Abstract
Although human malignant mesothelioma (HMM) is mainly caused by asbestos exposure, refractory ceramic fibres (RCFs) have been classified as possibly carcinogenic to humans on the basis of their biological effects in rodents’ lung and pleura and in cultured cells. Hence, further investigations are needed to clarify the mechanism of fibre-induced carcinogenicity and to prevent use of harmful particles. In a previous study, mesotheliomas were found in hemizygous Nf2 (Nf2+/−) mice exposed to asbestos fibres, and showed similar alterations in genes at the Ink4 locus and in Trp53 as described in HMM. Here we found that Nf2+/− mice developed mesotheliomas after intra-peritoneal inoculation of a RCF sample (RCF1). Clinical features in exposed mice were similar to those observed in HMM, showing association between ascite and mesothelioma. Early passages of 12 mesothelioma cell cultures from ascites developed in RCF1-exposed Nf2+/− mice demonstrated frequent inactivation by deletion of genes at the Ink4 locus, and low rate of Trp53 point and insertion mutations. Nf2 gene was inactivated in all cultures. In most cases, co-inactivation of genes at the Ink4 locus and Nf2 was found and, at a lower rate, of Trp53 and Nf2. These results are the first to identify mutations in RCF-induced mesothelioma. They suggest that nf2 mutation is complementary of p15Ink4b, p16Ink4a and p19Arf or p53 mutations and show similar profile of gene alterations resulting from exposure to ceramic or asbestos fibres in Nf2+/− mice, also consistent with the one found in HMM. These somatic genetic changes define different pathways of mesothelial cell transformation.
Keywords: Animals; Ascites; Mineral Fibers; Neurofibromin 2; Tumor Suppressor Protein p53; Cells, Cultured; Ceramics; Cyclin-Dependent Kinase Inhibitor Proteins; Cyclin-Dependent Kinase Inhibitor p16; Disease Models, Animal; Mesothelioma; Mice; Mice, Knockout
Introduction
In humans, malignant mesothelioma is caused by exposure to natural mineral fibres, asbestos and erionite (1,2). Despite widespread use of synthetic man-made fibres, especially asbestos fibres’ substitutes developed following asbestos ban, no occurrence of mesothelioma has been found in workers exposed to man-made fibres (3). Only benign pleural pathology has been recently observed in a morbidity study carried out at European refractory ceramic fibre (RCF) plants, enrolling workers involved in the production of ceramic fibres and associated activities (4). Actually, it may presently remains difficult to establish whether there is a link between ceramic fibre exposure and human malignant mesothelioma (HMM), as most workers have been previously exposed to asbestos fibres, and duration of exposure to man-made fibres alone is still limited in view of the long latency period associated with the occurrence of mesothelioma. Nevertheless, RCFs have been recently classified as possibly carcinogenic to humans (group 2B) by International Agency for Research on Cancer, regarding the induction of lung cancer in rats and pleural mesotheliomas in hamsters in nose-only long-term experiments (3,5–7). In addition, genetic and related effects have been described in cultured cells such as micronucleus formation, chromosome breakage, induction of DNA damage and production of reactive oxygen species (8–13). These observations encourage researches to clarify the mechanism of fibre-induced carcinogenicity and to prevent use of harmful particles. To that purpose, determination of the biological features of RCFs-induced mesothelioma in comparison with those of mesotheliomas resulting from exposure to other factors is of great interest.
Several studies have emphasized the differences in mutational pattern of mesotheliomas depending on the type of carcinogen (14–17), and mutation frequencies and mutation patterns appear to discriminate between carcinogens in other types of tumours both in mice and in humans (18–20). In a previous paper, we reported a model of mesothelioma supported by the generation of mesothelioma in mice harbouring hemizygosity in the Nf2 gene (Nf2+/− mice), as inactivation of this gene has been reported in HMM (2,21). When these mice were exposed to asbestos fibres (asb-Nf2+/− mice), they developed mesotheliomas having histological and genetic characteristics similar to those of HMM (22). That is, epithelial, fusiform and biphasic type mesotheliomas were found, and both frequency and type of genetic alterations in tumour suppressor genes Trp53, p16/Cdkn2a, p15/ Cdkn2b, p19/Arf and Nf2 were similar to those reported in orthologous human genes in mesothelioma (23). The aim of the present work was to determine whether RCFs produce mesotheliomas in Nf2+/− mice and to characterize mesothelioma cells. We found that histology of mesotheliomas developed in mice following exposure to RCFs (ceram-Nf2+/− mice) was consistent with that of asbestos-induced mesothelioma but some differences were seen in the frequency of histological subtypes. The profile of genetic somatic alterations found in tumour suppressor genes in primary cultures of mesothelioma cells obtained from ascitic fluids was similar to the one detected in mesothelioma cells from asb-Nf2+/− mice.
This report is the first to identify genetic somatic changes associated to RCF-induced mesothelioma in experimental animals. Discrimination between asbestos-induced and ceramic fibre-induced mesotheliomas cannot be made on the basis of histological features, and genetic alterations remain closely similar suggesting that some features of mesothelioma might be more specifically related to the effects of fibres.
Materials and methods
Animal experiment
Heterozygous (Nf2+/−) mice in the FVB/N background, knockout in exon 3 in one allele of the Nf2 gene, were exposed to RCFs according to the same procedure as used with crocidolite fibres (22,24). RCF sample (RCF1) was kindly provided by Thermal Insulation Manufacturer’s Association (TIMA) Inc. (Stamford, CT, USA). The mean arithmetic length and diameter were 22.4 ± 19.0 μm and 1.1 ± 0.8 μm, respectively, according to TIMA data sheets. Fibres were suspended in sterile 0.9% NaCl and homogenized by sonication as described previously. Intraperitoneal inoculation consisted in 300 μl injections of saline or fibres in control (ctrl-Nf2+/−) and ceram-Nf2+/− groups, respectively. Three milligrams of RCF1 were inoculated in two injections in a lag time of 2 months. There were 55 ceram-Nf2+/− mice in the RCF1-exposed groups and 33 ctrl-Nf2+/− mice in the saline group.
Clinicopathology
Mice were observed for clinical signs, morbidity and mortality throughout the study. Animals were euthanized by CO2 inhalation (25) when illness signs were detected. Those were cachexia, dyspnea, cyanosis (nose and tail), large increase in abdomen volume or apparent tumour. All the killed animals were systematically necropsied. Macroscopic lesions like organ adherence, nodules and ascites were noted. For histopathologic analysis, organs from the abdominal and thoracic cavities were proceeded according to standard methods. Forty-seven ceram-Nf2+/− and 28 crtl-Nf2+/− mice were available for histological investigation.
Establishment of cell cultures from ascites
When present, ascitic fluid was collected and cultured as reported previously (22). Mesothelioma cells were routinely cultured in RPMI 1640 medium with glutamax and 25 mM N-2-hydroxyethylpiperazine-N′-2-ethanesulfonic acid, supplemented with 8% fetal calf serum, 50 IU/ml penicillin and 50 mg/ml streptomycin, all from Life Technologies (Cergy Pontoise, France). Details on culturing procedure are reported in (23). All morphological and genetic analyses were carried out with cultures of <10 passages. For immunocytochemical characterization, cells were subcultured in 8-chamber Lab-Tek chamber slides (Dutcher, Strasbourg, France), and treated with relevant antibodies as described previously (23). Mesothelial cell characterization was made on the basis of co-expression of cytokeratin, vimentin and mesothelin. The following primary antibodies were used: rabbit polyclonal and mouse monoclonal antibodies against cytokeratin and vimentin, respectively, both from Dako (Trappes, France) and mesothelin (CAK1) from Signet laboratories (Eurobio, Les Ulis, France). Negative controls for cytokeratin were carried out using normal rabbit serum and normal goat serum, both from Dako, and mouse IgG1 (Dako) was used for vimentin and mesothelin.
Finally, 12 murine cell cultures were established from tumoral ascites from ceram-Nf2+/− mice (nos 129, 145, 146, 164, 170, 178, 186, 201, 225, 248, 255 and 277). The neoplastic state of these cultures was assessed by subcutaneous inoculation of 2 × 106 cells in Swiss nu/nu nude mice (Charles River Laboratories, L’Arbresle, France). All cultures produced tumours within a delay of 3–10 days after inoculation.
Polymerase chain reaction analyses of mesothelioma cell cultures from ceram-Nf2+/− mice
Genomic DNA (gDNA) was extracted from murine cell cultures as described previously (23). Polymerase chain reaction (PCR) analysis was performed with a GeneAmp 9700 apparatus (Perkin-Elmer, Courtaboeuf, France). The Nf2+ and Nf2− alleles were amplified from gDNA (1 μg) with Taq DNA polymerase (Invitrogen, Cergy-Pontoise, France) using mP1, mP2 and mP3 primers, whereas the Ink4a exons 1α (p16/Cdkn2a), 1β (p19/Arf) and 2 (common to p16/Cdkn2a and p19/Arf) were, respectively, amplified using MTS1F10A/ MTS1R10A, MTS1F4B/MTS1R5B and mCDKN2A-Ex2S/mCDKN2A-Ex2AS primers (Table I). p15/Cdkn2b (locus Ink4b) exons 1 and 2 were, respectively, amplified using mCDKN2B-Ex1S/mCDKN2B-Ex1AS and mCDKN2B-Ex2S/ mCDKN2B-Ex2AS primers (Table I). After an initial denaturation step at 94°C for 2 min, PCRs were carried out for 35 cycles including denaturation at 94°C for 30 s, annealing at the appropriate temperature for 30 s and extension at 72°C for 30 s. The final extension step was performed at 72°C for 5 min. Positive control was gDNA extracted from Mice ErythroLeukemia cell line. TCRa amplification was used as qualitative PCR control. Amplification products were analysed after migration on a 1.5% agarose gel and ethidium bromide staining.
Table I.
Nf2, p15/Cdkn2b, p16/Cdkn2a, p19/Arf, β-actin, TCRa and Gapdh genes PCR primers
| Primers and probes | Genes | Template | Sequences | Length (bp) | Tm (°C) | References |
|---|---|---|---|---|---|---|
| mP1 | Nf2 | gDNA | 5′-GCCTGCTCTTTACT-3′ | |||
| mP2 | 5′-CAGTGTGGAAGTGTTTGTGGTC-3′ | 250 (Nf2+) | 55 | (24) | ||
| mP3 | 5′-GTGTTGGATCATGATGTTTCG-3′ | 380 (Nf2−) | ||||
| mNf2S (ex1) | Nf2 | cDNA | 5′-CATGAGCTTCAGCTCACTCAAGAGGAAG-3′ | 338 (Nf2−) | 55 | (24) |
| mNf2AS (ex5) | 5′-ATCCCCGCTTGTGCACAGAGGGGTCATAG-3′ | 461 (Nf2+) | ||||
| mMTS1F10A (ex1α) | p16/Cdkn2a | gDNA | 5′-AGCGAACTCGAGGAGAGCC-3′ | |||
| mMTS1R10A (ex1α) | 5′-GGGCAGCAGCAACAACAA-3′ | 286 | 54 | (26) | ||
| mMTS1F4B (ex1β) | p19/Arf | gDNA | 5′-CGGCGCTTCTCACCTCGCTTG-3′ | |||
| mMTS1R5B (ex1β) | 5′-TCCTCTCTAGCCTCAACAACATGTT-3′ | 215 | 54 | (26) | ||
| mCDKN2A-Ex2S (ex2) | Common to p16/Cdkn2a-p19/Arf | gDNA | 5′-TCAACTACGGTGCAGATTCG-3′ | 404 | 55 | a |
| mCDKN2A-Ex2AS (intr3 | 5′-GGGTGGGTAAAATGGGAACT-3′ | |||||
| mCDKN2B-Ex1S (ex1) | p15/Cdkn2b | gDNA | 5′-GTTGGGCGGCAGCAGTGAC-3′ | 118 | 54 | a |
| mCDKN2B-Ex1AS (ex1) | 5′-CCTCCCGAAGCGGTTCAG-3′ | |||||
| mCDKN2B-Ex2S (ex2) | p15/Cdkn2b | gDNA | 5′-GTCATGATGATGGGCAGCG-3′ | 260 | 54 | a |
| mCDKN2B-Ex2AS (ex2) | 5′-CAATCTCCAGTGGCAGCGTG-3′ | |||||
| TCRA-S | TCRa | gDNA | 5′-TGACTCCCAAATCAATGTG-3′ | 136 | 53 | (27) |
| TCRA-AS | 5′-GCAGGTGAAGCTTGTCTG-3′ | |||||
| mBactin-S | β-actin | cDNA | 5′-AGAGGGAAATCGTGCGTGAC-3′ | 138 | 56 | (28) |
| mBactin-AS | 5′-CAATAGTGATGACCTGGCCGT-3′ | |||||
| Nf2-ex3Fc | 5′-TGATGTTTCGAAGGAAGAACCA-3′ | |||||
| Nf2-ex3Rc | Nf2 exon 3 | gDNA | 5′-TGAACTAGCTCCTCCTCAGCATT-3′ | 81 | 60 | b |
| Nf2-ex3Pc | VIC-5′-TTACTTTCACTTCCTGGCCAAATTT- TATCCTG-3′-TAMRA | |||||
| GAPDH-Fc | ||||||
| GAPDH-Rc | Gapdh | gDNA | Provided by Applied Biosystems | — | 60 | — |
| GAPDH-Pc |
AS: anti-sense; S: sense; ex: exon; int: intron and Tm: annealing temperature.
Primers were designed with assistance of the online software ‘Primer3’ (http://norp5424b.hsc.usc.edu/genetools.html).
Primers and probe were designed with assistance of Primer Express® ABI PRISM® (Applied Biosystems).
Primers and probes provided and used for quantitative real-time PCR carried out with a ABI PRISM® 7900HT SDS (Applied Biosystems).
Reverse transcription–PCR analyses of mesothelioma cell cultures from ceram-Nf2+/− mice
Total RNA isolated from scrapped cells and crushed kidneys was extracted using the RNA Plus extraction solution (Q-Biogene, Illkirch, France) and reverse transcribed with oligodT (Promega, Charbonnières, France) using the Superscript II RT kit (Invitrogen) according to the manufacturer’s recommendations. PCR analysis was performed with a GeneAmp 9700 apparatus (Perkin-Elmer) using 2 μl of cDNA and mNf2S/mNf2AS primers (Table I). After an initial denaturation step at 94°C for 2 min, 35 cycles were performed including steps of denaturation at 94°C for 30 s, annealing at 55°C for 30 s and extension at 72°C for 30 s. The final extension step was continued for 5 min. Controls were mRNAs extracted from kidney (K) from Nf2+/+ (K+/+) and Nf2+/− (K+/−) mice, respectively. β-Actin amplification (Stratagene, Amsterdam Zuidoost, the Netherlands) was used as a qualitative control. PCR products were analysed on a 1.5% agarose gel and visualized by ethidium bromide staining.
Mutational analysis of Trp53 in mesothelioma cell cultures ceram-Nf2+/− mice
Mutations of Trp53 exons 2–11 were screened by DNA sequencing. gDNA was extracted from cell cultures using a standard phenol–chloroform extraction procedure. DNA amplification was performed by PCR with a combination of forward and reverse primers (Table II) and Taq polymerase Hot Star (Qiagen, Courtaboeuf, France). PCR was carried out with a GeneAmp 9700 apparatus (Perkin-Elmer). After an initial denaturation step at 95°C for 10 min, PCRs were carried out for 40 cycles including denaturation at 95°C for 30 s, annealing at 58°C for 30 s and extension at 72°C for 30 s. Extension during the final step was continued for 10 min. PCR products were purified with distilled water through columns Millipore genomics, checked for quality and quantified prior sequencing. Sequencing PCR was performed on purified PCR products using forward or reverse primer located in the exon (Table II) and BigDye Terminator v1.1 cycle sequencing kit (Applied Biosystems, Courtaboeuf, France) on a GeneAmp 9700 (Perkin-Elmer). PCRs were carried out for 25 cycles including denaturation at 96°C for 10 s, annealing at 55°C for 5 s and extension at 60°C for 4 min. Sequencing PCR products were purified with distilled water through Resin Sephadex fine G50 into columns Multiscreen and analysed on an ABI PRISM 3100 Genetic analyser (Applied Biosystems). Analyses were performed using Factura and Autoassembler softwares (Applied Biosystems).
Table II.
Primers for mutational analysis of Trp53 genea
| Exons | Forward primers | Reverse primers | Length (bp) | Sequence primers |
|---|---|---|---|---|
| 2 | 5′-GACAAGTTATGCATCCATACAG-3′ | 5′-ACTGTTTTTGTTTCTCTCAGGC-3′ | 212 | Reverse |
| 3–4 | 5′-CCTGGGATAAGTGAGATTCTG-3′ | 5′-GAGAAAAAGAGGCATTGAAAGG-3′ | 508 | Reverse |
| 5–6 | 5′-CCACCTTGACACCTGATCGT-3′ | 5′-ATCAGTCTAGGCTGGAGTCA-3′ | 501 | Forward |
| 7 | 5′-TTCCCGGCTGCTGCAGGTC-3′ | 5′-CGGGACTCGTGGAACAGAA-3′ | 258 | Forward |
| 8–9 | 5′-GGCCTAGTTTACACACAGTC-3′ | 5′-GAGACAGAGGCAATAATGGGT-3′ | 429 | Reverse |
| 10 | 5′-GTGAAAGTGGTTGTGTGACC-3′ | 5′-TAGGGTGCAGCCCTAAGCA-3′ | 283 | Forward |
| 11 | 5′-CCAAACTGCTAGCTCCCATC-3′ | 5′-GAGACCTGACAACTATCAACC-3′ | 324 | Forward |
Primers were designed with assistance of the online software ‘Primer3’ (http://norp5424b.hsc.usc.edu/genetools.html).
Protein expression analyses of mesothelioma cell cultures ceram-Nf2+/− mice
Total proteins were extracted from scrapped cells using Radio Immuno Precipitation Assay (RIPA) buffer (Euromedex, Mundolsheim, France) containing protease inhibitors (Complete, Roche Molecular Biochemicals, Meylan, France). Lysates were incubated for 30 min on ice and cleared by centrifugation for 15 min at 4°C and 16 000g. Protein concentration of the supernatants was assessed by the Lowry’s procedure (Bio-Rad, Marnes la Coquette, France).
For western blot analysis, 20 μg of proteins were denaturated in Laemmli buffer by boiling for 5 min and separated on a reducing 10–12% sodium dodecyl sulfate–polyacrylamide gel. Proteins were blotted to nitrocellulose membranes by electrotransfer according to a procedure already described (23).
Protein detection was performed with primary antibodies raised against nf2 (A-19), p15Ink4b (M-20), p16Ink4a (M-156) and p53 (FL-393) all from TEBU Santa Cruz (Le Perray en Yvelines, France); p19Arf (ab-80) from Abcam (Cambridge, UK) and α-tubulin (CP06) from Calbiochem (VWR International, Strasbourg, France). Secondary antibodies were anti-goat IgG–horseradish peroxidase, anti-rabbit IgG–horseradish peroxidase and anti-mouse IgG–horseradish peroxidase (TEBU from Santa Cruz). Protein detection was performed using enhanced chemiluminescence reagents (GE Healthcare Biosciences, Orsay, France). Controls were proteins extracted from mice erythroleukemia cell line. α-Tubulin expression served as a control for equivalent protein loading.
Statistical analysis
Statistical analysis was performed with the Statview software (Abacus concept, Berkeley, CA). Survival curves were drawn using the Kaplan–Meier method and compared using the log-rank (Mantelcox) test. Comparison of tumour and ascite incidence between exposed and control groups was made using Fisher’s exact test.
Results
Clinicopathology
Viability of mice was reduced after exposure to RCF1, in comparison with controls. Median of survival after fibre exposure was 478 days in ceram-Nf2+/− mice group and 629 days in ctrl-Nf2+/− group (P = 0.0012). Ascitic fluid was observed in a total of 24 mice, 23 ceram-Nf2+/− and one ctrl-Nf2+/− mice.
Histological examination identified peritoneal lesions consistent with peritoneal mesothelioma in 26 (55%) ceram-Nf2+/− mice. Peritoneal hyperplasia was evidenced in two ceram-Nf2+/− mice. Two mesotheliomas (7.1%) were observed in the ctrl-Nf2+/− group. A cellular and mild fibrosis was often observed in ceramic-exposed animals. No fibrosis was found in ctrl-Nf2+/− mice.
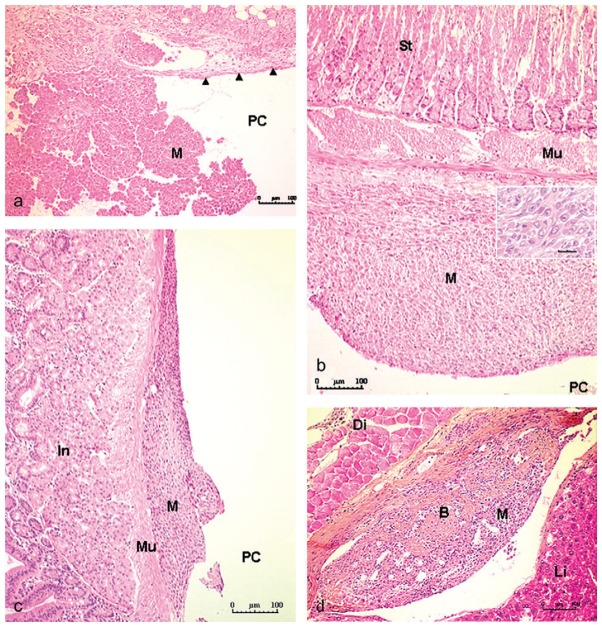
Histological features of peritoneal tumours were similar to those observed in HMM, showing epithelial, fusiform or biphasic pattern. In ceram-Nf2+/− mice, there were 7 epithelial, 10 fusiform and 9 bi-phasic mesotheliomas. Four mesotheliomas, three of fusiform pattern and one biphasic, exhibited bone metaplasia in ceram-Nf2+/− mice. Figure 1 illustrates the different mesothelioma morphologies. Mesotheliomas found in ctrl-Nf2+/− mice were similar to those found in the exposed animals. Both were biphasic mesotheliomas, one with a bone metaplasia.
Fig. 1.
Histology of peritoneal mesotheliomas found in ceram-Nf2+/− mice. (a) Epithelial mesothelioma (M) exhibiting a typical papillary pattern. Well-differentiated mesothelial cells proliferate into the peritoneal cavity (PC). Arrowheads: adjacent mesothelium. Grid represents 100 μm. (b) Epithelial mesothelioma (M). Dense and nodular proliferation at the surface of stomachal peritoneum, invading the peritoneal cavity (PC). Mu: stomachal muscle and St: stomachal mucosa. Grid represents 100 μm. Insert: higher magnification of tumour. Bar represents 2 μm. (c) Fusiform mesothelioma (M) at the surface of intestine peritoneum. In, intestinal mucosa; Mu: intestinal muscle and PC: peritoneal cavity. Grid represents 100 μm. (d) Typical bone differentiation (B) of a mesothelioma (M). Tumour growth is seen at the vicinity of the diaphragm (Di). Li: liver. Grid represents 100 μm.
In the ceram-Nf2+/− group, first mesothelioma was detected 223 days after inoculation. In ctrl-Nf2+/− group, mesotheliomas were found 450 and 676 days after inoculation.
Other than peritoneal tumours were detected in 19 ceram-Nf2+/− mice and 18 ctrl-Nf2+/− mice, the most frequently observed was lung papillary carcinoma as already described (22,24). Other tumours less frequently observed were carcinomas of the urogenital or digestive system, lacrimal gland or pineal gland. No difference was found between groups of exposed and control mice. In ceram-Nf2+/− group, most ascites (91%) were associated with mesothelioma. In parallel, most of mesotheliomas (80.8%) were associated with the production of ascitic fluid. Ascitis was present in one ctrl-Nf2+/− with mesothelioma.
Analysis of p15/Cdkn2b, p16/Cdkn2a and p19/Arf in mesothelioma cell cultures from ceram-Nf2+/− mice
Analysis of p15/Cdkn2b showed amplification of exons 1 and 2 in four mesothelioma cell cultures: 145, 146, 186 and 255, and lack of amplification indicative of intragenic deletion in the remaining cultures after PCR analysis of gDNA (Figure 2a). Investigation of Ink4a locus showed a normal status of p16/Cdkn2a and p19/Arf in the same cultures (i.e. 145, 146, 186 and 255) as demonstrated by the amplification of exons 1α (p16/Cdkn2a), 1β (p19/Arf) and 2 (common to p16/ Cdkn2a and p19/Arf). Accordingly, p15Ink4b, p16Ink4a and p19Arf protein expression was found only in cultures showing PCR gene amplification (Figure 2b and c).
Fig. 2.
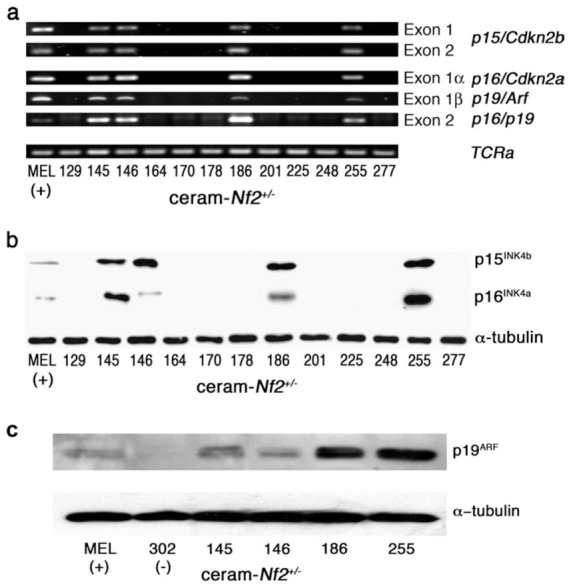
Analysis of p15/Cdkn2b (Ink4b locus), p16/Cdkn2a and p19/Arf (Ink4a/Arf locus) in mesothelioma cell cultures obtained from ascites developed in ceram-Nf2+/− mice. (a) Analysis of p15/Cdkn2b, p16/Cdkn2a and p19/Arf genes by PCR. Exons 1 and 2 (p15/Cdkn2b), 1α (p16/Cdkn2a), 1β (p19/Arf) and 2 (common to p16/Cdkn2a and p19/Arf) were amplified by PCR using appropriate primer sets (Table I). Loss of exons suggests that Ink4a and Ink4b inactivation occurred by loss of a part or whole chromosome. Only four cultures (145, 146, 186 and 255) demonstrated normal exon amplification. Positive control (+) was gDNA extracted from mice erythroleukemia cell line. TCRa amplification was used as PCR control. (b) Analysis of p15Ink4b and p16Ink4a protein expression by western blots. Each antibody used for immunoblot was specific for a non-conserved epitope of the corresponding protein. p15Ink4b and p16Ink4a proteins were detected in four cell cultures (145, 146, 186 and 255). Positive control (+) was proteins extracted from mice erythroleukemia cell line. α-Tubulin was used as control for equivalent protein loading. (c) Analysis of p19Arf protein expression by western blots. p19Arf protein expression was studied in cultures showing gene amplification and in a negative mesothelioma culture (302) obtained from an asb-Nf2+/− mice. p19Arf was accordingly expressed in cultures where p19/Arf exons were present. Positive control (+) was proteins extracted from mice erythroleukemia cell line. α-Tubulin was used as control for equivalent protein loading.
Analysis of Trp53 status in mesothelioma cell cultures from ceram-Nf2+/− mice
In order to determine the status of Trp53 in the murine mesothelioma cell cultures, direct sequencing of exons 2–11 was made from gDNA. Mutations were detected in 25% (3/12) mesothelioma cell cultures (no. 145, 186, 255). In culture 145, a mutation was detected in exon 7 at codon 236 (706A → G, N236D), changing the encoded asparagine into aspartic acid. In culture 186, a duplication (dup827–838) was detected in exon 8 from codon 276 to 281 introducing a sequence RNRR. In culture 255, a mutation occurred in exon 10 at codon 333 (997A → T, K333X), changing the encoded lysine into a stop codon (Figure 3a). Accordingly, a strong p53 protein expression was observed in cultures 145 and 186, whereas no band was detected in culture 255 (Figure 3b).
Fig. 3.
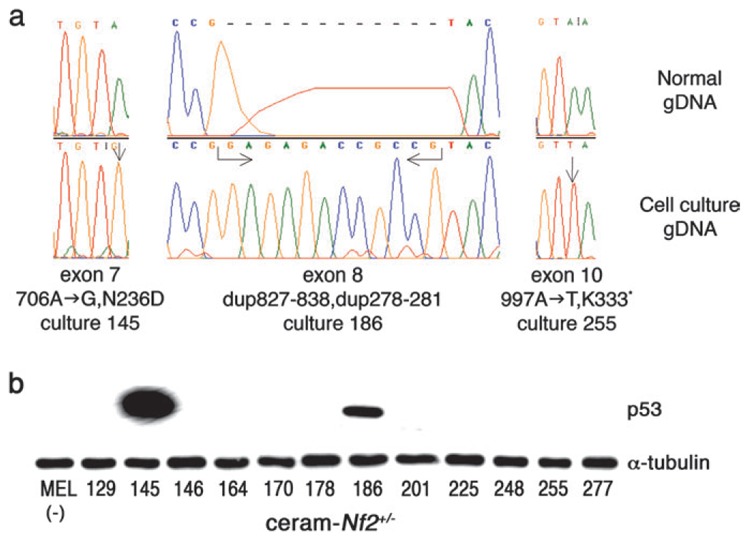
Analysis of Trp53 in mesothelioma cell cultures obtained from ascites developed in ceram-Nf2+/− mice. (a) Mutations in the Trp53 in murine mesothelioma cell cultures 145, 186 and 255. A missense mutation was found in culture 145, whereas in culture 255 the mutation introduced a stop codon. A duplication was detected in culture 186. Arrows indicate the position of the mutations. gDNA sequence was compared with data provided in GeneBank (top: normal gDNA and bottom: gDNA from mesothelioma cells). (b) Analysis of p53 protein expression by western blots. p53 was found to be expressed in cultures 145 and 186 in agreement with protein stabilization related to the occurrence of gene mutation. Negative control (−) was proteins extracted from mice erythroleukemia cell line. α-Tubulin expression served as a control for equivalent protein loading.
Analysis of loss of heterozygosity of Nf2 in mesothelioma cell cultures from ceram-Nf2+/− mice
Loss of heterozygosity of Nf2 was found in 83.5% (10/12) mesothelioma cell cultures obtained from ceram-Nf2+/− mice, on the basis of loss of exon 3 of the wild-type Nf2 allele (Nf2+) by PCR analysis (Figure 4a). Nevertheless, only a faint transcript, but no protein expression was found in culture 178, and neither transcript nor protein expression in culture 201 (Figure 4b and c).
Fig. 4.
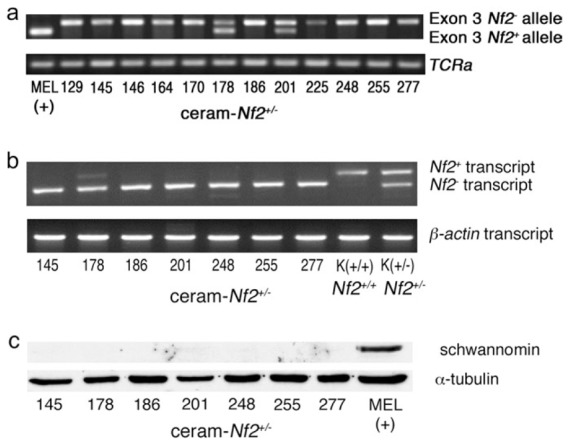
Analysis of loss of heterozygosity of exon 3 in Nf2 by PCR, and of gene expression in murine mesothelioma cell cultures obtained from ascites developed in ceram-Nf2+/− mice. (a) Analysis of loss of heterozygosity of exon 3 in Nf2 gene by PCR. Loss of heterozygosity was found in all but two cell cultures (178 and 201). Positive control (+) was gDNA extracted from mice erythroleukemia cell line. TCRa amplification was used as PCR control. (b) Reverse transcription–PCR analysis of Nf2 mRNA transcripts. mRNA was reverse transcribed and cDNA was amplified using appropriate primers (Table I). Seven cultures were analysed, demonstrating that culture 178 and 201 express faint or no Nf2+ transcript respectively. Controls were mRNAs extracted from kidney (K) from Nf2+/+ (K+/+) and Nf2+/− (K+/−) mice, respectively. β-Actin cDNA was used as reverse transcription–PCR control. (c) nf2 protein expression of schwannomin (or merlin) by western blots. Schwannomin expression was studied in cultures showing exon 3 amplification (178 and 201) and in five negative cultures used as negative controls. Schwannomin was not found to be expressed in these cultures. Positive control (+) was proteins extracted from mice erythroleukemia cell line. α-Tubulin expression served as a control for equivalent protein loading.
In order to validate Nf2 loss of heterozygosity results obtained with cell cultures, microdissection was performed on both frozen and paraffin tissue sections but sound data were not provided regarding the presence of normal cells and the limited amount of material available from the slides (data not shown). In five cases (mice 145, 178, 186, 255 and 277), frozen samples of tumours were available. Quantitative real-time PCR was carried out using especially designed primers for amplification of wild-type exon 3 of the Nf2 gene (Table I). Gapdh was used as reference gene and the 2−ΔΔCt method of data analysis was applied (29). A loss of heterozygosity was detected in tumours from 145, 186, 255 and 277 mice and amplification was found in tumour from 178 mouse. These results are consistent with data obtained with cultures from the corresponding ascites (Figure 4c). Amplification associated with lack of protein expression in culture 178 may be indicative of chromosome rearrangement.
Discussion
This study has demonstrated the development of mesotheliomas in hemizygous Nf2+/− mice exposed to RCFs. This finding is consistent with the tumoral potency of RCFs demonstrated previously in rats and hamsters (3,5–7). Accordingly, survival in ceram-Nf2+/− mice was significantly shortened in comparison with ctrl-Nf2+/− mice. In our series, first mesothelioma occurred after a delay of 7 months after inoculation. In ctrl-Nf2+/− mice, two mesotheliomas were detected after delays post-saline inoculation of 14.5 and 22.5 months, respectively. The low rate of spontaneous mesotheliomas in ctrl-Nf2+/− mice is consistent with data published elsewhere, in agreement with the potential role of Nf2 gene in the development of mesothelioma (24). The ability of RCFs to produce mesotheliomas in Nf2+/− mice does not appear to be the consequence of allelic inactivation of Nf2+/− gene, as mesotheliomas were also found in wild-type counterparts (data not shown). Nevertheless, this constitutive genetic modification probably contributes to the incidence of mesotheliomas, as the percentage of mesotheliomas was lower in RCF-exposed wild-type counterparts (42.5%, 47 mice).
Clinical features of RCF-induced mesotheliomas were similar to those reported in HMM, with symptoms of fluid accumulation being an indicator of mesothelioma formation, as most of ascites (91%) were associated with mesothelioma. Only two ascites were produced without occurrence of mesothelioma, in mice showing peritoneal inflammation and leiomyosarcoma, respectively.
Histological analysis revealed that mesotheliomas induced in Nf2+/− mice were of the same types as described in HMM, and in murine asbestos-induced mesotheliomas (22,30). However, it must be noted that the distribution of morphological subtypes differed from that of HMM. Human epithelial mesothelioma represents ~60% of the cases, the remaining being approximately equally distributed between fusiform and biphasic pattern. Epithelial morphology was less frequent in RCF1-induced murine mesotheliomas (27%), whereas both fusiform and biphasic patterns were more frequent (38.4 and 34.6%, respectively). In comparison with previously reported crocidolite-induced murine mesotheliomas, percentage of fusiform subtype was lower, at the benefit of biphasic phenotype.
In the literature, mesothelioma morphology in animals following exposure to ceramic fibres is not always quoted. Among the small number of mesotheliomas observed after inhalation in rats and hamsters, epithelial, sarcomatous and biphasic types were found (5,7,31). According to our knowledge, no data has been reported in mice, except that tumour nodules with surface papillary formations and more solid areas composed of spindle and polygonal cells were depicted in crocidolite-exposed mice (32).
Apart from mesothelioma, a mild fibrotic reaction was observed in ceram-Nf2+/− mice, in agreement with data reported by others in the lungs of RCF-exposed animals (5,33).
Molecular studies have been carried out on primary cultures of cells grown from ascitic fluids. It must be noted that ascites are representative of mesotheliomas occurring in ceram-Nf2+/− mice, as they were present in 80.8% of mesotheliomas diagnosed from histological sections. Mesotheliomas without ascite production were either early epithelial tumour (two cases) or invasive fusiform mesotheliomas (three cases). Moreover, a substantial number of ascites (52%) provided cell cultures, accounting for 50% of the mesotheliomas, thus entailing a fairly good representation of the tumour spectrum.
Gene analysis showed that mesotheliomas induced in ceram-Nf2+/− mice present similar tumour suppressor gene alterations, as described previously in murine and human asbestos-induced mesothelioma (2,21,23). That is, co-deletion of p16/Cdkn2a, p19/Arf and p15/Cdkn2b is predominant in mesotheliomas developed in ceram-Nf2+/− mice (66.7%) as found in abs-Nf2+/− mice (81.8%) and in humans (22,23). Moreover, Trp53 mutations in mice were found approximately at similar rates of 25% in ceram-Nf2+/− mice and 18.2% in abs-Nf2+/− mice (23). In this context, deletions found in Nf2 and in Ink4 genes result in the absence of the encoded proteins. As far as Trp53 is concerned, mutations in mice at codon 333 have not been reported (18,34). As the generated stop codon is localized in the oligomerization domain of the protein, it is probably that it produces a loss of function. In the corresponding human codon, two truncating mutations have been described (35,36). In these publications, no investigation of the truncated protein function was made, but it is usually assumed that frameshift and nonsense mutations are deleterious for Trp53, and prediction of mutation in the oligomerization domain suggest a loss of function (35,37).
In conclusion, clinicopathological features and molecular changes in neoplasia induced by RCF1 sample in mice are consistent with mesothelioma and similar to those observed in asbestos-induced mesothelioma. The recent observation of similarity between peritoneal and pleural HMM demonstrated by high correlation of gene expression in these two tumour types (38) is an additional validation of the present model of malignant mesothelioma. Genetic alterations in tumour suppressor genes suggest that mesothelial cell transformation can follow at least two major routes involving G1/S transition and Nf2-regulated pathways, associating genes at the Ink4 locus or Trp53, and suggesting that Nf2 mutation is complementary of Ink4 or Trp53 mutation. These results show that these somatic genetic changes are features of the mechanism of mesothelial cell transformation, and may suggest a relationship with the effect of mineral fibres.
Acknowledgments
This work was supported by Institut National de la Santé de la Recherche Médicale (INSERM) and University Paris-Val de Marne funds, grants from Ministère de l’Emploi et de la Solidarité no. 1D004C, Ministère de l’Environnement no. AC008B, Agence Nationale de la Recherche n°05 9 31/ANR 05 SEST 029-01, Ligue Nationale contre le Cancer, Cancéropôle Ile-de-France and Legs Poix. P.A. was a fellow from Association de la Recherche pour le Cancer.
Abbreviations
- gDNA
genomic DNA
- HMM
human malignant mesothelioma
- PCR
polymerase chain reaction
- RCF
refractory ceramic fibre
Footnotes
Conflict of Interest Statement: None declared.
References
- 1.Pasetto R, et al. Mesothelioma associated with environmental exposures. Med Lav. 2005;96:330–337. [PubMed] [Google Scholar]
- 2.Robinson BW, et al. Advances in malignant mesothelioma. N Engl J Med. 2005;353:1591–1603. doi: 10.1056/NEJMra050152. [DOI] [PubMed] [Google Scholar]
- 3.IARC. Man-made mineral fibres. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol. 81. IARC; Lyon: 2002. [PMC free article] [PubMed] [Google Scholar]
- 4.Cowie HA, et al. An epidemiological study of the respiratory health of workers in the European refractory ceramic fibre industry. Occup Environ Med. 2001;58:800–810. doi: 10.1136/oem.58.12.800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mast RW, et al. Studies on the chronic toxicity (inhalation) of four types of refractory ceramic fiber in male fischer 344 rats. Inhal Toxicol. 1995;7:425–467. doi: 10.3109/08958379509015208. [DOI] [PubMed] [Google Scholar]
- 6.Mast RW, et al. Multiple-dose chronic inhalation toxicity study of size-separated kaolin refractory ceramic fiber in male fisher 344 rats. Inhal Toxicol. 1995;7:469–502. doi: 10.3109/08958379509015209. [DOI] [PubMed] [Google Scholar]
- 7.McConnell EE, et al. Chronic inhalation study of a kaolin-based refractory ceramic fiber in Syrian golden hamsters. Inhal Toxicol. 1995;7:503–532. doi: 10.3109/08958379509015209. [DOI] [PubMed] [Google Scholar]
- 8.Donaldson K, et al. Superoxide anion release by alveolar macrophages exposed to respirable industrial fibres: modifying effect of fibre opsonisation. Exp Toxicol Pathol. 1995;47:229–231. doi: 10.1016/S0940-2993(11)80253-8. [DOI] [PubMed] [Google Scholar]
- 9.Dopp E, et al. Induction of micronuclei, hyperdiploidy and chromosomal breakage affecting the centric/pericentric regions of chromosomes 1 and 9 in human amniotic fluid cells after treatment with asbestos and ceramic fibers. Mutat Res. 1997;377:77–87. doi: 10.1016/s0027-5107(97)00062-6. [DOI] [PubMed] [Google Scholar]
- 10.Hart GA, et al. Cytotoxicity of refractory ceramic fibres to Chinese hamster ovary cells in culture. Toxicol In Vitro. 1992;6:317–326. doi: 10.1016/0887-2333(92)90021-i. [DOI] [PubMed] [Google Scholar]
- 11.Ohyama M, et al. The chemiluminescent response from human monocyte-derived macrophages exposed to various mineral fibers of different sizes. Ind Health. 2000;38:289–293. doi: 10.2486/indhealth.38.289. [DOI] [PubMed] [Google Scholar]
- 12.Wang QE, et al. Biological effects of man-made mineral fibers (I)–reactive oxygen species production and calcium homeostasis in alveolar macrophages. Ind Health. 1999;37:62–67. doi: 10.2486/indhealth.37.62. [DOI] [PubMed] [Google Scholar]
- 13.Wang QE, et al. Biological effects of man-made mineral fibers (II)–-their genetic damages examined by in vitro assay. Ind Health. 1999;37:342–347. doi: 10.2486/indhealth.37.342. [DOI] [PubMed] [Google Scholar]
- 14.Kociok N, et al. DNA fingerprint analysis reveals differences in mutational patterns in experimentally induced rat peritoneal tumors, depending on the type of environmetal mutagen. Cancer Genet Cytogenet. 1999;111:71–76. doi: 10.1016/s0165-4608(98)00221-0. [DOI] [PubMed] [Google Scholar]
- 15.Unfried K, et al. P53 mutations in tumours induced by intraperitoneal injection of crocidolite asbestos and benzo[a]pyrene in rats. Exp Toxicol Pathol. 1997;49:181–187. doi: 10.1016/S0940-2993(97)80005-X. [DOI] [PubMed] [Google Scholar]
- 16.Unfried K, et al. Distinct spectrum of mutations induced by crocidolite asbestos: clue for 8-hydroxydeoxyguanosine-dependent mutagenesis in vivo. Cancer Res. 2002;62:99–104. [PubMed] [Google Scholar]
- 17.Ni Z, et al. Analysis of K-ras and p53 mutations in mesotheliomas from humans and rats exposed to asbestos. Mutat Res. 2000;468:87–92. doi: 10.1016/s1383-5718(00)00043-7. [DOI] [PubMed] [Google Scholar]
- 18.Jackson MA, et al. Genetic alterations in cancer knowledge system: analysis of gene mutations in mouse and human liver and lung tumors. Toxicol Sci. 2006;90:400–418. doi: 10.1093/toxsci/kfj101. [DOI] [PubMed] [Google Scholar]
- 19.Olivier M, et al. TP53 mutation spectra and load: a tool for generating hypotheses on the etiology of cancer. IARC Sci Publ. 2004;157:247–270. [PubMed] [Google Scholar]
- 20.Staib F, et al. TP53 and liver carcinogenesis. Hum Mutat. 2003;21:201–216. doi: 10.1002/humu.10176. [DOI] [PubMed] [Google Scholar]
- 21.Murthy SS, et al. Asbestos, chromosomal deletions, and tumor suppressor gene alterations in human malignant mesothelioma. J Cell Physiol. 1999;180:150–157. doi: 10.1002/(SICI)1097-4652(199908)180:2<150::AID-JCP2>3.0.CO;2-H. [DOI] [PubMed] [Google Scholar]
- 22.Fleury-Feith J, et al. Hemizygosity of Nf2 is associated with increased susceptibility to asbestos-induced peritoneal tumours. Oncogene. 2003;22:3799–3805. doi: 10.1038/sj.onc.1206593. [DOI] [PubMed] [Google Scholar]
- 23.Lecomte C, et al. Similar tumor suppressor gene alteration profiles in asbestos-induced murine and human mesothelioma. Cell Cycle. 2005;4:1862–1869. doi: 10.4161/cc.4.12.2300. [DOI] [PubMed] [Google Scholar]
- 24.Giovannini M, et al. Conditional biallelic Nf2 mutation in the mouse promotes manifestations of human neurofibromatosis type 2. Genes Dev. 2000;14:1617–1630. [PMC free article] [PubMed] [Google Scholar]
- 25.AVMA. 1993 report of the AVMA panel on euthanasia. J Am Vet Med Assoc. 1993;202:230–249. [PubMed] [Google Scholar]
- 26.Zhuang SM, et al. Inactivations of p16INK4a-alpha, p16INK4a-beta and p15INK4b genes in 2′,3′-dideoxycytidine- and 1,3-butadiene-induced murine lymphomas. Oncogene. 1998;16:803–808. doi: 10.1038/sj.onc.1201600. [DOI] [PubMed] [Google Scholar]
- 27.Broers AE, et al. Interleukin-7 improves T-cell recovery after experimental T-cell-depleted bone marrow transplantation in T-cell-deficient mice by strong expansion of recent thymic emigrants. Blood. 2003;102:1534–1540. doi: 10.1182/blood-2002-11-3349. [DOI] [PubMed] [Google Scholar]
- 28.Giulietti A, et al. An overview of real-time quantitative PCR: applications to quantify cytokine gene expression. Methods. 2001;25:386–401. doi: 10.1006/meth.2001.1261. [DOI] [PubMed] [Google Scholar]
- 29.Livak KJ, et al. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 30.Churg A, et al. Mesothelioma. In: William D, Travis EB, Konrad Müller-Hermelink H, Harris Curtis C, editors. Pathology and Genetics of the Lung, Pleura, Thymus and Heart. Vol. 10. WHO Publications; Lyon: 2004. pp. 128–140. [Google Scholar]
- 31.Lee K, et al. Biological studies on inorganic potassium titanate fibers. Biological Effects of Man-Made Mineral Fibers; Proceedings of a WHO/IARC Conference; Copenhagen: World Health Organization; 1984. pp. 323–333. [Google Scholar]
- 32.Davis MR, et al. Establishment of a murine model of malignant mesothelioma. Int J Cancer. 1992;52:881–886. doi: 10.1002/ijc.2910520609. [DOI] [PubMed] [Google Scholar]
- 33.Smith DM, et al. Long-term health effects in hamsters and rats exposed chronically to man-made vitreous fibres. Ann Occup Hyg. 1987;31:731–754. doi: 10.1093/annhyg/31.4b.731. [DOI] [PubMed] [Google Scholar]
- 34.GAC database. Genetic alterations in cancer. 2006. [2 March 2007 date Last accessed]. http://dir-apps.niehs.nih.gov/gac/
- 35.Hartmann A, et al. p53 gene mutations inside and outside of exons 5–8: the patterns differ in breast and other cancers. Oncogene. 1995;10:681–688. [PubMed] [Google Scholar]
- 36.Nogales FF, et al. Multifocal intrafollicular granulosa cell tumor of the ovary associated with an unusual germline p53 mutation. Mod Pathol. 2004;17:868–873. doi: 10.1038/modpathol.3800133. [DOI] [PubMed] [Google Scholar]
- 37.Soussi T. The p53 database. 2005. [2 March 2007, date Last accessed]. http://p53.free.fr/
- 38.Borczuk AC, et al. Molecular profiling of malignant peritoneal mesothelioma identifies the ubiquitin-proteasome pathway as a therapeutic target in poor prognosis tumors. Oncogene. 2006;26:610–617. doi: 10.1038/sj.onc.1209809. [DOI] [PubMed] [Google Scholar]