Abstract
Objective
To compare the risk of incident hyperlipidemia in early rheumatoid arthritis (ERA) patients after initiation of various disease modifying anti-rheumatic drugs (DMARDs).
Methods
We conducted a cohort study using insurance claims data (2001–2012) in ERA patients. ERA was defined by the absence of any RA diagnosis or DMARD prescriptions for 12 months. Four mutually exclusive groups were defined based on DMARD initiation, TNF-α inhibitors ± non-biologic (nb) DMARDs, methotrexate ± non-hydroxycholorquine nbDMARDs, hydroxychloroquine ± non-methotrexate nbDMARDs, and other nbDMARDs only. The primary outcome was incident hyperlipidemia, defined by a diagnosis and a prescription for a lipid-lowering agent. For the subgroup of patients with laboratory results available, change in lipid levels was assessed. Multivariable Cox proportional hazard models and propensity score (PS) decile stratification with asymmetric trimming were used to control for confounding.
Results
Of the 17,145 ERA patients included in the study, 364 developed incident hyperlipidemia. The adjusted hazard ratios (95% CI) for hyperlipidemia were 1.41 (0.99–2.00) for TNF-α inhibitors, 0.81 (0.63–1.04) for hydroxychloroquine, and 1.33 (0.95–1.84) for other nbDMARDs compared with methotrexate in the full cohort, while 1.18 (0.80–1.73), 0.75 (0.58–0.98) and 1.41 (1.01–1.98), respectively in the PS trimmed cohort. In the subgroup analysis, hydroxychloroquine use showed significant reduction in low density lipoprotein (−8.9 mg/dl, 95% CI −15.8, −2.0), total cholesterol (−12.3 mg/dl, 95% CI −19.8, −4.8) and triglyceride (−19.5 mg/dl, 95% CI −38.7, −0.3) levels from baseline compared with methotrexate.
Conclusion
Use of hydroxychloroquine may be associated with a lower risk of hyperlipidemia among ERA patients.
INTRODUCTION
Epidemiological studies consistently demonstrate a higher risk of cardiovascular diseases (CVD) in patients with rheumatoid arthritis (RA) compared to the general population (1–3). Both traditional CVD risk factors and systemic inflammation likely contribute to the development of CVD in RA patients (4, 5). Hyperlipidemia is one of the most important modifiable risk factor for CVD in the general population (6). Despite the increased CVD risk in patients with RA, several studies suggest that RA patients may have lower total cholesterol and low density lipoprotein (LDL) compared to patients without RA (7, 8). Reports of inverse association between inflammatory markers and lipid parameters may explain this phenomenon (9, 10).
The comparative effect of various disease modifying anti-rheumatic drugs (DMARDs) on the risk of hyperlipidemia is poorly understood. The majority of evidence regarding DMARDs and hyperlipidemia in RA patients comes from small observational studies evaluating the effects of individual DMARD treatments on lipid levels. Several studies demonstrate reductions in LDL and total cholesterol after treatment with hydroxychloroquine (11–15). The data on the effect of methotrexate on lipid levels is inconsistent. A few studies report a neutral effect of methotrexate on LDL (14, 16), while a recent study observed an increase in LDL after methotrexate treatment (17). HDL levels are noted to increase after methotrexate treatment resulting in an improved atherogenic index (as defined by total cholesterol/HDL) (16, 17). A recent meta-analysis including data from 15 small observational cohort studies shows elevation of LDL and total cholesterol after treatment with tumor necrosis factor (TNF)-α inhibitors along with an increase in HDL levels; these changes stabilize over time resulting in no effect on the atherogenic index (18). Increases in LDL, total cholesterol and HDL resulting in improvement in the atherogenic index after initiation of TNF-α inhibitor treatment are also reported in a recent cohort study conducted using health insurance claims data (19).
CVD risk management in RA patients remains a significant concern and it is vital to understand the effect of DMARDs on various traditional CVD risk factors. To date, limited data are available comparing the risk of incident hyperlipidemia following treatment with DMARDs in RA patients. We therefore evaluated the association between various DMARDs and the risk of incident hyperlipidemia among patients with early RA. All subjects in the study were enrollees of two large U.S. commercial insurance programs. Based on the evidence present in the literature, we hypothesized that the use of hydroxychloroquine would be associated with a lower risk, while the use of TNF-α inhibitors would be associated with a higher risk of hyperlipidemia compared to the use of methotrexate.
METHODS
Study design and data source
We conducted a cohort study to evaluate the association between DMARD use and incident hyperlipidemia, using claims data (January 2001–September 2012) from two commercial US health plans - WellPoint and United HealthCare. These databases contain longitudinal claims information including medical diagnoses, procedures, hospitalizations, physician visits, and pharmacy dispensing on more than 40 million fully-insured subscribers with medical and pharmacy coverage across the United States. Both data sources also contain longitudinal information on outpatient laboratory test results for a subgroup of patients. These databases have been utilized in previous published studies (20–24). Personal identifiers were removed from the dataset before the analysis to protect subject confidentiality. The study protocol was approved by the Institutional Review Board of Brigham and Women’s Hospital.
Study cohort
We identified RA patients using a previously validated algorithm requiring at least 2 claims with ICD-9-CM diagnosis codes of 714.xx at least 7 days but no more than 365 days apart and at least 1 filled prescription for a DMARD after the first RA diagnosis. This algorithm had a positive predictive value of 86.2% in a validation study (25). In addition, we required the identified RA cases to have at least 365 days of continuous enrollment in their health plans prior to the first RA diagnosis for assessment of baseline characteristics. To minimize confounding by duration of RA and by duration of DMARD use, we excluded all patients who filled prescription for a DMARD in 365 days prior to the first observed RA diagnosis to focus on early RA cases who are naïve to DMARDs. The date of their first DMARD dispensing was defined as the index date and the period of 365 days prior to the index date was defined as the baseline period, during which all the covariates were measured. We excluded patients with a diagnosis of hyperlipidemia (ICD-9-CM code 272.xx) or use of any lipid lowering agents during the baseline period. We also excluded patients with existing CVD including myocardial infarction, angina, heart failure, cerebrovascular disease, or other forms of chronic heart disease. To study the effects of methotrexate and hydroxychloroquine individually, we further excluded patients who initiated both of these drugs on the index date. Finally, we excluded patients who started treatment with non-TNF-α inhibitor biologic agents (abatacept, rituximab, anakinra, and tocilizumab) to focus on biologic agents with the same mechanism of action.
We also identified subgroups of the patients who had at least one laboratory result available in the 365 days prior to the index date and at least one in the 365 days after the index date for each of the following tests: LDL, HDL, total cholesterol, and triglycerides. Individual test results were identified using logical observation identifiers names and codes (26).
Drug exposure
Patients were categorized into one of the four following mutually exclusive groups based on their DMARD initiation on the index date, 1) TNF-α inhibitors with or without other non-biologic DMARDs, 2) methotrexate without hydroxychloroquine nor TNF-α inhibitors, 3) hydroxychloroquine without methotrexate nor TNF-α inhibitors, and 4) other non-biologics without methotrexate, hydroxychloroquine nor TNF-α inhibitors. TNF-α inhibitors included adalimumab, certolizumab, etanercept, infliximab, and golimumab. Other non-biologic DMARDs included auranofin, injectable gold, penicillamine, sulfasalazine, minocycline, azathioprine, leflunomide, cyclophosphamide and cyclosporine. Methotrexate was selected as the reference exposure since it is the most commonly used DMARD for the treatment for RA. To ensure the mutual exclusivity of the groups during the entire follow-up period, we censored patients with the following DMARD switches: other non-biologic DMARDs to methotrexate or hydroxychloroquine or biologics, methotrexate to hydroxychloroquine or biologics, hydroxychroloquine to methotrexate or biologics, and TNF-α inhibitors to abatacept, rituximab, tocilizumab, or anakinra. Patients were followed until discontinuation of their index DMARD, outcome occurrence, disenrollment from the health plan, or study end point. Discontinuation was defined as no filled prescription for 90 days after accounting for the day supply of the previous prescription.
Study outcome
The primary outcome of interest was incident hyperlipidemia defined with at least one diagnosis of hyperlipidemia (ICD-9-CM code 272.xx) and a new prescription for a lipid lowering agent (including atorvastatin, fluvastatin, lovastatin, pravastatin, rosuvastatin, simvastatin, ezetimibe, cholestyramine, colestipol, colesevelam, gemfibrozil, fenofibrate, and clofibrate). The date of the first lipid lowering agent dispensing was defined as the outcome date.
The secondary outcome of interest was change in laboratory values of lipid parameters including LDL, HDL, total cholesterol and triglycerides in the subgroup of patients for whom we had these measures available. We assessed this change prior to initiation of any lipid lowering agents in these patients in order to evaluate the effect of DMARDs on lipid levels exclusively.
Covariates
Several covariates including patient age, gender, cardiovascular risk factors including hypertension, diabetes mellitus, smoking, and obesity, comorbidity score (27), antihypertensive medication use (angiotensin converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, calcium channel blockers, and diuretics), non-steroidal anti-inflammatory drug use, steroid use, any hospitalization, any emergency room visit, number of physician visits and number of prescriptions filled were measured during the baseline period. Obesity was determined using ICD-9 diagnosis code of 278.0x. Similarly, smoking status was ascertained by ICD-9 diagnosis codes of 305.1, 649.0x, 989.84 or V15.82, or use of anti-smoking drugs: varenicline, bupropion, nicotine or procedure codes indicating smoking cessation counselling or treatment (CPT codes- 99406, 99407, S9075, S9453). In a subgroup of the patients with outpatient laboratory data available, we also identified the pre-index levels of c-reactive protein (CRP) measured either using routine CRP testing or high-sensitivity CRP testing.
Statistical analysis
Descriptive statistics were used to summarize patient characteristics for the 4 exposure groups. Incidence rates for the development of hyperlipidemia were calculated for each exposure group and 95% confidence intervals (CI) were derived using Poisson distribution. Unadjusted survival curves were constructed using the Kaplan-Meier method stratified by exposure group. Cox proportional hazards (PH) models were used to generate crude and adjusted hazard ratios (HRs). The proportional hazard assumption was tested and met for all the models (28). An as-treated approach, achieved by the treatment-based censoring mechanisms discussed above, was used for the time to event analyses. To further control for confounding by indication, we used propensity score (PS) stratification method after asymmetric trimming as an additional analysis. This method has been shown to result in less unmeasured confounding by removing non-comparable patients between the exposure groups of interest from the analysis (29). Separate multivariable logistic regression models that contained all the patient factors that were available for every member of the full cohort (described under ‘Covariates’ above) calculated PS as the predicted probability of initiating each of the three exposures, hydroxychloroquine, TNF-α inhibitors, and other non-biologics, versus initiating methotrexate. We used cut-points of 2.5th percentile in patients treated with the exposure of interest (hydroxychloroquine, TNF-α inhibitors, or other non-biologics) and 97.5th percentile in patients treated with the reference treatment (methotrexate) of the PS distribution for asymmetric trimming (29). PS decile-stratified analysis was performed for each pairwise comparison after trimming.
For the secondary outcome analysis, pre-index mean lipid levels were compared to post-index mean lipid levels using paired t-tests. We then evaluated the effect of DMARD use on each individual lipid parameter using linear regression models with DMARD use as the exposure of interest and post-index laboratory parameter measurement as the outcome. In addition to the covariates listed above, the pre-index laboratory parameter measurement was also added to each model (30).
To evaluate the robustness of our findings, we conducted several sensitivity analyses. First, we analyzed the data by carrying forward the index DMARD exposure until the date of the outcome or health plan disenrollment or study end point to simulate an intention-to-treat (ITT) analysis. Next, to check for the possible impact of differential censoring on our findings, we repeated our main analysis (as treated) after truncating the follow-up at 12 months. Next, to evaluate the potential for time-varying confounding by steroid use, we modeled steroids use as a monthly time-varying covariate in an extended Cox regression model. Further, we controlled for baseline systemic inflammation in a subgroup of patients for whom we had pre-index CRP laboratory data available. Finally, we repeated our main analysis after excluding patients with traditional CVD risk factors, diabetes or hypertension, to rule out the possibility of differential intensities of follow-up (medical surveillance bias) (31).
RESULTS
Cohort Selection
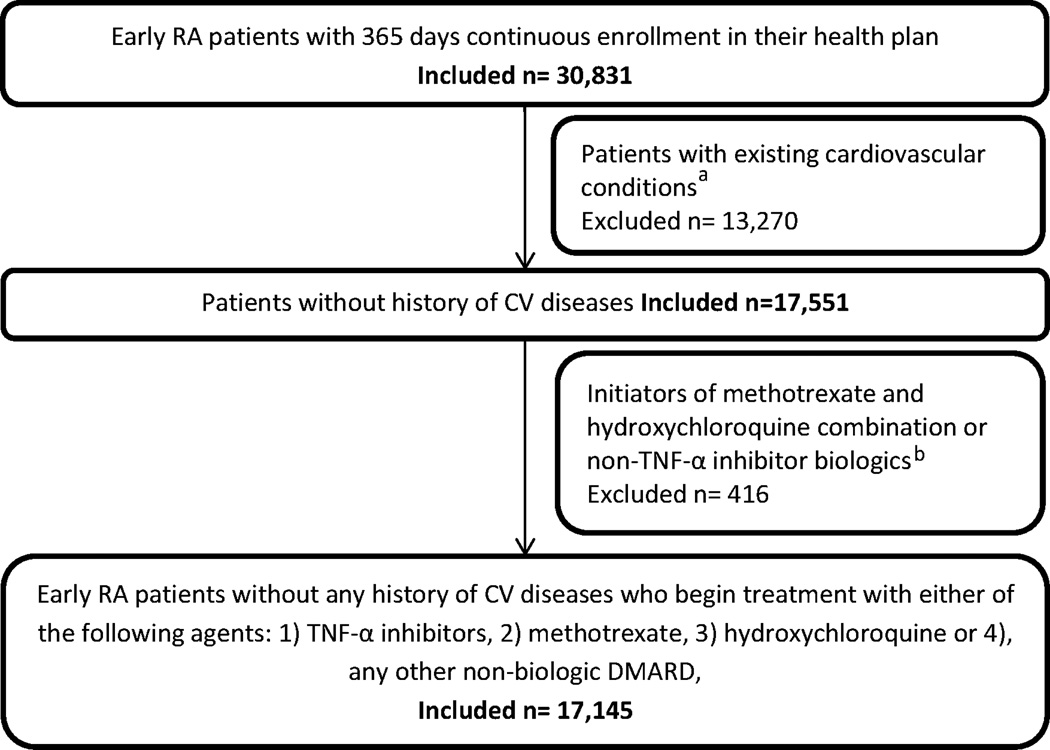
We identified 30,831 new RA patients who had at least 365 days of continuous enrollment prior to the index date. We then excluded patients with existing hyperlipidemia or cardiovascular disease or lipid lowering agent use (n=13,270) and patients beginning treatment with either methotrexate-hydroxychloroquine combination or non-TNF-α inhibitor biologics (n=416). Thus our final sample consisted of 17,145 new RA patients who initiated treatment with one of the four DMARD group of our interest (Figure 1). The majority (46.32%) of the patients in our cohort initiated treatment with methotrexate, followed by hydroxychloroquine (35.75%), other non-biologics (12.04%) and TNF-α inhibitors (5.89%). In the other non-biologics group, majority of the patients initiated treatment with sulfasalazine (75.6%), leflunomide (15.4%), or azathioprine (6.8%).
Figure 1.
Patient selection flow chart
a including hyperlipidemia or lipid lowering agent use or prior diagnosis of myocardial infarction, chronic heart failure, cerebrovascular events, angina, or other forms of chronic heart diseases
b Non-TNF-α-inhibitor biologics include abatacept, rituximab, anakinra, and tocilizumab.
Patient Characteristics
The four exposure groups had similar distributions of age, hypertension, and diabetes (Table 1). Frequency of obesity, smoking, cardiovascular drug use, and pain medication use was lower in TNF-α inhibitor initiators, while hydroxychroloquine initiators had a higher proportion of females. Among the subgroup of patients with pre-index outpatient CRP results available, hydroxychloroquine initiators had a lower proportion of patients with high CRP levels (defined as >3 mg/L). The distribution of CRP levels was similar across the rest of the three patient subgroups.
Table 1.
Baseline characteristics of the cohort by DMARD use
| Variable | n (%) | ||||
|---|---|---|---|---|---|
| TNF-α inhibitorsa | Methotrexate | Hydroxychloroquine | Other non-biologic DMARDsa |
||
| Total patients | 1010 (5.89) | 7941 (46.32) | 6130 (35.75) | 2064 (12.04) | |
| Female | 703 (69.60) | 5810 (73.16) | 5153 (84.06) | 1387 (67.20) | |
| Age (mean±SD) | 45.08 ± 13.71 | 47.81 ± 12.08 | 45.45 ± 12.12 | 45.29 ± 12.07 | |
| Follow-up days (mean±SD) | 471.22 ± 455.57 | 290.9 ± 333.24 | 284.05 ± 326.17 | 218.7 ± 263.42 | |
| Cardiovascular risk factors and comorbidities | |||||
| Hypertension | 190 (18.81) | 1760 (22.16) | 1311 (21.39) | 418 (20.25) | |
| Obesity | 29 (2.87) | 389 (4.9) | 269 (4.39) | 87 (4.22) | |
| Diabetes | 62 (6.14) | 449 (5.65) | 318 (5.19) | 109 (5.28) | |
| Smoking | 74 (7.33) | 793 (9.99) | 637 (10.39) | 232 (11.24) | |
| Comorbidity scoreb (mean±SD) | 0.32 ± 0.94 | 0.23 ± 0.89 | 0.34 ± 1.01 | 0.38 ± 1.15 | |
| Cardiovascular drug use | |||||
| ACE inhibitors | 49 (4.85) | 586 (7.38) | 418 (6.82) | 145 (7.03) | |
| ARBs | 32 (3.17) | 342 (4.31) | 285 (4.65) | 80 (3.88) | |
| Beta blockers | 48 (4.75) | 568 (7.15) | 450 (7.34) | 152 (7.36) | |
| Calcium channel blockers | 43 (4.26) | 456 (5.74) | 389 (6.35) | 113 (5.47) | |
| Diuretics | 70 (6.93) | 977 (12.3) | 709 (11.57) | 213 (10.32) | |
| Pain medications | |||||
| COX-2 inhibitors | 107 (10.59) | 1022 (12.87) | 718 (11.71) | 296 (14.34) | |
| Nonselective NSAIDs | 420 (41.58) | 4686 (59.01) | 3394 (55.37) | 1204 (58.33) | |
| Steroids | 431 (42.67) | 4529 (57.03) | 3068 (50.05) | 1130 (54.75) | |
| Cumulative steroid dose in milligramsc (mean±SD) | 1025.13 ± 1360 | 725.01 ± 1251 | 584.84 ± 706.76 | 775.79 ± 1104 | |
| Healthcare use in prior year | |||||
| Hospitalization | 143 (14.16) | 993 (12.50) | 774 (12.63) | 305 (14.78) | |
| Emergency room visit | 186 (18.42) | 1801 (22.68) | 1344 (21.93) | 471 (22.82) | |
| Number of distinct prescriptions filled (mean±SD) | 6.43 ± 5.82 | 7.39 ± 5.41 | 8.22 ± 6.07 | 8.26 ± 6.22 | |
| Number of physician visits (mean±SD) | 9.03 ± 7.81 | 8.43 ± 6.11 | 9.58 ± 6.73 | 9.87 ± 7.5 | |
| Laboratory data | |||||
| CRP tests ordered | 548 (54.25) | 4548 (57.27) | 3622 (59.08) | 1216 (58.91) | |
| CRP test results available (% of tests ordered) | 169 (30.83) | 1214 (26.69) | 957 (26.42) | 305 (25.08) | |
| CRP levels (% of results available) | |||||
| Low (<1 mg/L) | 34 (20.11) | 245 (20.18) | 291 (30.40) | 70 (22.95) | |
| Moderate (1–3 mg/L) | 37 (21.89) | 233 (19.19) | 229 (23.93) | 68 (22.29) | |
| High (>3 mg/L) | 98 (58.00) | 736 (60.62) | 437 (45.67) | 167 (54.76) | |
Abbreviations: ACE- Angiotensin converting enzyme, ARB- Angiotensin receptor bloockers, COX- Cyclo oxygenase, DMARDs- Disease modifying anti-rheumatic drugs, CRP- C-reactive protein, NSAIDS- non steroidal anti-inflammatory drugs, SD- standard deviation, TNF- Tumor necrosis factor
TNF-α inhibitors include adalimumab, certolizumab, etanercept, infliximab, and golimumab. Other non-biologic DMARDs include auranofin, injectable gold, penicillamine, sulfasalazine, minocycline, azathioprine, leflunomide, cyclophosphamide, and cyclosporine.
This score was computed after weighting 20 comorbid conditions and can range from −2 to 26. The range observed in our cohort was −2 to 14.
Cumulative dose measured among patients who used any steroid in the 365-day baseline period.
Risk of Incident Hyperlipidemia
Table 2 shows the number of new hyperlipidemia cases identified and total years of follow-up for each of the four exposure groups. The incidence rate for hyperlipidemia was the highest for other non-biologic initiators (36.4 cases per 1,000 person-years, 95% CI 26.5–48.7) and lowest for hydroxychloroquine (20.1 cases per 1,000 person-years, 95% CI 16.3–24.6). The unadjusted time to event curve demonstrated statistically significant differences (p for log-rank test=0.002) across the fours exposure groups (Appendix Figure 1). In the adjusted Cox regression (Table 2), a trend toward increased risk of hyperlipidemia was observed among TNF-α inhibitor initiators compared with methotrexate (HR 1.41, 95% CI 0.99–2.00). The HR for incident hyperlipidemia in other non-biologic DMARDs initiators was 1.33 (95% CI 0.95–1.84), while the HR in hydroxychloroquine initiators was 0.81 (95% CI 0.63–1.04) compared to methotrexate.
Table 2.
Incidence rates, unadjusted and adjusted risk of hyperlipidemia among early RA patients based on DMARD use- Full cohort
| DMARD use | Sample size |
Events | Follow-up (person- years) |
Incidence rate and 95% CIb (per 1000 person years) |
Unadjusted HR (95% CI) |
Adjustedc HR (95% CI) |
|---|---|---|---|---|---|---|
| Methotrexate | 7941 | 183 | 6329 | 28.9 (24.9–33.4) | Reference | Reference |
| TNF-α inhibitorsa | 1010 | 40 | 1303 | 30.7 (21.9–41.8) | 1.08 (0.76–1.52) | 1.41 (0.99–2.00) |
| Hydroxychloroquine | 6130 | 96 | 4770 | 20.1 (16.3–24.6) | 0.70 (0.54–0.89) | 0.81 (0.63–1.04) |
| Other non-biologic DMARDsa | 2064 | 45 | 1237 | 36.4 (26.5–48.7) | 1.25 (0.90–1.74) | 1.33 (0.95–1.84) |
Abbreviations: CI- Confidence interval, DMARDs- Disease modifying anti-rheumatic drugs, HR- Hazard ratio, TNF- Tumor necrosis factor.
TNF-α inhibitors include adalimumab, certolizumab, etanercept, infliximab, and golimumab. Other non-biologic DMARDs include auranofin, injectable gold, penicillamine, sulfasalazine, minocycline, azathioprine, leflunamide, cyclophosphamide and cyclosporine.
95% confidence interval derived from Poisson distribution.
Adjusted for age, gender, cardiovascular risk factors and comorbidities, cardiovascular drug use, pain medications and healthcare use in the prior year in a cox proportional hazard regression model.
PS distributions for the treatment groups in each comparison demonstrated substantial overlap (Appendix Figure 2). In the PS trimmed cohort (Table 3), hydroxychloroquine was associated with a reduction in the risk of hyperlipidemia compared to methotrexate (HR 0.75, 95% 0.58–0.98), while other non-biologic DMARDs were associated with an increase in the risk of hyperlipidemia compared to methotrexate (HR 1.41, 95% CI 1.01–1.98). Initiation of TNF-α inhibitor was not associated with an increase in the risk of hyperlipidemia compared with methotrexate in this analysis (HR 1.18, 95% CI 0.80–1.73).
Table 3.
Adjusted risk of hyperlipidemia based on DMARD use- Propensity score trimmed cohort
| Comparisona | Treatment | Sample size |
Events | HR (95% CI)b |
|---|---|---|---|---|
| TNF-α inhibitors vs Methotrexate | Methotrexate | 7177 | 158 | Reference |
| TNF-α inhibitors | 874 | 33 | 1.18 (0.80–1.73) | |
| Hydroxychloroquine vs Methotrexate | Methotrexate | 7232 | 164 | Reference |
| Hydroxychloroquine | 5632 | 91 | 0.75 (0.58–0.98) | |
| Other non-biologic DMARDs vs Methotrexate | Methotrexate | 7205 | 160 | Reference |
| Other DMARDs | 1893 | 44 | 1.41 (1.01–1.98) |
Abbreviations: CI- Confidence interval, DMARDs- Disease modifying anti-rheumatic drugs, HR- Hazard ratio, TNF- Tumor necrosis factor.
TNF-α inhibitors include adalimumab, certolizumab, etanercept, infliximab, and golimumab. Other non-biologic DMARDs include auranofin, injectable gold, penicillamine, sulfasalazine, minocycline, azathioprine, leflunamide, cyclophosphamide and cyclosporine.
Propensity score decile stratification was used to derive hazard ratios after trimming.
Changes in blood lipid levels
Methotrexate and TNF-α inhibitor treatment groups had non-significant changes in LDL and total cholesterol from baseline, while hydroxychloroquine group showed significant and substantial reduction in LDL and total cholesterol after treatment (Table 4). HDL levels significantly increased from baseline after treatment with methotrexate and hydroxychloroquine. In the adjusted analyses, hydroxychloroquine was associated with statistically significant reductions in mean LDL, total cholesterol and triglycerides compared to methotrexate.
Table 4.
Changes in lipid parameters based on DMARD use
|
Parameter (unit) |
DMARD use |
Cohort size |
Pre-index mean |
Post-index mean |
Change from pre-index to post-index |
Post-index mean change in lipid values compared to methotrexatec |
95% CI |
|---|---|---|---|---|---|---|---|
| LDL (mg/dl) |
Methotrexate | 129 | 112.1 | 111.8 | −0.3 | Ref. | – |
| TNF-α inhibitorsa | 25 | 113.6 | 115.6 | 2.0 | 2.7 | −8.3,13.7 | |
| Hydroxychloroquine | 101 | 113.4 | 103.9 | −9.5b | −8.9 | −15.8,−2.0d | |
| Other non-biologic DMARDsa | 28 | 123.2 | 121.6 | −1.6 | 1.1 | −9.3,11.5 | |
| HDL (mg/dl) |
Methotrexate | 130 | 56.5 | 59.2 | 2.7b | Ref. | – |
| TNF-α inhibitorsa | 28 | 53.0 | 52.5 | −0.5 | −3.5 | −9.1,2.1 | |
| Hydroxychloroquine | 100 | 56.8 | 59.5 | 2.7b | −0.3 | −4.0,3.3 | |
| Other non-biologic DMARDsa | 25 | 52.5 | 51.2 | −1.3 | −4.8 | −10.5,1.0 | |
| Total Cholesterol (mg/dl) |
Methotrexate | 149 | 189.3 | 193.7 | 4.4 | Ref. | – |
| TNF-α inhibitorsa | 32 | 196.3 | 200.8 | 4.5 | 1.6 | −10,13.3 | |
| Hydroxychloroquine | 121 | 196.1 | 187.1 | −9.0b | −12.3 | −19.8,−4.8d | |
| Other non-biologic DMARDsa | 33 | 205.3 | 197.6 | −7.7 | −8.9 | −20.3,2.4 | |
| Triglycerides (mg/dl) |
Methotrexate | 145 | 113.8 | 128.0 | 14.2b | Ref. | – |
| TNF-α inhibitorsa | 31 | 134.7 | 142.0 | 7.3 | 0.0 | −29.6,29.6 | |
| Hydroxychloroquine | 116 | 126.3 | 114.2 | −12.1b | −19.5 | −38.7,−0.3d | |
| Other non-biologic DMARDsa | 33 | 156.7 | 132.0 | −24.7 | −25.3 | −53.9,3.2 |
Abbreviations: CI- Confidence interval, DMARDs- Disease modifying anti-rheumatic drugs, HDL- High density lipoproteins, LDL- Low density lipoproteins, TNF- Tumor necrosis factor
TNF-α inhibitors include adalimumab, certolizumab, etanercept, infliximab, and golimumab. Other non-biologic DMARDs include auranofin, injectable gold, penicillamine, sulfasalazine, minocycline, azathioprine, leflunamide, cyclophosphamide and cyclosporine.
p<0.05 compared to baseline mean in a paired t-test.
Differences adjusted for age, gender, cardiovascular risk factors and comorbidities, cardiovascular drug use, pain medications, healthcare use in the prior year, and pre-index values in a linear regression model.
p<0.05 compared to methotrexate in a linear regression model.
Sensitivity Analyses
The results of our sensitivity analyses using an ITT approach, truncated follow-up at 12 months, steroids use as time-varying covariate and, patients without traditional CVD risk factors were similar to our original findings from the full cohort (Figure 2). However, the analysis where we controlled for CRP resulted estimates with very wide confidence intervals because of reduced sample size.
Figure 2.
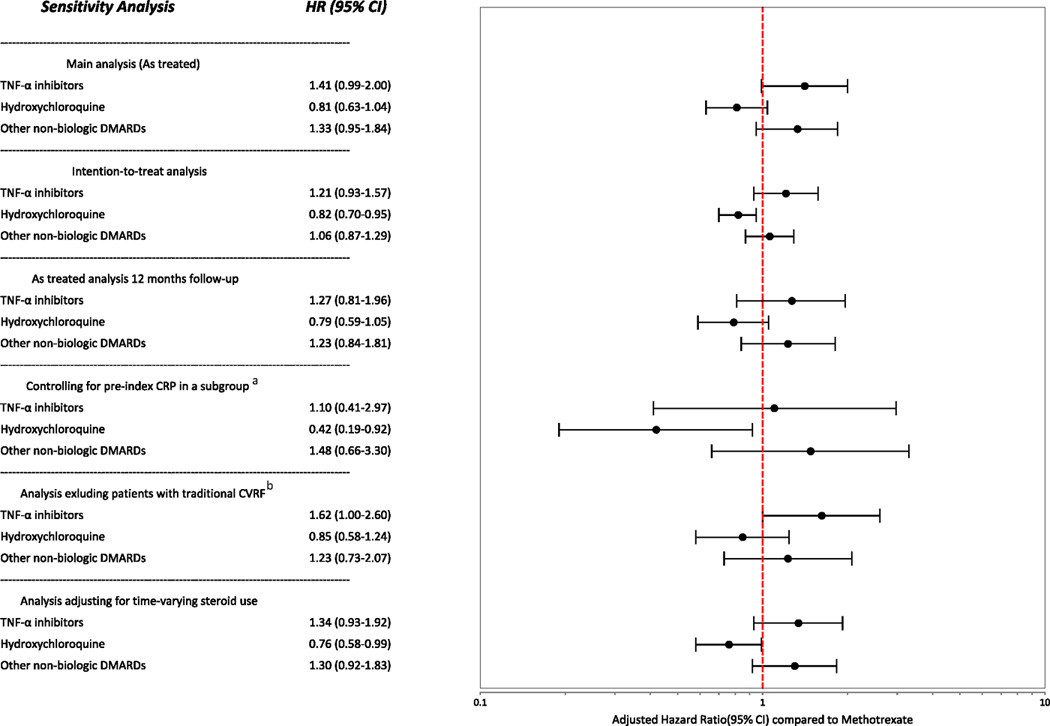
Sensitivity analyses
a This subgroup analysis was conducted in 2,644 patients for whom we have pre-index hsCRP results available (See Table 1). Total number for events for this group was 51.
b CVRF- Cardiovascular risk factors. This analysis was conducted after excluding all the patients with a pre-index diagnosis of diabetes or hypertension or pre-index use of any cardiovascular medications (sample size=11,613, number of events=164).
DISCUSSION
In this large cohort of patients with early RA, we observed that hydroxychlorquine use was associated with a trend towards reduced risk of hyperlipidemia when compared to methotrexate. In line with these findings, we observed that hydroxychloroquine use was associated with significant reduction in total cholesterol and LDL levels when compared to methotrexate in a subgroup analysis. Meanwhile, our primary multivariable analysis showed a trend toward increased risk of hyperlipidemia in initiators of TNF-α inhibitors compared to methotrexate. However, this association was attenuated in the PS stratified analysis which excluded non-comparable patients from each pairwise comparison.
Hydroxychloroquine may be associated with a reduction in LDL and total cholesterol based on its potential role in cholesterol metabolism (32). This hypothesis has been evaluated in several studies of RA patients (11–15) and systemic lupus erythematosus patients (33, 34) and results of those studies have been generally concordant suggesting significant reduction in LDL and total cholesterol after treatment with hydroxychloroquine. Our study provides further support to this hypothesis by documenting the potential association between hydroxychloroquine and a reduced risk of hyperlipidemia. This finding of lower trend for the risk of hyperlipidemia after hydroxychloroquine treatment compared to methotrexate was consistent across a variety of sensitivity analyses and was statistically significant in the PS trimmed cohort (Table 3), lending further credence to this observation.
The discrepancy in the effect of TNF-α inhibitors on the risk of hyperlipidemia between the full cohort and the PS trimmed cohort deserves discussion. We presented the estimates from the full cohort as the primary analysis in order to utilize data from each early RA patient who initiated treatment with a DMARD. However, while interpreting the estimates from the full cohort, one must be cautious of the possibility of residual confounding. The PS trimmed cohort excludes patients that are non-comparable between the exposure groups from each pairwise comparison and thus further tries to minimize residual confounding (29). However, since the PS trimmed cohort is not a clinically well-defined cohort, the generalizability of its finding may be an issue. Future research using clinical data on RA severity may further reduce confounding by indication which is inherent in all observational studies.
The relationship between TNF-α inhibitors and lipids in RA patients is complex. Two previous meta-analyses observed an increase in total cholesterol levels after TNF-α inhibitors treatment (18, 35). It has been suggested that higher levels of inflammation may reduce total cholesterol levels in RA patients, and control of inflammation with TNF-α inhibitors may lead to “normalization” of these levels (36). This hypothesis is supported by data demonstrating no changes in lipid levels among non-responders of TNF-α inhibitors (37). Control of inflammation associated with TNF-α inhibitor use is linked with physiologic changes reducing CVD risk, e.g. improved endothelial function and lower insulin resistance (38, 39). Thus, the control of inflammation with TNF-α inhibitors may offset any increased CVD risk from modest elevations in total cholesterol levels, thereby resulting in a net reduced risk for CVD (40).
Although the role of inflammation in the risk of CVD in RA has gained the most attention over the last decade, traditional cardiovascular risk factors such a hyperlipidemia, diabetes and hypertension, continue to play crucial role in the development of CVD in RA patients (5). Therefore, understanding RA treatments associated with favorable changes in traditional CV risk factors can inform CV risk management in RA. In line with previous studies, this study supports the potential role of hydroxychloroquine, alone or in combination with other DMARDs in reducing the risk of hyperlipidemia in RA patients.
The present study has several strengths. First, this is the only cohort study, to our knowledge, that compares the risk of incident hyperlipidemia between individual DMARDs. Second, the study findings are generalizable as the study population is representative of the US commercially insured population. Third, we used rigorous pharmacoepidemiology methods such as the new-user design to minimize prevalent user bias arising from ignoring the effect of differential duration of drug use on the outcome (41) and PS stratification to address confounding by indication (29). We also excluded users of lipid lowering agents at baseline to avoid mixing of effects of these agents with the effect of DMARDs on hyperlipidemia. Further, we designed several sensitivity analyses to evaluate the robustness of our findings. Results from the majority of our sensitivity analyses were similar to the main analysis providing evidence against biases due to changes in treatments post-index (ITT analysis), differential censoring (analysis truncated at 12 months for everyone), and differential surveillance (analysis excluding patients with traditional CVD risk factors). However, we did observe unstable estimates with wide confidence intervals for CRP controlled analyses. We postulate that the differences between estimates from this analysis and our main results may be attributed to the small subgroups of patients included in this analysis. Therefore, we advise readers to be cautious interpreting findings from this sensitivity analysis. Finally, we conducted a secondary analysis of patients with results from lipid parameter laboratory tests to confirm our findings.
This study also has several limitations. First, as with any other observational study, confounding by indication is possible. To address this limitation, we used an active comparator (i.e. methotrexate) for all our comparisons and used PS stratification method after asymmetric trimming as an additional analysis. Moreover, concerns of confounding by indication are limited while evaluating unintended treatment effects as in our case (42). Second, although our multivariable analyses were adjusted for more than 15 covariates, this study may be subject to residual confounding by factors not captured in the claims data such as body weight, physical activity or and dietary habits. As there was no data available on disease activity measures in the study database, our study may suffer from residual confounding by disease activity if there is a strong association between disease activity and lipid levels. The measured confounders for disease activity, such as steroid use in the full cohort and CRP levels in the subgroup with laboratory data, suggested lower disease activity among patients treated with hydroxychloroquine. Therefore, this limitation may be an important one for the hydroxychloroquine-methotrexate comparison. Third, as we defined RA, hyperlipidemia and other covariates based on diagnosis codes and/or prescription, misclassification bias is possible. To minimize this bias, we used a validated algorithm to identify RA (25) and combined medical claims with pharmacy claims to define the outcome of interest, hyperlipidemia. Finally, the subgroup analysis included very small number of patients with lipid levels available for TNF-α inhibitors and other non-biologic DMARDs. Therefore, the subgroup analysis may be underpowered to detect changes in lipid levels for these two exposure groups.
In conclusion, initiation of hydroxychloroquine may be associated with a lower risk of incident hyperlipidemia among early RA patients compared with methotrexate initiation. A possible increase in the risk of hyperlipidemia in TNF-α inhibitor initiators was noted in our primary analysis, but not in the PS stratified and subgroup analysis. More research is needed to evaluate the effect of TNF-α inhibitors on the risk of hyperlipidemia.
Supplementary Material
SIGNIFICANCE AND INNOVATIONS.
Patients with rheumatoid arthritis are at a higher risk of developing cardiovascular diseases (CVD). Hyperlipidemia is one of the most important CVD risk factors. This study is the first attempt to longitudinally evaluate the comparative risk of developing hyperlipidemia after initiation of various disease modifying anti-rheumatic drugs.
This study reveals that use of hydroxychloroquine may be associated with a lower risk of developing hyperlipidemia in patients with early RA compared to methotrexate.
Acknowledgments
Conflict of interest/Financial disclosures:
Dr. Solomon is supported by the NIH grants K24 AR055989, P60 AR047782, and R01 AR056215. Solomon receives research grants from Amgen and Lilly. He serves in unpaid roles on studies sponsored by Pfizer, Novartis, Lilly, and Bristol Myers Squibb. He also receives royalties from UpToDate.com.
Dr. Kim is supported by the NIH grant K23 AR059677. She receives research support from Pfizer and tuition support for the Pharmacoepidemiology Program at the Harvard School of Public Health partially funded by the Pharmaceutical Research and Manufacturers of America (PhRMA) foundation.
Dr. Liao is supported by the NIH grant K08 AR060257 and the Harold and Duval Bowen Fund.
Dr. Desai reports owning Biogen Idec stock due to spouse’s employment.
Funding:
This study was not funded by any institution.
Other acknowledgements:
We acknowledge the contributions of Jun Liu, MD, MSc (Division of Pharmacoepidemiology and Pharamcoeconomics, Brigham and Women’s Hospital) in data management and cleaning.
REFERENCES
- 1.Maradit-Kremers H, Crowson CS, Nicola PJ, Ballman KV, Roger VL, Jacobsen SJ, et al. Increased unrecognized coronary heart disease and sudden deaths in rheumatoid arthritis: a population-based cohort study. Arthritis Rheum. 2005;52(2):402–411. doi: 10.1002/art.20853. [DOI] [PubMed] [Google Scholar]
- 2.Solomon DH, Karlson EW, Rimm EB, Cannuscio CC, Mandl LA, Manson JAE, et al. Cardiovascular morbidity and mortality in women diagnosed with rheumatoid arthritis. Circulation. 2003;107(9):1303. doi: 10.1161/01.cir.0000054612.26458.b2. [DOI] [PubMed] [Google Scholar]
- 3.Fischer LM, Schlienger RG, Matter C, Jick H, Meier CR. Effect of rheumatoid arthritis or systemic lupus erythematosus on the risk of first-time acute myocardial infarction. Am J Cardiol. 2004;93(2):198–200. doi: 10.1016/j.amjcard.2003.09.037. [DOI] [PubMed] [Google Scholar]
- 4.Solomon DH, Kremer J, Curtis JR, Hochberg MC, Reed G, Tsao P, et al. Explaining the cardiovascular risk associated with rheumatoid arthritis: traditional risk factors versus markers of rheumatoid arthritis severity. Ann Rheum Dis. 2010;69(11):1920–1925. doi: 10.1136/ard.2009.122226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kremers HM, Crowson CS, Therneau TM, Roger VL, Gabriel SE. High ten-year risk of cardiovascular disease in newly diagnosed rheumatoid arthritis patients: a population-based cohort study. Arthritis Rheum. 2008;58(8):2268–2274. doi: 10.1002/art.23650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wilson PW, D'Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. Prediction of coronary heart disease using risk factor categories. Circulation. 1998;97(18):1837–1847. doi: 10.1161/01.cir.97.18.1837. [DOI] [PubMed] [Google Scholar]
- 7.Myasoedova E, Crowson CS, Kremers HM, Fitz-Gibbon PD, Therneau TM, Gabriel SE. Total cholesterol and LDL levels decrease before rheumatoid arthritis. Ann Rheum Dis. 2010;69(7):1310–1314. doi: 10.1136/ard.2009.122374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liao KP, Cai T, Gainer VS, Cagan A, Murphy SN, Liu C, et al. Lipid and lipoprotein levels and trends in rheumatoid arthritis compared to the general population. Arthritis Care Res (Hoboken) 2013 doi: 10.1002/acr.22091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boyer JF, Bongard V, Cantagrel A, Jamard B, Gottenberg JE, Mariette X, et al. Link between traditional cardiovascular risk factors and inflammation in patients with early arthritis: results from a French multicenter cohort. Arthritis Care Res (Hoboken) 2012;64(6):872–880. doi: 10.1002/acr.21623. [DOI] [PubMed] [Google Scholar]
- 10.Hashizume M, Yoshida H, Koike N, Suzuki M, Mihara M. Overproduced interleukin 6 decreases blood lipid levels via upregulation of very-low-density lipoprotein receptor. Ann Rheum Dis. 2010;69(4):741–746. doi: 10.1136/ard.2008.104844. [DOI] [PubMed] [Google Scholar]
- 11.Munro R, Morrison E, McDonald AG, Hunter JA, Madhok R, Capell HA. Effect of disease modifying agents on the lipid profiles of patients with rheumatoid arthritis. Ann Rheum Dis. 1997;56(6):374–377. doi: 10.1136/ard.56.6.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morris SJ, Wasko MC, Antohe JL, Sartorius JA, Kirchner HL, Dancea S, et al. Hydroxychloroquine use associated with improvement in lipid profiles in rheumatoid arthritis patients. Arthritis Care Res (Hoboken) 2011;63(4):530–534. doi: 10.1002/acr.20393. [DOI] [PubMed] [Google Scholar]
- 13.Wallace DJ, Metzger AL, Stecher VJ, Turnbull BA, Kern PA. Cholesterol-lowering effect of hydroxychloroquine in patients with rheumatic disease: reversal of deleterious effects of steroids on lipids. Am J Med. 1990;89(3):322–326. doi: 10.1016/0002-9343(90)90345-e. [DOI] [PubMed] [Google Scholar]
- 14.Seriolo B, Fasciolo D, Garnero A, Accardo S. The influence of hydroxychloroquine, methotrexate and the combination of both on lipid profile in patients with rheumatoid arthritis (Abstract) Arthritis Rheum. 1999;1999:S81-S. [Google Scholar]
- 15.Kerr G, Aujero M, Richards J, Sayles H, Davis L, Cannon G, et al. Associations of hydroxychloroquine use with lipid profiles in rheumatoid arthritis: Pharmacologic implications. Arthritis Care Res (Hoboken) 2014 doi: 10.1002/acr.22341. Published ahead of print. [DOI] [PubMed] [Google Scholar]
- 16.Georgiadis AN, Voulgari PV, Argyropoulou MI, Alamanos Y, Elisaf M, Tselepis AD, et al. Early treatment reduces the cardiovascular risk factors in newly diagnosed rheumatoid arthritis patients. Semin Arthritis Rheum. 2008;38(1):13–19. doi: 10.1016/j.semarthrit.2007.09.008. [DOI] [PubMed] [Google Scholar]
- 17.Navarro-Millan I, Charles-Schoeman C, Yang S, Bathon JM, Bridges SL, Jr, Chen L, et al. Changes in lipoproteins associated with methotrexate or combination therapy in early rheumatoid arthritis: results from the treatment of early rheumatoid arthritis trial. Arthritis Rheum. 2013;65(6):1430–1438. doi: 10.1002/art.37916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van Sijl AM, Peters MJ, Knol DL, de Vet RH, Sattar N, Dijkmans BA, et al. The effect of TNF-alpha blocking therapy on lipid levels in rheumatoid arthritis: a meta-analysis. Semin Arthritis Rheum. 2011;41(3):393–400. doi: 10.1016/j.semarthrit.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 19.Curtis JR, John A, Baser O. Dyslipidemia and changes in lipid profiles associated with rheumatoid arthritis and initiation of anti-tumor necrosis factor therapy. Arthritis Care Res (Hoboken) 2012;64(9):1282–1291. doi: 10.1002/acr.21693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Patorno E, Bohn RL, Wahl PM, Avorn J, Patrick AR, Liu J, et al. Anticonvulsant medications and the risk of suicide, attempted suicide, or violent death. JAMA. 2010;303(14):1401–1409. doi: 10.1001/jama.2010.410. [DOI] [PubMed] [Google Scholar]
- 21.Solomon DH, Massarotti E, Garg R, Liu J, Canning C, Schneeweiss S. Association between disease-modifying antirheumatic drugs and diabetes risk in patients with rheumatoid arthritis and psoriasis. JAMA. 2011;305(24):2525–2531. doi: 10.1001/jama.2011.878. [DOI] [PubMed] [Google Scholar]
- 22.Schneeweiss S, Doherty M, Zhu S, Funch D, Schlienger RG, Fernandez-Vidaurre C, et al. Topical treatments with pimecrolimus, tacrolimus and medium- to high-potency corticosteroids, and risk of lymphoma. Dermatology. 2009;219(1):7–21. doi: 10.1159/000209289. [DOI] [PubMed] [Google Scholar]
- 23.Seeger JD, Loughlin J, Eng PM, Clifford CR, Cutone J, Walker AM. Risk of thromboembolism in women taking ethinylestradiol/drospirenone and other oral contraceptives. Obstet Gynecol. 2007;110(3):587–593. doi: 10.1097/01.AOG.0000279448.62221.a8. [DOI] [PubMed] [Google Scholar]
- 24.Wang FT, Mast TC, Glass RJ, Loughlin J, Seeger JD. Effectiveness of the pentavalent rotavirus vaccine in preventing gastroenteritis in the United States. Pediatrics. 2010;125(2):e208–e213. doi: 10.1542/peds.2009-1246. [DOI] [PubMed] [Google Scholar]
- 25.Kim SY, Servi A, Polinski JM, Mogun H, Weinblatt ME, Katz JN, et al. Validation of rheumatoid arthritis diagnoses in health care utilization data. Arthritis Res Ther. 2011;13(1):R32. doi: 10.1186/ar3260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schneeweiss S, Rassen JA, Glynn RJ, Myers J, Daniel GW, Singer J, et al. Supplementing claims data with outpatient laboratory test results to improve confounding adjustment in effectiveness studies of lipid-lowering treatments. BMC Med Res Methodol. 2012;12:180. doi: 10.1186/1471-2288-12-180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gagne JJ, Glynn RJ, Avorn J, Levin R, Schneeweiss S. A combined comorbidity score predicted mortality in elderly patients better than existing scores. J Clin Epidemiol. 2011;64(7):749–759. doi: 10.1016/j.jclinepi.2010.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Allison PD. Survival analysis using SAS: a practical guide. SAS Institute; 2012. [Google Scholar]
- 29.Stürmer T, Rothman KJ, Avorn J, Glynn RJ. Treatment effects in the presence of unmeasured confounding: dealing with observations in the tails of the propensity score distribution—a simulation study. Am J Epidemiol. 2010;172(7):843–854. doi: 10.1093/aje/kwq198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Morgan SL, Winship C. Counterfactuals and causal inference: Methods and principles for social research. Cambridge University Press; 2007. [Google Scholar]
- 31.Sica GT. Bias in Research Studies. Radiology. 2006;238(3):780–789. doi: 10.1148/radiol.2383041109. [DOI] [PubMed] [Google Scholar]
- 32.Beynen AC, van der Molen AJ, Geelen MJ. Inhibition of hepatic cholesterol biosynthesis by chloroquine. Lipids. 1981;16(6):472–474. doi: 10.1007/BF02535017. [DOI] [PubMed] [Google Scholar]
- 33.Kavanaugh A, Adams-Huet B, Jain R, Denke M, McFarlin J. Hydroxychloroquine Effects on Lipoprotein Profiles (the HELP trial): A Double-Blind, Randomized, Placebo-Controlled, Pilot Study In Patients With Systemic Lupus Erythematosus. J Clin Rheumatol. 1997;3(1):3–8. [PubMed] [Google Scholar]
- 34.Cairoli E, Rebella M, Danese N, Garra V, Borba EF. Hydroxychloroquine reduces low-density lipoprotein cholesterol levels in systemic lupus erythematosus: a longitudinal evaluation of the lipid-lowering effect. Lupus. 2012;21(11):1178–1182. doi: 10.1177/0961203312450084. [DOI] [PubMed] [Google Scholar]
- 35.Daien CI, Duny Y, Barnetche T, Daures JP, Combe B, Morel J. Effect of TNF inhibitors on lipid profile in rheumatoid arthritis: a systematic review with meta-analysis. Ann Rheum Dis. 2012;71(6):862–868. doi: 10.1136/annrheumdis-2011-201148. [DOI] [PubMed] [Google Scholar]
- 36.Choy E, Sattar N. Interpreting lipid levels in the context of high-grade inflammatory states with a focus on rheumatoid arthritis: a challenge to conventional cardiovascular risk actions. Ann Rheum Dis. 2009;68(4):460–469. doi: 10.1136/ard.2008.101964. [DOI] [PubMed] [Google Scholar]
- 37.Jamnitski A, Visman IM, Peters MJ, Dijkmans BA, Voskuyl AE, Nurmohamed MT. Beneficial effect of 1-year etanercept treatment on the lipid profile in responding patients with rheumatoid arthritis: the ETRA study. Ann Rheum Dis. 2010;69(11):1929–1933. doi: 10.1136/ard.2009.127597. [DOI] [PubMed] [Google Scholar]
- 38.Hurlimann D, Forster A, Noll G, Enseleit F, Chenevard R, Distler O, et al. Anti-tumor necrosis factor-alpha treatment improves endothelial function in patients with rheumatoid arthritis. Circulation. 2002;106(17):2184–2187. doi: 10.1161/01.cir.0000037521.71373.44. [DOI] [PubMed] [Google Scholar]
- 39.Tam LS, Tomlinson B, Chu TT, Li TK, Li EK. Impact of TNF inhibition on insulin resistance and lipids levels in patients with rheumatoid arthritis. Clinical rheumatology. 2007;26(9):1495–1498. doi: 10.1007/s10067-007-0539-8. [DOI] [PubMed] [Google Scholar]
- 40.Solomon DH, Curtis JR, Saag KG, Lii J, Chen L, Harrold LR, et al. Cardiovascular risk in rheumatoid arthritis: comparing TNF-alpha blockade with nonbiologic DMARDs. Am J Med. 2013;126(8):730 e9–730 e17. doi: 10.1016/j.amjmed.2013.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ray WA. Evaluating medication effects outside of clinical trials: new-user designs. Am J Epidemiol. 2003;158(9):915–920. doi: 10.1093/aje/kwg231. [DOI] [PubMed] [Google Scholar]
- 42.Psaty BM, Siscovick DS. Minimizing bias due to confounding by indication in comparative effectiveness research: the importance of restriction. JAMA. 2010;304(8):897–898. doi: 10.1001/jama.2010.1205. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.