above results showed that uc.48+ siRNA inhibited pain behaviors by increasing the mechanical hyperalgesia threshold in DM rats.
Fig. 2.
Effect of uc.48+ siRNA on the mechanical withdrawal threshold or thermal withdrawal latency in DM rats. a The mechanical withdrawal threshold (MWT) was measured. The MWT in DM rats treated with uc.48+ siRNA was higher than that in DM rats (p < 0.001). There was no difference between the DM group and DM + scramble siRNA group (DM + negative control siRNA: DM + NC si group) (p > 0.05). The upper limit of mechanical withdrawal threshold (MWT) detection was 26.0 g. Mean ± SEM, n = 8. ***p < 0.001 compared to the control group; ### p < 0.001 compared to the DM group. b The thermal withdrawal latency (TWL) was measured. The TWL in DM rats treated with uc.48+ siRNA was higher than that in DM rats (p < 0.001). There was no difference between the DM group and DM + scramble siRNA group (DM + NC si group) (p > 0.05). The upper limit of the thermal withdrawal latency detection was 30 s. Mean ± SEM, n = 8. ***p < 0.001 compared to the control group; ### p < 0.01 compared to the DM group
The thermal withdrawal latency (TWL) was also measured. The TWL in the DM group was decreased by 46 % compared to that in the control group (p < 0.001). The TWL in DM rats treated with uc.48+ siRNA was increased by 46.5 % compared with that in the DM rats (p < 0.001) (Fig. 2b). There was no difference between the DM group and DM + NC siRNA group (p > 0.05). The above results revealed that uc.48+ siRNA treatment also elevated the thermal hyperalgesia threshold in DM rats, suggesting that uc.48+ siRNA treatment may relieve neuropathic pain.
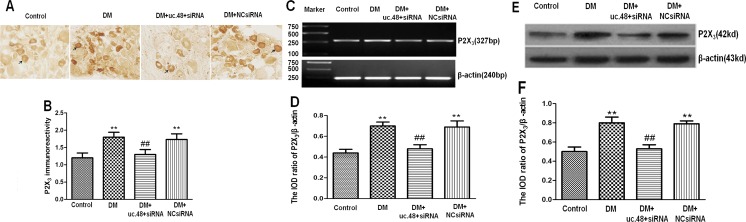
Effects of uc.48+ siRNA on the expression levels of P2X3 immunoreactivity, mRNA, or protein in the DRG of DM rats
The immunoreactivities of the P2X3 receptor in the DRG were detected using immunohistochemistry. Image analysis revealed that the intensity values (integrated optical density) of P2X3 expression in the DM group were increased by 1.6-fold compared to those in the control group (p < 0.01, n = 8 for each group). The intensity values of P2X3 expression in DM rats treated with uc.48+ siRNA were significantly decreased by 1.5-fold compared to those in the rats in the DM group (p < 0.01, n = 8 for each group). No difference in the intensity of DRG P2X3 expression was found between the DM group and DM + scramble siRNA group (p > 0.05, n = 8 for each group) (Fig. 3a, b). These results indicated that uc.48+ siRNA treatment might decrease the expression of DRG P2X3 immunoreactivities in DM rats.
Fig. 3.
Effect of uc.48+ siRNA on the expression of P2X3 immunoreactivity, mRNA, or protein in the DRG of DM rats. a, b P2X3 immunoreactivity in the DRG was detected using immunohistochemistry. Image analysis revealed that P2X3 expression (integrated optical density) in the DM group was higher than that in the control group (p < 0.01, n = 8 for each group). The P2X3 expression in DM rats treated with uc.48+ siRNA was significantly lower than that in the DM group. There were no differences in P2X3 expression between the DM group and DM + scramble siRNA group (DM + NC si group). Mean ± SEM, n = 8. **p < 0.01 compared to the control group; ## p < 0.01 compared to the DM group. c, d The expression of P2X3 mRNA was examined using RT-PCR. Image analysis revealed that the levels (integrated optical density) of P2X3 mRNA (normalized to each β-actin internal control) in the DM group were higher than those in the control group. The levels of P2X3 mRNA in DM rats treated with uc.48+ siRNA were significantly decreased compared to those in the DM group. There were no differences in the levels of P2X3 mRNA between the DM group and DM + scramble siRNA group (DM + NC si group) (p > 0.05). Mean ± SEM, n = 8. **p < 0.01 compared to the control group; ## p < 0.01 compared to the DM group. e, f The expression of P2X3 protein in the DRG was examined using western blotting analyses. Image analysis revealed that the levels (integrated optical density) of P2X3 protein (normalized to each β-actin internal control) in the DM group were higher than those in the control group. The levels of P2X3 protein in DM rats treated with uc.48+ siRNA were significantly lower than those in the DM group. There were no differences in the levels of P2X3 protein between the DM group and DM + scramble siRNA group (DM + NC si group). Mean ± SEM, n = 8. **p < 0.01 compared to the control group; ## p < 0.01 compared to the DM group
The expression levels of P2X3 mRNA in the DRG of each group were studied by RT-PCR. Using image analysis, the levels (average optical density) of P2X3 mRNA in the DM group were about 1.7-fold higher than those in the control group (p < 0.05, n = 8 for each group). The levels of P2X3 mRNA in DM rats treated with uc.48+ siRNA were significantly decreased by 1.5-fold compared to those in the DM group (p < 0.05, n = 8 for each group). There were no differences in the expression of P2X3 mRNA when comparing the DRG from the DM group and DM + scramble siRNA group (p > 0.05, n = 8 for each group) (Fig. 3c, d). These results indicate that uc.48+ siRNA treatment may reduce the upregulated expression of P2X3 mRNA in the DRG of DM rats.
The expression of P2X3 protein in the DRG was further studied by western blotting. Using image analysis, the levels (average optical density) of P2X3 protein (normalized to each β-actin internal control) in the DM group were about 1.6-fold higher than those in the control group (p < 0.05, n = 8 for each group). The levels of P2X3 protein in DM rats treated with uc.48+ siRNA were about 1.6-fold lower than those in the DM group (p < 0.05, n = 8 for each group). There were no differences in the expression of P2X3 protein when comparing the DRG from the DM group and DM + scramble siRNA group (p > 0.05, n = 8 for each group) (Fig. 3e, f). These results reveal that uc.48+ siRNA treatment may decrease the expression of DRG P2X3 protein in DM rats.
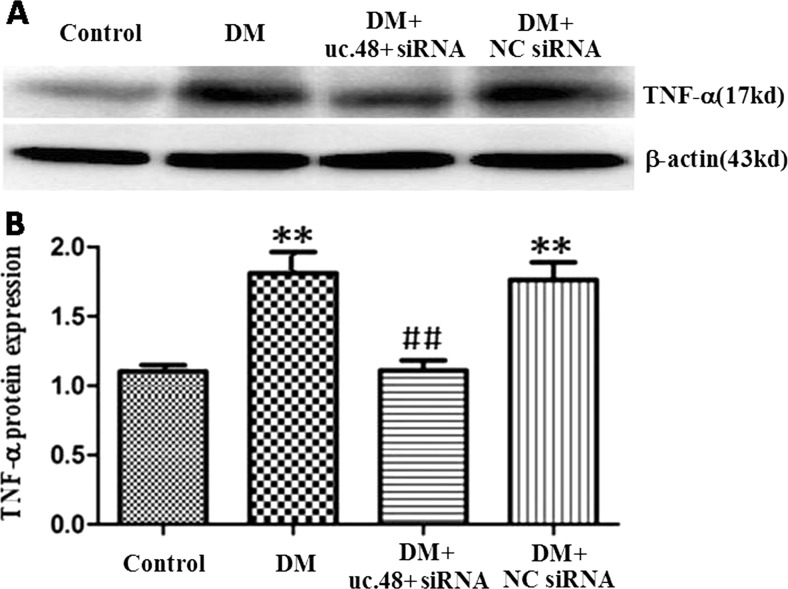
Effects of uc.48+ siRNA on the expression of TNF-α protein in the DRG of DM rats
The levels of TNF-α protein in the DRG were analyzed by western blotting. Using image analysis, the levels of TNF-α protein (normalized to each β-actin internal control) in the DM group were significantly augmented by 1.6-fold compared to those in the control group (p < 0.01). The relative levels of TNF-α protein in the DM + uc.48+ siRNA group were 1.6-fold lower than those in the DM group (p < 0.01) (Fig. 4). There were no differences in the expression of TNF-α protein when comparing the DRG from the DM group and DM + scramble siRNA group (p > 0.05, n = 8 for each group) (Fig. 4).
Fig. 4.
a, b Effects of uc.48+ siRNA on the expression of TNF-α protein in the DRG of DM rats. Image analysis revealed that the levels (integrated optical density) of TNF-α protein (normalized to each β-actin internal control) in the DM group were higher than those in the control group. The levels of TNF-α protein in DM rats treated with uc.48+ siRNA were significantly lower than those in the DM group. There were no differences in TNF-α protein expression between the DRG from the DM group and DM + NC si group. Mean ± SEM, n = 8. **p < 0.01 compared to the control group; ## p < 0.01 compared to the DM group
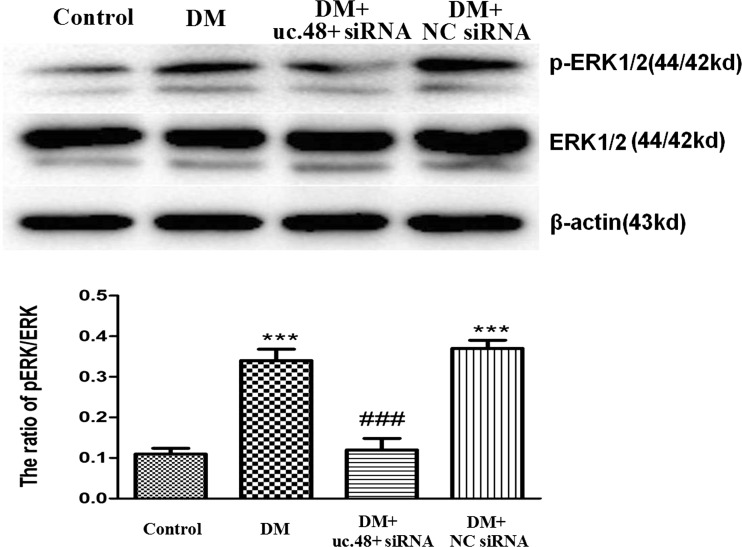
Effects of uc.48+ siRNA on the levels of ERK1/2 and p-ERK1/2 in the DRG of DM rats
The phosphorylation and activation of ERK1/2 participated in neuropathic pain. The levels of ERK1/2 and p-ERK1/2 in the DRG were analyzed by western blotting. The integrated optical density (IOD) ratio of ERK1/2 to β-actin was not significantly different between the four groups (p > 0.05). However, the IOD ratio of p-ERK1/2 to ERK1/2 (n = 8) in the DM group was 3.5-fold higher than that in the control group (p < 0.001, n = 8 for each group) (Fig. 5). The results indicated that the role of ERK phosphorylation in the DRG was related to the P2X3 receptor-mediated hyperalgesia in DM rats.
Fig. 5.
Effects of uc.48+ siRNA on the levels of ERK1/2 and p-ERK1/2 in the DRG of DM rats. The levels of ERK1/2 and p-ERK1/2 in the DRG were analyzed using western blotting analyses. The integrated optical density (IOD) ratio of ERK1/2 to β-actin was not significantly different between the four groups (p > 0.05). The IOD ratio of p-ERK1/2 to ERK in the DM group was higher than that in the control group. The IOD ratio of p-ERK1/2 to ERK in the DM rats treated with uc.48+ siRNA was significantly lower than that in the DM group. There was no difference in the IOD ratio of p-ERK1/2 to ERK between the DM group and DM + scramble siRNA group. Mean ± SEM, n = 8. ***p < 0.001 compared to the control group; ### p < 0.001 compared to the DM group
In addition, we examined whether the administration of uc.48+ siRNA could affect the phosphorylation of ERK in the DM DRG. The IOD ratio of p-ERK1/2 to ERK1/2 in the DM rats treated with uc.48+ siRNA was significantly reduced by 3.5-fold compared to that in the DM group (p < 0.001, n = 8 for each group). There were no differences in the IOD ratio of p-ERK1/2 to ERK1/2 between the DM group and DM + scramble siRNA group (p > 0.05, n = 8 for each group) (Fig. 5). Taken together, these results suggest that uc.48+ siRNA treatment may contribute to decreasing the phosphorylation and activation of ERK1/2 in the DRG of DM rats to alleviate P2X3 receptor-mediated hyperalgesia.
Discussion
Up to 90 % of eukaryotic genomes are transcribed, generating a large population of non-coding RNAs (ncRNAs) [11, 13]. The functions of most lncRNAs are not known; there is evidence that some lncRNAs have roles in physiological processes. The reports showed that the dysregulated expression of lncRNAs participated in the onset and progression of many pathological conditions [18–21]. In this study, the results showed that the expression of lncRNA uc.48+ in the DM DRG was much higher than that in the control. It suggested that lncRNA uc.48+ was involved in the pathophysiological process of DM DRG. The results also showed that the concentration levels of serum uc.48+ in T2DM patients with DNP complication were much higher compared to those in control healthy subjects. This finding further made sure that uc.48+ might be related to the pathological changes of T2DM. LncRNAs are being revealed as important components of gene regulatory networks [23–28]. It was reported that lncRNAs may control protein targeting to genomic loci and epigenetic silencing and may serve as scaffolds for multiple proteins [28]. It is not clear whether there is a protein target for lncRNA uc.48+ in the pathophysiological process of DM DRG.
A common phenotype of diabetic peripheral neuropathy is DNP [6, 49, 50]. DNP predominantly involves the lower limbs, specifically the foot, and in some cases, the upper extremities may be involved, including the fingertips and palms [5, 6, 9, 51, 52]. This study demonstrated that the threshold values of mechanical and thermal pain sensitivity in the DM rat models were decreased compared to those in the control rats. These results indicate that thermal hyperalgesia and abnormal mechanical hyperalgesia are enhanced in DM rats following the diabetic peripheral neuropathic lesions. In addition, the MWT and TWL in DM rats treated with uc.48+ siRNA were significantly higher than those in the DM model rats. The results suggested that uc.48+ siRNA treatment has an inhibitory action on mechanical and thermal hyperalgesia in DM rats. What is the mechanism by which uc.48+ siRNA treatment produces an inhibitory action on mechanical and thermal hyperalgesia in the DM rats?
In a genome-wide analysis of gene expression in STZ-induced diabetic rats, the confirmed gene changes were associated with inflammation [5, 6]. DM is often accompanied by a low-grade inflammation. ATP increases inflammatory-mediated pain [29–35]. The P2X3 receptor is expressed in the small-diameter primary afferent neurons and has an important role in pain transmission [30, 31, 35–39]. Studies have also shown that damage to peripheral nerves in the neuropathic pain state can increase the expression of the P2X3 receptor [30, 31, 35–37]. Our experiments showed that the expression of P2X3 protein and mRNA in the DM DRG is increased compared to controls. This is similar to other reports, which showed that the P2X3 receptor mediated mechanical allodynia in diabetic neuropathic rats [41, 42]. Meanwhile, our studies also revealed that the expression levels of P2X3 protein and mRNA in DM rats treated with lncRNA uc.48+ siRNA were significantly decreased in the DRG compared to DM rats. Therefore, uc.48+ siRNA treatment may relieve the P2X3 receptor-mediated DNP in DRG primary sensory neurons.
Pro-inflammatory cytokines, such as TNF-α, have an important role in the initiation and maintenance of neuropathic pain [53]. Our results showed that the expression of TNF-α in the DRG of DM rats was increased compared to that in the control group. Previous reports indicated that TNF-α potentiated P2X3 receptor-mediated pain sensations [54]. The expression of TNF-α in the DRG of DM rats treated with uc.48+ siRNA was significantly decreased compared to that in the DM group. Pro-inflammatory cytokines exacerbate mechanical and thermal hyperalgesia in the DM rats. The expression of TNF-α in the DRG of DM rats was decreased by uc.48+ siRNA treatment, which relieved P2X3 receptor-mediated DNP in DM rats. Activation of the ERK pathway is involved in P2X receptor-mediated pain transmission [54, 55]. Our results revealed that the IOD ratio of p-ERK1/2 to ERK1/2 in the DRG of the DM group was higher compared to that in the control group. After the administration of uc.48+ siRNA, the IOD ratio of p-ERK1/2 to ERK1/2 in DM rats treated with uc.48+ siRNA was significantly decreased compared to that in the DM group. Thus, uc.48+ siRNA treatment may decrease the phosphorylation and activation of ERK1/2 in the DRG of DM rats and then relieve P2X3 receptor-mediated hyperalgesia.
Conclusions
In summary, uc.48+ siRNA treatment decreased the upregulated expression of the P2X3 receptor and the phosphorylation and activation of ERK1/2 in the DM DRG. Then, the thermal and mechanical hyperalgesia in DM rats was decreased. The results indicate that uc.48+ siRNA treatment may alleviate the DNP by inhibiting the excitatory transmission mediated by the P2X3 receptor in DRG primary sensory neurons.
Acknowledgments
This study was supported by grants (nos. 31560276, 81570735, 81560219, 81171184, 31060139, 81360136, 81360140, 81560529, and 81200853) from the National Natural Science Foundation of China, a grant (no. 20151122040105) from the Technology Pedestal and Society Development Project of Jiangxi Province, a grant (no. 20142BAB205028) from the Natural Science Foundation of Jiangxi Province, and grants (nos. GJJ13155 and GJJ14319) from the Educational Department of Jiangxi Province.
Compliance with ethical standards
The procedures were approved by the Animal Care and Use Committee of Nanchang University Medical School. The IASP’s ethical guidelines for pain research in animals were followed. All animals were treated in accordance with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research in China. Following local ethics committee approval, written informed consent was obtained from each individual.
Conflict of interest
The authors declare that they have no competing interests.
Footnotes
Shouyu Wang and Hong Xu are joint first authors.
References
- 1.Pruimboom L, van Dam AC. Chronic pain: a non-use disease. Med Hypotheses. 2007;68:506–511. doi: 10.1016/j.mehy.2006.08.036. [DOI] [PubMed] [Google Scholar]
- 2.Schmader KE, Baron R, Haanpaa ML, Mayer J, O’Connor AB, Rice AS, Stacey B. Treatment considerations for elderly and frail patients with neuropathic pain. Mayo Clin Proc. 2010;85:S26–S32. doi: 10.4065/mcp.2009.0646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Treede RD, Jensen TS, Campbell JN, Cruccu G, Dostrovsky JO, Griffin JW, Hansson P, Hughes R, Nurmikko T, Serra J. Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology. 2008;70:1630–1635. doi: 10.1212/01.wnl.0000282763.29778.59. [DOI] [PubMed] [Google Scholar]
- 4.Whiting DR, Guariguata L, Weil C, Shaw J. IDF diabetes atlas: global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res Clin Pract. 2011;94:311–321. doi: 10.1016/j.diabres.2011.10.029. [DOI] [PubMed] [Google Scholar]
- 5.Callaghan BC, Cheng HT, Stables CL, Smith AL, Feldman EL. Diabetic neuropathy: clinical manifestations and current treatments. Lancet Neurol. 2012;11:521–534. doi: 10.1016/S1474-4422(12)70065-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Schreiber AK, Nones CF, Reis RC, Chichorro JG, Cunha JM. Diabetic neuropathic pain: physiopathology and treatment. World J Diabetes. 2015;6:432–444. doi: 10.4239/wjd.v6.i3.432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Obrosova IG. Diabetes and the peripheral nerve. Biochim Biophys Acta. 2009;1792:931–940. doi: 10.1016/j.bbadis.2008.11.005. [DOI] [PubMed] [Google Scholar]
- 8.Tesfaye S, Boulton AJ, Dyck PJ, Freeman R, Horowitz M, Kempler P, Lauria G, Malik RA, Spallone V, Vinik A, Bernardi L, Valensi P. Diabetic neuropathies: update on definitions, diagnostic criteria, estimation of severity, and treatments. Diabetes Care. 2010;33:2285–2293. doi: 10.2337/dc10-1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davies M, Brophy S, Williams R, Taylor A. The prevalence, severity, and impact of painful diabetic peripheral neuropathy in type 2 diabetes. Diabetes Care. 2006;29:1518–1522. doi: 10.2337/dc05-2228. [DOI] [PubMed] [Google Scholar]
- 10.Tavakoli M, Malik RA. Management of painful diabetic neuropathy. Expert Opin Pharmacother. 2008;9:2969–2978. doi: 10.1517/14656560802498149. [DOI] [PubMed] [Google Scholar]
- 11.Ponting CP, Belgard TG. Transcribed dark matter: meaning or myth? Hum Mol Genet. 2010;19:R162–R168. doi: 10.1093/hmg/ddq362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Stein LD. Human genome: end of the beginning. Nature. 2004;431:915–916. doi: 10.1038/431915a. [DOI] [PubMed] [Google Scholar]
- 13.Costa FF. Non-coding RNAs: meet thy masters. Bioessays. 2010;32:599–608. doi: 10.1002/bies.200900112. [DOI] [PubMed] [Google Scholar]
- 14.Bartel DP. MicroRNAs: target recognition and regulatory functions. Cell. 2009;136:215–233. doi: 10.1016/j.cell.2009.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chitwood DH, Timmermans MC. Small RNAs are on the move. Nature. 2010;467:415–419. doi: 10.1038/nature09351. [DOI] [PubMed] [Google Scholar]
- 16.Louro R, Smirnova AS, Verjovski-Almeida S. Long intronic noncoding RNA transcription: expression noise or expression choice? Genomics. 2009;93:291–298. doi: 10.1016/j.ygeno.2008.11.009. [DOI] [PubMed] [Google Scholar]
- 17.Ponjavic J, Ponting CP, Lunter G. Functionality or transcriptional noise? Evidence for selection within long noncoding RNAs. Genome Res. 2007;17:556–565. doi: 10.1101/gr.6036807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Batista PJ, Chang HY. Long noncoding RNAs: cellular address codes in development and disease. Cell. 2013;152:1298–1307. doi: 10.1016/j.cell.2013.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Di Gesualdo F, Capaccioli S, Lulli M. A pathophysiological view of the long non-coding RNA world. Oncotarget. 2014;5:10976–10996. doi: 10.18632/oncotarget.2770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Qureshi IA, Mehler MF. Long non-coding RNAs: novel targets for nervous system disease diagnosis and therapy. Neurotherapeutics. 2013;10:632–646. doi: 10.1007/s13311-013-0199-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wapinski O, Chang HY. Long noncoding RNAs and human disease. Trends Cell Biol. 2011;21:354–361. doi: 10.1016/j.tcb.2011.04.001. [DOI] [PubMed] [Google Scholar]
- 22.Sauvageau M, Goff LA, Lodato S, Bonev B, Groff AF, Gerhardinger C, Sanchez-Gomez DB, Hacisuleyman E, Li E, Spence M, Liapis SC, Mallard W, Morse M, Swerdel MR, D’Ecclessis MF, Moore JC, Lai V, Gong G, Yancopoulos GD, Frendewey D, Kellis M, Hart RP, Valenzuela DM, Arlotta P, Rinn JL. Multiple knockout mouse models reveal lincRNAs are required for life and brain development. Elife. 2013;2:e1749. doi: 10.7554/eLife.01749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guil S, Esteller M. RNA-RNA interactions in gene regulation: the coding and noncoding players. Trends Biochem Sci. 2015;40:248–256. doi: 10.1016/j.tibs.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 24.Hu W, Alvarez-Dominguez JR, Lodish HF. Regulation of mammalian cell differentiation by long non-coding RNAs. EMBO Rep. 2012;13:971–983. doi: 10.1038/embor.2012.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kaikkonen MU, Lam MT, Glass CK. Non-coding RNAs as regulators of gene expression and epigenetics. Cardiovasc Res. 2011;90:430–440. doi: 10.1093/cvr/cvr097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ponting CP, Oliver PL, Reik W. Evolution and functions of long noncoding RNAs. Cell. 2009;136:629–641. doi: 10.1016/j.cell.2009.02.006. [DOI] [PubMed] [Google Scholar]
- 27.Rapicavoli NA, Qu K, Zhang J, Mikhail M, Laberge RM, Chang HY. A mammalian pseudogene lncRNA at the interface of inflammation and anti-inflammatory therapeutics. Elife. 2013;2:e762. doi: 10.7554/eLife.00762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rinn JL, Chang HY. Genome regulation by long noncoding RNAs. Annu Rev Biochem. 2012;81:145–166. doi: 10.1146/annurev-biochem-051410-092902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Burnstock G. Pathophysiology and therapeutic potential of purinergic signaling. Pharmacol Rev. 2006;58:58–86. doi: 10.1124/pr.58.1.5. [DOI] [PubMed] [Google Scholar]
- 30.Burnstock G. Physiology and pathophysiology of purinergic neurotransmission. Physiol Rev. 2007;87:659–797. doi: 10.1152/physrev.00043.2006. [DOI] [PubMed] [Google Scholar]
- 31.Burnstock G. Purinergic receptors and pain. Curr Pharm Des. 2009;15:1717–1735. doi: 10.2174/138161209788186335. [DOI] [PubMed] [Google Scholar]
- 32.Burnstock G. Purinergic signalling: from discovery to current developments. Exp Physiol. 2014;99:16–34. doi: 10.1113/expphysiol.2013.071951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gao Y, Xu C, Liang S, Zhang A, Mu S, Wang Y, Wan F. Effect of tetramethylpyrazine on primary afferent transmission mediated by P2X3 receptor in neuropathic pain states. Brain Res Bull. 2008;77:27–32. doi: 10.1016/j.brainresbull.2008.02.026. [DOI] [PubMed] [Google Scholar]
- 34.Gao Y, Liu H, Deng L, Zhu G, Xu C, Li G, Liu S, Xie J, Liu J, Kong F, Wu R, Li G, Liang S. Effect of emodin on neuropathic pain transmission mediated by P2X2/3 receptor of primary sensory neurons. Brain Res Bull. 2011;84:406–413. doi: 10.1016/j.brainresbull.2011.01.017. [DOI] [PubMed] [Google Scholar]
- 35.Liang S, Xu C, Li G, Gao Y. P2X receptors and modulation of pain transmission: focus on effects of drugs and compounds used in traditional Chinese medicine. Neurochem Int. 2010;57:705–712. doi: 10.1016/j.neuint.2010.09.004. [DOI] [PubMed] [Google Scholar]
- 36.Burnstock G. Purinergic P2 receptors as targets for novel analgesics. Pharmacol Ther. 2006;110:433–454. doi: 10.1016/j.pharmthera.2005.08.013. [DOI] [PubMed] [Google Scholar]
- 37.Novakovic SD, Kassotakis LC, Oglesby IB, Smith JA, Eglen RM, Ford AP, Hunter JC. Immunocytochemical localization of P2X3 purinoceptors in sensory neurons in naive rats and following neuropathic injury. Pain. 1999;80:273–282. doi: 10.1016/S0304-3959(98)00225-5. [DOI] [PubMed] [Google Scholar]
- 38.Ueno S, Moriyama T, Honda K, Kamiya H, Sakurada T, Katsuragi T. Involvement of P2X2 and P2X3 receptors in neuropathic pain in a mouse model of chronic constriction injury. Drug Dev Res. 2003;59:104–111. doi: 10.1002/ddr.10208. [DOI] [Google Scholar]
- 39.Zhang A, Gao Y, Zhong X, Xu C, Li G, Liu S, Lin J, Li X, Zhang Y, Liu H, Linag S. Effect of sodium ferulate on the hyperalgesia mediated by P2X3 receptor in the neuropathic pain rats. Brain Res. 2010;1313:215–221. doi: 10.1016/j.brainres.2009.11.067. [DOI] [PubMed] [Google Scholar]
- 40.Lin J, Li G, Den X, Xu C, Liu S, Gao Y, Liu H, Zhang J, Li X, Liang S. VEGF and its receptor-2 involved in neuropathic pain transmission mediated by P2X(2)(/)(3) receptor of primary sensory neurons. Brain Res Bull. 2010;83:284–291. doi: 10.1016/j.brainresbull.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 41.Burnstock G, Novak I. Purinergic signalling and diabetes. Purinergic Signal. 2013;9:307–324. doi: 10.1007/s11302-013-9359-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Xu GY, Li G, Liu N, Huang LY. Mechanisms underlying purinergic P2X3 receptor-mediated mechanical allodynia induced in diabetic rats. Mol Pain. 2011;7:60. doi: 10.1186/1744-8069-7-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hanani M, Blum E, Liu S, Peng L, Liang S. Satellite glial cells in dorsal root ganglia are activated in streptozotocin-treated rodents. J Cell Mol Med. 2014;18:2367–2371. doi: 10.1111/jcmm.12406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Messinger RB, Naik AK, Jagodic MM, Nelson MT, Lee WY, Choe WJ, Orestes P, Latham JR, Todorovic SM, Jevtovic-Todorovic V. In vivo silencing of the Ca(V)3.2 T-type calcium channels in sensory neurons alleviates hyperalgesia in rats with streptozocin-induced diabetic neuropathy. Pain. 2009;145:184–195. doi: 10.1016/j.pain.2009.06.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu H, Wu B, Jiang F, Xiong S, Zhang B, Li G, Liu S, Gao Y, Xu C, Tu G, Peng H, Liang S, Xiong H. High fatty acids modulate P2X(7) expression and IL-6 release via the p38 MAPK pathway in PC12 cells. Brain Res Bull. 2013;94:63–70. doi: 10.1016/j.brainresbull.2013.02.002. [DOI] [PubMed] [Google Scholar]
- 46.Liu J, Li G, Peng H, Tu G, Kong F, Liu S, Gao Y, Xu H, Qiu S, Fan B, Zhu Q, Yu S, Zheng C, Wu B, Peng L, Song M, Wu Q, Li G, Liang S. Sensory-sympathetic coupling in superior cervical ganglia after myocardial ischemic injury facilitates sympathoexcitatory action via P2X7 receptor. Purinergic Signal. 2013;9:463–479. doi: 10.1007/s11302-013-9367-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gunduz O, Oltulu C, Buldum D, Guven R, Ulugol A. Anti-allodynic and anti-hyperalgesic effects of ceftriaxone in streptozocin-induced diabetic rats. Neurosci Lett. 2011;491:23–25. doi: 10.1016/j.neulet.2010.12.063. [DOI] [PubMed] [Google Scholar]
- 48.Xu C, Xu W, Xu H, Xiong W, Gao Y, Li G, Liu S, Xie J, Tu G, Peng H, Qiu S, Liang S. Role of puerarin in the signalling of neuropathic pain mediated by P2X3 receptor of dorsal root ganglion neurons. Brain Res Bull. 2012;87:37–43. doi: 10.1016/j.brainresbull.2011.10.007. [DOI] [PubMed] [Google Scholar]
- 49.Barrett AM, Lucero MA, Le T, Robinson RL, Dworkin RH, Chappell AS. Epidemiology, public health burden, and treatment of diabetic peripheral neuropathic pain: a review. Pain Med. 2007;8(Suppl 2):S50–S62. doi: 10.1111/j.1526-4637.2006.00179.x. [DOI] [PubMed] [Google Scholar]
- 50.Tesfaye S, Selvarajah D. Advances in the epidemiology, pathogenesis and management of diabetic peripheral neuropathy. Diabetes Metab Res Rev. 2012;28(Suppl- 1):8–14. doi: 10.1002/dmrr.2239. [DOI] [PubMed] [Google Scholar]
- 51.Galer BS, Gianas A, Jensen MP. Painful diabetic polyneuropathy: epidemiology, pain description, and quality of life. Diabetes Res Clin Pract. 2000;47:123–128. doi: 10.1016/S0168-8227(99)00112-6. [DOI] [PubMed] [Google Scholar]
- 52.Ziegler D. Painful diabetic neuropathy: advantage of novel drugs over old drugs? Diabetes Care. 2009;32(Suppl 2):S414–S419. doi: 10.2337/dc09-S350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Stein C, Clark JD, Oh U, Vasko MR, Wilcox GL, Overland AC, Vanderah TW, Spencer RH. Peripheral mechanisms of pain and analgesia. Brain Res Rev. 2009;60:90–113. doi: 10.1016/j.brainresrev.2008.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Burnstock G, Krugel U, Abbracchio MP, Illes P. Purinergic signalling: from normal behaviour to pathological brain function. Prog Neurobiol. 2011;95:229–274. doi: 10.1016/j.pneurobio.2011.08.006. [DOI] [PubMed] [Google Scholar]
- 55.Seino D, Tokunaga A, Tachibana T, Yoshiya S, Dai Y, Obata K, Yamanaka H, Kobayashi K, Noguchi K. The role of ERK signaling and the P2X receptor on mechanical pain evoked by movement of inflamed knee joint. Pain. 2006;123:193–203. doi: 10.1016/j.pain.2006.02.032. [DOI] [PubMed] [Google Scholar]