Abstract
Aim:
Recent evidence suggests that the essential amino acid leucine may be involved in systemic cholesterol metabolism. In this study, we investigated the effects of leucine supplementation on the development of atherosclerosis in apoE null mice.
Methods:
ApoE null mice were fed with chow supplemented with leucine (1.5% w/v) in drinking water for 8 week. Aortic atherosclerotic lesions were examined using Oil Red O staining. Plasma lipoprotein-cholesterol levels were measured with fast protein liquid chromatography. Hepatic gene expression was detected using real-time PCR and Western blot analyses.
Results:
Leucine supplementation resulted in 57.6% reduction of aortic atherosclerotic lesion area in apoE null mice, accompanied by 41.2% decrease of serum LDL-C levels and 40.2% increase of serum HDL-C levels. The body weight, food intake and blood glucose level were not affected by leucine supplementation. Furthermore, leucine supplementation increased the expression of Abcg5 and Abcg8 (that were involved in hepatic cholesterol efflux) by 1.28- and 0.86-fold, respectively, and significantly increased their protein levels. Leucine supplementation also increased the expression of Srebf1, Scd1 and Pgc1b (that were involved in hepatic triglyceride metabolism) by 3.73-, 1.35- and 1.71-fold, respectively. Consequently, leucine supplementation resulted in 51.77% reduction of liver cholesterol content and 2.2-fold increase of liver triglyceride content. Additionally, leucine supplementation did not affect the serum levels of IL-6, IFN-γ, TNF-α, IL-10 and IL-12, but markedly decreased the serum level of MCP-1.
Conclusion:
Leucine supplementation effectively attenuates atherosclerosis in apoE null mice by improving the plasma lipid profile and reducing systemic inflammation.
Keywords: amino acid, leucine supplementation, atherosclerosis, lipid metabolism, ABCG5, ABCG8, apoE null mice, inflammation
Introduction
Atherosclerosis (AS) is the leading cause of cardiovascular death worldwide. Inflammation, lipid deposition and foam cell formation are well-established pathological features of AS. The essential steps of AS development include the migration of circulating monocytes into the subendothelial arterial space and their differentiation into macrophages, the rapid uptake of modified low-density lipoprotein (LDL), and the subsequent formation of foam cells. Plasma lipoprotein levels are closely related to the onset and development of AS. High-density lipoprotein (HDL) levels have been reported to be negatively correlated with cardiovascular disease occurrence, which is due to the role of HDL in reverse cholesterol transport1. By contrast, LDL levels have been positively correlated with cardiovascular disease occurrence2 due to the ability of modified LDL to promote foam cell formation.
Leucine is one of the 8 essential amino acids. Previous studies have shown that leucine and its metabolites can stimulate skeletal muscle protein synthesis and inhibit skeletal muscle protein degradation3. Recent studies have shown that leucine is also involved in glucose and lipid metabolism. Zhang et al showed that leucine supplementation via drinking water significantly improved high fat diet-induced obesity and insulin resistance4. However, Cheng et al demonstrated that a leucine-deficient diet promoted the hydrolysis of white fat tissue and stimulated heat production, indicating that leucine impaired insulin sensitivity5. Another study by Newgard et al revealed that feeding mixed branched-chain amino acids to mice on a high-fat diet activated mammalian target of rapamycin (mTOR) signaling, which increased insulin resistance and reduced food intake6. Although many studies have reported distinct roles of leucine in different animal models of diabetes, clinical data have shown that plasma leucine levels are significantly increased in obese people with hyperlipidemia compared with non-obese people6. Furthermore, leucine supplementation decreased total and LDL cholesterol levels by 27% and 53%, respectively, in high-fat diet-treated mice4. These studies suggest that leucine may be involved in systemic cholesterol metabolism. Because plasma cholesterol, especially LDL cholesterol, play pivotal roles in AS development, we hypothesize that leucine supplementation inhibits AS development.
In this study, apoE null mice on a chow diet were provided leucine supplementation through drinking water for 8 weeks. The leucine-supplemented apoE null mice showed significantly attenuated progression of atherosclerotic plaques. Mechanistically, leucine improved the plasma lipid profile in apoE null mice by promoting ABCG5/ABCG8–mediated cholesterol efflux from the liver and inhibiting systemic inflammation.
Materials and methods
Animals
Female 14-week-old apoE null mice were provided by the Animal Center of Peking University Health Science Center (Beijing, China). ApoE null mice were fed with chow supplemented with or without 1.5% (w/v) leucine (Sigma, St Louis, MO, USA) in drinking water for 8 weeks. The treatment of the laboratory animals and the experimental protocols followed the guidelines of Peking University and were approved by the Institutional Authority for Laboratory Animal Care.
Analysis of atherosclerotic lesion area
Serial 6-μm-thick cryosections of mouse aortas, beginning at the aortic sinus, were collected every 400 μm. Sections were stained with Oil Red O. Oil Red O–positive areas in digitized color images of stained aortic sinus sections were quantified using ImagePro Plus 5.02 (Media Cybernetics Inc, MD, USA).
For en face analyses, aortas were longitudinally opened from the heart to the iliac arteries, and atherosclerotic lesions were stained with Oil Red O. Images of en face aortic lesions were obtained using an Olympus E-520 digital camera. Images were analyzed with ImagePro Plus 5.02; data are presented as percentages of total section/aorta area.
Plasma lipid analyses
Plasma total triglyceride and cholesterol contents, as well as HDL and LDL cholesterol levels, were measured with kits from BioSino Biotechnology & Science Inc (Beijing, China). Plasma lipoproteins were fractioned by fast protein liquid chromatography as previously described7.
Cell culture
Peritoneal macrophages were isolated as described8. Male 8-week-old C57BL/6J mice were intraperitoneally injected with 2 mL of 4% (w/v) thioglycollate broth (BD Biosciences Clontech, CA, USA). Three days later, the mice were anesthetized with ketamine/xylazine, and macrophages were obtained by peritoneal lavage with 10 mmol/L EDTA and 10% fetal bovine serum (FBS) in 8 mL of cold phosphate-buffered saline (PBS). Cells were plated at 1.0×106/mL RPMI-1640 with 10% FBS. After incubation for 3 h at 37°C, non-adherent cells were washed off, and adherent cells were collected for the experiments.
RNA extraction and real-time PCR
RNA from apoE null mice livers were isolated by RNA TRIP reagent (Applygen, Beijing, China); 1 μg of RNA was reverse transcribed by a reverse transcription system (Promega, Madison, WI). Then, the reaction mixture was amplified using real-time PCR. The amount of PCR products formed in each cycle was evaluated by SYBR Green I fluorescence. The primers used for mouse glucose/lipid metabolism-related genes are presented in Table 1. All amplification reactions used the Mx3000 Multiplex Quantitative PCR System (Stratagene, La Jolla, CA, USA). The results were analyzed using Stratagene Mx3000 software.
Table 1. Changes in expression of glucose/lipid metabolism related genes in the liver. bP<0.05, cP<0.01 vs Control.
| Gene name | Primers |
Fold changes |
||
|---|---|---|---|---|
| Right | Left | Control (n=8) | Leucine (n=9) | |
| Triglyceride metabolism | ||||
| Fasn (NM_007988) | GTTGGCCCAGAACTCCTGTA | GTCGTCTGCCTCCAGAGC | 1.54±0.57 | 1.53±0.41 |
| Scd1 (NM_009127) | CAGCCGAGCCTTGTAAGTTC | GCTCTACACCTGCCTCTTCG | 1.68±0.35 | 3.95±0.65c |
| Srebf1 (NM_011480) | TGGTTGTTGATGAGCTGGAG | GGCTCTGGAACAGACACTGG | 1.00±0.11 | 4.73±0.74c |
| ChREBP (NM_021455) | AACCACACACTGGGCTCTTC | CTTCAAAGGCCTCAAGTTGC | 0.64±0.13 | 0.13±0.02 |
| Lxralpha (NM_013839) | TGGAGAACTCAAAGATGGGG | TGAGAGCATCACCTTCCTCA | 0.84±0.08 | 1.02±0.08 |
| Lxrbeta (NM_009473) | AGAACTTGTGGGGGAAGACA | GGTGCAGTCATGAGCCCC | 0.90±0.09 | 1.10±0.07 |
| Pparg (NM_001127330) | TCTTCCATCACGGAGAGGTC | GATGCACTGCCTATGAGCAC | 1.14±0.10 | 1.75±0.27 |
| Ppard (NM_011145) | GGTCATAGCTCTGCCACCAT | ACTCAGAGGCTCCTGCTCAC | 1.11±0.37 | 2.07±0.82 |
| Pgc1a (NM_008904) | TGAGGACCGCTAGCAAGTTT | TGTAGCGACCAATCGGAAAT | 1.02±0.23 | 1.26±0.38 |
| Pgc1b (NM_133249) | GAGGTCAAGCTCTGGCAAGT | GCTCTCGTCCTTCTTCCTCA | 0.66±0.16 | 1.79±0.28c |
| Fatty acid transfer | ||||
| Slc27a1 (NM_011977) | AGCCGAACACGAATCAGAAC | TTCTGTGTGTACGTGGGTGG | 0.99±0.34 | 1.10±0.29 |
| Slc27a5 (NM_009512) | GGCCAAGGTAGAAGCAGTGA | CCAGTGTGCTGATTGTGGAT | 1.03±0.12 | 1.35±0.07 |
| Cd36 (NM_007643) | CCTGCAAATGTCAGAGGAAA | GCGACATGATTAATGGCACA | 0.65±0.14 | 0.75±0.16 |
| Fabp1 (NM_017399) | GATTTCTGACACCCCCTTGA | TGCAGAGCCAGGAGAACTTT | 0.93±0.33 | 1.29±0.22 |
| Cholesterol metabolism | ||||
| Hmgcr (NM_008255) | CACAATAACTTCCCAGGGGT | GGCCTCCATTGAGATCCG | 1.33±0.18 | 1.92±0.31 |
| Srebp2 (NM_033218) | CACAATATCATTGAAAAGCGCTACCGGTCC | TTTTCTGATTGGCCAGCTTCAGCACCATG | 0.86±0.10 | 1.33±0.21 |
| Abcg5 (NM_031884) | ACCTTACCCACGGTTCCTTTC | CGCCCTTTAGCGTGTTGTTC | 1.02±0.18 | 2.33±0.29c |
| Abcg8 (NM_026180) | AGCTGGGCATCCGAAATCTA | GTTGGGCAGCAGTTGGTCAT | 1.11±0.24 | 2.07±0.18c |
| Abcg1 (NM_009593) | TTTCCCAGAGATCCCTTTCA | ATCGAATTCAAGGACCTTTCC | 1.12±0.13 | 1.52±0.13b |
| Abca1 (NM_013454) | GCTGCAGGAATCCAGAGAAT | CATGCACAAGGTCCTGAGAA | 0.93±0.03 | 0.97±0.05 |
| Glucose metabolism | ||||
| G6pc (NM_008061) | GTGTCCAGGACCCACCAATA | ACTGTGGGCATCAATCTCCT | 0.85±0.11 | 0.34±0.09c |
| Pck1 (NM_011044) | TGTCTTCACTGAGGTGCCAG | CTGGATGAAGTTTGATGCCC | 0.66±0.18 | 1.05±0.11 |
Statistical analysis
Data are presented as the mean±SEM. Student's t-test was used to compare pairs (GraphPad Prism; GraphPad). P<0.05 was considered significant.
Results
Leucine supplementation reduces atherosclerotic lesions in apoE null mice
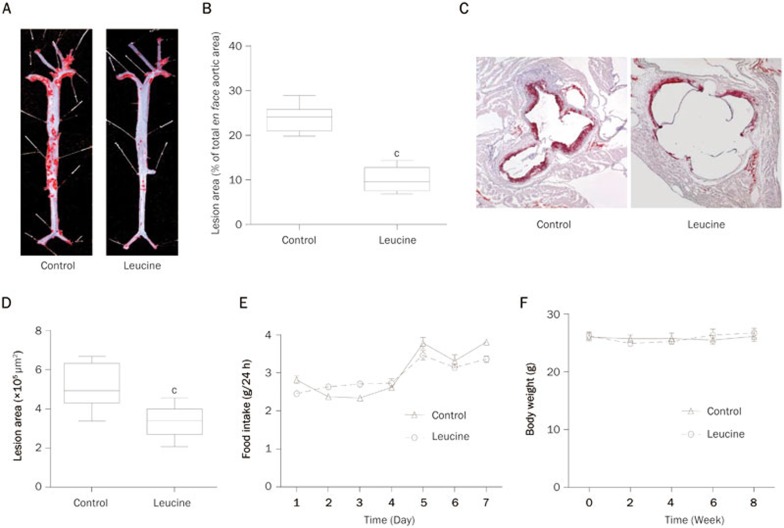
ApoE null mice were fed with chow and had free access to distilled water with or without supplementation of leucine (1.5% w/v) for 8 weeks. Atherosclerotic lesions were examined in these mice. Leucine supplementation significantly reduced the en face lesion of the entire aorta as shown by the Oil Red O staining (Figure 1A). The quantification of en face-prepared aortas revealed a significant 57.6% reduction in lesion area in leucine-treated apoE null mice (Figure 1B). We then stained the frozen slices of the aortic sinus with Oil Red O, which revealed a 40% lesion reduction in the leucine-treated mice (Figure 1C, 1D). Leucine supplementation by drinking water did not influence food intake in the mice (Figure 1E) or body weight (Figure 1F).
Figure 1.
Leucine reduced aortic atherosclerotic lesions in apoE null mice. ApoE null mice were fed with chow and supplemented with or without leucine (1.5%) in drinking water for 8 weeks. (A) Representative images of en face Oil Red O staining in the full aorta. (B) Relative quantification of lesion area in the control and leucine treatment mice; data are presented as the percentages of total en face aortic area. (C) Representative images of Oil Red O staining in the frozen section of the left ventricular outflow tract. (D) Quantification of lesion area in the control and leucine treatment mice, data are presented as the lesion areas (×105 μm2). (E) Food intake was measured at the first week of leucine supplementation. (F) Body weight was measured every two weeks. Data are presented as the mean±SEM. n=8 in control group. n=9 in leucine group. cP<0.01 vs control.
Leucine supplementation improves plasma lipid profile but not blood glucose levels in apoE null mice
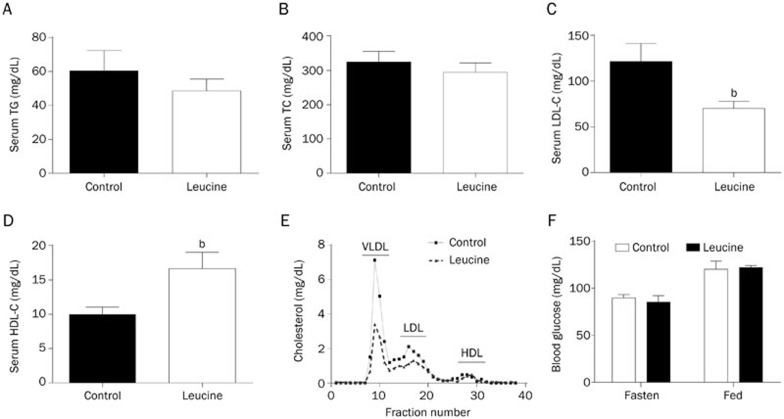
Inflammation, lipid deposition and foam cell formation are well-known features of AS. The plasma lipoprotein level is closely correlated with the onset and development of AS. Several studies have indicated that leucine participates in body lipid metabolism4,6. Therefore, we measured the total serum cholesterol, triglyceride, LDL-cholesterol, and HDL-cholesterol concentrations at the end of the leucine supplementation period. The results showed that the serum triglyceride and total cholesterol levels did not change after leucine supplementation (Figure 2A and 2B). Interestingly, the LDL-cholesterol level was significantly lower (Figure 2C), but the HDL-cholesterol level was significantly higher (Figure 2D) in the leucine-supplemented mice than those in the control mice. To confirm the improved lipid profile in mice supplemented with leucine, we performed fast protein liquid chromatography (FPLC) to detect the serum lipoproteins. The results revealed that VLDL-cholesterol and LDL-cholesterol levels were significantly reduced in the leucine group compared with the control group (Figure 2E). However, the fasting and fed blood glucose levels were not affected by leucine supplementation in apoE null mice fed with the chow diet (Figure 2F).
Figure 2.
Effect of leucine on plasma lipid profile and glucose level of apoE null mice. After leucine supplementation (1.5%) in drinking water for 8 weeks, (A) serum total triglyceride (TG), (B) total cholesterol (TC), (C) low-density lipoprotein cholesterol (LDL-C) and (D) high density lipoprotein cholesterol (HDL-C) concentrations were measured. (E) Distribution of cholesterol in plasma lipoproteins from control and leucine treated apoE null mice was measured by use of fast protein liquid chromatography (n=3). (F) Blood glucose levels at fasted and fed states were measured. Data are presented as the mean±SEM. n=8–9. bP<0.05 vs control.
Effect of leucine supplementation on hepatic triglyceride and total cholesterol contents
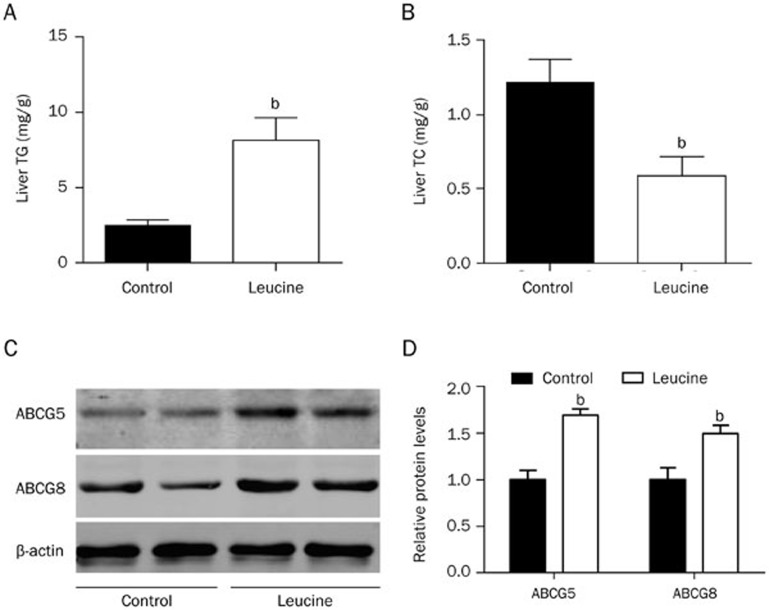
The liver is the main organ for VLDL synthesis and is the most important organ for lipid metabolism. Because leucine supplementation improved the plasma lipid profile, we next determined whether leucine supplementation affected the hepatic lipid content. The results showed that compared with control mice, apoE null mice supplemented with leucine had significantly increased triglyceride content (Figure 3A) but significantly decreased cholesterol content in the liver (Figure 3B). These results indicate that leucine supplementation modulated the hepatic lipid content.
Figure 3.
Lipid content and ABCG5/ABCG8 protein levels in the livers of apoE null mice. (A, B) After leucine supplementation (1.5%) in drinking water for 8 weeks, liver lipids were extracted. Triglyceride (TG) and total cholesterol (TC) contents were measured. (C, D) Western blot analyses detected liver protein levels of ABCG5 and ABCG8; protein levels relative to β-actin are shown. Data are presented as the mean±SEM. n=8–9. bP<0.05 vs control.
Effect of leucine supplementation on the expression of glucose/lipid metabolism-related genes in the liver
To investigate the mechanisms through which leucine supplementation improved the serum lipid profile, we detected glucose/lipid metabolism-related gene expression in the livers of the control and leucine-supplemented mice using real-time PCR. The mRNA levels of Abcg5 and Abcg8, which participate in hepatic cholesterol efflux to bile, were significantly increased in the liver tissue of the leucine-supplemented mice compared with the control group (Table 1). The mRNA levels of HMG-CoA reductase, the rate-limiting enzyme in cholesterol synthesis, and sterol regulatory element binding factor 2 (Srebp2) did not significantly change (Table 1). The mRNA levels of genes related to triglyceride metabolism, such as Srebf1, stearoyl-coenzyme A desaturase 1 (Scd1), and Pgc1b, significantly increased in the livers of the leucine group (Table 1), whereas those of other genes related to triglyceride synthesis, such as fatty acid synthase (Fasn), LXRa, LXRb, Pparg/PPARgamma, Ppard/PPARdelta, and Pgc1a, did not significantly change (Table 1). Genes that contribute to fatty acid uptake and transfer, including Slc27a1/Fatp1, Slc27a5/Fatp5, Fabp1 and Cd36, were not affected. The expression of the glucose-6-phosphatase gene (G6pc) was downregulated, whereas that of the phosphoenolpyruvate carboxykinase 1 (Pck1) gene was not changed. Additional Western blot analyses confirmed the increased ABCG5 and ABCG8 protein levels in the leucine-treated mice (Figure 3C, 3D). These results indicate that hepatic triglyceride synthesis and cholesterol efflux to bile are increased in apoE null mice supplemented with leucine.
Effect of leucine supplementation on the systemic inflammation of apoE null mice
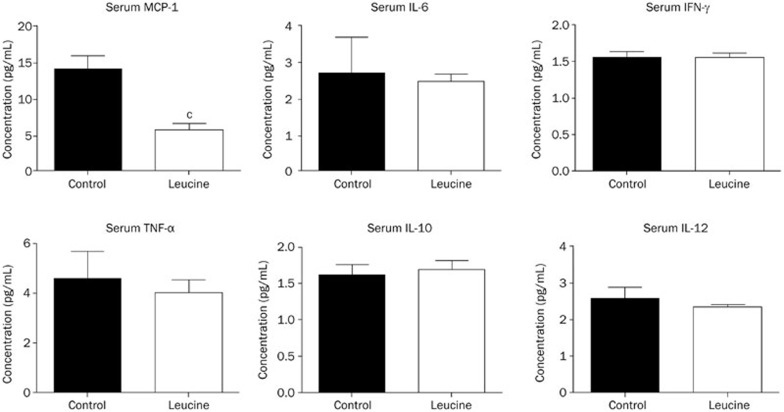
The above results indicated that leucine supplementation improved the plasma lipid profiles in apoE null mice. Next, we explored the effect of leucine supplementation on chronic systemic inflammation in apoE null mice. The results revealed that among the six examined cytokines [tumor necrosis factor (TNF)-α, monocyte chemotactic protein-1 (MCP)-1, interferan (IFN)-γ, interleukin (IL)-6, IL-12, and IL-10], the concentration of MCP-1 was significantly lower in the leucine group compared with the control group, whereas the concentrations of the other 5 cytokines were not significantly influenced (Figure 4).
Figure 4.
Effect of leucine on plasma inflammatory cytokine levels in apoE null mice. The serum inflammatory cytokines MCP-1, IL-6, IFN-γ, TNF-α, IL-10, IL-12 levels were measured. Data are presented as the mean±SEM. n=8–9. cP<0.01 vs control.
Discussion
The present work investigated the effect of leucine supplementation on atherosclerotic lesion development. The results showed that leucine supplementation through drinking water (1.5%) significantly reduced atherosclerotic lesion formation in apoE null mice, without changes in body weight, food intake or fasting/fed glucose concentration compared with the control group. This beneficial effect of leucine on AS may be attributed to an improved plasma lipid profile (increased HDL-C levels and decreased VLDL-C and LDL-C levels) and ameliorated inflammation. The increased expression levels of hepatic Abcg5 and Abcg8 potentially resulted in cholesterol efflux, the decreased liver cholesterol contents and subsequently, lowered serum VLDL-C and LDL-C levels. By contrast, hepatic Srebf1, Scd1 and Pgc1b expression levels increased and may have contributed to the increased hepatic triglyceride content.
Recently, several studies have demonstrated that compared with carbohydrate-rich diets with identical caloric contents, protein-rich diets promoted better glycemic control and weight loss9,10,11,12. Branched-chain amino acids (BCAA), especially leucine, are thought to play pivotal roles in these protein-rich diets13,14. Leucine, as an essential amino acid, stimulates protein synthesis in skeletal muscles15,16,17,18 and is known to be involved in glucose and lipid metabolism (however, conflicting reports regarding the metabolic role of leucine exist in the literature). Zhang et al reported that leucine supplementation through drinking water significantly reduced diet-induced obesity and insulin resistance and improved cholesterol metabolism in C57BL/6J mice4. In their study, the loss of fat mass was due to increased resting energy expenditure, which was caused by increased expressions of uncoupling protein 3 in white adipose tissue (WAT), brown adipose tissue (BAT), and skeletal muscle; food intake was unchanged between the control and leucine groups. Additionally, Zhang et al showed that leucine supplementation decreased hyperglycemia caused by a high-fat diet, and alleviated insulin resistance and lowered concentrations of glucagon, glucogenic amino acids, and glucose-6-phosphatase in the liver4. These beneficial effects of leucine on improving insulin resistance have been confirmed by several other studies13,14,19,20,21. By contrast, Cheng et al reported that a leucine-deficient diet significantly decreased fat mass in C57BL/6J mice5. They reported that the rapid fat mass loss caused by a leucine-deficient diet was due to decreased fatty acid synthesis in WAT and increased lipolysis and thermogenesis resulting from an upregulation of uncoupling protein 1 in BAT5. In other studies using leucine-deficient diets, hepatic insulin sensitivities were improved in leucine-deficient groups and were associated with GCN2/mTOR/S6K1 pathways22,23. However, the application of a leucine-deficient diet is controversial. Leucine is an essential amino acid that cannot be synthesized by the body and plays critical roles in metabolism24. The elimination of an essential amino acid will likely result in abnormal metabolic conditions as other chemicals will compensate for the deficiency. Cheng et al5 reported that leucine deprivation resulted in an activated sympathetic nervous system, elevated serum T3 concentration and increased Dio2 (T3 generator) mRNA levels in BAT compared with control groups. This result indicated that leucine-deprived mice demonstrated a hyperthyroid state. Thus, elevated resting thermogenesis, increased fat mobilization and decreased body weights were expected. Furthermore, Newgard et al6 reported that feeding mixed branched-chain amino acids activated mTOR and caused insulin resistance. The branched-chain amino acid supplementation reduced food intake in mice on high-fat diets, indicating an effect different from leucine supplementation. For this reason we used a chow diet with leucine supplementation.
Although contradictory effects of leucine supplementation on glucose metabolism have been reported in distinct models, studies have reported more consistent effects of leucine supplementation on lipid metabolism. Cheng et al6 demonstrated elevated plasma leucine level in obese humans with hyperlipidemia; Zhang et al4 showed that leucine supplementation alleviated hypercholesterolemia caused by high-fat diets. These studies indicate that leucine may be involved in body cholesterol metabolism.
There are two ways to maintain body cholesterol homeostasis. The first way is by reducing the intestinal absorption of cholesterol; the second way is by transporting excess cholesterol in peripheral tissues to the liver and excreting cholesterol into bile by reverse cholesterol transportation. Excess cholesterol in the liver is excreted either directly to bile or indirectly by conversion to bile acids25. The members of adenosine triphosphate (ATP) binding cassette transporter (ABC) superfamily ABCA1 and ABCG1 are two important factors in reverse cholesterol transportation26. These two members promote lipid efflux from hepatocytes and macrophages and provide a protective mechanism for the removal of excess cholesterol in the body. However, ABCA1 and ABCG1 do not encompass the mechanism of cholesterol efflux from the liver to the bile duct. ABCG5 and ABCG8 have recently been identified as members of the ABC superfamily G subfamily. They are transmembrane transporter proteins involved in reverse cholesterol transport and are known to reduce the absorption of intestinal steroids, promote the excretion of hepatic steroids and control the level of plasma steroids. ABCG5 and ABCG8 play key roles in maintaining the balance of steroids but not at the same levels as ABCA1 and ABCG127,28. ABCG5 and ABCG8 are highly expressed in the liver and the proximal small intestinal epithelial cells29. Unlike ABCA1, which is located on the basolateral side of hepatocytes, in situ hybridization has shown that ABCG5 and ABCG8 are arranged along the intestinal epithelial cell microvilli of the absorbing section of the bowel. This finding suggests that ABCG5 and ABCG8 participate in preventing the intestinal absorption of cholesterol and promoting cholesterol efflux from the liver30. Abcg5/Abcg8 transgenic mice31 and wild-type mice fed a high cholesterol diet30,32 or a synthetic liver X receptor (LXR) ligand T090131733,34 showed significantly increased cholesterol effluxes from the liver to the bile duct. Moreover, the knockout of Abcg533, Abcg835, or both36 may, in general, cause the opposite effect. In the present study, ABCG5 and ABCG8 expression levels were significantly increased in the livers of the leucine group when compared with those of the control group. Additionally, the levels of HMG-CoA reductase and Srebf2 mRNA did not change. Thus, the elevated level of cholesterol efflux and the unchanged cholesterol synthesis capacity contributed to the decreased cholesterol content in the liver of the leucine group. This effect may further result in the observed decreased hepatic VLDL production levels and the improved lipid profile in the leucine-treated mice.
However, genes associated with triglyceride metabolism, such as Srebf1, Scd1 and Pgc1b, were significantly upregulated, indicating enhanced triglyceride synthesis in the leucine-treated mice. By contrast, LDL and VLDL, which are associated with triglyceride transport from the liver to peripheral organs, were significantly decreased in the leucine group. Because the apoE null mice were unable to absorb LDL, the observed decreased serum LDL level was due to the decreased hepatic VLDL/LDL production. Thus, increased triglyceride synthesis and decreased hepatic VLDL/LDL secretion caused the accumulation of triglycerides in the livers of leucine-treated mice, whereas fatty acid uptake and transfer-related genes did not change.
In addition to a lipid metabolism disorder, atherosclerosis is also an inflammatory disease37. In this study, the MCP-1 concentration in the leucine group significantly decreased. Although this result may be attributed to the improved serum lipid profile, the reduced inflammation may have also partly contributed to the reduction in atherosclerotic lesions. Notably, similar to other BCAAs, the catabolism of leucine primarily occurs in skeletal muscle and to lesser extents in the small intestine, liver and kidneys. In these organs, arginine will be metabolized to citrulline and produce nitric monoxide3. Nitric monoxide is a vasodilator and an important protective factor of the cardiovascular system38. Many studies have shown that nitric oxide plays an important role in the inhibition of atherosclerosis39,40,41,42,43. Similarly, the beneficial role of leucine could be due to its metabolite nitric oxide.
Taken together, leucine supplementation attenuates AS in apoE null mice. It is likely that leucine reduces inflammation and improves the serum lipid profile by increasing hepatic cholesterol efflux through the up-regulation of Abcg5/Abcg8, leading to a reduction in atherosclerotic plaques. Thus, leucine supplementation may be a novel strategy for preventing AS.
Author contribution
Yang ZHAO, Xiao-yan DAI, Xian WANG and Ming-jiang XU designed research; Yang ZHAO, Xiao-yan DAI, Zhou ZHOU, Ge-xin ZHAO performed research; Yang ZHAO, Xiao-yan DAI, Xian WANG and Ming-jiang XU analyzed data; Yang ZHAO and Ming-jiang XU wrote the paper.
Acknowledgments
This study was supported by the National Basic Research Program of China (No 2011CB503904 and 2012CB518002), and the National Natural Science Foundation of China (No 31230035, 81270370, and 81470557).
References
- 1Castelli WP, Garrison RJ, Wilson PW, Abbott RD, Kalousdian S, Kannel WB. Incidence of coronary heart disease and lipoprotein cholesterol levels. The Framingham Study. JAMA 1986; 256: 2835–8. [PubMed] [Google Scholar]
- 2Dresel HA, Friedrich EA, Otto I, Waldherr R, Schettler G. The low density lipoprotein and low density lipoprotein receptors and their possible importance in the pathogenesis of atherosclerosis. Arzneimittelforschung 1985; 35: 1936–40. [PubMed] [Google Scholar]
- 3Wu G. Amino acids: metabolism, functions, and nutrition. Amino Acids 2009; 37: 1–17. [DOI] [PubMed] [Google Scholar]
- 4Zhang Y, Guo K, LeBlanc RE, Loh D, Schwartz GJ, Yu YH. Increasing dietary leucine intake reduces diet-induced obesity and improves glucose and cholesterol metabolism in mice via multimechanisms. Diabetes 2007; 56: 1647–54. [DOI] [PubMed] [Google Scholar]
- 5Cheng Y, Meng Q, Wang C, Li H, Huang Z, Chen S, et al. Leucine deprivation decreases fat mass by stimulation of lipolysis in white adipose tissue and upregulation of uncoupling protein 1 (UCP1) in brown adipose tissue. Diabetes 2010; 59: 17–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6Newgard CB, An J, Bain JR, Muehlbauer MJ, Stevens RD, Lien LF, et al. A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metab 2009; 9: 311–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7Kurano M, Hara M, Tsuneyama K, Okamoto K, Iso ON, Matsushima T, et al. Modulation of lipid metabolism with the overexpression of NPC1L1 in mouse liver. J Lipid Res 2012; 53: 2275–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8Dai XY, Cai Y, Sun W, Ding Y, Wang W, Kong W, et al. Intermedin inhibits macrophage foam-cell formation via tristetraprolin-mediated decay of CD36 mRNA. Cardiovasc Res 2014; 101: 297–305. [DOI] [PubMed] [Google Scholar]
- 9Skov AR, Toubro S, Ronn B, Holm L, Astrup A. Randomized trial on protein vs carbohydrate in ad libitum fat reduced diet for the treatment of obesity. Int J Obes Relat Metab Disord 1999; 23: 528–36. [DOI] [PubMed] [Google Scholar]
- 10Parker B, Noakes M, Luscombe N, Clifton P. Effect of a high-protein, high-monounsaturated fat weight loss diet on glycemic control and lipid levels in type 2 diabetes. Diabetes Care 2002; 25: 425–30. [DOI] [PubMed] [Google Scholar]
- 11Layman DK, Shiue H, Sather C, Erickson DJ, Baum J. Increased dietary protein modifies glucose and insulin homeostasis in adult women during weight loss. J Nutr 2003; 133: 405–10. [DOI] [PubMed] [Google Scholar]
- 12Layman DK, Boileau RA, Erickson DJ, Painter JE, Shiue H, Sather C, et al. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J Nutr 2003; 133: 411–7. [DOI] [PubMed] [Google Scholar]
- 13Leenders M, van Loon LJ. Leucine as a pharmaconutrient to prevent and treat sarcopenia and type 2 diabetes. Nutr Rev; 69: 675–89. [DOI] [PubMed] [Google Scholar]
- 14Layman DK, Walker DA. Potential importance of leucine in treatment of obesity and the metabolic syndrome. J Nutr 2006; 136: 319S–23S. [DOI] [PubMed] [Google Scholar]
- 15Dardevet D, Sornet C, Bayle G, Prugnaud J, Pouyet C, Grizard J. Postprandial stimulation of muscle protein synthesis in old rats can be restored by a leucine-supplemented meal. J Nutr 2002; 132: 95–100. [DOI] [PubMed] [Google Scholar]
- 16Shimomura Y, Yamamoto Y, Bajotto G, Sato J, Murakami T, Shimomura N, et al. Nutraceutical effects of branched-chain amino acids on skeletal muscle. J Nutr 2006; 136: 529S–32S. [DOI] [PubMed] [Google Scholar]
- 17Gomes-Marcondes MC, Ventrucci G, Toledo MT, Cury L, Cooper JC. A leucine-supplemented diet improved protein content of skeletal muscle in young tumor-bearing rats. Braz J Med Biol Res 2003; 36: 1589–94. [DOI] [PubMed] [Google Scholar]
- 18Anthony JC, Anthony TG, Layman DK. Leucine supplementation enhances skeletal muscle recovery in rats following exercise. J Nutr 1999; 129: 1102–6. [DOI] [PubMed] [Google Scholar]
- 19Nishitani S, Takehana K, Fujitani S, Sonaka I. Branched-chain amino acids improve glucose metabolism in rats with liver cirrhosis. Am J Physiol Gastrointest Liver Physiol 2005; 288: G1292–300. [DOI] [PubMed] [Google Scholar]
- 20Guo K, Yu YH, Hou J, Zhang Y. Chronic leucine supplementation improves glycemic control in etiologically distinct mouse models of obesity and diabetes mellitus. Nutr Metab (Lond) 2010; 7: 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21Hinault C, Mothe-Satney I, Gautier N, Lawrence JC Jr, Van Obberghen E. Amino acids and leucine allow insulin activation of the PKB/mTOR pathway in normal adipocytes treated with wortmannin and in adipocytes from db/db mice. FASEB J 2004; 18: 1894–6. [DOI] [PubMed] [Google Scholar]
- 22Guo F, Cavener DR. The GCN2 eIF2alpha kinase regulates fatty-acid homeostasis in the liver during deprivation of an essential amino acid. Cell Metab 2007; 5: 103–14. [DOI] [PubMed] [Google Scholar]
- 23Xiao F, Huang Z, Li H, Yu J, Wang C, Chen S, et al. Leucine deprivation increases hepatic insulin sensitivity via GCN2/mTOR/S6K1 and AMPK pathways. Diabetes; 60: 746–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24Nair KS, Short KR. Hormonal and signaling role of branched-chain amino acids. J Nutr 2005; 135: 1547S–52S. [DOI] [PubMed] [Google Scholar]
- 25Miller GJ, Miller NE. Plasma-high-density-lipoprotein concentration and development of ischaemic heart-disease. Lancet 1975; 1: 16–9. [DOI] [PubMed] [Google Scholar]
- 26Neufeld EB, Remaley AT, Demosky SJ, Stonik JA, Cooney AM, Comly M, et al. Cellular localization and trafficking of the human ABCA1 transporter. J Biol Chem 2001; 276: 27584–90. [DOI] [PubMed] [Google Scholar]
- 27Fitzgerald ML, Mujawar Z, Tamehiro N. ABC transporters, atheros-clerosis and inflammation. Atherosclerosis 2010; 211: 361–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28Matsuo M. ATP-binding cassette proteins involved in glucose and lipid homeostasis. Biosci Biotechnol Biochem 2010; 74: 899–907. [DOI] [PubMed] [Google Scholar]
- 29Graf GA, Li WP, Gerard RD, Gelissen I, White A, Cohen JC, et al. Coexpression of ATP-binding cassette proteins ABCG5 and ABCG8 permits their transport to the apical surface. J Clin Invest 2002; 110: 659–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30Repa JJ, Berge KE, Pomajzl C, Richardson JA, Hobbs H, Mangelsdorf DJ. Regulation of ATP-binding cassette sterol transporters ABCG5 and ABCG8 by the liver X receptors alpha and beta. J Biol Chem 2002; 277: 18793–800. [DOI] [PubMed] [Google Scholar]
- 31Yu L, Li-Hawkins J, Hammer RE, Berge KE, Horton JD, Cohen JC, et al. Overexpression of ABCG5 and ABCG8 promotes biliary cholesterol secretion and reduces fractional absorption of dietary cholesterol. J Clin Invest 2002; 110: 671–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32Yu L, Gupta S, Xu F, Liverman AD, Moschetta A, Mangelsdorf DJ, et al. Expression of ABCG5 and ABCG8 is required for regulation of biliary cholesterol secretion. J Biol Chem 2005; 280: 8742–7. [DOI] [PubMed] [Google Scholar]
- 33Plosch T, Bloks VW, Terasawa Y, Berdy S, Siegler K, Van Der Sluijs F, et al. Sitosterolemia in ABC-transporter G5-deficient mice is aggravated on activation of the liver-X receptor. Gastroenterology 2004; 126: 290–300. [DOI] [PubMed] [Google Scholar]
- 34Yu L, York J, von Bergmann K, Lutjohann D, Cohen JC, Hobbs HH. Stimulation of cholesterol excretion by the liver X receptor agonist requires ATP-binding cassette transporters G5 and G8. J Biol Chem 2003; 278: 15565–70. [DOI] [PubMed] [Google Scholar]
- 35Klett EL, Lu K, Kosters A, Vink E, Lee MH, Altenburg M, et al. A mouse model of sitosterolemia: absence of Abcg8/sterolin-2 results in failure to secrete biliary cholesterol. BMC Med 2004; 2: 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36Yu L, Hammer RE, Li-Hawkins J, Von Bergmann K, Lutjohann D, Cohen JC, et al. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc Natl Acad Sci U S A 2002; 99: 16237–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37Libby P. Inflammation and cardiovascular disease mechanisms. Am J Clin Nutr 2006; 83: 456S–60S. [DOI] [PubMed] [Google Scholar]
- 38Gewaltig MT, Kojda G. Vasoprotection by nitric oxide: mechanisms and therapeutic potential. Cardiovasc Res 2002; 55: 250–60. [DOI] [PubMed] [Google Scholar]
- 39Lowenstein CJ. Beneficial effects of neuronal nitric oxide synthase in atherosclerosis. Arterioscler Thromb Vasc Biol 2006; 26: 1417–8. [DOI] [PubMed] [Google Scholar]
- 40Momi S, Monopoli A, Alberti PF, Falcinelli E, Corazzi T, Conti V, et al. Nitric oxide enhances the anti-inflammatory and anti-atherogenic activity of atorvastatin in a mouse model of accelerated atherosclerosis. Cardiovasc Res 2012; 94: 428–38. [DOI] [PubMed] [Google Scholar]
- 41Muller G, Morawietz H. Nitric oxide, NAD(P)H oxidase, and atheros-clerosis. Antioxid Redox Signal 2009; 11: 1711–31. [DOI] [PubMed] [Google Scholar]
- 42Hayashi T, Iguchi A. Possibility of the regression of atherosclerosis through the prevention of endothelial senescence by the regulation of nitric oxide and free radical scavengers. Geriatr Gerontol Int 2010; 10: 115–30. [DOI] [PubMed] [Google Scholar]
- 43Li H, Forstermann U. Prevention of atherosclerosis by interference with the vascular nitric oxide system. Curr Pharm Des 2009; 15: 3133–45. [DOI] [PubMed] [Google Scholar]