Abstract
There are unprecedented epidemics of obesity, such as type II diabetes and non-alcoholic fatty liver diseases (NAFLD) in developed countries. A concerning percentage of American children are being affected by obesity and NAFLD. Studies have suggested that the maternal environment in utero might play a role in the development of these diseases later in life. In this study, we documented that inhibiting SIRT1 signaling in human fetal hepatocytes rapidly led to an increase in intracellular glucose and lipids levels. More importantly, both de novo lipogenesis and gluconeogenesis related genes were upregulated upon SIRT1 inhibition. The AKT/FOXO1 pathway, a major negative regulator of gluconeogenesis, was decreased in the human fetal hepatocytes inhibited for SIRT1, consistent with the higher level of gluconeogenesis. These results indicate that SIRT1 is an important regulator of lipid and carbohydrate metabolisms within human fetal hepatocytes, acting as an adaptive transcriptional response to environmental changes.
Introduction
SIRT1 (Silent mating type information regulation 2 homolog) belongs to the Sirtuins family of proteins and codes for a class III NAD-dependent histone deacetylase (HDACs). In mammals, the functions of SIRT1 have been essentially linked to the regulation of growth, apoptosis, metabolism and aging, responding to environmental cues through NAD+ levels [1–4]. SIRT1 deacetylates a wide range of targets, leading to epigenetic modifications of histones and modulation of transcription factors or metabolic enzymes [3]. Thus, SIRT1 has been thought to be a molecular link between the adaptive transcriptional response and the metabolic status [5, 6]. Recent studies performed on the liver of mice showed the key role of SIRT1 in the development of fatty liver through the regulation of proteins involved in lipid [7, 8] and carbohydrate metabolism [9–11]. These studies have highlighted the potential therapeutic use of SIRT1 in hepatic steatosis.
In recent years, the incidence of non-alcoholic fatty liver diseases (NAFLD) has increased dramatically in developed countries, present in more than 30% of the population in the U.S. It is associated with obesity, insulin resistance, and type II diabetes, and it predisposes the liver to the development of chronic inflammation and oxidative stress [12]. Nonalcoholic steatohepatitis or NASH is the combination of fatty infiltration of hepatocytes with the presence of inflammation. Ballooning degeneration of hepatocytes and increased Mallory’s hyaline inclusions often manifests this injury. Progressive inflammation in both pediatric and adult NASH patients can lead to scarring or fibrosis of the liver and, in severe cases, even cirrhosis and hepatocellular carcinomas (HCC). More importantly, pediatric liver steatosis has increased dramatically in the last 10 years, affecting more than 10% of American children [13]. Pediatric NAFLD is strongly associated with obesity and insulin resistance and its pathogenesis is not yet fully understood [14]. Most worrying is the fact that cardiovascular morbidity in children and teenagers are associated with NAFLD. Thus, understanding the pathogenesis, risk factors, and natural history of fatty liver disease is much needed to prevent youth at risk.
The role played by the environment in the development of NAFLD is very important despite potential genetic susceptibilities. Nicholas Hales and David Barker have proposed the “Thrifty phenotype hypothesis”, which speculate that the maternal nutrition during development may lead to type 2 diabetes, obesity, and the metabolic syndrome in the offspring later in life [15]. There have been several demonstrations of this “fetal origin of adult disease” in rodent models, testing the effect of caloric restriction or high fat diet on the first and second generations of offspring [16–19]. However, only few indications of such demonstrations have been observed in human [20, 21], thus, when and how the human body becomes susceptible to this environment remains unknown. Herein, we have investigated the role of SIRT1 in the lipid and carbohydrate metabolisms of human fetal hepatocytes and showed that a short-span inhibition of this protein lead to an upregulation of de novo lipogenesis and gluconeogenesis pathways in human fetal hepatocytes.
Material and Methods
Fetal human hepatocytes isolation and culture
The de-identified tissues were obtained from Magee Women’s Hospital (Pittsburgh, PA) and the University of Washington Department of Pediatrics, Division of Genetic Medicine, Laboratory of Developmental Biology (Seattle, WA) after obtaining a written informed consent by a protocol approved by the Human Research Review Committee of the University of Pittsburgh (Honest broker approval number HB015 and HB000836). Human fetal hepatocytes were isolated from fetal livers (Table 1) obtained after the termination of pregnancy performed at 20–23 weeks of gestation. Primary human fetal hepatocytes were isolated by digesting the tissue in EMEM (Lonza, Walkersville, MD), which contains 0.5 mg/ml of collagenase (Type XI, Sigma-Aldrich, Saint-Louis MO, Cat. #C7657), on a lab shaker for 40 minutes. Viability was assessed by trypan blue exclusion test and was routinely >85%. Fetal hepatocytes were plated at a density of 1.3x105 cells/cm2 on type I rat tail collagen coated 12 well plates (Corning, Corning, NY). Cells were cultured for 3 days with a DMEM medium (Gibco, Life Technologies, Carlsbad, CA, USA) containing 1X penstrep, 10-7M of insulin (Sigma-Aldrich, Saint-Louis, MO), and 5% bovine serum albumin (Gibco, Life Technologies, Carlsbad, CA, USA). The SIRT1 pharmacological inhibitor (Sirtinol) was purchased from Chayman Chemical (Ann Arbor, MI, USA) and was added to the cells at a concentration of 50uM every 24h from day 0 to 3.
Table 1. Donor Demographics.
| Tissue ID | Gestational age | Sex | Viability |
|---|---|---|---|
| Fetal liver 1 | 20 weeks | Male | 87% |
| Fetal liver 2 | 20 weeks | Female | 88% |
| Fetal liver 3 | 21 weeks | Unknown | 88% |
| Fetal liver 4 | 23 weeks | Female | 88% |
| Fetal liver 5 | 21 weeks | Male | 89% |
| Fetal liver 6 | 23 weeks | Female | 91% |
| Fetal liver 7 | 21 weeks | Female | 87% |
| Fetal liver 8 | 20 weeks | Male | 88% |
RNA extraction, Reverse Transcription and quantitative Polymerase Chain Reactions
Total RNA from the cultured human fetal hepatocytes were extracted using the RNeasy mini kit (Qiagen, Valencia, CA, USA) using the manufacturer's protocol. RNA was quantified by a spectrophotometer to assess the chemical purity and concentration of RNA. 2ug of RNA was mixed with random hexamers (Invitrogen, Carlsbad, CA), heated to 70°C for 5 minutes, and cooled to 4°C. RNA was then reverse transcribed using the Superscript III First-Strand Synthesis System (Invitrogen, Carlsbad, CA), following the manufacturer's protocol. Subsequently, qPCR was performed using TaqMan Fast Advanced Master Mix (Life Technologies, CA, USA) with a StepOnePlus system (Applied Biosystems, Foster City, CA) with the primers from Life Technologies (Carlsbad, CA, USA) described in Table 2. Expression of specific genes was normalized to an internal control Hypoxanthine-guanine phosphoribosyltransferase (HPRT) mRNA expression.
Table 2. List Primers.
| Gene Name | Gene Symbol | Reference (Life technologies) |
|---|---|---|
| acetyl-CoA carboxylase alpha | ACACA | Hs01046047_m1 |
| ELOVL fatty acid elongase 6 | ELOVL6 | Hs00907564_m1 |
| fatty acid synthase | FASN | Hs01005622_m1 |
| glucose-6-phosphatase, catalytic subunit | G6PC | Hs02560787_s1 |
| hypoxanthine phosphoribosyltransferase 1 | HPRT1 | Hs02800695_m1 |
| phosphoenolpyruvate carboxykinase 2 | PCK2 | Hs00388934_m1 |
| stearoyl-CoA desaturase | SCD | Hs01682761_m1 |
| sirtuin 1 | SIRT1 | Hs01009006_m1 |
| sterol regulatory element binding transcription factor 1 | SREBF1 | Hs01088691_m1 |
| acyl-CoA dehydrogenase | ACADM | Hs00936580_m1 |
| acyl-CoA oxidase 1 | ACOX1 | Hs01074241_m1 |
| ATP-binding cassette, sub-family A (ABC1), member 1 | ABCA1 | Hs01059118_m1 |
Metabolic content measurement
Cultured human fetal hepatocytes were washed with PBS and fixed with 4% PFA for 20min. Cells were stained with HCS LipidTOX red neutral lipid stain and Hoechst 33342 (Sigma-Aldrich, Saint-Louis, MO) for 30 min following the manufacturer's protocol. Triglycerides were extracted from fetal hepatocytes by mixing 10% of NP-40 to the cell pellets and by incubating at 80°C for 5min. The triglycerides were measured with a Triglyceride Quantification colorimetric assay (Biovision, Milpitas, CA) according to the manufacturer's instructions. Glucose concentration levels were measured from fetal hepatocytes medium by using the glucose autokit (Wako Chemicals, Richmond, VA).
Histological assessment
Cultured human fetal hepatocytes were washed with PBS and fixed with 4% PFA for 20min. Incubations with primary antibodies (1:100 mouse anti-SIRT1, (Santa Cruz Biotechnology #sc-74504), 1:100 rabbit anti-phospho Ser473-Akt (Cell signaling #4060) and 1:400 rabbit anti-phospho Ser256-FOXO1 (Abcam #ab26651)) were carried out overnight at 4°C. For the detection of the primary antibodies, secondary antibodies (1:250 Alexa Fluor 488 Donkey anti-Mouse IgG (Life technologies CA, USA #A-21202), and 1:250 Alexa Fluor 488 Goat anti-Rabbit IgG (Life technologies, CA, USA #A-11008)) were used followed by counterstaining with 0.2 ug/mL of Hoechst 33342 (Sigma-Aldrich, Saint-Louis, MO). Images were captured using a Zeiss microscope. Acquired images were processed and quantified using ImageJ 1.48v software.
Western Blot
A total of 30 μg of protein was resolved on Tris-HCl precast gels (Bio-Rad Laboratories, Hercules, CA) by SDSPAGE analysis. The proteins were transferred to polyvinylidene difluoride membranes. Membranes were incubated overnight at 4°C with the following primary antibodies: Mouse anti-SIRT1 sc-74504, Rabbit anti-glyceraldehyde-3-phosphate dehydrogenase sc-2577 (Santa Cruz Biotechnology, Santa Cruz, CA), Rabbit Akt #4691S, Rabbit Phospho-Akt-ser473#4060 (Cell signaling, Beverly, MA), Rabbit anti-FOXO1 ab70382, Rabbit Phospho-FOXO1-Ser256 ab26651, Mouse TORC2 ab105932 (Abcam, Cambridge, MA). The signal was amplified either by an anti-mouse or anti-rabbit HRP-conjugated secondary antibody (Millipore, Billerica, MA). Proteins were revealed using the SuperSignal West Pico Chemiluminescent Substrate (Thermo Fisher Scientific, San Jose, CA).
Statistical analyses
All statistical analyses were performed with Prism 6.0 Software (GraphPad Software, Inc., La Jolla, CA) using non-parametric Mann-Whitney test. Data were presented as mean ± SEM, with P < 0.05 considered as statistically significant.
Results
Inhibition of SIRT1 induces an increase in lipids and carbohydrates levels in human fetal hepatocytes
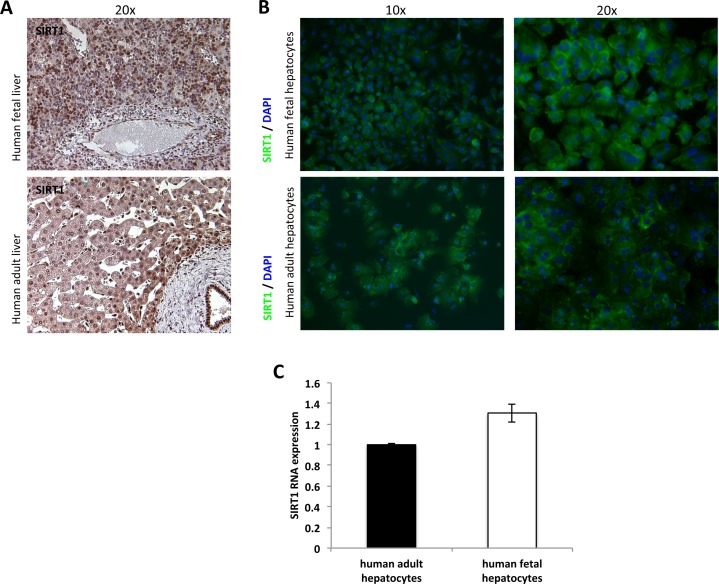
There are numerous studies that address the role of SIRT1 in liver metabolism [8, 22–24]. In mice, it has been shown that the deletion of SIRT1 leads to hepatic steatosis with normal and high fat diet [7, 22]. Moreover, mice models of hepatic steatosis display low levels of SIRT1 [25]. Both the metabolism of carbohydrates and lipids were found to be upregulated in these models. The expression of SIRT1 in adult liver has been described, however, SIRT1 expression and role in human fetal hepatocytes is unknown. Thus, we first assessed the expression of SIRT1 in human fetal hepatocytes. Immune-staining of SIRT1 in fetal and adult liver tissue demonstrated that SIRT1 is expressed in both tissues (Fig 1A). Immune-histological images of adult and fetal hepatocytes in culture also exhibited a clear expression of SIRT1, mainly cytoplasmic with some nuclear localization (Fig 1B). This was further confirmed by western blot analysis of SIRT1 in human fetal hepatocytes (S1 Fig) and with qPCR analysis of SIRT1 expression (Fig 1B). These results strongly indicate that SIRT1 is highly expressed during development in human fetal hepatocytes.
Fig 1. Expression of SIRT1 in human fetal and adult liver and isolated hepatocytes.
(A) Immunohistochemistry of SIRT1 in human fetal and adult liver, (B) Immunofluorescence staining of SIRT1 in fetal and adult hepatocytes after 2 days of culture. (C) Quantification of hepatic SIRT1 mRNA expression measured by qRT- PCR in human fetal and adult hepatocytes in vitro and in vivo (n≥3/group).
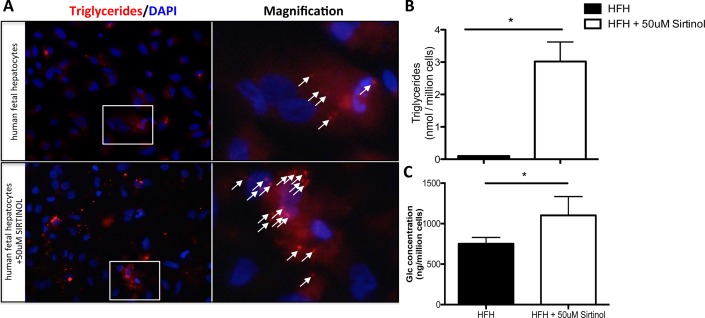
To characterize the role of SIRT1 in lipid and glucose metabolism of human fetal hepatocytes, we used a pharmacological inhibitor of SIRT1, Sirtinol. Human fetal hepatocytes were exposed to 50uM of Sirtinol for three days. Intracellular lipid accumulation was analyzed using a fluorescent probe (Fig 2A). As expected, normal human fetal hepatocytes exhibit almost no intracellular lipid. However, we observed a clear increase in intracellular lipid accumulation when SIRT1 was inhibited. This was further confirmed by measuring the intracellular triglycerides. Human fetal hepatocytes exposed to Sirtinol showed a 30-fold increase in triglycerides accumulation compared to control (Fig 2B). We then measured the intracellular glucose levels. Human fetal hepatocytes inhibited for SIRT1 exhibited a significantly higher level of glucose concentration compared to control (Fig 2C). Altogether, these results showed that Sirtinol exposure leads to an increase in lipid and glucose levels in human fetal hepatocytes after SIRT1 inhibition.
Fig 2. Lipids and glucose levels in human fetal hepatocytes after Sirt1 inhibition.
(A) Fluorescent staining of lipids (red droplets, white arrow) inside human fetal hepatocytes (HFH) with or without 50uM Sirtinol for 3 days (20x). (B) Triglycerides quantification of human fetal hepatocytes with or without +50uM Sirtinol for 3 days analyzed by a colorimetric assay. (C) Glucose concentration between normal HFH and HFH +50uM Sirtinol for 3 days. (n≥7/group; *, P < .05).
Inhibition of SIRT1 leads to an increased de novo lipogenesis in human fetal hepatocytes
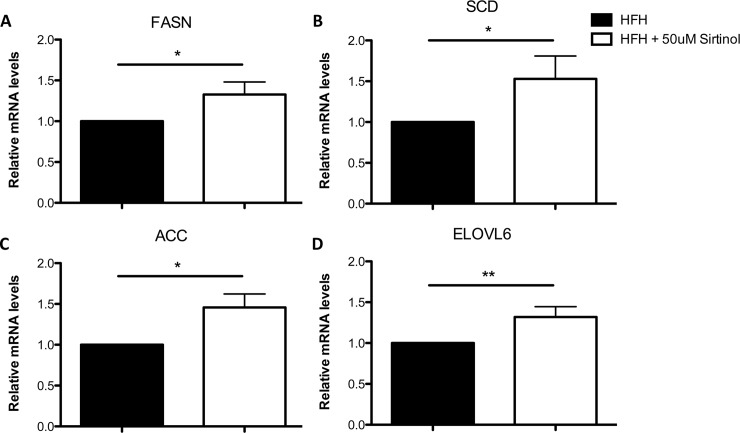
Two potential pathways could explain the increase in lipid content after SIRT1 inhibition: a decrease in β-oxidation and increase in de novo lipogenesis pathway. We examined the mRNA levels of two gene targets of β-oxidation, acyl-CoA dehydrogenase (ACADM) and acyl-CoA oxidase 1 (ACOX1). We could not detect any difference between human fetal hepatocytes with or without Sirtinol (S2A Fig). This can be explained by previous findings reporting that β-oxidation of fetal livers is low. Shortly after birth, exposure of fatty acids through the mother’s milk induces the activation β-oxidation through CPT1-a [26]. Moreover, SIRT1 liver inhibition in rodents has been shown to induce an increase in de novo lipogenesis, leading to hepatic steatosis [7]. Thus, we studied genes involved in de novo lipogenesis by performing qPCR 24h after Sirtinol exposure. We found that all four major genes involved in de novo—fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), stearoyl-CoA desaturase (SCD), and elongase of long chain fatty acids family 6 (ELOVL6)—were upregulated when the cells were exposed to SIRT1 inhibitor (Fig 3A, 3B, 3C and 3D). These findings are very consistent with previous published data showing an increase in de novo lipogenesis associated with SIRT1 inhibition in rodents [7] and explain the rapid increase in lipid production and accumulation of human fetal hepatocytes.
Fig 3. Increased activation of De Novo Lipogenesis related genes in human fetal hepatocytes after Sirt1 inhibition.
Expression of hepatic FASN, SCD, ACC and ELOVL6 mRNA measured by qRT-PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (N≥7/group; *, P < .05)
Inhibition of SIRT1 leads to an increased gluconeogenesis in human fetal hepatocytes through the impairment of AKT/FOXO1 pathway
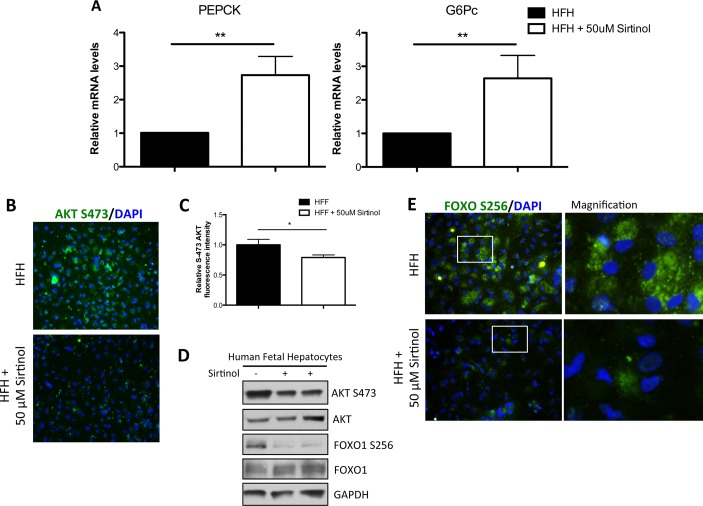
Previous studies have reported controversial results regarding changes in glucose production and use in rodent models with SIRT1-knockout [10, 11]. More recently, we reported that rodents carrying a liver-SIRT1 knockout exhibited hyperglycemia and had an increase in gluconeogenesis, which lead to insulin resistance [9]. Herein, we first investigated gluconeogenesis in human fetal hepatocytes by measuring the transcript of two main gluconeogenetic enzymes: phosphoenolpyruvate carboxykinase (PEPCK) and the catalytic subunit of glucose-6-phosphatase (G6Pc). Both exhibited approximately 3-fold increases, which was consistent with the increase in glucose concentration observed (Fig 4A). Inside the liver, the gluconeogenesis pathway is known to be under the control of AKT/FOXO1 [27]. Forkhead box O1 (FOXO1) usually enters the nucleus and eventually leads to the activation of genes linked to gluconeogenesis. However, the activation of AKT induces the phosphorylation of FOXO1, which lead to the exclusion of this protein out of the nucleus to the cytoplasm. This system controls the level of gluconeogenesis inside the cell [27]. Thus, to further explain the upregulaltion of the gluconeogenesis pathway, we first checked the activation of AKT at serine 473 through immunofluorescence staining of human fetal hepatocytes. AKT phosphorylation was found in both cells, but to a lower extend when Sirtinol was present (Fig 4B and 4C). Western blots analysis corroborated the decrease of AKT-S473 activation in human fetal hepatocytes inhibited for SIRT1 (Fig 4D). We then measured the phosphorylation of FOXO1, downstream of AKT. FOXO1 S256 decreases in human fetal hepatocytes with Sirtinol, which indicate a higher FOXO1 activity inside the nucleus (Fig 4D). As expected, immunofluorescence staining of FOXO S256 exhibited a cytoplasmic localization of the protein in both groups of human fetal hepatocytes (Fig 4E). Overall, these data demonstrates that the inhibition of SIRT1 causes an increase in gluconeogenesis, leading to a higher intracellular glucose concentration in human fetal hepatocytes, through the control of AKT/FOXO1 pathway.
Fig 4. Increased activation of Glucogenesis pathway in human fetal hepatocytes after exposure to Sirtinol.
(A) Expression of hepatic PEPCK and G6PC mRNA measured by qRT- PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (B) Immunofluorescence of S-473 AKT in human fetal hepatocytes with or without 50uM Sirtinol (10x). (C) Fluorescence intensity quantification for S-473 AKT signal (arbitrary units) using ImageJ (n≥7/group; *, P < .05). (D) Western blot analysis for S-473 AKT/AKT, S-256 FOXO1/FOXO1 in human fetal hepatocytes exposed to 50uM Sirtinol compared to controls. GAPDH was used as a loading control. (E) Immunofluorescence staining of S-256 FOXO1 in human fetal hepatocytes with or without 50uM Sirtinol (20x).
Discussion
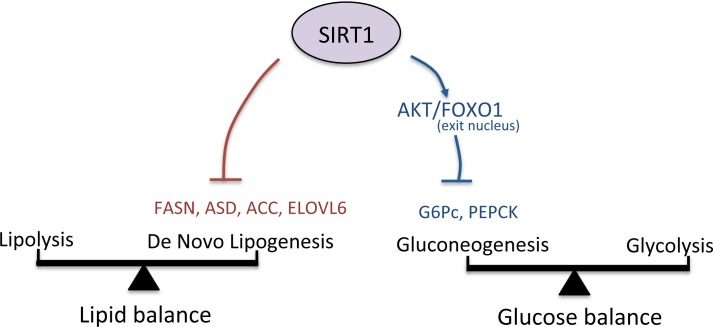
In the recent years, researchers have just started exploring and understanding the importance of SIRT1 in metabolism. SIRT1 acts as a metabolic “switch” linked to hepatic steatosis. To the best of our knowledge, this is the first report that showed the implication of SIRT1 in metabolic processes in human fetal hepatocytes. In this study, we observed that the inhibition of SIRT1 using a pharmacological agent for a short period of time leads to a rapid increase in lipid and carbohydrate concentrations. Moreover, we have confirmed a negative regulation of SIRT1 in de novo lipogenesis and gluconeogenesis through the AKT/FOXO1 pathway in human fetal hepatocytes. These results are coherent with previously studies on mice carrying a deletion of SIRT1 in the liver and developing liver steatosis. These results also show that human fetal hepatocytes are responsive to SIRT1 signals and emphasize SIRT1 role as a regulator of lipid and glucose balances in the liver (Fig 5). Altogether, this study confirmed the extensive role of SIRT1 in hepatocytes metabolism during development.
Fig 5. Representation of SIRT1 regulation of lipid and glucose balances in human fetal hepatocytes.
In normal conditions, SIRT1 inhibits De Novo Lipogenesis and Gluconeogenesis through the AKT/FOXO1 pathway inside human fetal hepatocytes, keeping a normal balance inside the cells. Upon SIRT1 inhibition, both the lipid and glucose balance will be disrupted, leading to an increase of lipid and carbohydrates levels in human fetal hepatocytes.
Previous studies have already documented an increase in lipogenesis and gluconeogenesis during fetal development [28, 29]. The maternal diet (fructose diet and high fat diet) imposed these metabolic changes on the fetus. It suggests that changes in the maternal diet can provide more nutrients availability, used as energy blocks by the fetus to sustain these metabolic pathways. Similarly, exposure to certain factors (such as a high fat diet) might affect SIRT1 in the liver and induce metabolic changes during development. In the long run (after birth), an increase production of lipids could render these offsprings more susceptible to the development of hepatic steatosis and hence hyperglycemia that is linked to insulin resistance and diabetes [30, 31]. Consequently, more sophisticated models to study human tissues overtime (infants and adults) are needed to address questions regarding the important role of SIRT1 and the molecular mechanistic upstream and downstream.
Our future studies will investigate which signaling pathways under SIRT1 influence the lipid metabolism on these cells. For instance, SIRT1 has been shown to deacetylate Liver X Receptor (LXR), which is a regulator of lipid homeostasis [32]. However, we were not able to detect differences in LXR gene targets: ATP-binding cassette, sub-family A (ABC1), member 1 or Srebp-1c (S2B Fig). This suggests that SIRT1 may acts through another pathway in human fetal hepatocytes. In the other hand, AKT/FOXO1 pathway is a major regulator of glucose production in response to insulin, nevertheless, it has been shown that SIRT1 represses gluconeogenesis during fasting, through CREB-regulated transcription coactivator 2 (CRTC2) deacetylation, leading to its degradation [33]. However, we did not find significant changes in CRTC2 expression when SIRT1 was inhibited (S2C Fig). These results support the main role of AKT/FOXO1 pathway to regulate gluconeogenesis under the control of SIRT1 in human fetal hepatocytes.
Numerous rodent studies have shown that the parental dietary may affect the development of diseases later in life, such as diabetes or obesity, by changing the epigenetic signature of the offspring [34]. Recently, studies performed on animal models reported that maternal high fat diet and obesity resulted in a decrease of SIRT1 mRNA in fetal livers [35, 36], highlighting the role of intrauterine environment on SIRT1 expression. However, animal models do not mimic the pathogenesis of human metabolic diseases and further studies on human tissues will be required to determine what controls SIRT1. It will be interesting to determine whether SIRT1 acts as an adaptive transcriptional response during human development. In this respect, SIRT1 could respond to external environmental cues such as maternal obesity and create the conditions to develop NAFLD. A better understanding of the central position of SIRT1 in hepatic metabolism will help in the comprehension of the metabolic diseases and will provide valuable information to prevent and treat human metabolic abnormalities.
Supporting Information
Western blot analysis of SIRT1 in human fetal liver cells (fibroblasts and hepatocytes). A human fetal fibroblasts cell line carrying a shMIR-SIRT1 activated upon doxycycline addition was used to assess the specificity of SIRT1 expression.
(TIF)
(A) Expression of hepatic ACOX1 and ACADM mRNA measured by qRT- PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (B) Expression of hepatic ABCA1 and SREBP-1C mRNA expression measured by qRT- PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (n≥7/group; *, P < .05) (C) Western blot analysis for CRCT2 in human fetal hepatocytes exposed to +50uM Sirtinol. GAPDH was used as a control.
(TIF)
Acknowledgments
Funding from the US National Institutes of Health (DK099257 to A.S.-G., UH3TR000503 and ES022606 subcontract to A.S.-G.), and the Competitive Medical Research Fund Program from UPMC Health System to A.S.-G. Laboratory of Developmental Biology was supported by NIH Award Number 5R24HD000836 from the Eunice Kennedy Shriver National Institute of Child Health & Human Development.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
Funding from the US National Institutes of Health (DK099257 to A.S.-G., UH3TR000503 and ES022606 subcontract to A.S.-G.), and the Competitive Medical Research Fund Program from UPMC Health System to A.S.-G. Laboratory of Developmental Biology was supported by NIH Award Number 5R24HD000836 from the Eunice Kennedy Shriver National Institute of Child Health & Human Development.
References
- 1.Bordone L, Guarente L. Calorie restriction, SIRT1 and metabolism: understanding longevity. Nat Rev Mol Cell Biol. 2005;6(4):298–305. 10.1038/nrm1616 . [DOI] [PubMed] [Google Scholar]
- 2.Christopher L. Brooks WG. How does SIRT1 affect metabolism, senescence and cancer? Nature Reviews Cancer. 2009;9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schug TT, Li X. Sirtuin 1 in lipid metabolism and obesity. Ann Med. 2011;43(3):198–211. Epub 2011/02/25. 10.3109/07853890.2010.547211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chang HC, Guarente L. SIRT1 and other sirtuins in metabolism. Trends Endocrinol Metab. 2014;25(3):138–45. Epub 2014/01/07. 10.1016/j.tem.2013.12.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rodgers JT, Lerin C, Gerhart-Hines Z, Puigserver P. Metabolic adaptations through the PGC-1 alpha and SIRT1 pathways. FEBS Lett. 2008;582(1):46–53. 10.1016/j.febslet.2007.11.034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Yamamoto M, Iguchi G, Fukuoka H, Suda K, Bando H, Takahashi M, et al. SIRT1 regulates adaptive response of the growth hormone–insulin-like growth factor-I axis under fasting conditions in liver. PNAS. 2013;110(37):14948–53. 10.1073/pnas.1220606110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang RH, Li C, Deng CX. Liver Steatosis and Increased ChREBP Expression in Mice Carrying a Liver Specific SIRT1 Null Mutation under a Normal Feeding Condition. International Journal of Biological Sciences. 2010;6(7):682–90. PubMed Central PMCID: PMC21103071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Purushotham A, Schug TT, Xu Q, Surapureddi S, Guo X, Li X. Hepatocyte-specific deletion of SIRT1 alters fatty acid metabolism and results in hepatic steatosis and inflammation. Cell Metab. 2009;9(4):327–38. Epub 2009/04/10. 10.1016/j.cmet.2009.02.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang RH, Kim HS, Xiao C, Xu X, Gavrilova O, Deng CX. Hepatic Sirt1 deficiency in mice impairs mTorc2/Akt signaling and results in hyperglycemia, oxidative damage, and insulin resistance. J Clin Invest. 2011;121(11):4477–90. Epub 2011/10/04. 10.1172/JCI46243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Joseph T. Ridgers CL, Wilhem Haas, Gygl Steven P., Bruce M. Spiegelman, Pere Pulgserver. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature. 2005;434(7029):113–8. doi: 10.1038/nature03314 . [DOI] [PubMed] [Google Scholar]
- 11.Erion DM, Yonemitsu S, Nie Y, Nagai Y, Gillum MP, Hsiao JJ, et al. SirT1 knockdown in liver decreases basal hepatic glucose production and increases hepatic insulin responsiveness in diabetic rats. Proc Natl Acad Sci U S A. 2009;106(27):11288–93. 10.1073/pnas.0812931106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cohen JC, Horton JD, Hobbs HH. Human fatty liver disease: old questions and new insights. Science. 2011;332(6037):1519–23. Epub 2011/06/28. 10.1126/science.1204265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Alisi A, Feldstein AE, Villani A, Raponi M, Nobili V. Pediatric nonalcoholic fatty liver disease: a multidisciplinary approach. Nat Rev Gastroenterol Hepatol. 2012;9(3):152–61. 10.1038/nrgastro.2011.273 . [DOI] [PubMed] [Google Scholar]
- 14.Nobili V, Alkhouri N, Alisi A, Della Corte C, Fitzpatrick E, Raponi M, et al. Nonalcoholic fatty liver disease: a challenge for pediatricians. JAMA Pediatr. 2015;169(2):170–6. Epub 2014/12/17. 10.1001/jamapediatrics.2014.2702 . [DOI] [PubMed] [Google Scholar]
- 15.Hales CN, Barker DJP. The thrifty phenotype hypothesis. British Medical Bulletin. 2001;60:5–20. [DOI] [PubMed] [Google Scholar]
- 16.Brenseke B, Bahamonde J, Talanian M, Kornfeind E, Daly J, Cobb G, et al. Mitigating or exacerbating effects of maternal-fetal programming of female mice through the food choice environment. Endocrinology. 2015;156(1):182–92. Epub 2014/11/12. 10.1210/en.2014-1523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fernandez-Twinn DS, Wayman A, Ekizoglou S, Martin MS, Hales CN, Ozanne SE. Maternal protein restriction leads to hyperinsulinemia and reduced insulin-signaling protein expression in 21-mo-old female rat offspring. Am J Physiol Regul Integr Comp Physiol. 2005;288(2):R368–73. Epub 2004/10/30. 10.1152/ajpregu.00206.2004 . [DOI] [PubMed] [Google Scholar]
- 18.Ozanne SE, Olsen GS, Hansen LL, Tingey KJ, Nave BT, Wang BT, et al. Early growth restriction leads to down regulation of protein kinase C zeta and insulin resistance in skeletal muscle. Journal of Endocrinology. 2003;177:235–41. [DOI] [PubMed] [Google Scholar]
- 19.Clive J. Petry MWD, Pawlak Dorota B., Ozanne Susan E. and Hales C. Nicholas. Diabetes in Old Male Offspring of Rat Dams Fed a Reduced Protein Diet. International Journal of Experimental Diabetes Research. 2001;2:139–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ravelli ACJ, van der Meulen JHP, Michels RPJ, Osmond C, Barker DJP, Hales CN, et al. Glucose tolerance in adults after prenatal exposure to famine. The Lancet. 1998;351(9097):173–7. 10.1016/s0140-6736(97)07244-9 [DOI] [PubMed] [Google Scholar]
- 21.Valdez R, Athens MA, Thompson GH, Bradshaw BS, Stern MP. Birthweight and adult health outcomes in a biethnic population in the USA. Diabetologia. 1994;37(6):624–31. [DOI] [PubMed] [Google Scholar]
- 22.Li Y, Xu S, Giles A, Nakamura K, Lee JW, Hou X, et al. Hepatic overexpression of SIRT1 in mice attenuates endoplasmic reticulum stress and insulin resistance in the liver. FASEB J. 2011;25(5):1664–79. Epub 2011/02/16. 10.1096/fj.10-173492 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Herranz D, Munoz-Martin M, Canamero M, Mulero F, Martinez-Pastor B, Fernandez-Capetillo O, et al. Sirt1 improves healthy ageing and protects from metabolic syndrome-associated cancer. Nat Commun. 2010;1:3 10.1038/ncomms1001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pfluger PT, Herranz D, Velasco-Miguel S, Serrano M, Tschop MH. Sirt1 protects against high-fat diet-induced metabolic damage. Proc Natl Acad Sci U S A. 2008;105(28):9793–8. 10.1073/pnas.0802917105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Deng XQ, Chen LL, Li NX. The expression of SIRT1 in nonalcoholic fatty liver disease induced by high-fat diet in rats. Liver Int. 2007;27(5):708–15. Epub 2007/05/15. 10.1111/j.1478-3231.2007.01497.x . [DOI] [PubMed] [Google Scholar]
- 26.Chatelain F, Kohl C, Esser V, McGarry JD, Girard J, Pegorier JP. Cyclic AMP and fatty acids increase carnitine palmitoyltransferase I gene transcription in cultured fetal rat hepatocytes. European Journal of Biochemistry. 1996;235:789–98. [DOI] [PubMed] [Google Scholar]
- 27.Puigserver P, Rhee J, Donovan J, Walkey CJ, Yoon JC, Oriente F, et al. Insulin-regulated hepatic gluconeogenesis through FOXO1–PGC-1a interaction. Nature. 2003;423:550–5. [DOI] [PubMed] [Google Scholar]
- 28.Clayton ZE, Vickers MH, Bernal A, Yap C, Sloboda DM. Early Life Exposure to Fructose Alters Maternal, Fetal and Neonatal Hepatic Gene Expression and Leads to Sex-Dependent Changes in Lipid Metabolism in Rat Offspring. PLoS One. 2015;10(11). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McCurdy CE, Bishop JM, Williams SM, Grayson BE, Smith MS, Friedman JE, et al. Maternal high-fat diet triggers lipotoxicity in the fetal livers of nonhuman primates. The Journal of Clinical Investigation. 119(2):323–35. 10.1172/JCI32661 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Browning JD, Horton JD. Molecular mediators of hepatic steatosis and liver injury. Journal of Clinical Investigation. 2004;114(2):147–52. 10.1172/jci200422422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Muoio DM, Newgard CB. Mechanisms of disease:Molecular and metabolic mechanisms of insulin resistance and beta-cell failure in type 2 diabetes. Nat Rev Mol Cell Biol. 2008;9(3):193–205. 10.1038/nrm2327 . [DOI] [PubMed] [Google Scholar]
- 32.Li X, Zhang S, Blander G, Tse JG, Krieger M, Guarente L. SIRT1 Deacetylates and Positively Regulates the Nuclear Receptor LXR. Molecular Cell. 2007;12(28):91–106. [DOI] [PubMed] [Google Scholar]
- 33.Liu Y, Dentin R, Chen D, Hedrick S, Ravnskjaer K, Schenk S, et al. A Fasting Inducible Switch Modulates Gluconeogenesis Via Activator-Coactivator Exchange. Nature. 2008;456(7219):269–73. 10.1038/nature07349 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rando OJ, Simmons RA. I'm eating for two: parental dietary effects on offspring metabolism. Cell. 2015;161(1):93–105. 10.1016/j.cell.2015.02.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Borengasser SJ, Kang P, Faske J, Gomez-Acevedo H, Blackburn ML, Badger TM, et al. High fat diet and in utero exposure to maternal obesity disrupts circadian rhythm and leads to metabolic programming of liver in rat offspring. PLoS One. 2014;9(1):e84209 10.1371/journal.pone.0084209 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Suter MA, Chen A, Burdine MS, Choudhury M, Harris RA, Lane RH, et al. A maternal high-fat diet modulates fetal SIRT1 histone and protein deacetylase activity in nonhuman primates. FASEB J. 2012;26(12):5106–14. 10.1096/fj.12-212878 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Western blot analysis of SIRT1 in human fetal liver cells (fibroblasts and hepatocytes). A human fetal fibroblasts cell line carrying a shMIR-SIRT1 activated upon doxycycline addition was used to assess the specificity of SIRT1 expression.
(TIF)
(A) Expression of hepatic ACOX1 and ACADM mRNA measured by qRT- PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (B) Expression of hepatic ABCA1 and SREBP-1C mRNA expression measured by qRT- PCR in human fetal hepatocytes exposed to +50uM Sirtinol compared to controls. (n≥7/group; *, P < .05) (C) Western blot analysis for CRCT2 in human fetal hepatocytes exposed to +50uM Sirtinol. GAPDH was used as a control.
(TIF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.