Abstract
Indirect challenges act to provoke bronchoconstriction by causing the release of endogenous mediators and are used to identify airway hyper-responsiveness. This paper reviews the historical development of challenges, with exercise, eucapnic voluntary hyperpnoea (EVH) of dry air, wet hypertonic saline, and with dry powder mannitol, that preceded their use in clinical practice. The first challenge developed for clinical use was exercise. Physicians were keen for a standardized test to identify exercise-induced asthma (EIA) and to assess the effect of drugs such as disodium cromoglycate. EVH with dry air became a surrogate for exercise to increase ventilation to very high levels. A simple test was developed with EVH and used to identify EIA in defence force recruits and later in elite athletes. The research findings with different conditions of inspired air led to the conclusion that loss of water by evaporation from the airway surface was the stimulus to EIA. The proposal that water loss caused a transient increase in osmolarity led to the development of the hypertonic saline challenge. The wet aerosol challenge with 4.5% saline, provided a known osmotic stimulus, to which most asthmatics were sensitive. To simplify the osmotic challenge, a dry powder of mannitol was specially prepared and encapsulated. The test pack with different doses and an inhaler provided a common operating procedure that could be used at the point of care. All these challenge tests have a high specificity to identify currently active asthma. All have been used to assess the benefit of treatment with inhaled corticosteroids. Over the 50 years, the methods for testing became safer, less complex, and less expensive and all used forced expiratory volume in 1 sec to measure the response. Thus, they became practical to use routinely and were recommended in guidelines for use in clinical practice.
Keywords: exercise, eucapnic hyperpnoea, hypertonic saline, dry powder mannitol, disodium cromoglycate, indirect challenges, airway hyperresponsiveness
‘Indirect challenges act by causing the release of endogenous mediators that cause airway smooth muscle to contract, with or without effect in inducing microvascular leakage. Because the responses to these challenges are modified or even completely inhibited by inhaled steroids, the airway response to these challenges may be a close reflection of active airway inflammation’ (1). This review covers some of the scientific history of the development of the ‘indirect’ challenges that have become established in clinical practice for bronchial provocation testing. These are exercise, eucapnic voluntary hyperpnoea of dry air, a wet aerosol of hypertonic saline, and a dry powder aerosol of mannitol (2).
Introduction
In the 1960s, there were several events that ultimately led to the development of ‘indirect’ challenge tests to identify airway hyperresponsiveness (AHR). The first was the recognition that children with asthma could have an ‘attack’ provoked by exercise, a non-immunological stimulus (3, 4). This ‘attack’ was identified by a transient increase in airways resistance after exercise. The terms first used to describe it were exercise-induced asthma (EIA) (5), exercise-induced bronchospasm (6), and exercise-induced bronchoconstriction (7).
Another event was the commercial release in 1968 of the drug disodium cromoglycate (DSCG) given by inhalation as a dry powder (8). The mode of action of DSCG, from in vitro studies, suggested that it stabilised the mast cell membrane, inhibiting the immunological (via IgE) release of histamine in response to inhaled allergen. DSCG had no direct action on airway smooth muscle (8), yet it was very effective in preventing both the early- and late-phase airway response to inhaled allergen (9, 10). At the time, allergen inhalation challenge, also an indirect challenge was used on a case-by-case basis particularly to identify sensitivity to an occupational exposure. These allergen challenges were difficult to standardize and unsuitable for use to evaluate DSCG, either in children or in large population studies. DSCG, however, had also been reported to inhibit EIA in adults (11, 12). The potential for children with EIA to benefit from DSCG was quickly recognised and investigators focused on developing a ‘standardized’ exercise test and on identifying an easy to measure index for change in lung function (13–16).
Exercise
In quest of a suitable protocol
An early protocol to identify EIA in children, in the USA, was 5 min of cycle ergometer exercise of sufficient intensity to reach a heart rate of 180 bpm (13, 17). The maximum decrease or fall in forced expiratory volume in 1 sec (FEV1) recorded in the 20 min after exercise, expressed as a percentage of the pre-exercise value, was used to measure the response. Eighteen (72%) asthmatics had a % fall ≥10% in FEV1. The response was characterised as mild <10% fall, moderate 10–25%, and severe >25%. Only four normal non-asthmatic subjects were tested and none had a sustained fall in FEV1.
As running was identified as more potent for provoking EIA than cycling, protocols were also developed for exercising on a motorised treadmill (18, 19). Peak expiratory flow (PEF) was often used, rather than FEV1, because it could be measured quickly during exercise to document a rise as well as a fall after exercise (20). The coefficient of variation for repeated measures of PEF was 4.1%. For running exercise, the upper limit for the % fall (mean+2SD) in PEF in normal non-asthmatic children was found to be 10% (21).
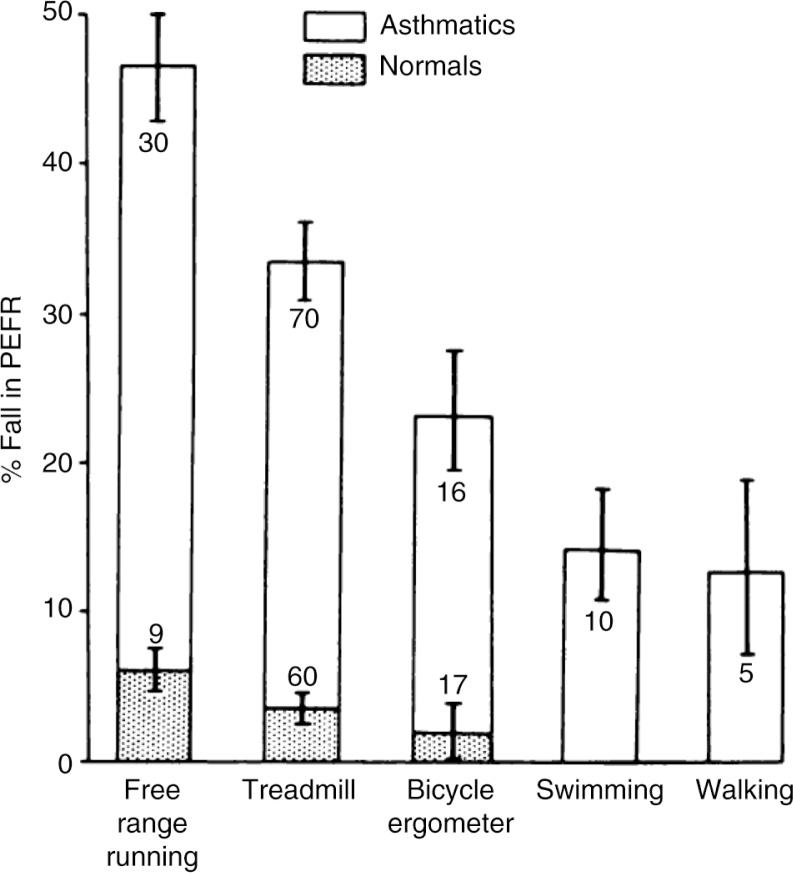
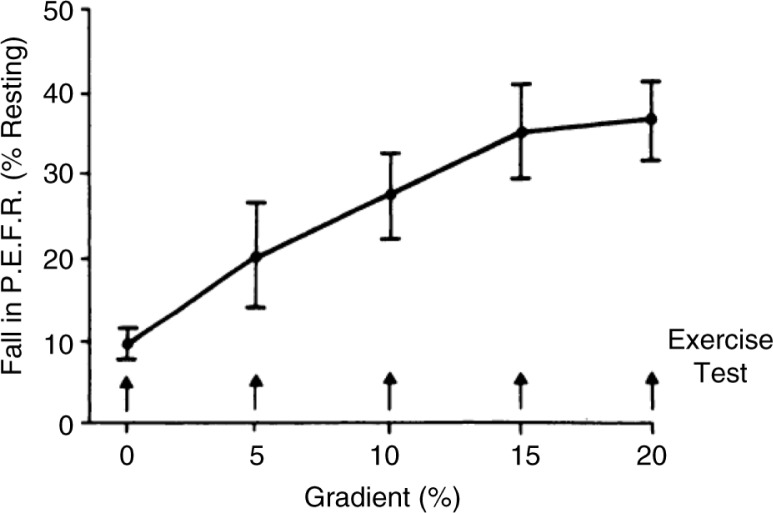
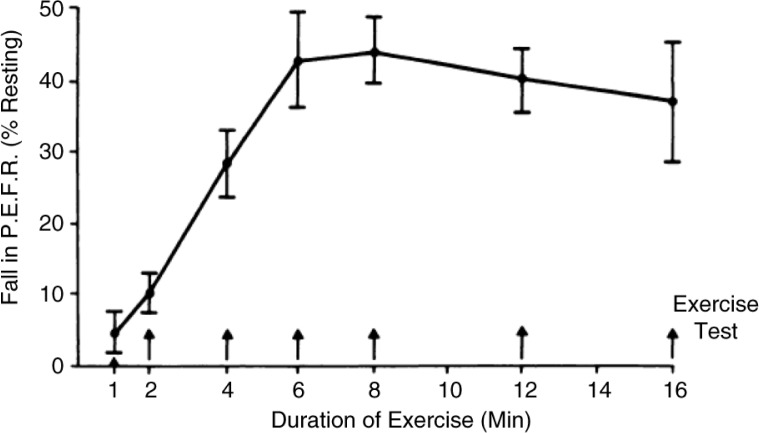
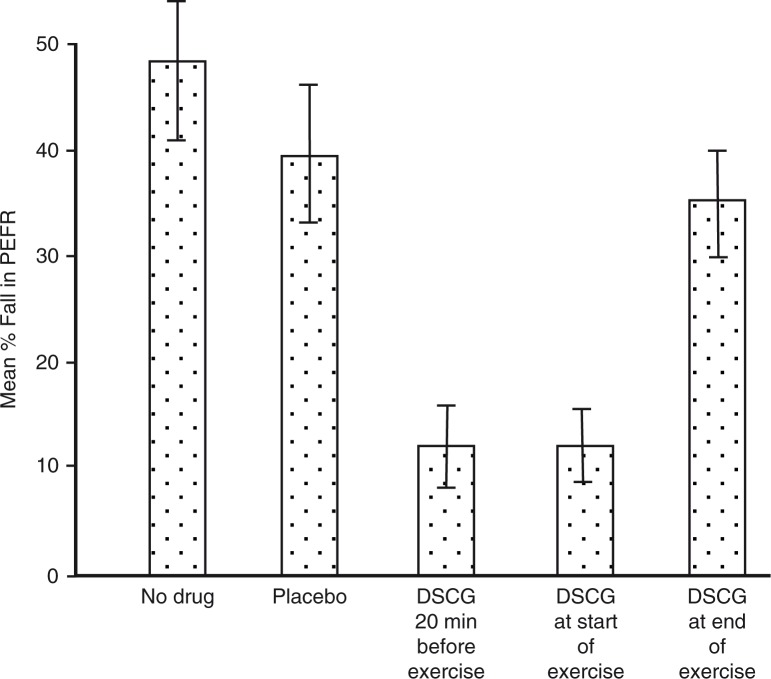
In addition to type of exercise (Fig. 1), the intensity (Fig. 2), and duration (Fig. 3) of the running exercise were also found to be important, as was the interval between repeated tests (14, 21). By 1973, Godfrey et al. (14) in the UK concluded that ‘the greatest amount of exercise-induced asthma is found after 6–8 min of steady-state running at a work rate equivalent to two thirds of work capacity. Exercise may be repeated every 2 h throughout the day without any diminution of its causing post-exercise bronchoconstriction.’ This ‘standardized’ test provided a model to test the efficacy and duration of action for DSCG in children (14). Importantly, the drug was shown to be effective when given at the start of exercise (Fig. 4) and to inhibit EIA even in those who were non-atopic (22, 23).
Fig. 1.
Exercise-induced asthma expressed as a % fall from baseline PEFR in groups of asthmatic and normal subjects. Although the individual subjects were not all the same in each group they were all working at the same relative work load. The numbers indicate the number of subjects. The bars indicate±SEM. Reproduced with permission from (14).
Fig. 2.
Effect of gradient (work load) on asthma, induced by treadmill running, at a constant speed for 6 min. Each point represents the mean of tests in nine subjects who performed each gradient on a separate day. The bars indicate±SEM. Reproduced with permission from (14).
Fig. 3.
Effect of duration of exercise on asthma, induced by treadmill running, at constant speed and slope. Each point represents the mean of tests in 10 subjects who performed each duration on a separate day. The response plateaued at 6–8 min. The bars indicate±SEM. Reproduced with permission from (14).
Fig. 4.
Disodium cromoglycate, taken 20 min, and at the start of running exercise and at the end of exercise compared with response after placebo and on a control day. The bars indicate±SEM. Adapted from (22).
A summary of research in EIA was published in 1974 following a symposium in Seattle, WA, USA. The contributors reported that EIA was most efficiently identified by measuring a fall in PEF or FEV1 of either 10% or 15% of the pre-exercise value (16, 24–26). The cycling was confirmed as poorly sensitive to identify EIA although it had some technical advantages (27). The use of a motorised treadmill was confirmed, and 6 min of running up a gradient of 10% at 5 km/h was recommended with the best reproducibility being obtained by repeating the test within 1 week (26). Free range running was also recognised as a useful test with the majority of those responding doing so within 5 min of ceasing exercise (28). In 1975, a 15% fall in FEV1 was identified as abnormal (24) though later studies identified a 10% cut off to define EIA (29).
While standardized protocols for exercise tests in children were published in the UK in the mid-1970s (14, 21, 30), the first clinical guidelines in the USA for testing both in adults and children were published in 1979 (31). These guidelines for adults proposed that the minimum equipment should provide for a continuous strip chart record of an ‘electrocardiogram’ and a stepped (progressive) protocol be used for the initial testing. Suggestions were also made for the steady-state exercise test of 5–8 min to be preceded by 4 min of increasing the speed and incline of the treadmill to reach the required heart rate (90% predicted maximum) and oxygen consumption (30–40 ml/min/kg) (32).
Influence of humidity
Over the years, some investigators had noted that there was a seasonal variation in severity in some subjects and it was suggested that ‘changing patterns of humidity, temperature and wind velocity’ maybe responsible for increased susceptibility to EIA (28, 33).
In 1976, Weinstein et al. (34) at an Academy of Allergy meeting in Puerto Rico reported inhibition of EIA in 10 of 13 subjects after inhaling an aerosol of ultrasonically nebulized normal saline via a mask during running exercise. This observation was quickly followed up by many groups wanting to confirm this finding using water, inspired as a gas, rather than normal saline as an aerosol.
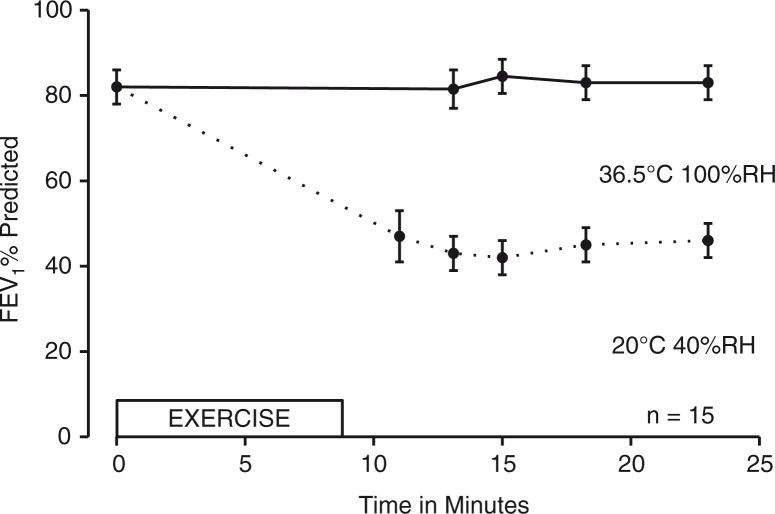
In 1977, Bar-Or et al. (35) had children run in a climate chamber at 25–26°C with high (90%) or low relative humidity (25%). The mean fall in FEV1 was 36.8% in the dry air and this was reduced to <10% in the humid condition. They were the first to highlight ‘the importance of monitoring and standardizing the climatic conditions in the laboratory’ (35). Later in 1977, Chen and Horton (36) used inspired air at body conditions (37°C and 100% RH) to inhibit EIA. They found full protection from EIA in four asthmatic subjects who had a >20% fall in FEV1 after walking exercise inhaling dry air 23°C 15% RH. They concluded that EIA ‘must relate to the loss of heat/or water from the respiratory tract during exercise’ (36). The inhibitory effect of inspiring air at body conditions was confirmed by others studying cycling (37) and running exercise and including subjects with severe EIA (Fig. 5) (38).
Fig. 5.
Mean±SEM values for FEV1 expressed as percentage of predicted before and after 5.5–8 min of treadmill running in 15 patients with severe EIA. Exercise was performed on two occasions 20 min apart with warm humid air as the first challenge (% fall 6.5±5.2 SD) and room air as the second (% fall 53.9±11.5 SD) (38). Figure reproduced with permission from (49).
Heat loss and water loss: thermal load or osmotic load
Attention then turned to the effects of inspired temperature as a determinant of the response to exercise. In 1977, Strauss et al. (39) reported enhancement of EIA when cold air at −8 to −15°C was inspired during 3–4 min of ‘exhausting leg work on a cycle ergometer’. In 1978, using the same exercise protocol, they reported the effects of inspiring air at ambient and body temperature when fully saturated and when relatively dry. They showed that water content of the inspired air was the major determinant of severity of EIA (37). Including the data from these two studies, the same group proposed that the ‘magnitude of EIA is directly proportional to the thermal load’ and can be measured in terms of respiratory heat exchange (40). Furthermore, they suggested that ‘the major stimulus for EIA is heat loss with subsequent airway cooling’ (41).
By 1982, Anderson et al. had also studied the effect of varying heat and water content of the inspired air (42). In contrast to Deal et al. (40), they found that asthmatics varied by up to a factor 3 in their sensitivity to the thermal load even when correcting for lung size. They confirmed the greatest severity of EIA when subjects inspired dry air and the least with humid air. An unexpected finding was that 12 subjects still had EIA after inhaling air of body conditions (37°C 44 mg H2O/L) during and after exercise. Knowing that excess water could provoke bronchoconstriction (43), they concluded that ‘the bronchoconstricting effect from water gain and water loss from the airways may be a change in tonicity of the fluid lining the respiratory tract’ (42). Similar findings were reported in 1985, from a study using voluntary hyperpnoea (44).
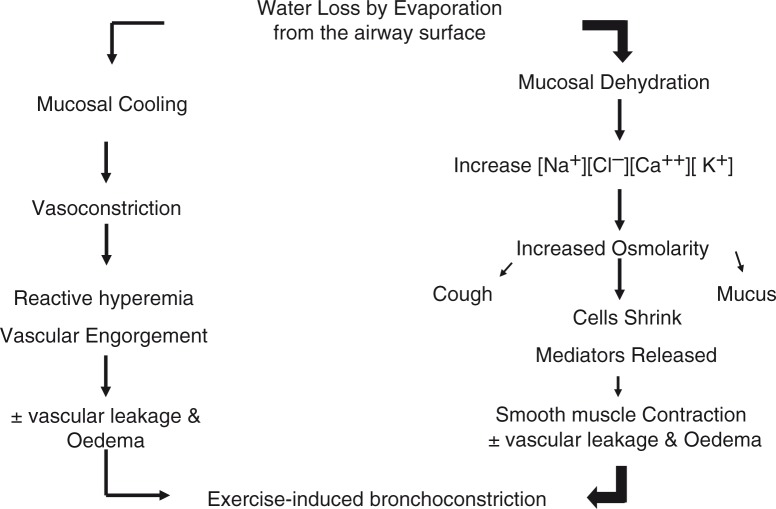
The 1982 study of Anderson et al. (42) had not included an exercise test with a low inspired air temperature because no enhancement of EIA had been found when cool air was inspired in a pilot study. A casual comment to a colleague, about this unexpected finding, led to the publication of data from a climate chamber study using air of 9°C and 36°C but the same inspired water content (9–10 mg H2O/L) (45). The severity of EIA was the same under both conditions where water loss, but not heat loss, was the same. It was argued that ‘the osmotic and not the cooling effects induced by vaporization of water was the important factor determining EIA’ (45). This proposal was presented in detail as a unifying hypothesis for EIA (46). The conclusion was that ‘there may be an important association between osmotic changes in the epithelium and the release of mediators from bronchial mucosal mast cells’ (Fig. 6) (46).
Fig. 6.
Evaporative water loss from the airways can lead to bronchoconstriction via airway cooling and rewarming and/or by airway dehydration and an increase in osmolarity of the airways. Dehydration is important for all temperatures of dry air, whereas cooling and rewarming will be additionally important when exercise is performed in subfreezing air conditions but become less so as warmer air is inspired. Reproduced with permission from (66).
To investigate further the airway cooling hypothesis, a study of the effects of hot dry air (32–40°C) was performed in children cycling for 8 min (47). Moderate-to-severe EIA (mean% fall FEV1 39.8%±22.3) occurred in 20 of the 22 subjects and the expired air temperature during exercise was higher than at rest (35.3°C versus 33.2°C) (47). This finding demonstrated that abnormal airway cooling was not occurring and thus not essential for EIA to occur. They proposed ‘water loss and not heat loss was the stimulus to EIA under these inspired air conditions’ (47). A similar conclusion in respect to respiratory heat loss was made by others using voluntary hyperpnoea (48).
The water per litre of expired air was found to be relatively constant between 31 and 33 mg/L when air over a wide range of temperature was inspired at 22–40°C (49). Providing the ventilation was high enough for long enough, then the rate and amount of water loss would be sufficient to provide an osmotic load. It was not feasible, however, to obtain a representative sample of surface liquid from the lower airways during exercise. That evaporative water loss could increase airway osmolarity was confirmed in the human nose (50). Mathematical models estimated that exercising under temperate conditions, 40% of the water lost would come from the lower airways (51).
Air condition during recovery and thermal gradients
In 1986, McFadden et al. (52) reported that the condition of the air inspired during recovery from exercise determined the magnitude of airway response in adults. They found that exercising for 4 min breathing cold air followed by 5 min breathing air of body conditions during recovery resulted in a greater fall in FEV1 in both asthmatic and normal subjects (52). They concluded that ‘in order to induce obstruction a thermal gradient seems to be necessary at the end of challenge so that the cooling brought about by hyperpnea is followed by rewarming when hyperpnea ceases’ (52). While airway cooling followed by rapid rewarming could amplify the airway response to water loss particularly in cold weather athletes (53), it could occur independently of the osmotic effects (Fig. 6) (54). A thermal gradient did not appear essential (55) because EIA occurred when breathing hot (≥36°C) dry air both during and after exercise when a thermal gradient would be minimal if not absent (37, 40, 45, 47, 56). Further flow rates often fall during exercise of 6–8 min duration and before any rewarming could occur (56). Finally, the observations (52) were not reproduced in children who exercised for 5.5–10 min (57). The laboratory protocols did not include air conditions during recovery from exercise, but they did recommend 6–8 min of vigorous exercise, rather than 4 min. The longer duration was known to enhance the magnitude of the airway response under both temperate and hot air dry air conditions (21, 56).
Defining the protocol for clinical practice
Once all the important factors determining the airway response to exercise were identified from research studies, protocols were established for use in clinical practice (58–60). In essence, these stated that to identify EIA it required the duration of exercise be 6–8 min and the exercise load, either cycling or running, be of sufficient intensity to raise ventilation to 40–60% of maximum voluntary ventilation (MVV), where MVV equals 35 times FEV1. This intensity was to be maintained for 4 min and the inspired air needed to contain <10 mg H2O/L. The nose was to be clipped and a suitable time period to have elapsed since last medication or vigorous exercise. The heart rate was required to be measured continuously and, in those over 40 years, an electrocardiogram was taken throughout exercise and for 5 min after its completion.
Over the years, heart rate became a surrogate for ventilation, as a measure of intensity of exercise. This outcome was unfortunate in that heart rate during exercise does not reflect ventilation or rate of respiratory water loss (61). The major determinants of EIA remain the level of ventilation reached and sustained during exercise and the water content of the inspired air.
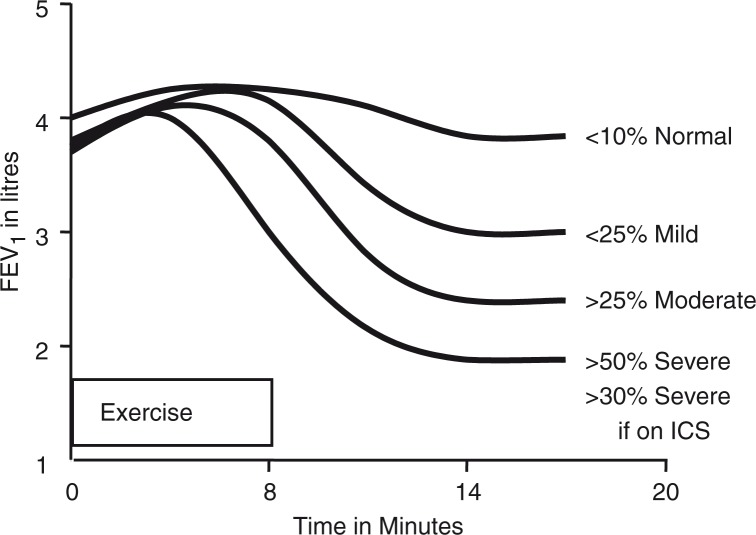
There has been some ‘fine tuning’ from subsequent studies. For example, a protocol for exercise in the field taking climatic conditions into account suggested a 15% fall in FEV1 to identify EIA in children (62, 63). A cut-off of a 13% fall in FEV1 was suggested by Godfrey after analysing published data in a ~1,000 normal children (64). Combining two indices of spirometry improved sensitivity to identify EIA (65). Levels of severity for EIA were suggested (66) (Fig. 7) and later adopted (67). The greater sensitivity of exercising, in the field, rather than in the laboratory was emphasised (68). The importance of ensuring that exercise load is appropriate in children and heart rate is preferably 95% during the last 4 min of exercise has been highlighted (69). The FEV0.5 was introduced to assess EIA in young children (70). The reproducibility of the response to a running exercise protocol demonstrated the need for two negative tests before EIA could be confidently excluded (71). A novel test to identify EIA in very young children was introduced involving exercising on a jumping castle in a cool environment (72).
Fig. 7.
Pattern of change in FEV1 after 8 min of vigorous exercise inspiring dry air at a ventilation exceeding 50% of maximum, in a normal healthy subject without exercise-induced bronchoconstriction (EIB) and in subjects with mild, moderate, or severe EIB. The severity of the response is based on the maximum fall in FEV1 in the 20 min after exercise expressed as a percentage of the baseline value. If a subject is taking inhaled corticosteroids on a daily basis, a post exercise fall in FEV1 of 30% or more would be considered severe. Reproduced with permission from (66).
Recommendations and protocols to identify EIA in clinical practice have been published by both the European (73) and American Academies of Allergy and Clinical Immunology (74) and the American Thoracic Society (67).
Eucapnic voluntary hyperpnoea of dry air
In the clinical laboratory, it became difficult to ensure that all subjects, under investigation for EIA, could actually exercise on a bicycle or a treadmill to a sufficient intensity and sustain a level of ventilation for long enough to provoke EIA and there was a high frequency of negative tests. These problems plus the safety measures required by guidelines to study adult subjects likely contributed to the disenchantment with exercise testing and enhanced the development of a surrogate test for identifying EIA.
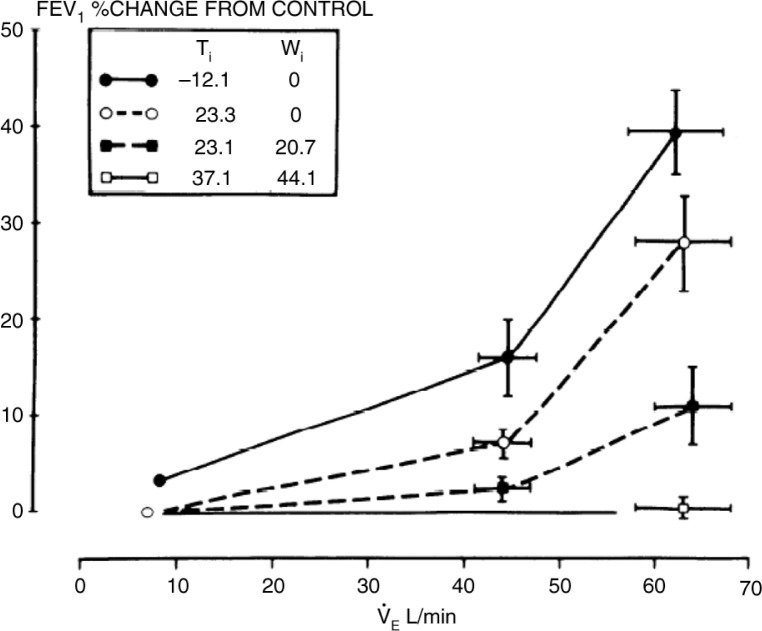
Studies in both children and adults (75–77) indicated that a major determinant of the severity of EIA was not the exercise itself but the rate of ventilation achieved and sustained during exercise. In 1979, Deal et al. (41) had reported equivalent bronchoconstriction with both exercise and hyperpnoea of dry air over a wide range of air conditions. These findings clearly demonstrated that humoral substances released during exercise were not relevant, and that ventilation and inspired air condition (Fig. 8) could explain much of the difference in severity of EIA between different forms of exercise (41). Thereafter, hyperpnoea with dry air presented an attractive option to identify EIA in that it reduced the cost of equipment substantially and obviated the need for trained personnel to supervise an exercise test.
Fig. 8.
Changes that developed in L/sec in FEV1 at various levels of ventilation (VE) and various inspired air conditions for temperature and water content (see inset). Values at extreme left of the graph represent effects of inhaling each gas mixture on lung function at rest as determined from previous studies in the same subjects. Reproduced with permission from (41).
In the early 1980s, a number of protocols were developed using voluntary hyperpnoea of dry air with added carbon dioxide (CO2) as a surrogate for exercise-induced hyperpnoea (78, 79). The CO2 was introduced in sufficient quantities to keep end-tidal CO2 (PETCO2) levels within the normal range. There were two major differences between the types of protocol developed. One difference was the temperature of the inspired air, either air at room temperature or air conditioned to subfreezing temperatures. The other difference was choice of either a single level of ventilation or multiple levels that progressively increased.
A cold air (−15°C) challenge with ventilation (VE) increasing every 3 min (7.5, 15, 30, and 60 L/min) was developed by O'Byrne et al. (80). They reported the value for respiratory heat exchange (RHE) that provoked a 20% fall in FEV1. This protocol was also used to study the inhibitory effects of DSCG (81). In contrast to O'Byrne et al., Aquilina (82) chose a single level of VE of 24×FEV1 L for 3 min and used both room temperature air and air at −10°C with the cold air causing a fall in FEV1 in all subjects that was >9% of the resting value.
The RHE index was impractical and hard to interpret for most investigators and after using the same protocol, the more simple index of ventilation to provoke a 20% fall in FEV1 was suggested by Weiss et al. (83). Others found that repeatability of the cold air test, as with exercise, was better between days rather than within a day (84). The protocol with progressive increases in ventilation was included in the 1993 document on standardized lung function testing (58).
Another protocol included in the same document (58) was a voluntary hyperpnoea test for children standardized for different ages and sizes (85). Zach et al. used a single level of ventilation equivalent to 75% MVV for 4 min at −17°C and reported changes in FEV1 as the being the most reproducible measurement (85). This protocol was used for many years (86) including in a survey of 5,697 children in Germany (87). It was later modified for use in very young children (2–5 years) with specific airway resistance being measured by whole body plethysmography, and the test was recommended to identify asthma (88).
All the protocols necessitated breath-by-breath monitoring of the PETCO2 with a rapid gas analyser and for many the conditioning of inspired air to subfreezing temperatures. Although several commercial devices became available to generate cold air at −10°C, they required excessively high volumes of compressed air adding to the cost of the test. These technical requirements made these protocols for voluntary hyperpnoea unattractive for use in clinical laboratories and a more simplified system was needed.
The simplified system was provided by using a constant level of CO2 in a gas mixture delivered at room temperature (89, 90). When the inspired air contained 4.89% CO2 and the FEV1 was >1.5 L, the PETCO2 remained in the eucapnic range of ~38–42 mmHg at ventilations between 40 and 105 L/min. Using this method, the changes in FEV1 following challenge were shown to be similar to that provoked, in the same subjects, by exercise at the same ventilation. It was called eucapnic voluntary hyperventilation and was really developed as a bronchial provocation test to identify EIA in potential recruits to the defence force. Over a series of studies, it was reported that 1) a 10% fall in FEV1 5–10 min after eucapnic voluntary hyperpnoea (EVH) was an asthmatic response (91). 2) The dosing schedule should be standardized at a single level of uninterrupted ventilation (92, 93). 3) Dry air was more sensitive to identify AHR than cold air and much more sensitive than cycling exercise, and 4) a fall in FEV1 >10% of the resting value after 5–6 min of hyperpnoea at 60–80% of MVV was diagnostic of AHR (94).
A higher level of ventilation (30 times FEV1) equivalent to 85% MVV was also suggested with the defence force recruits in mind. This ventilation is higher than the 50–65% MVV achieved during exercise by non-trained subjects and much closer to that achieved by elite athletes. For this reason, an update of the original protocol described by Argyros (92) was published (95) and recommended for identifying EIA in elite athletes for the Winter Games in Salt Lake City (2, 96, 97).
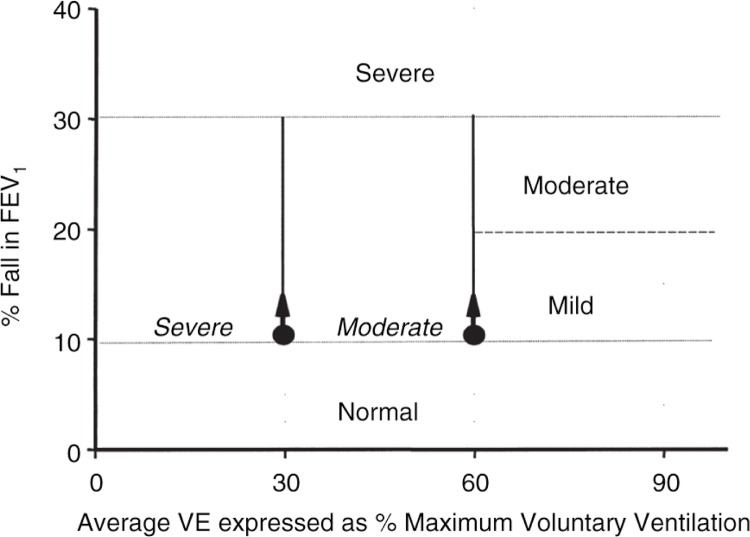
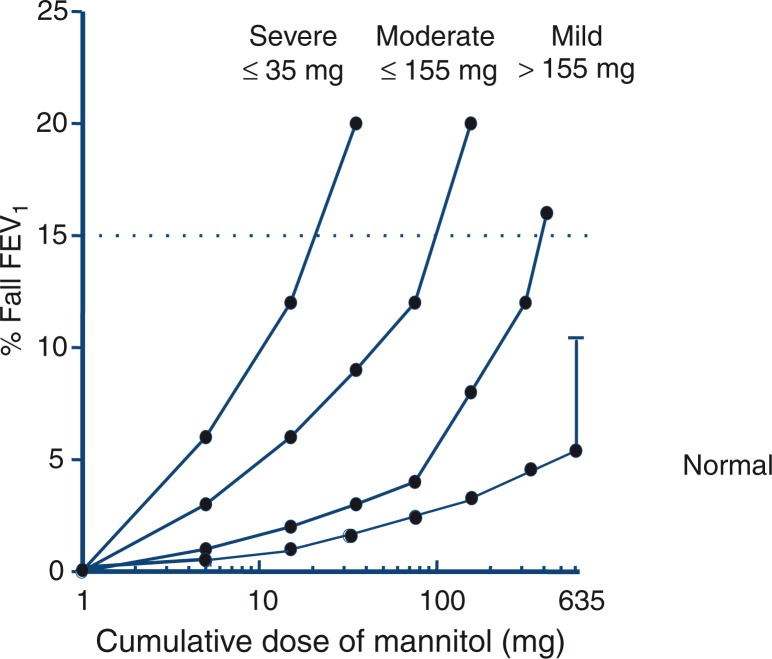
This test became known as the EVH test. It required a source of compressed gas at room temperature containing 21% O2, 4.9–5% CO2, and the remaining N2, and the subject was required to breathe this for 6 min at a single level of ventilation equivalent to 30 times the FEV1 (2, 92, 95).The severity of the response is described in Fig. 9, (2). A population of elite athletes was tested and the EVH found to be the best to identify potential for EIA (98, 99) and the test continued to be recommended for assessment of elite athletes (100).
Fig. 9.
The classification of severe, moderate, and mild is made on the level of ventilation required to induce a positive response. For example, ≥10% fall in FEV1. The response is severe if a positive response is obtained at 30% maximum voluntary ventilation (MVV), moderate if it is positive at 60% MVV, and mild if it is positive at 90% MVV. A fall in FEV1 greater than 30% whatever the ventilation would be regarded as severe. This plot can be used for a multistage or single-stage test. Maximum voluntary ventilation can be calculated on the predicted or actual FEV1 with MVV=35×FEV1. By relating the ventilation to a common predicted value, values between subjects can be compared. This also allows the intensity of exercise inducing the response to be estimated. Reproduced with permission from (2).
The required duration of EVH is shorter than exercise because the target ventilation is reached in seconds rather than minutes as occurs with exercise (61). The rate of water loss at target ventilation needs to exceed water replacement so that, over time, more airways are recruited into the conditioning process. A big advantage of EVH is that the target ventilation exceeds the maximum ventilation achievable during exercise so that false-negative tests are unlikely. In research studies of known asthmatics, however, it was recognised that the high level of ventilation and the duration of 6 min had the potential to cause large unwanted falls in FEV1. For this reason, the dosing protocol was modified to evaluate the effects of drugs (101, 102) to compare with other stimuli (103) and to assess subjects with symptoms of asthma in a clinic (104, 105). A valid clinical test for EIA is considered to be one where the ventilation reached and sustained is 60% of MVV or 21 times FEV1 (104) and is the one recommended for challenging known asthmatics (95).
In the last 10 years, a delivery system capable of monitoring and adjusting CO2 to maintain eucapnia during hyperpnoea up to 220 L/min, with a display screen, became commercially available (Eucapsys, SMTEC, Switzerland). This facilitated identification of EIA in athletes in the clinical laboratory because the ventilation was not limited to 105 L/min as was necessary when a constant level of 4.9–5% CO2 concentration was inspired. Other devices have also been used (Ailos Medicinsk Teknik, Karlstad, Sweden) and another recently developed in the USA (Rosenthal Hyperventilometer).
Aerosols of hypertonic saline
The idea to perform a challenge with an aerosol of hypertonic saline arose as a result of the report of the bronchoconstricting effects of inhaling an aerosol of water by Allegra and Bianco (106). They had used an ‘ultrasonic mist of distilled water’ delivering 2 ml/min via a face mask and reported a significant increase in specific airways resistance in asthmatics, but not normal subjects. No response occurred following the inhalation of normal saline. They reported the inhibitory effect of DSCG and proposed that mast cell release of histamine was consistent with the airway response (107–109).
Using the same nebulizer (MistO2gen EN 143 Timeter PA), Schoeffel et al. (43) administered increasing doses of both hypotonic and hypertonic saline aerosols and measured the airway response using FEV1 rather than airways resistance. The aerosol was inhaled via a Hans Rudolph 2700 valve, and the expired ventilation was measured using a Drager volume meter. Ten asthmatic subjects with EIA were studied. Initially, 5 or 10 L of the aerosol was inspired through the nebulizer and the FEV1 measured 30 sec later. When the fall in FEV1 was <10% of baseline, the volumes used in subsequent exposure were 20, 40, 80, 80, and 80 L until a 20% fall in FEV1 had occurred or 310 L had been inhaled. The airway response was also expressed as the volume to provoke a 20% fall in FEV1 from baseline (PV20). Schoeffel et al. (43) confirmed the earlier findings with distilled water and isotonic saline and were the first to report the bronchoconstricting effects of hypertonic aerosols of saline (2.7% and 3.6%) in subjects with asthma. They stated that the effect was likely due to osmolarity as inhaling an aerosol of 20% dextrose provoked similar changes in FEV1 (43). Citing that both hypo and hypertonicity resulted in release of histamine from mast cells (109, 110), they proposed that water movement in and out of the mast cell was the stimulus for mediator release. They concluded that ‘the method used for the challenge was rapid, simple and inexpensive and provides a new technique for the diagnosis of non-immunologically mediated bronchial hyperreactivity’ (43).
In the early studies, the challenge with hypertonic saline started with a 60-sec exposure and the test continued until a 20% fall in FEV1 or 30 ml had been delivered. Many asthmatic subjects were very sensitive to these aerosols and the initial exposure time was reduced to 30 sec and the maximum dose to 15.5 ml. The dose of aerosol delivered by the ultrasonic nebulizer was found to be constant, independent of air flow and directly related to expired volume so that time could also be used for a dosing schedule. Exposure times were 30 sec 1, 2, 4, and 8 min with FEV1 being measured in duplicate 60 sec after each exposure. The use of time made the method practical for use in clinical practice. The nebulizer unit with accompanying tubing, but not the valve, was weighed before and after challenge to calculate the total dose of aerosol delivered, and a dose–response curve was constructed. In 1983, the provoking dose of water or hypertonic saline to induce a 20% fall in FEV1 (PD20) replaced the (PV20). As the majority (80%) of asthmatic subjects responded in <9 min, this made the protocol feasible as a routine provocation test even though a minority of subjects required 20–25 min to respond. Consistent with exercise and hyperpnoea with cold air, the responses to both water and 3.6% saline responses were inhibited by DSCG (111). The method was published in detail in 1984 and 1985 (112–114).
Both hypo and hypertonic aerosol challenge tests were included in the Sterk document in 1993 (58). The distilled water test was used extensively for research, particularly for assessment of drugs (115–117). There were a number of findings however that probably contributed to it never becoming established in clinical practice. These included the finding it caused excessive cough and increased non-specific AHR (118–120). Further, it was found that the presence of permeant anions reduced cough making other aerosols more attractive (121).
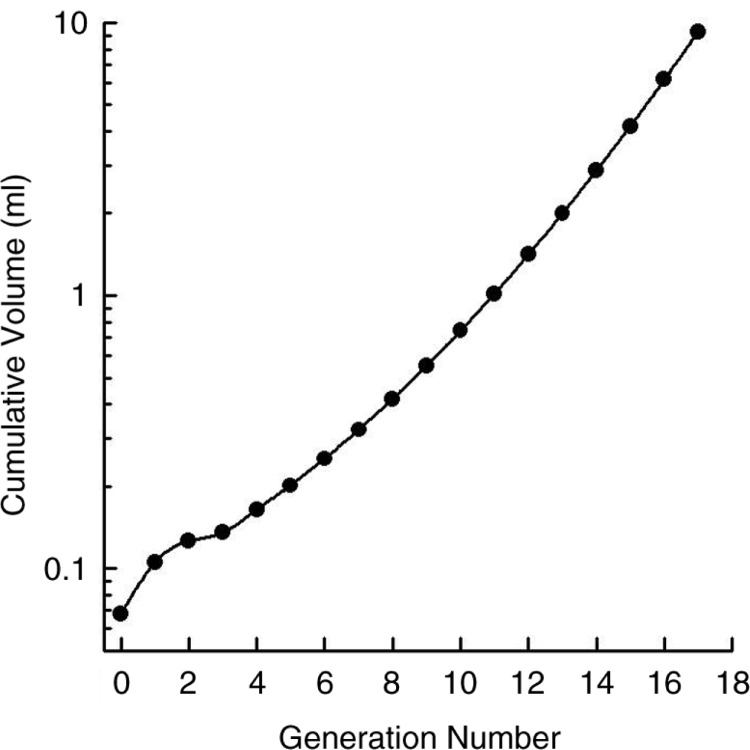
The development of the challenge with hypertonic saline aerosol continued because of its potential to mimic the osmotic effects of evaporative loss of water from the airway surface, a stimulus proposed to account for EIA (Fig. 6) (45, 46). This potential was shown to be realistic in 1983 when computer-generated calculations based on the airway dimensions of Weibel (122) revealed that the cumulative surface area of the first seven generations of airways was <400 cm2 (56) and the cumulative volume correspondingly small (Fig. 10) (123). For example, the cumulative volume of airway surface liquid for seven generations was estimated at 0.39 ml (46, 56, 124). From that calculation, it was immediately obvious that only a very small volume of water needed to be lost by evaporation during exercise, or a very small volume of hypertonic saline needed to be deposited on the surface of these airways to cause a marked increase in osmolarity of the airway surface.
Fig. 10.
The cumulative volume of airway surface liquid, in relation to the number of generations of airways assuming 10 µm for the periciliary fluid depth. Reproduced with permission from (123).
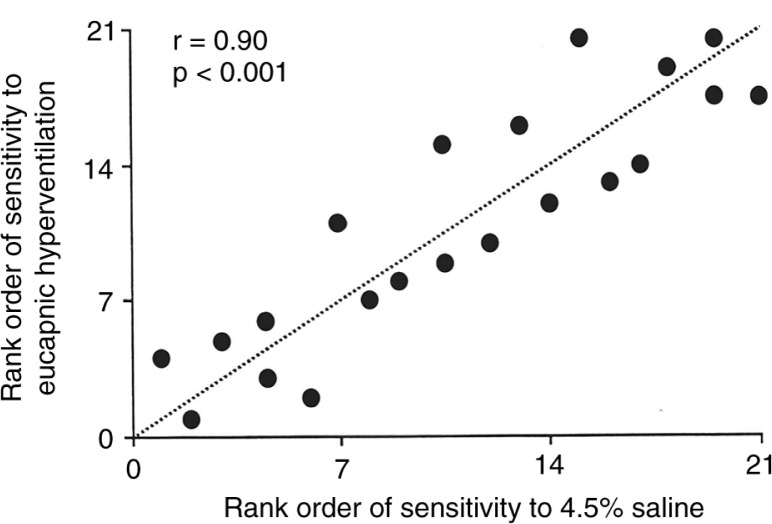
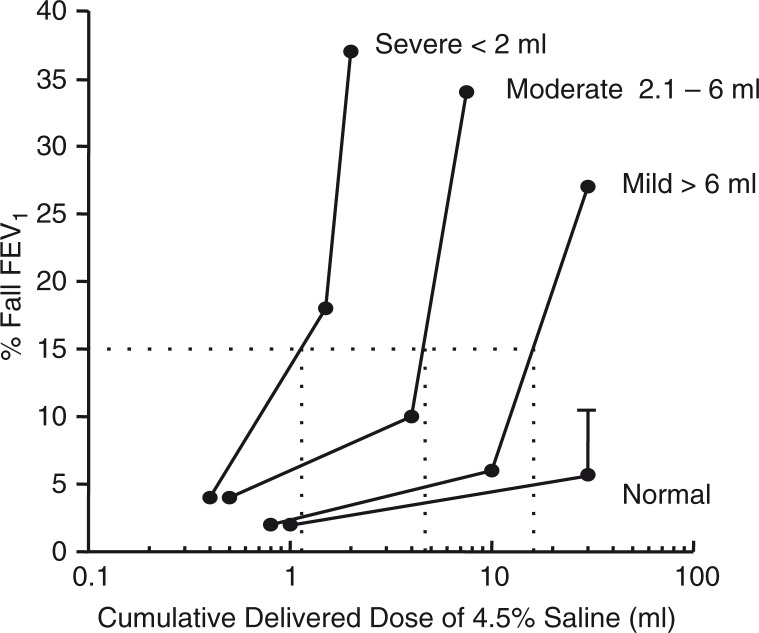
Continuing investigations, with different concentrations of saline, demonstrated it was rate of change of osmolarity that was important so concentration was increased from 3.6% (concentration of sea water) to 4.5% (125). The increase to 4.5% saline reduced the exposure time and the chance of false-negative tests and made the test practical for clinical use (126). Comparisons were made with exercise and EVH, and the sensitivity of subjects to the tests found to be concordant in most cases (Fig. 11) (124, 127–129). As inhaled corticosteroids were becoming more commonly prescribed at the time, there was interest in using a challenge test that had a high specificity to identify currently active asthma. Subsequently, the finding that sensitivity to 4.5% saline was linearly related to the % of mast cells obtained from brush biopsy of the airways (130) and it was reduced by treatment with ICS (131, 132) contributed to its adoption for clinical use (124, 133). The availability of data in healthy subjects resulted in a positive response to 4.5% saline test being reduced from 20% to 15% (Fig. 12) (2, 124). It was also an advantage that sputum could be harvested to assess cellular count at the same time (134, 135).
Fig. 11.
The Spearman's rank correlation illustrates the relationship between the sensitivity to eucapnic hyperpnoea with dry air and sensitivity to 4.5% saline in 21 subjects. Reproduced with permission from (124).
Fig. 12.
Classification of the response to hyperosmolar to 4.5% saline in terms of the provoking dose of aerosol required to induce a 15% fall in FEV1. The delivered dose is cumulative and is calculated by dividing the total dose delivered over the time of the challenge. For normal subjects, the mean plus 1 SD is shown. Reproduced with permission from (2) with data for normal subjects from (156).
Challenges with hypertonic saline aroused interest, not only from hospital-based clinicians but also from the defence forces, the underwater diving fraternity, occupational physicians, paediatricians, and epidemiologists. Suitability to dive was assessed in those with a past history of asthma (136). The hypertonic saline was used for assessing suitability for mild asthmatics to join the defence forces (137). The same protocol was used in a field survey in an occupational setting (138). In a study in children those who were positive to hypertonic saline were five times more likely to have EIA (139). The hypertonic saline became well characterised for use in children (140–142) and used along with inflammatory markers to identify asthma in children (143).
Dry powder aerosol of mannitol
By the mid-1990s, it was obvious that there were technical and hygienic limitations in generating dense aerosols from ultrasonic nebulizers in the laboratory environment. The test required filters and scales for weighing, and cleaning procedures took time and were cumbersome. Further, the particle size of the aerosol could change over the life of the piezoelectric crystal of the nebulizer. To simplify the use of hypertonic saline for identifying AHR, a dry powder was developed. Both sodium chloride (144) and mannitol were trialled (145). Mannitol was selected because it was a naturally occurring substance generally regarded as safe, commonly used as an excipient, stable at high levels of humidity and not absorbed to any significant extent by the gastrointestinal tract. Mannitol was known to stimulate the release of histamine from human lung mast cells in vitro (146). Importantly, the release of histamine was enhanced in the presence of anti-IgE and optimal at 32°C. The release of histamine occurred after an exposure to the hyperosmolar solution of only 60 sec (146, 147). Further, this release could be blocked by DSCG (148).
The mannitol powder was prepared by spray drying and was encapsulated and delivered in doses (5, 10, 20, 40, 80, 160, 160, and 160 mg) from a disposable dry powder inhaler. FEV1 was measured 60 sec after each dose and a 15% fall in FEV1 after inhaling 635 mg or less was taken as indicative of a positive response (Fig. 13) (2, 145). The sensitivity to mannitol was expressed as a PD15 and reactivity as the response–dose ratio (RDR), that is,% fall in FEV1 at the end of challenge divided by the cumulative dose of mannitol that achieved the fall. This index permitted all tests to be analysed, whether a PD15 was obtained or not, so it was useful to assess the beneficial response to inhaled corticosteroids (149).
Fig. 13.
Classification of the response to mannitol in terms of the provoking dose of powder delivered from the capsules required to induce a 15% fall in FEV1 (PD15). For normal subjects, the mean plus 1 SD is shown. Reproduced with permission from (2), with data for normal subjects from (156).
A series of clinical studies in Australia, Canada, Finland, and Switzerland established the potential for mannitol to become a simple point of care test to identify AHR consistent with currently active asthma and airway inflammation responsive to inhaled corticosteroids (145, 149–155). The intellectual property, owned by the public health service, was licensed to a pharmaceutical company. Two Phase 3 trials (156, 157) were carried out in two populations, one with an established diagnosis of asthma and the other in subjects with symptoms of asthma but no definite diagnosis. The Phase 3 trials for registration of mannitol required comparison with other tests, one being with the wet aerosol of 4.5% saline, (156) another running exercise and another methacholine chloride (157). The time to document a positive response to mannitol was 17.3 min and a negative response 26 min (156). The time for challenge and the high specificity (95%) to identify asthma made the mannitol test attractive to clinicians particularly in relation to decision to treat with ICS (156, 157). The false-negative tests contributing to a sensitivity of 60% occurred in those currently being treated with ICS or in subjects with mild asthma not requiring ICS (156, 158). When the use of steroids was taken into account, the sensitivity to identify asthma using the mannitol test rose to 89% (156).
The mannitol test received regulatory approval in Australia in 2006, the European countries between 2006 and 2008, and Korea and USA in 2010. The mannitol test kit of capsules and inhaler (known as Aridol™ or Osmohale™ Pharmaxis Ltd, Frenchs Forest, NSW, Australia) provides a common operating procedure for bronchial provocation across the globe and requires only the minimum of equipment including a spirometer and clock. The test was taken up into clinical practice and included in guidelines (67, 74, 159).
Since registration there have been many studies assessing mannitol responsiveness as both a diagnostic test to identify EIA (160–162) and a clinical diagnosis of asthma (163, 164). It has been compared with inflammatory biomarkers of asthma (165, 166). Several large studies using inhaled steroids have demonstrated usefulness for both down titrating (167) and up titrating dose based on AHR to mannitol (168).
Characteristics of all indirect challenges
The characteristics that make challenges with stimuli, that act indirectly, different, and potentially more useful in clinical practice than stimuli that act directly (e.g. methacholine) are summarised as follows. All the responses to indirect challenge tests 1) have a high specificity to identify people with current asthma (156, 157, 163, 164), 2) are inhibited by chronic treatment with inhaled corticosteroids (132, 149, 155, 169–177), 3) are inhibited by acute treatment with a leukotriene receptor antagonist or a 5-lipoxygenase inhibitor (154, 178–183), 4) are associated with the release of the specific mast cell mediator PGD2 and release of leukotriene E4 (184–191), 5) are inhibited by acute treatment with DSCG (81, 132, 188, 190, 192–195), and 6) are subject to a refractory period that is usually <3h (93, 196–201).
Conclusion
The scientific research that identified the stimulus to EIA, as evaporative loss of water from the airway surface, was fundamental to the development and standardization of these indirect challenge tests. The safety of these tests was established by their successful use in large numbers of subjects in Phase 3 trials, clinical trials, and in field studies. All these challenge tests have a high specificity to identify currently active asthma. All can be used to identify the need for treatment and for the assessment of response to inhaled corticosteroids and other anti-inflammatory drugs. Over the 50 years, the methods for testing became less complex, requiring less expensive equipment, using an easy measure to express sensitivity and reactivity. Thus, they became practical to use routinely and were recommended in guidelines for use in clinical practice.
Conflict of interest and funding
The author has not received any funding or benefits from industry or elsewhere to write this historical review. The author is the inventor of the mannitol test and receives 10% of the royalties paid to Sydney Local Area Health District by Pharmaxis Ltd.
References
- 1.Joos GF, O'Connor B, Anderson SD, Chung F, Cockcroft DW, Dahlen B, et al. Indirect airway challenges. Eur Respir J. 2003;21:1050–68. doi: 10.1183/09031936.03.00008403. [DOI] [PubMed] [Google Scholar]
- 2.Anderson SD, Brannan JD. Methods for ‘indirect’ challenge tests including exercise, eucapnic voluntary hyperpnea and hypertonic aerosols. Clin Rev Allergy Immunol. 2003;24:63–90. doi: 10.1385/CRIAI:24:1:27. [DOI] [PubMed] [Google Scholar]
- 3.Jones RS, Buston MH, Wharton MW. The effect of exercise on ventilatory function in the child with asthma. Brit J Dis Chest. 1962;56:78–86. doi: 10.1016/s0007-0971(62)80005-9. [DOI] [PubMed] [Google Scholar]
- 4.Jones RS, Wharton MJ, Buston MH. The place of physical exercise and bronchodilator drugs in the assessment of the asthmatic child. Arch Dis Childh. 1963;38:539–45. doi: 10.1136/adc.38.202.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McNeill RS, Nairn JR, Millar JS, Ingram CG. Exercise-induced asthma. Q J Med. 1966;35:55–67. [PubMed] [Google Scholar]
- 6.Sly RM, Heimlich EM, Busser RJ, Strick L. Exercise-induced bronchospasm: effect of adrenergic or cholinergic blockade. J Allergy. 1967;40:93–9. doi: 10.1016/0021-8707(67)90102-5. [DOI] [PubMed] [Google Scholar]
- 7.Fisher HK, Holton P, Buxton RSJ, Nadel JA. Mechanism of exercise-induced bronchoconstriction. Am Rev Resp Dis. 1970;101:885–96. doi: 10.1164/arrd.1970.101.6.885. [DOI] [PubMed] [Google Scholar]
- 8.Cox JS. Disodium cromoglycate (FPL 670) (‘Intal’): a specific inhibitor of reaginic antibody-antigen mechanisms. Nature. 1967;216:1328–9. doi: 10.1038/2161328a0. [DOI] [PubMed] [Google Scholar]
- 9.Altounyan REC. Inhibition of experimental asthma by a new compound, disodium cromoglycate. Acta Allerg. 1967;22:487. [Google Scholar]
- 10.Pepys J, Hargreave FE, Chan M, McCarthy DS. Inhibitory effects of disodium cromoglycate on allergen-inhalation tests. Lancet. 1968;2:134–8. doi: 10.1016/s0140-6736(68)90419-4. [DOI] [PubMed] [Google Scholar]
- 11.Davies SE. Effect of disodium cromoglycate on exercise-induced asthma. Br Med J. 1968;3:593–4. doi: 10.1136/bmj.3.5618.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Poppius H, Muttari A, Kreus KE, Karronen O, Viljanen A. Exercise asthma and disodium cromoglycate. Brit Med J. 1970;4:337–9. doi: 10.1136/bmj.4.5731.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pierson WE, Bierman CW, Stamm SJ. Cycloergometer-induced bronchospasm. J Allergy. 1969;43:136–44. doi: 10.1016/s0021-8707(69)80004-3. [DOI] [PubMed] [Google Scholar]
- 14.Godfrey S, Silverman M, Anderson SD. Problems of interpreting exercise-induced asthma. J Allergy Clin Immunol. 1973;52:199–209. doi: 10.1016/0091-6749(73)90058-4. [DOI] [PubMed] [Google Scholar]
- 15.Cropp GJ. Grading, time course, and incidence of exercise-induced airway obstruction and hyperinflation in asthmatic children. Pediatrics. 1975;56(5 pt-2 suppl):868–79. [PubMed] [Google Scholar]
- 16.Cropp GJA. Relative sensitivity of different pulmonary function tests in the evaluation of exercise-induced asthma. Pediatrics. 1975;56(5 Pt 2):860S–7S. [PubMed] [Google Scholar]
- 17.Eggleston PA, Bierman CW, Pierson WE, Stamm SJ, Van Arsdel PP., Jr A double blind trial of the effect of cromolyn sodium on exercise-induced bronchospasm. J Allergy Clin Immunol. 1972;50:57–63. doi: 10.1016/0091-6749(72)90079-6. [DOI] [PubMed] [Google Scholar]
- 18.Fitch KD, Morton AR. Specificity of exercise in exercise-induced asthma. Br Med J. 1971;4:577–81. doi: 10.1136/bmj.4.5787.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Anderson SD, Connolly N, Godfrey S. Comparison of bronchoconstriction induced by cycling and running. Thorax. 1971;26:396–401. doi: 10.1136/thx.26.4.396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wright BM, McKerrow CB. Maximum forced expiratory flow rate as a measure of ventilatory capacity: with a description of a new portable instrument for measuring it. Br Med J. 1959;2:1041–6. doi: 10.1136/bmj.2.5159.1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Silverman M, Anderson SD. Standardization of exercise tests in asthmatic children. Arch Dis Childh. 1972;47:882–9. doi: 10.1136/adc.47.256.882. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Silverman M, Andrea T. Time course of effect of disodium cromoglycate on exercise-induced asthma. Arch Dis Childh. 1972;47:419–22. doi: 10.1136/adc.47.253.419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Silverman M, Turner-Warwick M. Exercise-induced asthma: response to disodium cromoglycate in skin-test positive and skin-test negative subjects. Clin Allergy. 1972;2:137–42. doi: 10.1111/j.1365-2222.1972.tb01278.x. [DOI] [PubMed] [Google Scholar]
- 24.Bierman CW, Kawabori I, Pierson WE. Incidence of exercise-induced asthma in children. Pediatrics. 1975;56(5 Part 2):847–50. [PubMed] [Google Scholar]
- 25.Cropp GJ. The exercise bronchoprovocation test: standardization of procedures and evaluation of response. J Allergy Clin Immunol. 1979;64(6 pt 2):627–33. doi: 10.1016/0091-6749(79)90026-5. [DOI] [PubMed] [Google Scholar]
- 26.Godfrey S, Silverman M, Anderson SD. The use of the treadmill for assessing exercise-induced asthma and the effect of varying the severity and the duration of exercise. Paediatrics. 1975;56(5 Pt 2):893S–8S. [PubMed] [Google Scholar]
- 27.Eggleston PA. The cycloergometer as a system for studying exercise-induced asthma. Pediatrics. 1975;56(5 Part 2):899–903. [PubMed] [Google Scholar]
- 28.Pierson WE, Bierman CW. Free running test for exercise -induced bronchoconstriction. Pediatrics. 1975;56(5 Part 2):890–2. [PubMed] [Google Scholar]
- 29.Kattan M, Thomas CM, Keens TG, Mellis CM, Levison H. The response to exercise in normal and asthmatic children. J Pediatr. 1978;92:718–21. doi: 10.1016/s0022-3476(78)80135-8. [DOI] [PubMed] [Google Scholar]
- 30.Anderson SD, Silverman M, Konig P, Godfrey S. Exercise-induced asthma. A Review. Br J Dis Chest. 1975;69:1–39. doi: 10.1016/0007-0971(75)90053-4. [DOI] [PubMed] [Google Scholar]
- 31.Eggleston PA, Rosenthal RR, Anderson SD, Anderton R, Bierman CW, Bleecker ER, et al. Guidelines for the methodology of exercise challenge testing of asthmatics. J Allergy Clin Immunol. 1979;64:642–5. doi: 10.1016/0091-6749(79)90028-9. [DOI] [PubMed] [Google Scholar]
- 32.Eggleston PA. Laboratory evaluation of exercise-induced asthma: methodologic considerations. J Allergy Clin Immunol. 1979;64:604S–8S. doi: 10.1016/0091-6749(79)90022-8. [DOI] [PubMed] [Google Scholar]
- 33.Fitch KD, Morton AR, Blanksby BA. Effects of swimming training on children with asthma. Arch Dis Child. 1976;51:190–4. doi: 10.1136/adc.51.3.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Weinstein RE, Anderson JA, Kvale P, Sweet LC. Effects of humidification on exercise induced asthma (EIA) J Allergy Clin Immunol. 1976;57:A250–1. [Google Scholar]
- 35.Bar-Or O, Neuman I, Dotan R. Effects of dry and humid climates on exercise-induced asthma in children and preadolescents. J Allergy Clin Immunol. 1977;60:163–8. doi: 10.1016/0091-6749(77)90119-1. [DOI] [PubMed] [Google Scholar]
- 36.Chen WY, Horton DJ. Heat and water loss from the airways and exercise-induced asthma. Respiration. 1977;34:305–13. doi: 10.1159/000193842. [DOI] [PubMed] [Google Scholar]
- 37.Strauss RH, McFadden ER, Ingram RH, Deal EC, Jaeger JJ, Stearns D. Influence of heat and humidity on the airway obstruction induced by exercise in asthma. J Clin Invest. 1978;61:433–40. doi: 10.1172/JCI108954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Anderson SD, Daviskas E, Schoeffel RE, Unger SF. Prevention of severe exercise-induced asthma with hot humid air. Lancet. 1979;2:629. doi: 10.1016/s0140-6736(79)91681-7. [DOI] [PubMed] [Google Scholar]
- 39.Strauss RH, McFadden ER, Jr., Ingram RH, Jr., Jaeger JJ. Enhancement of exercise-induced asthma by cold air. N Engl J Med. 1977;297:743–7. doi: 10.1056/NEJM197710062971402. [DOI] [PubMed] [Google Scholar]
- 40.Deal EC, McFadden ER, Ingram RH, Strauss RH, Jaeger JJ. Role of respiratory heat exchange in production of exercise-induced asthma. J Appl Physiol Respir Environ Exerc Physiol. 1979;46:467–75. doi: 10.1152/jappl.1979.46.3.467. [DOI] [PubMed] [Google Scholar]
- 41.Deal EC, McFadden ER, Ingram RH, Jaeger JJ. Hyperpnea and heat flux: initial reaction sequence in exercise-induced asthma. J Appl Physiol Respir Environ Exerc Physiol. 1979;46:476–83. doi: 10.1152/jappl.1979.46.3.476. [DOI] [PubMed] [Google Scholar]
- 42.Anderson SD, Schoeffel RE, Follet R, Perry CP, Daviskas E, Kendall M. Sensitivity to heat and water loss at rest and during exercise in asthmatic patients. Eur J Respir Dis. 1982;63:459–71. [PubMed] [Google Scholar]
- 43.Schoeffel RE, Anderson SD, Altounyan RE. Bronchial hyperreactivity in response to inhalation of ultrasonically nebulised solutions of distilled water and saline. Br Med J. 1981;283:1285–7. doi: 10.1136/bmj.283.6302.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aitken ML, Marini JJ. Effect of heat delivery and extraction on airway conductance in normal and in asthmatic subjects. Am Rev Respir Dis. 1985;131:357–61. doi: 10.1164/arrd.1985.131.3.357. [DOI] [PubMed] [Google Scholar]
- 45.Hahn A, Anderson SD, Morton AR, Black JL, Fitch KD. A re-interpretation of the effect of temperature and water content of the inspired air in exercise-induced asthma. Am Rev Respir Dis. 1984;130:575–9. doi: 10.1164/arrd.1984.130.4.575. [DOI] [PubMed] [Google Scholar]
- 46.Anderson SD. Is there a unifying hypothesis for exercise-induced asthma? J Allergy Clin Immunol. 1984;73(5 Part 2):660–5. doi: 10.1016/0091-6749(84)90301-4. [DOI] [PubMed] [Google Scholar]
- 47.Anderson SD, Schoeffel RE, Black JL, Daviskas E. Airway cooling as the stimulus to exercise-induced asthma – a re-evaluation. Eur J Respir Dis. 1985;67:20–30. [PubMed] [Google Scholar]
- 48.Eschenbacher WL, Sheppard D. Respiratory heat loss is not the sole stimulus for bronchoconstriction induced by isocapnic hyperpnea with dry air. Am Rev Respir Dis. 1985;131:894–901. doi: 10.1164/arrd.1985.131.6.894. [DOI] [PubMed] [Google Scholar]
- 49.Anderson SD, Daviskas E. Pathophysiology of exercise-induced asthma: role of respiratory water loss. In: Weiler J, editor. Allergic and respiratory disease in sports medicine. New York: Marcel Dekker; 1997. pp. 87–114. [Google Scholar]
- 50.Togias AG, Proud D, Lichenstein LM, Adams GK, Norman PS, Kagey-Sobotka A, et al. The osmolality of nasal secretions increases when inflammatory mediators are released in response to inhalation of cold, dry air. Am Rev Respir Dis. 1988;137:625–9. doi: 10.1164/ajrccm/137.3.625. [DOI] [PubMed] [Google Scholar]
- 51.Daviskas E, Gonda I, Anderson SD. Local airway heat and water vapour losses. Respiration Physiology. 1991;84:115–32. doi: 10.1016/0034-5687(91)90023-c. [DOI] [PubMed] [Google Scholar]
- 52.McFadden ER, Lenner KA, Strohl KP. Postexertional airway rewarming and thermally induced asthma. J Clin Invest. 1986;78:18–25. doi: 10.1172/JCI112549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Anderson SD, Holzer K. Exercise-induced asthma: is it the right diagnosis in elite athletes? J Allergy Clin Immunol. 2000;106:419–28. doi: 10.1067/mai.2000.108914. [DOI] [PubMed] [Google Scholar]
- 54. Anderson SD, Daviskas E. The mechanism of exercise-induced asthma is …. J Allergy Clin Immunol. 2000;106:453–9. doi: 10.1067/mai.2000.109822. [DOI] [PubMed] [Google Scholar]
- 55.Anderson SD, Daviskas E. The airway microvasculature and exercise-induced asthma. Thorax. 1992;47:748–52. doi: 10.1136/thx.47.9.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Anderson SD, Daviskas E, Smith CM. Exercise-induced asthma: a difference in opinion regarding the stimulus. Allergy Proc. 1989;10:215–26. doi: 10.2500/108854189778960054. [DOI] [PubMed] [Google Scholar]
- 57.Smith CM, Anderson SD, Walsh S, McElrea M. An investigation of the effects of heat and water exchange in the recovery period after exercise in children with asthma. Am Rev Respir Dis. 1989;140:598–605. doi: 10.1164/ajrccm/140.3.598. [DOI] [PubMed] [Google Scholar]
- 58.Sterk PJ, Fabbri LM, Quanjer PH, Cockcroft DW, O'Byrne PM, Anderson SD, et al. Airway responsiveness: standardized challenge testing with pharmacological, physical and sensitizing stimuli in adults. Eur Respir J. 1993;6(Suppl 16):53–83. doi: 10.1183/09041950.053s1693. [DOI] [PubMed] [Google Scholar]
- 59.Folgering H, Palange P, Anderson S. Clinical exercise testing with reference to lung diseases: indications and protocols. Eur Respir Mon. 1997;6:51–71. [Google Scholar]
- 60.Crapo RO, Casaburi R, Coates AL, Enright PL, Hankinson JL, Irvin CG, et al. Guidelines for methacholine and exercise challenge testing – 1999. Am J Respir Crit Care Med. 2000;161:309–29. doi: 10.1164/ajrccm.161.1.ats11-99. [DOI] [PubMed] [Google Scholar]
- 61.Anderson SD, Lambert S, Brannan JD, Wood RJ, Koskela H, Morton AR, et al. Laboratory protocol for exercise asthma to evaluate salbutamol given by two devices. Med Sci Sports Exerc. 2001;33:893–900. doi: 10.1097/00005768-200106000-00007. [DOI] [PubMed] [Google Scholar]
- 62.Haby MM, Anderson SD, Peat JK, Mellis CM, Toelle BG, Woolcock AJ. An exercise challenge protocol for epidemiological studies of asthma in children: comparison with histamine challenge. Eur Respir J. 1994;7:43–9. doi: 10.1183/09031936.94.07010043. [DOI] [PubMed] [Google Scholar]
- 63.Haby MM, Peat JK, Mellis CM, Anderson SD, Woolcock AJ. An exercise challenge for epidemiological studies of childhood asthma: validity and repeatability. Eur Respir J. 1995;8:729–36. [PubMed] [Google Scholar]
- 64.Godfrey S, Springer C, Bar-Yishay E, Avital A. Cut-off points defining normal and asthmatic bronchial reactivity to exercise and inhalation challenges in children and young adults. Eur Respir J. 1999;14:659–68. doi: 10.1034/j.1399-3003.1999.14c28.x. [DOI] [PubMed] [Google Scholar]
- 65.Custovic A, Arifhodzic N, Robinson A, Woodcock A. Exercise testing revisited. The response to exercise in normal and atopic children. Chest. 1994;105:1127–32. doi: 10.1378/chest.105.4.1127. [DOI] [PubMed] [Google Scholar]
- 66.Freed AN, Anderson SD. Exercise-induced bronchoconstriction. Human models. In: Kay AB, editor. Allergy & allergic diseases. Oxford: Blackwell Scientific Publications; 2008. pp. 808–22. [Google Scholar]
- 67.Parsons JP, Hallstrand TS, Mastronade JG, Kaminsky DA, Rundell K, Hull J, et al. An official American Thoracic Society clinical practice guideline: exercise-induced bronchoconstriction. Am J Respir Crit Care Med. 2013;187:1016–27. doi: 10.1164/rccm.201303-0437ST. [DOI] [PubMed] [Google Scholar]
- 68.Rundell KW, Wilber RL, Szmedra L, Jenkinson DM, Mayers LB, Im J. Exercise-induced asthma screening of elite athletes: field vs laboratory exercise challenge. Med Sci Sports Exerc. 2000;32:309–16. doi: 10.1097/00005768-200002000-00010. [DOI] [PubMed] [Google Scholar]
- 69.Carlsen KH, Engh G, Mørk M. Exercise induced bronchoconstriction depends on exercise load. Respir Med. 2000;94:750–5. doi: 10.1053/rmed.2000.0809. [DOI] [PubMed] [Google Scholar]
- 70.Vilozni D, Bentur L, Efrati O, Barak A, Szeinberg A, Shoseyov D, et al. Exercise challenge test in 3–6 year old asthmatic children. Chest. 2007;132:497–503. doi: 10.1378/chest.07-0052. [DOI] [PubMed] [Google Scholar]
- 71.Anderson SD, Pearlman DS, Rundell KW, Perry CP, Boushey H, Sorkness CA, et al. Reproducibility of the airway response to an exercise protocol standardized for intensity, duration, and inspired air conditions, in subjects with symptoms suggestive of asthma. Respir Res. 2010;11:120. doi: 10.1186/1465-9921-11-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Van Leeuwen JC, Driessen JMM, de Jongh FHC, Anderson SD, Thio BJ. Measuring breakthrough exercise-induced bronchoconstriction in young asthmatic children using a jumping castle. J Allergy Clin Immunol. 2013;131:1427–9. doi: 10.1016/j.jaci.2012.10.014. [DOI] [PubMed] [Google Scholar]
- 73.Carlsen KH, Anderson SD, Bjermer L, Bonini S, Brusasco V, Canonica W, et al. Exercise-induced asthma, respiratory and allergic disorders in elite athletes: epidemiology, mechanisms and diagnosis: part I of the report from the Joint Task Force of the European Respiratory Society (ERS) and the European Academy of Allergy and Clinical Immunology (EAACI) in cooperation with GA2LEN. Allergy. 2008;63:387–403. doi: 10.1111/j.1398-9995.2008.01662.x. [DOI] [PubMed] [Google Scholar]
- 74.Weiler JM, Anderson SD, Randolph C, Bonini S, Craig T, Randolph C, et al. Pathogenesis, prevalence, diagnosis, and management of exercise-induced bronchoconstriction: a practice parameter. Ann Allergy Asthma Clin Immunol. 2010;105(6 suppl):S1–S47. doi: 10.1016/j.anai.2010.09.021. [DOI] [PubMed] [Google Scholar]
- 75.Chan-Yeung MMW, Vyas MN, Grzybowski S. Exercise-induced asthma. Am Rev Respir Dis. 1971;104:915–23. doi: 10.1164/arrd.1971.104.6.915. [DOI] [PubMed] [Google Scholar]
- 76.Zeballos RJ, Shturman-Ellstein R, McNally JF, Jr., Hirsch JE, Souhrada JF. The role of hyperventilation in exercise-induced bronchoconstriction. Am Rev Respir Dis. 1978;118:877–84. doi: 10.1164/arrd.1978.118.5.877. [DOI] [PubMed] [Google Scholar]
- 77.Kilham H, Tooley M, Silverman M. Running, walking and hyperventilation causing asthma in children. Thorax. 1979;34:582–6. doi: 10.1136/thx.34.5.582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kivity S, Souhrada JF, Melzer E. A dose-response-like relationship between minute ventilation and exercise-induced bronchoconstriction in young asthmatic patients. Eur J Respir Dis. 1980;61:342–6. [PubMed] [Google Scholar]
- 79.Kivity S, Souhrada JF. Hyperpnea: the common stimulus for bronchospasm in asthma during exercise and voluntary isocapnic hyperpnea. Respiration. 1980;40:169–77. doi: 10.1159/000194275. [DOI] [PubMed] [Google Scholar]
- 80.O'Byrne PM, Ryan G, Morris M, McCormack D, Jones NL, Morse JL, et al. Asthma induced by cold air and its relation to nonspecific bronchial hyperresponsiveness to methacholine. Am Rev Respir Dis. 1982;125:281–5. doi: 10.1164/arrd.1982.125.3.281. [DOI] [PubMed] [Google Scholar]
- 81.Latimer KM, Roberts R, Morris MM, Hargreave FE. Inhibition by sodium cromoglycate of bronchoconstriction stimulated by respiratory heat loss: comparison of pressurised aerosol and powder. Thorax. 1984;39:277–81. doi: 10.1136/thx.39.4.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Aquilina AT. Comparison of airway reactivity induced by histamine, methacholine, and isocapnic hyperventilation in normal and asthmatic subjects. Thorax. 1983;38(10):766–70. doi: 10.1136/thx.38.10.766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Weiss JW, Rossing TH, McFadden ER, Ingram RH. Relationship between bronchial responsiveness to hyperventilation with cold air and methacholine in asthma. J Allergy Clin Immunol. 1983;72:140–4. doi: 10.1016/0091-6749(83)90521-3. [DOI] [PubMed] [Google Scholar]
- 84.Tessier P, Cartier A, L'Archeveque J, Ghezzo J, Martin RR, Malo J-L. Within- and between-day reproducibility of isocapnic cold air challenges in subjects with asthma. J Allergy Clin Immunol. 1986;78(3 Pt 1):379–87. doi: 10.1016/0091-6749(86)90021-7. [DOI] [PubMed] [Google Scholar]
- 85.Zach M, Polgar G, Kump H, Kroisel P. Cold air challenge of airway hyperreactivity in children: practical application and theoretical aspects. Pediatr Res. 1984;18:469–78. doi: 10.1203/00006450-198405000-00016. [DOI] [PubMed] [Google Scholar]
- 86.Varga E-M, Eber E, Zach MS. Cold air challenge for measuring airway reactivity in children: lack of a late asthmatic reaction. Lung. 1990;168:267–72. doi: 10.1007/BF02719703. [DOI] [PubMed] [Google Scholar]
- 87.Nicolai T, Mutius EV, Reitmeir P, Wjst M. Reactivity to cold-air hyperventilation in normal and in asthmatic children in a survey of 5,697 schoolchildren in southern Bavaria. Am Rev Respir Dis. 1993;147:565–72. doi: 10.1164/ajrccm/147.3.565. [DOI] [PubMed] [Google Scholar]
- 88.Nielsen KG, Bisgaard H. Lung function response to cold air challenge in asthmatic and healthy children of 2–5 years of age. Am J Respir Crit Care Med. 2000;161:1805–9. doi: 10.1164/ajrccm.161.6.9905098. [DOI] [PubMed] [Google Scholar]
- 89.Rosenthal RR. Simplified eucapnic voluntary hyperventilation. J Allergy Clin Immunol. 1984;73:676–85. doi: 10.1016/0091-6749(84)90304-x. [DOI] [PubMed] [Google Scholar]
- 90.Phillips YY, Jaeger JJ, Laube BL, Rosenthal RR. Eucapnic voluntary hyperventilation of compressed gas mixture. A simple system for bronchial challenge by respiratory heat loss. Am Rev Respir Dis. 1985;131:31–5. doi: 10.1164/arrd.1985.131.1.31. [DOI] [PubMed] [Google Scholar]
- 91. Hurwitz KM, Argyros GJ, Roach JM, Eliasson AH, Phillips YY. Interpretation of eucapnic voluntary hyperventilation in the diagnosis of asthma. Chest. 1995;108:1240–5. doi: 10.1378/chest.108.5.1240. [DOI] [PubMed] [Google Scholar]
- 92.Argyros GJ, Roach JM, Hurwitz KM, Eliasson AH, Phillips YY. Eucapnic voluntary hyperventilation as a bronchoprovocation technique. Development of a standardized dosing schedule in asthmatics. Chest. 1996;109:1520–4. doi: 10.1378/chest.109.6.1520. [DOI] [PubMed] [Google Scholar]
- 93.Argyros GJ, Roach JM, Hurwitz KM, Eliasson AH, Phillips YY. The refractory period after eucapnic voluntary hyperventilation challenge and its effect on challenge technique. Chest. 1995;108:419–24. doi: 10.1378/chest.108.2.419. [DOI] [PubMed] [Google Scholar]
- 94.Eliasson AH, Phillips YY, Rajagopal KR, Howard RS. Sensitivity and specificity of bronchial provocation testing. An evaluation of four techniques in exercise-induced bronchospasm. Chest. 1992;102:347–55. doi: 10.1378/chest.102.2.347. [DOI] [PubMed] [Google Scholar]
- 95.Anderson SD, Argyros GJ, Magnussen H, Holzer K. Provocation by eucapnic voluntary hyperpnoea to identify exercise induced bronchoconstriction. Br J Sports Med. 2001;35:344–7. doi: 10.1136/bjsm.35.5.344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Anderson SD, Fitch K, Perry CP, Sue-Chu M, Crapo R, McKenzie D, et al. Responses to bronchial challenge submitted for approval to use inhaled beta2 agonists prior to an event at the 2002 Winter Olympics. J Allergy Clin Immunol. 2003;111:44–9. doi: 10.1067/mai.2003.1. [DOI] [PubMed] [Google Scholar]
- 97.Anderson SD, Kippelen P. The best physiological test to use, when evaluating asthma and sport- from a respiratory physiologist's point of view. LungeForum. 2004;14(3):6–13. [Google Scholar]
- 98.Dickinson J. Screening elite winter athletes for exercise induced asthma: a comparison of three challenge methods. Brit J Sports Med. 2006;40:179–82. doi: 10.1136/bjsm.2005.022764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Dickinson J, McConnell A, Whyte G. Diagnosis of exercise-induced bronchoconstriction: eucapnic voluntary hyperpnoea challenges identify previously undiagnosed elite athletes with exercise-induced bronchoconstriction. Br J Sports Med. 2010;45(14):1126–31. doi: 10.1136/bjsm.2010.072520. [DOI] [PubMed] [Google Scholar]
- 100.Fitch KD, Sue-Chu M, Anderson SD, Boulet L-P, Hancox RJ, McKenzie DC, et al. Asthma and the elite athlete: summary of the International Olympic Committee's Consensus Conference, Lausanne, Switzerland, January 22–24, 2008. J Allergy Clin Immunol. 2008;122:254–60. doi: 10.1016/j.jaci.2008.07.003. [DOI] [PubMed] [Google Scholar]
- 101.Smith CM, Anderson SD, Seale JP. The duration of action of the combination of fenoterol hydrobromide and ipratropium bromide in protecting against asthma provoked by hyperpnea. Chest. 1988;94:709–17. doi: 10.1378/chest.94.4.709. [DOI] [PubMed] [Google Scholar]
- 102.Rodwell LT, Anderson SD, du Toit J, Seale JP. Different effects of inhaled amiloride and furosemide on airway responsiveness to dry air challenge in asthmatic subjects. Eur Respir J. 1993;6:855–61. [PubMed] [Google Scholar]
- 103.Brannan JD, Koskela H, Anderson SD, Chew N. Responsiveness to mannitol in asthmatic subjects. Am J Respir Crit Care Med. 1998;158:1120–6. doi: 10.1164/ajrccm.158.4.9802087. [DOI] [PubMed] [Google Scholar]
- 104.Brummel NE, Mastronarde JG, Rittinger D, Philips G, Parsons JP. The clinical utility of eucapnic voluntary hyperventilation testing for the diagnosis of exercise-induced bronchospasm. J Asthma. 2009;46:683–6. doi: 10.1080/02770900902972178. [DOI] [PubMed] [Google Scholar]
- 105.Parsons JP, Kaeding C, Phillips G, Jarjoura D, Wadley G, Mastronarde JG. Prevalence of exercise-induced bronchospasm in a cohort of varsity college athletes. Med Sci Sports Exerc. 2007;39:1487–92. doi: 10.1249/mss.0b013e3180986e45. [DOI] [PubMed] [Google Scholar]
- 106.Allegra L, Bianco S. Non-specific broncho-reactivity obtained with an ultrasonic aerosol of distilled water. Eur J Respir Dis. 1980;61(Suppl 106):41–9. [PubMed] [Google Scholar]
- 107.Salvato G. Mast cells in bronchial connective tissue of man: their modification in asthma and after treatment with histamine liberator 48/80. Int Arch Allergy. 1961;18:348–58. doi: 10.1159/000229186. [DOI] [PubMed] [Google Scholar]
- 108.Patterson R, McKenna JM, Suszko IM, Solliday NH, Pruzansky JJ, Roberts M, et al. Living histamine containing cells from the bronchial lumes of humans. J Clin Invest. 1977;59:217–25. doi: 10.1172/JCI108631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kaliner M, Austen KF. Cyclic AMP, ATP and reversed anaphylactic histamine release from rat mast cells. J Immunology. 1974;112:664–74. [PubMed] [Google Scholar]
- 110.Findlay SR, Dvorak AM, Kagey-Sobotka A, Lichtenstein LM. Hyperosmolar triggering of histamine release from human basophils. J Clin Invest. 1981;67:1604–13. doi: 10.1172/JCI110195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Anderson SD, Schoeffel RE, Finney M. Evaluation of ultrasonically nebulised solutions as a provocation in patients with asthma. Thorax. 1983;38:284–91. doi: 10.1136/thx.38.4.284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Anderson SD, Schoeffel RE. A method for documenting bronchial hyperresponsiveness using ultrasonically nebulized water. Pract Cardiol. 1984;10:69–88. [Google Scholar]
- 113.Anderson SD. Bronchial challenge by ultrasonically nebulized aerosols. Clin Rev Allergy. 1985;3:427–39. [PubMed] [Google Scholar]
- 114.Anderson SD, Schoeffel RE. The inhalation of ultrasonically nebulized aerosols as a provocation test for asthma. In: Hargreave FE, Woolcock AJ, editors. Airway responsiveness: measurement and interpretation. Mississauga, Ontario: Astra Pharmaceuticals Canada Ltd; 1985. pp. 39–50. [Google Scholar]
- 115.Mattoli S, Foresi A, Corbo GM, Valente S, Ciappi G. The effect of indomethacin on the refractory period occurring after the inhalation of ultrasonically nebulized distilled water. J Allergy Clin Immunol. 1987;79:678–83. doi: 10.1016/s0091-6749(87)80165-3. [DOI] [PubMed] [Google Scholar]
- 116.Foresi A, Pelucchi A, Mastropasqua B, Cavigioli G, Carlesi RM, Marazzini L. Effect of inhaled furosemide and torasemide on bronchial response to ultrasonically nebulized distilled water in asthmatic subjects. Am Rev Respir Dis. 1992;146:364–8. doi: 10.1164/ajrccm/146.2.364. [DOI] [PubMed] [Google Scholar]
- 117.Groot CA, Lammers JW, Molema J, Festen J, van Herwaarden CL. Effect of inhaled beclomethasone and nedocromil sodium on bronchial hyperresponsiveness to histamine and distilled water. Eur Respir J. 1992;5:1075–82. [PubMed] [Google Scholar]
- 118.Mattoli S, Foresi A, Corbo GM, Valente S, Patalano F, Ciappi G. Increase in bronchial responsiveness to methacholine and late asthmatic response after the inhalation of ultrasonically nebulized distilled water. Chest. 1986;90:726–32. doi: 10.1378/chest.90.5.726. [DOI] [PubMed] [Google Scholar]
- 119.Smith CM, Anderson SD, Black JL. Methacholine responsiveness increases after ultrasonically nebulized water, but not after ultrasonically nebulized hypertonic saline. J Allergy Clin Immunol. 1987;79:85–92. doi: 10.1016/s0091-6749(87)80021-0. [DOI] [PubMed] [Google Scholar]
- 120.Kivity S, Poterman R, Schwarz Y, Soferman R, Topilsky M. Changes in sensitivity to methacholine after inhalation with distilled water: the role of the bronchoconstrictive response. Eur Respir J. 1995;8:253–6. doi: 10.1183/09031936.95.08020253. [DOI] [PubMed] [Google Scholar]
- 121.Eschenbacher WL, Boushey HA, Sheppard D. Alteration in osmolarity of inhaled aerosols cause bronchoconstriction and cough, but absence of a permeant anion causes cough alone. Am Rev Respir Dis. 1984;129:211–5. [PubMed] [Google Scholar]
- 122.Weibel ER. Morphometry of the human lung. Berlin: Springer; 1963. [Google Scholar]
- 123.Anderson SD, Daviskas E. Airway drying and exercise induced asthma. In: McFadden ER, editor. Exercise induced asthma – lung biology in health and disease. New York: Marcel Dekker; 1999. pp. 77–113. [Google Scholar]
- 124.Anderson SD, Smith CM, Rodwell LT, du Toit JI, Riedler J, Robertson CF. The use of non-isotonic aerosols for evaluating bronchial hyperresponsiveness. In: Spector S, editor. Provocation challenge procedures. New York: Marcel Dekker; 1995. pp. 249–78. [Google Scholar]
- 125.Anderson SD, Smith CM. The use of nonisotonic aerosols for evaluating bronchial hyperresponsiveness. In: Spector S, editor. Provocative challenge procedures. New York: Futura, Mount Kisco; 1989. pp. 227–52. [Google Scholar]
- 126.Smith CM, Anderson SD. Inhalation provocation tests using non-isotonic aerosols. J Allergy Clin Immunol. 1989;84(5 Pt 1):781–90. doi: 10.1016/0091-6749(89)90309-6. [DOI] [PubMed] [Google Scholar]
- 127.Smith CM, Anderson SD. Hyperosmolarity as the stimulus to asthma induced by hyperventilation? J Allergy Clin Immunol. 1986;77:729–36. doi: 10.1016/0091-6749(86)90419-7. [DOI] [PubMed] [Google Scholar]
- 128.Smith CM, Anderson SD. A comparison between the airway response to isocapnic hyperventilation and hypertonic saline in subjects with asthma. Eur Respir J. 1989;2:36–43. [PubMed] [Google Scholar]
- 129.Smith CM, Anderson SD. Inhalational challenge using hypertonic saline in asthmatic subjects: a comparison with responses to hyperpnoea, methacholine and water. Eur Respir J. 1990;3:144–51. [PubMed] [Google Scholar]
- 130.Gibson PG, Saltos N, Borgas T. Airway mast cells and eosinophils correlate with clinical severity and airway hyperresponsiveness in corticosteroid-treated asthma. J Allergy Clin Immunol. 2000;105:752–9. doi: 10.1067/mai.2000.105319. [DOI] [PubMed] [Google Scholar]
- 131.Rodwell LT, Anderson SD, Seale JP. Inhaled steroids modify bronchial responses to hyperosmolar saline. Eur Respir J. 1992;5:953–62. [PubMed] [Google Scholar]
- 132.Anderson SD, Dutoit JI, Rodwell LT, Jenkins CR. Acute effect of sodium cromoglycate on airway narrowing induced by 4.5-percent saline aerosol – outcome before and during treatment with aerosol corticosteroids in patients with asthma. Chest. 1994;105:673–80. doi: 10.1378/chest.105.3.673. [DOI] [PubMed] [Google Scholar]
- 133.Anderson SD. Indirect challenge tests: airway hyperresponsiveness in asthma: its measurement and clinical significance. Chest. 2010;138(2 Suppl):25S–30S. doi: 10.1378/chest.10-0116. [DOI] [PubMed] [Google Scholar]
- 134.Iredale MJ, Wanklyn SAR, Phillips IP, Krauz T, Ind PW. Non-invasive assessment of bronchial inflammation in asthma: no correlation between eosinophilia of induced sputum and bronchial responsiveness to inhaled hypertonic saline. Clin Exp Allergy. 1994;24:940–5. doi: 10.1111/j.1365-2222.1994.tb02725.x. [DOI] [PubMed] [Google Scholar]
- 135.Anderson SD, Gibson P. The use of aerosols of hypertonic saline and distilled water (fog) In: Barnes PJ, Grunstein MM, Leff A, Woolcock AJ, editors. Asthma. New York: Raven Press; 1997. pp. 1135–50. [Google Scholar]
- 136.Anderson SD, Brannan J, Trevillion L, Young IH. Lung function and bronchial provocation tests for intending divers with a history of asthma. SPUMS J. 1995;25:233–48. [Google Scholar]
- 137.Freed R, Anderson SD, Wyndham J. The use of bronchial provocation tests for identifying asthma. A review of the problems for occupational assessment and a proposal for a new direction. ADF Health. 2002;3:77–85. [Google Scholar]
- 138.Rabone S, Phoon WO, Anderson SD, Wan KC, Seneviratne M, Gutierrez L, et al. Hypertonic saline challenge in an adult epidemiological field survey. Occup Med. 1996;46:177–85. doi: 10.1093/occmed/46.3.177. [DOI] [PubMed] [Google Scholar]
- 139.Riedler J, Reade T, Dalton M, Holst DI, Robertson CF. Hypertonic saline challenge in an epidemiological survey of asthma in children. Am J Respir Crit Care Med. 1994;150(6 Pt 1):1632–9. doi: 10.1164/ajrccm.150.6.7952626. [DOI] [PubMed] [Google Scholar]
- 140.Riedler J, Robertson CF. Effect of tidal volume on the output and particle size distribution of hypertonic saline from an ultrasonic nebuliser. Eur Respir J. 1994;7:998–1002. [PubMed] [Google Scholar]
- 141.Riedler J, Reade T, Robertson CF. Repeatability of response to hypertonic saline aerosol in children with mild to severe asthma. Pediatr Pulmonol. 1994;18:330–6. doi: 10.1002/ppul.1950180511. [DOI] [PubMed] [Google Scholar]
- 142.Riedler J, Gamper A, Eder W, Oberfeld G. Prevalence of bronchial hyperresponsiveness to 4.5% saline and its relation to asthma and allergy symptoms in Austrian children. Eur Respir J. 1998;11:355–60. doi: 10.1183/09031936.98.11020355. [DOI] [PubMed] [Google Scholar]
- 143.de Meer G, Postma DS, Janssen NA, de Jongste JC, Brunekreef B. Bronchial hyper-responsiveness to hypertonic saline and blood eosinophilic markers in 8–13 year old school children. Clin Exp Allergy. 2004;34:1226–31. doi: 10.1111/j.1365-2222.2004.02017.x. [DOI] [PubMed] [Google Scholar]
- 144.Anderson SD, Spring J, Moore B, Rodwell LT, Spalding N, Gonda I, et al. The effect of inhaling a dry powder of sodium chloride on the airways of asthmatic subjects. Eur Respir J. 1997;10:2465–73. doi: 10.1183/09031936.97.10112465. [DOI] [PubMed] [Google Scholar]
- 145.Anderson SD, Brannan J, Spring J, Spalding N, Rodwell LT, Chan K, et al. A new method for bronchial-provocation testing in asthmatic subjects using a dry powder of mannitol. Am J Respir Crit Care Med. 1997;156(3 Pt 1):758–65. doi: 10.1164/ajrccm.156.3.9701113. [DOI] [PubMed] [Google Scholar]
- 146.Eggleston PA, Kagey-Sobotka A, Schleimer RP, Lichtenstein LM. Interaction between hyperosmolar and IgE-mediated histamine release from basophils and mast cells. Am Rev Respir Dis. 1984;130:86–91. doi: 10.1164/arrd.1984.130.1.86. [DOI] [PubMed] [Google Scholar]
- 147.Eggleston PA, Kagey-Sobotka A, Lichtenstein LM. A comparison of the osmotic activation of basophils and human lung mast cells. Am Rev Respir Dis. 1987;135:1043–8. doi: 10.1164/arrd.1987.135.5.1043. [DOI] [PubMed] [Google Scholar]
- 148.Flint KC, Leung KBP, Pearce FL, Hudspith BN, Brostoff J, Johnson NM. The hyperosmolar release of histamine from bronchoalveolar mast cells and its inhibition by sodium cromoglycate. Thorax. 1985;40(9):717. [Google Scholar]
- 149.Brannan JD, Koskela H, Anderson SD, Chan H-K. Budesonide reduces sensitivity and reactivity to inhaled mannitol in asthmatic subjects. Respirology. 2002;7:37–44. doi: 10.1046/j.1440-1843.2002.00357.x. [DOI] [PubMed] [Google Scholar]
- 150.Holzer K, Anderson SD, Chan H-K, Douglass J. Mannitol as a challenge test to identify exercise-induced bronchoconstriction in elite athletes. Am J Respir Crit Care Med. 2003;167:534–47. doi: 10.1164/rccm.200208-916OC. [DOI] [PubMed] [Google Scholar]
- 151.Subbarao P, Brannan JD, Ho B, Anderson SD, Chan H-K, Coates AL. Inhaled mannitol identifies methacholine-responsive children with active asthma. Pediatr Pulmonol. 2000;29:291–8. doi: 10.1002/(sici)1099-0496(200004)29:4<291::aid-ppul9>3.0.co;2-a. [DOI] [PubMed] [Google Scholar]
- 152.Leuppi JD, Salome CM, Jenkins CR, Koskela H, Brannan JD, Anderson SD, et al. Markers of airway inflammation and airway hyperresponsiveness in patients with well-controlled asthma. Eur Respir J. 2001;18:444–50. doi: 10.1183/09031936.01.00058601. [DOI] [PubMed] [Google Scholar]
- 153.Leuppi JD, Salome CM, Jenkins CR, Anderson SD, Xuan W, Marks GB, et al. Predictive markers of asthma exacerbations during stepwise dose-reduction of inhaled corticosteroids. Am J Respir Crit Care Med. 2001;163:406–12. doi: 10.1164/ajrccm.163.2.9912091. [DOI] [PubMed] [Google Scholar]
- 154.Brannan JD, Anderson SD, Gomes K, King GG, Chan H-K, Seale JP. Fexofenadine decreases sensitivity to and montelukast improves recovery from inhaled mannitol. Am J Respir Crit Care Med. 2001;163:1420–5. doi: 10.1164/ajrccm.163.6.2006019. [DOI] [PubMed] [Google Scholar]
- 155.Koskela HO, Hyvarinen L, Brannan JD, Chan HK, Anderson SD. Sensitivity and validity of three bronchial provocation tests to demonstrate the effect of inhaled corticosteroids in asthma. Chest. 2003;124:1341–9. doi: 10.1378/chest.124.4.1341. [DOI] [PubMed] [Google Scholar]
- 156.Brannan JD, Anderson SD, Perry CP, Freed-Martens R, Lassig AR, Charlton B. The safety and efficacy of inhaled dry powder mannitol as a bronchial provocation test for airway hyperresponsiveness: a phase 3 comparison study with hypertonic (4.5%) saline. Respir Res. 2005;6:144. doi: 10.1186/1465-9921-6-144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Anderson SD, Charlton B, Weiler JM, Nichols S, Spector SL, Pearlman DS, et al. Comparison of mannitol and methacholine to predict exercise-induced bronchoconstriction and a clinical diagnosis of asthma. Respir Res. 2009;10:4. doi: 10.1186/1465-9921-10-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Brannan JD, Perry CP, Anderson SD. Mannitol (AridolTM) test results in asthmatic adults receiving inhaled corticosteroids. J Allergy Clin Immunol. 2013;131:906–7. doi: 10.1016/j.jaci.2012.08.028. [DOI] [PubMed] [Google Scholar]
- 159.GINA. Strategy for asthma management and prevention. 2015. chapter 1(5). Available from: http://wwwginasthma/org/local/uploads/files/GINA_Report2015_Trackedpdf.
- 160.Kersten ET, Driessen JM, van der Berg JD, Thio BJ. Mannitol and exercise challenge tests in asthmatic children. Pediatr Pulmonol. 2009;44:655–61. doi: 10.1002/ppul.21034. [DOI] [PubMed] [Google Scholar]
- 161. Barben J, Roberts M, Chew N, Carlin JB, Robertson CF. Repeatability of bronchial responsiveness to mannitol dry powder in children with asthma. Pediatr Pulmonol. 2003;36:490–4. doi: 10.1002/ppul.10378. [DOI] [PubMed] [Google Scholar]
- 162.Barben J, Kuehni C, Strippoli M-P, Schiller B, Hammer J, Trachsel D. Mannitol dry powder challenge in comparison with exercise-testing in children. Pediatric Pulmonol. 2011;46:842–8. doi: 10.1002/ppul.21453. [DOI] [PubMed] [Google Scholar]
- 163.Sverrild A, Porsbjerg C, Thomsen SF, Backer V. Diagnostic properties of inhaled mannitol in the diagnosis of asthma: a population study. J Allergy Clin Immunol. 2009;124:928–32. doi: 10.1016/j.jaci.2009.06.028. [DOI] [PubMed] [Google Scholar]
- 164.Backer V, Sverrild A, Ulrik CS, Bodtger U, Seersholm N, Porsbjerg C. Diagnostic work-up in patients with possible asthma referred to a university hospital. Eur Clin Respir J. 2015;2:27768. doi: 10.3402/ecrj.v2.27768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Porsbjerg C, Lund TK, Pedersen L, Backer V. Inflammatory subtypes in asthma are related to airway hyperresponsiveness to mannitol and exhaled NO. J Asthma. 2009;46:606–12. doi: 10.1080/02770900903015654. [DOI] [PubMed] [Google Scholar]
- 166.Anderson WJ, Lipworth BJ. Relationship of mannitol challenge to methacholine challenge and inflammatory markers in persistent asthmatics receiving inhaled corticosteroids. Lung. 2012;190:513–21. doi: 10.1007/s00408-012-9396-6. [DOI] [PubMed] [Google Scholar]
- 167.Clearie KL, Jackson CM, Fardon TC, Williamson PA, Vaidyanathan S, Burns P, et al. Supervised step-down of inhaled corticosteroids in the community-an observational study. Respir Med. 2011;105:558–65. doi: 10.1016/j.rmed.2010.10.004. [DOI] [PubMed] [Google Scholar]
- 168.Lipworth BJ, Short PM, Williamson PA, Clearie KL, Fardon TC, Jackson CM. A randomized primary care trial of steroid titration against mannitol in persistent asthma: STAMINA trial. Chest. 2012;141:607–15. doi: 10.1378/chest.11-1748. [DOI] [PubMed] [Google Scholar]
- 169.Henriksen JM, Dahl R. Effects of inhaled budesonide alone and in combination with low-dose terbutaline in children with exercise-induced asthma. Am Rev Respir Dis. 1983;128:993–7. doi: 10.1164/arrd.1983.128.6.993. [DOI] [PubMed] [Google Scholar]
- 170.Henriksen JM. Effect of inhalation of corticosteroids on exercise induced asthma: randomised double blind crossover study of budesonide in asthmatic children. Brit Med J. 1985;291:248–9. doi: 10.1136/bmj.291.6490.248-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Vathenen AS, Knox AJ, Wisniewski A, Tattersfield AE. Effect of inhaled budesonide on bronchial reactivity to histamine, exercise, and eucapnic dry air hyperventilation in patients with asthma. Thorax. 1991;46:811–6. doi: 10.1136/thx.46.11.811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Pedersen S, Hansen OR. Budesonide treatment of moderate and severe asthma in children: a dose-response study. J Allergy Clin Immunol. 1995;95(1 Pt 1):29–33. doi: 10.1016/s0091-6749(95)70149-4. [DOI] [PubMed] [Google Scholar]
- 173.Jonasson G, Carlsen KH, Hultquist C. Low-dose budesonide improves exercise-induced bronchospasm in schoolchildren. Pediatr Allergy Immunol. 2000;11:120–5. doi: 10.1034/j.1399-3038.2000.00067.x. [DOI] [PubMed] [Google Scholar]
- 174.du Toit JI, Anderson SD, Jenkins CR, Woolcock AJ, Rodwell LT. Airway responsiveness in asthma: bronchial challenge with histamine and 4.5% sodium chloride before and after budesonide. Allergy Asthma Proc. 1997;18:7–14. doi: 10.2500/108854197778612817. [DOI] [PubMed] [Google Scholar]
- 175.Aldridge RE, Hancox RJ, Robin Taylor D, Cowan JO, Winn MC, Frampton CM, et al. Effects of terbutaline and budesonide on sputum cells and bronchial hyperresponsiveness in asthma. Am J Respir Crit Care Med. 2000;161:1459–64. doi: 10.1164/ajrccm.161.5.9906052. [DOI] [PubMed] [Google Scholar]
- 176.Hofstra WB, Neijens HJ, Duiverman EJ, Kouwenberg JM, Mulder PG, Kuethe MC, et al. Dose-response over time to inhaled fluticasone propionate: treatment of exercise- and methacholine-induced bronchoconstriction in children with asthma. Pediatr Pulmonol. 2000;29:415–23. doi: 10.1002/(sici)1099-0496(200006)29:6<415::aid-ppul1>3.0.co;2-7. [DOI] [PubMed] [Google Scholar]
- 177.Subbarao P, Duong M, Adelroth E, Otis J, Obminski G, Inman M, et al. Effect of ciclesonide dose and duration of therapy on exercise-induced bronchoconstriction in patients with asthma. J Allergy Clin Immunol. 2006;117:1008–13. doi: 10.1016/j.jaci.2005.11.048. [DOI] [PubMed] [Google Scholar]
- 178.Israel E, Dermarkarian R, Rosenberg M, Sperling R, Taylor G, Rubin P, et al. The effects of a 5-lipoxygenase inhibitor on asthma induced by cold, dry air. N Engl J Med. 1990;323:1740–4. doi: 10.1056/NEJM199012203232505. [DOI] [PubMed] [Google Scholar]
- 179.Meltzer SS, Hasday JD, Cohn J, Bleecker ER. Inhibition of exercise-induced bronchospasm by zileuton: a 5-lipoxygenase inhibitor. Am J Respir Crit Care Med. 1996;153:931–5. doi: 10.1164/ajrccm.153.3.8630575. [DOI] [PubMed] [Google Scholar]
- 180.Leff JA, Busse WW, Pearlman D, Bronsky EA, Kemp J, Hendeles L, et al. Montelukast, a leukotriene-receptor antagonist, for the treatment of mild asthma and exercise-induced bronchoconstriction. N Engl J Med. 1998;339:147–52. doi: 10.1056/NEJM199807163390302. [DOI] [PubMed] [Google Scholar]
- 181.Coreno A, Skowronski M, Kotaur C, McFadden ER. Comparative effects of long-acting b2-agonists, leukotriene antagonists, and a 5-lipoxygenase inhibitor on exercise-induced asthma. J Allergy Clin Immunol. 2000;106:500–6. doi: 10.1067/mai.2000.109425. [DOI] [PubMed] [Google Scholar]
- 182.Edelman JM, Turpin JA, Bronsky EA, Grossman J, Kemp JP, Ghannam AF, et al. Oral montelukast compared with inhaled salmeterol to prevent exercise-induced bronchoconstriction. A randomized, double-blind trial. Exercise Study Group. Ann Intern Med. 2000;132:97–104. doi: 10.7326/0003-4819-132-2-200001180-00002. [DOI] [PubMed] [Google Scholar]
- 183.Rundell K, Spiering BA, Baumann JM, Evans TM. Effects of montelukast on airway narrowing from eucapnic voluntary hyperventilation and cold air exercise. Br J Sports Med. 2005;39:232–6. doi: 10.1136/bjsm.2004.014282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Reiss TF, Hill JB, Harman E, Zhang J, Tanaka WK, Bronsky E, et al. Increased urinary excretion of LTE4 after exercise and attenuation of exercise-induced bronchospasm by montelukast, a cysteinyl leukotriene receptor antagonist. Thorax. 1997;52:1030–5. doi: 10.1136/thx.52.12.1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.O'Sullivan S, Roquet A, Dahlén B, Larsen F, Eklund A, Kumlin M, et al. Evidence for mast cell activation during exercise-induced bronchoconstriction. Eur Respir J. 1998;12:345–50. doi: 10.1183/09031936.98.12020345. [DOI] [PubMed] [Google Scholar]
- 186.Brannan JD, Gulliksson M, Anderson SD, Chew N, Kumlin M. Evidence of mast cell activation and leukotriene release after mannitol inhalation. Eur Respir J. 2003;22:491–6. doi: 10.1183/09031936.03.00113403. [DOI] [PubMed] [Google Scholar]
- 187.Misso NLA, Aggarwal S, Phelps S, Beard R, Thompson PJ. Urinary leukotriene E4 and 9a,11b,-prostaglandin F2 concentrations in mild, moderate and severe asthma, and in healthy subjects. Clin Exp Allergy. 2004;34:624–31. doi: 10.1111/j.1365-2222.2004.1912.x. [DOI] [PubMed] [Google Scholar]
- 188.Brannan JD, Gulliksson M, Anderson SD, Chew N, Seale JP, Kumlin M. Inhibition of mast cell PGD2 release protects against mannitol-induced airway narrowing. Eur Respir J. 2006;27:944–50. doi: 10.1183/09031936.06.00078205. [DOI] [PubMed] [Google Scholar]
- 189.Hallstrand TS, Moody MW, Wurfel MM, Schwartz LB, Henderson WR, Aitken ML. Inflammatory basis of exercise-induced bronchoconstriction. Am J Respir Crit Care Med. 2005;172:679–86. doi: 10.1164/rccm.200412-1667OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Kippelen P, Larsson J, Anderson SD, Brannan JD, Dahlen B, Dahlen SE. Effect of sodium cromoglycate on mast cell mediators during hyperpnea in athletes. Med Sci Sports Exerc. 2010;42:1853–60. doi: 10.1249/MSS.0b013e3181da4f7d. [DOI] [PubMed] [Google Scholar]
- 191.Kippelen P, Larsson J, Anderson SD, Brannan JD, Delin I, Dahlen B, et al. Acute effects of beclomethasone on hyperpnea-induced bronchoconstriction. Med Sci Sports Exerc. 2010;42:273–80. doi: 10.1249/MSS.0b013e3181b541b1. [DOI] [PubMed] [Google Scholar]
- 192.Chan-Yeung M. The effect of Sch 1000 and disodium cromoglycate on exercise-induced asthma. Chest. 1977;71:320–3. doi: 10.1378/chest.71.3.320. [DOI] [PubMed] [Google Scholar]
- 193.Tullett WM, Tan KM, Wall RT, Patel KR. Dose-response effect of sodium cromoglycate pressurised aerosol in exercise induced asthma. Thorax. 1985;40:41–4. doi: 10.1136/thx.40.1.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Juniper EF, Kline PA, Morris MM, Hargreave FE. Airway constriction by isocapnic hyperventilation of cold, dry air: comparison of magnitude and duration of protection by nedocromil sodium and sodium cromoglycate. Clin Allergy. 1987;17:523–8. doi: 10.1111/j.1365-2222.1987.tb02048.x. [DOI] [PubMed] [Google Scholar]
- 195.Boulet L-P, Turcotte H, Tennina S. Comparative efficacy of salbutamol, ipratropium and cromoglycate in the prevention of bronchospasm induced by exercise and hyperosmolar challenges. J Allergy Clin Immunol. 1989;83:882–7. doi: 10.1016/0091-6749(89)90101-2. [DOI] [PubMed] [Google Scholar]
- 196.Edmunds A, Tooley M, Godfrey S. The refractory period after exercise-induced asthma: its duration and relation to the severity of exercise. Am Rev Respir Dis. 1978;117:247–54. doi: 10.1164/arrd.1978.117.2.247. [DOI] [PubMed] [Google Scholar]
- 197.Schoeffel RE, Anderson SD, Gillam I, Lindsay DA. Multiple exercise and histamine challenge in asthmatic patients. Thorax. 1980;35:164–70. doi: 10.1136/thx.35.3.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Wilson NM, Barnes PJ, Vickers H, Silverman M. Hyperventilation-induced asthma: evidence for two mechanisms. Thorax. 1982;37:657–62. doi: 10.1136/thx.37.9.657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Rosenthal RR, Laube BL, Hood DB, Norman PS. Analysis of refractory period after exercise and eucapnic voluntary hyperventilation challenge. Am Rev Respir Dis. 1990;141:368–72. doi: 10.1164/ajrccm/141.2.368. [DOI] [PubMed] [Google Scholar]
- 200.Belcher NG, Rees PJ, Clark TJM, Lee TH. A comparison of the refractory periods induced by hypertonic airway challenge and exercise in bronchial asthma. Am Rev Respir Dis. 1987;135:822–5. doi: 10.1164/arrd.1987.135.4.822. [DOI] [PubMed] [Google Scholar]
- 201.Larsson J, Perry CP, Anderson SD, Brannan JD, Dahlen S-E, Dahlen B. The occurrence of refractoriness and mast cell mediator release following mannitol-induced bronchoconstriction. J Appl Physiol. 2011;110:1029–35. doi: 10.1152/japplphysiol.00978.2010. [DOI] [PubMed] [Google Scholar]