Abstract
[Purpose] This study evaluated the effects of topical oxygen therapy on the hind limb wounds of rats under ischemic conditions. [Subjects and Methods] Twelve injured rats were treated with topical oxygen on skin wounds located on the hind limb and compared with twelve injured control rats. Indexes including gross morphology of the wound, wound healing time, wound healing rate, and histological and immunohistochemical staining of sections of wound tissue were examined at different time points after intervention. [Results] The wound healing time was shorter in the topical oxygen therapy group than the control group. The wound healing rate and granulation tissue formation in the topical oxygen therapy group showed significant improvement on days 3, 7, and 14. Through van Gieson staining, the accumulation of collagen fiber in the topical oxygen therapy group was found to have improved when compared with the control group on day 7. Through semiquantitative immunohistochemical staining, many more new vessels were found in the topical oxygen therapy group compared with the model control group on day 7. [Conclusion] The results of the experiment showed that topical oxygen therapy improved ischemic wound healing.
Key words: Wound healing, Animal model, Topical oxygen therapy
INTRODUCTION
Normal wound healing involves a series of sequential stages, including hemostasis, inflammation, remodeling of granulation tissue, and reepithelialization1, 2). In general, wounds can recover soon after they occur. However, in clinical practice, the healing process is subject to the combined influence of various factors, such as diabetes, pressure ulcers, radiation injuries, peripheral vascular diseases, and venous stasis diseases. These cause blood vessel breaks and low supplies and high demands of oxygen, leading to hypoxia3, 4), which in turn results in delayed healing. Oxygen is a key requirement for wound healing5). Oxygen and reactive oxygen species are involved in all stages of wound healing, modulating cell migration, adhesion, proliferation, neovascularization, remodeling, and apoptosis6, 7). The present research discovered that increasing the oxygen delivered to tissues in the area of a wound could accelerate wound healing.
Oxygen therapy has been applied in the clinical treatment of wounds since the 1960s8). There are two popular methods of oxygen-based therapies, namely, HBO (hyperbaric oxygen) and TOT (topical oxygen therapy). HBO increase local tissue oxygen by improving the blood oxygen concentration9). At present, HBO is an FDA-approved treatment for chronic wounds10). Compared with HBO, TOT has the following advantages: the required facilities are simpler, it is much cheaper, and it involves no risk of hyperbaric oxygen toxicity in organs. Thus it is drawing increasing attention from medical researchers11, 12). However, there are controversies regarding the effectiveness of topical oxygen therapy in treating chronic wounds and the work parameters for widespread application13). Further research is still needed.
In order to study the effects of TOT on the healing of ischemic wounds, a hind limb ischemic wound model was prepared by ligation of the abdominal aorta, and a comparison was made between a TOT group and model control group. The present experiment aimed to explore the effects of TOT on wound healing and molecular mechanisms by examination of the gross morphology and pathology.
SUBJECTS AND METHODS
We used an ischemic wound model. Twenty-four Sprague-Dawley rats of either sex weighing 180–250 g (Zhongshan University Experimental Animal Center) were housed in a temperature controlled animal holding room with a 12-hour light/dark cycle. They were fasted for 8 hours with free access to water before the experiment. The experimental protocol was approved by the Ethics Committee for Animal Research, Jinan University, China. All animals received humane care in compliance with the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientifical Purposes.
After anesthesia achieved by intraperitoneal injection of chloral hydrate (10 mL/kg body weight), the rats were fixed supine on an animal experiment table, and then the belly hair was picked off. An incision about 2.5 cm long was made in the middle of the bilateral anterior superior iliac spine. The abdomen was opened, the intestine and other organs were pushed to the left, and the abdominal aorta was exposed. The abdominal aorta was tied where it divided into the left and right common iliac artery, and the abdominal incision was then sutured. The hind limb hair was shaved, and a full-thickness square-shaped skin wound (d=1.1 cm) was made with a hole puncher on the hind limb.
We used the device shown in Fig. 1 to perform TOT. The TOT group received topical oxygen therapy daily (4 L/min, 30 min/day, 1 time/day). The model control group only received routine dressing changes.
Fig. 1.

A rat with the topical oxygen device on a limb The device supplied 100% oxygen at 1 atm of pressure
Wound area healing rate is the most direct indicator for assessment of wound healing. On days 3, 7, 10, and 14 after treatment, the wounds were photographed with a digital camera, and the ImageJ software was used to measure the wound area and calculate the wound healing rate at each time point. The formula used was as follows: the wound healing rate = (initial wound area –time point area) / initial wound area × 100%. We decided that the wound was healed when the wound healing rate was greater than 95%. We observed the gross morphology of the wounds every day and studied wound healing with hematoxylin and eosin (HE) and van Gieson staining (VG) and immunohistochemistry.
On days 3, 7, 10, and 14, three rats were taken from each group randomly. The wound area and a little normal skin around the wound were removed and put in 10% neutral buffer formalin. Three tissue sections with thicknesses of 5 um were taken from each sample, one for histomorphology, one for van Gieson staining, and one for histochemistry.
The data were analyzed by using the SPSS software (version 13.0) and were expressed as the mean ± standard deviation. The group differences were analyzed by a variance analysis of the repeated measurements. A p<0.05(*) was considered statistically significant. All differences referred to in the text have been statistically verified.
RESULTS
According to the results of comparing changes in wound size at each time point, in the topical oxygen therapy group, obvious growth of granulation tissues could be seen on days 2–3, and it was bright red, soft, moist, and prone to bleeding. Obvious wound contraction was observed on days 4–7. The wound area decreased obviously on days 7–10. Then the wound healed on days 12–14. For the model control group, the granulation tissues grew more slowly. Growth of granulation tissue was visible on days 4–5, and it was pink, soft, moist, and prone to bleeding. Wound secretions were much greater in the control group than in the TOT group. The wound showed an obvious contraction on days 6–9, and it healed on days 14–16. Both group’s wounds were flat, with no obvious scar left on the skin afterwards (Fig. 2). The wound healing rates of the two groups showed significant differences on days 3, 7, 14 (Table 2). The average healing time was 12.8 + 0.6 days in the TOT group and 14.7 + 0.8 days in the model control group (Table 1). The difference was statistically different (p<0.05); that is to say, the wounds of the TOT group healed faster than those of the model control group.
Fig. 2.
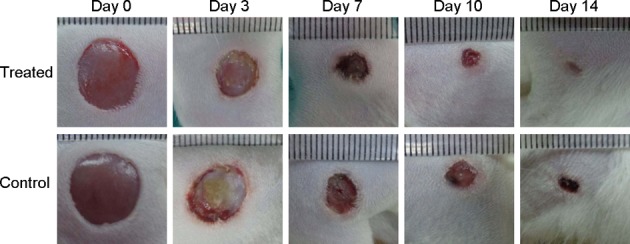
Typical macroscopic results showing that the wounds of the oxygen group healed faster than those of the control group
Table 2. Comparison of the wound healing rate.
| Day 3 | Day 7 | Day 10 | Day 14 | |
|---|---|---|---|---|
| Control group (n=6) | 45.1±1.9 | 79.4±2.1 | 87.8±1.4 | 97.8±1.5 |
| Treated group (n=6) | 37.3±4.4** | 74.0±2.5** | 85.6±2.7 | 94.5±2.8* |
Values are presented as the mean±SD.
Tested by independent-samples t-test (*p<0.05; **p<0.01)
Treated group: topical oxygen therapy
Table 1. Comparison of wound healing time.
| Group | Healing time |
|---|---|
| Control group | 12.8±0.6 |
| Treated group | 14.7±0.8* |
Values are presented as the mean±SD. Tested by independent-samples t-test (*p<0.05). Treated group: topical oxygen therapy
According to observation of the histomorphological changes, we found that new granulation tissues were formed by collagen fibers, fibroblasts, and capillaries in the first three days in both groups. There was a large number of inflammatory cells in both groups, but there were not so many new capillaries in the model control group compared with the TOT group. Both groups were in a significant hyperplastic state on days 7–10, but there were still a large number of inflammatory cells and some small arteries and veins in the model control group. However, the TOT group had a much smaller amount of inflammatory cells in the granulation tissue and showed a rapid increase in small arteries and veins. By day 14, the wounds in the TOT group were completely covered by the epithelium, and large number of mature granulation tissues and skin appendages were under the epithelium. However, the wounds in the control group had not yet completely healed by day 14, and a large number of small blood vessels and disordered collagen fibers were found in the granulation tissue (Fig. 3).
Fig. 3.
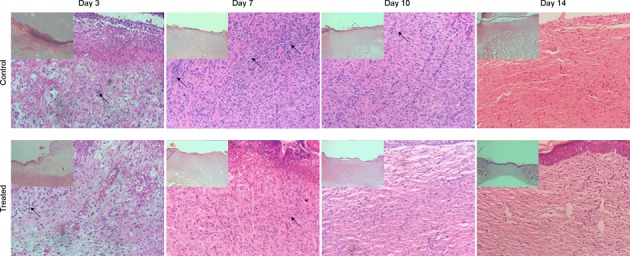
HE staining showed that the granulation tissues grew better in the TOT group than the model control group. On days 7–10, the inflammatory cells decreased in the TOT group; however, there were still large numbers of inflammatory cells in the control group
According to the results of VG staining, there were very few collagen fibers growing on the first three days in both groups. The collagen fibers gradually increased on days 3–7, but there were much more collagen fibers in the TOT group than the model control group on day 7. The collagen fibers in the TOT group were neatly arranged, and the structure was integrated; in contrast, the collagen fibers in the model control group were fewer and disorganized. However, there was no significant difference between the two groups on days 10–14 (Fig. 4).
Fig. 4.
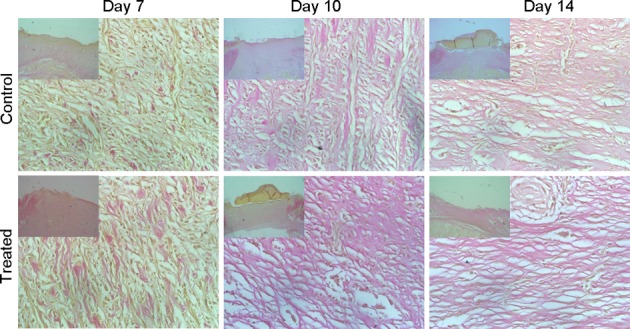
Collagen fibers in the wounds. On day 7, there were many more collagen fibers in the TOT group
According to the results of comparing the expressions of CD31 at each time point. Only very few new vessels could be found at the bottom of the wounds on the third day in both groups. Angiogenesis increased significantly in both groups on days 3–10, but it was much more significant in the TOT group than in the control group on day 7. On day 14, many vessels in the TOT group were occluded, and the wounds were healed. However, there were still many vessels that were large and not occluded in the model control group (Fig. 5, Table 3). But the number of vessels in the two groups showed no significant difference.
Fig. 5.
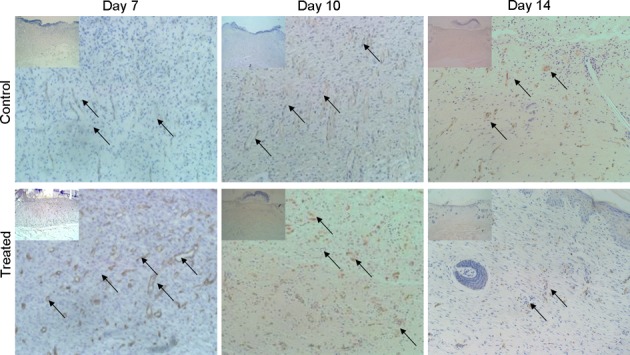
The expression of CD31 showed that on day 7, there were many more new vessels in the TOT group than the model control group; however, on days 10-14, there were no significant differences between the two groups
Table 3. Immunohistochemical reaction of CD31 in the wound.
| Day 7 | Day 10 | Day 14 | |
|---|---|---|---|
| Control group | ++ | +++ | ++ |
| Treated group | ++++ | +++ | + |
DISCUSSION
With a rapid increase in the number of patients with diabetes and other diseases, more and more people are suffering from diabetic wounds, pressure ulcers, leg ulcers, and other problem wounds. Although there are a variety of different chronic wounds and the treatments for them are not the same, this study found that in a variety of chronic wounds, blood vessel rupture and the localized hypoxic environment caused by peripheral vascular disease were important factors that restrict wound healing14). The oxygen balance (oxygen supply is balanced with oxygen demand) was broken. According to one study, the oxygen pressure in the center of a wound is only 0–10 mmHg15, 16). This condition has a great impact on collagen deposition, neovascularization, and bactericidal function.
In a preliminary experiment, we constructed a simulated wound model to study the following factors, which could affect the wound surface oxygen concentration, with the use of TOT: the manner of providing oxygen, oxygen flow, the equipment space, dressing, and water evaporation. Then we selected the best work parameters: wrapping TOT devices around the wound, an oxygen flow of 4 L/min, and use of normal saline to make the oxygen fluid moist. We found that covering a wound with a dressing would not lead to oxygen concentration reduction. So in clinical practice, we do not need to open the dressing for patients who need TOT 2–3 times a day.
Topical oxygen therapy promotes wound healing. As can be seen from the healing time of the two groups, the TOT group was significantly ahead of the model control group (p<0.05), and the differences in healing rate on days 3, 7, and 10 between the two groups were significant. Pathological examination revealed that the inflammatory reaction was significantly mitigated and that the formation of collagen fibers, new vessels, and granulation tissues was more advanced compared with the model control group. It can be concluded that topical oxygen therapy can promote wound healing.
Infection is an important factor that affects wound healing. Inflammation increases once a wound is infected. Generally speaking, normal inflammation has a positive effect on wound healing post trauma, but excessive activation of the inflammatory response does a great damage to wound healing, as it can cause synthesis and release of excessive inflammatory factors. Topical oxygen therapy could promote wound healing by controlling bacterial infection and regulating wound inflammation by increasing the oxygen pressure in the wound. Hopf conducted a prospective study on the relation between wound infection and subcutaneous oxygen concentration in 130 patients and found that the subcutaneous oxygen concentration was inversely proportional to the likelihood of wound infection, so oxygen can reduce the probability of wound infection and promote the healing of a wound17). It is widely recognized that neutrophils, macrophages, monocytes, and endothelial cells can produce reactive oxygen species ROS, oxygen free radicals, and special enzymes to fight infection and other microbial breeding after a wound occurs. NADPH oxidase is a key factor in the generation of ROS, oxygen free radicals, and H2O2, and this process is a highly oxygen-dependent process (Km between 40–80 mmHg). Previous studies found that neutrophils would fail to kill bacteria if the partial pressure of oxygen was less than 40 mmHg18). Topical oxygen therapy inhibits bacterial breeding, reduces wound infection, enhances the bacteria killing function of neutrophils and macrophages10). So it can promote wound healing.
Both angiogenesis and collagen deposition are important factors in the formation of granulation tissues. Although it is widely accepted that a certain degree of hypoxia plays a positive role in angiogenesis in various kinds of acute and chronic wounds because hypoxia can start the ROS system and promote the expression of VEGF mRNA19,20,21). However, this does not mean that the longer and more serious the hypoxic environment, the better. Besides, there is a negative feedback mechanism of VEGF expression in the body, so the expression of VEGF genes was inhibited to a certain degree. According to the research of Chaudhuri, oxygen has the potential to induce vascular endothelial growth factor mRNA levels in endothelial cells and macrophages7). Meanwhile, tissue regeneration requires a large oxygen and energy supply, and sustained severe hypoxia can promote the expression of VEGF, but it also leads to the obstruction of vascular growth because of a lack of sufficient oxygen and energy. According to one previous study, the oxygen partial pressure in the center of a wound is only 0–10 mmHg22). In that study, the wound was in a severe hypoxic state, and this slowed down or even stopped fibroblast proliferation, collagen synthesis, and granulation tissue formation. One could also say that it delayed wound healing. Previous studies showed that the amount of collagen deposition had a positive correlation with the content of oxygen and that the best oxygen content for fibroblast synthesis of collagen fibers was 30–40 mmHg. According to one study, increasing the oxygen partial pressure 2 mm below the wound subsurface could effectively promote collagen deposition and angiogenesis and promote wound healing11). But we need to accept that the topical oxygen therapy only improves the condition of hypoxia in the wound bed; it does not change the state from hypoxia to the normal.
Covering of the wound by the crawling epithelium is an important process in wound healing. Loo and Halliwell found that H2O2 could promote keratinocyte proliferation and epithelial crawling distance23). However O’Toole obtained different results24). From our experiment, we found that TOT could clearly promote wound epithelium crawling. On day 14, the wounds in the TOT group were completely covered by the epithelium, but some granulation tissues were still uncovered in the control group.
In conclusion, we used an ischemic hind limb wound model in rats to test the effects of topical oxygen therapy. From the research, we found that TOT could promote ischemic wound healing by reducing wound infection and stimulating collagen deposition and angiogenesis by increasing the local oxygen partial pressure. We could not detect the wound oxygen concentration during and after treatment due to an instrument limitation. So the most beneficial oxygen level and a means of maintaining the oxygen balance in different stages of wound healing remain to be determined and need to be probed in the future.
Acknowledgments
Guangdong Province Foundation for Nature Science (S2013010015264) is gratefully acknowledged for funding this research. We would like to thank Shenzhen Kangyi Biotechnology Co., Ltd., for contributing the TOT device.
REFERENCES
- 1.Bitto A, Minutoli L, Altavilla D, et al. : Simvastatin enhances VEGF production and ameliorates impaired wound healing in experimental diabetes. Pharmacol Res, 2008, 57: 159–169. [DOI] [PubMed] [Google Scholar]
- 2.Jeon JK, Park SK, Lee JH: Effects of high voltage pulsed current stimulation with a visible contraction intensity on expression of TGF-β1 and synthesis of type I collagen in wound-induced white rats. J Phys Ther Sci, 2015, 27: 1485–1490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tandara AA, Mustoe TA: Oxygen in wound healing—more than a nutrient. World J Surg, 2004, 28: 294–300. [DOI] [PubMed] [Google Scholar]
- 4.Sen CK: Wound healing essentials: let there be oxygen. Wound Repair Regen, 2009, 17: 1–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Leaper D: Perfusion, oxygenation and warming. Int Wound J, 2007, 4: 4–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Black J: Tissue oxygen perfusion and pressure ulcer healing. Plast Surg Nurs, 2000, 20: 10–14. [DOI] [PubMed] [Google Scholar]
- 7.Chaudhuri V, Potts BR, Karasek MA: Mechanisms of microvascular wound repair I. Role of mitosis, oxygen tension, and I-kappa B. In Vitro Cell Dev Biol Anim, 2006, 42: 308–313. [DOI] [PubMed] [Google Scholar]
- 8.Hunt TK, Ellison EC, Sen CK: Oxygen: at the foundation of wound healing—introduction. World J Surg, 2004, 28: 291–293. [DOI] [PubMed] [Google Scholar]
- 9.Yan D, Shan J, Ze Y, et al. : The effects of combined hyperbaric oxygen therapy on patients with post-stroke depression. J Phys Ther Sci, 2015, 27: 1295–1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gordillo GM, Roy S, Khanna S, et al. : Topical oxygen therapy induces vascular endothelial growth factor expression and improves closure of clinically presented chronic wounds. Clin Exp Pharmacol Physiol, 2008, 35: 957–964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rodriguez PG, Felix FN, Woodley DT, et al. : The role of oxygen in wound healing: a review of the literature. Dermatol Surg, 2008, 34: 1159–1169. [DOI] [PubMed] [Google Scholar]
- 12.Sibbald RG, Woo KY, Queen D: Wound bed preparation and oxygen balance—a new component? Int Wound J, 2007, 4: 9–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tracey AK, Alcott CJ, Schleining JA, et al. : The effects of topical oxygen therapy on equine distal limb dermal wound healing. Can Vet J, 2014, 55: 1146–1152. [PMC free article] [PubMed] [Google Scholar]
- 14.Wu L, Xia YP, Roth SI, et al. : Transforming growth factor-beta1 fails to stimulate wound healing and impairs its signal transduction in an aged ischemic ulcer model: importance of oxygen and age. Am J Pathol, 1999, 154: 301–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jonsson K, Jensen JA, Goodson WH, 3rd, et al. : Tissue oxygenation, anemia, and perfusion in relation to wound healing in surgical patients. Ann Surg, 1991, 214: 605–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.LaVan FB, Hunt TK: Oxygen and wound healing. Clin Plast Surg, 1990, 17: 463–472. [PubMed] [Google Scholar]
- 17.Hopf HW, Hunt TK, West JM, et al. : Wound tissue oxygen tension predicts the risk of wound infection in surgical patients. Arch Surg, 1997, 132: 997–1004, discussion 1005. [DOI] [PubMed] [Google Scholar]
- 18.Hohn DC, MacKay RD, Halliday B, et al. : Effect of O2 tension on microbicidal function of leukocytes in wounds and in vitro. Surg Forum, 1976, 27: 18–20. [PubMed] [Google Scholar]
- 19.Sen CK, Khanna S, Babior BM, et al. : Oxidant-induced vascular endothelial growth factor expression in human keratinocytes and cutaneous wound healing. J Biol Chem, 2002, 277: 33284–33290. [DOI] [PubMed] [Google Scholar]
- 20.Hickey MM, Simon MC: Regulation of angiogenesis by hypoxia and hypoxia-inducible factors. Curr Top Dev Biol, 2006, 76: 217–257. [DOI] [PubMed] [Google Scholar]
- 21.Chen CH, Wang JH, Lu SN, et al. : Characteristics of adefovir resistance in patients with or without lamivudine-resistant hepatitis B virus treated with adefovir: a 4-year experience. Liver Int, 2011, 31: 206–214. [DOI] [PubMed] [Google Scholar]
- 22.Patel V, Chivukula IV, Roy S, et al. : Oxygen: from the benefits of inducing VEGF expression to managing the risk of hyperbaric stress. Antioxid Redox Signal, 2005, 7: 1377–1387. [DOI] [PubMed] [Google Scholar]
- 23.Loo AE, Halliwell B: Effects of hydrogen peroxide in a keratinocyte-fibroblast co-culture model of wound healing. Biochem Biophys Res Commun, 2012, 423: 253–258. [DOI] [PubMed] [Google Scholar]
- 24.O’Toole EA, Marinkovich MP, Peavey CL, et al. : Hypoxia increases human keratinocyte motility on connective tissue. J Clin Invest, 1997, 100: 2881–2891. [DOI] [PMC free article] [PubMed] [Google Scholar]


