Abstract
Bipolar disorder is prevalent, with high risks of disability, substance abuse and premature mortality. Treatment responses typically are incomplete, especially for depressive components, so that many cases can be considered “treatment resistant.” We reviewed reports on experimental treatments for such patients: there is a striking paucity of such research, mainly involving small incompletely controlled trials of add-on treatment, and findings remain preliminary. Encouraging results have been reported by adding aripiprazole, bupropion, clozapine, ketamine, memantine, pramipexole, pregabalin, and perhaps tri-iodothyronine in resistant manic or depressive phases. The urgency of incomplete responses in such a severe illness underscores the need for more systematic, simpler, and better controlled studies in more homogeneous samples of patients.
Keywords: Bipolar disorder, depression, experimental treatments, mania, treatment-resistance.
INTRODUCTION
Bipolar disorder is a persistent, episodic and debilitating condition with an estimated lifetime prevalence of over 2.0%, including both types I (with mania) and II (with hypomania) [1, 2]. Bipolar disorder is associated with recurring episodes of mania, hypomania, mixed manic-depressive states, or psychosis, as well as prominent major depression and dysthymia, as well as prevalent anxiety symptoms—all leading to high risks of potentially severe functional impairment, substance abuse, and high rates of suicide, accidents, and increased mortality from co-occurring medical illnesses—all despite use of available pharmacological and psychosocial treatments [1, 3-8]. The depressive components of the disorder have been especially difficult to treat successfully and they account for three-quarters of the nearly 50% of weeks of follow-up with treatment that include clinically significant residual morbidity [3, 9].
Consensus guidelines and expert recommendations usually advocate use of monotherapy in the treatment of bipolar disorder patients whenever possible, with adjunctive therapy indicated when a patient relapses on maintenance treatment [5, 10, 11]. In reality, unsatisfactory responses to available treatments for bipolar disorder are very prevalent, especially for bipolar depression, and empirical use of various, largely untested, combinations of treatments is the rule [5, 12-14]. Clinical responses that are particularly poor are often labeled as evidence of “treatment resistance,” although the term is defined, imprecisely, by varied numbers and types of treatment trials, responses, and periods of observation [9, 15-18].
We have proposed a working definition of treatment resistance as involving responses considered clinically unsatisfactory following at least two trials of dissimilar medicinal treatments in presumably adequate doses and durations, within a specific phase of bipolar illness (manic, depressive, or mixed), or for “breakthrough” symptoms that emerge despite previous apparently effective maintenance treatment, and excluding patients who are intolerant of a treatment regimen and, to the extent possible, those who are not adherent to recommended treatment [19]. The present overview considers experimental interventions for treatment resistance found in any phase of bipolar disorder, as indicated by clinically unsatisfactory responses to current treatments based on accepted community standards and on expert guidelines and recommendations, as cited above.
METHODS
We searched the digitized medical research literature for reports related to pharmacological treatments of treatment-resistance in bipolar disorder patients using the MedLine/PubMed database of the U.S. National Center for Biotechnology Information (NCBI; http://www.ncbi.nlm.nih.gov/entrez/query.fcgi), and limiting the search to reports in English. We used the following search terms in various combinations: bipolar, treatment, drug or medication resistant, resistance, or refractory, and difficult to treat. We initially considered reports of meta-analyses, systematic reviews, randomized controlled trials (RCTs), naturalistic and retrospective studies, case series, and case reports. Hand-searching further considered references cited in reports initially identified by computer-searching. Authors reviewed the abstracts of identified reports, and full reports of articles that met entry criteria were reviewed in detail by at least two authors, who extracted relevant details and resolved disagreements by consensus. Minimal entry criteria included study subjects diagnosed with a bipolar disorder based on an international diagnostic standard, usually the American Psychiatric Association Diagnostic and Statistical Manual of Mental Disorders (DSM, editions III, IV, or -5), or the World Health Organization’s International Classification of Diseases (ICD, editions 9 or 10). In view of the paucity of reports on this topic, their consideration did not require specific numbers of subjects, randomization, or controls. Table 1 includes studies that specified trials in treatment-resistant subjects, but the text includes some additional studies with interesting leads developed among bipolar disorder patients who were not necessarily treatment-resistant.
Table 1.
Therapeutic trials for treatment-resistant bipolar disorder.
| Report | Design (Level of Evidence) | Treatment Resistance | Failed Trials | Experimental Treatments | Subjects | Females (%) | Age (yrs) | Onset Age | Treated (mos) | Findings | Comments | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Antipsychotics | |||||||||||||||
| Clozapine (CLZ) | |||||||||||||||
| Banov et al. 1994 [21] | Chart review (4) | ––– | ––– | ––– | 52 BD 81 SzAff 14 UP 40 Sz 6 BD-NOS | ––– | ––– | ––– | 18.7 | Best for mania; better social function; discontinue if ≥1 prior depression | CLZ effective | ||||
| Calabrese et al. 1996 [22] | Open, Prospective (4) | Failed Li, ACs + ≥2 APs | >3 | CLZ only | 10 BD 15 SzAff | ––– | ––– | ––– | 3.25 | Improvements: 72% (YMRS), 32% (BPRS) | CLZ effective (esp. in BD) | ||||
| Suppes et al. 1999 [23] | Open, Prospective (4) | Failed 2 MSs | ≥2 | Add CLZ | 26 BD-I 12 SzAff | 58.0 | 38.0 | 17.0 | 12.0 | Improvements: BPRS, CGI, BRMS | CLZ effective | ||||
| Ciapparelli et al. 2003 [24] | Open, prospective (4) | ––– | ––– | ––– | 34 Sz 30 SzAff 37 BD | ––– | ––– | ––– | ––– | Improvements: (BPRS, GAF, functioning), more in BD & SzAff | CLZ effective | ||||
| Chang et al. 2006 [25] | Chart review (4) | ––– | ––– | ––– | 51 BD | ––– | ––– | ––– | ––– | Fewer hospital days in 90%; fewer episodes (all types) | CLZ effective | ||||
| Aripiprazole (APZ) | |||||||||||||||
| Ketter et al. 2006 [31] | Open, Prospective (4) | ––– | ––– | Add APZ (15.3mg/d) (3.2 Rxs/case) | 11 BD-I 15 BD-II 4 BD-NOS | 70.0 | 44.4 | 21.5 | 2.80 | Improvements: CGI, GAF; 13% remit | APZ effective | ||||
| Kemp et al. 2007 [32] | Open, Prospective (4) | Failed ≥12 wks (MSs + ADs) | >2 | Add APZ (15 mg/d) to APs (75%), ADs (67%), AEDs (50%); 3.7 Rxs/case | 4 BD-I 7 BD-II 1 BD-NOS | 58.3 | 48.3 | ––– | 2.00 | 33% improved ≥50% (MADRS) | ––– | ||||
| Benedetti et al. 2010 [33] | Open, Prospective (4) | Failed ≥2 MSs or APs | ≥2 | Add APZ (6.8 mg/d) to CLZ (293 mg/d) | 1 BD 6 SzAff | 57.1 | 39.0 | ––– | 0.50 | Improved psychosis (BPRStotal+thought disorder+anergy) | APZ+CLZ effective, not more ADRs | ||||
| Olanzapine (OLZ) | |||||||||||||||
| Vieta et al. 2001 [35] | Open, Prospective (4) | Failed Li & ≥1 MS (CBZ±VPA, 6mos) | ≥2 | Add OLZ (12 mg/d) to: Li (70%), AEDs (56%), ADs (48%), APs (31%), ±others (82%) | 23 BD | 43.5 | 39.9 | 24.2 | 10.8 | 100% improved (CGI) | ––– | ||||
| Chen et al. 2011 [34] | Open, prospective (4) | Failed ≥2 MSs or APs (no OLZ) | ≥2 | OLZ only (5–40 mg/d) | 18 BD I | 38.9 | 44.4 | ––– | 3.00 | 88%: ≥50% reduction (YMRS), 78%: remission | OLZ effective | ||||
| Antipsychotics | |||||||||||||||
| Quetiapine (QTP) | |||||||||||||||
| Ahn et al. 2011 [36] | Open, prospective (4) | Failed QTP or LTG + other Rxs | ≥2 | QTP (188 mg/d) to: LTG (228 mg/d) or LTG (204 mg/d) to QTP (208 mg/d) ± Other Rxs | 15 BD-I 22 BD-II 1 BP-NOS | 73.7 | 40.1 | 18.4 | 3.00 | 100% improved (CGI, GAF) 18% dropped out (side-effects) 21% required other Rxs | ––– | ||||
| Anticonvulsants | |||||||||||||||
| Eslicarbazepine (sLIC) | |||||||||||||||
| Nath et al. 2012 [38] | Case report (4) | ––– | –– | Add sLIC (800 mg/d) to QTP | 1 BD | 0 | ––– | ––– | 6.00 | Improved (YMRS) ≤3 wks; remit 6 mos | ––– | ||||
| Pregabalin (PGB) | |||||||||||||||
| Conesa et al. 2012 [43] | Case report (4) | Failed multiple trials (MSs & APs) | ––– | Add PGB (225mg/d) to VPA, HAL, CLZ | 1 BD | 0 | 46.0 | 20.0 | 10.0 | Psychosis+ mood remits; more illness-aware | Rapid response | ||||
| Schaffer et al. 2013 [44] | Open, Prospective (4) | ––– | ––– | Add PGB (to 3.3 Rxs/case) | 58 BD | 79.0 | 47.0 | ––– | 18.0 + 36.0 | 41% respond; 10% stable 36 mos | PGB safe & effective | ||||
| Topiramate (TPM) | |||||||||||||||
| Vieta et al. 2002 [51] | Open, Prospective (4) | Failed >2 trials (MSs ± others) | ≥2 | Add TPM (202 mg/d) | 28 BD-I 3 BD-II 2 BD-NOS 1 SzAff | 67.6 | 42.0 | ––– | 6.0 | 58% improved ≥50% (YMRS, HDRS, CGI); 44% fewer episodes | ––– | ||||
| Antidepressants | |||||||||||||||
| Bupropion (BUP) | |||||||||||||||
| Erfurth et al. 2002 [16] | Open, Prospective 94) | Failed >2 trials (MS+AD) | ≥2 | Add BUP to: ADs (92%), APs (15%), Li (8%), ACs (31%) (1.5 Rxs/case) | 7 BD 4 UP | 61.5 | 48.4 | ––– | 1.0 | 64% improved ≥50% (MADRS) | No manic switch | ||||
| Glutamate antagonists | |||||||||||||||
| Ketamine (KTM) | |||||||||||||||
| Diazgranados et al. 2010 [65] | Randomized v PBO, blinded add-on (1) | Failed ≥1 trial (AD + MS) | ≥1 | Add KTM v PBO crossover | 18 BD | 66.7 | 47.9 | 20.3 | 0.5 | ≥50% improved MADRS: KTM: 71%, PBO: 6% | KTM safe & effective | ||||
| Cusin et al. 2012 [66] | Case reports (4) | Failed multiple trials | >3 | Add IM KTM | 2 BD | 100 | 52.5 | ––– | 5.5 | Improved symptoms & function | ––– | ||||
| Glutamate antagonists | |||||||||||||||
| Ketamine (KTM) | |||||||||||||||
| Zarate et al. 2012 [63] | Randomized v PBO (1) | Failed ≥1 trial of AD & MS | >2 | IV KTM v PBO 2 wks crossover | 9 BD-I 6 BD-II | 53.3 | 46.7 | 16.1 | 0.5 | ≥50% improved MADRS: KTM 79% v PBO 0%; (43% in 1 d) | ––– | ||||
| Lara et al. 2013 [67] | Case series (4) | Failed ≥4 mono- or combination treatments | ≥4 | Add SL KTM (10 mg every 2–7 d) | 2 BD-I 12 BD-II 12 MDD | 69.2 | 45.2 | ––– | ––– | Mood improved: 77% (31% after 1 dose) | KTM well tolerated | ||||
| Memantine (MEM) | |||||||||||||||
| Agarwal & Tripathi 2009 [68] | Case report (4) | Failed multiple trials | ≥2 | Add MEM (10mg/d) to VPA (1500mg/d) & CLZ (350mg/d) | 1 BD | 0 | 42.0 | ––– | 1.5 | Achieved remission | MEM effective | ||||
| Koulopoulos et al. 2010 [69] | Open, Prospective (4) | Failed ≥2 trials (MS or APDs) | ≥2 | Add MEM (10–30 mg/d) | 18 BD | 77.8 | 42.0 | ––– | 6.0 | 72% very much improved (CGI) | MEM effective | ||||
| Koulopoulos et al. 2012 [70] | Open, Prospective (4) | Failed ≥2 trials (MS or APDs) | ≥2 | Add MEM (10–30mg/d) | 40 BD | ––– | 49.0 | ––– | 12.0 | 73% very much improved (CGI) | MEM effective | ||||
| Serra et al. 2014 [71] | Open, mirror-image (4) | Failed ≥2 trials over 3 yrs | >2 | Add MEM (20–30 mg/d) | 17 BD-I 13 BD-II | 70.0 | 46.9 | ––– | 36.0 | Marked improvements: CGI, episodes | MEM effective; no switches | ||||
| Cholinesterase Inhibitor | |||||||||||||||
| Donepezil (DPZ) | |||||||||||||||
| Burt et al. 1999 [73] | Chart review (4) | Failed ≥2 trials (MSs or ADs) | ≥2 | Add DPZ | 11 BD | 63.6 | 39.2 | ––– | ––– | 54% responded; 27% slightly improved (CGI) | ––– | ||||
| Evins et al. 2006 [74] | Randomized v PBO (1) | YMRS ≥15 after trial of Li, VPA or CBZ ≥2wks | ≥1 | Add DPZ (5–10 mg/d) v PBO | 11 BD | 81.8 | 39.0 | ––– | 3.5 | Not improved | DPZ ineffective | ||||
| Dopamine agonists | |||||||||||||||
| Pramipexole (PPX) | |||||||||||||||
| Sporn et al. 2000 [83] | Chart review (4) | ––– | -–– | Add PPX (0.7mg/d) | 12 BD 20 UP | ––– | ––– | ––– | 6.1 | Effective in 50% of BD (CGI) | 1 transient hypomania | ||||
| Perugi et al. 2001 [84] | Chart review (4) | Failed ≥8 wks MSs+ADs | ––– | Add PPX (0.75-1.5 mg/d) or RPN (1.5–5.0 mg/d) | 18 BD-II | ––– | ––– | ––– | 4.4 | 40% responded (CGI) | ––– | ||||
| Cassano et al. 2004 [85] | Open, Prospective (4) | Failed ≥4wks AD | ≥1 | Add PPX (0.75-1.5mg/d) | 2 BD-I 9 BD-II 12 UP | 69.6 | 52.8 | 35.1 | 7.0 | 60.9% remit: MADRS, CGI | 2 switches | ||||
| Dopamine agonists | |||||||||||||||
| Pramipexole (PPX) | |||||||||||||||
| Goldberg et al. 2004 [82] | Randomized v PBO (1) | Failed ≥2 trials (ADs+MSs) | >2 | Add PPX (1.7mg/d) or PBO to: AEDs (91%), Li (27%); (1.2 Rxs/case) | 22 BD | 50.0 | 42.1 | ––– | 1.5 | Improved ≥50% (HDRS): 67% PPX, 20% PBO | 1 switch | ||||
| Ropinirole (RPN) | |||||||||||||||
| Perugi et al. 2001 [84] | Chart review (4) | Failed ≥8w (MSs + ADs) | ––– | Add RPN (1.5–5.0 or PPX (0.75–1.5 mg/d) | 18 BD-II | ––– | ––– | ––– | 4.4 | 40% responded (CGI) | ––– | ||||
| Psychostimulants | |||||||||||||||
| Modafinil (MOD) | |||||||||||||||
| Dell’Osso et al. 2013 [86] | Chart review (4) | Residual depressive symptoms | ––– | Add MOD (38%) or PPX (62%) | 27 BD-I 28 BD-II 8 BD-NOS | 60.3 | 43.5 | 18.6 | 19.0 | Intolerability: MOD<PPX (by 3x) | MOD better-tolerated | ||||
| Parker & Brotchie 2010 [89] | Case series (4) | ––– | ––– | Add (30) or only treatment (20): methyphenidate or d-amphetamine | 27 BD 23 UP | ––– | 49.1 | ––– | 14.2 | 34% improved | No ratings; 18%: ADRs | ||||
| Calcium Channel Blocker | |||||||||||||||
| Diltiazem (DLT) | |||||||||||||||
| Silverstone & Birkett 2000 [97] | Open, mirror-image (4) | Failed >2 trials (MS±others) | ≥2 | Add DLT to Li (25%), AC (88%), ADs (1.7/case), AP (12%) | 8 BD | 100 | ––– | ––– | 6.0 +6.0 | Improvements noted | ––– | ||||
| Opioid | |||||||||||||||
| Oxycodone (OXC) | |||||||||||||||
| Schiffman & Gitlin 2012 [100] | Case report (4) | Failed ≥2 trials | ––– | Add OXC (50 µg/d) to MS, AD or AP | 1 BD | 100 | 54.0 | ––– | 12.0 | Improved (1 wk), remitted ≤1 mo for 7 mo | ––– | ||||
| Thyroid hormones | |||||||||||||||
| Triiodothyronine (T3) | |||||||||||||||
| Kelly & Lieberman 2009 [102] | Chart review (4) | Failed ≥2 trials | ≥2 | Add T3 (90.4 µg/d) | 125 BD-II 34 BD-NOS | 37.7 | 45.5 | ––– | ––– | 85% improved (CGI); 33% remitted (GAF) | T3 effective; no switches | ||||
| Thyroxine (T4) | |||||||||||||||
| Bauer et al. 2002 [103] | Open, prospective (4) | Failed ≥2 trials | ≥2 | Add T4 (379 µg/d) | 9 BD-I 4 BD-II 4 SzAff 4 UP | 76.2 | 47.6 | 32.6 | 51.4 | 52.4% very, 19.0% much improved: (CGI, episodes, hospital) | BD best | ||||
| Thyroid hormones | |||||||||||||||
| Thyroxine (T4) | |||||||||||||||
| Stamm et al. | Randomized | Depressed | ≥2 | Add T4 (to 300 | 35 BD-I | 51.6 | 44.9 | ––– | 1.5 | Minor T4 v PBO | T4 | ||||
| 2014 [104] | v PBO (1) | despite | µg/d) | 27 BD-II | differences | ineffective | |||||||||
| multiple | v PBO | ||||||||||||||
| agents | |||||||||||||||
Abbreviations: Name codes of test agents self-defined above; AC = anticonvulsant; AD = antidepressant; ADRs = adverse drug-associated responses; AED = anti-epileptic drugs; AP = antipsychotic; BD = Bipolar Disorder; BPRS = Brief Psychiatric Rating Scale; BRMS = Bech-Rafaelsen Mania Scale; CBZ = carbamazepine; CGI = Clinical Global Impression; d = day; GAF = Global Assessment of Functioning; HAL= haloperidol; HDRS = Hamilton Depression Rating Scale; IV = intravenous; Li = lithium; MADRS = Montgomery-Åsberg Depression Rating Scale; mo = month; MS = mood-stabilizer; PBO = placebo; QTP = quetiapine; RXs = treatments; SL = sublingual; Sz = Schizophrenia; SzAff = Schizoaffective Disorder; UP = Unipolar Depression; VPA = sodium valproate; YMRS = Young Mania Rating Scale. Levels of evidence: 1 (randomised controlled trials), 2 (non-randomised controlled trials), 3 (observational studies with controls), 4 (observational studies without controls) (Source: US Department of Health and Human Services).
RESULTS
The search process considered a total of 100 potentially useful reports, of which 38 satisfied entry criteria and provided data reported here. The preferred reporting items for systematic reviews and meta-analyses (PRISMA) flowchart for this review is shown in Fig. 1. These reports are characterized by their design, demographic and clinical characteristics of subjects, and their main findings (Table 1). Several types of psychotropic drug treatments were considered, as follows.
Fig. (1).
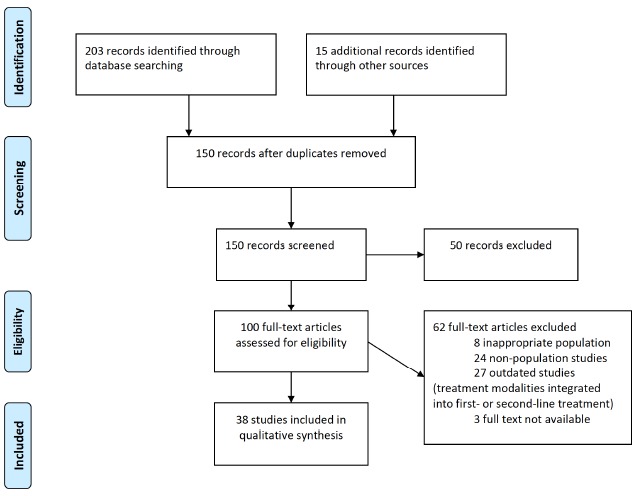
Preferred reporting items for systematic reviews and meta-analyses (PRISMA) flowchart illustrating methodological steps in identifying empirical studies to be included in this systematic review.
Atypical Antipsychotics
Virtually all antipsychotic drugs have potent and rapid efficacy in acute mania, although modern, “second-generation,” or “atypical” agents (with relatively low risks of adverse neurological effects) currently are usually preferred, particularly for long-term treatment of bipolar disorder patients, owing to the efficacy of some such agents in acute bipolar depression as well as growing evidence of long-term mood-stabilizing effects [5, 20]. We found reports on the use of four atypical antipsychotics in the treatment of resistant bipolar disorder.
Clozapine lacks regulatory approval for use in any phase of bipolar disorder, but has some evidence of efficacy in the treatment of refractory mania, with or without psychotic symptoms, given alone or adjunctively with a standard mood-stabilizing treatment such as lithium carbonate or a putatively mood-stabilizing anticonvulsant [21-25]. The risk-benefit profile in long-term treatment of bipolar disorder with clozapine needs to be assessed carefully, due to its risk of agranulocytosis, carditis, ileus, seizures, and other potentially life-threatening adverse effects [5]. It is also of interest to consider whether it may provide antisuicidal effects in bipolar disorder as are reported to occur (and have regulatory approval) in patients diagnosed with schizophrenia [26, 27]. Clozapine also may reduce abuse of alcohol and other substances, and should be evaluated for such effects in bipolar disorder patients [28].
Aripiprazole has been tested in at least two, short-term, placebo-controlled, monotherapy trials in acute mania. Both found greater reduction in mania symptom ratings than with a placebo, without greater risk of adverse effects or of early discontinuation [29, 30]. Three small, open trials added aripiprazole to other treatments in treatment resistant bipolar disorder (including in bipolar depression), and found some evidence for beneficial effects [31, 32], including one that included subjects who had not responded well to a trial of clozapine [33]. Among adverse effects, aripiprazole has a substantial risk of inducing akathisia-like restlessness, which can interact badly in agitated or manic bipolar disorder patients [31, 32].
Olanzapine was tried in an open, prospective trial for mania that had not responded satisfactorily to at least two mood-stabilizers or antipsychotics, and yielded more than 50% reduction in mania ratings in 88.5% of the 18 cases [34]. A long-term trial added olanzapine to other mood-stabilizing treatments for at least 6 months and found improvement in Clinical Global Impression (CGI) scores in all 23 patients, with reductions in relapses and hospitalizations [35].
Quetiapine (in doses averaging 188 mg/day) was combined with the anticonvulsant lamotrigine in an open trial in 38 cases of treatment-resistant bipolar depression for 3 months. Small improvements in CGI ratings by an average of 1 point were noted. Adverse effects including excessive sedation led 17.9% of the cases to require discontinuation of quetiapine [36].
Anticonvulsants
Several drugs used clinically as anticonvulsants for epileptic patients have been found to exert antimanic effects. These include carbamazepine and valproic acid salts, which also are used empirically (“off-label”) for long-term reduction of recurrences of bipolar illness, although without regulatory approval. In addition, lamotrigine can reduce long-term recurrences of depression in bipolar disorder patients, although it is impractical for short-term use due to the need for slow dose-increases to limit risk of dermatological reactions. In addition, it seems to have little antimanic efficacy in short- or long-term applications. The carbamazepine analog oxcarbazepine, and gabapentin have been used empirically to treat bipolar disorder patients, even though both lack empirical evidence of efficacy [5].
Eslicarbazepine (S-[+]-licarbazepine) is a relatively new anticonvulsant approved for adjunctive use in epilepsy. It is chemically related to carbamazepine and oxcarbazepine (all dibenzazepines) and is the principal active metabolite of oxcarbazepine. It appears to be relatively well tolerated [37]. Its use has been reported in at least one case of refractory mania, with apparent benefit and needs to be studied further [38].
Pregabalin is a structural analog of the principal inhibitory amino acid neurotransmitter of the central nervous system, ã-aminobutyric acid (GABA), which acts through voltage-dependent calcium channels and, among other functions, limits release of the neurotransmitters glutamate and norepinephrine [39, 40]. It is effective in epilepsy with partial seizures and has beneficial effects on anxiety and certain types of chronic pain, especially in fibromyalgia and some kinds of neuropathic pain [40]. In small numbers of cases, pregabalin has been reported anecdotally to increase responses to quetiapine in acute mania [41], as well as to decrease depressive symptoms associated with anxiety disorders [42]. Adding pregabalin to other antimanic agents also was associated with improvement in a case of treatment-resistant, acute mania [43], as well as initially in 41% of 58 such patients in an open-label trial, with sustained benefit in 10% for up to 3 years [44]. This anticonvulsant also may have beneficial effects in limiting abuse of central depressants including alcohol and benzodiazepines [45, 46]. Given that patients with bipolar disorder commonly have co-occurring substance abuse, pregabalin might be studied specifically for patients with these dual diagnoses.
Topiramate is a structurally novel anticonvulsant (a methylethyldienefructopyranose) with pharmacodynamic similarities to valproate and carbamazepine, including potentiation of GABA, reduced activity of glutamate as a cerebral excitatory neurotransmitter, and blockade of neuronal sodium and calcium ion channels [47]. Its lack of association with weight-gain has encouraged its empirical, usually adjunctive, use in the treatment of psychiatric disorders with weight-promoting drugs [48]. However, evidence of its having acute antimanic, antidepressant, or long-term mood-stabilizing effects in bipolar disorder was not found in several well-designed, controlled trials [49,50]. We found one early, uncontrolled trial that suggested possible long-term stabilizing effects when topiramate was added to standard treatments in 34 cases treatment-resistant, broadly defined bipolar disorders for up to six months [51], but the finding has not been sustained, and topiramate appears no longer to be of interest for the treatment of otherwise treatment-resistant bipolar disorder.
Antidepressants
Antidepressant use in bipolar depression has been highly controversial, based on inconsistent as well as remarkably limited evidence of short-term efficacy and lack of evidence for substantial long-term protective effectiveness against recurrences of depressive phases in bipolar disorder [52-55]. There also have been concerns that mood-elevating agents may induce potentially dangerous states of manic excitation, particularly in bipolar I disorder patients, although the available evidence indicates that drug-associated increases above the high spontaneous rates of mood-switching are far lower than is widely believed [56]. In addition, there is no evidence that antidepressants alter the risk of commonly encountered suicidal behavior in bipolar disorder patients, particularly in younger years [7].
Despite these uncertainties, there is some evidence that antidepressants can yield useful, short-term antidepressant effects in bipolar disorder, especially when given cautiously at initially low and slowly increased doses of short-acting agents, with a mood-stabilizing treatment in place, probably selectively for depressed bipolar disorder patients lacking in current agitation or hypomanic symptoms [53-55]. In addition, specific antidepressants vary in their association with manic switching. Among agents of high risk are older tricyclic antidepressants and the modern serotonin-norepinephrine potentiating agent venlafaxine, whereas serotonin reuptake inhibitors (SRIs) and the mild stimulant-antidepressant bupropion appear to have lower risks [56]. Bupropion may appear to be better tolerated in part owing to its regulatory approval for use in relatively low doses to limit risk of inducing epileptic seizures [7].
A small, unblinded trial involving both bipolar and unipolar depressed patients added bupropion to a variety of other, previously unsuccessful psychopharmacological agents, and observed improvements in ratings of depressive symptoms by ≥50% within four weeks in 7/11 cases, with no newly-emerging mania or hypomania [16]. Also, open-label, randomized addition of bupropion in low doses (150 mg/day) to aripiprazole plus sodium valproate in 7 depressed bipolar disorder patients (not necessarily treatment-resistant) yielded reductions in the abuse of cocaine compared to 5 similar, treatment-as-usual, comparison subjects [57].
Remarkably, despite the prominence of unresolved depression among bipolar I and II disorder patients, and the massive investigation of antidepressants in unipolar depression since the 1950s, there are very few randomized, controlled trials of older or newer antidepressants in any phase of depressive morbidity in bipolar disorder patients, with or without evidence of treatment resistance [52, 55]. In part, this lack of investigation may reflect exaggerated concerns about the risks of inducing mania or hypomania, especially among type I bipolar disorder patients [58].
Glutamatergic Agents
Glutamate is the principal excitatory, cerebral amino acid neurotransmitter and is involved in synaptic plasticity, learning and memory, among many other functions. There is increasing evidence that the glutamatergic system may play a role in the pathophysiology of bipolar disorder [59, 60]. Several types of drugs exert effects mediated through glutamatergic systems, with particular attention given to the N-methyl-D-asparate (NMDA) type of glutamate receptor.
An NMDA antagonist of interest is the potentially hallucinogenic, dissociative veterinary anesthetic agent ketamine, a phenylcyclohexanone, which also exerts effects on monoamine transport and at opioid receptors [61]. In addition to its anesthetic and analgesic effects [62], ketamine also has striking and rapid effects on mood, particularly to reverse depression, often very rapidly [63, 64]. Two small but double-blinded, randomized, crossover, placebo-controlled trials found that intravenous infusion of ketamine rapidly improved depressive symptoms in cases of refractory bipolar depression. One trial achieved beneficial responses in 71% of 18 subjects following single doses, compared to 6% of controls [65]. The other study also found robust improvement in depression after infusion of ketamine, with reduction of suicidal ideation in 15 severely depressed bipolar disorder patients who had not responded to at least one previous treatment trial [63]. An uncontrolled experience with two bipolar disorder patients with treatment-resistant depression observed responses to intramuscularly injected, adjunctive ketamine after not responding to its oral or intranasal administration or to other treatments; both patients were maintained successfully with injections every other week for nearly six months [66]. Another series of 14 bipolar and 12 unipolar patients with treatment-resistant depression were given adjunctive ketamine sublingually; 77% of the 26 patients showed evidence of improvement and tolerated addition of ketamine well [67].
Memantine has NMDA receptor antagonist activity and is used in the treatment of Alzheimer dementia. It has been reported to have beneficial effects in a case report of treatment-unresponsive bipolar disorder [68]. Additional open-label, add-on trials, including a six-year, mirror-image study, have observed favorable effects for up to one to three years [69-71] We also found one, small, randomized, placebo-controlled trial comparing addition of memantine (to 20 mg/day; n=14) or placebo (n=15) to lamotrigine (≥100 mg/day) in bipolar depressed patients who were not necessarily treatment-resistant. Memantine was associated with superior early improvements in depression ratings that
were not sustained for 8 weeks [72]. These findings, together, encourage further study of memantine in randomized, controlled trials.
Anticholinesterases
Other agents used to treat dementia have also been considered for treatment-resistant bipolar disorder, including centrally active cholinesterase antagonists aimed at potentiating the actions of cerebral acetylcholine. One of these, donepezil, showed some preliminary benefits [73], but when studied in a placebo-controlled trial, was not helpful in refractory mania when added to standard therapy [74]. This agent also has shown suggestions of improved cognition in bipolar disorder patients, but at the risk of emotional destabilization, especially in bipolar I cases [75]. Additional case reports also support the impression that donepezil may induce or worsen mania [76].
Dopamine Agonists
Compounds with dopamine-enhancing activity have been used as augmenting agents in treatment-resistant cases of unipolar and bipolar major depression. One of these, the benzthiazole pramipexole, acts as an agonist of D2 and D3 dopamine receptors in forebrain and has been used successfully to treat Parkinson disease, Ekbom’s restless legs syndrome, and to suppress prolactin production in the anterior pituitary [77, 78]. It may also have antidepressant effects [79, 80], including in treatment-resistant unipolar and bipolar depression [81]. A double-blinded, randomized, placebo-controlled trial tested pramipexole as an add-on agent in treatment-resistant bipolar depression [82]. More than half of the participants improved clinically within 6 weeks of adding pramipexole, and the drug was quite well tolerated, with a reported risk of mood-switching of 4.5% [82]. In addition, three, small, uncontrolled chart reviews or case series provide evidence for possible long-term effectiveness of pramipexole in bipolar disorder patients (not necessarily treatment-resistant, and including some unipolar depressed cases) in trials lasting 4–7 months [83-85]. One of these uncontrolled studies included treatment with another dopamine agonist, the indolone ropinerole added to the treatment regimens of depressed bipolar disorder patients who had been poorly responsive to other treatments [84]. Dopamine agonists may be of value in bipolar II as well as bipolar I depression, and risks of inducing mania or hypomania appear to be moderate [83, 84, 86].
Use of dopamine agonists in bipolar disorder patients may carry particular risks of emotional destabilization following their discontinuation, given reports of a dopamine agonist withdrawal syndrome (“DAWS”) in Parkinson disease patients, which included agitation and other prominent psychiatric symptoms [87].
Psychostimulants
Methylphenidate and amphetamines inhibit the physiological inactivation of released dopamine by neuronal reuptake, to increase actions of the neurotransmitter. Stimulants were often used for the treatment of major depressive disorder before the discovery of monoamine inhibitors and of tricyclic antidepressants in the 1950s, although their benefits were limited and adverse effects and risks of abuse led to their virtual abandonment for this purpose [5,88]. Such drugs have been considered for use in cases of otherwise treatment-resistant depression, including in an uncontrolled study of 50 treatment-resistant depressed patients of various types, of whom one-third showed apparent benefit; 1/27 (3.7%) of the bipolar disorder cases became manic or hypomanic [89].
Modafinil and its active R-enantiomer, armodafinil are mild stimulant-like agents with complex neuropharmacological actions that include inhibition of dopamine reuptake, similar to other stimulants, and they are used primarily to treat narcolepsy [90,91]. Given that depression is frequently associated with fatigue and somnolence, modafinil has been considered as a potentially useful adjunct to other treatments for depression, including in bipolar disorder. Two randomized, double-blinded, placebo-controlled trials for bipolar depression (not treatment resistant) found that adjunctive modafinil (100–200 mg/day) and armodafinil (150 mg/d) were superior to placebo in reducing depressive symptoms, with little risk of switching into mania or hypomania within six weeks [92, 93]. In addition, modafinil (626 mg/day) and pramipexole were given adjunctively without blinding or controls, with 3.5 other drugs/person for up to 1.5 years in 63 treatment-resistant bipolar disorder outpatients; modafinil yielded somewhat superior benefits for bipolar depression, based on clinical ratings, with approximately three-fold better tolerability of modafinil [94].
As with direct dopamine agonists, stimulants including anti-narcolepsy agents require further study for their safety on discontinuation in bipolar disorder patients as well as to clarify their efficacy in various phases of the disorder.
Calcium Channel Antagonists
Calcium channels have been implicated in the neurobiology of bipolar disorder [95]. The functioning of cell membrane calcium channels in the central nervous system can be altered by such drugs as the calcium channel antagonists developed primarily to treat hypertension [96]. One such agent, the chemically complex heterocyclic diltiazem, has been considered for treatment-resistant bipolar disorder. A small, uncontrolled, mirror-image study comparing morbidity in six months before versus during addition of diltiazem to unsuccessful, ongoing mood-stabilizing treatments appeared to add to long-term stabilization [97]. However, these findings were not supported by several later studies, leaving unresolved whether such drugs might contribute to the treatment of bipolar disorder [98].
Other Agents
Other drugs considered for the treatment of refractory bipolar disorder include analgesic opioids and thyroid hormones. There is some evidence that opioids may be beneficial in unipolar depression [99], and there is at least one case report of possible value of adding the opioid oxycodone to other treatments that had been unsuccessful for bipolar depression [100]. Opioids are unlikely to provoke mania, but their risks of producing dependency and withdrawal reactions, as well as other adverse effects, have severely limited interest in their use for mood disorders, especially in bipolar disorder with its high risk of co-occurring substance abuse [1].
Thyroid hormones have been used adjunctively in treatment-resistant, non-bipolar major depression with inconsistent evidence of efficacy, especially for tri-iodothyroine (T3) [101]. An uncontrolled, retrospective chart review evaluated effects of adding tri-iodothyronine (90.4 µg/day) to complex maintenance regimens of 159 treatment-refractory bipolar disorder patients, and found improvement in 85% of cases [102]. In an open-label trial in 13 cases of treatment-resistant depression in bipolar, unipolar, and schizoaffective disorders, L-thyroxine (T4) was given for up to a year in high doses (379 µg/day); 71% improved substantially by several measures, with better responses in the bipolar disorder subjects [103]. A recent, randomized trial tested effects of adding L-thyroxine in doses up to 300 µg/day for six weeks to complex regimens in 62 bipolar disorder patients who remained depressed; there were only minor differences from placebo controls [104]. These several findings are largely inconclusive, but suggest that tri-iodothyronine requires further study.
CONCLUSIONS
Bipolar disorder is a prevalent condition with a very large disease burden that includes high social and economic costs, substance abuse, disability, high suicidal risks and increased all-cause mortality rates, incomplete control of long-term morbidity, and especially poor control of depressive components of the disorder. Despite its high prevalence of treatment-resistance, studies of pharmacological treatment options in bipolar disorder remain remarkably scarce, highly variable in the quality of their designs, and largely inconclusive. Most studies reviewed involved relatively small numbers of patients, often admixtures of bipolar (I, II, or unspecified) with unipolar and schizoaffective disorder diagnoses, varying definitions of treatment-resistance, inconsistent definition of and selection by initial clinical states, imprecisely defined aims, and complex treatment regimens to which test agents were added. Encouraging results in apparent treatment-resistant bipolar disorder have been reported by adding clozapine, aripiprazole, pregabalin, bupropion, ketamine, memantine, pramipexole, and perhaps tri-iodothyronine to ongoing, sometimes already complex, regimens. The high prevalence of unresolved morbidity, especially of depressive components, in bipolar disorder requires far more experimental therapeutic trials of consistently better quality, involving coherent sampling, randomization, placebo controls, and simpler treatment regimens. Promising extant short-term findings should be pursued with trials continued for at least a year.
ACKNOWLEDGEMENTS
Supported, in part, by a grant from the Bruce J. Anderson Foundation and by the McLean Private Donors Bipolar Disorder Research Fund (to RJB).
CONFLICT OF INTEREST
None of the authors nor any close family member has any current financial relationships with the pharmaceutical or biotechnology industries.
REFERENCES
- 1.Goodwin F.K., Jamison K.R. Manic Depressive Illness. 2nd ed. New York: Oxford University Press; 2007. [Google Scholar]
- 2.Merikangas K.R., Akiskal H.S., Angst J., Greenberg P.E., Hirschfeld R.M., Petukhova M., Kessler R.C. Lifetime and 12-month prevalence of bipolar spectrum disorder in the National Comorbidity Survey replication. Arch. Gen. Psychiatry. 2007;64(5):543–552. doi: 10.1001/archpsyc.64.5.543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Baldessarini R.J., Salvatore P., Khalsa H.M., Gebre-Medhin P., Imaz H., GonzAlez-Pinto A., Perez J., Cruz N., Maggini C., Tohen M. Morbidity in 303 first-episode bipolar I disorder patients. Bipolar Disord. 2010;12(3):264–270. doi: 10.1111/j.1399-5618.2010.00812.x. [DOI] [PubMed] [Google Scholar]
- 4.Baldessarini R.J., Vieta E., Calabrese J.R., Tohen M., Bowden C.L. Bipolar depression: overview and commentary. Harv. Rev. Psychiatry. 2010;18(3):143–157. doi: 10.3109/10673221003747955. [DOI] [PubMed] [Google Scholar]
- 5.Baldessarini R.J. Chemotherapy in Psychiatry. 3rd ed. New York: Springer Press; 2013. [Google Scholar]
- 6.Baldessarini R.J., PA(c)rez J., Salvatore P., Trede K., Maggini C. History of bipolar manic-depressive disorder. In: Yildiz A., Nemeroff C., Ruiz P., editors. Bipolar Disorder: Millennium Update. New York: Oxford University Press, New York; 2014. in press. [Google Scholar]
- 7.Tondo L., Baldessarini R.J. Suicide in bipolar disorder. In: Yildiz A., Nemeroff C., Ruiz P., editors. Bipolar Disorder: Millennium Update. New York: Oxford University Press, New York; 2014. in press. [Google Scholar]
- 8.VAzquez G.H., Baldessarini R.J., Tondo L. Co-occurrence of anxiety and bipolar disorders: clinical and therapeutic overview. Depress. Anxiety. 2014;31(3):196–206. doi: 10.1002/da.22248. [DOI] [PubMed] [Google Scholar]
- 9.Tondo L., VAzquez G.H., Baldessarini R.J. Treatment-resistant depression in bipolar disorder. Curr. Psychiatry Rep. 2014;16:431. doi: 10.1007/s11920-013-0431-y. [DOI] [PubMed] [Google Scholar]
- 10.NICE (UK National Institute for Health and Clinical Excellence) Bipolar Disorder, guideline 38. London: NICE; 2006. [accessible at: guidance.nice.org.uk/cg38, accessed 20 Aug 2014] [Google Scholar]
- 11.Yatham L.N., Kennedy S.H., Parikh S.V. CANMAT and ISBD collaborative update of guidelines for the management of patients with bipolar disorder. Bipolar Disord. 2013;15:1–44. doi: 10.1111/bdi.12025. [DOI] [PubMed] [Google Scholar]
- 12.Baldessarini R.J., Henk H.J., Sklar A.R., Chang J., Leahy L.F. Psychotropic medications for bipolar disorder patients in the United States: polytherapy and adherence. Psychiatr. Serv. 2008;59:1175–1183. doi: 10.1176/ps.2008.59.10.1175. [DOI] [PubMed] [Google Scholar]
- 13.Centorrino F., Ventriglio A., Vincenti A., Talamo A., Baldessarini R.J. Changes in medication practices for hospitalized psychiatric patients: 2009 versus 2004. Hum. Psychopharmacol. 2010;25(2):179–186. doi: 10.1002/hup.1095. [DOI] [PubMed] [Google Scholar]
- 14.Parikh S.V., LeBlanc S.R., Ovanessian M.M. Advancing bipolar disorder: key lessons from the Systematic Treatment Enhancement Program for Bipolar Disorder (STEP-BD). Can. J. Psychiatry. 2010;55(3):136–143. doi: 10.1177/070674371005500304. [DOI] [PubMed] [Google Scholar]
- 15.Dell?(tm)Osso B., Mundo E., D?(tm)Urso N., Pozzoli S., Buoli M., Ciabatti M., Rosanova M., Massimini M., Bellina V., Mariotti M., Altamura A.C. Augmentative repetitive navigated transcranial magnetic stimulation (rTMS) in drug-resistant bipolar depression. Bipolar Disord. 2009;11(1):76–81. doi: 10.1111/j.1399-5618.2008.00651.x. [DOI] [PubMed] [Google Scholar]
- 16.Erfurth A., Michael N., Stadtland C., Arolt V. Bupropion as add-on strategy in difficult-to-treat bipolar depressive patients. Neuropsychobiology. 2002;45(Suppl. 1):33–36. doi: 10.1159/000049259. [DOI] [PubMed] [Google Scholar]
- 17.GonzAlez-Isasi A., EcheburAa E., Mosquera F., IbAAez B., Aizpuru F., GonzAlez-Pinto A. Long-term efficacy of a psychological intervention program for patients with refractory bipolar disorder: a pilot study. Psychiatry Res. 2010;176(2-3):161–165. doi: 10.1016/j.psychres.2008.06.047. [DOI] [PubMed] [Google Scholar]
- 18.Pacchiarotti I., Mazzarini L., Colom F. Treatment-resistant bipolar depression: towards a new definition. 2009. [DOI] [PubMed]
- 19.Poon S.H., Sim K., Sum M.Y., Kuswanto C.N., Baldessarini R.J. Evidence-based options for treatment-resistant adult bipolar disorder patients. Bipolar Disord. 2012;14(6):573–584. doi: 10.1111/j.1399-5618.2012.01042.x. [DOI] [PubMed] [Google Scholar]
- 20.Stahl S. Essential Osychopharmacology. 4th ed. Cambridge: Cambridge University Press; 2013. [Google Scholar]
- 21.Banov M.D., Zarate C.A., Jr, Tohen M., Scialabba D., Wines J.D., Jr, Kolbrener M., Kim J.W., Cole J.O. Clozapine therapy in refractory affective disorders: polarity predicts response in long-term follow-up. J. Clin. Psychiatry. 1994;55(7):295–300. [PubMed] [Google Scholar]
- 22.Calabrese J.R., Kimmel S.E., Woyshville M.J., Rapport D.J., Faust C.J., Thompson P.A., Meltzer H.Y. Clozapine for treatment-refractory mania. Am. J. Psychiatry. 1996;153(6):759–764. doi: 10.1176/ajp.153.6.759. [DOI] [PubMed] [Google Scholar]
- 23.Suppes T., Webb A., Paul B., Carmody T., Kraemer H., Rush A.J. Clinical outcome in a randomized 1-year trial of clozapine versus treatment as usual for patients with treatment-resistant illness and a history of mania. Am. J. Psychiatry. 1999;156(8):1164–1169. doi: 10.1176/ajp.156.8.1164. [DOI] [PubMed] [Google Scholar]
- 24.Ciapparelli A., Dell?(tm)Osso L., Bandettini di Poggio A., Carmassi C., Cecconi D., Fenzi M., Chiavacci M.C., Bottai M., Ramacciotti C.E., Cassano G.B. Clozapine in treatment-resistant patients with schizophrenia, schizoaffective disorder, or psychotic bipolar disorder: a naturalistic 48-month follow-up study. J. Clin. Psychiatry. 2003;64(4):451–458. doi: 10.4088/JCP.v64n0416. [DOI] [PubMed] [Google Scholar]
- 25.Chang J.S., Ha K.S., Young Lee K., Sik Kim Y., Min Ahn Y. The effects of long-term clozapine add-on therapy on the rehospitalization rate and the mood polarity patterns in bipolar disorders. J. Clin. Psychiatry. 2006;67(3):461–467. doi: 10.4088/JCP.v67n0318. [DOI] [PubMed] [Google Scholar]
- 26.Meltzer H.Y., Alphs L., Green A.I., Altamura A.C., Anand R., Bertoldi A., Bourgeois M., Chouinard G., Islam M.Z., Kane J., Krishnan R., Lindenmayer J.P., Potkin S. International Suicide Prevention Trial Study Group. Clozapine treatment for suicidality in schizophrenia: International Suicide Prevention Trial (InterSePT). Arch. Gen. Psychiatry. 2003;60(1):82–91. doi: 10.1001/archpsyc.60.1.82. [DOI] [PubMed] [Google Scholar]
- 27.Hennen J., Baldessarini R.J. Suicidal risk during treatment with clozapine: a meta-analysis. Schizophr. Res. 2005;73(2-3):139–145. doi: 10.1016/j.schres.2004.05.015. [DOI] [PubMed] [Google Scholar]
- 28.Zhornitsky S., Rizkallah E., Pampoulova T., Chiasson J.P., Stip E., RomprA(c) P.P., Potvin S. Antipsychotic agents for the treatment of substance use disorders in patients with and without comorbid psychosis. J. Clin. Psychopharmacol. 2010;30(4):417–424. doi: 10.1097/JCP.0b013e3181e7810a. [DOI] [PubMed] [Google Scholar]
- 29.Keck P.E., Jr, Marcus R., Tourkodimitris S., Ali M., Liebeskind A., Saha A., Ingenito G. Aripiprazole Study Group. A placebo-controlled, double-blind study of the efficacy and safety of aripiprazole in patients with acute bipolar mania. Am. J. Psychiatry. 2003;160(9):1651–1658. doi: 10.1176/appi.ajp.160.9.1651. [DOI] [PubMed] [Google Scholar]
- 30.Sachs G., Sanchez R., Marcus R., Stock E., McQuade R., Carson W., Abou-Gharbia N., Impellizzeri C., Kaplita S., Rollin L., Iwamoto T. Aripiprazole Study Group. Aripiprazole in the treatment of acute manic or mixed episodes in patients with bipolar I disorder: a 3-week placebo-controlled study. J. Psychopharmacol. (Oxford) 2006;20(4):536–546. doi: 10.1177/0269881106059693. [DOI] [PubMed] [Google Scholar]
- 31.Ketter T., Wang P., Chandler R., Culver J., Alarcon A. Adjunctive aripiprazole in treatment-resistant bipolar depression. 2006. [DOI] [PubMed]
- 32.Kemp D.E., Gilmer W.S., Fleck J., Straus J.L., Dago P.L., Karaffa M. Aripiprazole augmentation in treatment-resistant bipolar depression: early response and development of akathisia. Prog. Neuropsychopharmacol. Biol. Psychiatry. 2007;31(2):574–577. doi: 10.1016/j.pnpbp.2006.12.009. [DOI] [PubMed] [Google Scholar]
- 33.Benedetti A., Di Paolo A., Lastella M., Casamassima F., Candiracci C., Litta A., Ciofi L., Danesi R., Lattanzi L., Del Tacca M., Cassano G.B. Augmentation of clozapine with aripiprazole in severe psychotic bipolar and schizoaffective disorders: a pilot study. Clin. Pract. Epidemol Ment. Health. 2010;6:30–35. doi: 10.2174/1745017901006010030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Chen J., Muzina D.J., Kemp D.E., Conroy C., Chan P., Serrano M.B., Ganocy S.J., Fang Y., Calabrese J.R., Gao K. Safety and efficacy of olanzapine monotherapy in treatment-resistant bipolar mania: a 12-week open-label study. Hum. Psychopharmacol. 2011;26(8):588–595. doi: 10.1002/hup.1249. [DOI] [PubMed] [Google Scholar]
- 35.Vieta E., Reinares M., Corbella B., Benabarre A., Gilaberte I., Colom F., MartA-nez-ArAn A., GastA3 C., Tohen M. Olanzapine as long-term adjunctive therapy in treatment-resistant bipolar disorder. J. Clin. Psychopharmacol. 2001;21(5):469–473. doi: 10.1097/00004714-200110000-00002. [DOI] [PubMed] [Google Scholar]
- 36.Ahn Y.M., Nam J.Y., Culver J.L., Marsh W.K., Bonner J.C., Ketter T.A. Lamotrigine plus quetiapine combination therapy in treatment-resistant bipolar depression. Ann. Clin. Psychiatry. 2011;23(1):17–24. [PubMed] [Google Scholar]
- 37.Benes J., Parada A., Figueiredo A.A., Alves P.C., Freitas A.P., Learmonth D.A., Cunha R.A., Garrett J., Soares-da-Silva P. Anticonvulsant and sodium channel-blocking properties of novel 10,11-dihydro-5H-dibenz[b,f]azepine-5-carboxamide derivatives. J. Med. Chem. 1999;42(14):2582–2587. doi: 10.1021/jm980627g. [DOI] [PubMed] [Google Scholar]
- 38.Nath K., Bhattacharya A., Praharaj S. Eslicarbazepine acetate in the management of refractory bipolar disorder. 2012. [DOI] [PubMed]
- 39.Micheva K., Taylor C., Smith S. Pregabalin reduces the release of synaptic vesicles from cultured hippocampal neurons. 2006. [DOI] [PubMed]
- 40.Martinotti G., Lupi M., Sarchione F., Santacroce R., Salone A., De Berardis D., Serroni N., Cavuto M., Signorelli M., Aguglia E., Valchera A., Iasevoli F., Di Giannantonio M. The potential of pregabalin in neurology, psychiatry and addiction: a qualitative overview. Curr. Pharm. Des. 2013;19(35):6367–6374. doi: 10.2174/13816128113199990425. [DOI] [PubMed] [Google Scholar]
- 41.Oulis P., Florakis A.A., Tzanoulinos G., Papadimitriou G.N. Adjunctive pregabalin to quetiapine in acute mania. Clin. Neuropharmacol. 2009;32(3):174. doi: 10.1097/WNF.0b013e31818a651a. [DOI] [PubMed] [Google Scholar]
- 42.Stein D.J., Baldwin D.S., Baldinetti F., Mandel F. Efficacy of pregabalin in depressive symptoms associated with generalized anxiety disorder: a pooled analysis of 6 studies. Eur. Neuropsychopharmacol. 2008;18(6):422–430. doi: 10.1016/j.euroneuro.2008.01.004. [DOI] [PubMed] [Google Scholar]
- 43.Conesa M., Rojo L., Plumed J., Livianos L. Pregabalin in the treatment of refractory bipolar disorders. 2012. [DOI] [PMC free article] [PubMed]
- 44.Schaffer L.C., Schaffer C.B., Miller A.R., Manley J.L., Piekut J.A., Nordahl T.E. An open trial of pregabalin as an acute and maintenance adjunctive treatment for outpatients with treatment resistant bipolar disorder. J. Affect. Disord. 2013;147(1-3):407–410. doi: 10.1016/j.jad.2012.09.005. [DOI] [PubMed] [Google Scholar]
- 45.Martinotti G., Di Nicola M., Tedeschi D., Mazza M., Janiri L., Bria P. Efficacy and safety of pregabalin in alcohol dependence. Adv. Ther. 2008;25(6):608–618. doi: 10.1007/s12325-008-0066-2. [DOI] [PubMed] [Google Scholar]
- 46.Oulis P., Konstantakopoulos G. Pregabalin in the treatment of alcohol and benzodiazepines dependence. 2010. [DOI] [PMC free article] [PubMed]
- 47.Shank R.P., Gardocki J.F., Vaught J.L., Davis C.B., Schupsky J.J., Raffa R.B., Dodgson S.J., Nortey S.O., Maryanoff B.E. Topiramate: preclinical evaluation of structurally novel anticonvulsant. Epilepsia. 1994;35(2):450–460. doi: 10.1111/j.1528-1157.1994.tb02459.x. [DOI] [PubMed] [Google Scholar]
- 48.Gabriel A. Adjunctive topiramate treatment in refractory obese bipolar patients: a descriptive open label study. Eat. Weight Disord. 2007;12(1):48–53. doi: 10.1007/BF03327772. [DOI] [PubMed] [Google Scholar]
- 49.Levy N.A., Janicak P.G. Calcium channel antagonists for the treatment of bipolar disorder. Bipolar Disord. 2000;2(2):108–119. doi: 10.1034/j.1399-5618.2000.020204.x. [DOI] [PubMed] [Google Scholar]
- 50.Vasudev K., Macritchie K., Geddes J., Watson S., Young A. Topiramate for acute affective episodes in bipolar disorder. Cochrane Database Syst. Rev. 2006;CD003384(1):CD003384. doi: 10.1002/14651858.cd003384.pub2. [DOI] [PubMed] [Google Scholar]
- 51.Vieta E., Torrent C., Garcia-Ribas G. Use of topiramate in treatment-resistant bipolar spectrum disorders. 2002. [DOI] [PubMed]
- 52.Pacchiarotti I., Bond D.J., Baldessarini R.J., Nolen W.A., Grunze H., Licht R.W., Post R.M., Berk M., Goodwin G.M., Sachs G.S., Tondo L., Findling R.L., Youngstrom E.A., Tohen M., Undurraga J., GonzAlez-Pinto A., Goldberg J.F., Yildiz A., Altshuler L.L., Calabrese J.R., Mitchell P.B., Thase M.E., Koukopoulos A., Colom F., Frye M.A., Malhi G.S., Fountoulakis K.N., VAzquez G., Perlis R.H., Ketter T.A., Cassidy F., Akiskal H., Azorin J.M., ValentA- M., Mazzei D.H., Lafer B., Kato T., Mazzarini L., MartA-nez-Aran A., Parker G., Souery D., Ozerdem A., McElroy S.L., Girardi P., Bauer M., Yatham L.N., Zarate C.A., Nierenberg A.A., Birmaher B., Kanba S., El-Mallakh R.S., Serretti A., Rihmer Z., Young A.H., Kotzalidis G.D., MacQueen G.M., Bowden C.L., Ghaemi S.N., Lopez-Jaramillo C., Rybakowski J., Ha K., Perugi G., Kasper S., Amsterdam J.D., Hirschfeld R.M., Kapczinski F., Vieta E. The International Society for Bipolar Disorders (ISBD) task force report on antidepressant use in bipolar disorders. Am. J. Psychiatry. 2013;170(11):1249–1262. doi: 10.1176/appi.ajp.2013.13020185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tondo L., Baldessarini R.J., VAzquez G., Lepri B., Visioli C. Clinical responses to antidepressants among 1036 acutely depressed patients with bipolar or unipolar major affective disorders. Acta Psychiatr. Scand. 2013;127(5):355–364. doi: 10.1111/acps.12023. [DOI] [PubMed] [Google Scholar]
- 54.VAzquez G.H., Tondo L., Undurraga J., Baldessarini R.J. Overview of antidepressant treatment of bipolar depression. Int. J. Neuropsychopharmacol. 2013;16(7):1673–1685. doi: 10.1017/S1461145713000023. [DOI] [PubMed] [Google Scholar]
- 55.VAzquez G.H., Tondo L., Undurraga J., Zaratiegui R., Baldessarini R.J. Pharmacological treatment for bipolar depression. 2014.
- 56.Tondo L., VAzquez G.H., Baldessarini R.J. Mania associated with antidepressant-treatment: comprehensive meta-analytic review. 2010. [DOI] [PubMed]
- 57.Sepede G., Di lorio G., Lupi M., Sarchione F., Acciavatti T., Fiori F., Santacroce R., Martinotti G., Gambi F., Di Giannantonio M. Bupropion as an add-on therapy in depressed bipolar disorder type I patients with comorbid cocaine dependence. Clin. Neuropharmacol. 2014;37(1):17–21. doi: 10.1097/WNF.0000000000000011. [DOI] [PubMed] [Google Scholar]
- 58.Undurraga J., Baldessarini R.J., ValentA- M., Pacchiarotti I., Tondo L., VAzquez G., Vieta E. Bipolar depression: clinical correlates of receiving antidepressants. J. Affect. Disord. 2012;139(1):89–93. doi: 10.1016/j.jad.2012.01.027. [DOI] [PubMed] [Google Scholar]
- 59.Eastwood S.L., Harrison P.J. Markers of glutamate synaptic transmission and plasticity are increased in the anterior cingulate cortex in bipolar disorder. Biol. Psychiatry. 2010;67(11):1010–1016. doi: 10.1016/j.biopsych.2009.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jun C., Choi Y., Lim S.M., Bae S., Hong Y.S., Kim J.E., Lyoo I.K. Disturbance of the glutamatergic system in mood disorders. Exp. Neurobiol. 2014;23(1):28–35. doi: 10.5607/en.2014.23.1.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kohrs R., Durieux M.E. Ketamine: teaching an old drug new tricks. Anesth. Analg. 1998;87(5):1186–1193. doi: 10.1111/aas.12377. [INSERT FIGURE 001]. [DOI] [PubMed] [Google Scholar]
- 62.McNicol E.D., Schumann R., Haroutounian S. A systematic review and meta-analysis of ketamine for the prevention of persistent post-surgical pain. Acta Anaesthesiol. Scand. 2014;58(10):1199–1213. doi: 10.1111/aas.12377. [DOI] [PubMed] [Google Scholar]
- 63.Zarate C.A., Jr, Brutsche N.E., Ibrahim L., Franco-Chaves J., Diazgranados N., Cravchik A., Selter J., Marquardt C.A., Liberty V., Luckenbaugh D.A. Replication of ketamine?(tm)s antidepressant efficacy in bipolar depression: a randomized controlled add-on trial. Biol. Psychiatry. 2012;71(11):939–946. doi: 10.1016/j.biopsych.2011.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Fond G., Loundou A., Rabu C., Macgregor A., LanA on C., Brittner M., Micoulaud-Franchi J.A., Richieri R., Courtet P., Abbar M., Roger M., Leboyer M., Boyer L. Ketamine administration in depressive disorders: a systematic review and meta-analysis. Psychopharmacology (Berl.) 2014;231(18):3663–3676. doi: 10.1007/s00213-014-3664-5. [DOI] [PubMed] [Google Scholar]
- 65.Diazgranados N., Ibrahim L., Brutsche N.E., Newberg A., Kronstein P., Khalife S., Kammerer W.A., Quezado Z., Luckenbaugh D.A., Salvadore G., Machado-Vieira R., Manji H.K., Zarate C.A., Jr A randomized add-on trial of an N-methyl-D-aspartate antagonist in treatment-resistant bipolar depression. Arch. Gen. Psychiatry. 2010;67(8):793–802. doi: 10.1001/archgenpsychiatry.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cusin C., Hilton G.Q., Nierenberg A.A., Fava M. Long-term maintenance with intramuscular ketamine for treatment-resistant bipolar II depression. Am. J. Psychiatry. 2012;169(8):868–869. doi: 10.1176/appi.ajp.2012.12020219. [DOI] [PubMed] [Google Scholar]
- 67.Lara D.R., Bisol L.W., Munari L.R. Antidepressant, mood stabilizing and procognitive effects of very low dose sublingual ketamine in refractory unipolar and bipolar depression. Int. J. Neuropsychopharmacol. 2013;16(9):2111–2117. doi: 10.1017/S1461145713000485. [DOI] [PubMed] [Google Scholar]
- 68.Agarwal V., Tripathi A. Memantine in the management of a clinically challenging case of bipolar disorder. Indian J. Psychiatry. 2009;51(2):137–138. doi: 10.4103/0019-5545.49455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Koukopoulos A., Reginaldi D., Serra G., Koukopoulos A., Sani G., Serra G. Antimanic and mood-stabilizing effect of memantine as an augmenting agent in treatment-resistant bipolar disorder. 2010. [DOI] [PubMed]
- 70.Koukopoulos A., Serra G., Koukopoulos A.E., Reginaldi D., Serra G. The sustained mood-stabilizing effect of memantine in the management of treatment resistant bipolar disorders: findings from a 12-month naturalistic trial. J. Affect. Disord. 2012;136(1-2):163–166. doi: 10.1016/j.jad.2011.09.040. [DOI] [PubMed] [Google Scholar]
- 71.Serra G., DE Chiara L., Koukopoulos A.E., Koukopoulos A., Serra G., Kahn D.A. Memantine in the treatment and prophylaxis of bipolar II disorder and comorbid fibromyalgia: a case report. J. Psychiatr. Pract. 2014;20(3):232–236. doi: 10.1097/01.pra.0000450324.44661.12. [DOI] [PubMed] [Google Scholar]
- 72.Anand A., Gunn A.D., Barkay G., Karne H.S., Nurnberger J.I., Mathew S.J., Ghosh S. Early antidepressant effect of memantine during augmentation of lamotrigine inadequate response in bipolar depression: a double-blind, randomized, placebo-controlled trial. Bipolar Disord. 2012;14(1):64–70. doi: 10.1111/j.1399-5618.2011.00971.x. [DOI] [PubMed] [Google Scholar]
- 73.Burt T., Sachs G.S., Demopulos C. Donepezil in treatment-resistant bipolar disorder. Biol. Psychiatry. 1999;45(8):959–964. doi: 10.1016/S0006-3223(98)00320-5. [DOI] [PubMed] [Google Scholar]
- 74.Eden Evins A., Demopulos C., Nierenberg A., Culhane M.A., Eisner L., Sachs G. A double-blind, placebo-controlled trial of adjunctive donepezil in treatment-resistant mania. Bipolar Disord. 2006;8(1):75–80. doi: 10.1111/j.1399-5618.2006.00243.x. [DOI] [PubMed] [Google Scholar]
- 75.Kelly T. Is donepezil useful for improving cognitive dysfunction in bipolar disorder? J. Affect. Disord. 2008;107(1-3):237–240. doi: 10.1016/j.jad.2007.07.027. [DOI] [PubMed] [Google Scholar]
- 76.Leung J.G. Donepezil-induced mania. Consult Pharm. 2014;29(3):191–195. doi: 10.4140/TCP.n.2014.191. [DOI] [PubMed] [Google Scholar]
- 77.Kvernmo T., HA rtter S., Burger E. Review of the receptor-binding and pharmacokinetic properties of dopamine agonists. 2006. [DOI] [PubMed]
- 78.Antonini A., Barone P., Ceravolo R., Fabbrini G., Tinazzi M., Abbruzzese G. Role of pramipexole in the management of Parkinson?(tm)s disease. CNS Drugs. 2010;24(10):829–841. doi: 10.2165/11585090-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 79.Zarate C.A., Jr, Payne J.L., Singh J. Pramipexole for bipolar II depression: placebo-controlled proof of concept study. 2004. [DOI] [PubMed]
- 80.Goss A.J., Kaser M., Costafreda S.G., Sahakian B.J., Fu C.H. Modafinil augmentation therapy in unipolar and bipolar depression: a systematic review and meta-analysis of randomized controlled trials. J. Clin. Psychiatry. 2013;74(11):1101–1107. doi: 10.4088/JCP.13r08560. [DOI] [PubMed] [Google Scholar]
- 81.Cusin C., Iovieno N., Iosifescu D.V., Nierenberg A.A., Fava M., Rush A.J., Perlis R.H. A randomized, double-blind, placebo-controlled trial of pramipexole augmentation in treatment-resistant major depressive disorder. J. Clin. Psychiatry. 2013;74(7):e636–e641. doi: 10.4088/JCP.12m08093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Goldberg J.F., Burdick K.E., Endick C.J. Preliminary randomized, double-blind, placebo-controlled trial of pramipexole added to mood stabilizers for treatment-resistant bipolar depression. Am. J. Psychiatry. 2004;161(3):564–566. doi: 10.1176/appi.ajp.161.3.564. [DOI] [PubMed] [Google Scholar]
- 83.Sporn J., Ghaemi S.N., Sambur M.R., Rankin M.A., Recht J., Sachs G.S., Rosenbaum J.F., Fava M. Pramipexole augmentation in the treatment of unipolar and bipolar depression: a retrospective chart review. Ann. Clin. Psychiatry. 2000;12(3):137–140. doi: 10.3109/10401230009147102. [DOI] [PubMed] [Google Scholar]
- 84.Perugi G., Toni C., Ruffolo G., Frare F., Akiskal H. Adjunctive dopamine agonists in treatment-resistant bipolar II depression: an open case series. Pharmacopsychiatry. 2001;34(4):137–141. doi: 10.1055/s-2001-15872. [DOI] [PubMed] [Google Scholar]
- 85.Cassano P., Lattanzi L., Soldani F., Navari S., Battistini G., Gemignani A., Cassano G.B. Pramipexole in treatment-resistant depression: an extended follow-up. Depress. Anxiety. 2004;20(3):131–138. doi: 10.1002/da.20038. [DOI] [PubMed] [Google Scholar]
- 86.Dell?(tm)Osso B., Ketter T.A. Assessing efficacy/effectiveness and safety/tolerability profiles of adjunctive pramipexole in bipolar depression: acute versus long-term data. Int. Clin. Psychopharmacol. 2013;28(6):297–304. doi: 10.1097/YIC.0b013e3283639015. [DOI] [PubMed] [Google Scholar]
- 87.Rabinak C.A., Nirenberg M.J. Dopamine agonist withdrawal syndrome in Parkinson disease. Arch. Neurol. 2010;67(1):58–63. doi: 10.1001/archneurol.2009.294. [DOI] [PubMed] [Google Scholar]
- 88.Abbasowa L., Kessing L.V., Vinberg M. Psychostimulants in moderate to severe affective disorder: a systematic review of randomized controlled trials. Nord. J. Psychiatry. 2013;67(6):369–382. doi: 10.3109/08039488.2012.752035. [DOI] [PubMed] [Google Scholar]
- 89.Parker G., Brotchie H. Do the old psychostimulant drugs have a role in managing treatment-resistant depression? 2010. [DOI] [PubMed]
- 90.Minzenberg M.J., Carter C.S. Modafinil: a review of neurochemical actions and effects on cognition. Neuropsychopharmacology. 2008;33(7):1477–1502. doi: 10.1038/sj.npp.1301534. [DOI] [PubMed] [Google Scholar]
- 91.Loland C.J., Mereu M., Okunola O.M., Cao J., Prisinzano T.E., Mazier S., Kopajtic T., Shi L., Katz J.L., Tanda G., Newman A.H. R-modafinil (armodafinil): a unique dopamine uptake inhibitor and potential medication for psychostimulant abuse. Biol. Psychiatry. 2012;72(5):405–413. doi: 10.1016/j.biopsych.2012.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Calabrese J.R., Ketter T.A., Youakim J.M., Tiller J.M., Yang R., Frye M.A. Adjunctive armodafinil for major depressive episodes associated with bipolar I disorder: a randomized, multicenter, double-blind, placebo-controlled, proof-of-concept study. J. Clin. Psychiatry. 2010;71(10):1363–1370. doi: 10.4088/JCP.09m05900gry. [DOI] [PubMed] [Google Scholar]
- 93.Frye M., Grunze H., Suppes T. Placebo-controlled evaluation of adjunctive modafinil in the treatment of bipolar depression. 2007. [DOI] [PubMed]
- 94.Dell?(tm)osso B., Timtim S., Hooshmand F., Miller S., Wang P.W., Hill S.J., Portillo N., Ketter T.A. Superior chronic tolerability of adjunctive modafinil compared to pramipexole in treatment-resistant bipolar disorder. J. Affect. Disord. 2013;150(1):130–135. doi: 10.1016/j.jad.2012.11.030. [DOI] [PubMed] [Google Scholar]
- 95.Nurnberger J.I., Jr, Koller D.L., Jung J., Edenberg H.J., Foroud T., Guella I., Vawter M.P., Kelsoe J.R. Psychiatric Genomics Consortium Bipolar Group. Identification of pathways for bipolar disorder: a meta-analysis. JAMA Psychiatry. 2014;71(6):657–664. doi: 10.1001/jamapsychiatry.2014.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Fleckenstein A. History of calcium antagonists. Circ. Res. 1983;52(2 Pt 2):I3–I16. [PubMed] [Google Scholar]
- 97.Silverstone P.H., Birkett L. Diltiazem as augmentation therapy in patients with treatment-resistant bipolar disorder: a retrospective study. J. Psychiatry Neurosci. 2000;25(3):276–280. [PMC free article] [PubMed] [Google Scholar]
- 98.Casamassima F., Hay A.C., Benedetti A., Lattanzi L., Cassano G.B., Perlis R.H. L-type calcium channels and psychiatric disorders: A brief review. Am. J. Med. Genet. B. Neuropsychiatr. Genet. 2010;153B(8):1373–1390. doi: 10.1002/ajmg.b.31122. [DOI] [PubMed] [Google Scholar]
- 99.Berrocoso E., SAnchez-BlAzquez P., GarzA3n J., Mico J.A. Opiates as antidepressants. Curr. Pharm. Des. 2009;15(14):1612–1622. doi: 10.2174/138161209788168100. [DOI] [PubMed] [Google Scholar]
- 100.Schiffman J.E., Gitlin M.J. Adjunctive oxycodone for the treatment of refractory bipolar depression. J. Clin. Psychiatry. 2012;73(7):992. doi: 10.4088/JCP.11cr07565. [DOI] [PubMed] [Google Scholar]
- 101.Lasser R.A., Baldessarini R.J. Thyroid hormones in depressive disorders: a reappraisal of clinical utility. Harv. Rev. Psychiatry. 1997;4(6):291–305. doi: 10.3109/10673229709030557. [DOI] [PubMed] [Google Scholar]
- 102.Kelly T., Lieberman D.Z. The use of triiodothyronine as an augmentation agent in treatment-resistant bipolar II and bipolar disorder NOS. J. Affect. Disord. 2009;116(3):222–226. doi: 10.1016/j.jad.2008.12.010. [DOI] [PubMed] [Google Scholar]
- 103.Bauer M. Thyroid hormone augmentation with levothyroxine in bipolar depression. Bipolar Disord. 2002;4(Suppl. 1):109–110. doi: 10.1034/j.1399-5618.4.s1.59.x. [DOI] [PubMed] [Google Scholar]
- 104.Stamm T.J., Lewitzka U., Sauer C., Pilhatsch M., Smolka M.N., Koeberle U., Adli M., Ricken R., Scherk H., Frye M.A., Juckel G., Assion H.J., Gitlin M., Whybrow P.C., Bauer M. Supraphysiologic doses of levothyroxine as adjunctive therapy in bipolar depression: a randomized, double-blind, placebo-controlled study. J. Clin. Psychiatry. 2014;75(2):162–168. doi: 10.4088/JCP.12m08305. [DOI] [PubMed] [Google Scholar]


