Abstract
Background
Opioids have long been regarded as the most effective drugs for treatment of severe acute and chronic pain. Unfortunately, their therapeutic efficacy and clinical utility has been limited because of central and peripheral side effects.
Methods
To determine the therapeutic value of peripheral mu-opioid receptors as a target for neuropathic pain treatment, we examined the effects of DALDA, a hydrophilic, peripherally acting mu-opioid receptor agonist, in male and female rats with spinal nerve ligation-induced neuropathic pain. We also utilized behavioral, pharmacologic, electrophysiologic, and molecular biologic tools to characterize DALDA's possible mechanisms of action in male rats.
Results
DALDA, administered subcutaneously, had 70 times greater efficacy for inhibiting thermal (n=8–11/group) than mechanical hypersensitivity (n=6–8/group) in male rats. The pain inhibitory effects of DALDA on mechanical and heat hypersensitivity were abolished in animals pretreated with systemic methylnaltrexone (n=7–9/group), a peripheral mu-opioid receptor antagonist. In spinal wide-dynamic range neurons, systemic DALDA inhibited C-fiber–mediated, but not A-fiber–mediated, response in neuropathic male rats (n=13). In primary sensory neurons, DALDA inhibited the capsaicin-induced [Ca2+] increase more than the β-alanine–induced [Ca2+] increase (n=300); capsaicin and β-alanine activate subpopulations of neurons involved in the signaling of heat and mechanical pain, respectively. DALDA-treated rats (n=5–8/group) did not exhibit motor deficits and locomotor impairment suggesting that it does not induce central side effects.
Conclusion
These findings suggest that DALDA may represent a potential alternative to current opioid therapy for the treatment of neuropathic pain and is likely to be associated with minimal adverse effects.
Keywords: DALDA, mu-opioid receptor, peripheral nervous system, neuropathic pain, wide-dynamic range neuron, dorsal root ganglion
Introduction
Chronic neuropathic pain is prevalent among 6–8% of the population. In patients attending pain clinics, the incidence is as high as 25–51.9%.1 Moreover, chronic pain causes considerable social and economic burden and leads to high healthcare costs and lost productivity.2,3 Patients with neuropathic pain resulting from nerve injuries present with varying degrees of mechanical and heat hyperalgesia. These manifestations are also observed in animal models of neuropathic pain.4,5 Mechanical and heat hypersensitivities involve different peripheral and central mechanisms and hence may require different treatment strategies.4,6,7 In the past several years, a number of drugs have been developed for the treatment of neuropathic pain, but no single agent is uniformly effective, and opioids remain some of the most commonly used drugs. However, their therapeutic utility is limited considerably by their severe central and peripheral side effects, including sedation, dizziness, respiratory depression, constipation, nausea, vomiting, tolerance, and physical dependence.8 These adverse effects may lead to opioid discontinuation and contribute to under-dosing and inadequate analgesia in patients with neuropathic pain.9 They are a primary reason that opioids have been downgraded from a second-line to a third-line class of drugs in the recent recommendations for the pharmacological treatment of neuropathic pain.10 Hence, alternative therapies that lack central and peripheral adverse effects are important for the treatment of neuropathic pain.
Activating opioid receptors in the peripheral nervous system may offer an opportunity for treating certain chronic pain conditions while avoiding their central side effects.11,12 However, whether peripheral opioids affect mechanical and heat hypersensitivity differently is unclear. Additionally, little is known about their cellular mechanisms, such as which subpopulations of primary sensory neurons are targeted by peripherally acting opioids to inhibit different neuropathic pain modalities. Dermorphin is a natural heptapeptide mu-opioid receptor (MOR) agonist found in amphibian skin.13,14 Degradation of dermorphin by peptidases produces the N-terminal tetrapeptide H-Tyr-DAla-Phe-Gly-OH.14,15 Additional amino acid substitutions to this tetrapeptide led to the development of dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA), a highly selective MOR agonist.16–18 Intriguingly, DALDA is highly hydrophilic because it carries three net positive charges at physiologic pH. Moreover, it is metabolically more stable than its other analogs and exhibits a restrictive penetration into the CNS after systemic drug administration.17 These properties make DALDA a promising drug candidate for the treatment of chronic neuropathic pain with reduced risk of central side effects.
Critical to the use of peripherally acting opioids is an understanding of the analgesic properties and mechanisms that underlie the therapeutic effects of DALDA. Therefore, the purpose of this research was to improve our understanding of the cellular mechanisms involved in peripheral opioid analgesia and the development of peripheral opioids with minimal central side effects. In this study, we characterized the efficacy of systemic DALDA to attenuate mechanical and heat hypersensitivity in nerve-injured rats, investigated its site and mechanism of action, and assessed its safety profile.
Materials and Methods
Animals
Male and female Sprague-Dawley rats (200–350 g; Harlan Bioproducts for Science, Indianapolis, IN) were housed under optimal laboratory conditions with a 12-hour light/dark cycle and free access to food and water. Animals were acclimatized to laboratory conditions before the tests. All behavioral experiments were carried out between 9:00 a.m. and 5:00 p.m. by an investigator blinded to the drug assignment. The experimental protocols were approved by the Animal Care and Use Committee of Johns Hopkins University and complied with the National Institutes of Health Guide for the Use of Experimental Animals to ensure minimal animal use and discomfort.
Drugs
DALDA was purchased from US Biologicals (Salem, MA), and methylnaltrexone bromide (MNTX) and D-Phe-Cys-Tyr-D-Trp-Orn-Thr-Pen-Thr-NH2 (CTOP) were purchased from Sigma-Aldrich (St. Louis, MO). Other drugs were purchased from Sigma-Aldrich or Tocris Bioscience (Bristol, UK). Stock solutions were freshly prepared as instructed by the manufacturer.
Neuropathic pain model
L5 spinal nerve ligation (SNL) surgery was used for the induction of neuropathic pain in rats. The procedure was a modification of that described in our previous studies.19,20
Intrathecal catheter implantation
After rats were anesthetized (2% isoflurane), a small slit was cut in the atlanto-occipital membrane, and a 6–7-cm piece of saline-filled PE-10 tubing was inserted. We confirmed intrathecal drug delivery by injecting lidocaine (400 µg/20 µl, Hospira, Lake Forest, IL), which resulted in a temporary motor paralysis of the lower limbs.19,20
Animal behavioral tests
Animals were allowed to recover from surgery for 2 weeks before any behavior testing was done. Animals were acclimatized and habituated to the test environment. All procedures have been described in our previous studies.19–21
von Frey hair test
To assess mechanical hypersensitivity to punctuate mechanical stimuli, we measured paw withdrawal threshold (PWT) to von Frey filaments. Each filament (0.38–13.1 g) was applied to the test area on the plantar surface of the hindpaw for 4–6 seconds according to the up-down method.21–23
Hargreaves test
To test for signs of heat hypersensitivity, we used the Hargreaves test, which measures paw-withdrawal latency (PWL) to radiant heat stimuli. Radiant heat was applied to the plantar surface of each hindpaw three times (3–5-minute interval) with a plantar stimulator analgesia meter (IITC model 390, Woodland Hills, CA). A cut off time of 20 seconds was used to prevent tissue damage.
Rota-rod test
We used the rota-rod test to assess the well-known motor impairment side effects of opioids. Rats were acclimatized and trained on a rotating rod (Ugo Basile, Italy) that accelerated from 0 to 30 rpm in 180 seconds. On the day of testing, rat performance on the rod was measured before (pre-drug baseline) and 45 minutes after administration of DALDA or morphine. The time (in seconds) that each animal remained on the accelerating rod without falling was recorded.19,24
Open field test
The open field test was used to assess the effect of systemic DALDA administration on spontaneous exploration and locomotor activity of rats. Rats were placed in an open field chamber (73 × 45 cm rectangular plastic box with a wall height of 33 cm) for 10 minutes. Their behavior was video recorded, and parameters such as total distance travelled; mean travel speed; and number of border periphery, internal periphery, and center crossings were analyzed by SMART 3 software (Panlab Harvard Apparatus).
Spinal dorsal horn recordings
In anesthetized rats, we performed tracheotomy, mechanical ventilation, and extracellular recordings of single dorsal horn neuronal activity as described in our previous studies.20,21 Briefly, a laminectomy was performed at vertebral levels T12-L1 corresponding to lumbar enlargements at spinal segments L3-S1. During neurophysiologic recording, animals were paralyzed with intraperitoneal (i.p.) pancuronium bromide (0.15 mg/kg, Elkins-Sinn Inc., Cherry Hill, NJ) to facilitate controlled ventilation. Only wide-dynamic range (WDR) neurons with defined receptive fields (RFs) in the plantar region of the hind paw were studied. The cutaneous RFs of WDR neurons were mapped, and a single site (most sensitive site) near the center of the RF was chosen for application of test stimuli. Analog data were collected with a real-time, computer-based data acquisition and processing system (CED Spike 2, Cambridge, UK). WDR cells were identified by their characteristic responses.20,21 The evoked responses of WDR neurons to a series of mechanical (brushing, graded von Frey monofilaments: 1–15 g, Stoelting Co., Wood Dale, IL) and electrical test stimuli (0.1–10 mA, 2 milliseconds) were examined. The WDR neuronal response to a supra-threshold electrical stimulus consists of an early A-component (0–100 milliseconds) and a later C-component (100–500 milliseconds).
Dorsal root ganglion (DRG) neuronal culture and calcium imaging
Experiments were conducted as we have described previously.19,25,26 Briefly, DRGs from rats were collected in cold DH10 medium and treated with enzyme solution at 37°C. Neurons were loaded with Fura 2-acetomethoxyl ester (Molecular Probes, Eugene, OR) for 45 minutes in the dark at room temperature.25,26 After being washed, cells were imaged at 340 and 380 nm excitation for detection of intracellular free calcium. Calcium imaging assays were performed by an experimenter blind to drug treatment. For isolectin IB4 labeling studies, dissociated DRG neurons were cultured in an incubator at 37°C. After 24–48 hours, neuron cultures were treated with fluorescein-labeled Griffoniasimplicifolia lectin I-isolectin B4 (1:500; Vector Laboratories) for 10 minutes at room temperature and rinsed for 3 minutes. Then these cultures were loaded with Fura 2-acetomethoxyl ester. After calcium imaging assays, we selected small-diameter DRG neurons (<25 µm) to analyze the changes in calcium concentration ([Ca2+]). Cells were divided into two groups: IB4− and IB4+ neurons.
Statistical Analysis
To establish the dose-response functions, we normalized PWT and PWL data by calculating maximum possible effect (MPE) values, as PWTs are often at the cut off values in naïve animals. MPE values for inhibiting mechanical hypersensitivity were calculated with the equation: MPE (%) = [1 − (Cutoff PWT − Post-drug PWT)/(Cutoff PWT − Pre-drug PWT)] × 100, where cutoff PWT = 21.5 g. Because PWLs in naïve animals do not reach the cutoff value (20 seconds), MPE values for inhibiting heat hypersensitivity were calculated with the equation: MPE (%) = [1 − (Pre-injury PWL − Post-drug PWL)/(Pre-injury PWL − Pre-drug PWL)] × 100. Thus, the MPE value for inhibiting heat hyperalgesia may exceed 100%. In electrophysiology studies, we compared the number of action potentials evoked by test stimuli between pre-drug and post-drug conditions. For analysis of windup, we plotted the C-component of WDR neurons evoked by each stimulus against the stimulation number in a train of 16 stimuli. We compared the A- and C-component produced by graded electrical stimuli and total C-component in response to windup stimulation between pre- and post-drug conditions.
There were no data missing for any of the variables. The methods for statistical comparisons in each study are given in the figure legends. The number of animals used in each study was based on our experience with similar studies. We randomized animals to the different treatment groups and blinded the experimenter to drug treatment to reduce selection and observation bias. STATISTICA 6.0 software (StatSoft, Inc., Tulsa, OK) was used to conduct all statistical analyses. The Tukey honestly significant difference post-hoc test was used to compare specific data points. Bonferroni correction was applied for multiple comparisons. Two-tailed tests were performed; p<0.05 was considered significant in all tests.
Results
DALDA-induced attenuation of mechanical hypersensitivity involves activation of peripheral mu-opioid receptors
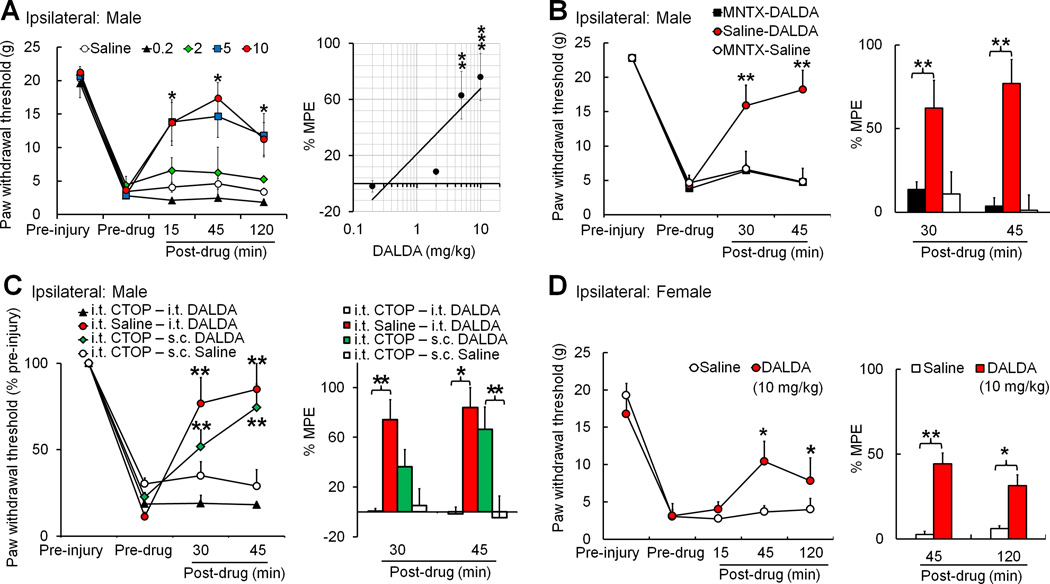
To investigate the therapeutic utility of DALDA on neuropathic pain, we first examined the effects of systemic administration of DALDA on SNL-induced mechanical hypersensitivity. Compared to pre-injury baseline, paw withdrawal threshold (PWT) of nerve-injured (ipsilateral) hind paw to mechanical stimuli was significantly decreased at 2–3 weeks post-SNL. Subcutaneous administration of DALDA in male SNL rats dose-dependently increased the ipsilateral PWTs at 15, 45, and 120 minutes (0.2–10 mg/kg, n=6–8/dose), compared to the pre-drug baseline (Fig. 1A). The magnitude and duration of DALDA-induced anti-allodynic effects increased with dose. We calculated the peak MPE at 45 minutes post-drug to establish the dose-response function and calculated the ED50 (dose estimated to produce 50% MPE) as 4.2 mg/kg. MPEs of 5 and 10 mg/kg doses were significantly higher than the MPE of vehicle (saline, n=11, Fig. 1A). Contralateral PWT did not change significantly after nerve injury or drug treatment (data not shown). Next, to identify the site of action of DALDA, we utilized peripheral and central MOR antagonists. We pretreated nerve-injured male rats with systemic methylnaltrexone (5 mg/kg i.p.), a peripherally acting MOR-preferring antagonist, 10 minutes before DALDA treatment (10 mg/kg, s.c., n=7). Methylnaltrexone, but not saline, completely blocked the anti-allodynic effects of systemic DALDA on mechanical hypersensitivity in male SNL rats (Fig. 1B, n=7). In contrast, pretreatment with intrathecal CTOP (0.5 µg/10 µl, n=7), a highly selective MOR antagonist, did not prevent the anti-allodynic effects of systemic DALDA (Fig. 1C). The same CTOP pretreatment blocked spinal opioid analgesia induced by intrathecal injection of DALDA (0.5 µg/10 µl, n=5), suggesting that CTOP effectively blocks MOR activation in the spinal cord. DALDA (10 mg/kg, s.c.) also significantly increased the ipsilateral PWTs from pre-drug baseline in female rats at 2–3 weeks post-SNL (n=6, Fig. 1D). There was a trend that peak MPE at 45 min after DALDA treatment in female rats (44.2 ± 13.3%, 10 mg/kg, s.c.) is lower than that in male rats (75.9 ± 13.8%, n=7), but the difference did not reach statistical significance (p=0.11, Student’s t-test).
Figure 1. Systemic administration of dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) inhibits mechanical hypersensitivity in nerve-injured male rats through activation of peripheral opioid receptors.
(A) Left: Subcutaneous (s.c.) injection of DALDA (0.2, 2, 5, 10 mg/kg, n=6–8/dose) dose-dependently inhibited mechanical allodynia in male rats at 2–3 weeks after spinal nerve ligation (SNL), as indicated by the significant increase in ipsilateral paw withdrawal threshold (PWT). The time course shows peak drug effect at 45 minutes after injection. *p<0.05 versus pre-drug (5, 10mg/kg), two-way mixed model ANOVA. Right: The maximum possible effect (MPE) of DALDA at 45 minutes post-drug was calculated. MPE (%) = [1 − (Pre-injury PWT − Post-drug PWT)]/[Pre-injury PWT − Pre-drug PWT] × 100. **p<0.01, ***p<0.001 versus saline group (n=11), one-way ANOVA. (B) Left: Pretreatment with an intraperitoneal (i.p.) injection of methylnaltrexone (MNTX, 5 mg/kg, 10-minute pretreatment, n=7), but not saline (n=7), blocked the inhibitory effect of DALDA (10 mg/kg, s.c.) on mechanical hypersensitivity in male SNL rats. Injection of methylnaltrexone (5 mg/kg, i.p., n=7) followed by saline (s.c.) did not change PWT from pre-drug baseline. Right: The MPEs at 30 minutes and 45 minutes after the second drug administration were calculated for each group. (C) Left: Intrathecal (i.th.) pretreatment with the highly selective MOR antagonist D-Phe-Cys-Tyr-D-Trp-Orn-Thr-Pen-Thr-NH2 (CTOP, 5 µg/10 µl, n=5), but not saline (n=5), blocked inhibition of mechanical hypersensitivity by intrathecal DALDA (0.5 µg/10 µl) in male SNL rats. However, the same CTOP treatment did not block inhibition of mechanical hypersensitivity by systemic DALDA (10 mg/kg, s.c., n=7). Intrathecal CTOP (5 µg/10 µl, n=5) followed by saline injection (s.c.) did not change PWT from pre-drug baseline. (D) Left: DALDA (10 mg/kg, n=6, s.c.), but not saline (n=6), also inhibited mechanical allodynia in female rats at 2–3 weeks after SNL. Right: The MPE at 45 and 120 minutes post-drug was calculated. (B–D) Left: *p<0.05, **p<0.01 versus pre-drug, two-way mixed model ANOVA. Right: *p<0.05, **p<0.01 versus saline, one-way ANOVA. Data are expressed as mean ± SEM.
Less DALDA is required to attenuate thermal hyperalgesia than to inhibit mechanical allodynia
To further examine the effect of systemic DALDA on heat hyperalgesia in nerve-injured rats, we measured PWL with the Hargreaves test before and after drug treatment. At 2–3 weeks post-SNL, PWL was significantly decreased in the ipsilateral hind paw (Fig. 2A), but not in the contralateral hind paw (Fig. 2B). Systemic DALDA dose-dependently attenuated this SNL-induced thermal hyperalgesia in male rats (0.02, 0.1, 0.2, 2 mg/kg, s.c., n=8–11/group). We used the peak MPEs for systemic DALDA to reverse heat hyperalgesia at 45 minutes post-drug to establish the dose-response function and calculate ED50 (Fig. 2C). The ED50 (0.06 mg/kg) was significantly lower than that required to inhibit mechanical allodynia (4.2 mg/kg) in male SNL rats. MPEs for 0.1, 0.2, and 2 mg/kg doses were all significantly higher than MPE for the vehicle-treated group (saline, n=11, Fig. 2C). An intraperitoneal injection of methylnaltrexone (5 mg/kg), but not saline (n=9/group), 10 minutes before DALDA injection completely blocked the anti-hyperalgesic effect of high-dose DALDA (2 mg/kg, s.c., Fig. 2D). Drug treatment did not significantly alter PWL of the contralateral hindpaw (Fig. 2E). These findings suggest that peripheral MOR activation mediates systemic DALDA-induced inhibition of both mechanical and heat hyperalgesia in nerve-injured male rats. However, inhibition of mechanical hypersensitivity requires a 70-fold greater dose of DALDA than inhibition of heat hyperalgesia. DALDA (10 mg/kg, s.c.) also increased the ipsilateral PWLs from pre-drug baseline in female rats (n=6, Fig. 2F), but did not significantly alter PWL of the contralateral hindpaw (Fig. 2G).
Figure 2. Systemic administration of dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) inhibits heat hypersensitivity in nerve-injured male rats at lower doses than those required to inhibit mechanical hypersensitivity.
(A) At 2–3 weeks after spinal nerve ligation (SNL) in male rats, subcutaneous (s.c.) injection of DALDA (0.02, 0.1, 0.2, 2 mg/kg, n=8–11/group) dose-dependently increased the ipsilateral paw withdrawal latency (PWL) as compared to that at pre-drug baseline. *p<0.05 versus pre-drug (0.1. 0.2, 2 mg/kg), two-way mixed model ANOVA. (B) The PWL in the contralateral hindpaw did not change after nerve injury or drug treatment. (C) The maximum possible effect (MPE) of DALDA at 45 minutes post-drug was calculated. MPE (%) = [1 − (Pre-injury PWL − Post-drug PWL)/(Pre-injury PWL − Pre-drug PWL)] × 100. **p<0.01, ***p<0.001 versus saline group (n=11), one-way ANOVA. (D) Pretreatment with an intraperitoneal (i.p.) injection of methylnaltrexone (MNTX, 5 mg/kg, n=9), but not saline (n=9), 10 minutes before DALDA (2 mg/kg, s.c.) blocked inhibition of heat hyperalgesia by DALDA in male rats. Methylnaltrexone (5 mg/kg) followed by saline (s.c.) did not change PWL from pre-drug baseline (n=9). *p<0.05 versus pre-drug, #p<0.05 versus MNTX+DALDA group, two-way mixed model ANOVA. (E) The PWL of the contralateral hindpaw did not change after nerve injury or drug treatment. (F–G) DALDA (10 mg/kg, n=6, s.c.), but not saline (n=6), also inhibited heat hypersensitivity in female rats at 2–3 weeks after SNL. *p<0.05 versus pre-drug, #p<0.05 versus saline, two-way mixed model ANOVA. Data are expressed as mean ± SEM.
Systemic DALDA inhibits the C-component of spinal WDR neurons more potently than the A-component
To further confirm the site of action and delineate the cellular mechanisms involved in DALDA's modality-preferred inhibition of neuropathic pain, we examined the changes in spinal WDR neuronal response before and after systemic DALDA treatment in male SNL rats. WDR neurons receive converging afferent inputs in both low-threshold A-fibers and high-threshold C-fibers (presumably nociceptive). Yet, the A- and C-fiber–mediated responses to natural stimulation are not readily differentiated in WDR neurons. In contrast, WDR neuronal response to a supra-threshold electrical stimulus (2 milliseconds) can be separated as a short latency A-fiber component (0–100 milliseconds) and a longer latency C-fiber component (100–500 milliseconds, Fig. 3A), based on calculated conduction velocities. This unique feature of WDR neuronal response allows us to differentiate the drug effects on A- and C-fiber–mediated activities in the same neuron. We then characterized the effects of DALDA on responses of WDR cells to graded intracutaneous electrical stimuli (0.1–10 mA, 2 milliseconds) and to windup-inducing stimuli (16 pulses, 0.5 Hz, supra-C-fiber activation threshold, 2 milliseconds) in nerve injured rats. An electrical search stimulus was applied through a pair of fine needles inserted subcutaneously at the central area of the hind paw. Systemic DALDA (10 mg/kg, i.p.) significantly decreased the C-component, but not the A-component, of WDR neurons to graded intracutaneous electrical stimuli (Fig. 3B–D, n=13). The stimulus-response functions of C-component (Fig. 3D), but not A-component (Fig. 3C), were significantly inhibited by DALDA in WDR neurons. We separated A-component into Aβ- (0–25 milliseconds) and Aδ-mediated responses (25–100 milliseconds) based on calculated conduction velocities. The total numbers of Aβ- and Aδ-mediated responses to graded electrical stimuli (0.1–10 mA) after DALDA treatment (Aβ: 72.1 ± 5.3; Aδ: 29.9 ± 6.3) were not significant different from pre-drug baseline (Aβ: 79.2 ± 4.9; Aδ: 29.3 ± 6.8, p>0.05, paired t-test). Saline treatment (n=10) did not affect WDR neuron response (Fig. 3B).
Figure 3. Systemic dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) selectively inhibits the C-component of WDR neurons to electrical stimulation, an effect that is blocked by methylnaltrexone.
(A) An analog recording of wide-dynamic range (WDR) neuronal responses to the 1st, 4th, 8th, and 16th stimulus of a train of intracutaneous electrical stimuli (0.5 Hz, 16 pulses, 2.0 milliseconds, supra-C-fiber activation threshold) that induces windup. WDR neuronal responses display A- and C-components to an intracutaneous electrical stimulus. At 2–3 weeks after spinal nerve ligation (SNL) in male rats, windup of C-component was inhibited at 30–45 minutes after systemic administration of DALDA (10 mg/kg, i.p.). (B) The total A-component and C-component to graded intracutaneous electrical stimuli (0.1–10 mA, 2.0 milliseconds) and the total C-component to windup-inducing stimuli after treatment of SNL rats with saline (i.p., n=10), DALDA (10 mg/kg, i.p., n=13), and methylnaltrexone (MNTX, 5 mg/kg, i.p., 15-minute pretreatment, n=9) followed by DALDA (10 mg/kg). **p<0.01 versus saline group, one-way ANOVA. (C) The stimulus-response (S-R) function of the A-component of WDR neuronal response to graded intracutaneous electrical stimuli (0.1–10 mA, 2 milliseconds) before and 30–45 minutes after systemic injection of DALDA (upper panel, 10 mg/kg, n=9, i.p.) or methylnaltrexone (MNTX, 5 mg/kg, i.p., 15-minute pretreatment) with DALDA (lower panel, 10 mg/kg, n=8, i.p.). (D) The S-R function of C-component to graded intracutaneous electrical stimuli in each group. (E) Windup of C-component of WDR neurons to a train of intracutaneous electrical stimuli (0.5 Hz, 16 pulses) before and after drug treatment. The C-component to 0.5 Hz stimulation was plotted against the stimulation sequence number of each trial. C-E: *p<0.05 versus pre-drug, two-way repeated measures ANOVA. Data are expressed as mean ± SEM.
Repetitive electrical stimuli that activate C-fibers may induce temporal summation and transiently enhance the excitability of dorsal horn neurons, a phenomenon called windup (0.5 Hz, Fig. 3A). Windup is most prominent in C-fiber–mediated responses of WDR neurons. Total C-component to windup stimuli was significantly decreased at 30–45 minutes after systemic administration of DALDA (10 mg/kg, i.p., Fig. 3B), compared to that at pre-drug baseline. DALDA also significantly inhibited the windup function (Fig. 3E). Saline treatment (n=10) did not significantly change the A- or C-component of WDR neuronal response to graded electrical stimuli (Fig. 3B) or the windup function (data not shown). Importantly, pretreatment with systemic methylnaltrexone (5 mg/kg, i.p., 15-minute pretreatment, n=9) completely blocked systemic DALDA-induced inhibition of C-component and windup (Figs. 3B,D,E). These in vivo electrophysiologic findings suggest that systemic administration of DALDA inhibits the C-component of WDR neurons predominantly and the A-component to a much lesser extent in SNL rats. Further, the inhibition of C-component by systemic DALDA likely occurs through activation of peripheral opioid receptors.
Mechanical hypersensitivity is a characteristic manifestation of neuropathic pain.27–29 WDR neurons responded to brush stimuli and showed increased firing rates to increasing intensities of punctuate mechanical stimuli (Fig. 4A–C). Although systemic DALDA inhibited mechanical allodynia, which is considered to be mediated by A-fibers, the average responses of WDR neurons to brush stimulation and the stimulus-response functions to graded punctuate mechanical stimuli were not significantly changed after systemic DALDA treatment (10 mg/kg, i.p., n=13), as compared to pre-drug baseline (Fig. 4B). Yet, individual WDR neurons responded differently to DALDA treatment. Of 13 neurons, 6 showed a total response to graded mechanical stimuli that was decreased to <74% of pre-drug level, which is more than two SD less than the mean value after saline treatment (122.2 ± 24.1%, mean ± SD, n=8, Fig. 4D).Yet, two neurons paradoxically showed an increase in response to >170% of pre-drug level. Thus, quantitative mechanical testing showed different patterns of response to systemic DALDA treatment in WDR neurons of SNL rats. Saline treatment did not affect WDR neuronal response to mechanical stimulation (Fig. 4C, n=8).
Figure 4. Effects of systemic dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) on the responses of wide-dynamic range (WDR) neurons to mechanical stimulation in nerve-injured rats.
(A) Left: Peri-stimulus time histograms (bin size: 0.2 seconds) show an example of WDR neuronal response to punctuate mechanical stimuli (1.0–15.0 g von Frey probe, 5 seconds, applied to the skin receptive field) that did not change after subcutaneous (s.c.) injection of DALDA (10 mg/kg, non-responding). Right: An example of another WDR neuron that shows decreased response after DALDA treatment (responding). (B) At 2–3 weeks after spinal nerve ligation (SNL) in male rats, the response to brushing stimuli and the stimulus-response functions of WDR neuronal response to graded mechanical stimuli were not significantly different from pre-drug baseline at 30–45 minutes after DALDA injection (10 mg/kg, s.c., n=13). (C) Injection of saline did not change WDR neuronal response to mechanical stimuli in SNL rats (n=8). (D) The total response of each WDR neuron to graded mechanical stimuli was plotted for each group (as % pre-drug value). Inhibitory cells were defined as neurons that had a post-drug response that was <74% of pre-drug level (below red dashed line), which is more than two SD less than the mean of the saline group (122 ± 24%, mean ± SD, n=8). The facilitatory cells were defined as neurons that had a post-drug response >170% of pre-drug level (above blue dashed line). Black bar: mean response. Data are expressed as mean + SEM. APs, action potentials.
DALDA differentially affects the excitation of TRPV1- and MrgD-expressing DRG neurons
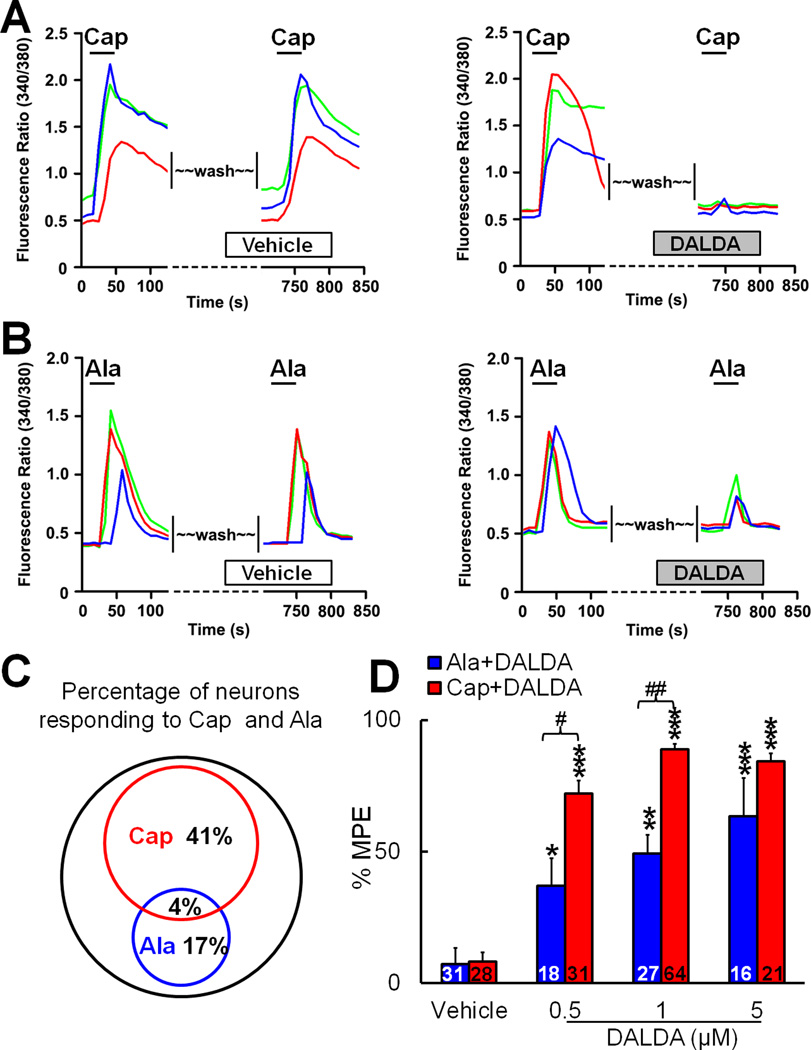
Recent studies have suggested that activation of TRPV1-expressing and MrgD-expressing DRG neurons is critical to heat and mechanical pain signaling, respectively.30–33 Therefore, we examined whether the modality preference we observed in our behavioral tests after systemic DALDA administration is paralleled by differential effects on the excitation of these two subpopulations of primary sensory neurons. We performed calcium-imaging studies to examine the effects of DALDA on the [Ca2+] increase induced by capsaicin, which activates TRPV1+neurons, and β-alanine, which activates MrgD+ neurons (Fig. 5A,B). Bath application of capsaicin (0.5 µM) increased [Ca2+] in 41% of DRG neurons, and β-alanine (1mM) increased [Ca2+] in 17% of DRG neurons (n=300; Fig. 5C). Only 4% of DRG neurons responded to both capsaicin and β-alanine, suggesting very small co-localization of TRPV1 and MrgD in DRG neurons. After a 10-minute washout of the first drug, a second application of capsaicin or β-alanine produced only a slightly smaller increase in [Ca2+], suggesting minimal desensitization. However, pretreatment with DALDA (10 minutes, bath application) dose-dependently blocked the [Ca2+] increase to the second application of capsaicin and β-alanine (Fig. 5D). Importantly, MPEs for 0.5 and 1 µM DALDA to inhibit the capsaicin-induced [Ca2+] increase were significantly higher than those to inhibit the β-alanine-induced increase (Fig. 5D). The IC50 of DALDA to inhibit the capsaicin-induced increase in [Ca2+] was 0.14 µM, whereas that to inhibit β-alanine-induced activation was 1.33 µM (p<0.01, Student's t-test). Thus, DALDA preferentially inhibits the increase in [Ca2+] evoked by capsaicin in DRG neurons.
Figure 5. Dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) inhibits capsaicin- and β-alanine–induced increases in [Ca2+] in dorsal root ganglion (DRG) neurons.
(A) Left: Representative traces from calcium-imaging assays show the increase of [Ca2+] in cultured DRG neurons in response to the first and second bath application of capsaicin (Cap, 0.5 µM). Neurons were washed (10 minutes) with vehicle before the second Cap application. Right: Pretreatment with DALDA (1 µM, 10 minutes, bath application) blocked the increase in [Ca2+] induced by the second application of Cap. (B) Left: Representative traces from calcium-imaging assays show the increase of [Ca2+] in cultured DRG neurons in response to the first and second bath application of β-alanine (Ala, 1 mM). Neurons were washed with vehicle before the second Ala application. Right: Pretreatment with DALDA (1 µM, 10 minutes) partially reduced the increase in [Ca2+] induced by the second application of Ala. (C) The Venn diagram of calcium responses illustrates the proportion of Cap- and Ala-responsive DRG neurons (n=300). The sizes of the circles are proportional to the sizes of the cell populations. (D) Quantification of calcium-imaging assays. DALDA (0.5, 1, 5 µM) dose-dependently inhibited Cap- and Ala-induced increases in [Ca2+]. MPE (%) = [(Pre-DALDA) − (Post-DALDA)]/(Pre-DALDA)] × 100. Numbers of neurons in each group are indicated. *p<0.05, **p<0.01,***p<0.001 versus vehicle group; #p<0.05, ##p<0.01 versus Ala-DALDA group, one-way ANOVA. Data are expressed as mean + SEM.
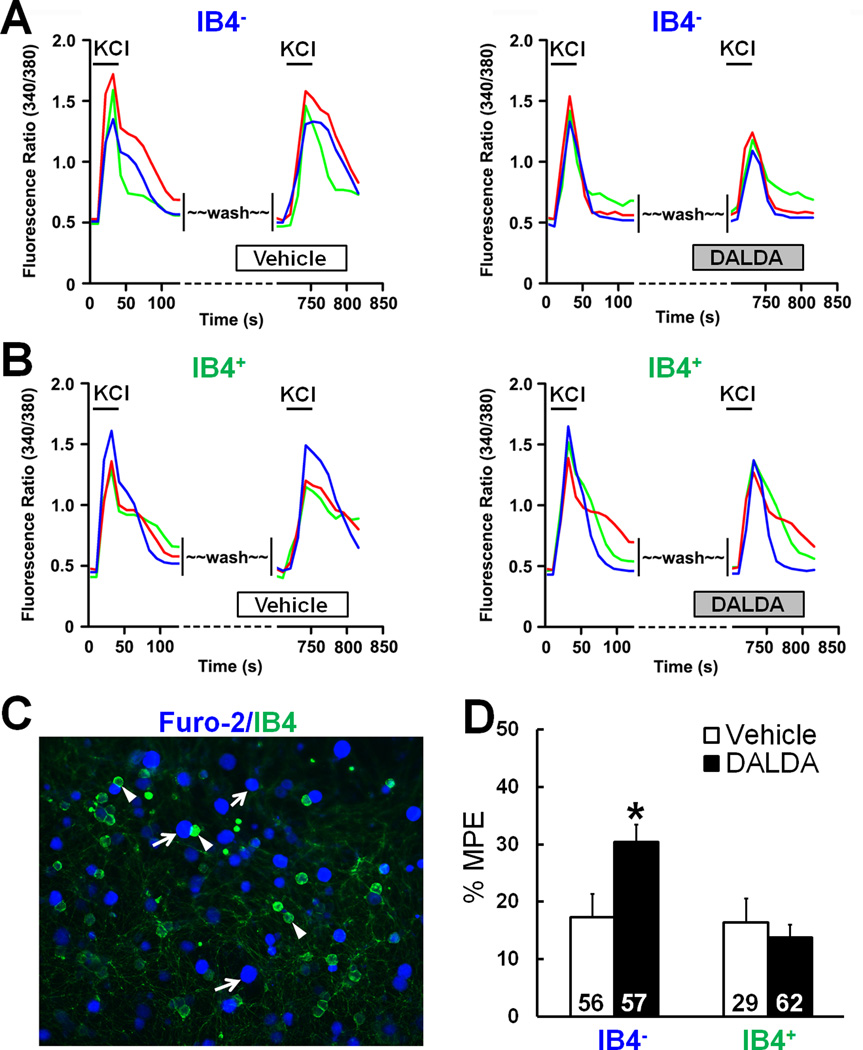
TRPV1 is highly expressed in peptidergic DRG neurons, whereas MrgD is found mostly in the non-peptidergic subpopulation. IB4 is a histochemical marker for non-peptidergic DRG neurons, which can be labeled with IB4-FITC in culture. Bath application of KCl (30 mM), which strongly depolarizes the cell membrane, induced a robust increase in [Ca2+] in both IB4+ and IB4− neurons loaded with Fura 2-acetomethoxy ester dye (Fig. 6A–C). As a control, sequential treatment with KCl caused only a modest reduction in the [Ca2+] increase. Importantly, DALDA (n=57), but not vehicle (n=56), significantly inhibited the KCl-evoked [Ca2+] increase in IB4− neurons (Fig. 6D), which are likely peptidergic cells. However, in IB4+ neurons, DALDA (n=62) was no more effective than vehicle (n=29).
Figure 6. Dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) inhibits potassium chloride (KCl)-induced increases in [Ca2+] in isolectin IB4− dorsal root ganglion (DRG) neurons.
(A) Left: Representative traces from calcium-imaging assays show the increase of [Ca2+] in isolectin IB4− DRG neurons after the first and second bath application of KCl (30 mM). Neurons were washed with vehicle (10 minutes) before the second KCl application. Right: The [Ca2+] increase in response to the second application of KCl was reduced in IB4− neurons by pretreatment with DALDA (1 µM, 10 minutes, bath application). (B) DALDA did not reduce the KCl-evoked increase in [Ca2+] in IB4+ neurons. (C) Fluorescent image of DRG neurons after they were labeled with IB4-FITC and loaded with Fura 2-acetomethoxyl ester. Examples of IB4− (purple) and IB4+ (green) neurons are marked with arrows and arrowheads, respectively. (D) Quantification of calcium-imaging assays. DALDA significantly inhibited the KCl-induced increase in [Ca2+] in IB4− neurons, but not in IB4+ neurons. MPE (%) = [(Pre-DALDA) − (Post-DALDA)]/(Pre-DALDA)] × 100. Data are expressed as mean + SEM. *p<0.05 versus vehicle group, Student's t-test.
Systemic DALDA retains the ability to inhibit mechanical hypersensitivity in nerve-injured rats pretreated with resiniferatoxin (RTX)
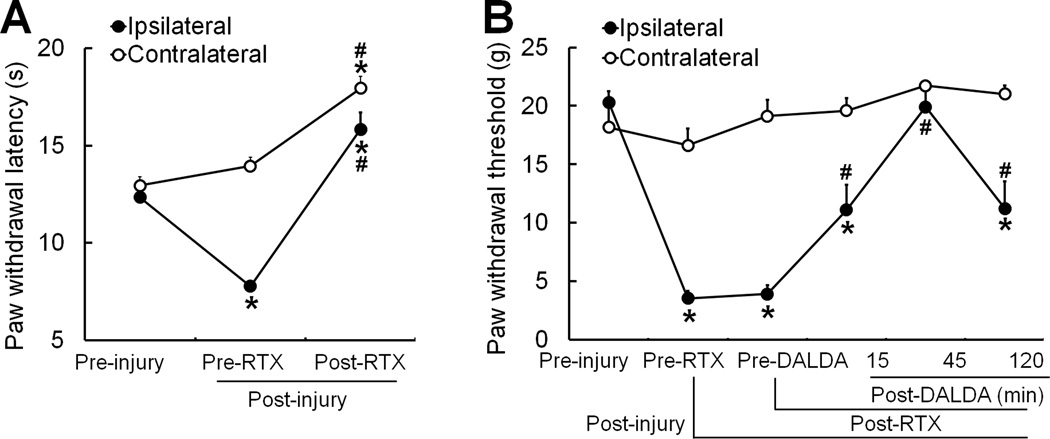
To further delineate the role of TRPV1+ neurons in DALDA-induced amelioration of mechanical and thermal hypersensitivity in nerve-injured rats, we injected adult male rats 7days post-SNL with RTX (0.1 mg/kg, i.p., n=9), a highly potent and selective TRPV1 agonist that desensitizes TRPV1 receptor and decreases the excitability of TRPV1-expressing neurons. At 7–9 days after RTX treatment, heat hypersensitivity was abolished in SNL rats (Fig. 7A). Systemic RTX, but not vehicle (data not shown), further induced heat analgesia, as indicated by increases in PWLs of both injured and uninjured hind paws to levels greater than the pre-injury baseline (Fig. 6A). However, RTX treatment had no effect on nerve injury-induced mechanical hypersensitivity. Importantly, systemic DALDA (10 mg/kg, s.c.) was still able to inhibit mechanical hypersensitivity in RTX-treated SNL rats, as indicated by a significant increase in PWT at 15–120 minutes post-injection (Fig. 7B, n=9). These data clearly indicate that systemic DALDA has different cellular targets for inhibiting nerve injury-induced mechanical and thermal hypersensitivity.
Figure 7. Systemic dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) retains ability to inhibit mechanical hypersensitivity in nerve-injured rats that receive resiniferatoxin (RTX).
(A) The paw withdrawal latency (PWL) of the ipsilateral hindpaw was significantly decreased in male rats on day 7 after spinal nerve ligation (SNL, pre-RTX). On days 7–9 after intraperitoneal (i.p.) injection of RTX (0.1 mg/kg, n=9), both ipsilateral and contralateral PWLs were significantly increased from the pre-RTX and pre-injury level. (B) Rats showed significant reduction in ipsilateral paw withdrawal thresholds (PWTs) on day 7 post-SNL. Systemic RTX treatment (0.1 mg/kg, i.p., n=9) did not alter SNL-induced mechanical allodynia. However, systemic administration of DALDA (10 mg/kg, s.c.) significantly attenuated mechanical allodynia even after RTX treatment. *p<0.05 versus pre-injury, #p<0.05 versus pre-RTX, one-way repeated measures ANOVA. Data are expressed as mean + SEM.
Systemic DALDA does not cause opioid-related side effects
Finally, to determine if systemic DALDA produces well-known opioid-related side effects such as motor incoordination and locomotor impairment, we compared male SNL rats treated with DALDA to those treated with morphine. In the rota-rod test, the fall time of rats treated with systemic DALDA (10, 20 mg/kg, s.c.) was not significantly decreased at 45 minutes after injection, as compared to pre-DALDA baseline and that in saline-treated rats (Fig. 8A,B, n=5–8/group). In contrast, rats treated with morphine (10 mg/kg, s.c., n=8, Fig. 8B) showed a significant reduction in fall time. Additionally, in the open field test, total distance traveled (in 10 minutes, Fig. 8C,D), number of center crossings (Fig. 8E), mean travel speed and number of entries at the border and internal periphery (data not shown) were unaffected at 45 minutes after DALDA treatment (10 mg/kg, s.c.) as compared to those at pre-drug baseline and in the saline-treated group (n=5–8/group). Thus, nerve-injured rats displayed normal activity level, gross locomotion, and exploration habits after systemic DALDA treatment. In contrast, all activities were markedly reduced in morphine-treated rats (10 mg/kg, s.c., n=5/group) as compared to pretreatment baseline and saline- and DALDA-treated groups.
Figure 8. Systemic dermorphin [D-Arg2, Lys4] (1–4) amide (DALDA) does not induce opioid-related side effects and does not affect exploration activity of rats.
(A) In the rota-rod test, neither DALDA (10 mg/kg, s.c., n=5) nor saline (n=8) decreased fall time (i.e., time on the rod) in spinal nerve ligation (SNL) male rats at 45 minutes post-injection, as compared to that at baseline. (B) Morphine (10 mg/kg, s.c., n=8), but not a higher dose of DALDA (20 mg/kg, n=8), did induce motor dysfunction on the rota-rod test at 45 minutes post-injection. (C) Examples of SNL rat exploration activity (10 minutes' duration) in the open field test before and 45 minutes after injection of saline, DALDA (10 mg/kg, s.c.), and morphine (10 mg/kg, s.c., n=5/group). (D–E) In the open field test, morphine (10 mg/kg, s.c.), but not DALDA (10 mg/kg, s.c.) or saline (n=5/group), reduced the total distance traveled in 10 minutes and the number of center crossings by SNL rats at 45 minutes after injection. *p<0.05, **p<0.01 versus pretreatment, paired t-test. Data are expressed as mean + SEM.
Discussion
The therapeutic utility of centrally-penetrating MOR agonists in neuropathic pain treatment is limited by central adverse effects. In this study, we utilized various behavioral, pharmacologic, in vivo electrophysiologic, and molecular biologic tools to demonstrate the peripherally restricted, modality-preferred anti-hyperalgesic effects of systemic DALDA. We further delineated potential underlying cellular mechanisms that will help to establish the therapeutic utility of peripherally acting opioids for the treatment of neuropathic pain.
Our in vivo electrophysiologic recordings revealed that DALDA predominantly inhibited C-fiber inputs, which signal thermal and noxious mechanical information, to WDR neurons. These findings complement those of the animal behavioral studies, which also showed that systemic DALDA more effectively inhibited heat hypersensitivity (mediated by C-fibers) than mechanical hypersensitivity (likely mediated by A-fibers). Although the A-component of WDR neurons to electrical simulation was not reduced, a subgroup of WDR neurons showed decreased responses to natural mechanical stimulation after systemic DALDA treatment. The reason for this discrepancy is unclear, but it is possible that systemic DALDA may change A-fiber conduction properties, which can be revealed when WDR neurons receive a prolonged barrage of afferent inputs produced by mechanical stimuli (5-second), but may not be observed with a short (2-millisecond) high-intensity electrical pulse. Systemic DALDA also inhibited windup of the C-component, reflecting a short-term neuronal sensitization to repetitive noxious inputs that occurs during natural stimulation of C-fibers.34–36 Thus, systemic DALDA may also ameliorate the neuronal sensitization that underlies development of hyperalgesia by inhibiting peripheral noxious inputs.37–41
Although pharmacokinetic data are unavailable for subcutaneous DALDA administration in nerve-injured rats, our findings indicate that DALDA may not appreciably accumulate in CNS after systemic administration. Importantly, systemic DALDA-induced inhibition of both mechanical and heat hyperalgesia was blocked by systemic methylnaltrexon, but was unaffected by intrathecal CTOP, which blocks spinal MORs over a prolonged period. Since activation of MORs on dorsal horn neurons would reduce WDR neuronal excitability and inhibit their responses to both A- and C-fiber inputs, the finding that A-component in WDR neurons was not reduced by DALDA may also imply that systemic DALDA does not activate spinal MORs. Although systemic DALDA may activate MOR in the brain regions to induce pain inhibition, the following findings suggest that DALDA inhibits neuropathic pain primarily through peripheral mechanisms in current study. Activation of MORs in CNS would induce both anti-hyperalgesia and anti-nociception. DALDA is highly specific to MOR and is 14-fold more potent than morphine.16 However, unlike morphine, which often induces anti-nociception, systemic DALDA normalized heat hyperalgesia without producing anti-nociception (e.g., PWL above pre-injury baseline) in nerve-injured rats. Additionally, contralateral PWT and PWL in SNL rats did not increase after systemic DALDA treatment. If DALDA had entered the CNS, it would have induced anti-nociception. Additionally, there is also an absence of any demonstrable CNS-related side effects known to morphine in SNL rats after systemic DALDA treatment.
We further examined the mechanisms for modality-preferred pain inhibition by systemic DALDA in male SNL rats. Different peripheral and central mechanisms are involved in mechanical and heat hypersensitivity. Both animal behavioral and in vivo electrophysiologic findings point to a peripheral site of action for systemic DALDA. Multimodal nociceptors in the peripheral nervous system express a wide array of ion channels and receptors that transduce intense thermal, mechanical, chemical, or cold stimuli into electrical activity.42,43 Some receptors are segregated into different subsets of DRG neurons, such as the “heat receptor” TRPV1 and the “cold receptor” TRPM8. Mas-related G-protein-coupled receptors (e.g., MrgC, D) also are distributed in a mutually exclusive fashion in DRG neurons.30,33,44,45 Thus, different subpopulations of primary sensory neurons may contribute to the modality-specific differences in DALDA’s effects on mechanical and thermal hyperalgesia.30,46 Small-diameter DRG neurons, which are presumably nociceptive, generally can be separated into peptidergic and non-peptidergic subpopulations. The vanilloid receptor TRPV1 is well known for its role in heat pain signaling. TRPV1 and MOR are more highly expressed and co-localized in peptidergic DRG neurons (likely IB4−) than in the non-peptidergic subpopulation. DALDA shows high binding affinity and is highly selectivity for MORs with a selectivity ratio Ki-delta/Ki-mu of 11,400.16–18 Therefore, DALDA may preferably inhibit heat-sensing TRPV1+ neurons by activating MORs. This preference may partially explain the greater efficacy of systemic DALDA to inhibit heat hypersensitivity than mechanical hypersensitivity. In line with this notion, DALDA significantly inhibited the [Ca2+] increase induced by KCl in IB4−, but not in IB4+ neurons. Further, DALDA induced significantly greater inhibition of the [Ca2+] increase evoked by capsaicin, which activates TRPV1, than that evoked by β-alanine, which activates MrgD. TRPV1+ neurons play a dominant role in heat nociception and hyperalgesia, whereas MrgD+ neurons are important to mechanical pain signaling.30,31,47 MrgD is expressed mostly in non-peptidergic neurons and rarely co-localize with TRPV1. Indeed, our results also suggest that only 4% of DRG neurons respond to both capsaicin and β-alanine. Systemic treatment with RTX, which selectively decreases the excitability of TRPV1+ neurons,48,49 produced a prolonged reversal of heat hypersensitivity, but not mechanical hypersensitivity, in nerve-injured rats. Interestingly, RTX-treatment did not affect the attenuation of mechanical allodynia by systemic administration of DALDA. This finding suggests that systemic DALDA ameliorates mechanical and heat hypersensitivity via different cellular targets. Together, these findings suggest a potential cellular mechanism by which DALDA preferentially inhibits heat hypersensitivity.
Finally, to study the safety profile of DALDA, we tested DALDA-treated rats in rota-rod and open-field test. In contrast to morphine, systemic DALDA did not impair motor coordination of rats in the rota-rod test, even at the highest doses tested. Additionally, whereas morphine-treated rats exhibited reductions in distance traveled and travel speed in the open field test, the locomotor function of DALDA-treated rats appeared unchanged from baseline. Additional findings from other investigators suggest that DALDA produces only a transient, minor increase in blood pressure and does not affect maternal respiratory, hemodynamic, or metabolic functions, further suggesting that DALDA has a minimal side-effect profile.50 Nevertheless, DALDA may share other PNS side effects (e.g., constipation, vomiting, dry mouth) known to peripherally acting mu-opioids following repetitive and long term drug treatment. The pharmacokinetic, PNS side effects and influences on bowel function of DALDA need to be systematic and carefully investigated in future, in order to fully characterize the pharmacological properties of this molecule. Our recent study suggested that repeated use of loperamide for alleviating neuropathic mechanical hypersensitivity may lead to the development of tolerance, possibly at peripheral opioid receptors.51 Because different MOR agonists induce different magnitudes of receptor internalization, desensitization, and tolerance processes, it remains to be tested whether DALDA leads to the development of analgesic tolerance or opioid-induced hyperalgesia.
Systemic DALDA (10 mg/kg, s.c.) also alleviated mechanical and heat hypersensitivity in female rats after nerve injury. Intriguingly, there was a trend that MPE for DALDA to inhibit mechanical hypersensitivity in female rats was lower than that in male rats, suggesting possible gender based differences. Since estrous cycles of female may profoundly affect pain response and drug effects,52–54 future studies need to characterize gender difference and determine effects of estrous cycles on the efficacy and mechanisms of pain inhibition by DALDA. Such studies will help to fully establish the clinical usefulness of peripherally acting opioids for a therapeutic formulation. In summary, our findings suggest that systemic administration of DALDA attenuates both mechanical and heat hypersensitivity in nerve-injured rats through activation of MORs at peripheral but not central sites. Further, the efficacy of DALDA to inhibit heat hypersensitivity is greater than that to inhibit mechanical hypersensitivity in male rats. Because it does not affect CNS function, DALDA may pose minimal risk for central dose-limiting adverse effects and have low addiction or abuse potential. Hence, DALDA may represent a promising therapeutic alternative to currently used opioids for the treatment of neuropathic pain.
Acknowledgements
The authors thank Claire F. Levine, MS (scientific editor, Department of Anesthesiology/CCM, Johns Hopkins University), for editing the manuscript.
Disclosure of funding: This study was supported by grants from the National Institutes of Health (Bethesda, Maryland, USA): NS26363 (S.N.R.) and NS70814 (Y.G.). This study was also subsidized by a seed grant from the Johns Hopkins Blaustein Pain Research Fund (Y.G.) and a grant from the National Natural Science Foundation of China: 81428008 (Y.W.). X.D. is a faculty of the Howard Hughes Medical Institute. This work was facilitated by the Pain Research Core funded by the Blaustein Fund and the Neurosurgery Pain Research Institute at the Johns Hopkins University. Ronen Shechter received funding from Ruth L. Kirschstein National Research Service Award- Sponsoring institution: Johns Hopkins University. Grant No. 2T32GM075774-7
Footnotes
The authors declare no competing interests.
Authors' contributions: V.T., F.Y., and S-Q.H. performed most of the experiments and were involved in writing a draft manuscript. R.S., C.Z., B.S., T.Z., and V.T. performed or assisted with portions of the experiments. Y.W. and X.D. were involved in experimental design, data analysis, and interpretation. Y.G. and S.N.R. designed and directed the project and wrote the final manuscript.
References
- 1.Mathieson S, Maher CG, Terwee CB, Folly de CT, Lin CW. Neuropathic pain screening questionnaires have limited measurement properties. A systematic review. J. Clin. Epidemiol. 2015;68(8):957–966. doi: 10.1016/j.jclinepi.2015.03.010. [DOI] [PubMed] [Google Scholar]
- 2.Attal N, Bouhassira D. Pharmacotherapy of neuropathic pain: which drugs, which treatment algorithms? Pain. 2015;156(Suppl 1):S104–S114. doi: 10.1097/01.j.pain.0000460358.01998.15. [DOI] [PubMed] [Google Scholar]
- 3.Gewandter JS, Dworkin RH, Turk DC, Farrar JT, Fillingim RB, Gilron I, Markman JD, Oaklander AL, Polydefkis MJ, Raja SN, Robinson JP, Woolf CJ, Ziegler D, Ashburn MA, Burke LB, Cowan P, George SZ, Goli V, Graff OX, Iyengar S, Jay GW, Katz J, Kehlet H, Kitt RA, Kopecky EA, Malamut R, McDermott MP, Palmer P, Rappaport BA, Rauschkolb C, Steigerwald I, Tobias J, Walco GA. Research design considerations for chronic pain prevention clinical trials: IMMPACT recommendations. Pain. 2015;156(7):1184–1197. doi: 10.1097/j.pain.0000000000000191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Campbell JN, Meyer RA. Mechanisms of neuropathic pain. Neuron. 2006;52:77–92. doi: 10.1016/j.neuron.2006.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kim KJ, Yoon YW, Chung JM. Comparison of three rodent neuropathic pain models. Exp. Brain Res. 1997;113:200–206. doi: 10.1007/BF02450318. [DOI] [PubMed] [Google Scholar]
- 6.Woolf CJ, Mannion RJ. Neuropathic pain: aetiology, symptoms, mechanisms, and management. Lancet. 1999;353:1959–1964. doi: 10.1016/S0140-6736(99)01307-0. [DOI] [PubMed] [Google Scholar]
- 7.Backonja MM. Neuropathic pain therapy: from bench to bedside. Semin. Neurol. 2012;32:264–268. doi: 10.1055/s-0032-1329204. [DOI] [PubMed] [Google Scholar]
- 8.Rosenblum A, Marsch LA, Joseph H, Portenoy RK. Opioids and the treatment of chronic pain: controversies, current status, and future directions. Exp. Clin. Psychopharmacol. 2008;16:405–416. doi: 10.1037/a0013628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Benyamin R, Trescot AM, Datta S, Buenaventura R, Adlaka R, Sehgal N, Glaser SE, Vallejo R. Opioid complications and side effects. Pain Physician. 2008;11:S105–S120. [PubMed] [Google Scholar]
- 10.Finnerup NB, Attal N, Haroutounian S, McNicol E, Baron R, Dworkin RH, Gilron I, Haanpaa M, Hansson P, Jensen TS, Kamerman PR, Lund K, Moore A, Raja SN, Rice AS, Rowbotham M, Sena E, Siddall P, Smith BH, Wallace M. Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol. 2015;14:162–173. doi: 10.1016/S1474-4422(14)70251-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Stein C, Clark JD, Oh U, Vasko MR, Wilcox GL, Overland AC, Vanderah TW, Spencer RH. Peripheral mechanisms of pain and analgesia. Brain Res. Rev. 2009;60:90–113. doi: 10.1016/j.brainresrev.2008.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vadivelu N, Mitra S, Hines RL. Peripheral opioid receptor agonists for analgesia: a comprehensive review. J. Opioid. Manag. 2011;7:55–68. doi: 10.5055/jom.2011.0049. [DOI] [PubMed] [Google Scholar]
- 13.Montecucchi PC, de CR, Erspamer V. Identification of dermorphin and Hyp6-dermorphin in skin extracts of the Brazilian frog Phyllomedusa rhodei. Int. J. Pept. Protein Res. 1981;17:316–321. doi: 10.1111/j.1399-3011.1981.tb01997.x. [DOI] [PubMed] [Google Scholar]
- 14.Scalia S, Salvadori S, Marastoni M, Bortolotti F, Tomatis R. Reversed-phase HPLC study on the in vitro enzymic degradation of dermorphin. Peptides. 1986;7:247–251. doi: 10.1016/0196-9781(86)90221-4. [DOI] [PubMed] [Google Scholar]
- 15.Sasaki Y, Hosono M, Matsui M, Fujita H, Suzuki K, Sakurada S, Sakurada T, Kisara K. On the degradation of dermorphin and D-Arg2-dermorphin analogs by a soluble rat brain extract. Biochem. Biophys. Res. Commun. 1985;130:964–970. doi: 10.1016/0006-291x(85)91709-7. [DOI] [PubMed] [Google Scholar]
- 16.Shimoyama M, Shimoyama N, Zhao GM, Schiller PW, Szeto HH. Antinociceptive and respiratory effects of intrathecal H-Tyr-D-Arg-Phe-Lys-NH2 (DALDA) and [Dmt1] DALDA. J. Pharmacol. Exp. Ther. 2001;297:364–371. [PubMed] [Google Scholar]
- 17.Schiller PW, Nguyen TM, Chung NN, Lemieux C. Dermorphin analogues carrying an increased positive net charge in their "message" domain display extremely high mu opioid receptor selectivity. J. Med. Chem. 1989;32:698–703. doi: 10.1021/jm00123a035. [DOI] [PubMed] [Google Scholar]
- 18.Zimmerman DM, Leander JD. Selective opioid receptor agonists and antagonists: research tools and potential therapeutic agents. J. Med. Chem. 1990;33:895–902. doi: 10.1021/jm00165a002. [DOI] [PubMed] [Google Scholar]
- 19.He SQ, Li Z, Chu YX, Han L, Xu Q, Li M, Yang F, Liu Q, Tang Z, Wang Y, Hin N, Tsukamoto T, Slusher B, Tiwari V, Shechter R, Wei F, Raja SN, Dong X, Guan Y. MrgC agonism at central terminals of primary sensory neurons inhibits neuropathic pain. Pain. 2014;155:534–544. doi: 10.1016/j.pain.2013.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Guan Y, Liu Q, Tang Z, Raja SN, Anderson DJ, Dong X. Mas-related G-protein-coupled receptors inhibit pathological pain in mice. Proc. Natl. Acad. Sci. U. S. A. 2010;107:15933–15938. doi: 10.1073/pnas.1011221107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shechter R, Yang F, Xu Q, Cheong YK, He SQ, Sdrulla A, Carteret AF, Wacnik PW, Dong X, Meyer RA, Raja SN, Guan Y. Conventional and kilohertz-frequency spinal cord stimulation produces intensity- and frequency-dependent inhibition of mechanical hypersensitivity in a rat model of neuropathic pain. Anesthesiology. 2013;119:422–432. doi: 10.1097/ALN.0b013e31829bd9e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods. 1994;53:55–63. doi: 10.1016/0165-0270(94)90144-9. [DOI] [PubMed] [Google Scholar]
- 23.Dixon WJ. Efficient analysis of experimental observations. Annu. Rev. Pharmacol. Toxicol. 1980;20:441–462. doi: 10.1146/annurev.pa.20.040180.002301. [DOI] [PubMed] [Google Scholar]
- 24.Brittain JM, Duarte DB, Wilson SM, Zhu W, Ballard C, Johnson PL, Liu N, Xiong W, Ripsch MS, Wang Y, Fehrenbacher JC, Fitz SD, Khanna M, Park CK, Schmutzler BS, Cheon BM, Due MR, Brustovetsky T, Ashpole NM, Hudmon A, Meroueh SO, Hingtgen CM, Brustovetsky N, Ji RR, Hurley JH, Jin X, Shekhar A, Xu XM, Oxford GS, Vasko MR, White FA, Khanna R. Suppression of inflammatory and neuropathic pain by uncoupling CRMP-2 from the presynaptic Ca(2)(+) channel complex. Nat. Med. 2011;17:822–829. doi: 10.1038/nm.2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Han L, Ma C, Liu Q, Weng HJ, Cui Y, Tang Z, Kim Y, Nie H, Qu L, Patel KN, Li Z, McNeil B, He S, Guan Y, Xiao B, LaMotte RH, Dong X. A subpopulation of nociceptors specifically linked to itch. Nat. Neurosci. 2013;16:174–182. doi: 10.1038/nn.3289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Q, Tang Z, Surdenikova L, Kim S, Patel KN, Kim A, Ru F, Guan Y, Weng HJ, Geng Y, Undem BJ, Kollarik M, Chen ZF, Anderson DJ, Dong X. Sensory Neuron-Specific GPCR Mrgprs Are Itch Receptors Mediating Chloroquine-Induced Pruritus. Cell. 2009;39(7):1353–1365. doi: 10.1016/j.cell.2009.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Campbell JN, Meyer RA. Mechanisms of neuropathic pain. Neuron. 2006;52:77–92. doi: 10.1016/j.neuron.2006.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Abrahamsen B, Zhao J, Asante CO, Cendan CM, Marsh S, Martinez-Barbera JP, Nassar MA, Dickenson AH, Wood JN. The cell and molecular basis of mechanical, cold, and inflammatory pain. Science. 2008;321:702–705. doi: 10.1126/science.1156916. [DOI] [PubMed] [Google Scholar]
- 29.Baron R. Mechanisms of disease: neuropathic pain--a clinical perspective. Nat. Clin. Pract. Neurol. 2006;2:95–106. doi: 10.1038/ncpneuro0113. [DOI] [PubMed] [Google Scholar]
- 30.Cavanaugh DJ, Lee H, Lo L, Shields SD, Zylka MJ, Basbaum AI, Anderson DJ. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc. Natl. Acad. Sci. U. S. A. 2009;106:9075–9080. doi: 10.1073/pnas.0901507106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Basbaum AI, Bautista DM, Scherrer G, Julius D. Cellular and molecular mechanisms of pain. Cell. 2009;139:267–284. doi: 10.1016/j.cell.2009.09.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang H, Zylka MJ. Mrgprd-expressing polymodal nociceptive neurons innervate most known classes of substantia gelatinosa neurons. J. Neurosci. 2009;29:13202–13209. doi: 10.1523/JNEUROSCI.3248-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zylka MJ. Nonpeptidergic circuits feel your pain. Neuron. 2005;47:771–772. doi: 10.1016/j.neuron.2005.09.003. [DOI] [PubMed] [Google Scholar]
- 34.Hughes AM, Rhodes J, Fisher G, Sellers M, Growcott JW. Assessment of the effect of dextromethorphan and ketamine on the acute nociceptive threshold and wind-up of the second pain response in healthy male volunteers. Br. J. Clin. Pharmacol. 2002;53:604–612. doi: 10.1046/j.1365-2125.2002.01602.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Magerl W, Wilk SH, Treede RD. Secondary hyperalgesia and perceptual wind-up following intradermal injection of capsaicin in humans. Pain. 1998;74:257–268. doi: 10.1016/s0304-3959(97)00177-2. [DOI] [PubMed] [Google Scholar]
- 36.Staud R, Robinson ME, Price DD. Temporal summation of second pain and its maintenance are useful for characterizing widespread central sensitization of fibromyalgia patients. J. Pain. 2007;8:893–901. doi: 10.1016/j.jpain.2007.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Vikman KS, Kristensson K, Hill RH. Sensitization of dorsal horn neurons in a two-compartment cell culture model: wind-up and long-term potentiation-like responses. J. Neurosci. 2001;21:RC169. doi: 10.1523/JNEUROSCI.21-19-j0004.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Eide PK. Wind-up and the NMDA receptor complex from a clinical perspective. Eur. J. Pain. 2000;4:5–15. doi: 10.1053/eujp.1999.0154. [DOI] [PubMed] [Google Scholar]
- 39.Nackley AG, Zvonok AM, Makriyannis A, Hohmann AG. Activation of cannabinoid CB2 receptors suppresses C-fiber responses and windup in spinal wide dynamic range neurons in the absence and presence of inflammation. J. Neurophysiol. 2004;92:3562–3574. doi: 10.1152/jn.00886.2003. [DOI] [PubMed] [Google Scholar]
- 40.Ren K. Wind-up and the NMDA receptor: from animal studies to humans. Pain. 1994;59:157–158. doi: 10.1016/0304-3959(94)90067-1. [DOI] [PubMed] [Google Scholar]
- 41.Herrero JF, Laird JM, Lopez-Garcia JA. Wind-up of spinal cord neurones and pain sensation: much ado about something? Prog. Neurobiol. 2000;61:169–203. doi: 10.1016/s0301-0082(99)00051-9. [DOI] [PubMed] [Google Scholar]
- 42.Julius D, Basbaum AI. Molecular mechanisms of nociception. Nature. 2001;413:203–210. doi: 10.1038/35093019. [DOI] [PubMed] [Google Scholar]
- 43.Patapoutian A, Tate S, Woolf CJ. Transient receptor potential channels: targeting pain at the source. Nat. Rev. Drug Discov. 2009;8:55–68. doi: 10.1038/nrd2757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Dong X, Han S, Zylka MJ, Simon MI, Anderson DJ. A diverse family of GPCRs expressed in specific subsets of nociceptive sensory neurons. Cell. 2001;106:619–632. doi: 10.1016/s0092-8674(01)00483-4. [DOI] [PubMed] [Google Scholar]
- 45.Zylka MJ, Rice FL, Anderson DJ. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron. 2005;45:17–25. doi: 10.1016/j.neuron.2004.12.015. [DOI] [PubMed] [Google Scholar]
- 46.Braz JM, Nassar MA, Wood JN, Basbaum AI. Parallel "pain" pathways arise from subpopulations of primary afferent nociceptor. Neuron. 2005;47:787–793. doi: 10.1016/j.neuron.2005.08.015. [DOI] [PubMed] [Google Scholar]
- 47.Rau KK, McIlwrath SL, Wang H, Lawson JJ, Jankowski MP, Zylka MJ, Anderson DJ, Koerber HR. Mrgprd enhances excitability in specific populations of cutaneous murine polymodal nociceptors. J. Neurosci. 2009;29:8612–8619. doi: 10.1523/JNEUROSCI.1057-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ossipov MH, Bian D, Malan TP, Jr, Lai J, Porreca F. Lack of involvement of capsaicin-sensitive primary afferents in nerve-ligation injury induced tactile allodynia in rats. Pain. 1999;79:127–133. doi: 10.1016/s0304-3959(98)00187-0. [DOI] [PubMed] [Google Scholar]
- 49.King T, Qu C, Okun A, Mercado R, Ren J, Brion T, Lai J, Porreca F. Contribution of afferent pathways to nerve injury-induced spontaneous pain and evoked hypersensitivity. Pain. 2011;152:1997–2005. doi: 10.1016/j.pain.2011.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Clapp JF, III, Kett A, Olariu N, Omoniyi AT, Wu D, Kim H, Szeto HH. Cardiovascular and metabolic responses to two receptor-selective opioid agonists in pregnant sheep. Am. J. Obstet. Gynecol. 1998;178:397–401. doi: 10.1016/s0002-9378(98)80032-x. [DOI] [PubMed] [Google Scholar]
- 51.He SQ, Yang F, Perez FM, Xu Q, Shechter R, Cheong YK, Carteret AF, Dong X, Sweitzer SM, Raja SN, Guan Y. Tolerance develops to the antiallodynic effects of the peripherally acting opioid loperamide hydrochloride in nerve-injured rats. Pain. 2013;154:2477–2486. doi: 10.1016/j.pain.2013.07.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sorge RE, Mapplebeck JC, Rosen S, Beggs S, Taves S, Alexander JK, Martin LJ, Austin JS, Sotocinal SG, Chen D, Yang M, Shi XQ, Huang H, Pillon NJ, Bilan PJ, Tu Y, Klip A, Ji RR, Zhang J, Salter MW, Mogil JS. Different immune cells mediate mechanical pain hypersensitivity in male and female mice. Nat. Neurosci. 2015;18:1081–1083. doi: 10.1038/nn.4053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mogil JS. Sex differences in pain and pain inhibition: multiple explanations of a controversial phenomenon. Nat. Rev. Neurosci. 2012;13:859–866. doi: 10.1038/nrn3360. [DOI] [PubMed] [Google Scholar]
- 54.Taves S, Berta T, Liu DL, Gan S, Chen G, Kim YH, Van dV, Laufer S, Ji RR. Spinal inhibition of p38 MAP kinase reduces inflammatory and neuropathic pain in male but not female mice: Sex-dependent microglial signaling in the spinal cord. Brain Behav. Immun. 2015 doi: 10.1016/j.bbi.2015.10.006. S0889-1591(15)30032-5. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]