Abstract
Cardiac magnetic resonance imaging of the pressure overloaded right ventricle (RV) of precapillary pulmonary hypertension (PH) patients, exhibits late gadolinium enhancement at the interventricular insertion regions, a phenomenon which has been linked to focal fibrosis. Native T1-mapping is an alternative technique to characterize myocardium and has the advantage of not requiring the use of contrast agents. The aim of this study was to characterize the myocardium of idiopathic pulmonary arterial hypertension (IPAH), systemic scleroderma related PH (PAH-Ssc) and chronic thromboembolic PH (CTEPH) patients using native T1-mapping and to see whether native T1-values were related to disease severity. Furthermore, we compared native T1-values between the different precapillary PH categories. Native T1-mapping was performed in 46 IPAH, 14 PAH-SSc and 10 CTEPH patients and 10 control subjects. Native T1-values were assessed using regions of interest at the RV and LV free wall, interventricular septum and interventricular insertion regions. In PH patients, native T1-values of the interventricular insertion regions were significantly higher than the native T1-values of the RV free wall, LV free wall and interventricular septum. Native T1-values at the insertion regions were significantly related to disease severity. Native T1-values were not different between IPAH, PAH-Ssc and CTEPH patients. Native T1-values of the interventricular insertion regions are significantly increased in precapillary PH and are related to disease severity. Native T1-mapping can be developed as an alternative technique for the characterization of the interventricular insertion regions and has the advantage of not requiring the use of contrast agents.
Keywords: T1-mapping, Myocardium, Non-contrast T1, Pulmonary hypertension
Introduction
Precapillary pulmonary hypertension (PH) is characterized by an increase in pulmonary vascular resistance (PVR) and right ventricular (RV) adaption to the increased load is one of the main determinants of patient outcome [1]. A well-established method to non-invasively characterize the myocardium is the assessment of late gadolinium enhancement (LGE) by cardiac magnetic resonance imaging (CMRI). Previous studies in PH patients showed delayed enhancement of the interventricular insertion regions [2–5] and linked this phenomenon to focal fibrosis [4, 6]. In addition, LGE was shown to correlate with disease severity [2, 4, 5]. A major drawback of the assessment of LGE is the need for the administration of the contrast agent gadolinium. Its toxicity due to depositions in other parts of the body, for example in the brain [7], is not completely understood, but gadolinium administration is contra-indicated in patients with renal insufficiency. Moreover, the LGE technique is not suitable to detect more diffuse myocardial pathologies since the calculation of the threshold of abnormal enhancement depends on a reference area of myocardium [8].
An alternative technique to characterize the myocardium is native T1-mapping. T1-mapping quantifies the T1 relaxation time per pixel tissue and different tissues show a characteristic range of T1-values [9–11]. Native T1-values increase when the heart is affected by edema and diffuse or focal fibrosis [12–20]. To quantify native T1-values, there is no need for a reference area of myocardium, making it possible to directly quantify the total myocardium. Moreover, native T1-values can be determined without the use of contrast agents [10].
Therefore, the aim of this study was to characterize the myocardium of idiopathic pulmonary arterial hypertension (IPAH), systemic scleroderma related PH (PAH-Ssc) and chronic thromboembolic PH (CTEPH) patients using native T1-mapping and to see whether native T1-values were related to disease severity. Furthermore, since the difference in presence of histologically confirmed myocardial fibrosis between these forms of precapillary PH vary between studies [21–23], we compared native T1-values between the different precapillary PH categories.
Methods
Subjects
We retrospectively analyzed all available native T1-mapping data of IPAH patients (n = 46), PAH-SSc patients (n = 14) and CTEPH patients (n = 10) scanned between June 2011 and March 2014 in the VU University Medical Center. Data was acquired in the context of an ongoing prospective research program to investigate the role of CMRI in the evaluation of PH patients. The study was approved by The Medical Ethics Review Committee of the VU University Medical Center. Since this study did not fall within the scope of the Medical Research Involving Human Subjects Act (WMO), the study was approved without requirement of an informed consent statement. IPAH, PAH-SSc and CTEPH were diagnosed according to ATS/ERS guidelines [24]. Both treatment naïve patients and patients under optimal PAH-therapies were included in the study. Patients with left sided heart failure and congenital heart disease were excluded from this study. Furthermore, native T1-mapping was performed in ten healthy volunteers, who gave written informed consent for usage of the data for this study.
CMRI protocol
Native T1-mapping was acquired on a Siemens 1.5 T Avanto scanner. A single breath-hold Modified Look-Locker Inversion-recovery (MOLLI) pulse sequence was used on a mid-ventricular short axis imaging plane. Three, three, and five non-segmented images were acquired at end-diastole within 17 heart beats to sample the recovery of longitudinal magnetization after the inversion pulse. Minimal inversion time was 100 ms [25]. Inplane motion correction was applied. Motion correction was applied by exploiting the known exponential form of inversion recovery and treating the motion and inversion recovery as a joint estimation problem. This was performed with the generation of a series of motion free synthetic inversion recovery images which were used at each inversion time for registration with the measured MOLLI images [26].
CMRI analyses
Native T1-values were assessed using regions of interest (ROIs) at the interventricular insertion regions, the RV free wall, LV free wall, interventricular septum and interventricular insertion regions on mid-ventricular short axis T1-maps. ROIs were manually drawn as illustrated in Fig. 1. ROIs were carefully assessed and the borders of the myocardium were avoided to prevent partial volume effects due to surrounding tissue or the blood pool. ROIs in the RV free wall could be accurately positioned in all patients in the inferior part of the RV free wall (Fig. 1). We attempted to draw ROIs of the total RV free wall, but this was not feasible in the majority of patients because the RV free wall was too thin. Native T1-values of the RV wall could not be assessed in the control subjects because in all control subjects the RV wall was too thin. T1-values of the interventricular insertion regions were averaged over the inferior and superior insertion regions. Ventricular volumes were assessed as described before [1].
Fig. 1.

Schematic illustration of ROIs. Schematic illustration of a mid-ventricular short-axis image. ROIs of the different regions are marked with colors. ROIs of the LV free wall (red) covered the total LV free wall. ROIs of the RV free wall (blue), were only analyzed in the inferior part of the RV free wall. Attempts to cover the total RV free wall failed because in a majority of the patients the free wall was too thin, resulting in unreliable native T1-values due to partial volume effects. ROIs of the interventricular septum are marked in yellow. ROIs of the interventricular insertion regions are marked in green
Statistical methods
Data are presented as mean ± standard deviation, unless stated differently. In PH patients, native T1-values between the RV free wall, LV free wall, interventricular septum and interventricular insertion regions were compared using repeated measures one-way ANOVA with Bonferroni post-hoc correction. In control subjects, native T1-values between the LV free wall, interventricular septum and interventricular insertion regions were compared using repeated measures one-way ANOVA with Bonferroni post-hoc correction.
Native T1-values of the different regions of the myocardium between IPAH, PAH-SSc and CTEPH patients and healthy controls were compared using one-way ANOVA with Bonferroni post-hoc correction.
In PH patients, Pearson correlation analysis were applied to assess the relation between native T1-values and hemodynamic and cardiac parameters of disease severity: right atrial pressure (RAP), mean pulmonary artery pressure (mPAP), pulmonary vascular resistance (PVR), cardiac index (CI), RV stroke volume index (SVI), RV end-diastolic volume index (RVEDVI), RV end-systolic volume index (RVESVI), RV ejection fraction (RVEF) and NT Pro-BNP.
All analyses were performed using SPSS for Windows version 20.0 and Graphpad Prism for Windows version 5.0. p values <0.05 were considered statistically significant.
Results
Patient characteristics, hemodynamics and standard CMRI measurements are summarized in Table 1. Control subjects were significantly younger than PH patients (20 ± 1 and 54 ± 16 p < 0.001). 40% of the control subjects were female. Heart rate (HR) was not significantly different between control subjects (74 ± 11 bpm) and PH patients (79 ± 14 bpm).
Table 1.
Patient characteristics
| N = 70 (IPAH n = 46; PAH-SSc n = 14; CTEPH n = 10) | Control subjects (n = 10) | p value | |
|---|---|---|---|
| Female (%) | 73 | 40 | 0.04 |
| Age (years) | 54 ± 16 | 20 ± 1 | <0.001 |
| Heart rate (bpm) | 79 ± 14 | 74 ± 11 | 0.280 |
| Hemodynamics | |||
| mPAP (mmHg) | 47 ± 13 | – | |
| PAWP (mmHg) | 8 ± 3 | – | |
| PVR (dyn s/cm5) | 634 ± 342 | – | |
| RAP (mmHg) | 7 ± 4 | – | |
| CI (L/min/m2) | 3 ± 1 | – | |
| CMRI | |||
| RVEDVI (mL/m2) | 82 ± 38 | – | |
| RVESVI (mL/m2) | 51 ± 37 | – | |
| RVSVI (mL/m2) | 30 ± 12 | – | |
| RVEF (%) | 42 ± 16 | – | |
| LVEF (%) | 65 ± 11 | – | |
| NT ProBNP (ng/L) | 1393 ± 2283 | – |
mPAP mean pulmonary artery pressure, PAWP pulmonary arterial wedge pressure, PVR pulmonary vascular resistance, RAP right atrial pressure, CI cardiac index, CMRI cardiac magnetic resonance imaging, RVEDVI indexed right ventricular end-diastolic volume, RVESVI indexed right ventricular end-systolic volume, RVSVI right ventricular stroke volume index, RVEF right ventricular ejection fraction, LVEF left ventricular ejection fraction
Native T1-values in PH patients and control subjects
In PH patients, native T1-values of the interventricular insertion regions (1060 ± 70 ms) were significantly higher than the native T1-values of the RV free wall, LV free wall and interventricular septum (Figs. 2, 3). Native T1-values of the RV free wall (996 ± 69 ms) were not significantly different from the native T1-values of the LV free wall (977 ± 60 ms) and interventricular septum (1009 ± 48 ms). Native T1-values of the LV free wall were significantly lower than the native T1-values of the interventricular septum.
Fig. 2.
Native T1-values (ms) of the RV free wall, LV free wall, interventricular septum and the interventricular insertion regions. Data is presented as mean and standard error of the mean. a Regional differences in the myocardium of control subjects. No regional differences were found in control subjects. b Regional differences in the myocardium of PH patients. T1-values of the RV free wall of PH patients were not different from T1-values of the LV free wall and interventricular septum. *T1-values of the interventricular septum of PH patients were significantly higher compared to native T1-values of the LV free wall. #Native T1-values of the interventricular insertion regions of PH patients were significantly higher compared to native T1-values of the RV free wall, LV free wall and interventricular septum
Fig. 3.
Native T1-maps showing increased native T1-values at the interventricular insertion regions. Two examples of native T1-maps of a control subject (a) and an IPAH (b) patient. The white arrows indicate the increased native T1-values at the interventricular insertion regions
In control subjects, no differences were found between native T1-values of the LV free wall (961 ± 26 ms), interventricular septum (958 ± 23 ms) and interventricular insertion regions (957 ± 27 ms) (Fig. 2).
Relation between native T1-values of the interventricular insertion regions and disease severity in PH patients
In PH patients, native T1-values of the interventricular insertion regions were significantly related to RAP (Pearson r = 0.310; p = 0.01), RVEDVI (Pearson r = 0.376; p = 0.001), RVESVI (Pearson r = 0.358 p = 0.002), RVEF (Pearson r = −0.282; p = 0.018) and NT pro-BNP (Pearson r = 0.392; p = 0.001), but not to CI, PVR, RVSVI and mPAP (Fig. 4).
Fig. 4.
Correlations between native T1-values of the interventricular insertion regions and RVEDVI, RVESVI, RVEF and RAP in PH patients. Native T1-values of the interventricular insertion regions of PH patients were significantly related to RVEDVI, RVESVI, RVEF and RAP. RVEDVI right ventricular end-diastolic volume index, RVESVI right ventricular end-systolic volume index, RVEF right ventricular ejection fraction, RAP right atrial pressure
Comparison of native T1-values between IPAH, CTEPH and PAH-Ssc patients and control subjects
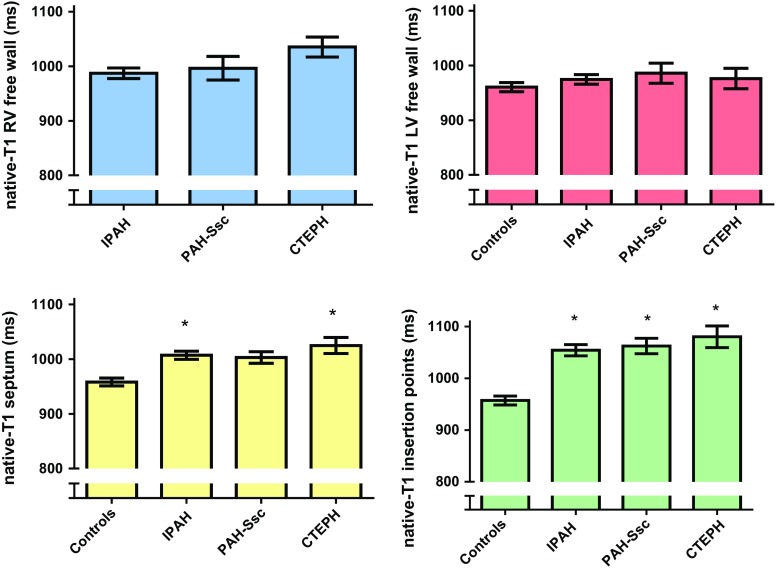
Native T1-values of the RV free wall, LV free wall, interventricular septum and the interventricular insertion regions were not significantly different between IPAH, CTEPH and PAH-SSc patients.
Native T1-values of the LV free wall were not significantly different between control subjects and PH patients. Native T1-values of the interventricular septum was significantly higher in IPAH and CTEPH patients compared to control subjects. Native T1-values of the interventricular insertion regions were significantly higher in all PH categories compared to control subjects (Fig. 5).
Fig. 5.
Comparison of native T1-values between IPAH, PAH-SSc and CTEPH patients and control subjects. Data is presented as mean and standard error of the mean. The Y-axis Native T1-values of the RV free wall (blue), LV free wall (red), interventricular septum (yellow) and interventricular insertion regions (green) were not significantly different between IPAH, PAH-SSc and CTEPH patients. Native T1-values of the LV free wall was not significantly different between control subjects and PH patients. Native T1-values of the interventricular septum was significantly higher in IPAH and CTEPH patients compared to control subjects. Native T1-values of the interventricular insertion regions were significantly increased in all PH categories compared to control subjects
Discussion
This is the first study using the native T1-mapping technique to characterize the myocardium in precapillary PH patients. In PH patients, native T1-values of the interventricular insertion regions were significantly increased compared to the native T1-values of the RV free wall, LV free wall and interventricular septum. Furthermore, native T1-values of the interventricular insertion regions were related to disease severity.
Increased native T1-values of the interventricular insertion regions
LGE studies in PH showed the late enhancement of the interventricular insertion regions and it has been suggested that this phenomenon most likely reflect locally increased focal fibrosis [2–5]. This suggestion was strengthened by McCann et al. [4] by finding increased focal fibrosis at the interventricular insertion regions in the histology of the myocardium of two PH patients. Bull et al. [12] linked the native T1-values to histological findings in patients with severe aortic stenosis and found a good correlation between native T1-values and the collagen volume fraction. Furthermore, a recent study investigating native T1-values in a chronic PH animal model also found increased native T1-values at the interventricular insertion regions. In this study, the PH group showed increased interstitial collagen at the interventricular insertion regions compared to the sham group [27]. Therefore, the increased native T1-values of the interventricular insertion regions most likely reflect locally increased focal fibrosis. However, McCann et al. [4] also described edema at the interventricular insertion regions, which also can contribute to an increase in native T1-values [17]. It is suggested that predilection for fibrosis to develop at the interventricular insertion regions is caused by mechanical stress due to the bowing of the interventricular septum into the LV [2] which is often seen in precapillary PH patients. However, late enhancement at the interventricular insertion regions is also described in patients with hypertrophic cardiomyopathy [3, 28] indicating that this phenomenon is not specific for a pressure overloaded RV.
Increased native T1-values of the interventricular insertion regions were moderately, but significantly related to disease severity. This is in line with previous LGE studies showing comparable correlations between the late enhancement of the interventricular insertion regions and RVEF, RV volumes and mPAP [2, 4, 5]. Furthermore, similar correlations were found between native T1-values of the interventricular insertion regions and measures of disease severity [27]. The correlations between native T1-values of the interventricular insertion regions and RV functional measures and NT-proBNP is probably related to the associated increase in RV wall tension. It has been shown that RV wall tension is associated with the delay in time to peak shortening of the RV, causing the leftward shift of the interventricular septum during late RV systole [29]. The right-to-leftward shifting of the interventricular septum probably increases the mechanical shear stress on the interventricular insertion regions.
In a recent study, native T1-values were assessed in the RV of healthy subjects and the authors found increased native T1-values in the RV free wall compared to the LV free wall and suggested that this finding could be due to a higher collagen content of the RV free wall [30]. In the PH patients in our study, native T1-values of the RV free wall were not significantly higher than those of the LV free wall and were in the same range as the native T1-values of the RV free wall in healthy subjects [30]. We could not assess the native T1-values of the RV wall in our control subjects because the wall was too thin. Kawel-Boehm et al. [30] could assess native T1-values of the RV free wall of healthy subjects because they performed native T1-mapping at the end-systolic phase.
Native T1-values of the LV free wall were not significantly different between PH patients and control subjects and were in the same range as native T1-values of the LV free wall conducted in a large cohort of healthy subjects of the same age [11]. This is in line with LGE studies showing no late enhancement in the myocardium of precapillary PH patients apart from the interventricular insertion regions [2–5].
Native T1-values between IPAH, PAH-SSc and CTEPH patients
A recent study of Ntusi et al [18] found increased native T1-values of the total myocardium in no-PH systemic sclerosis patients compared to controls. We found no differences in native T1-values between IPAH, PAH-SSc and CTEPH patients. The presence of myocardial fibrosis found in these different forms of precapillary PH differ between studies. No differences in myocardial fibrosis of RV free wall tissue were found between controls, IPAH and PAH-SSc patients [21], while others found an increased amount of myocardial fibrosis in PAH patients compared to no-PH controls [22]. Ntusi et al. [18] included both limited cutaneous (lcSSc) and diffuse cutaneous systemic sclerosis (dcSSc) patients, while in our study we only included patients with lcSSc. It is known that cardiac involvement of systemic sclerosis is much higher in dcSSc patients compared to lcSSc patients [31], which can explain the differences in results.
Limitations
We could not assess native T1-values of the total RV free wall, since in most patients the RV free wall was too thin to avoid partial volume effects. Only the inferior part of the RV free wall could be accurately assessed in all patients. Although we did not assess the total RV free wall, large variances in native T1-values between different regions of the RV free wall are not expected in a pressure overloaded RV.
Our study is a retrospective analysis and we included both treatment naïve and treated PH patients. From our data we cannot rule out any treatment effects on the measured native T1-values.
The control subjects were significantly younger compared to PH patients. It is known that age can influence native T1-values of the myocardium, with higher native T1-values found at a younger age [11]. Since native T1-values of the interventricular insertion regions were significantly increased in PH patients compared to control subjects, this only strengthens our findings.
The MOLLI technique as we applied is known to slightly underestimate native T1 values at higher heart rates [32]. The main conclusions in this study rely on regional differences, and are therefore not affected. Moreover, heart rates were not different between patients and controls. Another potential confounder is the effect of the T2 relaxation time: the absolute T1 values obtained with the MOLLI technique defer from their actual values in tissues with short T2 [33]. Whether or not T2-changes play a role in our study, is still to be explored. A technique such as SASHA [34] is more accurate, however, the values obtained with this SACHA technique show a larger variability and the MOLLI method performs more precisely [35].
Conclusions
Native T1-values of the interventricular insertion regions are increased compared to native T1-values of the LV free wall, RV free wall and interventricular septum in patients with precapillary PH and are related to disease severity. Native-T1 values are not different between IPAH, PAH-SSc and CTEPH patients. Native T1-mapping can be an alternative for the characterization of the interventricular insertion regions without the use of contrast agents.
Acknowledgments
The authors thank Andreas Greiser for making the MOLLI pulse sequence available.
Funding
Anton Vonk-Noordegraaf was financially supported by the NWO, Vidi Grant, Project Number 91.796.306.
Compliance with ethical standards
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by The Medical Ethics Review Committee of the VU University Medical Center. Since this study did not fall within the scope of the Medical Research Involving Human Subjects Act (WMO), the study was approved without requirement of an informed consent statement.
Contributor Information
Onno A. Spruijt, Email: o.spruijt@vumc.nl
J. Tim Marcus, Phone: +31 20 4440179, Email: jt.marcus@vumc.nl.
References
- 1.van de Veerdonk MC, Kind T, Marcus JT, Mauritz GJ, Heymans MW, Bogaard HJ, Boonstra A, Marques KM, Westerhof N, Vonk-Noordegraaf A. Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. J Am Coll Cardiol. 2011;58:2511–2519. doi: 10.1016/j.jacc.2011.06.068. [DOI] [PubMed] [Google Scholar]
- 2.Blyth KG, Groenning BA, Martin TN, Foster JE, Mark PB, Dargie HJ, Peacock AJ. Contrast enhanced-cardiovascular magnetic resonance imaging in patients with pulmonary hypertension. Eur Heart J. 2005;26:1993–1999. doi: 10.1093/eurheartj/ehi328. [DOI] [PubMed] [Google Scholar]
- 3.McCann GP, Beek AM, Vonk-Noordegraaf A, van Rossum AC. Delayed contrast-enhanced magnetic resonance imaging in pulmonary arterial hypertension. Circulation. 2005;112:e268. doi: 10.1161/CIRCULATIONAHA.104.512848. [DOI] [PubMed] [Google Scholar]
- 4.McCann GP, Gan CT, Beek AM, Niessen HW, Vonk Noordegraaf A, van Rossum AC. Extent of MRI delayed enhancement of myocardial mass is related to right ventricular dysfunction in pulmonary artery hypertension. Am J Roentgenol. 2007;188:349–355. doi: 10.2214/AJR.05.1259. [DOI] [PubMed] [Google Scholar]
- 5.Sanz J, Dellegrottaglie S, Kariisa M, Sulica R, Poon M, O’Donnell TP, Mehta D, Fuster V, Rajagopalan S. Prevalence and correlates of septal delayed contrast enhancement in patients with pulmonary hypertension. Am J Cardiol. 2007;100:731–735. doi: 10.1016/j.amjcard.2007.03.094. [DOI] [PubMed] [Google Scholar]
- 6.Moon JC, Reed E, Sheppard MN, Elkington AG, Ho SY, Burke M, Petrou M, Pennell DJ. The histologic basis of late gadolinium enhancement cardiovascular magnetic resonance in hypertrophic cardiomyopathy. J Am Coll Cardiol. 2004;43:2260–2264. doi: 10.1016/j.jacc.2004.03.035. [DOI] [PubMed] [Google Scholar]
- 7.McDonald RJ, McDonald JS, Kallmes DF, Jentoft ME, Murray DL, Thielen KR, Williamson EE, Eckel LJ. Intracranial gadolinium deposition after contrast-enhanced MR imaging. Radiology. 2015;275:772–782. doi: 10.1148/radiol.15150025. [DOI] [PubMed] [Google Scholar]
- 8.Kwong RY, Farzaneh-Far A. Measuring myocardial scar by CMR. J Am Coll Cardiol Img. 2011;4:157–160. doi: 10.1016/j.jcmg.2010.12.004. [DOI] [PubMed] [Google Scholar]
- 9.Burt JR, Zimmerman SL, Kamel IR, Halushka M, Bluemke DA. Myocardial T1 mapping: techniques and potential applications. Radiographics. 2014;34:377–395. doi: 10.1148/rg.342125121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Moon JC, Messroghli DR, Kellman P, Piechnik SK, Robson MD, Ugander M, Gatehouse PD, Arai AE, Friedrich MG, Neubauer S, Schulz-Menger J, Schelbert EB. Myocardial T1 mapping and extracellular volume quantification: a Society for Cardiovascular Magnetic Resonance (SCMR) and CMR Working Group of the European Society of Cardiology consensus statement. J Cardiovasc Magn Reson. 2013;15:92. doi: 10.1186/1532-429X-15-92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Piechnik SK, Ferreira VM, Lewandowski AJ, Ntusi NA, Banerjee R, Holloway C, Hofman MB, Sado DM, Maestrini V, White SK, Lazdam M, Karamitsos T, Moon JC, Neubauer S, Leeson P, Robson MD. Normal variation of magnetic resonance T1 relaxation times in the human population at 1.5 T using ShMOLLI. J Cardiovasc Magn Reson. 2013;15:13. doi: 10.1186/1532-429X-15-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bull S, White SK, Piechnik SK, Flett AS, Ferreira VM, Loudon M, Francis JM, Karamitsos TD, Prendergast BD, Robson MD, Neubauer S, Moon JC, Myerson SG. Human non-contrast T1 values and correlation with histology in diffuse fibrosis. Heart. 2013;99:932–937. doi: 10.1136/heartjnl-2012-303052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dall’Armellina E, Piechnik SK, Ferreira VM, Si QL, Robson MD, Francis JM, Cuculi F, Kharbanda RK, Banning AP, Choudhury RP, Karamitsos TD, Neubauer S. Cardiovascular magnetic resonance by non contrast T1-mapping allows assessment of severity of injury in acute myocardial infarction. J Cardiovasc Magn Reson. 2012;14:15. doi: 10.1186/1532-429X-14-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ferreira VM, Piechnik SK, Dall’Armellina E, Karamitsos TD, Francis JM, Choudhury RP, Friedrich MG, Robson MD, Neubauer S. Non-contrast T1-mapping detects acute myocardial edema with high diagnostic accuracy: a comparison to T2-weighted cardiovascular magnetic resonance. J Cardiovasc Magn Reson. 2012;14:42. doi: 10.1186/1532-429X-14-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ferreira VM, Piechnik SK, Dall’Armellina E, Karamitsos TD, Francis JM, Ntusi N, Holloway C, Choudhury RP, Kardos A, Robson MD, Friedrich MG, Neubauer S. T(1) mapping for the diagnosis of acute myocarditis using CMR: comparison to T2-weighted and late gadolinium enhanced imaging. J Am Coll Cardiol Img. 2013;6:1048–1058. doi: 10.1016/j.jcmg.2013.03.008. [DOI] [PubMed] [Google Scholar]
- 16.Ferreira VM, Piechnik SK, Dall’Armellina E, Karamitsos TD, Francis JM, Ntusi N, Holloway C, Choudhury RP, Kardos A, Robson MD, Friedrich MG, Neubauer S. Native T1-mapping detects the location, extent and patterns of acute myocarditis without the need for gadolinium contrast agents. J Cardiovasc Magn Reson. 2014;16:36. doi: 10.1186/1532-429X-16-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Maestrini V, Treibel TA, White SK, Fontana M, Moon JC. T1 mapping for characterization of intracellular and extracellular myocardial diseases in heart failure. Curr Cardiovasc Imaging Rep. 2014;7:9287. doi: 10.1007/s12410-014-9287-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ntusi NA, Piechnik SK, Francis JM, Ferreira VM, Rai AB, Matthews PM, Robson MD, Moon J, Wordsworth PB, Neubauer S, Karamitsos TD. Subclinical myocardial inflammation and diffuse fibrosis are common in systemic sclerosis—a clinical study using myocardial T1-mapping and extracellular volume quantification. J Cardiovasc Magn Reson. 2014;16:21. doi: 10.1186/1532-429X-16-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Puntmann VO, D’Cruz D, Smith Z, Pastor A, Choong P, Voigt T, Carr-White G, Sangle S, Schaeffter T, Nagel E. Native myocardial T1 mapping by cardiovascular magnetic resonance imaging in subclinical cardiomyopathy in patients with systemic lupus erythematosus. Circ Cardiovasc Imaging. 2013;6:295–301. doi: 10.1161/CIRCIMAGING.112.000151. [DOI] [PubMed] [Google Scholar]
- 20.Puntmann VO, Voigt T, Chen Z, Mayr M, Karim R, Rhode K, Pastor A, Carr-White G, Razavi R, Schaeffter T, Nagel E. Native T1 mapping in differentiation of normal myocardium from diffuse disease in hypertrophic and dilated cardiomyopathy. J Am Coll Cardiol Img. 2013;6:475–484. doi: 10.1016/j.jcmg.2012.08.019. [DOI] [PubMed] [Google Scholar]
- 21.Overbeek MJ, Mouchaers KT, Niessen HM, Hadi AM, Kupreishvili K, Boonstra A, Voskuyl AE, Belien JA, Smit EF, Dijkmans BC, Vonk-Noordegraaf A, Grunberg K (2010) Characteristics of interstitial fibrosis and inflammatory cell infiltration in right ventricles of systemic sclerosis-associated pulmonary arterial hypertension. Int J Rheumatol. doi:10.1155/2010/604615 [DOI] [PMC free article] [PubMed]
- 22.Rain S, Handoko ML, Trip P, Gan CT, Westerhof N, Stienen GJ, Paulus WJ, Ottenheijm CA, Marcus JT, Dorfmuller P, Guignabert C, Humbert M, Macdonald P, Dos Remedios C, Postmus PE, Saripalli C, Hidalgo CG, Granzier HL, Vonk-Noordegraaf A, van der Velden J, de Man FS. Right ventricular diastolic impairment in patients with pulmonary arterial hypertension. Circulation. 2013;128(2016–25):1–10. doi: 10.1161/CIRCULATIONAHA.113.001873. [DOI] [PubMed] [Google Scholar]
- 23.Ruiter G, van de Veerdonk MC, Bogaard HJ, Wong YY, Marcus JT, Lammertsma AA, Westerhof N, van der Laarse WJ, de Man FS, Vonk-Noordegraaf A. The interventricular septum in pulmonary hypertension does not show features of right ventricular failure. Int J Cardiol. 2014;173:509–512. doi: 10.1016/j.ijcard.2014.03.064. [DOI] [PubMed] [Google Scholar]
- 24.Galie N, Hoeper MM, Humbert M, Torbicki A, Vachiery JL, Barbera JA, Beghetti M, Corris P, Gaine S, Gibbs JS, Gomez-Sanchez MA, Jondeau G, Klepetko W, Opitz C, Peacock A, Rubin L, Zellweger M, Simonneau G. Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respir J. 2009;34:1219–1263. doi: 10.1183/09031936.00139009. [DOI] [PubMed] [Google Scholar]
- 25.Messroghli DR, Greiser A, Frohlich M, Dietz R, Schulz-Menger J. Optimization and validation of a fully-integrated pulse sequence for modified look-locker inversion-recovery (MOLLI) T1 mapping of the heart. J Magn Reson Imaging. 2007;26:1081–1086. doi: 10.1002/jmri.21119. [DOI] [PubMed] [Google Scholar]
- 26.Xue H, Shah S, Greiser A, Guetter C, Littmann A, Jolly MP, Arai AE, Zuehlsdorff S, Guehring J, Kellman P. Motion correction for myocardial T1 mapping using image registration with synthetic image estimation. Magn Reson Med. 2012;67:1644–1655. doi: 10.1002/mrm.23153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Garcia-Alvarez A, Garcia-Lunar I, Pereda D, Fernandez-Jimenez R, Sanchez-Gonzalez J, Mirelis JG, Nuno-Ayala M, Sanchez-Quintana D, Fernandez-Friera L, Garcia-Ruiz JM, Pizarro G, Aguero J, Campelos P, Castella M, Sabate M, Fuster V, Sanz J, Ibanez B. Association of myocardial T1-mapping CMR with hemodynamics and RV performance in pulmonary hypertension. J Am Coll Cardiol Img. 2015;8:76–82. doi: 10.1016/j.jcmg.2014.08.012. [DOI] [PubMed] [Google Scholar]
- 28.Choudhury L, Mahrholdt H, Wagner A, Choi KM, Elliott MD, Klocke FJ, Bonow RO, Judd RM, Kim RJ. Myocardial scarring in asymptomatic or mildly symptomatic patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2002;40:2156–2164. doi: 10.1016/S0735-1097(02)02602-5. [DOI] [PubMed] [Google Scholar]
- 29.Marcus JT, Gan CT, Zwanenburg JJ, Boonstra A, Allaart CP, Gotte MJ, Vonk-Noordegraaf A. Interventricular mechanical asynchrony in pulmonary arterial hypertension: left-to-right delay in peak shortening is related to right ventricular overload and left ventricular underfilling. J Am Coll Cardiol. 2008;51:750–757. doi: 10.1016/j.jacc.2007.10.041. [DOI] [PubMed] [Google Scholar]
- 30.Kawel-Boehm N, Dellas Buser T, Greiser A, Bieri O, Bremerich J, Santini F. In-vivo assessment of normal T1 values of the right-ventricular myocardium by cardiac MRI. Int J Cardiovasc Imaging. 2013;30:323–328. doi: 10.1007/s10554-013-0326-3. [DOI] [PubMed] [Google Scholar]
- 31.Hunzelmann N, Genth E, Krieg T, Lehmacher W, Melchers I, Meurer M, Moinzadeh P, Muller-Ladner U, Pfeiffer C, Riemekasten G, Schulze-Lohoff E, Sunderkoetter C, Weber M, Worm M, Klaus P, Rubbert A, Steinbrink K, Grundt B, Hein R, Scharffetter-Kochanek K, Hinrichs R, Walker K, Szeimies RM, Karrer S, Muller A, Seitz C, Schmidt E, Lehmann P, Foeldvari I, Reichenberger F, Gross WL, Kuhn A, Haust M, Reich K, Bohm M, Saar P, Fierlbeck G, Kotter I, Lorenz HM, Blank N, Grafenstein K, Juche A, Aberer E, Bali G, Fiehn C, Stadler R, Bartels V. The registry of the German Network for Systemic Scleroderma: frequency of disease subsets and patterns of organ involvement. Rheumatology. 2008;47:1185–1192. doi: 10.1093/rheumatology/ken179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Piechnik SK, Ferreira VM, Dall’Armellina E, Cochlin LE, Greiser A, Neubauer S, Robson MD. Shortened Modified Look-Locker Inversion recovery (ShMOLLI) for clinical myocardial T1-mapping at 1.5 and 3 T within a 9 heartbeat breathhold. J Cardiovasc Magn Reson. 2010;12:69. doi: 10.1186/1532-429X-12-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chow K, Flewitt JA, Pagano JJ, Green JD, Friedrich MG, Thompson RB. T2-dependent errors in MOLLI T1 values: simulations, phantoms, and in-vivo studies. J Cardiovasc Magn Reson. 2012;14(Suppl 1):281. doi: 10.1186/1532-429X-14-S1-P281. [DOI] [Google Scholar]
- 34.Chow K, Flewitt JA, Green JD, Pagano JJ, Friedrich MG, Thompson RB. Saturation recovery single-shot acquisition (SASHA) for myocardial T(1) mapping. Magn Reson Med. 2013;71:2082–2095. doi: 10.1002/mrm.24878. [DOI] [PubMed] [Google Scholar]
- 35.Roujol S, Weingartner S, Foppa M, Chow K, Kawaji K, Ngo LH, Kellman P, Manning WJ, Thompson RB, Nezafat R. Accuracy, precision, and reproducibility of four T1 mapping sequences: a head-to-head comparison of MOLLI, ShMOLLI, SASHA, and SAPPHIRE. Radiology. 2014;272:683–689. doi: 10.1148/radiol.14140296. [DOI] [PMC free article] [PubMed] [Google Scholar]