Abstract
The use of synthetic materials to repair craniofacial defects is increasing today and will increase further in the future. Because of the complexity of the anatomy in the head and neck region, reconstruction and augmentation of this area pose a challenge to the surgeon. This review discusses key facts and applications of traditional reconstruction bone substitutes, also offering comparative information. It then describes the properties and clinical applications of bioactive-glass (B-G) and its variants in oral and maxillofacial surgery, and provides clinical findings. The discussion of each compound includes a description of its composition and structure, the advantages and shortcomings of the material, and its current uses in the field of osteoplastic and reconstructive surgery. With a better understanding of the available alloplastic implants, the surgeon can make a more informed decision as to which implant would be most suitable in a particular patient.
Keywords: osteoplastic and reconstructive surgery, alloplastic materials, bioactive glasses
Successful repair of skull and facial defects, whether arising from trauma, tumor resection, or congenital disorders, continues to be a major challenge to oral and maxillofacial surgeons. Techniques vary, depending on the nature of the defect and quantity of tissue required. Reconstructions with autologous hard tissues have a long history with good clinical outcomes.1 However, bone autografts have certain disadvantages, such as limited availability when the volume of the defect is sizable, reduced dimensional stability together with an additional surgical site for the bone harvest, which prolongs operation time, convalescence, and donor-site morbidity.2 Recently, several new alloplastic materials have expanded our arsenal of reconstructive options. Their benefits are not only the avoidance of donor-site morbidity but also the increasing availability and ready usage of biomaterials, three-dimensional stability, their cost effectiveness in reduction of the operating time, known composition and safety.3 Nevertheless, a limited number of these products approach equivalence to autologous transplants.4 Failure of most implants originates at the interface between the biomaterial and its host tissue.5 This means that surface activity of the alloplast is critical in evaluating its suitability for bone replacement, particularly if the area to be reconstructed will be subject to stress loading. Bioactive implants provide a potential solution to the problem of interface failure.5 A bioactive material is defined as one that elicits a specific biological response and forms a living bond with the host tissues,5 rather than forming an interfacial layer of scar tissue. This process prevents formation of a fibrous capsule surrounding the implant by the adhesion of repair tissues. Hench et al5 reported the first bioactive material in 1971. They tested whether a phosphate containing silica-based material could fulfill the criterion of tissue bonding. The major breakthrough was that a degradable glass of the composition SiO2 45, Na2O 24.5, CaO 24.5, and P2O5 6 in weight percent (wt%), later termed 45S5, formed a bond with living bone so strong that it could not be removed without breaking the bone.5 The selection of the composition was ideal; the low silica content made the glass easy to melt but also gave it the ability to form a surface layer of hydroxyapatite (HA) chemically and structurally similar to the mineral phase of human bone, following the immediate release of ionic species from the bulk material in contact with body fluids.6 It has been shown that the glass dissolves at a rate equal to that at which new host tissue is remodeled, serving as a biocompatible interface along which bone cells migrate due to its osteogenic properties.5 Notably, the constituents are physiological chemicals found in the body, typically silicium, sodium, potassium, magnesium, oxygen, calcium, and phosphorus. According to several studies, the concentration of these chemicals never rise to levels that could disturb the adjacent tissues.6 7 In this respect, biologically active glasses have equally demonstrated considerable antibacterial8 and angiogenesis-promoting characteristics.9 The antimicrobial properties exhibited are likely multifactorial involving the creation of a local alkaline environment and the resistance of the material to bacterial adhesion and biofilm formation.10 11 Integration of mesenchymal stem cells and various growth factors such as vascular endothelial growth factor serve to augment the materials ability to repair tissue defects and stimulate neovascularization.12 Hench's 45S5 bioactive-glass (B-G), known also as Bioglass®, as it was trademarked by the University of Florida, and its variant products have been used in a variety of different shapes such as plates, rods, or rigid devices as well as in the form of particulate in many medical applications (Fig. 1).13 14 15 16 17 18 19 20 21 22 Clinically, these surface-reactive glass–ceramic biomaterials have been thoroughly investigated by oral and maxillofacial surgeons (Table 1). This article reviews their use both in craniomaxillofacial trauma and bone reconstructive surgery, as well as in oral surgery, starting with the former.
Fig. 1.
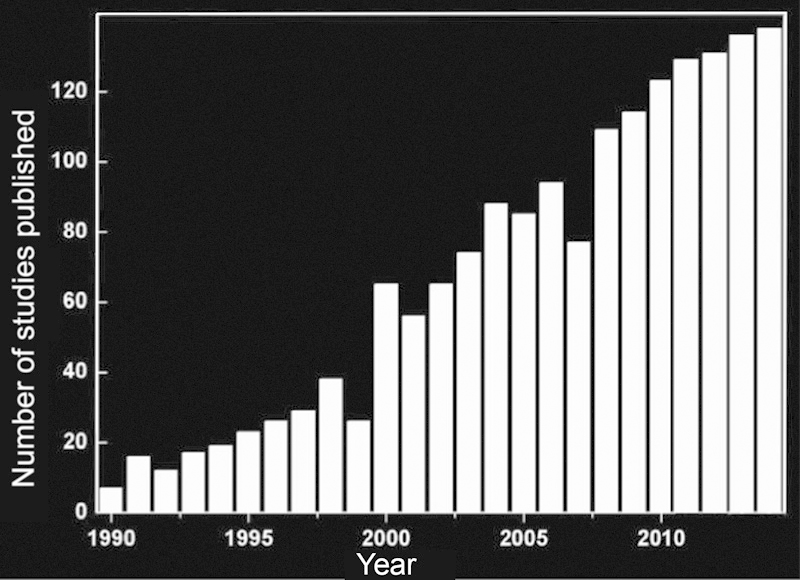
Number of studies published per year in the field of biologically active glass ceramics (B–G and its variants). Compiled from a literature search in Web of Science performed in December 2014.
Table 1. Prospective randomized clinical studies evaluating the efficacy of B-G and its variants in craniomaxillofacial bone reconstruction and oral surgery.
| Title | Author | Journal | Scope | Patients and methods | Results | Conclusion |
|---|---|---|---|---|---|---|
| Using 45S5 Bioglass cones as endosseous ridge maintenance implants to prevent alveolar ridge resorption: a 5-year evaluation | Stanley et al | Int J Oral Maxillofac Implants 1997;12(1):95–105 | To examine the effectiveness of B-G 45S5 cones as space fillers after removal of tooth roots to delay the resorption of alveolar ridges | Retrospective review of 168 implants in 20 recalled patients with a mean postimplantation interval of 63.2 mo | High rate (85.7%) of cone retention after 5 y | Placement of B-G 45S5 into fresh sockets promoted alveolar ridge preservation |
| Particulate bioglass as a grafting material in the treatment of periodontal intrabony defects | Zamet et al | J Clin Periodontol 1997;24(6):410–418 | To evaluate the effects of B-G 45S5 in the treatment of periodontal intrabony defects | Retrospective review of 20 patients. Clinical follow-up measurements were recorded at baseline, 3 mo and 1 y. Standardized radiographs for computer-assisted densitometric image analysis (CADIA) were taken at baseline, immediately postoperatively and at 1 y | CADIA data showed a significant increase in radiographic density and volume between the defects treated with B-G 45S5 when compared with those treated with surgical debridement only. Probing pocket depth and attachment level showed significant in sites treated with B-G 45S5 | Results demonstrated the efficiency of B-G 45S5 as an adjunct to conventional surgery in the treatment of intrabony defects |
| Comparison of Bioactive glass synthetic bone graft particles and open debridement in the treatment of human periodontal defects. A clinical study | Froum et al | J Periodontol 1998;69(6):698–709 | To compare the repair response of B-G 45S5 and open debridement in the treatment of human periodontal osseous defects | Retrospective review of 16 patients. Radiographs and soft tissue presurgical measurements were repeated at 6, 9, and 12 mo. At 12 mo, all sites were surgically re-entered to record osseous measurements | Significantly greater mean probing depth reduction was noted, and clinical attachment level gain was significantly improved in the B-G 45S5 group compared with the controls. Also, there was significantly less gingival recession, and defect fill reduction was significantly greater in the B-G 45S5 sites compared with the control sites | B-G 45S5 showed significant improvements in clinical parameters compared with open flap debridement |
| Clinical evaluation of bioactive glass in the treatment of periodontal osseous defects in humans | Lovelace et al | J Periodontol 1998;69(9):1027–1035 | To compare the use of B-G 45S5 to demineralized freeze-dried bone allograft (DFDBA) in the treatment of human periodontal osseous defects | Paired osseous defects in 15 patients with moderate to advanced adult periodontitis were randomly selected to receive grafts of B-G 45S5 or DFDBA. Clinical follow-up measurements were taken the day of surgery and at the 6-mo re-entry surgery | The results indicated that both treatments provided soft and hard tissue improvements when compared with baseline. No statistical difference was found when comparing sites grafted with B-G 45S5 to the use of DFDBA | This study showed that B-G 45S5 was capable of producing results in the short term (6 mo) similar to that of DFDBA when used in moderate to deep intrabony periodontal defects |
| Alveolar ridge reconstruction and/or preservation using root form bioglass cones | Yilmaz et al | J Clin Periodontol 1998;25(10):832–839 | To investigate the efficacy of root form B-G 45S5 cones implanted extraction sites to avoid deformities of the residual alveolar ridge in the maxillary anterior region | Retrospective review of 16 patients. Alveolar ridge width and height measurements were obtained using study casts preoperatively, immediately postoperatively, and at 3 and 12 mo after operation | No dehiscences were detected. The differences between preoperative original ridge height and width and postoperative measurements were not statistically significant | Results demonstrated the efficiency of this method in preserving alveolar ridges following tooth extraction, particularly relevant in relation to preparation for subsequent restorative treatment |
| A bioactive glass particulate in the treatment of molar furcation invasions | Anderegg et al | J Periodontol 1999;70(4):384–387 | To evaluate the effects of B-G 45S5 in the treatment of mandibular molar furcation invasion defects. Patients received surgical therapy using B-G 45S5 compared with open flap debridement alone | Retrospective review of 15 patients. Clinical follow-up measurements were recorded at 3 and 6 mo postoperatively | The results of therapy were statistically significant in the defects treated with B-G 45S5 | Results demonstrated the efficiency of B-G 45S5 in the treatment of class II furcation defects regarding the clinical parameters of probing depth reduction and the reduction in bleeding on probing |
| Histological observations on biopsies harvested following sinus floor elevation using a bioactive glass material of narrow size range | Tadjoedin et al | Clin Oral Implants Res 2000;11(4):334–344 | To evaluate the bone augmenting capacity of B-G 45S5 particles in human sinus floor elevations | Retrospective review of 10 patients. Bilateral grafting was performed using a 1:1 mixture of autogenous bone particles (from iliac crest) and B-G 45S5 particles at one side (experimental side), and bone particles only at the other side (control side, split mouth design). Histomorphometrical measurements were repeated at 4, 6, and 16 mo from bone biopsies |
B-G 45S5 particles transformed and became excavated with time, starting at 4 mo, and their centers gradually filled with bone tissue. All B-G 45S5 particles had disappeared by resorption at 16 mo after grafting and had been replaced by bone tissue. Parameters of bone turnover indicated that bone remodeling was very active at both sides | Results demonstrated that a 1:1 mixture of autogenous bone/B-G 45S5 particles seemed a promising alternative to autogenous bone only, when low amounts of bone tissue are available for sinus augmentation |
| Reconstruction of orbital floor fractures using bioactive glass | Kinnunen et al | J Craniomaxillofac Surg 2000;28(4):229–234 | To compare the use of B-G S53P4 implants with autogenous cartilage grafts for the repair of orbital floor defects after trauma | Retrospective review of 28 patients performed from 1995 to 1999. Clinical and radiological follow-up between 2 and 5 y after surgery | Postoperative tomograms showed adequate maintenance of orbital and maxillary sinus volume without any evidence of resorption or complications in either group | Bioactive glass implants were well tolerated and seemed to be a promising repair material for orbital floor fractures as well as the use of autologous bone alone. Their use provided favorable healing, caused new bone formation, and led to less morbidity as no donor-site operation was needed |
| Clinical comparison of bioactive glass bone replacement graft material and expanded polytetrafluoroethylene barrier membrane in treating human mandibular molar class II furcations | Yukna et al | J Periodontol 2001;72(2):125–133 | To examine the response of mandibular molar class II furcations to treatment with either B-G 45S5 or expanded polytetrafluoroethylene (ePTFE) barrier membrane | Retrospective evaluation until surgical reentry at 6 mo in 27 pairs of mandibular molars in 27 patients with moderate to advanced periodontitis | Follow-up measurements demonstrated essentially similar clinical results with both treatments for bone and soft tissue changes | Equal clinical results with B-G 45S5 and ePTFE barriers. B-G 45S5 was associated with simpler application and required no additional material removal procedures |
| Maxillary sinus floor augmentation using bioactive glass granules and autogenous bone with simultaneous implant placement | Cordioli et al | Clin Oral Implants Res 2001;12(3):270–278 | To evaluate the use of B-G 45S5 combined with autogenous bone (4:1 ratio) as grafting material for maxillary sinus augmentation with simultaneous implant placement | Unilateral or bilateral sinus augmentation was performed in 12 patients. Presurgical CT scans and core biopsy specimens were evaluated between 9 and 12 mo | An increase in mineralized tissue height of 7.1 ± 1.6 mm was evident when comparing the presurgical and 9–12 mo CT scans. Histological evaluation yielded a mean of 30.6 ± 5.7% of bone tissue in the grafted sites | B-G 45S5 with autologous bone graft used in one-stage sinus augmentation yielded sufficient quality and volume of mineralized tissue for predictable simultaneous implant placement in patients with 3–5 mm of bone height before grafting |
| Effects of pretreatment clinical parameters on bioactive glass implantation in intrabony periodontal defects | Park et al | J Periodontol 2001;72(6):730–740 | To examine the effectiveness of B-G 45S5 implantation in intrabony periodontal defects | Retrospective evaluation 6 mo after surgery in 38 intrabony defects from 38 patients with chronic periodontitis | Comparative observation between preoperative and postoperative clinical parameters (probing depth, clinical attachment level, bone probing depth, and gingival recession) showed significantly greater improvements after B-G 45S5 implantation | Use of B-G 45S5 in flap operations resulted in significantly greater improvements over flap operation alone |
| Repair of orbital floor fractures with bioactive glass implants | Aitasalo et al | J Oral Maxillofac Surg 2001;59(12):1390–1395; discussion 1395–1396 | To examine the effectiveness of B-G S53P4 implants for the repair of orbital floor defects caused by blunt facial trauma | Retrospective review of 36 patients performed from 1995 to 1999. Clinical and radiological follow-up at 1, 3, and 12 mo after surgery | No foreign body reaction and no inflammation in the bone or soft tissue. No sign of resorption or infection, nor postoperative extrusion, hemorrhage, or displacement. New bone formation around the implants. Good functional and cosmetic results at the 1-y follow-up | B-G S53P4 well-tolerated material in orbital floor reconstruction. It provided a favorable environment for an uncomplicated healing process |
| Clinical evaluation of an enamel matrix protein derivative combined with a bioactive glass for the treatment of intrabony periodontal defects in humans | Sculean et al | J Periodontol 2002;73(4):401–408 | To compare the treatment of deep intrabony defects with a combination of enamel matrix protein derivative (EMD) and B-G 45S5 to B-G 45S5 alone | Retrospective review of 28 patients with chronic periodontitis. Soft tissue measurements (evaluation of probing depth, clinical attachment level, and gingival recession) were made at baseline and at 1 y following therapy | No differences in any of the investigated parameters were observed at baseline between the two groups. Healing was uneventful in all patients. At 1 y after therapy, no statistically significant differences in any of the investigated parameters were observed between the test and control groups | The combination of EMD and B-G 45S5 did not seem to additionally improve the clinical outcome of the therapy with B-G 45S5 alone |
| Bioactive glass granules as a bone adjunctive material in maxillary sinus floor augmentation | Turunen et al | Clin Oral Implants Res 2004;15(2):135–141 | To compare the use of B-G S53P4 granules mixed with autologous bone (AB) chips harvested from the iliac crest (1:1 mixture) to bone alone in bilateral sinus floor augmentation procedure | Retrospective review of 17 patients. Biopsies for histological, scanning electron microscopy (SEM), and energy dispersive X-ray (EDX) analyses were taken after 21, 34, 49, and 62 wk | Histological evaluation and histomorphometrical analysis performed from the SEM images revealed similar results between the two groups. EDX analysis showed a tight contact and chemical bonding between the glass and bone | B-G S53P4 granules could be used together with AB chips as bone adjunctive material in maxillary sinus floor augmentation procedures, thus decreasing the amount of bone needed |
| Bioactive glass S53P4 in frontal sinus obliteration: a long-term clinical experience | Peltola et al | Head Neck 2006;28(9):834–841 | To use B-G S53P4 as obliteration material in a series of osteoplastic frontal sinus operations on patients suffering from chronic frontal sinusitis | Retrospective review of 42 patients. Clinical and histopathologic follow-up at 1, 5, and 10 y after surgery | Histopathologic samples revealed bone formation. Fourier-transform infrared (FTIR) studies showed bone produced by B-G S53P4 to be similar to natural frontal bone. Microbiologic cultures obtained with histologic samples revealed no growth of bacteria | Accurate obliteration of sinuses achieved in 39 patients and uneventful recovery in 92% of the patients showed that B-G S53P4 was a reliable frontal sinus obliteration material, providing favorable conditions for total bony sinus obliteration |
| Bioabsorbable membrane and bioactive glass in the treatment of intrabony defects in patients with generalized aggressive periodontitis: results of a 5-year clinical and radiological study | Mengel et al | J Periodontol 2006;77(10):1781–1787 | To compare the long-term effectiveness of bioabsorbable membrane Gore Resolut XT (RXT) (Gore Resolut® XT W.L. Gore & Associates Inc., Newark, DE) and B-G 45S5 in the treatment of intrabony defects in patients with generalized aggressive periodontitis | Clinical and radiological evaluation before surgery, at 6 mo and every year for 5 y after surgery of 16 patients: 22 intrabony defects treated with RXT and 20 treated with B-G 45S5 | Comparative observation of clinical parameters (plaque index, gingival index, probing depth, bleeding on probing, gingival recession, clinical attachment level, and tooth mobility) at baseline and every year for 5 y showed highly significant improvements with both groups | No significant difference between the use of RXT membrane and B-G 45S5 in the treatment of intrabony defects in patients with generalized aggressive periodontitis. Both regenerative materials gave good results in the treatment of intrabony periodontal defects |
| Four-year results of a prospective-controlled clinical study evaluating healing of intrabony defects following treatment with an enamel matrix protein derivative alone or combined with a bioactive glass | Sculean et al | J Clin Periodontol 2007;34(6):507–513 | To evaluate the 4-y results following regenerative periodontal surgery at intrabony defects with either a combination of enamel matrix protein derivative (EMD) and B-G 45S5 or with EMD alone | Retrospective review of 25 patients randomly treated with either EMD or B-G 45S5, 13 treated with EMD alone (control). Measurements were recorded at baseline, at 1 and 4 y following therapy | Evaluation of clinical attachment level, probing depth, and gingival recession showed no statistically significant differences in any of the investigated parameters at 1 and 4 y between the treatment groups | Results indicated that the clinical improvements obtained with both regenerative modalities could be maintained over a period of 4 y |
| Clinical evaluation of platelet-rich plasma and bioactive glass in the treatment of intra-bony defects | Demir et al | J Clin Periodontol 2007;34(8):709–715 | To evaluate the effect of B-G 45S5 with and without platelet-rich plasma (PRP) on the clinical healing of intrabony defects | Retrospective review of 29 patients. Intrabony defects were randomly treated with either PRP/B-G 45S5 or B-G 45S5 alone. Clinical parameters were recorded at baseline and repeated 9 mo after surgery and surgical re-entries were also performed |
Pocket depth reduction, clinical attachment gain, and defect fill were noted in both groups. None of the differences between the two treatment modalities were statistically significant | Results showed that both treatment modalities were effective in the treatment of intrabony defects. The results also showed that using PRP with B-G 45S5 has no additional benefit in the reduction of pocket depth, clinical attachment gain, and defect fill |
| A cost-effectiveness evaluation of enamel matrix derivatives alone or in conjunction with regenerative devices in the treatment of periodontal intra-osseous defects | Listl et al | J Clin Periodontol 2010;37(10):920–927 | To identify the most cost-effective approach to treatment of infrabony lesions with enamel matrix derivatives (EMD) | Costs and clinical outcomes of 12 different treatment techniques (including flap operation, EMD alone, and EMD in association with B-G 45S5) were compared | The most cost-effective treatment option was identified on the basis of the maximum net benefit criterion. The maximum net benefit was achieved by treatment with EMD in conjunction with B-G 45S5 |
If EMD use is indicated, EMD in conjunction with either B-G 45S5 is more cost effective than EMD alone |
| Comparison of bioactive glass coated and hydroxyapatite coated titanium dental implants in the human jaw bone | Mistry et al | Aust Dent J 2011;56(1):68–75 | To evaluate and compare the behavior of hydroxyapatite and B-G 45S5 coated implants (62 implants) in osseous tissue following implantation in 31 patients | B-G 45S5 coating was applied by vitreous enameling technique. The outcome was assessed up to 12 mo after prosthetic loading using different clinical and radiological parameters | B-G 45S5 coating materials were nontoxic and biocompatible. Overall results showed that B-G 45S5 coated implants were as equally successful as hydroxyapatite in achieving osseointegration and supporting final restorations | B-G 45S5 is a good alternative coating material for dental implants, possibly allowing wider case selection criteria together with improved integration rates even in the more challenging osteoporotic and medically compromised patients |
| Autogenous cortical bone and bioactive glass grafting for treatment of intraosseous periodontal defects | Sumer et al | Eur J Dent 2013;7(1):6–14 | To compare the effectiveness of autogenous cortical bone (ACB) and B-G 45S5 grafting for the regenerative treatment of intraosseous periodontal defects | Via a split-mouth design, 15 chronic periodontitis patients who had probing pocket depths (PPDs) of ≥6 mm following initial periodontal therapy were randomly assigned to receive two treatments in contralateral areas of the dentition: ACB grafting and B-G 45S5 grafting. The parameters compared in the patients were preoperative and 6-month postoperative PPDs, clinical attachment levels (CALs), and radiographic alveolar bone heights | Both treatment modalities resulted in significant changes in postoperative measurements when compared with preoperative values. PPDs were decreased, CALs were increased, and radiographic alveolar bone heights were increased in patients treated with both ACB grafting and B-G 45S5 grafting. Differences between the treatments were not statistically significant | Both ACB and B-G 45S5 grafting led to significant improvements in clinical and radiographic parameters 6 mo postoperatively. These results suggest that either an ACB graft, which is completely safe with no associated concerns about disease transmission and immunogenic reactions, or a B-G 45S5 graft, which has an unlimited supply, can be selected for regenerative periodontal treatment |
| Adipose stem cells used to reconstruct 13 cases with cranio-maxillofacial hard-tissue defects | Sándor et al | Stem Cells Transl Med 2014;3(4):530–540 | To examine the effectiveness of B-G 45S5 scaffolds seeded with adipose-derived stem cells (ASCs) for the repair of craniomaxillofacial hard-tissue defects. Autologous adipose tissue was harvested from the anterior abdominal wall, and adipose-derived stem cells were cultured, expanded, and then seeded onto resorbable scaffold materials for subsequent reimplantation into hard-tissue defects | Retrospective review of 13 patients. Hard-tissue defects at four anatomically different sites, namely, frontal sinus (3 cases), cranial bone (5 cases), mandible (3 cases), and nasal septum (2 cases). Follow-up time ranged from 12 to 52 mo |
Successful integration of the construct to the surrounding skeleton was noted in 10 of the 13 cases. Two cranial defect cases in which nonrigid resorbable containment meshes were used sustained bone resorption to the point that they required the procedure to be redone. One septal perforation case failed outright at 1 y because of the postsurgical resumption of the patient's uncontrolled nasal picking habit | Results indicated that the clinical improvements obtained with the use of (ASCs) seeded in B-G 45S5 scaffolds could be maintained over a period of 4 y |
Calvarial Defects Reconstruction
Calvarial bone reconstruction is adopted to repair large skull bone defects resulting from the treatment of tumors, infections, trauma, intracranial hemorrhage, or infarction. These defects cause discomfort as well as other functional problems to patients. Reconstructive surgery serves to protect the brain, provides a satisfying aesthetic outcome, and frequently involves the use of preserved allogenous bone grafts, as not enough autologous hard tissues are available. However, the inherent drawbacks of this approach strongly urge clinicians and researchers to explore alternative treatment options.23 Currently, several alloplastic materials, such as metals, plastics, ceramics, and fiber-reinforced composites (FRCs) are used for the reconstruction of skull bone defects under compromised healing conditions.24 Metallic implants have been used alone25 or combined with bone dust or HA cement to enhance bone ingrowth and integrity in cranial bone reconstructions.26 Polymethylmethacrylate (PMMA), polyethylene, and polyether ketone have been used in various formulations.27 Although acrylic resins are readily available for immediate usage and can be easily shaped either with manual or handpiece instruments during the operation, they do not attach chemically to surrounding bone and, later on, foreign-body reactions have been observed.27 In cranioplasty, late plate exposure and alloplast displacement, because of lack of incorporation at the donor site, are practical disadvantages associated with the use of these materials, in which case the removal becomes necessary with a technically challenging operation.27
Previous studies have reported on 45S5 B-G particles mixed with autologous bone particles harvested from cranial burr holes as an adjunct to cranial vault reconstruction.28 29 This was done to reconstruct full-thickness defects in two young patients, when minimal spontaneous bone regeneration was expected. On follow-up computer tomography (CT) scans, these patients demonstrated conversion of the majority of the reconstructed defect to bone density within 6 months. At 4-year follow-up, both patients had stable reconstruction, and there was no need for reoperation or biopsy of the biomaterial.
A promising new development was represented by the use of customized porous implants, made of a supporting FRCs23 or PMMA30 framework and modified B-G (S53P4), to combine workability of resinous materials with innate bioactive/antimicrobial properties of the latter in calvarial23 and midface bone30 reconstructions. Beneficial effects of S53P4 particles with the composition SiO2 53, Na2O 23, CaO 20, and P2O5 4.0 wt% have been demonstrated experimentally10 11 and put to use for various clinical indications.13 14 16 31 32 33 In the manufacturing process, prototyping models of the defect regions were created with additive manufacturing technology and these templates were used to prepare the patient-specific implants (Fig. 2).23 30 The last mentioned were perforated with holes 1.5 mm in diameter to enhance body fluid perfusion, coated with S53P4 granules of 0.5 to 0.8 mm in size and the application of prostheses (overall average thickness of 3.5 mm) was done with either biodegradable fixation screws or sutures. In longitudinal clinical and radiological examinations, with follow-up times up to 4 years23 and 5 years,30 normal progressive wound healing with diminishing postoperative clinical symptoms was observed and the implants retained their original position regardless of the fixation type, providing the expected functional as well as aesthetic outcome at all time points. Furthermore, there were no long-term complications such as implant-induced skull resorption, or later inflammatory reactions and acute toxicities systemically. Readers should also be aware that there was no donor-site morbidity because the custom-made implants did not need any graft operations. The perforated structure of the implants favored tissue ingrowth, yet providing solid flexural modulus and mechanical strength, but the bioactive coating was regarded as the key attribute, especially for those patients with a history of previous reconstruction material removal because of infection.23 The strong porous structure of the composite implants mimicked surrounding bone, while S53P4 particles on their surface enabled new hard tissue formation by osteoblasts. The growth and maturation of natural bone in the porous inner layers provided firm adhesion, which in turn prevented long-term complications such as alloplast displacement. Finally, the synthetic implants did not evoke artifacts in postoperative imaging modalities for tumor follow-up. The bioactive coating was permeable to X-rays and performed in CT imaging (Fig. 2) as well as methacrylate alone.23 30 The same applies to magnetic resonance imaging (MRI), as S53P4 is nonmagnetic.
Fig. 2.
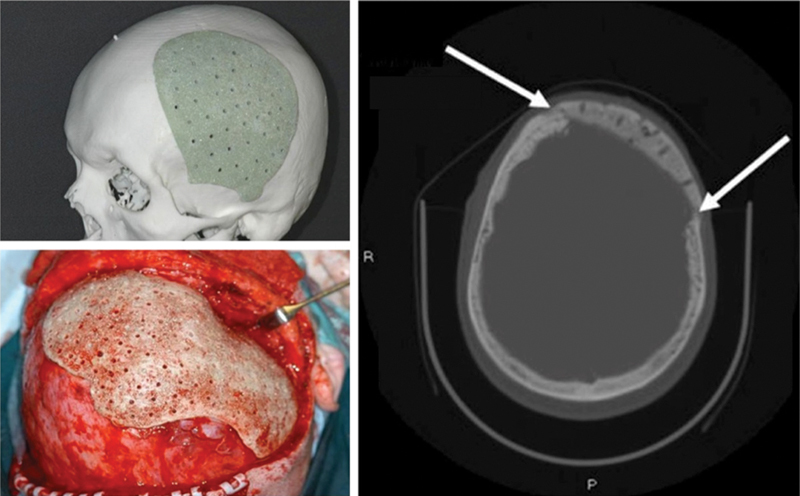
Additive manufacturing model of a large left calvarial bone defect with a tailor-made PMMA and B-G (S53P4) implant before operation (above, left). Intraoperative picture of a bioactive composite implant adjusted to its correct position in the calvarial bone defect. Note the 1.5 mm perforations to enhance tissue growth into the alloplastic material (below, left). CT scan of a left temporal bone defect 2 years after reconstruction (right). Implant is in the correct position in the skull. New bone formation between implant and surrounding bone is seen (white arrows). CT, computed tomography; PMMA, polymethylmethacrylate. (Adapted from Peltola et al.30)
Surgical Approaches to Orbit and Facial Skeleton
Today, a remarkable share of clinical challenges regarding the fronto-orbital area that require obliterative and reconstructive surgery can be resolved with biomaterials. Such clinical conditions can be chronic suppurative frontal sinusitis, mucoceles, pyoceles, complicated fractures (especially those involving the frontal sinus drainage pathway), and tumors (such as carcinoma). Frontal bone osteomyelitis with bone necrosis and frontal sinus diseases with orbital or intracranial extension after tumor removal are also presumed to be conditions for reconstruction with biomaterials. Defects after subcranial tumor resection in the anterior skull base often need a reconstruction material, and this is another field for the use of tissue replacement materials.
The earlier widely used autogenic tissues are associated with donor-site morbidity, whereas allogenic, animal-derived graft materials and bank tissues have a risk of biohazard infections.34 Microvascular free flaps can have aesthetic limitations. Endoscopic Lothrop-type procedures have been recommended to resolve the problems with chronic frontal sinusitis and, recently, even to avoid chronic frontal sinusitis after osteoplastic flap failure.34 Despite these advances in frontal sinus surgery, there are still problematic cases where the osteoplastic procedure with obliteration is preferable.
In bone reconstruction with alloplasts, the most important practical factors are an adequate knowledge of their composition along with awareness of specific advantages and disadvantages. Synthetic materials are available in any time and amount, whereas large reconstructions and reoperations may limit the use of autogenic grafts. They are manufactured under controlled conditions, and thus the exact compositions are well known. Another obvious advantage is avoidance of having to harvest autogenic tissues. This decreases the total time needed for surgery, and also the length of wounds is reduced. In experimental studies comparing synthetic biomaterials, B-Gs produced more new bone over the same period than materials such as HA and tricalcium phosphate,35 and the bone produced by the first was shown to be more similar to natural frontal bone. HA cement was associated with unsatisfactory long-term results due to exposure of the material, delayed inflammatory reactions, and infections originating in the frontal sinus area.36 37 Other distinctive qualities of B-Gs are ability to remain where placed even with adjacent suctioning, hemostasis, and incorporation into host bone without the fibrous encapsulation encountered with most other synthetic materials.38
Accurate filling with saline liquid-moistened B-G granules (S53P4 formula, 0.5–0.8 and 0.8–1.0 mm in size) was used successfully for contour restoration of the facial skeleton and frontal bone reconstruction,39 40 41 42 43 with uneventful outcomes or any evidence of resorption. Indeed, the B-G granule remnants seemed to maintain perdurable and uniform bony reconstruction.34 When using biologically active materials, it is advisable to know their resorption behavior, and this should closely match the bone formation rate at the reconstruction or implant sites. The slower degradation rate and lack of resorption presented by S53P4 may be due to glass composition, which has higher silica content than the parent 45S5 B-G.
Separate similar studies were performed with tailor-made, rigid B-G plates which were fitted into place for the repair of orbital floor fractures, with good maintenance of globe position during follow-up periods of 5 years,44 as well as after fronto-orbital tumor resections, with the only difference that in this case, implants were manufactured casting glass of the S53P4 composition.34 42 43 45 Without exception, the biomaterials used were well tolerated, with good functional and aesthetic outcomes (Figs. 3 and 4). The B-G plate met most of the criteria for a beneficial orbital wall reconstruction material. Besides, it represented the only orbital reconstruction material with antibacterial properties, which may be the reason why acute or late infections were not reported.45 This is a very important aspect in orbital wall reconstructions, where a connection between the orbit and the maxillary sinus is common. In fact, infections originating from the maxillary sinus can lead to the need to remove the reconstruction material.45 In histologic studies, special attention was paid to new bone formation and conversion of B-G.34 The results were in agreement with the findings of other studies,7 in which bonding between the glass implant surface and host tissues was formed by attachment of collagen fibers to newly formed silica-rich containing layers. Although B-G is a slowly biodegradable material, the plates were firmly attached to orbital bone structures when histological samples were harvested, indicating immobilization and incorporation to the orbital bone even without specific screw fixation.45
Fig. 3.
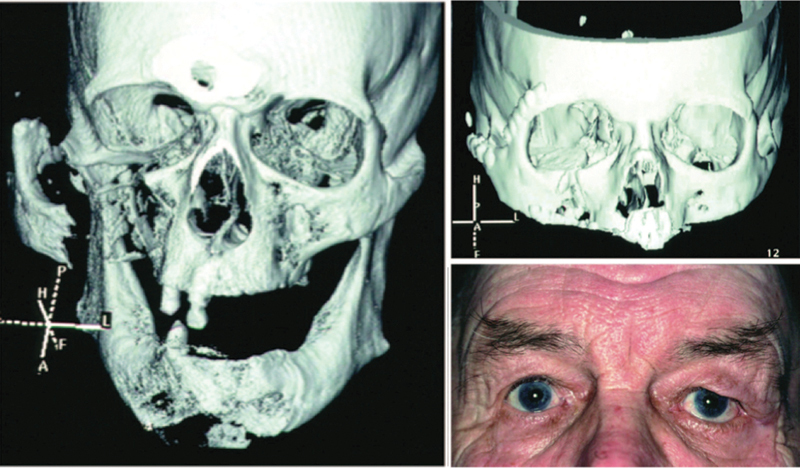
Three-dimensional CT scan of a complicated fracture of the maxillary sinus and zygomatic arch (left). CT at 1 year after reconstruction of the orbit (above, right) with the B-G implant. Photograph of the patient obtained 3 years after surgery (below, right). (Adapted from Aitasalo and Peltola.34)
Fig. 4.
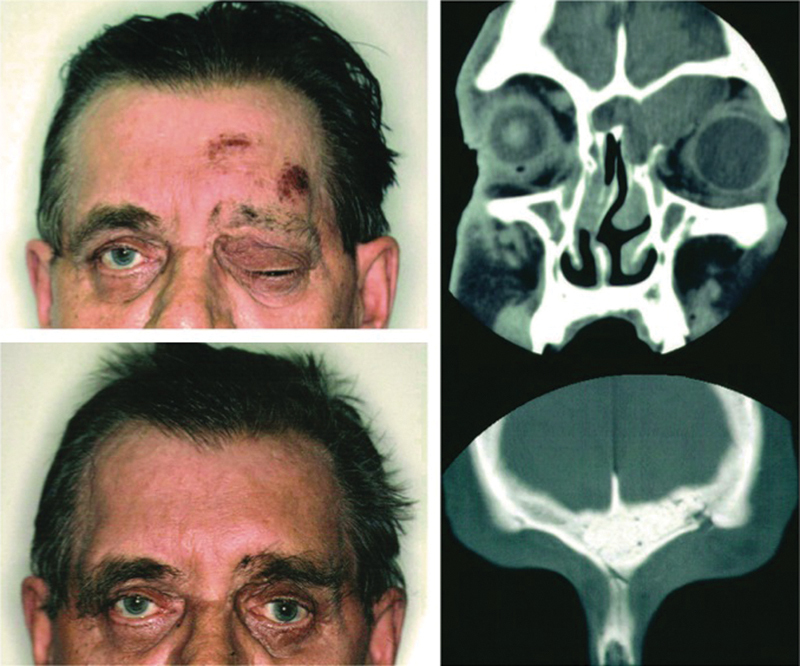
Mucocele of the left frontal sinus and orbit. Photograph of the patient (above, left) and sinus CT scan in coronal projection (above, right) of the patient before surgery. Photograph of the patient (below, left) and CT scan (below, right) of the patient after frontal sinus obliteration, with B-G and calvarial bone transplantation in the roof of the left orbit. (Adapted from Aitasalo and Peltola.34)
Paranasal Sinuses Surgery
The maxillofacial area is a unique challenge to the surgeon because it is related to infection-sensitive structures such as the paranasal sinuses, upper respiratory tract, and oral cavity. Notably, frequent infections, inflammatory disorders, posttraumatic conditions, and tumors affect the frontal sinus and present unique requirements for successful surgical treatment. Indications for frontal sinus obliteration include problematic conditions that cannot be resolved with functional endoscopic sinus surgery techniques due to irreversibly damaged natural drainage. Such indications can include chronic suppurative frontal sinusitis, mucoceles, pyoceles, complex fractures (especially those involving the drainage pathway), and tumors, such as osteomas.46
Finding a reliable, biocompatible, and safe material for frontal sinus obliteration has proven difficult. Abdominal fat is well established as a versatile and reliable material in frontal sinus obliteration.46 In addition, pericranial flaps are interesting alternatives in frontal sinus obliteration.46 Compared with HA cement, frontal sinus obliteration with autogenic fat is more cost effective but requires a longer operation time.46 However, a risk of donor-site morbidity is related to abdominal fat and autogenic bone transplantation.
Promising results have been described with the use of B-G in chronically infected nasal septum perforations19 and after canal wall down mastoidectomy.47 B-G-ceramic middle ear implants in ossicular chain reconstructions also showed good tolerance after 8 years.18 The material appeared to have a broader antimicrobial effect than HA, inhibiting their growth,48 and S53P4 glass granules (0.5–0.8 and 0.8–1.0 mm in diameter) moistened in sterile physiologic saline were subsequently used to obliterate the frontal sinuses with favorable results (Fig. 3).31 34 40 46 Moistened small granules of B-G are adherent and easy to handle for filling the cavity completely. Hence, a second operation needed for harvesting of autogenous material can be avoided. Repeated CT or MRI scans of frontal sinuses are the only way to accurately monitor the degree of obliteration and identify the patient at risk for recurrent disease after an osteoplastic procedure. A 5-year clinical follow-up study revealed uneventful outcomes,40 and bone formation with no loss of volume was reported in the obliterated frontal sinuses over a 10-year period.31 Peltola et al46 characterized the long-term histological healing process in B-G obliterated frontal sinuses. Special attention was given to osteogenesis, occurrence of connective tissue, and possible long-term structural changes in the remaining obliteration materials. The authors demonstrated formation of histologically normal bone without connective tissue in direct contact with the biomaterial particles, in the absence of inflammatory changes or foreign-body reactions. The antibacterial properties of B-G provide extraordinarily favorable conditions and are important factors in obliteration material, that is, in chronic infected frontal sinuses, with a relative risk of reinfection. The predictable healing process and the occlusion of the sinuses decrease the risk of relapse. Laboratory studies showed no abnormal changes in inflammatory parameters or in liver and kidney functions, confirming that B-G is a safe, stable material for permanent clinical frontal sinus obliteration.31
B-G particles have also been used to modify the maxillary sinuses. Maxilla grafting for elevation of the maxillary sinus floor with composite grafts of granules of S53P4 glass (80–90 wt%) and autologous bone (10–20 wt%) was shown to be as good as the treatment with autogenous bone alone, yielding the same quality and volume of mineralized tissue when a reasonable healing period is allowed.33 49 50 51 52 53 The use of autogenous bone was dictated by its osteogenic potential related to the number of surviving osteoblasts and osteoinductive effect brought about by the release of bone morphogenic proteins and other growth factors, which have the capacity to accelerate deposition of new bone along the graft material.52 Notwithstanding, the amount of bone needed was considerably decreased and donor-site morbidity alleviated. Using histomorphometric analysis, Tadjoedin et al53 found that the composite B-G mixture accelerated healing time for bone regeneration to 6 months, compared with 12 months for bone graft alone. Both treatment regimens resulted in stable bone at the reconstructed site. Accelerated bone healing for elevation of the maxillary sinus floor can allow for simultaneous bone augmentation of the sinus floor and placement of titanium implants for dental restoration in patients who would otherwise have insufficient maxillary bone for implant placement. Finally, this combination of findings provides further support for the conceptual premise that the antimicrobial activity of B-G against sinus pathogens31 48 might contribute to the resolution of inflammatory responses and provide extraordinarily favorable conditions for an uneventful healing process.49 53
Conclusion
Acting as a resorbable framework in which bone cells can grow,54 55 biologically active glasses are an important consideration when choosing the optimal biomaterial to be used as a bone substitute in craniomaxillofacial applications. Their bioactive properties allow for an osteoproductive environment in which the bone–biomaterial interface is uniquely stronger than it would be with other forms of alloplastic materials.
A review of the present literature supports clinical applications of prefabricated implants made of B-G ceramics, as has been used in calvarial and midface bone reconstructions as well as for the repair of orbital floor fractures. Also, present experience with B-G in particulate form, preferably mixed with small amount of autogenous bone, support the convenience of this biomaterial over alternate forms of synthetic graft materials. This protocol has been highly successful for elevation of the maxillary sinus floor in preparation for titanium implant placement in the atrophic maxilla. A similar protocol may prove useful in reconstruction of other areas of the head and neck.
Footnotes
Disclosure The authors deny any financial affiliations (e.g., employment, direct payment, stock holdings, retainers, consultantships, patent licensing arrangements, or honoraria) or involvement with any commercial organization with direct financial interest in the subject or materials discussed in this article. The authors are responsible for the content and writing of this article.
References
- 1.Beederman M, Alkureishi L W, Lam S, Warnke P, Reid R R. Exchange hybrid cranioplasty using particulate bone graft and demineralized bone matrix: the best of both worlds. J Craniofac Surg. 2014;25(2):451–454. doi: 10.1097/SCS.0000000000000491. [DOI] [PubMed] [Google Scholar]
- 2.Almaiman M, Al-Bargi H H, Manson P. Complication of anterior iliac bone graft harvesting in 372 adult patients from May 2006 to May 2011 and a literature review. Craniomaxillofac Trauma Reconstr. 2013;6(4):257–266. doi: 10.1055/s-0033-1357510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Eppley B L Alloplastic implantation Plast Reconstr Surg 199910461761–1783., quiz 1784–1785 [DOI] [PubMed] [Google Scholar]
- 4.Smeets R, El-Moawen A, Jung O. et al. From bench to application: current practices in tissue engineering and its realisation at maxillofacial units in Germany, Austria and Switzerland. J Craniomaxillofac Surg. 2014;42(7):1128–1132. doi: 10.1016/j.jcms.2014.01.043. [DOI] [PubMed] [Google Scholar]
- 5.Hench L L, Splinter R J, Allen W C, Greenlee T K. Bonding mechanisms at the interface of ceramic prosthetic materials. J Biomed Mater Res. 1971;5(6):117–141. [Google Scholar]
- 6.Hench L L. The story of Bioglass. J Mater Sci Mater Med. 2006;17(11):967–978. doi: 10.1007/s10856-006-0432-z. [DOI] [PubMed] [Google Scholar]
- 7.Wilson J, Pigott G H, Schoen F J, Hench L L. Toxicology and biocompatibility of bioglasses. J Biomed Mater Res. 1981;15(6):805–817. doi: 10.1002/jbm.820150605. [DOI] [PubMed] [Google Scholar]
- 8.Stoor P, Söderling E, Grenman R. Interactions between the bioactive glass S53P4 and the atrophic rhinitis-associated microorganism Klebsiella ozaenae. J Biomed Mater Res. 1999;48(6):869–874. doi: 10.1002/(sici)1097-4636(1999)48:6<869::aid-jbm16>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 9.Day R M. Bioactive glass stimulates the secretion of angiogenic growth factors and angiogenesis in vitro. Tissue Eng. 2005;11(5-6):768–777. doi: 10.1089/ten.2005.11.768. [DOI] [PubMed] [Google Scholar]
- 10.Leppäranta O, Vaahtio M, Peltola T. et al. Antibacterial effect of bioactive glasses on clinically important anaerobic bacteria in vitro. J Mater Sci Mater Med. 2008;19(2):547–551. doi: 10.1007/s10856-007-3018-5. [DOI] [PubMed] [Google Scholar]
- 11.Munukka E, Leppäranta O, Korkeamäki M. et al. Bactericidal effects of bioactive glasses on clinically important aerobic bacteria. J Mater Sci Mater Med. 2008;19(1):27–32. doi: 10.1007/s10856-007-3143-1. [DOI] [PubMed] [Google Scholar]
- 12.Rahaman M N, Day D E, Bal B S. et al. Bioactive glass in tissue engineering. Acta Biomater. 2011;7(6):2355–2373. doi: 10.1016/j.actbio.2011.03.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lindfors N C, Heikkilä J T, Koski I, Mattila K, Aho A J. Bioactive glass and autogenous bone as bone graft substitutes in benign bone tumors. J Biomed Mater Res B Appl Biomater. 2009;90(1):131–136. doi: 10.1002/jbm.b.31263. [DOI] [PubMed] [Google Scholar]
- 14.McAndrew J, Efrimescu C, Sheehan E, Niall D. Through the looking glass; bioactive glass S53P4 (BonAlive®) in the treatment of chronic osteomyelitis. Ir J Med Sci. 2013;182(3):509–511. doi: 10.1007/s11845-012-0895-5. [DOI] [PubMed] [Google Scholar]
- 15.Pernaa K, Koski I, Mattila K. et al. Bioactive glass S53P4 and autograft bone in treatment of depressed tibial plateau fractures - a prospective randomized 11-year follow-up. J Long Term Eff Med Implants. 2011;21(2):139–148. doi: 10.1615/jlongtermeffmedimplants.v21.i2.40. [DOI] [PubMed] [Google Scholar]
- 16.Sarin J, Grénman R, Aitasalo K, Pulkkinen J. Bioactive glass S53P4 in mastoid obliteration surgery for chronic otitis media and cerebrospinal fluid leakage. Ann Otol Rhinol Laryngol. 2012;121(9):563–569. doi: 10.1177/000348941212100901. [DOI] [PubMed] [Google Scholar]
- 17.Abramovich S, Hannan S A, Huins C T. et al. Prospective cohort comparison of bioactive glass implants and conchal cartilage in reconstruction of the posterior canal wall during tympanomastoidectomy. Clin Otolaryngol. 2008;33(6):553–559. doi: 10.1111/j.1749-4486.2008.01813.x. [DOI] [PubMed] [Google Scholar]
- 18.Reck R, Störkel S, Meyer A. Bioactive glass-ceramics in middle ear surgery. An 8-year review. Ann N Y Acad Sci. 1988;523:100–106. doi: 10.1111/j.1749-6632.1988.tb38504.x. [DOI] [PubMed] [Google Scholar]
- 19.Stoor P, Grénman R. Bioactive glass and turbinate flaps in the repair of nasal septal perforations. Ann Otol Rhinol Laryngol. 2004;113(8):655–661. doi: 10.1177/000348940411300811. [DOI] [PubMed] [Google Scholar]
- 20.Stoor P, Pulkkinen J, Grénman R. Bioactive glass S53P4 in the filling of cavities in the mastoid cell area in surgery for chronic otitis media. Ann Otol Rhinol Laryngol. 2010;119(6):377–382. doi: 10.1177/000348941011900603. [DOI] [PubMed] [Google Scholar]
- 21.Sándor G K, Numminen J, Wolff J. et al. Adipose stem cells used to reconstruct 13 cases with cranio-maxillofacial hard-tissue defects. Stem Cells Transl Med. 2014;3(4):530–540. doi: 10.5966/sctm.2013-0173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Profeta A C. Dentine bonding agents comprising calcium-silicates to support proactive dental care: origins, development and future. Dent Mater J. 2014;33(4):443–452. doi: 10.4012/dmj.2013-267. [DOI] [PubMed] [Google Scholar]
- 23.Aitasalo K M, Piitulainen J M, Rekola J, Vallittu P K. Craniofacial bone reconstruction with bioactive fiber-reinforced composite implant. Head Neck. 2014;36(5):722–728. doi: 10.1002/hed.23370. [DOI] [PubMed] [Google Scholar]
- 24.Neovius E, Engstrand T. Craniofacial reconstruction with bone and biomaterials: review over the last 11 years. J Plast Reconstr Aesthet Surg. 2010;63(10):1615–1623. doi: 10.1016/j.bjps.2009.06.003. [DOI] [PubMed] [Google Scholar]
- 25.Winder J, Cooke R S, Gray J, Fannin T, Fegan T. Medical rapid prototyping and 3D CT in the manufacture of custom made cranial titanium plates. J Med Eng Technol. 1999;23(1):26–28. doi: 10.1080/030919099294401. [DOI] [PubMed] [Google Scholar]
- 26.Ducic Y. Titanium mesh and hydroxyapatite cement cranioplasty: a report of 20 cases. J Oral Maxillofac Surg. 2002;60(3):272–276. doi: 10.1053/joms.2002.30575. [DOI] [PubMed] [Google Scholar]
- 27.Stelnicki E J, Ousterhout D K. Prevention of thermal tissue injury induced by the application of polymethylmethacrylate to the calvarium. J Craniofac Surg. 1996;7(3):192–195. doi: 10.1097/00001665-199605000-00006. [DOI] [PubMed] [Google Scholar]
- 28.Gosain A K; Plastic Surgery Educational Foundation DATA Committee. Bioactive glass for bone replacement in craniomaxillofacial reconstruction Plast Reconstr Surg 20041142590–593. [DOI] [PubMed] [Google Scholar]
- 29.Gosain A K; Plastic Surgery Educational Foundation DATA Committee. Biomaterials for reconstruction of the cranial vault Plast Reconstr Surg 20051162663–666. [DOI] [PubMed] [Google Scholar]
- 30.Peltola M J, Vallittu P K, Vuorinen V, Aho A A, Puntala A, Aitasalo K M. Novel composite implant in craniofacial bone reconstruction. Eur Arch Otorhinolaryngol. 2012;269(2):623–628. doi: 10.1007/s00405-011-1607-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Peltola M, Aitasalo K, Suonpää J, Varpula M, Yli-Urpo A. Bioactive glass S53P4 in frontal sinus obliteration: a long-term clinical experience. Head Neck. 2006;28(9):834–841. doi: 10.1002/hed.20436. [DOI] [PubMed] [Google Scholar]
- 32.Stoor P, Söderling E, Grénman R. Bioactive glass S53P4 in repair of septal perforations and its interactions with the respiratory infection-associated microorganisms Haemophilus influenzae and Streptococcus pneumoniae. J Biomed Mater Res. 2001;58(1):113–120. doi: 10.1002/1097-4636(2001)58:1<113::aid-jbm170>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 33.Turunen T, Peltola J, Yli-Urpo A, Happonen R P. Bioactive glass granules as a bone adjunctive material in maxillary sinus floor augmentation. Clin Oral Implants Res. 2004;15(2):135–141. doi: 10.1111/j.1600-0501.2004.00989.x. [DOI] [PubMed] [Google Scholar]
- 34.Aitasalo K M Peltola M J Bioactive glass hydroxyapatite in fronto-orbital defect reconstruction Plast Reconstr Surg 200712071963–1972., discussion 1973–1974 [DOI] [PubMed] [Google Scholar]
- 35.Peltola M J, Aitasalo K M, Suonpää J T, Yli-Urpo A, Laippala P J, Forsback A P. Frontal sinus and skull bone defect obliteration with three synthetic bioactive materials. A comparative study. J Biomed Mater Res B Appl Biomater. 2003;66(1):364–372. doi: 10.1002/jbm.b.10023. [DOI] [PubMed] [Google Scholar]
- 36.Moreira-Gonzalez A, Jackson I T, Miyawaki T, Barakat K, DiNick V. Clinical outcome in cranioplasty: critical review in long-term follow-up. J Craniofac Surg. 2003;14(2):144–153. doi: 10.1097/00001665-200303000-00003. [DOI] [PubMed] [Google Scholar]
- 37.Verret D J, Ducic Y, Oxford L, Smith J. Hydroxyapatite cement in craniofacial reconstruction. Otolaryngol Head Neck Surg. 2005;133(6):897–899. doi: 10.1016/j.otohns.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 38.Shapoff C A Alexander D C Clark A E Clinical use of a bioactive glass particulate in the treatment of human osseous defects Compend Contin Educ Dent 1997184352–354., 356, 358 passim [PubMed] [Google Scholar]
- 39.Dusková M, Smahel Z, Vohradník M. et al. Bioactive glass-ceramics in facial skeleton contouring. Aesthetic Plast Surg. 2002;26(4):274–283. doi: 10.1007/s00266-002-1032-z. [DOI] [PubMed] [Google Scholar]
- 40.Peltola M, Suonpää J, Aitasalo K, Varpula M, Yli-Urpo A, Happonen R P. Obliteration of the frontal sinus cavity with bioactive glass. Head Neck. 1998;20(4):315–319. doi: 10.1002/(sici)1097-0347(199807)20:4<315::aid-hed6>3.0.co;2-1. [DOI] [PubMed] [Google Scholar]
- 41.Peltola M, Suonpää J, Aitasalo K. et al. Experimental follow-up model for clinical frontal sinus obliteration with bioactive glass (S53P4) Acta Otolaryngol Suppl. 2000;543:167–169. doi: 10.1080/000164800454305. [DOI] [PubMed] [Google Scholar]
- 42.Kinnunen I, Aitasalo K, Pöllönen M, Varpula M. Reconstruction of orbital floor fractures using bioactive glass. J Craniomaxillofac Surg. 2000;28(4):229–234. doi: 10.1054/jcms.2000.0140. [DOI] [PubMed] [Google Scholar]
- 43.Aitasalo K Kinnunen I Palmgren J Varpula M Repair of orbital floor fractures with bioactive glass implants J Oral Maxillofac Surg 200159121390–1395., discussion 1395–1396 [DOI] [PubMed] [Google Scholar]
- 44.Thompson I D. Singapore: World Scientific; 2011. Clinical applications of bioactive glasses for maxillofacial repair; pp. 77–96. [Google Scholar]
- 45.Peltola M, Kinnunen I, Aitasalo K. Reconstruction of orbital wall defects with bioactive glass plates. J Oral Maxillofac Surg. 2008;66(4):639–646. doi: 10.1016/j.joms.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 46.Peltola M J, Aitasalo K M, Aho A J, Tirri T, Suonpää J T. Long-term microscopic and tissue analytical findings for 2 frontal sinus obliteration materials. J Oral Maxillofac Surg. 2008;66(8):1699–1707. doi: 10.1016/j.joms.2007.11.020. [DOI] [PubMed] [Google Scholar]
- 47.Della Santina C C, Lee S C. Ceravital reconstruction of canal wall down mastoidectomy: long-term results. Arch Otolaryngol Head Neck Surg. 2006;132(6):617–623. doi: 10.1001/archotol.132.6.617. [DOI] [PubMed] [Google Scholar]
- 48.Stoor P, Söderling E, Salonen J I. Antibacterial effects of a bioactive glass paste on oral microorganisms. Acta Odontol Scand. 1998;56(3):161–165. doi: 10.1080/000163598422901. [DOI] [PubMed] [Google Scholar]
- 49.Stavropoulos A, Sima C, Sima A, Nyengaard J, Karring T, Sculean A. Histological evaluation of healing after transalveolar maxillary sinus augmentation with bioglass and autogenous bone. Clin Oral Implants Res. 2012;23(1):125–131. doi: 10.1111/j.1600-0501.2011.02161.x. [DOI] [PubMed] [Google Scholar]
- 50.Rickert D, Slater J J, Meijer H J, Vissink A, Raghoebar G M. Maxillary sinus lift with solely autogenous bone compared to a combination of autogenous bone and growth factors or (solely) bone substitutes. A systematic review. Int J Oral Maxillofac Surg. 2012;41(2):160–167. doi: 10.1016/j.ijom.2011.10.001. [DOI] [PubMed] [Google Scholar]
- 51.Tadjoedin E S, de Lange G L, Lyaruu D M, Kuiper L, Burger E H. High concentrations of bioactive glass material (BioGran) vs. autogenous bone for sinus floor elevation. Clin Oral Implants Res. 2002;13(4):428–436. doi: 10.1034/j.1600-0501.2002.130412.x. [DOI] [PubMed] [Google Scholar]
- 52.Cordioli G, Mazzocco C, Schepers E, Brugnolo E, Majzoub Z. Maxillary sinus floor augmentation using bioactive glass granules and autogenous bone with simultaneous implant placement. Clinical and histological findings. Clin Oral Implants Res. 2001;12(3):270–278. doi: 10.1034/j.1600-0501.2001.012003270.x. [DOI] [PubMed] [Google Scholar]
- 53.Tadjoedin E S, de Lange G L, Holzmann P J, Kulper L, Burger E H. Histological observations on biopsies harvested following sinus floor elevation using a bioactive glass material of narrow size range. Clin Oral Implants Res. 2000;11(4):334–344. doi: 10.1034/j.1600-0501.2000.011004334.x. [DOI] [PubMed] [Google Scholar]
- 54.Liu X, Rahaman M N, Fu Q. Bone regeneration in strong porous bioactive glass (13-93) scaffolds with an oriented microstructure implanted in rat calvarial defects. Acta Biomater. 2013;9(1):4889–4898. doi: 10.1016/j.actbio.2012.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Jones J R. Review of bioactive glass: from Hench to hybrids. Acta Biomater. 2013;9(1):4457–4486. doi: 10.1016/j.actbio.2012.08.023. [DOI] [PubMed] [Google Scholar]


