Abstract
Staphylococcus aureus (S. aureus) is notorious for its ability to acquire antibiotic-resistance, and antibiotic-resistant S. aureus has become a wide-spread cause of high mortality rate. Novel antimicrobials capable of eradicating S. aureus cells including antibiotic-resistant ones are thus highly desired. Membrane-active bactericides and species-specific antimicrobials are two promising sources of novel anti-infective agents for fighting against bacterial antibiotic-resistance. We herein show that Ca2+ and Mg2+, two alkaline-earth-metal ions physiologically essential for diverse living organisms, both disrupt model S. aureus membranes and kill stationary-phase S. aureus cells, indicative of membrane-activity. In contrast to S. aureus, Escherichia coli and Bacillus subtilis exhibit unaffected survival after similar treatment with these two cations, indicative of species-specific activity against S. aureus. Moreover, neither Ca2+ nor Mg2+ lyses mouse red blood cells, indicative of hemo-compatibility. This works suggests that Ca2+ and Mg2+ may have implications in targeted eradication of S. aureus pathogen including the antibiotic-resistant ones.
Staphylococcus aureus (S. aureus) is a Gram-positive bacterium notorious for its ability to acquire antibiotic-resistance1. Antibiotic-resistant strains of S. aureus have emerged as a widespread cause of both hospital- and community-associated infections, leading to high mortality rate1,2,3. For example, methicillin-resistant S. aureus is estimated to cause >11,000 deaths per year in the United States4. It is thus imperative to discover/develop antimicrobials that are both active against S. aureus including antibiotic-resistant strains and less prone to evoke resistance.
Antimicrobial peptides (AMPs) are nature’s antibiotics still in action despite of their evolutionarily ancient origins. Many AMPs act by impairing the barrier function of bacterial membranes5,6,7,8,9,10,11, a generic mode that appears to be more difficult for bacteria to circumvent than the metabolic-targeting modes of conventional antibiotics12. By capturing the structural features common to most AMPs (i.e., being simultaneously cationic and amphiphilic), synthetic mimics of AMPs (SMAMPs) have demonstrated similar in vitro antibacterial activity and membrane-destabilizing modes as do AMPs13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42. Despite of the great potentials, AMPs and SMAMPs are effort- and, often, cost-consuming to produce, which has significantly hindered their pharmaceutical development. Alternative to AMPs and SMAMPs, species-specific antimicrobials have recently been proposed as a promising source of anti-infective agents that are less prone to evoke resistance43. Therefore, antimicrobials that are readily available and simultaneously membrane-active and species-specific against S. aureus are highly desired.
Metal ions are readily available, and certain transition-metal ions (e.g., Cu2+, Hg2+, Zn2+, and Cd2+) have demonstrated wide-spectrum antibacterial efficacy to varying extent44,45. A best known example might be Ag+ ion, which is active against both Gram-negative and –positive bacteria46,47. Nevertheless, use of heavy metal ions as disinfectants may adversely impact the host and/or the environment. Besides, the action modes by which these heavy metal ions achieve their antibacterial activity remain elusive. Having these concerns in mind, we hence turn to non-transition metal ions, in efforts to find candidates for specifically disrupting S. aureus membranes.
Calcium and magnesium ions (Ca2+ and Mg2+) are two alkaline-earth-metal ions (M2+) physiologically essential to almost all living organisms48. Upon binding with cardiolipin (CL), a major lipid component in S. aureus membranes49,50, M2+ (M = Ca, Mg) forms M2+-CL complexes of negative curvature (Fig. 1a)41,51, and negative curvature is a physical parameter necessary for a variety of membrane-destabilization processes as has been validated for those induced by AMPs and SMAMPs11,39,40,41,42. We therefore hypothesize that Ca2+ and Mg2+ may be membrane-active, species-specific agent against S. aureus. To test this hypothesis, we perform vesicle membrane permeabilization assays and antibacterial assays and find that, at ≤40 mM, both Ca2+ and Mg2+ disrupt model S. aureus membranes and kill stationary phase S. aureus cells, indicative of membrane-activity. In contrast to S. aureus, Escherichia coli and Bacillus subtilis exhibit unaffected survival after similar treatment with these two cations, indicative of species-specific activity against S. aureus. Moreover, within the tested dose range, neither Ca2+ nor Mg2+ is hemolytic against mouse red blood cells, indicative of good hemo-compatibility. Collectively, these results suggest that Ca2+ and Mg2+ may have implications in targeted eradication of S. aureus pathogen including antibiotic-resistant ones.
Figure 1.
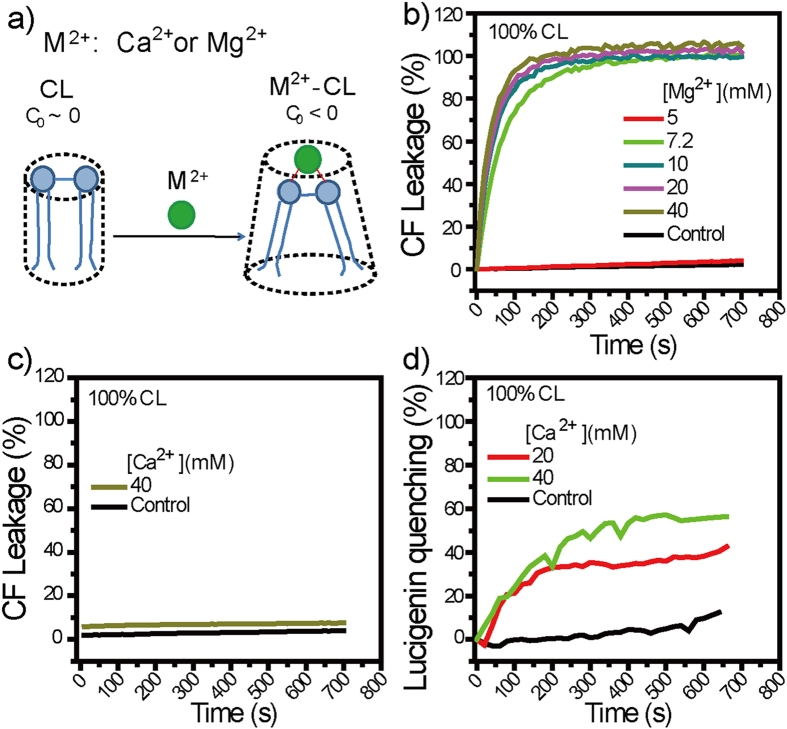
(a) Binding of M2+ (M = Ca, Mg) with cardiolipin (CL), the major lipid component in S. aureus, converts the originally zero-intrinsic-curvature (C0 ~ 0) lipid into M2+-CL complexes with negative intrinsic curvature (C0 < 0). (b–d) Dye leakage assays using large unilamellar vesicles (LUVs) composed of 100% CL as a first-order model for S. aureus membranes. (b) Mg2+ at ≥7.2 mM caused significant carboxyl fluorescein (CF) leakage. (c,d) Ca2+, though (c) unable to cause detectable CF leakage, caused (d) appreciable quenching in the fluorescence intensity of intravesicular lucigenin. Controls are samples assayed similarly but without M2+ additions.
Results and Discussion
M2+ (M = Ca, Mg) binds with CL to form M2+-CL complexes of negative curvature (Fig. 1a)41,51, and negative curvature promote membrane destabilization as validated for cases with AMPs and SMAMPs11,39,40,41,42. We hence evaluated whether Ca2+ and Mg2+ destabilizes S. aureus membranes, using mono-component large unilamellar vesicles (LUVs) composed of CL as our first order model of S. aureus membranes and performing dye leakage assays39,41,52,53,54,55,56,57. To dissect the effect of M2+ dose from those of ionic strength and osmolarity, we use M2+-supplemented HEPES buffers (Table 1) which have ionic strength and osmolarity kept almost constant but varying MCl2 concentration; these same buffers are used for all experiments throughout this work. The first dye probe we used is carboxyl fluorescein (CF), a membrane-impermeant molecule with a hydrodynamic diameter of ~1 nm and negatively charges at physiological pH58,59,60,61,62. Mg2+, once ≥7.2 mM, caused ~100% CF leakage from CL LUVs (Fig. 1b), indicative of Mg2+-induced CF efflux across CL membranes. Ca2+, though unable to cause appreciable CF leakage up to 40 mM (Fig. 1c), induced >30% lucigenin quenching (Fig. 1d) once ≥20 mM in similar assays but with CF being replaced with lucigenin—a membrane-impermeant, fluorescent Cl−-indicator of similar molecular diameter as CF63,64,65, indicative of Ca2+-induced Cl− influx across membranes. Obviously, both Ca2+ and Mg2+ permeabilize CL membranes. That their distinct abilities to releases CF from CL LUVs correlate with the difference in water channel diameter of inverted hexagonal (HII) structures they induced in CL membranes (1.50 nm versus 2.42 nm)51 further suggest that they may cause the observed membrane permeabilization by binding with CL to form negative-curvature M2+-CL complexes (M = Ca, Mg).
Table 1. Mg2+ and Ca2+ doses in 10 mM HEPES buffera.
| M2+ (mM) | HEPES (mM) | NaCl (mM) | Sucrose (mM) |
|---|---|---|---|
| 0 | 10 | 170 | 0 |
| 5 | 10 | 155 | 0 |
| 10 | 10 | 140 | 15 |
| 20 | 10 | 110 | 45 |
| 40 | 10 | 50 | 105 |
aHEPES, NaCl, and sucrose were supplemented to help keep the pH, final ionic strength and final osmolarity constant at 7.4, 170 mM, and 325–340 RT (where R is the gas constant and T is ambient temperature, and ideal solutions are assumed), respectively. Same buffers were used for all experiments through this work unless specified otherwise.
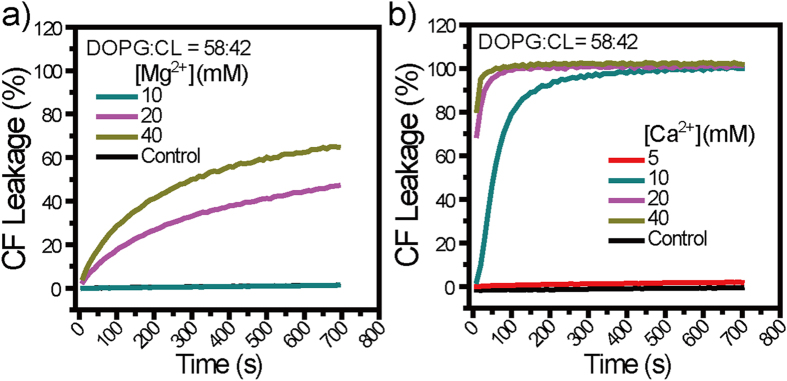
In addition to CL, phosphoglycerol (PG) is another major lipid component in S. aureus membranes49,50. To assess whether Ca2+ and Mg2+ permeabilize S. aureus membranes in which CL is diluted by PG, we use binary LUVs composed of DOPG:CL = 58:42 as a more realistic model for S. aureus membranes57 and perform similar CF leakage assays as above. From DOPG:CL = 58:42 LUVs, Ca2+ at ≥10 mM caused >90% CF leakage within 300 s after its addition, as compared to undetectable CF leakage caused by that at 5 mM (Fig. 2a), indicative of a minimum threshold Ca2+ concentration of 10 mM. Similarly, Mg2+ at ≥20 mM caused ≥40% CF leakage at 700 s after its addition, as compared to undetectable CF leakage by that at ≤10 mM (Fig. 2b), indicative of a minimum threshold Mg2+ concentration of 20 mM. Obviously, both Ca2+ and Mg2+ permeabilize model S. aureus membranes despite that CL content is diluted by PG but, for them to do so, certain minimal threshold concentrations are required.
Figure 2.
(a,b) Dye leakage assays using DOPG: CL = 58:42 LUVs as a more realistic model for S. aureus membranes. Obviously, both (a) Mg2+ and (b) Ca2+ caused significant CF leakage from DOPG: CL = 58:42 LUVs and, to do so, their minimum threshold concentrations are 20 and 10 mM, respectively. Controls are samples assayed similarly but without M2+ addition.
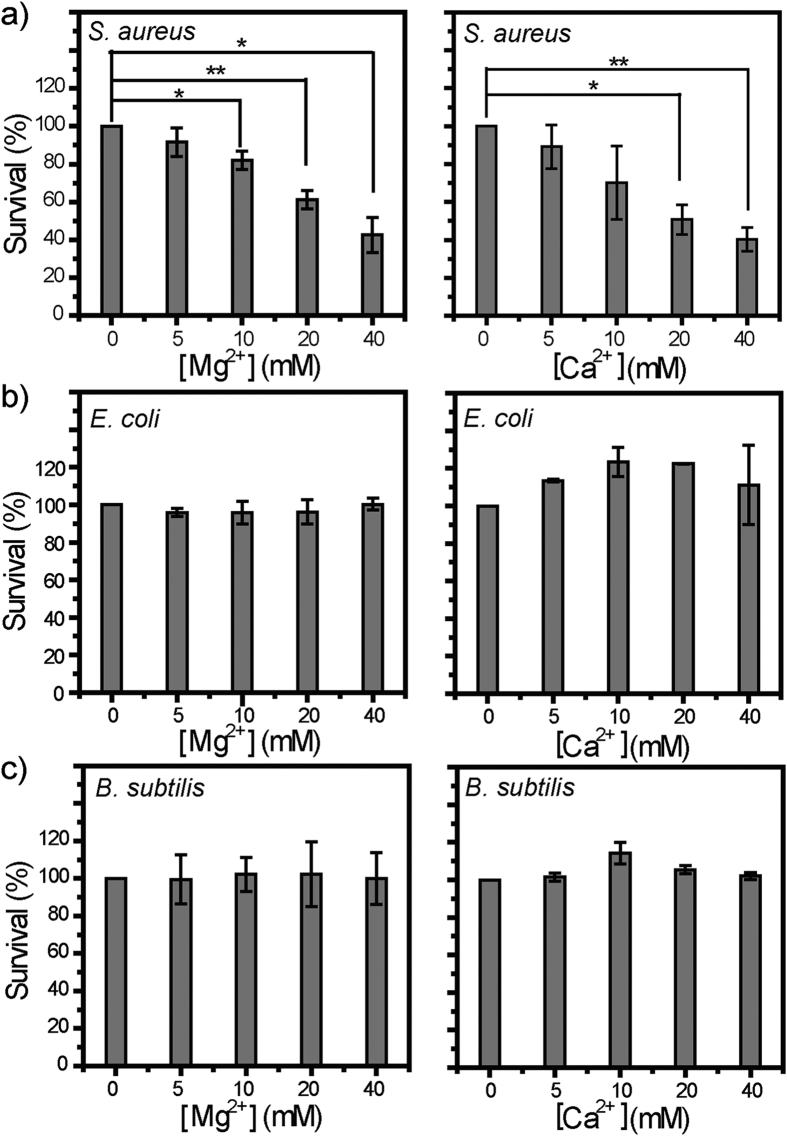
Both Ca2+ and Mg2+ are active against model S. aureus membranes. Does that necessarily correspond to antibacterial activity against S. aureus cells? To assess this, we evaluated the bactericidal activities of Ca2+ and Mg2+ by performing antibacterial plate killing assays. Note that bacterial cells in stationary phase are more resistant to environmental stresses and antibiotics than counterparts in logarithmic phase12,66,67. We hence used S. aureus cells in stationary phase, rather than those in logarithmic phase as normally do, for antibacterial assays. Our results (Fig. 3a) reveal that, after 40-min treatment with either Ca2+ or Mg2+, S. aureus cells exhibit viability loss to varying extent in a dose-dependent manner, with a maximal viability loss of ~60% observed at M2+ concentration of 40 mM, the highest dose tested. It is noteworthy that a relative loss of 60% in viability ratio corresponds to an absolute number density of ~3 × 105 CFU/mL (colony-forming units per milliliter) in bacterial cells killed. Taken together, these observations suggest that both Ca2+ and Mg2+ are definitively bactericidal against S. aureus.
Figure 3.
Plate killing assays against stationary-phase cells of (a) S. aureus, (b) E. coli, and (c) B. subtilis. After 40-min co-incubation with Mg2+ (left) and Ca2+ (right) at ≤40 mM, S. aureus cells exhibited dose-dependent loss in viability. In contrast, the viability of E. coli and B. subtilis cells is barely impacted in similar assays. Data points are reported as mean ± standard deviation. *and** indicate p < 0.05 and p < 0.01, respectively.
Closer examinations on both membrane permeabilizaiton experiments and antibacterial assays above suggest that Ca2+ and Mg2+ may be membrane-active against S. aureus. The minimal M2+ dose required for killing significant (p < 0.05) percentage of inoculated S. aureus cells are 10 and 20 mM for Ca2+ and Mg2+, respectively (Fig. 3a), which correlate well with the respective minimal threshold M2+ dose for these two cations to induce appreciable CF release from model S. aureus membranes (Fig. 2a,b), suggesting that Ca2+ and Mg2+ may kill S. aureus cells by disrupting their membranes.
In stark contrast to their definitive activity against S. aureus, Ca2+ and Mg2+ barely affect the viability of E. coli or B. subtilis (Fig. 3b,c), despite that B. subtilis is a Gram-positive bacterium as is S. aureus. Both E. coli and B. subtilis contain no/low CL in their membranes68. Thus, high CL content in S. aureus membrane may account for the observed activity of Ca2+ and Mg2+.
With species-specific antibacterial activity, MCl2 solutions are distinct from their corresponding MO powder slurries, which are wide-spectrum disinfectants69,70,71,72,73 used by human population of different cultures. Moreover, the observed activity of MCl2 solutions suggests that M2+ (M = Ca, Mg) ions may play contributive, rather than negligible, roles in the activity of their corresponding metal oxide (MO) powder slurries against S. aureus. To inhibit/kill ≥50% inoculated cells requires MO powder slurries of MO power dose at a few mg/mL, which corresponds to 1–100 mM70,71,74,75. Frequently, M2+ (M = Ca, Mg) ions produced via MO dissociation are viewed as negligible factors in the antibacterial activities of MO powder slurries74,76,77, due to inactivity of both the supernatant of MgO powder slurry and the MCl2 solutions at concentrations 10-fold of the MO powder solubility values70,72. Note that M2+ concentrations even 10-fold of MO powder solubility values are still <10 mM, which is within the barely-active dose range (Fig. 3a). Moreover, presence of S. aureus cells may actively retrieve free M2+ to form M2+-CL complexes, a process which may promote MO dissociation and thus shift the effective M2+ concentrations into the bactericidal range.
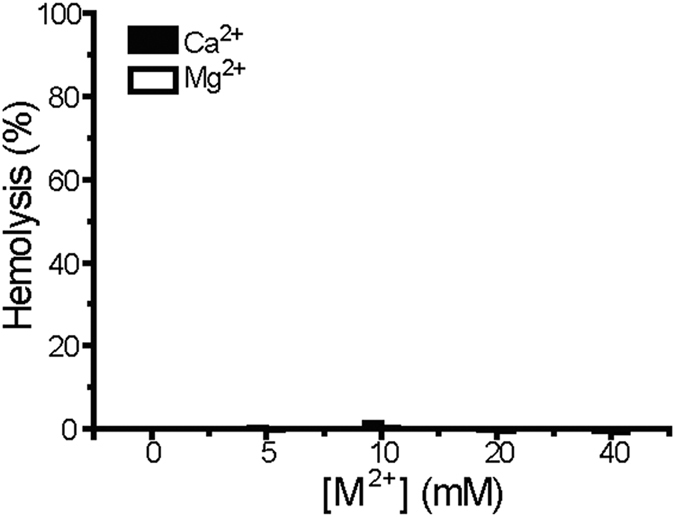
As potential antimicrobial agents, toxicity to host cells is a major concern. To preliminarily evaluate the toxicity of Mg2+ and Ca2+ ions, we performed hemolytic assays against mouse red blood cells. Within the tested concentration range (0–40 mM), neither Mg2+ nor Ca2+ caused >5% hemolysis (Fig. 4), indicative of good hemo-compatibility. Combined with the antibacterial assays (Fig. 3), these results suggest that Mg2+ and Ca2+ may preferentially eradiate S. aureus cells without affecting other bacteria or mammalian cells in the same niche.
Figure 4. Up to 40 mM, neither Mg2+ nor Ca2+ caused detectable hemolysis against mouse red blood cells.
Data points are reported as mean ± standard deviation.
Conclusion
In summary, we found that Ca2+ and Mg2+ may be membrane-active, species-specific bactericidal agent against S. aureus. Moreover, within the tested concentration range, both Ca2+ and Mg2+ lack hemolytic toxicity. This work suggests that Ca2+ and Mg2+ may have implications in targeted eradication of S. aureus pathogen including antibiotic-resistant ones.
Methods
Materials
S. aureus (ATCC 25923), E. coli (ATCC 25922) and B. subtilis (ATCC 6051) were purchased from American Type Culture Collection (ATCC) (Virginia, USA). Lipids used in this work, DOPG (1,2-dioleoyl-snglycero-3-[phospho-rac-(1-glycerol)] (sodium salt)), and TOCL (1,1′,2,2′-Tetraoleoyl Cardiolipin, Sodium Salt) were purchased from Avanti Polar Lipids (Alabama, USA) and used without further purification. Carboxyl fluorescein (CF) was purchased from Sigmal-Aldrich (Shanghai, China). Dehydrated Mueller-Hinton (MH) medium formulation and dehydrated Tryptic Soy Broth (TSB) medium formulation were purchased from Qingdao Hope Bio-Technology (Qingdao, China). All other reagents were purchased from Sinopharm Chemical Reagent Company (Shanghai, China). All reagents were used as supplied unless specified otherwise.
Large Unilamellar Vesicle (LUV) Preparations
LUVs composed of 100% CL and DOPG:CL = 58:42 were used as model cellular membranes for S. aureus57 and prepared via extrusion. Into a glass vial, CL stock solution was added with or without stock solutions of DOPG; all lipid stock solutions were in chloroform at 20 mg/mL. The resulting lipid mixture was dried under gentle N2 flow, desiccated in vacuum overnight, and rehydrated with CF (40 mM CF) or lucigenin (1 mM lucigenin, 50 mM NaNO3) solutions at 45 °C for 2 h. The resultant solution was subjected to five freeze-thaw cycles and subsequently extruded through a 0.4-μm Nucleopore polycarbonate membrane (Whatman) for 21 times using a mini-extruder (Avanti Polar Lipids). External CF or lucigenin was removed by gel filtration (Sephadex G-25, GE healthcare) using HEPES buffer A (10 mM HEPES, 150 mM NaCl, pH = 7.4) as eluent.
Characterizations on Membrane-Permeabilization
Fluorescence emission intensity It for CF (λex = 492 nm, λem = 518 nm) was monitored as a function of time (t). Into a fluorimeter sample cuvette, we added expected CF-preloaded LUV suspension (900 μL) and, at 200 s after initiation of It recording, M2+-containing HEPES solutions (1800 μL in total, Table 1), to a final lipid concentration of 0.1 mM. At 900 s after initiation of It recording (i.e., at 700 s after M2+ addition), 50 μL 10% triton was added, to yield I∞ which indicates 100% CF leakage. The percentages of CF leakage were calculated as leakage (%) = (It − I0)/(I∞ − I0) × 100, where I0 is the fluorescence intensity immediately (<10 s) after M2+ addition. Controls are samples assayed similarly but treated with M2+-absent solution (i.e., 10 mM HEPES, 150 mM NaCl, pH 7.4).
Antibacterial Assays
The antibacterial activities of Ca2+ and Mg2+ were evaluated by performing classic plate killing assays against stationary-phase bacterial cells. For each bacterial strain, 3–5 individual bacterial colonies were inoculated into fresh sterile trypticase soy broth (TSB) and incubated at 37 °C with shaking (200 rpm) for 16 h to stationary phase. Bacterial cells were then harvested and washed twice with sterile HEPES buffer A (10 mM HEPES, 150 mM NaCl, pH 7.4) via centrifugation (2,500 rpm, Eppendorf 5810R) for 1 min and, within 15 min, adjusted with sterile HEPES buffer A (10 mM HEPES, 150 mM NaCl, pH 7.4) to ~1.5 × 106 CFU/mL and inoculated (50 μL) into each zero-dilution well (150 μL in total) of a preset 96-well microplate.
Expected amounts of divalent cation stock solutions (10 mM HEPES, 150 mM CaCl2 or MgCl2, pH 7.4), sucrose stock solution (10 mM HEPES, 400 mM sucrose, pH 7.4), NaCl stock solution (10 mM HEPES, 500 mM NaCl, pH 7.4), and HEPES buffer B (10 mM HEPES, pH 7.4) were added into each zero-dilution well of a 96-well plate; all solutions were sterilized via filtering. After bacterial inoculation, final inculum size in each zero-dilution well was ~5 × 105 CFU/mL and final buffer compositions in the zero-dilution wells are summarized in Table 1.
The microplate was then incubated at 37 °C with shaking (200 rpm) for 40 min. Serial 10-fold dilutions were subsequently made with sterile HEPES buffer A (10 mM HEPES, 150 mM NaCl, pH 7.4). Each dilution (20 μL) was plated onto MH agar plates, which were then incubated at 37 °C overnight to give visible colonies. Inoculum size was indicated by control samples containing untreated bacteria. Each trial was performed in triplicate, and the reported results are the averages of two independent trials.
Hemolysis assays
Mouse blood was withdrawn from healthy mice obtained from the Animal Center of Anhui Medical University; the animal treatment was performed in compliance with the guidelines for the care and use of research animals established by the Animal Care and Use Committee at University of Science and Technology of China, and the experimental protocol was approved by the Animal Care and Use Committee at University of Science and Technology of China. Fresh mouse blood (200 μL) was washed with sterile HEPES buffer (10 mM HEPES, 150 mM NaCl, pH = 7.4) (800 μL) and washed for three times with sterile HEPES buffer via centrifuge at 900 rcf for 5 min, and the pellet was re-suspended into sterile HEPES buffer (1,000 μL) to yield the mouse red blood cell (mRBC) stock suspension for hemolysis assays. The mRBC stock suspension (200 μL) and MCl2-containing HEPES solution (400 μL) were added into each centrifuge cups. After the incubation at 37 °C for 40 min with shaking at 250 rpm, the centrifuge cups were centrifuged at 900 rcf for 5 min, and the supernatant (100 μL) of each cup was transferred into a well of a 96-well microplate. Hemolysis was monitored by measuring the absorbance of the released hemoglobin at optical density at 414 nm, OD414. Controls included HEPES buffer (300 μL) and mRBC suspension (200 μL) treated with triton X-100 (50%, 100 μL) to provide reference for 0% and 100% hemolysis, respectively. Each hemolysis assay trial was carried out in triplicate, and the reported results are the averages of two independent trials.
Statistical Analysis
Statistical comparisons were carried out by performing student t test analysis with the statistical software package BioMedCalc (version 2.9). p values of <0.05 and <0.01 indicate statistical difference and statistically significant difference, respectively.
Additional Information
How to cite this article: Xie, Y. and Yang, L. Calcium and Magnesium Ions Are Membrane-Active against Stationary-Phase Staphylococcus aureus with High Specificity. Sci. Rep. 6, 20628; doi: 10.1038/srep20628 (2016).
Acknowledgments
We thank Zhi Zhao and Kan Hu for assistance on fluorimeter experiments. This work was supported in part by the National Natural Science Foundation of China (Grants 11074178 and 21174138), the Youth Innovation Promotion Association of Chinese Academy of Sciences, and Anhui Education Department (KJ2013A267).
Footnotes
Author Contributions L.Y. conceived the idea, Y.X. conducted the research, and Y.X. and L.Y. analyzed the data and wrote the paper.
References
- Chambers H. F. & DeLeo F. R. Waves of resistance: Staphylococcus aureus in the antibiotic era. Nat. Rev. Microbiol. 7, 629–641, doi: 10.1038/nrmicro2200. (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore C. E., Segal S., Berendt A. R., Hill A. V. S. & Day N. P. J. Lack of association between toll-like receptor 2 polymorphisms and susceptibility to severe disease caused by Staphylococcus aureus. Clin. Diagn. Lab. Immunol. 11, 1194–1197, doi: 10.1128/Cdli.11.6.1194-1197.2004 (2004). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wertheim H. F. L. & Verbrugh H. A. Global prevalence of meticillin-resistant Staphylococcus aureus. Lancet 368, 1866 (2006). [DOI] [PubMed] [Google Scholar]
- Antibiotic Resistance Threats in the United States, 2013. Report by the U.S. Centers for Dieseas Control and Prevention.
- Shai Y. From Innate Immunity to de-Novo Designed Antimicrobial Peptides. Curr. Pharm. Des. 8, 715–725, doi: 10.2174/1381612023395367 (2002). [DOI] [PubMed] [Google Scholar]
- Yeaman M. R. & Yount N. Y. Mechanisms of Antimicrobial Peptide Action and Resistance. Pharmacol. Rev. 55, 27–55 (2003). [DOI] [PubMed] [Google Scholar]
- Wimley W. C. Describing the Mechanism of Antimicrobial Peptide Action with the Interfacial Activity Model. ACS Chem. Biol. 5, 905–917, doi: 10.1021/cb1001558 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hancock R. E. W. & Sahl H.-G. Antimicrobial and host-defense peptides as new anti-infective therapeutic strategies. Nat. Biotechnol. 24, 1551–1557, doi: 10.1038/nbt1267 (2006). [DOI] [PubMed] [Google Scholar]
- Brogden K. A. Antimicrobial Peptides: Pore Formers or Metabolic Inhibitors in Bacteria? Nat. Rev. Microbiol. 3, 238–250 (2005). [DOI] [PubMed] [Google Scholar]
- Zasloff M. Antimicrobial Peptides of Multicellular Organisms Nature 415, 389–395 (2002). [DOI] [PubMed] [Google Scholar]
- Schmidt N. W. et al. Criterion for Amino Acid Composition of Defensins and Antimicrobial Peptides Based on Geometry of Membrane Destabilization. J. Am. Chem. Soc. 133, 6720–6727, doi: 10.1021/ja200079a (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hurdle J. G., O’Neill A. J., Chopra I. & Lee ,. R. E. Targeting bacterial membrane function: an underexploited mechanism for treating persistent infections. Nat. Rev. Microbiol. 9, 62–75, doi: 10.1038/nrmicro2474 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oren Z. & Shai Y. Selective Lysis of Bacteria but Not Mammalian Cells by Diastereomers of Melittin: Structure-Function Study. Biochemistry 36, 1826 –1835 (1997). [DOI] [PubMed] [Google Scholar]
- Chen Y. et al. Rational Design of a-Helical Antimicrobial Peptides with Enhanced Activities and Specificity/Therapeutic Index. J. Biol. Chem. 280, 12316–12329 (2005). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Won H.-S., Jung S.-J., Kim H. E., Seo M.-D. & Lee B.-J. Systematic Peptide Engineering and Structural Characterization to Search for the Shortest Antimicrobial Peptide Analogue of Gaegurin 5. J. Biol. Chem. 279, 14784–14791 (2004). [DOI] [PubMed] [Google Scholar]
- Fernandez-Lopez S. et al. Antibacterial agents based on the cyclic d,l-[alpha]-peptide architecture. Nature 412, 452–455 (2001). [DOI] [PubMed] [Google Scholar]
- Hamuro Y., Schneider J. P. & DeGrado W. F. De Novo Design of Antibacterial beta-Peptides J. Am. Chem. Soc. 121, 12200–12201 (1999). [Google Scholar]
- Porter E. A., Wang X., Lee H.-S., Weisblum B. & Gellman S. H. Antibiotics: Non-haemolytic β-amino-acid Oligomers. Nature 404, 565–565 (2000). [DOI] [PubMed] [Google Scholar]
- Liu D. & DeGrado W. F. De Novo Design, Synthesis, and Characterization of Antimicrobial beta-Peptides J. Am. Chem. Soc. 123, 7553–7559 (2001). [DOI] [PubMed] [Google Scholar]
- Porter E. A., Weisblum B. & Gellman S. H. Mimicry of Host-Defense Peptides by Unnatural Oligomers: Antimicrobial β-Peptides. J. Am. Chem. Soc. 124, 7324–7330 (2002). [DOI] [PubMed] [Google Scholar]
- Hayouka Z. et al. Interplay among Subunit Identity, Subunit Proportion, Chain Length, and Stereochemistry in the Activity Profile of Sequence-Random Peptide Mixtures. J. Am. Chem. Soc. 135, 11748–11751, doi: 10.1021/ja406231b (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapireddy S. et al. RTD-1Mimic Containing γPNA Scaffold Exhibits Broad-Spectrum Antibacterial Activities. J. Am. Chem. Soc. 134, 4041–4044, doi: 10.1021/ja211867j (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patch J. A. & Barron A. E. Helical Peptoid Mimics of Magainin-2 Amide. J. Am. Chem. Soc. 125, 12092–12093 (2003). [DOI] [PubMed] [Google Scholar]
- Tew G. N. et al. De novo Design of Biomimetic Antimicrobial Polymers. Proc. Natl. Acad. Sci. USA 99, 5110–5114 (2002). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu D. et al. Nontoxic Membrane-Active Antimicrobial Arylamide Oligomers. Angew. Chem. Int. Ed. 43, 1158–1162 (2004). [DOI] [PubMed] [Google Scholar]
- Tew G. N., Clements D., Tang H., Arnt L. & Scott R. W. Antimicrobial activity of an abiotic host defense peptide mimic. Biochim. Biophys. Acta, Biomembranes 1758, 1387–1392 (2006). [DOI] [PubMed] [Google Scholar]
- Choi S. et al. De novo design and in vivo activity of conformationally restrained antimicrobial arylamide foldamers. Proc. Natl. Acad. Sci. USA 106, 6968–6973, doi: 10.1073/pnas.0811818106 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radzishevsky I. S. et al. Improved antimicrobial peptides based on acyl-lysine oligomers. Nat. Biotechnol. 25, 657–659, doi: 10.1038/nbt1309. (2007). [DOI] [PubMed] [Google Scholar]
- Thaker H. D., Cankaya A., Scott R. W. & Tew G. N. Role of Amphiphilicity in the Design of Synthetic Mimics of Antimicrobial Peptides with Gram-Negative Activity. ACS Med. Chem. Lett. 4, 481–485, doi: 10.1021/ml300307b (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuroda K. & DeGrado W. F. Amphiphilic Polymethacrylate Derivatives as Antimicrobial Agents. J. Am. Chem. Soc. 127, 4128–4129 (2005). [DOI] [PubMed] [Google Scholar]
- Palermo E. F., Sovadinova I. & Kuroda, K. Structural Determinants of Antimicrobial Activity and Biocompatibility in Membrane-Disrupting Methacrylamide Random Copolymers. Biomacromolecules 10, 3098–3107, doi: 10.1021/bm900784x (2009). [DOI] [PubMed] [Google Scholar]
- Lienkamp K. et al. Antimicrobial Polymers Prepared by ROMP with Unprecedented Selectivity: A Molecular Construction Kit Approach. J. Am. Chem. Soc. 130, 9836–9843, doi: 10.1021/ja801662y (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ilker M. F., Nüsslein K., Tew G. N. & Coughlin E. B. Tuning the Hemolytic and Antibacterial Activities of Amphiphilic Polynorbornene Derivatives. J. Am. Chem. Soc. 126, 15870–15875 (2004). [DOI] [PubMed] [Google Scholar]
- Mowery B. P. et al. Mimicry of Antimicrobial Host-Defense Peptides by Random Copolymers. J. Am. Chem. Soc. 129, 15474–15476, doi: 10.1021/ja077288d (2007). [DOI] [PubMed] [Google Scholar]
- Sambhy V., Peterson B. R. & Sen A. Antibacterial and Hemolytic Activities of Pyridinium Polymers as a Function of the Spatial Relationship between the Positive Charge and the Pendant Alkyl Tail. Angew. Chem. Int. Ed. 47, 1250–1254, doi: 10.1002/anie.200702287 (2008). [DOI] [PubMed] [Google Scholar]
- Oda Y., Kanaoka S., Sato T., Aoshima S. & Kuroda K. Block versus Random Amphiphilic Copolymers as Antibacterial Agents. Biomacromolecules 12, 3581–3591, doi: 10.1021/bm200780r (2011). [DOI] [PubMed] [Google Scholar]
- Qiao Y. et al. Highly dynamic biodegradable micelles capable of lysing Gram-positive and Gram-negative bacterial membrane. Biomaterials 33, 1146–1153, doi: http://dx.doi.org/10.1016/j.biomaterials.2011.10.020 (2012). [DOI] [PubMed] [Google Scholar]
- Jiang Y. et al. Acid-Activated Antimicrobial Random Copolymers: A Mechanism-Guided Design of Antimicrobial Peptide Mimics. Macromolecules 46, 3959–3964, doi: 10.1021/ma400484b (2013). [DOI] [Google Scholar]
- Yang L. et al. Synthetic Antimicrobial Oligomers Induce a Composition-Dependent Topological Transition in Membranes. J. Am. Chem. Soc. 129, 12141–12147 (2007). [DOI] [PubMed] [Google Scholar]
- Yang L. et al. Mechanism of a prototypical synthetic membrane-active antimicrobial: Efficient hole-punching via interaction with negative intrinsic curvature lipids Proc. Natl. Acad. Sci. USA 105, 20595–20600, doi: 10.1073/pnas.0806456105 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Som A., Yang L., Wong G. C. L. & Tew G. N. Divalent Metal Ion Triggered Membrane Activity in Cardiolipin Vesicles by Antimicrobial Molecule. J. Am. Chem. Soc. 131, 15102–15103 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu K. et al. A Critical Evaluation of Random Copolymer Mimesis of Homogeneous Antimicrobial Peptides. Macromolecules 46, 1908–1915, doi: 10.1021/ma302577e (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perros M. A sustainable model for antibiotics. Science 347, 1062–1064, doi: 10.1126/science.aaa3048 (2015). [DOI] [PubMed] [Google Scholar]
- Borkow G. & Gabbay J. Copper as a biocidal tool. Curr. Med. Chem. 12, 2163–2175, doi: 10.2174/0929867054637617 (2005). [DOI] [PubMed] [Google Scholar]
- Aghatabay N. M. et al. Synthesis, characterization and antimicrobial activity of Fe(II), Zn(II), Cd(II) and Hg(II) complexes with 2,6-bis(benzimidazol-2-yl) pyridine ligand. Eur. J. Med. Chem. 42, 205–213, doi: 10.1016/j.ejmech.2006.09.023 (2007). [DOI] [PubMed] [Google Scholar]
- Chernousova S. & Epple M. Silver as Antibacterial Agent: Ion, Nanoparticle, and Metal. Angew. Chem. Int. Ed. 52, 1636–1653, doi: 10.1002/anie.201205923 (2013). [DOI] [PubMed] [Google Scholar]
- Jung W. K. et al. Antibacterial Activity and Mechanism of Action of the Silver Ion in Staphylococcus aureus and Escherichia coli. Appl. Environ. Microbiol. 74, 2171–2178, doi: 10.1128/aem.02001-07 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cutinell C. & Galdiero F. Ion-Binding Properties of Cell Wall of Staphylococcus Aureus. J. Bacteriol. 93, 2022–& (1967). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Short S. A. & White D. C. Metabolism of Phosphatidylglycerol, Lysylphosphatidylglycerol, and Cardiolipin of Staphylococcus aureus. J. Bacteriol. 108, 219–226 (1971). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai M. et al. Staphylococcus aureus requires cardiolipin for survival under conditions of high salinity. BMC Microbiol. 11, 13 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rand R. P. & Sengupta S. Cardiolipin Forms Hexagonal Structures with Divalent Cations. Biochim. Biophys. Acta, Biomembranes 225, 484−+ (1972). [DOI] [PubMed] [Google Scholar]
- Som A. & Tew G. N. Influence of Lipid Composition on Membrane Activity of Antimicrobial Phenylene Ethynylene Oligomers. J. Phys. Chem. B 112, 3495–3502 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei G. et al. Lipid Composition Influences the Membrane-Disrupting Activity of Antimicrobial Methacrylate Co-polymers. J. Biomater. Sci. Polym. Ed. 22, 2041–2061, doi: 10.1163/092050610x530982 (2011). [DOI] [PubMed] [Google Scholar]
- Michael R., Niesman B. K. & Gholam A. Peyman. Encapsulation of Sodium Fluorescein for Dye Release Studies. Invest. Ophthalmol. Vis. Sci. 33, 2113–2119 (1992). [PubMed] [Google Scholar]
- Chin W. et al. Biodegradable Broad-Spectrum Antimicrobial Polycarbonates: Investigating the Role of Chemical Structure on Activity and Selectivity. Macromolecules 46, 8797–8807, doi: 10.1021/ma4019685 (2013). [DOI] [Google Scholar]
- Ferdani R. et al. Transport of chloride and carboxyfluorescein through phospholipid vesicle membranes by heptapeptide amphiphiles. Org. Biomol. Chem. 5, 2423–2432, doi: 10.1039/b705544g (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epand R. F. et al. Dual Mechanism of Bacterial Lethality for a Cationic Sequence-Random Copolymer that Mimics Host-Defense Antimicrobial Peptides. J. Mol. Biol. 379, 38–50 (2008). [DOI] [PubMed] [Google Scholar]
- Mukherjee S. et al. Antibacterial membrane attack by a pore-forming intestinal C-type lectin. Nature 505, 103–107, doi: 10.1038/nature12729 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yandek L. E. et al. Mechanism of the cell-penetrating peptide transportan 10 permeation of lipid bilayers. Biophys. J. 92, 2434–2444, doi: 10.1529/biophysj.106.100198 (2007). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delon A., Usson Y., Derouard J., Biben T. & Souchier C. Continuous photobleaching in vesicles and living cells: a measure of diffusion and compartmentation. Biophys. J. 90, 2548–2562, doi: 10.1529/biophysj.105.069815 (2006). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel H., Tscheka C. & Heerklotz H. Characterizing vesicle leakage by fluorescence lifetime measurements. Soft Matter 5, 2849, doi: 10.1039/b908524f (2009). [DOI] [Google Scholar]
- Breukink E. et al. Lipid II Is an Intrinsic Component of the Pore Induced by Nisin in Bacterial Membranes. J. Biol. Chem. 278, 19898–19903, doi: 10.1074/jbc.M301463200 (2003). [DOI] [PubMed] [Google Scholar]
- Richard Maskiewicz D. S. & Thomas C. Bruice. Chemiluminescent Reactions of Lucigenin.1.Reactions of Lucigenin with Hydrogen Peroxide. J. Am. Chem. Soc. 101, 5347–5354 (1978). [Google Scholar]
- Chen Y. A Mechanism for Tamoxifen-mediated Inhibition of Acidification. J. Biol. Chem. 274, 18364–18373, doi: 10.1074/jbc.274.26.18364 (1999). [DOI] [PubMed] [Google Scholar]
- William A., Harrell J., Marie Liesel Bergmeyer, Peter Y. Zavalij & Jeffery T. Davis. Ceraminde-Mediated Transport of Chloride and Bicarbonate Across Phospholipid Membranes. Chem. Commun. 46, 3950–3952 (2010). [DOI] [PubMed] [Google Scholar]
- Matin A., Lynch S. & Benoit M. Increased bacterial resistance and virulence in simulated microgravity and its molecular basis. Gravit. Space Res. 19, (2007). [Google Scholar]
- Anderl J. N., Zahller J., Roe F. & Stewart P. S. Role of nutrient limitation and stationary-phase existence in Klebsiella pneumoniae biofilm resistance to ampicillin and ciprofloxacin. Antimicrob. Agents Chemother. 47, 1251–1256, doi: 10.1128/Aac.47.4.1251-1256.2003 (2003). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamp J. A. F. O. d., Redai I. & Deenen L. L. M. v. Phospholipid Composition of Bacillus subtilis. J. Bacteriol. 99, 298–303 (1969). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto O., Ohira T., Alvarez K. & Fukuda M. Antibacterial characteristics of CaCO3-MgO composites. Mater. Sci. Eng. B Adv. 173, 208–212, doi: 10.1016/j.mseb.2009.12.007 (2010). [DOI] [Google Scholar]
- Sawai J. Quantitative evaluation of antibacterial activities of metallic oxide powders (ZnO, MgO and CaO) by conductimetric assay. J. Microbiol. Methods 54, 177–182, doi: 10.1016/S0167-7012(03)00037-X (2003). [DOI] [PubMed] [Google Scholar]
- Sawai J., Himizu K. & Yamamoto O. Kinetics of bacterial death by heated dolomite powder slurry. Soil Biol. Biochem. 37, 1484–1489, doi: 10.1016/j.soilbio.2005.01.011 (2005). [DOI] [Google Scholar]
- Sawai J. et al. Antibacterial characteristics of magnesium oxide powder. World J. Microbiol. Biotechnol. 16, 187–194, doi: 10.1023/A:1008916209784 (2000). [DOI] [Google Scholar]
- Sawai J., Shiga H. & Kojima H. Kinetic analysis of the bactericidal action of heated scallop-shell powder. Int. J. Food Microbiol. 71, 211–218, doi: 10.1016/S0168-1605(01)00619-5 (2001). [DOI] [PubMed] [Google Scholar]
- Sawai J., Shiga H. & Kojima H. Kinetic analysis of death of bacteria in CaO powder slurry. Int. Biodeter. Biodegr. 47, 23–26, doi: 10.1016/S0964-8305(00)00115-3 (2001). [DOI] [Google Scholar]
- Bae D. H., Yeon J. H., Park S. Y., Lee D. H. & Ha S. D. Bactericidal effects of CaO (scallop-shell powder) on foodborne pathogenic bacteria. Arch. Pharm. Res. 29, 298–301, doi: 10.1007/Bf02968574 (2006). [DOI] [PubMed] [Google Scholar]
- Siqueira J. F. & Lopes H. P. Mechanisms of antimicrobial activity of calcium hydroxide: a critical review. Int. Endod. J. 32, 361–369, doi: 10.1046/j.1365-2591.1999.00275.x (1999). [DOI] [PubMed] [Google Scholar]
- Sawai J. et al. Hydrogen peroxide as an antibacterial factor in zinc oxide powder slurry. J. Ferment. Bioeng. 86, 521–522, doi: 10.1016/S0922-338x(98)80165-7 (1998). [DOI] [Google Scholar]