Abstract
Fibroblasts are cells widely used in cell culture, both for transient primary cell culture or permanent as transformed cell lines. Lately, fibroblasts become cell sources for use in disease modeling after cell reprogramming because it is easily accessible in the body. Fibroblasts in patients will maintain all genetic background during reprogramming into induced pluripotent stem cells. In spite of their large use, fibroblasts are obtained after an invasive procedure, a superficial punch skin biopsy, collected under patient’s local anesthesia. Taking into consideration the minimum patient’s discomfort during and after the biopsy procedure, as well as the aesthetics aspect, it is essential to reflect on the best site of the body for the biopsy procedure combined with the success of getting robust fibroblast cultures in the lab. For this purpose, we compared the efficiency of four biopsy sites of the body (skin from eyelid, back of the ear, abdominal cesarean scar and groin). Cell proliferation assays and viability after cryopreservation were measured. Our results revealed that scar tissue provided fibroblasts with higher proliferative rates. Also, fibroblasts from scar tissues presented a higher viability after the thawing process.
Keywords: Skin biopsy, Fibroblast, Primary cell culture, Scar, Eyelid, Ear, Groin
Introduction
Fibroblasts were first described at the end of the nineteenth century and are the most common cell type from connective tissue in animals, presenting an elongated morphology and extended cellular processes, with a fusiform shape (Virchow 1858; Duvall 1879). Fibroblasts are mesenchymal-derived cell types, important in several physiological process, such as synthesis of extracellular matrix (ECM), epithelial differentiation, regulation of inflammation and wound healing (Tarin and Croft 1969). In addition, fibroblasts are also responsible for secretion of growth factors and work as scaffolds for several other cell types, acting as the main mediator cell for tissue fibrosis and scar formation (Parsonage et al. 2005; Tomasek et al. 2002).
Fibroblasts are easily cultured and maintained in vitro. Historically, human fibroblast lines established from patients biopsies have been used in a number of studies to elucidate the pathogenesis of several diseases (Villegas and McPhaul 2005). Ever since the establishment of HeLa cells by George Gey in 1951 (Jones et al. 1971), scientists established several other fibroblast lines for different use, such as the fibroblasts NIH3T3 from Swiss mouse derived embryos, in which fibroblast were transformed with the antigen T from SV40 virus, turning them into a growing stable cell line (Todaro and Green, 2010), the cell line HEK293T, used in the production of viral vectors (Acrani et al. 2010), or primary cultures of fibroblasts derived from mouse embryos (MEF) used as feeder for embryonic stem cells culture (Odell et al. 2010.; vom Brocke, et al. 2006).
In addition, fibroblasts derived from skin are frequently used to produce induced pluripotent stem cells, or iPSCs (Takahashi et al. 2007), a powerful tool that allows production of other kinds of desired cells, which is now being widely used for disease modeling in vitro (Freitas et al. 2012; Ring et al. 2012; Chang et al. 2011; Marchetto et al. 2010; Dimos et al., 2008; Park et al. 2008). The use of iPSC technique for disease modeling is particularly important because it makes possible to generate the cell target of the disease by still keeping the genetic background from the patient, which is extremely important—no matter if the disease is monogenetic, with an identified mutation or if the genetic cause remains unknown.
So far, fibroblast culture methods seem well established but little is known about the ideal site for harvesting. Furthermore, biopsy produces a small scar and it is interesting to reconcile the collection site with aesthetics. In this article, we aim to verify the effectiveness of human body sites for biopsy collection and subsequent cell culture establishment, as well as the comfort and aesthetics for the patient. Tangible sites that were tested in this work are places commonly involved in plastic surgery for aesthetic repairs, such as eyelids, ears and abdomen. In addition, we include the groin area, taking advantage that this is an area not normally exposed.
Methods
Skin samples and cell culture obtaining
Skin samples from the back of ears, eyelid, abdomen scar and groin tissue were collected from female patients (ages ranges 40–47 years old), two patients for each site, by plastic surgery following consent under protocols approved by the participants institutions. The skin slices were approximately 0.3 cm2 square, but for the eyelid it was just a thin line, 0.2 × 0.2 cm. After removal, subcutaneous and adipose tissues attached to the skin zone were detached leaving just epidermis and dermis. The pieces of skin were placed in a conical tube containing DMEM/F12 (LGC Biotechnology, São Paulo, Brazil), 5× antibiotic antimycotic solution (Sigma Aldrich, St. Louis, MO, USA—penicillin/streptomycin/amphotericin; final 500 U/mL, 0.5 and 1.25 μg/mL, respectively) and placed on ice for transportation. In the lab, skin samples were washed in PBS containing antibiotic antimycotic solution (same concentration as used for transport—Sigma) for 3 times by gently shaking, then minced and transferred to culture dishes containing DMEM/F12, 10 % of Fetal Bovine Serum (FBS, Invitrogen, Carlsbad, CA, USA), 2 mM l-glutamine (Invitrogen), 1× MEM NEAA (Invitrogen), 2× antibiotic antimycotic solution (penicillin/streptomycin/amphotericin—final concentration of 200 U/mL, 0.2 and 0.5 μg/mL, respectively—Sigma) and maintained in an incubator with temperature at 37 °C and atmosphere of 5 % CO2. After 1 week, explant cultures had their medium changed every 2–3 days. Next, cells were expanded when confluent.
MTT assay
Approximately 1 × 104 cells were platted in quadruplicate on a 96 well/plate and MTT (Thiazolyl Blue Tetrazolium—Sigma-Aldrich, MO, USA) (5 mg/mL) was added 2, 5 and 9 days after plating and incubated for 3 h at 37 °C. After this period, MTT product was precipitated and solubilized using DMSO. The formazan product formed was measured in a spectrophotometer (μ-Quant Plate Reader, BioTek Instruments Inc, Winooski, VT, USA) at 550 nm. The growth rates were plotted in a graphic using Graph Pad program (GraphPad Software, San Diego, CA, USA).
Viability assay using trypan blue dye
Viability of cells after cryopreservation was evaluated using cell counting after trypan blue staining. Cells were diluted in trypan blue (1:10—Invitrogen) counted in a Neubauer chamber and frozen in 10 % DMSO and 90 % FBS and stored in a freezer at −80 °C for 1 week. After that time, cells were quickly thawed, washed, resuspended in the same media described above and immediately counted using trypan blue to dye dead cells, the same conditions were used for the first cell counting. The data obtained were plotted using GraphPad.
Results
Fibroblast cell lines
Biopsies were performed at distinct sites of healthy adult women volunteers after signing an informed consent. The tissues were subjected to explant method for cell culture establishment, after removal of adipose tissue and washed with antibiotics and antifungal solutions. The areas selected were: back of the ear (Fig. 1a), eyelids (Fig. 1b), abdomen (pre-existent cesarean scar—Fig. 1c), and groin (Fig. 1d). The sites for biopsies were defined considering the easy access and the aesthetics, but the biopsy could be carried out in any place defined by surgeon. After culture procedures described above, fibroblasts started to detach and spread out on the surface of the cell plate within 1 week; few cells were noted. After 2–3 weeks under the same culture conditions cell confluence was about 90 %. In despite of being from different skin sites, cultured fibroblast presented very similar morphology, as noted in Fig. 1a–c. Concerning the biopsy from groin area, the explant method did not generated fibroblast spreading out and Fig. 1d shows a piece of adherent skin using the same culture conditions used for the other skin areas.
Fig. 1.
Body sites selected for the harvesting of fibroblast cells: back of ears, eyelid, cesarean scar and groin. a Fibroblast cultured from the back of the ears (otoplasty) and, b fibroblast cultured from Eyelid (blepharoplasty), c fibroblast cultured from Cesarean Scar (abdomen) e, d Skin tissue from Groin under cell culture conditions, which did not spread cells. Cells were cultured in DMEM/F12, 10 % of FBS, 2 mm l-glutamine, 2× antibiotic antimycotic solution MEM NEAA, respectively) and maintained in an incubator with temperature at 37 °C and atmosphere of 5 % CO2. a–c magnification of 100×, scale bar 400 μm. d magnification of 40×, scale bar 1000 μm
MTT assay
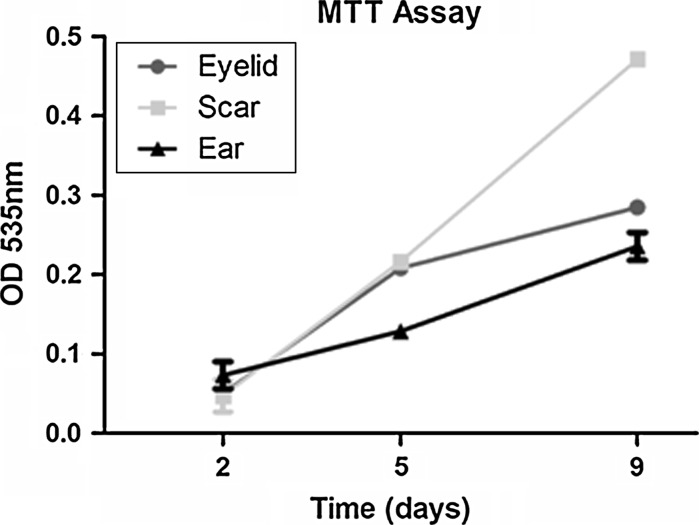
MTT were performed as described by Carmichael et al. (1987) and the assay was carried out by 9 days, with MTT measurement on days 2, 5 and 9 after plating. The assay is dependent on the cellular reduction of MTT by the mitochondrial dehydrogenase of viable cells to a blue formazan product that can be measured spectrophotometrically. In this assay was observed constant proliferation of fibroblasts from eyelid, ear and scar. However, fibroblasts from abdominal scar exhibited a higher proliferation rate when compared with fibroblasts from the two other sites (Fig. 2).
Fig. 2.
Proliferation rate of fibroblast cells from scar, eyelid and ear. The data represent the mean ± SD of absorbance at 535 nm. The experiments were performed in quadruplicate on every 3–4 days
Cryopreservation assay
One of the most important points in cell culture is the capacity to stock cells at very low temperatures and rescue them at the desired time. In order to check the cryopreservation capacity, cells were frozen and thawed and then counted using trypan blue dye to evaluate viability counting death cells stained in blue and bright alive cells. Cells obtained from eyelid revealed a viability of 67 %, from the back of the ear 51.5 % and from the scar 81 %. Thus, comparing fibroblasts from the three biopsied sites, fibroblasts from scar were the ones which presented higher rates of live cells while the fibroblasts from the back of the ear presented equivalent number of live and death cells (Fig. 3).
Fig. 3.
Percentage of viable or dead cells after thawing
Discussion
Fibroblasts represent a very interesting cell type. They can be easily isolated from different body sites and are easily cultured in vitro. Although biopsies are considered an invasive procedure, patient-derived skin fibroblasts have been reported as reliable sources for medical research purposes, with afford advantages over transformed cell lines. Since the primary explants methods, initiated by Harrison (1907) and Carrel (1912) (Carrel 1912; Harrison 1907), many techniques have been developed and are widely used.
In mammals, fibroblasts are highly heterogeneous. To enhance the successful establishment of a fibroblast cell line, several factors are involved, such as the time from biopsy to establishment of tissue culture and the body region (Villegas and McPhaul 2005). A comparative study made with gene expression of 50 fetal and adult human cultured fibroblasts derived from skin of 16 different body sites displayed distinct transcriptional patterns, suggesting that fibroblasts from different body sites should be considered distinct cell types, which reflect the fact that these cells are probably related to diverse physiological processes (Chang et al. 2002). Such heterogeneity may create interpretation problems when comparing fibroblasts derived from different body parts, from different subjects. Thus, it is important to consider what is the purpose of each research.
The use of fibroblasts in medical research allowed studies that helped to elucidate the pathogenesis of several diseases. Since the development of iPSCs (Takahashi et al. 2007), fibroblasts have been largely used in modeling diseases (Freitas et al. 2012; Martinez-Santamaria et al. 2012; Marchetto et al. 2010; Dimos et al. 2008; Park et al. 2008).
In this work, we aimed to evaluate the effectiveness of the establishment of primary fibroblast cell lines from eyelid, back of the ear, abdominal cesarean scar and groin samples, considering also the easy access and the aesthetics, especially because these are body sites targeted for plastic surgeries or are not usually exposed. Our results inferred that fibroblasts could be easily obtained by explant cultures using skin from eyelid, back of the ear and scar. Regardless of these three sites they provided good cultured cells, fibroblasts obtained from scar revealed higher proliferation rate and higher viability (81 %) after thawing when compared with fibroblasts from eyelid and back of the ear. A possible explanation for our observation could be provided taking a better look on wound healing process. During wound healing, fibroblasts are stimulated by several factors, especially by secreted factors present in damaged tissue, including growth factors, like transforming growth factor-β (TGFβ), epidermal growth factor (EGF), platelet-derived growth factor (PDGF) and fibroblast growth factor 2 (FGF2), released from injured epithelial cells and infiltrating mononuclear cells, such as monocytes and macrophages (Zeisberg et al. 2000).
In the healing process, fibroblasts invade the lesion, generate ECM to serve as a scaffold for other cells, and possess cytoskeletal elements that facilitate contractions of healing wounds, essential to diminish the size of healing and approximate the margins of the lesion (Tomasek et al. 2002). So far, fibroblasts are considered the main mediators of tissue fibrosis and scar formation (Kalluri and Zeisberg 2006). In addition, in wound healing, fibroblasts secret higher levels of ECM constituents and proliferate more than in healthy organs (Muller and Rodemann 1991; Castor et al. 1979). This amplified activity suggests cell activation processes (Castor et al. 1979). Once the wound is repaired, is not elucidated if the activated fibroblasts revert to a resting phenotype, go on an apoptosis process or if the tissue is repopulated by resting fibroblasts from the adjacent tissue (Tomasek et al. 2002). Conversely, in tissue fibrosis, fibroblasts keep their activated state, often until organ death. So fibroblasts isolated from fibrotic tissue maintain their activated phenotype when cultured in vitro (Orimo et al. 2005; Muller and Rodemann 1991). Considering all of that information, it seems to be expected that fibroblasts from scar presented an advantage regarding proliferative rate and survival advantages compared with fibroblasts obtained from other sites used in this work, but more studies are necessary to verify this findings, including other body sites and expression profile of fibroblast factors. Last, based on our results, fibroblasts obtained from scars could be excellent candidates for cell banking, for genetic diseases studies and for cellular reprogramming.
Conclusions
In this work we present feasible body sites for skin biopsy, as eyelid, back of the ear, groin and abdominal scar considering fibroblast cell isolation. Our results revealed that fibroblasts isolated from abdominal scar presented higher cell proliferation rates and also higher cell viability after cryopreservation. This seems to be in agreement with the physiology of wound repair, but more studies should be carried out in order to verify the reproducibility of this finding, even collecting more samples from the same individual, including scar biopsy. In addition, considering the easy transport and culture protocol, we inferred that fibroblasts are an excellent cell source and good candidates for research as well as for cell banking and further studies using cell modeling.
Acknowledgments
We are thankful for USP, UCSD and FAPESP for the financial support provided for this research.
References
- Acrani GO, Gomes R, Proença-Módena JL, da Silva AF, Carminati PO, Silva ML, Santos RI, Arruda E (2010) Apoptosis induced by Oropouche virus infection in HeLa cells is dependent on virus protein expression. Virus Res 149:56–63 [DOI] [PubMed]
- Carmichael J, DeGraff WG, et al. Evaluation of a tetrazolium-based semiautomated colorimetric assay: assessment of chemosensitivity testing. Cancer Res. 1987;47:936–942. [PubMed] [Google Scholar]
- Carrel A. On the permanent life of tissues outside of the organism. J Exp Med. 1912;15:516–528. doi: 10.1084/jem.15.5.516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castor CW, Wilson SM, Heiss PR, Seidman JC. Activation of lung connective tissue cells in vitro. Am Rev Respir Dis. 1979;120:21. doi: 10.1164/arrd.1979.120.1.101. [DOI] [PubMed] [Google Scholar]
- Chang HY, Chi JT, Dudoit S, Bondre C, van de Rijn M, Botstein D, Brown PO. Diversity, topographic differentiation, and positional memory in human fibroblasts. Proc Natl Acad Sci USA. 2002;99:12877–12882. doi: 10.1073/pnas.162488599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang T, Zheng W, Tsark W, Bates S, Huang H, Lin RJ, Yee JK (2011) Brief report: phenotypic rescue of induced pluripotent stem cell-derived motoneurons of a spinal muscular atrophy patient. Stem Cells 29:2090–2093 [DOI] [PubMed]
- Dimos JT, Rodolfa KT, Niakan KK, Weisenthal LM, Mitsumoto H, Chung W, Croft GF, Saphier G, Leibel R, Goland R, Wichterle H, Henderson CE, Eggan K (2008) Induced pluripotent stem cells generated from patients with ALS can be differentiated into motor neurons. Science 321:1218–1221 [DOI] [PubMed]
- Duvall M. Atlas d’Embryologie. Paris: Masson; 1879. [Google Scholar]
- Freitas BCG, Trujillo CA, Carromeu C, Yusupova M, Herai R, Muotri AR (2012) Stem cells and modeling of autism spectrum disorders. Exp Neurol. doi:10.1016/j.expneurol.2012.09.017 [DOI] [PMC free article] [PubMed]
- Harrison RG. Observations on the living developing nerve fiber. Proc Soc Exp Biol Med. 1907;4:140–143. doi: 10.3181/00379727-4-98. [DOI] [Google Scholar]
- Jones HW Jr, McKusick VA, Harper PS, Wuu KD (1971) George Otto Gey. (1899–1970). The HeLa cell and a reappraisal of its origin. Obstet Gynecol 38:945–949 [PubMed]
- Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6:392–401. doi: 10.1038/nrc1877. [DOI] [PubMed] [Google Scholar]
- Marchetto MC, Carromeu C, Acab A, Yu D, Yeo GW, Mu Y, Chen G, Gage FH, Muotri AR (2010) A model for neural development and treatment of Rett syndrome using human induced pluripotent stem cells. Cell 143:527–539 [DOI] [PMC free article] [PubMed]
- Martinez-Santamaria L, Guerrero-Aspizua S, Del Río M (2012) Skin bioengineering: preclinical and clinical applications. Actas Dermosifiliogr 103:5–11 [DOI] [PubMed]
- Muller GA, Rodemann HP. Characterization of human renal fibroblasts in health and disease. I. Immunophenotyping of cultured tubular epithelial cells and fibroblasts derived from kidneys with histologically proven interstitial fibrosis. Am J Kidney Dis. 1991;17:680–683. doi: 10.1016/S0272-6386(12)80351-9. [DOI] [PubMed] [Google Scholar]
- Odell A, Askham J, Whibley C, Hollstein M (2010) How to become immortal: let MEFs count the ways. Aging (Albany NY) 2:160–165 [DOI] [PMC free article] [PubMed]
- Orimo A, Gupta PB, Sgroi DC, Arenzana-Seisdedos F, Delaunay T, Naeem R, Carey VJ, Richardson AL, Weinberg RA (2005) Stromal fibroblasts present in invasive human breast carcinomas promote tumor growth and angiogenesis through elevated SDF-1/CXCL12 secretion. Cell 121:335–348 [DOI] [PubMed]
- Park IH, Arora N, et al. Disease-specific induced pluripotent stem cells. Cell. 2008;134:877–886. doi: 10.1016/j.cell.2008.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsonage G, Filer AD, et al. A stromal address code defined by fibroblasts. Trends Immunol. 2005;26:150–156. doi: 10.1016/j.it.2004.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ring KL, Tong LM, et al. Direct reprogramming of mouse and human fibroblasts into multipotent neural stem cells with a single factor. Cell Stem Cell. 2012;11:100–109. doi: 10.1016/j.stem.2012.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- Tarin D, Croft CB. Ultrastructural features of wound healing in mouse skin. J Anat. 1969;105:189–190. [PubMed] [Google Scholar]
- Todaro GJ, Green H. Quantitative studies of the growth of mouse embryo cells in culture and their development into established lines. J Cell Biol. 2010;17:299–313. doi: 10.1083/jcb.17.2.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomasek JJ, Gabbiani G, et al. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol. 2002;3:349–363. doi: 10.1038/nrm809. [DOI] [PubMed] [Google Scholar]
- Villegas J, McPhau Nl (2005) Establishment and culture of human skin fibroblasts. Curr Protoc Mol Biol Chapter 28: Unit 28 3 [DOI] [PubMed]
- Virchow R (1858) Die Cellularpathologie in Ihrer Begruendung auf Physiologische und Pathologische Gewebelehre. Hirschwald, A., Berlin, Germany [PubMed]
- vom Brocke J, Schmeiser HH, et al. MEF immortalization to investigate the ins and outs of mutagenesis. Carcinogenesis. 2006;27:2141–2147. doi: 10.1093/carcin/bgl101. [DOI] [PubMed] [Google Scholar]
- Zeisberg M, Strutz F, Muller GA. Role of fibroblast activation in inducing interstitial fibrosis. J Nephrol. 2000;13:S111–S120. [PubMed] [Google Scholar]