Abstract
Background. Mesenchymal stem cells are the most commonly used seed cells in biomedical research and tissue engineering. Their secretory proteins have also been proven to play an important role in tissue healing. Methods. We isolated adipose stem cells and placental stem cells and performed analysis examining characteristics. The secretory proteins were extracted from conditioned medium and analyzed by MALDI-TOF/TOF. The antiaging effect of conditioned mediums was evaluated by the results of facial skin application. Results. Adipose stem cells and placental stem cells were found to be very similar in their surface markers and multipotency. The specific proteins secreted from adipose stem cells were more adept at cell adhesion, migration, wound healing, and tissue remodeling, while the proteins secreted by placental stem cells were more adept at angiogenesis, cell proliferation, differentiation, cell survival, immunomodulation, and collagen degradation. While these two types of conditioned medium could improve the facial index, the improvement of Melanin index after injection of the adipose stem cell conditioned medium was much more significant. Conclusion. The results suggest that the secreted proteins are ideal cell-free substances for regeneration medicine, especially in the antiaging field.
1. Introduction
Mesenchymal stem cells are a type of adult stem cell with the attributes of self-renewing and multipotency. They can be used in biomedical research, new drug development, and toxicology studies, and so forth [1]. Currently this type of cell is a focal point in clinical research and application [2–4].
The phenomenon that mesenchymal stem cells could improve distal injury organ healing without cell migration and cell differentiation indicates that the cell exosomes play an important role in tissue and organ repair [5–7]. In addition to cell differentiation and proliferation, the secreted proteins also represent an important extension of the stem cells' function [8, 9].
In recent years, adipose stem cells (ASCs) have received great attention in regenerative medicine. The research shows that ASCs are both abundant and varying in activities within our bodies [10]. The ASCs also have very similar characteristics and classification with bone marrow mesenchymal stem cells. Therefore, the proliferation, differentiation, and secretome characteristics of ASCs are considered advantageous in tissue protection, antiapoptosis, and cell replacement [11]. In addition, fat tissue is easily harvested in plastic surgery, making ASC an accessible and ideal kind of adult stem cell.
ASCs have the problem of being limited in cosmetics and clinical application. Due to the requirement of having a large quantity for clinical application, we also chose to use placental stem cells (PSCs) in this secretomics research [12]. Placental tissue is a crucial accessory in protection, nutrition, respiration, and excretion of the embryo. Researchers isolated mesenchymal stem cells from mature placenta and found that PSCs express the typical genes of mesenchymal stem cells as well as some other specific gene [13]. In addition, the placenta is an important nutrient for the development of the fetus and can secrete many developmental factors, such as granulocyte colony-stimulating factor (G-CSF), granulocyte-macrophage colony-stimulating factor (GM-CSF), macrophage colony-stimulating factor (M-CSF), and stem cell factor (SCF) [14]. From this, we speculate that PSCs have unique characteristic of their secretome.
The characterization of secretomics of mesenchymal stem cells was unclear because of the differences in culture condition, culture medium, expansion time, and so forth. In our research, we utilized a method for the extraction of secretory proteins suitable for clinical application and analysis of the secretomes of ASCs and PSCs. Furthermore, we examined the antiaging ability of their secretion factors in human facial skin.
2. Methods
The human trials in this study obtained the informed consent of the volunteers according to the Helsinki Declaration and were approved by the Society of Medical Ethics in Jiangsu Province.
2.1. Isolation of Adipose Stem Cells and Placental Stem Cells
A single sample of 100 mL of adipose tissue was first harvested from a 45-year-old female cosmetic surgery patient. The adipose tissue was washed three times with sterile phosphate-buffered saline solution (PBS). The washed tissue was cut into pieces and digested by collagenase (type I collagenase, Worthington, USA) for forty minutes at 37°C with gentle agitation. Enzyme activity was neutralized with Dulbecco Modified Eagle Medium (DMEM) supplemented with 2% fetal bovine serum (FBS), and the tissues were filtered with 150 μm filters. After centrifugation at 300 g for 10 minutes, the cell pellet was resuspended in DMEM containing 2% human platelet lysate (hPL) and 1% penicillin-streptomycin, cultured, and expanded at 37°C in 5% CO2 incubators.
The placental tissue was washed with PBS to remove the blood. The tissue was minced into small pieces and incubated with 0.2% collagenase II for one hour at 37°C to allow for thorough digestion and filtered through a 250 μm metal sieve to remove the tissue fragments. The cells were collected by centrifugation at 300 g for 10 min and washed three times with PBS. The cell pellet was then suspended in DMEM with 2% hPL and 1% penicillin-streptomycin and incubated at 37°C in 5% CO2 incubators.
2.2. Characteristics of Adipose Stem Cells and Placental Stem Cells
To confirm multipotency, the differentiability of the cells into osteoblasts, adipocytes, and chondrocytes was analyzed. Both types of cells were plated at a density of 3 × 104 cells/cm2 in dishes at passage 3. Once the cells were 90% confluent, the completed medium was substituted with inducing medium (osteogenic medium: completed medium supplemented with 50 μM ascorbic acid, 0.1 μM dexamethasone, and 10 mM b-glycerolphosphate; adipogenic medium: complete medium supplemented with 1.0 μM dexamethasone, 10 mg/mL insulin, 100 μM indomethacin, and 500 μM IBMX; chondrogenic medium: completed medium with 1% penicillin-streptomycin, 1% ITS (insulin, transferrin, and selenium), 100 nM dexamethasone, 50 μg/mL L-ascorbic acid 2-phosphate, 10 ng/mL TGFβ1, and 500 ng/mL BMP6). For up to 20 days, the cells were stained with Oil Red O for adipogenic induction, Alizarin Red S for osteogenic induction, or Safranin O for chondrogenic induction. For fluorescence activated cell sorting (FACS) analysis, the cultured cells were collected and incubated with CD29-fluorescein isothiocyanate (FITC), CD34-FITC, CD71-FITC, and CD90-FITC for 30 min at 4°C and then analyzed using flow cytometry.
2.3. Extraction of Secretory Protein in Conditioned Medium (CM)
The ASCs and PSCs culture dishes were washed three times with PBS and cultured overnight in culture medium consisting of DMEM, Nutrient Mixture F-12 (DMEM/F-12), and 0.2% hPL resulting in about 6–8 × 106 cells. After 24 hours, the cultured medium was collected and fresh serum-free medium was added. The CM was filtered using a 0.22 μm filter, centrifuged with Amicon Ultra 15 mL (MWCO 3 kD, Millipore) at 4°C 4000 g for 30 minutes, and then concentrated about 5 times using ultrafiltration membrane of 3.5 kD with polyethylene glycol at 4°C. Overall, the CM was concentrated about 15 times in the above process. The initial concentration was 1000 mL of 0.389 ± 0.04 mg/mL and the final concentration was 70 mL of 5.989 ± 0.07 mg/mL. Freeze-dry powder was prepared in sterilized penicillin bottles using the Lyophilizer (Boyikang Corp., Beijing, China).
2.4. MALDI-TOF/TOF Analysis
Protein identification experiments were performed at positive mode using the MALDI-TOF/TOF 7090 system. In-gel tryptic digestion of the proteins was performed using an in-gel digestion kit following the protocol recommended by the manufacturer (Shimadzu Kratos, Kyoto). Before deposition onto a MALDI plate, all samples were desalted and concentrated with C18 ZipTip following the recommended protocol (Millipore, Massachusetts). Peptide extracts were eluted in a concentrated solution of 2,5-dihydroxybenzoic acid (12.5 mg/mL) with 50% acetonitrile and 0.1% trifluoroacetic acid in water and spotted onto the MALDI target plate. The TOF spectra were recorded in the reflector mode with a mass range from 500 to 2000 Da. Each spectrum was the cumulative average of 200 laser shots. The spectra were calibrated with the trypsin autodigestion ion peaks m/z (842.51 and 2211.10). The peptide mass fingerprints (PMF) were obtained using the Mascot search engine with a tolerance of 100 ppm and one missed cleavage site.
2.5. Application in Facial Antiaging
18 young volunteers were randomly divided into 3 groups with 6 in each group: ASC-CM group, PSC-CM group, and control group. The CM of ASC or PSC was dissolved into injectable hyaluronic acid (HA) and injected into the facial skin of each person with DermaQueen equipment (Seoul, Korea). The injected depth was 0.1 mm, the interval was about 2 mm, and the volume was 2 μL in each point. The ASC-CM group was injected with HA containing 2 mg/mL ASC-CM, the PSC-CM group was injected with HA containing 2 mg/mL PSC-CM, and the control group was injected with HA containing 0.2% hPL. 15 days after injection, the facial skin of each volunteer was tested with Courage & Khazaka Electronic GmbH (Cologne, Germany). The detection indexes included Erythema, Melanin, Glossymeter, TEWAmeter, and Corneometer. The intensity of skin redness was measured using the Erythema index (Erythema). The Melanin index represents the darkness of the skin (Melanin). Glossymeter is used to measure gloss on skin. TEWAmeter is used to assess the transepidermal water loss (TEWL) and evaluated the water barrier function of the skin. Corneometer can determine the water content in skin.
2.6. Statistical Analysis
The measurement data was described using mean and standard deviation and the difference was analyzed using paired t-test and analysis of variance (ANOVA). All the analyses were performed by Stata (12.0 version) using p < 0.05 as statistically significant.
3. Results
3.1. Characterization of Adipose Stem Cells and Placental Stem Cells
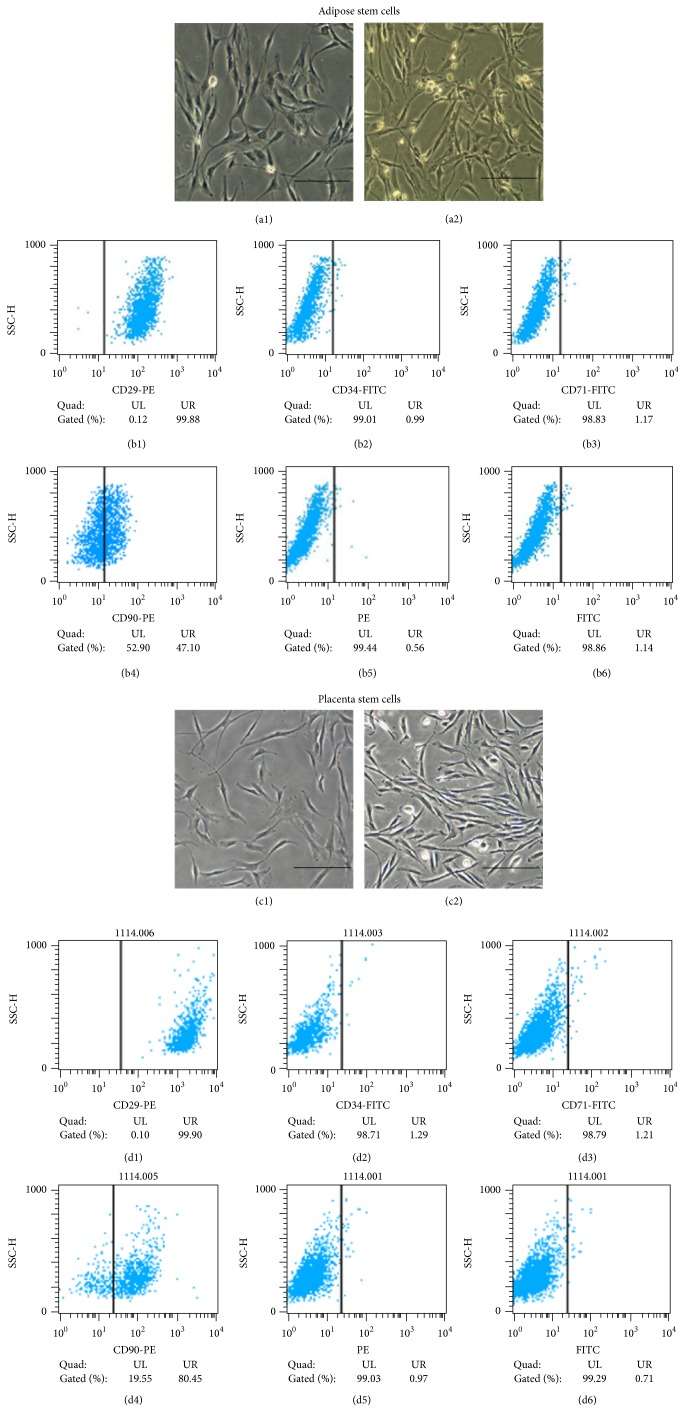
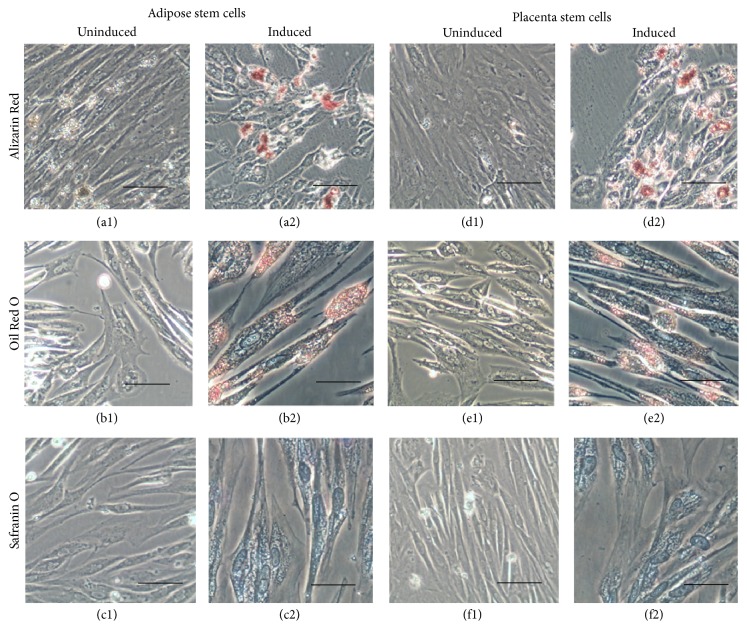
ASCs and PSCs showed the same morphological features as fibroblast-like adherent cells in passage 1 and passage 3 (Figure 1). In FACS analysis, the two types of cells expressed the same surface proteins, such as CD29, CD34, and CD71. In this study, the expression of CD90 was 80.45% in PSC and 47.1% in ASC (Figure 1). In the multipotency analysis, both types of cells successfully differentiated into chondrocytes, adipocytes, and osteocytes (Figure 2). The experiment showed that these two types of cells have very similar characteristic and function consistent with the characteristics of mesenchymal stem cells.
Figure 1.
Characterization of adipose stem cells and placenta stem cells. Both cells showed typical fibroblast-like morphology at passage 1 (a1, c1) and passage 3 (a2, c2). The flow cytometry analysis confirmed that adipose stem cells express CD29 (99.88%, b1) and CD90 (52.9%, b4) but do not express CD34 (0.99%, b2) and CD71 (1.17%, b3); the placenta stem cells express CD29 (99.9%, d1) and CD90 (80.45%, d4) but do not express CD34 (1.29%, d2) and CD71 (1.21%, d3). The FITC (b6, d6) and PE (b5, d5) were performed as blank control. UL refers to upper left quadrant and UR refers to upper right quadrant. Scale bar = 100 μm.
Figure 2.
Multipotent differentiation of adipose stem cells and placenta stem cells. After 20 days of induction, the cells were stained with Alizarin Red, Oil Red, and Safranin O. Both kinds of cells could differentiate into osteocytes (a2, d2), adipocytes (b2, e2), and chondrocytes (c2, f2) successfully. And the uninduced groups were cells without induction as negative control (a1–f1). Scale bar = 50 μm.
3.2. Characterization of Secretory Proteins
In our MALDI-TOF/TOF analysis, 11 proteins were identified in the CM of both types of cells: vascular endothelial growth factor (VEGF), M-CSF, stromal cell-derived factor-1 (SDF-1), transforming growth factor-β (TGF-β), tumor necrosis factor-α (TNF-α), interleukin-1 (IL-1), interleukin-6 (IL-6), hepatocyte growth factor (HGF), insulin-like growth factor-1 (IGF-1), metalloproteinase-2 (MMP-2), and interleukin-8 (IL-8) (Table 1). 11 proteins were exclusively identified in the PSC-CM: monocyte chemotactic protein 1 (MCP-1) (also referred to as chemokine ligand 2, CCL2), SCF, basic fibroblast growth factor (FGF-2), fibroblast growth factor 7 (FGF-7), angiopoietin-1 (ANGP-1), placental growth factor (PGF), adrenomedullin (AM), plasminogen activator (PA), platelet-derived growth factor (PDGF), metalloproteinase-1 (MMP-1), and metalloproteinase-9 (MMP-9) (Table 2). 10 proteins were exclusively identified in ASC-CM: acidic fibroblast growth factor (FGF-1), G-CSF, CM-CSF, pigment epithelium-derived factor (PEDF), metalloproteinase inhibitor 1 (TIMP-1), plasminogen activator inhibitor (PAI), connective tissue growth factor (CTGF), collagen-1, collagen-6, and fibronectin (FN) (Table 3). The details of peptide sequence identification are shown in Supplement 1 of the Supplementary Material available online at http://dx.doi.org/10.1155/2016/7315830.
Table 1.
The characterization of secretory proteins in both ASC-CM and PSC-CM.
| Protein name | UniProtKB ID |
Gene | Molecular function | Biological process | Subcellular location | Family | Function |
|---|---|---|---|---|---|---|---|
| Vascular endothelial growth factor | P15692 | VEGFA | Developmental protein, growth factor, mitogen | Angiogenesis, differentiation | Secreted | PDGF/VEGF growth factor family | Growth factor active in angiogenesis, vasculogenesis, and endothelial cell growth. Induces endothelial cell proliferation, promotes cell migration, inhibits apoptosis, and induces permeabilization of blood vessels |
|
| |||||||
| Macrophage colony-stimulating factor | P09603 | CSF1 | Cytokine, growth factor | Immunity, inflammatory response, and innate immunity | Cell membrane, membrane, secreted | U/A | Cytokine that plays an essential role in the regulation of survival, proliferation, and differentiation of hematopoietic precursor cells, especially mononuclear phagocytes, such as macrophages and monocytes |
|
| |||||||
| Stromal cell-derived factor-1 | P48061 | CXCL12 | Cytokine, growth factor | Chemotaxis | Secreted | Chemokine CC family | Chemoattractant active on T-lymphocytes, monocytes, but not neutrophils |
|
| |||||||
| Transforming growth factor-β | P01137 | TGFB1 | Growth factor, mitogen | U/A | Extracellular matrix, secreted | TGF-beta family | Multifunctional protein that controls proliferation, differentiation, and other functions in many cell types |
|
| |||||||
| Tumor necrosis factor-α | P01375 | TNF | Cytokine | U/A | Cell membrane, membrane, secreted | Tumor necrosis factor family | It is mainly secreted by macrophages and can induce cell death of certain tumor cell lines |
|
| |||||||
| Interleukin-1 | P01583 | IL1A | Cytokine, mitogen, and pyrogen | Inflammatory response | Secreted | IL-1 family | Produced by activated macrophages; IL-1 stimulates thymocyte proliferation by inducing IL-2 release, B-cell maturation and proliferation, and fibroblast growth factor activity |
|
| |||||||
| Interleukin-6 | P05231 | IL-6 | Cytokine, growth factor | Acute phase | Secreted | IL-6 family | Cytokine with a wide variety of biological functions. It is a potent inducer of the acute phase response. Plays an essential role in the final differentiation of B-cells into Ig-secreting cells involved in lymphocyte and monocyte differentiation |
|
| |||||||
| Hepatopoietin-A | P14210 | HGF | Growth factor, serine protease homolog | U/A | U/A | Peptidase S1 family | Is potent mitogen for mature parenchymal hepatocyte cells, seems to be a hepatotrophic factor, and acts as a growth factor for a broad spectrum of tissues and cell types |
|
| |||||||
| Insulin growth factor-1 | P05019 | IGF1 | Growth factor | U/A | Secreted | Insulin family | The insulin-like growth factors, isolated from plasma, are structurally and functionally related to insulin but have a much higher growth-promoting activity |
|
| |||||||
| Matrix metalloproteinase-2 | P08253 | MMP2 | Hydrolase, metalloprotease, and protease | Angiogenesis, collagen degradation | Cytoplasm, extracellular matrix, membrane, mitochondrion, nucleus, secreted | Peptidase M10A family | Ubiquitous metalloproteinase that is involved in diverse functions such as remodeling of the vasculature, angiogenesis, tissue repair, tumor invasion, inflammation, and atherosclerotic plaque rupture |
|
| |||||||
| Interleukin-8 | P10145 | CXCL8 | Cytokine | Chemotaxis, inflammatory response | Secreted | Chemokine CxC family | IL-8 is a chemotactic factor that attracts neutrophils, basophils, and T-cells, but not monocytes. It is also involved in neutrophil activation |
Table 2.
The characterization of specific secretory proteins in PSC-CM.
| Protein name | UniProtKB ID | Gene | Molecular function | Biological process | Subcellular location | Family | Function |
|---|---|---|---|---|---|---|---|
| Monocyte chemotactic protein 1 | P13500 | CCL2 | Cytokine | Chemotaxis, inflammatory response | Secreted | Chemokine CC family | Chemotactic factor that attracts monocytes and basophils but not neutrophils or eosinophils. Augments monocyte antitumor activity |
|
| |||||||
| Kit ligand | P21583 | KITLG | Growth factor | Cell adhesion | Cell membrane, cytoplasm, cytoskeleton, and membrane, secreted | SCF family | Ligand for the receptor-type protein-tyrosine kinase KIT. Plays an essential role in the regulation of cell survival and proliferation, hematopoiesis, stem cell maintenance, gametogenesis, mast cell development, migration and function, and melanogenesis |
|
| |||||||
| Fibroblast growth factor 2 | P09038 | FGF2 | Developmental protein, growth factor, mitogen | Angiogenesis, differentiation | Nucleus, secreted | Heparin-binding growth factors family | Plays an important role in the regulation of cell survival, cell division, angiogenesis, cell differentiation, and cell migration |
|
| |||||||
| Fibroblast growth factor 7 | P21781 | FGF7 | Growth factor, mitogen | U/A | Secreted | Heparin-binding growth factors family | Plays an important role in the regulation of embryonic development, cell proliferation, and cell differentiation |
|
| |||||||
| Angiopoietin-1 | Q15389 | ANGPT1 | Developmental protein | Angiogenesis, differentiation | Secreted | U/A | Plays an important role not only in the regulation of angiogenesis, endothelial cell survival, proliferation, migration, adhesion and cell spreading, and reorganization of the actin cytoskeleton, but also in the maintenance of vascular quiescence. Required for normal angiogenesis and heart development during embryogenesis |
|
| |||||||
| Placenta growth factor | P49763 | PGF | Developmental protein, growth factor, mitogen | Angiogenesis, differentiation | Secreted | PDGF/VEGF growth factor family | Growth factor active in angiogenesis and endothelial cell growth, stimulating their proliferation and migration |
|
| |||||||
| Adrenomedullin | P35318 | ADM | Hormone | U/A | Secreted | Adrenomedullin family | AM and PAMP are potent hypotensive and vasodilator agents. Numerous actions have been reported, most related to the physiologic control of fluid and electrolyte homeostasis |
|
| |||||||
| Plasminogen activator | P00749 | PLAU | Hydrolase, protease, and serine protease | Blood coagulation, fibrinolysis, hemostasis, and plasminogen activation | Secreted | Peptidase S1 family | Specifically cleaves the zymogen plasminogen to form the active enzyme plasmin |
|
| |||||||
| Platelet-derived growth factor | P04085 | PDGFA | Developmental protein, growth factor, mitogen | U/A | Secreted | PDGF/VEGF growth factor family | Growth factor that plays an essential role in the regulation of embryonic development, cell proliferation, cell migration, survival, and chemotaxis |
|
| |||||||
| Matrix metalloproteinase-1 | P03956 | MMP1 | Hydrolase, metalloprotease, and protease | Collagen degradation, host-virus interaction | Extracellular matrix, secreted | Peptidase M10A family | Cleaves collagens of types I, II, and III at one site in the helical domain. Also cleaves collagens of types VII and X |
|
| |||||||
| Matrix metalloproteinase-9 | P14780 | MMP9 | Hydrolase, metalloprotease, and protease | Collagen degradation | Extracellular matrix, secreted | Peptidase M10A family | Cleaves type IV and type V collagen into large C-terminal three-quarter fragments and shorter N-terminal one-quarter fragments |
Table 3.
The characterization of specific secretory proteins in ASC-CM.
| Protein name | UniProtKB ID | Gene | Molecular function | Biological process | Subcellular location | Family | Function |
|---|---|---|---|---|---|---|---|
| Fibroblast growth factor-1 | P05230 | FGF1 | Developmental protein, growth factor, mitogen | Angiogenesis, differentiation | Cytoplasm, nucleus, secreted | Heparin-binding growth factors family | Plays an important role in the regulation of cell survival, cell division, angiogenesis, cell differentiation, and cell migration |
|
| |||||||
| Granulocyte colony-stimulating factor | P09919 | CSF3 | Cytokine, growth factor | U/A | Secreted | IL-6 family | Granulocyte/macrophage colony-stimulating factors are cytokines that act in hematopoiesis by controlling the production, differentiation, and function of 2 related white cell populations of the blood, the granulocytes, and the monocytes-macrophages |
|
| |||||||
| Granulocyte-macrophage colony-stimulating factor | P04141 | CSF2 | Cytokine, growth factor | U/A | Secreted | GM-CSF family | Cytokine that stimulates the growth and differentiation of hematopoietic precursor cells from various lineages, including granulocytes, macrophages, eosinophils, and erythrocytes |
|
| |||||||
| Pigment epithelium-derived factor | P36955 | SERPINF1 | U/A | U/A | Secreted | Serpin family | Neurotrophic protein; induces extensive neuronal differentiation in retinoblastoma cells |
|
| |||||||
| Metalloproteinase inhibitor 1 | P01033 | TIMP-1 | Growth factor, metalloenzyme inhibitor, metalloprotease inhibitor, and protease inhibitor | U/A | Secreted | Protease inhibitor I35 family | Metalloproteinase inhibitor that functions by forming one to one complexes with target metalloproteinases, such as collagenases, and irreversibly inactivates them by binding to their catalytic zinc cofactor |
|
| |||||||
| Plasminogen activator inhibitor 1 | P05121 | SERPINE1 | Protease inhibitor, serine protease inhibitor | U/A | Secreted | Serpin family | Serine protease inhibitor |
|
| |||||||
| Connective tissue growth factor | P29279 | CTGF | U/A | Cell adhesion, DNA synthesis | Extracellular matrix, secreted | CCN family | Major connective tissue mitoattractant secreted by vascular endothelial cells. Promotes proliferation and differentiation of chondrocytes |
|
| |||||||
| Collagen alpha-2(I) chain | P08123 | COL1A2 | U/A | U/A | Extracellular matrix, secreted | Fibrillar collagen family | Type I collagen is a member of group I collagen (fibrillar forming collagen) |
|
| |||||||
| Collagen alpha-1(VI) chain | P12109 | COL6A1 | U/A | Cell adhesion | Extracellular matrix, secreted | Type VI collagen family | Collagen VI acts as a cell-binding protein |
|
| |||||||
| Fibronectin | P02751 | FN1 | U/A | Acute phase, angiogenesis, cell adhesion, and cell shape | Extracellular matrix, secreted | U/A | Fibronectins bind cell surfaces and various compounds including collagen, fibrin, heparin, DNA, and actin. Fibronectins are involved in cell adhesion, cell motility, opsonization, wound healing, and maintenance of cell shape |
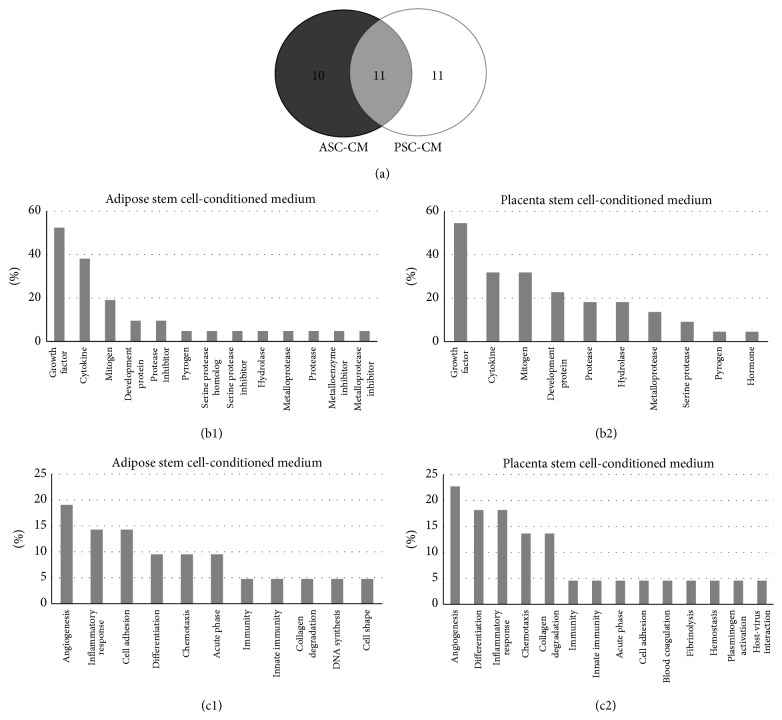
According to the molecular function information in the UniProt database, we found that, in the ASC-CM, 52.4% of total protein types were growth factors, 38.1% were cytokines, and 19% were mitogens. In the PSC-CM, 54.5% of total protein types were growth factors, 31.8% were cytokines, 31.8% were mitogens, 22.7% were development proteins, 18.9% were proteases, and 18.9% were hydrolases (Figure 3). Analyzing according to biological processes, we found that in the ASC-CM 19% of total protein types had angiogenesis function, 14.3% were involved in inflammatory response, 14.3% had cell adhesion function, 9.5% had cell differentiation function, 9.5% had chemotaxis function, and 9.5% were involved in acute phase response. In the PSC-CM, 22.7% of total protein types had angiogenesis function, 18.2% were involved in inflammatory response, 18.2% had cell differentiation function, 13.7% had chemotaxis function, and 13.7% had collagen degradation function (Figure 3). Almost all of the examined proteins were secreted proteins; some of these proteins are also found in the extracellular matrix, cell membrane, nucleus, and so forth.
Figure 3.
Characteristics of secretory proteins. 11 proteins were identified in both ASC-CM and PSC-CM, 11 proteins were specific in PSC-CM, and 10 proteins were in ASC-CM (a). The molecular function information showed that 52.4% of total protein types of ASC-CM were growth factor (b1); 54.5% of PSC-CM was growth factor (b2). The biological process information showed that 19% of total protein types of ASC-CM had angiogenesis function (c1); 22.7% of total protein types of PSC-CM had angiogenesis function (c2).
The protein-protein interactions of secreted proteins of ASC and PSC were analyzed by STRING 10.0. The interaction network was drawn with the thickness of connecting lines representing the strength of evidence (Figure 4). We found that all proteins could interact with others; some proteins had even more active functions for multiple interaction points. The relatively more active proteins in both kinds of CM were IL6, IL8, MMP-2, TGF-β, VEGF, IGF-1, and SDF-1 (also known as CXCL12). In the ASC-CM, the unique active proteins were CTGF, FN, TIMP-1, and PEDF (also known as SERPINF1). In the PSC-CM, the unique active proteins were FGF-2, CCL2, FGF-7, MMP-1, and MMP-9. This research shows that most of active proteins in the ASC-CM and the PSC-CM are the same. Comparatively, the ASC-CM had more proteins involved in the function of cell adhesion promotion, metalloproteinase inhibition, and plasminogen activator; the PSC-CM had more proteins involved in the function of cell proliferation promotion, differentiation and migration, inflammatory response, and collagen degeneration.
Figure 4.
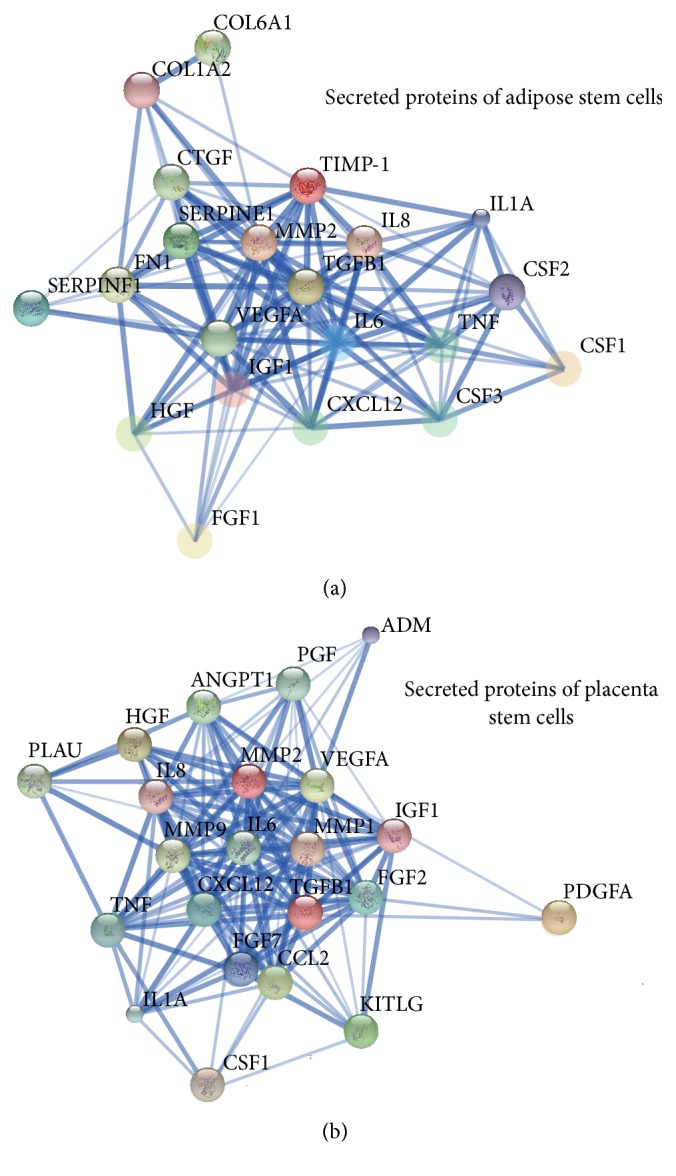
The analysis of protein-protein interaction of secreted proteins of ASC and PSC. The thickness of line represents the strength of evidence. All proteins could interact with others whether in ASC-CM (a) or PSC-CM (b). The relative active proteins in two kinds of CM were IL6, IL8, MMP-2, TGFB1, VEGFA, IGF-1, and CXCL12 (SDF-1). In ASC-CM, the specific active proteins were CTGF, FN, TIMP-1, and SERPINE-1 (PEDF). And in PSC-CM, the specific active proteins were FGF-2, CCL2, FGF-7, MMP-1, and MMP-9.
3.3. Antiaging Function in Human Facial Skin
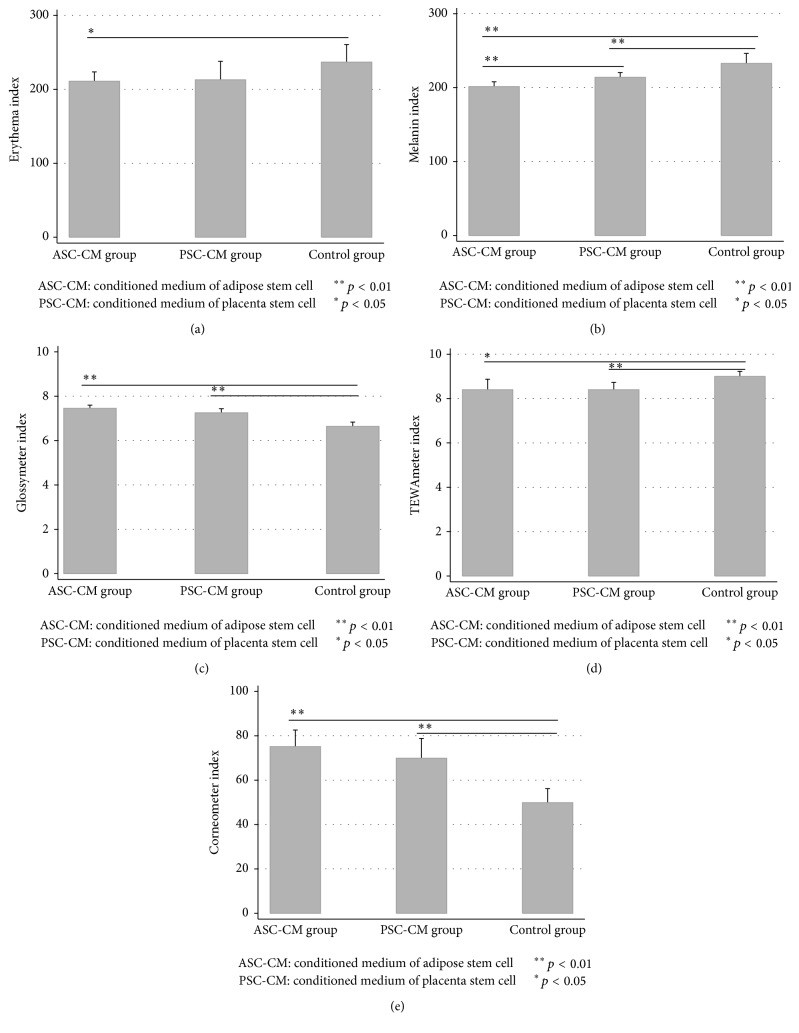
There were no significant differences in the facial indexes between the three groups before injection. 15 days after injection, indexes of Erythema, Melanin, Glossymeter, TEWAmeter, and Corneometer of the ASC-CM group showed improvement compared to the control group. Indexes of Melanin, Glossymeter, TEWAmeter, and Corneometer in PSC-CM group also demonstrated improvement compared to the control group. Only the Melanin index of the ASC-CM group was significantly lower than that of the PSC-CM group (Figure 5). According to ANOVA analysis, there were significant differences among three groups in Erythema index (F = 10.23, p < 0.01), Melanin index (F = 47.97, p < 0.01), Glossymeter (F = 85.13, p < 0.01), TEWAmeter (F = 13.69, p < 0.01), and Corneometer (F = 47.42, p < 0.01), respectively.
Figure 5.
Antiaging functions of ASC-CM and PSC-CM in human facial skin. 15 days after injection, all indexes in ASC-CM group were more improved than those in control group (a–e). All indexes except Erythema in PSC-CM group were more improved than control group (b–e). Only the Melanin index in ASC-CM group was significantly lower than PSC-CM group (b).
4. Discussion
In this study, ASCs and PSCs were used as seed cells because adipose tissue and placenta are ideal stem cell sources in daily medical practice. Furthermore, ASCs and PSCs are also ideal substitutes for autologous bone marrow mesenchymal stem cells due to their similar characteristics. In our study, these two kinds of cells demonstrated similar surface markers and multipotency as other mesenchymal stem cells; these results are the same as those of previous studies.
Utilizing CM is a novel cell-free treatment strategy for clinical applications; concentrated CM has partial function of mesenchymal stem cells [15, 16]. Accumulating evidence indicates that the healing effects of MSCs are mainly related to their unique paracrine properties [17]. In our previous research, ASC-CM was proven to have the ability to promote full-thickness defect skin model healing and human skin laser injury healing [18]. In another research, ASC-CM promoted mouse liver regeneration and increased albumin expression [19]. There has been far less basic and clinical research with PSCs and their CM. In in vitro research, the secretory factors of human chorion-derived stem cells are shown to enhance the activation of human fibroblasts and influence wound healing [20].
hPL was used as the serum-free medium for our cell culture because hPL does not induce an immune response when applied in vivo and provides adequate nutrition for cell culture [21–23]. In this research, hPL did not affect the expression of cell surface markers or the characteristics of multipotent differentiation. Therefore, hPL is an ideal cell culture medium supplement in serum-free and cell-free research.
In MALDI-TOF/TOF analysis, growth factor proteins accounted for 52.4% and 54.5% of the total secreted proteins in ASC-CM and PSC-CM, respectively. These growth factors can stimulate cell growth, proliferation, differentiation, and maturation [24, 25]. For example, VEGF and FGF promote the formation of artery and vessel [26, 27]. Cytokines, which are important cell signaling molecules, accounted for 38.1% and 31.8% of total secreted proteins in ASC-CM and PSC-CM, respectively. Generally, cytokines are associated with hematopoietic cells and immune system cells; they include chemokine, interferon, interleukin, lymphokine, and tumor necrosis factor [28, 29]. Cytokines are important in modulating the balance between humoral and cell-based immune responses and could be a neutral term to affect cell proliferation [30]. However, some cytokines promote cell division as growth factors, such as G-CSF, M-CSF, and GM-CSF. Mitogen is a chemical substance that stimulates mitosis [31]. Development proteins play an important role in the development of embryos. In addition, many secretory proteins have more than one molecular function. In the analysis of biological processes, we found that proteins involved with angiogenesis and inflammation comprised a major portion in both kinds of CM. However, in the ASC-CM, there were a greater proportion of cell adhesion proteins; in the PSC-CM there was a greater proportion of proteins related to differentiation, chemotaxis, and collagen degradation. This may indicate that ASC-CM is better suited for cell adhesion and that PSC-CM is more appropriate for promoting cell differentiation and chemotaxis.
In the analysis of the protein-protein interaction network, the proteins in the inner layer of the network are considered to have more active protein function. The active proteins in both types of CM included IL6, IL8, MMP2, TGFB1, VEGFA, IGF-1, and CXCL12. Interleukins are a group of cytokines that promotes the development and differentiation of T and B lymphocytes and hematopoietic cells [32]. IL-6 and IL-8 promote hematopoietic stem cell proliferation and differentiation and regulate immune response and chemotaxis [33, 34]. MMP2 is involved in the breakdown of extracellular matrix, such as during embryonic development, reproduction, and tissue remodeling [35]. TGF-B is a multifunctional protein that controls cells proliferation, differentiation, and other functions [36]. VEGFA is active in angiogenesis, vasculogenesis, and endothelial cell growth [37]. IGF has growth-promoting activity [38]. CXCL12 is strongly chemotactic for lymphocytes and mesenchymal stem cells and plays an important role in angiogenesis [39]. The above indicates that the functions of angiogenesis, cell proliferation, differentiation, inflammatory response, and chemotaxis were found in proteins of both kinds of CM.
The unique active proteins in ASC-CM include CTGF, FN, TIMP-1, and SERPINE-1 (PAI). CTGF is a major connective tissue mitoattractant [40]. Fibronectins promote cell motility, adhesion, and increased wound healing [41]. TIMP-1 inhibits metalloproteinase irreversibly by binding to its catalytic zinc cofactor [42]. SERPINE-1 is a type of serine protease inhibitor [43]. Overall, these proteins are more adept at cell adhesion, migration, wound healing, and tissue remodeling.
The unique active proteins in PSC-CM include FGF2, FGF7, CCL2, MMP1, and MMP9. FGF2 and FGF7 play an important role in the regulation of embryonic development, cell survival, cell division, angiogenesis, cell differentiation, and cell migration [44]. CCL2 is a chemotactic factor that attracts monocytes and basophils and augments monocyte antitumor activity [45]. MMP1 and MMP9 cleave types I, II, III, IV, V, VII, and X collagen [46]. The above indicates that proteins unique to PSC-CM are more adept at angiogenesis, cell proliferation, differentiation, cell survival, immunomodulation, and collagen degradation.
Facial aging is a complex process that affects both 3D shape and textures, such as dryness, coarseness, lost luster, and wrinkles. ASCs are most commonly used in plastic surgery as seed cells. ASC-CM has also been proven to play an important role in the prevention of photoaging dermal cells; our previous study has proven that TGF-b is an essential protein in antiaging. In order to compare the effects of ASC-CM and PSC-CM in skin aging, we injected both CM into facial skin and analyzed the facial skin index. After the injection, both CM greatly improved the facial indexes, such as Erythema and Melanin. ASC-CM and PSC-CM were only different in the index of Melanin. Although the secretory proteins of CM of both cell types were detected, the mechanism of antiaging still requires further inquiry.
Supplementary Material
The details of peptide sequence identification by MALDI-TOF/TOF analysis: The peptide mass fingerprints (PMF) were obtained using the Mascot search engine with a tolerance of 100 ppm and one missed cleavage site. The peptide sequence of fingerprints and matched protein names were showed in this Table. Mr (expt) showed the molecular mass of peptide in experiment; Mr(calc) showed the molecular mass of peptide in calculation. Position indicated the position of detected peptide in protein. 1.1 showed the peptides harvest from placental stem cells' conditioned medium; 1.2 showed the peptides harvest from adipose stem cells' conditioned medium.
Acknowledgment
All the authors give thanks to Bingrong Zhou for his valuable experimental help.
Conflict of Interests
The authors have declared that no competing interests exist.
Authors' Contribution
Yan Xu and Shilei Guo contributed equally to this paper.
References
- 1.Zolochevska O., Shearer J., Ellis J., et al. Human adipose-derived mesenchymal stromal cell pigment epithelium-derived factor cytotherapy modifies genetic and epigenetic profiles of prostate cancer cells. Cytotherapy. 2014;16(3):346–356. doi: 10.1016/j.jcyt.2013.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Trounson A., McDonald C. Stem cell therapies in clinical trials: progress and challenges. Cell Stem Cell. 2015;17(1):11–22. doi: 10.1016/j.stem.2015.06.007. [DOI] [PubMed] [Google Scholar]
- 3.Isakson M., de Blacam C., Whelan D., McArdle A., Clover A. J. Mesenchymal stem cells and cutaneous wound healing: current evidence and future potential. Stem Cells International. 2015;2015:12. doi: 10.1155/2015/831095.831095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Munir H., McGettrick H. M. Mesenchymal stem cell therapy for autoimmune disease: risks and rewards. Stem Cells and Development. 2015;24(18):2091–2100. doi: 10.1089/scd.2015.0008. [DOI] [PubMed] [Google Scholar]
- 5.Zhao Y., Sun X., Cao W., et al. Exosomes derived from human umbilical cord mesenchymal stem cells relieve acute myocardial ischemic injury. Stem Cells International. 2015;2015:12. doi: 10.1155/2015/761643.761643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kang K., Ma R., Cai W., et al. Exosomes secreted from CXCR4 overexpressing mesenchymal stem cells promote cardioprotection via Akt signaling pathway following myocardial infarction. Stem Cells International. 2015;2015:14. doi: 10.1155/2015/659890.659890 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shabbir A., Cox A., Rodriguez-Menocal L., Salgado M., Badiavas E. V. Mesenchymal stem cell exosomes induce proliferation and migration of normal and chronic wound fibroblasts, and enhance angiogenesis in vitro. Stem Cells and Development. 2015;24(14):1635–1647. doi: 10.1089/scd.2014.0316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Clabaut A., Grare C., Léger T., Hardouin P., Broux O. Variations of secretome profiles according to conditioned medium preparation: the example of human mesenchymal stem cell-derived adipocytes. Electrophoresis. 2015;36(20):2587–2593. doi: 10.1002/elps.201500086. [DOI] [PubMed] [Google Scholar]
- 9.Gallina C., Turinetto V., Giachino C. A new paradigm in cardiac regeneration: the mesenchymal stem cell secretome. Stem Cells International. 2015;2015:10. doi: 10.1155/2015/765846.765846 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Francesco F., Ricci G., D'Andrea F., Nicoletti G. F., Ferraro G. A. Human adipose stem cells: from bench to bed-side. Tissue Engineering Part B Reviews. 2015;21(6):572–584. doi: 10.1089/ten.TEB.2014.0608. [DOI] [PubMed] [Google Scholar]
- 11.Busser H., Najar M., Raicevic G., et al. Isolation and characterization of human mesenchymal stromal cell subpopulations: comparison of bone marrow and adipose tissue. Stem Cells and Development. 2015;24(18):2142–2157. doi: 10.1089/scd.2015.0172. [DOI] [PubMed] [Google Scholar]
- 12.Oliveira M. S., Barreto-Filho J. B. Placental-derived stem cells: culture, differentiation and challenges. World Journal of Stem Cells. 2015;7(4):769–775. doi: 10.4252/wjsc.v7.i4.769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fukuchi Y., Nakajima H., Sugiyama D., Hirose I., Kitamura T., Tsuji K. Human placenta-derived cells have mesenchymal stem/progenitor cell potential. STEM CELLS. 2004;22(5):649–658. doi: 10.1634/stemcells.22-5-649. [DOI] [PubMed] [Google Scholar]
- 14.Dame J. B., Christensen R. D., Juul S. E. The distribution of granulocyte-macrophage colony-stimulating factor and its receptor in the developing human fetus. Pediatric Research. 1999;46(4):358–366. doi: 10.1203/00006450-199910000-00002. [DOI] [PubMed] [Google Scholar]
- 15.Danieli P., Malpasso G., Ciuffreda M. C., et al. Conditioned medium from human amniotic mesenchymal stromal cells limits infarct size and enhances angiogenesis. Stem Cells Translational Medicine. 2015;4(5):448–458. doi: 10.5966/sctm.2014-0253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shen C., Lie P., Miao T., et al. Conditioned medium from umbilical cord mesenchymal stem cells induces migration and angiogenesis. Molecular Medicine Reports. 2015;12(1):20–30. doi: 10.3892/mmr.2015.3409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Li F., Zhao S. Z. Mesenchymal stem cells: potential role in corneal wound repair and transplantation. World Journal of Stem Cells. 2014;6(3):296–304. doi: 10.4252/wjsc.v6.i3.296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhou B.-R., Xu Y., Xu Y., et al. The effect of conditioned media of adipose-derived stem cells on wound healing after ablative fractional carbon dioxide laser resurfacing. BioMed Research International. 2013;2013:9. doi: 10.1155/2013/519126.519126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lee S. K., Lee S. C., Kim S.-J. A novel cell-free strategy for promoting mouse liver regeneration: utilization of a conditioned medium from adipose-derived stem cells. Hepatology International. 2015;9(2):310–320. doi: 10.1007/s12072-014-9599-4. [DOI] [PubMed] [Google Scholar]
- 20.Kim M. K., Seo B. F., Kim K. J., Lee S.-J., Ryu Y. H., Rhie J. W. Secretory factors of human chorion-derived stem cells enhance activation of human fibroblasts. Cytotherapy. 2015;17(3):301–309. doi: 10.1016/j.jcyt.2014.10.007. [DOI] [PubMed] [Google Scholar]
- 21.Li C. Y., Wu X. Y., Tong J. B., et al. Comparative analysis of human mesenchymal stem cells from bone marrow and adipose tissue under xeno-free conditions for cell therapy. Stem Cell Research &; Therapy. 2015;6, article 55 doi: 10.1186/s13287-015-0066-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shih D. T.-B., Burnouf T. Preparation, quality criteria, and properties of human blood platelet lysate supplements for ex vivo stem cell expansion. New Biotechnology. 2015;32(1):199–211. doi: 10.1016/j.nbt.2014.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krieger J. E., Blande I. S., Bassaneze V., et al. Adipose tissue mesenchymal stem cell expansion in animal serum-free medium supplemented with autologous human platelet lysate. Transfusion. 2009;49(12):2680–2685. doi: 10.1111/j.1537-2995.2009.02346.x. [DOI] [PubMed] [Google Scholar]
- 24.Zhang X., Wang Y., Gao Y., et al. Maintenance of high proliferation and multipotent potential of human hair follicle-derived mesenchymal stem cells by growth factors. International Journal of Molecular Medicine. 2013;31(4):913–921. doi: 10.3892/ijmm.2013.1272. [DOI] [PubMed] [Google Scholar]
- 25.Qian D., Gong J., He Z., et al. Bone marrow-derived mesenchymal stem cells repair necrotic pancreatic tissue and promote angiogenesis by secreting cellular growth factors involved in the SDF-1α/CXCR4 axis in rats. Stem Cells International. 2015;2015:20. doi: 10.1155/2015/306836.306836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Oh M. K., Park H. J., Lee J. H., Bae H. M., Kim I. S. Single chain precursor prohaptoglobin promotes angiogenesis by upregulating expression of vascular endothelial growth factor (VEGF) and VEGF receptor2. FEBS Letters. 2015;589(9):1009–1017. doi: 10.1016/j.febslet.2015.03.006. [DOI] [PubMed] [Google Scholar]
- 27.Ito Y., Tsurushima H., Sato M., et al. Angiogenesis therapy for brain infarction using a slow-releasing drug delivery system for fibroblast growth factor 2. Biochemical and Biophysical Research Communications. 2013;432(1):182–187. doi: 10.1016/j.bbrc.2013.01.013. [DOI] [PubMed] [Google Scholar]
- 28.Schürch C. M., Riether C., Ochsenbein A. F. Cytotoxic CD8+ T cells stimulate hematopoietic progenitors by promoting cytokine release from bone marrow mesenchymal stromal cells. Cell Stem Cell. 2014;14(4):460–472. doi: 10.1016/j.stem.2014.01.002. [DOI] [PubMed] [Google Scholar]
- 29.Búfalo M. C., Bordon-Graciani A. P., Conti B. J., de Assis Golim M., Sforcin J. M. The immunomodulatory effect of propolis on receptors expression, cytokine production and fungicidal activity of human monocytes. Journal of Pharmacy and Pharmacology. 2014;66(10):1497–1504. doi: 10.1111/jphp.12279. [DOI] [PubMed] [Google Scholar]
- 30.Li P., Spolski R., Liao W., Leonard W. J. Complex interactions of transcription factors in mediating cytokine biology in T cells. Immunological Reviews. 2014;261(1):141–156. doi: 10.1111/imr.12199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Arthur J. S. C., Ley S. C. Mitogen-activated protein kinases in innate immunity. Nature Reviews Immunology. 2013;13(9):679–692. doi: 10.1038/nri3495. [DOI] [PubMed] [Google Scholar]
- 32.Szklarczyk D., Franceschini A., Wyder S., et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Research. 2015;43(1):D447–D452. doi: 10.1093/nar/gku1003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lee G.-L., Chang Y.-W., Wu J.-Y., et al. TLR 2 induces vascular smooth muscle cell migration through cAMP response element-binding protein-mediated interleukin-6 production. Arteriosclerosis, Thrombosis, and Vascular Biology. 2012;32(11):2751–2760. doi: 10.1161/ATVBAHA.112.300302. [DOI] [PubMed] [Google Scholar]
- 34.Ruan Y., Ji X., Wen M., Zhu X., Fu X. Interleukin 8 enhances the immune response of ducks to avian influenza vaccine. Acta Virologica. 2014;58(4):356–358. doi: 10.4149/av_2014_04_356. [DOI] [PubMed] [Google Scholar]
- 35.Ferrer M., Rodriguez H., Zara L., Yu Y., Xu W., Oko R. MMP2 and acrosin are major proteinases associated with the inner acrosomal membrane and may cooperate in sperm penetration of the zona pellucida during fertilization. Cell and Tissue Research. 2012;349(3):881–895. doi: 10.1007/s00441-012-1429-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vaidya A., Kale V. P. TGF-β signaling and its role in the regulation of hematopoietic stem cells. Systems and Synthetic Biology. 2015;9(1):1–10. doi: 10.1007/s11693-015-9161-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kofler N. M., Simons M. Angiogenesis versus arteriogenesis: neuropilin 1 modulation of VEGF signaling. F1000Prime Reports. 2015;7, article 26 doi: 10.12703/p7-26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Livingstone C. Insulin-like growth factor-I (IGF-I) and clinical nutrition. Clinical Science. 2013;125(6):265–280. doi: 10.1042/CS20120663. [DOI] [PubMed] [Google Scholar]
- 39.Shi J., Wei Y., Xia J., et al. CXCL12-CXCR4 contributes to the implication of bone marrow in cancer metastasis. Future Oncology. 2014;10(5):749–759. doi: 10.2217/fon.13.193. [DOI] [PubMed] [Google Scholar]
- 40.Dendooven A., Gerritsen K. G., Nguyen T. Q., Kok R. J., Goldschmeding R. Connective tissue growth factor (CTGF/CCN2) ELISA: a novel tool for monitoring fibrosis. Biomarkers. 2011;16(4):289–301. doi: 10.3109/1354750x.2011.561366. [DOI] [PubMed] [Google Scholar]
- 41.Schwarzbauer J. E., DeSimone D. W. Fibronectins, their fibrillogenesis, and in vivo functions. Cold Spring Harbor Perspectives in Biology. 2011;3(7) doi: 10.1101/cshperspect.a005041.a005041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Du S., Mao G., Zhu T., Luan Z., Du Y., Gu H. TIMP1 in conditioned media of human adipose stromal cells protects neurons against oxygen-glucose deprivation injury. Neuroscience Letters. 2015;584:56–59. doi: 10.1016/j.neulet.2014.09.045. [DOI] [PubMed] [Google Scholar]
- 43.Yasar Yildiz S., Kuru P., Toksoy Oner E., Agirbasli M. Functional stability of plasminogen activator inhibitor-1. The Scientific World Journal. 2014;2014:11. doi: 10.1155/2014/858293.858293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Inchovska M., Ogneva V., Martinova Y. Role of FGF1, FGF2 and FGF7 in the development of the pancreas from control and streptozotocin-treated hamsters. Cell Proliferation. 2006;39(6):537–550. doi: 10.1111/j.1365-2184.2006.00410.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bauer D., Redmon N., Mazzio E., et al. Diallyl disulfide inhibits TNFα induced CCL2 release through MAPK/ERK and NF-Kappa-B signaling. Cytokine. 2015;75(1):117–126. doi: 10.1016/j.cyto.2014.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Van Doren S. R. Matrix metalloproteinase interactions with collagen and elastin. Matrix Biology. 2015;44–46:224–231. doi: 10.1016/j.matbio.2015.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
The details of peptide sequence identification by MALDI-TOF/TOF analysis: The peptide mass fingerprints (PMF) were obtained using the Mascot search engine with a tolerance of 100 ppm and one missed cleavage site. The peptide sequence of fingerprints and matched protein names were showed in this Table. Mr (expt) showed the molecular mass of peptide in experiment; Mr(calc) showed the molecular mass of peptide in calculation. Position indicated the position of detected peptide in protein. 1.1 showed the peptides harvest from placental stem cells' conditioned medium; 1.2 showed the peptides harvest from adipose stem cells' conditioned medium.