Abstract
A rapid and sensitive (100%) matrix-assisted laser desorption ionization−time of flight mass spectrometry (MALDI-TOF MS) assay was developed to detect OXA-48-type producers, using 161 previously characterized clinical isolates. Ertapenem was monitored to detect carbapenem resistance, and temocillin was included in the assay as a marker for OXA-48-producers. Structural analysis of temocillin is described. Data are obtained within 60 min.
TEXT
Carbapenemase-producing Enterobacteriaceae (CPE) are increasingly being isolated worldwide. As these bacteria are difficult to detect and control, their spread is considered a public health concern (1, 2). Dissemination of the group D carbapenemases, particularly, blaOXA-48 isolates, is of major concern (3–5). These isolates are characterized by low carbapenem MICs and often go undetected by most phenotypic methods.
The development of mass spectrometry techniques, such as matrix-assisted laser desorption ionization−time of flight mass spectrometry (MALDI-TOF MS), has yielded sensitivity with faster turnaround times (6–10). The aim of this study was to determine whether a MALDI-TOF MS ertapenem assay can be used to identify and classify CPE producers, with particular emphasis on OXA-48-type isolates. We propose the use of temocillin as a resistance marker for OXA-48-type enzymes (11), as isolates harboring these types of carbapenemases show very high levels of resistance to temocillin (12, 13), and there is no specific inhibitor for group D carbapenemases. Phenylboronic acid (PBA) and dipicolinic acid (DPA) are used as inhibitors of, respectively, group A and B carbapenemases.
A total of 161 characterized nonrepeat Enterobacteriaceae isolates, 117 of which carried a carbapenemase enzyme, were tested (Table 1). Seventy-two of the 161 isolates were genotypically characterized by the Antibiotic Resistance Surveillance Program of the Spanish National Center of Microbiology (14), and the remaining 89 were characterized by the Microbiology Service of the A Coruña University Hospital Complex (15). The following isolates were identified: 79 Klebsiella pneumoniae, 16 Klebsiella. oxytoca, 22 Enterobacter cloacae, 5 Enterobacter aerogenes, 35 Escherichia coli, 3 Serratia marcescens, and 1 Citrobacter freundii.
TABLE 1.
Results of the comparison of the combined disk method and the MALDI-TOF MS method with specific A and B class inhibitors for detecting Enterobacteriaceae suspected of carbapenemase production
| β-Lactamase class | No. of isolates per β-lactamasea | MALDI-TOF MS (% [no.]) assayb |
MALDI-TOF MS avg time of ERTf hydrolysis detection (min) | TEMc analysis |
Interpreted as class D carbapenemase positive by MALDI-TOF MS (no.) | |||
|---|---|---|---|---|---|---|---|---|
| PBAd analysis: interpreted as class A carbapenemase positive | DPAe analysis: interpreted as class B carbapenemase positive | Resistanceg (<12 mm) | TEM sensitivity by MALDI-TOF MSh | Average time of TEM hydrolysis detection (min) | ||||
| None | 5 | |||||||
| SHV | 5 | |||||||
| EBC | 5 | |||||||
| K-1 | 5 | |||||||
| FOX | 1 | |||||||
| CMY-2 | 4 | |||||||
| DHA | 5 | |||||||
| TEM | 1 | |||||||
| CTX-M | 13 | 7 (1/13) | 1/1 | 30 | ||||
| KPC | 20 | 100 (20/20) | 50 | 50 | ||||
| VIM | 30 | 100 (30/30) | 30 | 70 (19/30) | 100 (19/19) | 35 | ||
| NDM | 7 | 100 (7/7) | 55 | 14(1/7) | 1/1 | 60 | ||
| IMP | 15 | 87 (13/15) | 80 | 27 (4/15) | 100 (4/4) | 45 | ||
| OXA | 45 | 60 | 100 (45/45) | 100 (45/45) | 15 | 100 (45/45) | ||
A total of 161 isolates (44 noncarbapenemase producers and 117 carbapenemase isolates), distributed as indicated, were studied.
Number of strains that yielded a positive result in the carbapenemase group classification by MALDI-TOF.
TEM, temocillin.
PBA, phenylboronic acid.
DPA, dipicolinic acid.
ERT, ertapenem.
Temocillin resistance percentage calculated on the zone diameter around temocillin table (Rosco).
Sensitivity of detection of temocillin resistance by MALDI-TOF MS. The resistance detected by temocillin disks (Rosco) was used as a reference.
All isolates were analyzed by the disk diffusion method (KPC/MBL and OXA-48 confirm kit; Rosco Diagnostica A/S, Taastrup, Denmark) with disks containing meropenem (10 μg), meropenem plus PBA, meropenem plus DPA, meropenem plus cloxacillin (CL), and temocillin (30 μg). A 0.5-McFarland suspension of the isolates on Mueller-Hinton II agar plates (Becton Dickinson, USA) was used, followed by an overnight incubation of the plates at 37°C. The susceptibility cutoff used for temocillin was <12 mm (12).
For the MALDI-TOF MS assay, isolates were previously grown overnight at 37°C in Trypticase soy agar with 5% sheep blood (TSA II) (Becton Dickinson). The hydrolysis of ertapenem (0.5 mg/ml Invaz [Merck & Co., Inc.], 0.001% SDS, 50 mM NH4HCO3 [pH 7.0]) (6, 9) and temocillin (1.5 mg/ml Negaban [Eumedica], 0,001% SDS, 50 mM NH4HCO3 [pH 7.0]) (9) was evaluated after resuspension of the bacteria that filled a 1-μl inoculation loop in 10 μl of the antibiotic solution in an Eppendorf tube and incubation of the suspension at 37°C under agitation. When inhibitors were used, bacteria were resuspended in 5 μl of ertapenem (1 mg/ml Invaz, 0.001% SDS, 50 mM NH4HCO3 [pH 7.0]) and in 5 μl of the corresponding inhibitor solution: PBA (2 mg/ml; Sigma-Aldrich, Munich, Germany) for group A and DPA (3 mg/ml; Sigma-Aldrich) for group B carbapenemases.
Incubation times of 15, 30, 60, 90, and 180 min were tested for each isolate. The Eppendorf tubes were then centrifuged for 2 min at 13,000 × g at room temperature, and 1 μl of the supernatant was applied under 1 μl of α-cyano-4-hydroxycinnamic acid (HCCA), which is used as the matrix in the MALDI-TOF MS target. After drying of the matrix, measurements were performed (6).
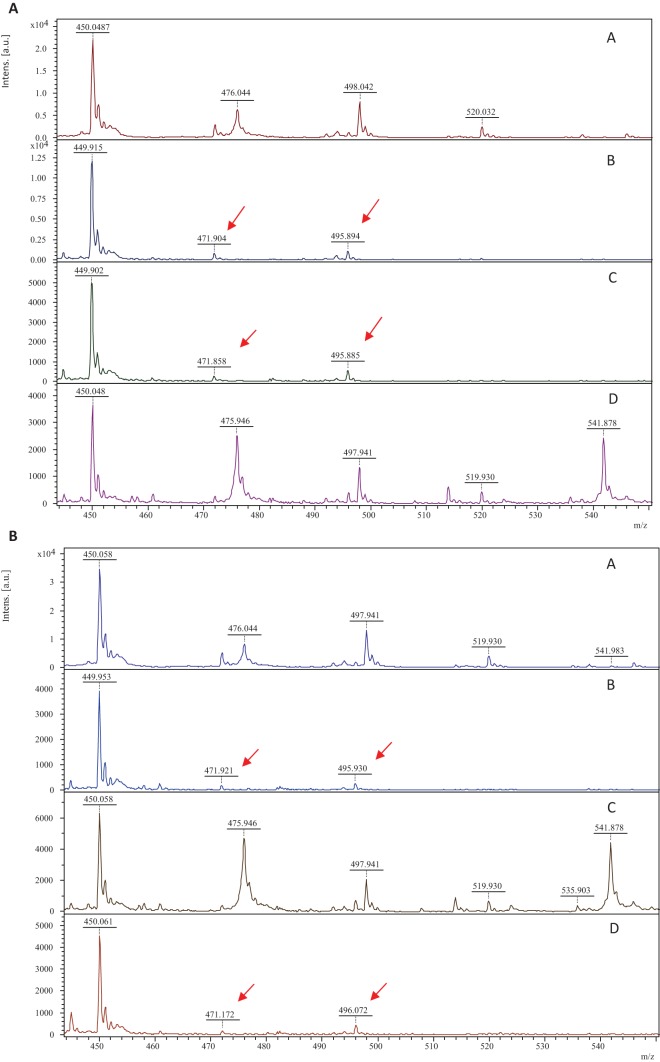
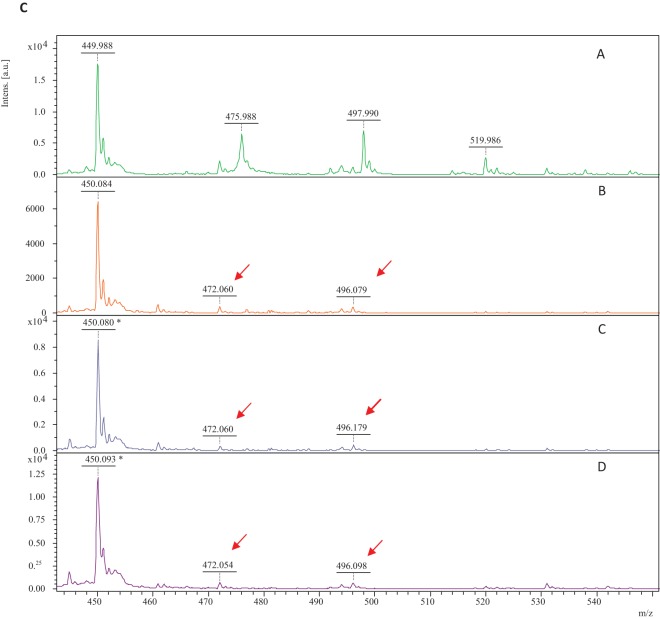
MALDI-TOF MS analysis, calibration, and spectra processing (flexAnalysis software; Bruker Daltonics GmbH, Germany) were carried out as previously described by Oviaño et al. (10). The ertapenem spectrum was interpreted according to Sparbier et al. (6). For group A carbapenemase classification, mass peaks of ertapenem must reappear when PBA is added, and when DPA is added, mass peaks of the hydrolyzed form must remain, unlike for group B carbapenemases (Fig. 1a and b). For group D carbapenemase detection, ertapenem hydrolysis mass peaks must remain, regardless of the type of inhibitor added (Fig. 1c).
FIG 1.
(a) Ertapenem mass spectra generated by group A carbapenemases: ertapenem mass spectra (negative control in the experiment) (spectrum A); mass spectra of ertapenem plus a blaKPC-2 E. cloacae strain after 60 min of exposure (spectrum B); ertapenem plus blaKPC-2 E. cloacae plus DPA (spectrum C); and ertapenem plus blaKPC-2 E. cloacae plus PBA (spectrum D). The peaks indicated (↙) are representative of ertapenem and its hydrolyzed forms. (b) Ertapenem mass spectra generated by group B carbapenemases: ertapenem mass spectra (negative control in the experiment) (spectrum A); mass spectra of ertapenem plus a blaVIM-1 E. coli strain after 15 min of exposure (spectrum B); ertapenem plus blaVIM-1 E. coli plus DPA (spectrum C); and ertapenem plus blaVIM-1 E. coli plus PBA (spectrum D). The peaks indicated (↙) are representative of ertapenem and its hydrolyzed forms. (c) Ertapenem mass spectra generated by group D carbapenemases: ertapenem mass spectra (negative control in the experiment) (spectrum A); mass spectra of ertapenem plus a blaOXA-48 K. pneumoniae strain after 90 min of exposure (spectrum B); ertapenem plus blaOXA-48 K. pneumoniae plus DPA (spectrum C); and ertapenem plus blaOXA-48 K. pneumoniae plus PBA (spectrum D). The peaks indicated (↙) are representative of ertapenem and its hydrolyzed forms. Intens., intensity; a.u., arbitrary units.
None of the sensitive β-lactamase carrier isolates, AmpC-type isolates, or extended-spectrum β-lactamase [ESBL]-type isolates were classified as carbapenemase positive in the MALDI-TOF MS assay. Twenty-seven of the 44 isolates (61%) were found to be resistant to ertapenem by phenotypic methods (disk diffusion and Etest), but none of them hydrolyzed ertapenem (Table 1). These isolates were characterized as blaSHV-1 (n = 4), blaK-1 (n = 5), blaEBC (n = 5), blaDHA (n = 2), and blaCTX-M (n = 11).
The average time for detection of ertapenem hydrolysis in class A carbapenemases by MALDI-TOF MS was 50 min, with 100% sensitivity. For detection of class B carbapenemases, the average time for ertapenem hydrolysis detection ranged from 30 to 80 min, and the sensitivity was 96% with a specificity of 100%. Within class B, VIM-type and NDM-type carbapenemases were accurately detected with 100% sensitivity and IMP-type producers with 87% sensitivity. The two undetected isolates were IMP-22-type E. cloacae. For identification of class D carbapenemases, MALDI-TOF MS exhibited 100% sensitivity for detecting ertapenem hydrolysis for all of the isolates tested, in an average time of 60 min, with no inhibition shown for any of the inhibitors (Table 1). To confirm the classification, temocillin hydrolysis was also evaluated.
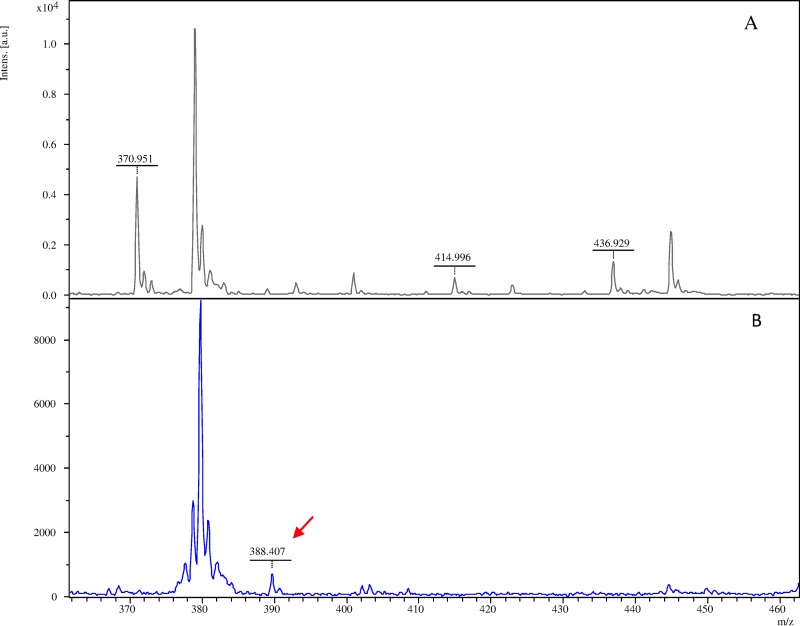
Structural analysis and experimental data on the temocillin MS spectrum revealed the following: the molecular ion peak for temocillin [M + H]+ at 415 Da, the sodium adduct [M + Na]+ at 437 Da, and the metabolite corresponding to the elimination of the carboxyl group [Mdecarb + H]+ at 371 Da (Fig. 2). The main temocillin species detected correspond to the decarboxylated form, for which the mass peak tends to be more intense (around a 75% relative difference in the area under the peak) than for the sodium adduct and the molecular ion peak. The hydrolyzed form seems to be very unstable, as it was not detected in the spectrum: only a small peak for the hydrolyzed decarboxylated form [Mhydrol/decarb + H]+ was sometimes detected at 389 Da. Temocillin resistance was determined either by the complete disappearance of temocillin peaks or by disappearance of the temocillin peaks plus the appearance of the hydrolyzed form. The relative intensity of the mass peaks within the same compound and spectrum is not relevant for interpretation of the resistance pattern.
FIG 2.
Temocillin mass spectra generated by OXA-48-type isolates (group D carbapenemases). Mass spectra of temocillin (A) and its hydrolyzed form (B) 15 min after exposure to a blaOXA-48 K. pneumoniae strain. The mass peaks indicated are representative of temocillin and its hydrolyzed form (↙).
All OXA-48-type enzymes (45/45, 100% sensitivity) were resistant to temocillin, and the MALDI-TOF MS assay was able to detect temocillin hydrolysis for all OXA-48 producers tested within 15 min. Temocillin resistance was also observed in 24 MBL producers and in a CTX-M producer (Table 1) (16). Thus, for accurate detection of the carbapenemase class, a carbapenem plus inhibitors must be included in the assay. However, the MALDI-TOF MS assay yields more accurate identification because the time required for temocillin hydrolysis can be used as a highly sensitive marker. The time required for detection of temocillin hydrolysis in OXA-48-type enzymes is 15 min, and the time needed for the other carbapenemases to reach a positive result is longer in all cases (more than 30 min), except for 3 VIM-1 K. pneumoniae isolates. In areas where OXA-48 producers are prevalent, the MALDI-TOF MS assay with inclusion of temocillin alone is highly sensitive, with a specificity of 93% (45/48) for a 15-min test. The method also helps to discount the possible presence of CPE, because of its high negative predictive value (100%).
The MALDI-TOF MS assay showed an overall sensitivity of 98% (115/117) and an overall specificity of 100% (117/117) for detecting carbapenemase producers. In comparison with the disk diffusion method, the MALDI-TOF MS assay shows the same sensitivity and specificity (100%) for detecting group A and D carbapenemases, but the results are produced faster (24 h earlier). For detection of group B carbapenemases, the disk diffusion assay exhibits a sensitivity of 80% (42/52) and the MALDI-TOF MS assay exhibits a sensitivity of 96%.
The assay was also performed on 8 blood cultures that were genotypically characterized as blaOXA-48 K. pneumoniae, obtained during an outbreak in August in the A Coruña University Hospital, giving an unequivocal pattern of an OXA-type isolate. The tests yielded promising results for future applications.
The biochemical Carba NP test yielded poor detection of OXA-48-type enzymes (sensitivity of around 80%), in contrast to the proposed method, which shows 100% sensitivity (9, 17, 18). The vast majority of articles reporting other MALDI-TOF MS assays focus on detecting carbapenemases (6, 7, 9), and few focus on detecting metallo-β-lactamases (19) or oxacillinases (20). However, we report here for the first time a MALDI-TOF MS assay that enables detection and classification into Ambler classes A, B, and D simultaneously. Detection of OXA-48-type producers is particularly improved: other MALDI-TOF MS assays including ertapenem do not yield positive results within 2 h because they require an incubation time of 24 h (8). OXA-48-type producers have been detected within 90 min in imipenem-based assays (21) and within 120 min in meropenem-based assays (9); however, because inhibitors or any other resistance marker such as temocillin were not included, the isolates could only be classified as carbapenemase producers and could not be further identified as OXA-48-type producers without the support of genotypic methods. In the proposed new assay, ertapenem hydrolysis occurs in 60 min and temocillin hydrolysis in 15 min for OXA-48-type producers. This is due to the combined use in the reaction buffer of NH4HCO3, which enhances ertapenem sensitivity (9), and SDS, which helps to lyse slime-producing isolates. In areas where OXA-48 producers are prevalent, inclusion of ertapenem and temocillin yields a reliable and ready to use assay for clinical practice. We suggest first testing the two antibiotics at 60 and 15 min, respectively. The assay shows 100% sensitivity and 93% specificity for detecting OXA-48-producers. If ertapenem plus inhibitors is tested at 60 min and temocillin at 15 min, the specificity reaches 100%. In areas where other resistance patterns are predominant, we suggest testing ertapenem plus inhibitors after 90 min. Informatics tools for automatic spectrum interpretation, such as STAR-BL prototype software (Bruker Daltonics GmbH), might be helpful for nonexperienced users to facilitate interpretation of the spectrum. Furthermore, they allow the calculation of the hydrolysis ratio of the antibiotics which might a useful tool for future studies (22).
ACKNOWLEDGMENTS
We thank Sebastian van de Velde, Scientific Manager at Eumedica S.A., for providing us with temocillin (Negaban) and Soria-Melguizo S.A. for technical support.
Funding Statement
This work was supported by grants from the following organizations: Fondo de Investigación Sanitaria (grants PI12/01242 to J.O. and PI12/00512 to G.B.); the Antibiotic Resistance Surveillance Programme of the Spanish Centro Nacional de Microbiología, Instituto de Salud Carlos III, Ministerio de Economía y Competitividad; the Plan Nacional de I+D+I 2008–2011; and the Instituto de Salud Carlos III, Subdirección General de Redes y Centros de Investigación Cooperativa, Ministerio de Economía y Competitividad, Spanish Network for Research in Infectious Diseases (REIPI RD12/0015), cofinanced by European Development Regional Fund “A Way to Achieve Europe” ERDF. The funders had no role in the study design, data collection, interpretation, or the decision to submit the work for publication.
REFERENCES
- 1.Nordmann P, Poirel L. 2014. The difficult-to-control spread of carbapenemase producers among Enterobacteriaceae worldwide. Clin Microbiol Infect 20:821–830. doi: 10.1111/1469-0691.12719. [DOI] [PubMed] [Google Scholar]
- 2.Cantón R, Akóva M, Carmeli Y, Giske CG, Glupczynski Y, Gniadkowski M, Livermore DM, Miriagou V, Naas T, Rossolini GM, Samuelsen Ø, Seifert H, Woodford N, Nordmann P, European Network on Carbapenemases . 2012. Rapid evolution and spread of carbapenemases among Enterobacteriaceae in Europe. Clin Microbiol Infect 18:413–431. doi: 10.1111/j.1469-0691.2012.03821.x. [DOI] [PubMed] [Google Scholar]
- 3.Pfeifer Y, Schlatterer K, Engelmann E, Schiller RA, Frangenberg HR, Stiewe D, Holfelder M, Witte W, Nordmann P, Poirel L. 2012. Emergence of OXA-48-type carbapenemase-producing Enterobacteriaceae in German hospitals. Antimicrob Agents Chemother 56:2125–2128. doi: 10.1128/AAC.05315-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pitart C, Solé M, Roca I, Fabrega A, Vila J, Marco F. 2011. First outbreak of a plasmid-mediated carbapenem-hydrolyzing OXA-48-lactamase in Klebsiella pneumoniae in Spain. Antimicrob Agents Chemother 55:4398–4401. doi: 10.1128/AAC.00329-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Potron A, Poirel L, Nordmann P. 2014. Derepressed transfer properties leading to the efficient spread of the plasmid encoding carbapenemase OXA-48. Antimicrob Agents Chemother 58:467–471. doi: 10.1128/AAC.01344-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sparbier K, Schubert S, Weller U, Boogen C, Kostrzewa M. 2012. Matrix-assisted laser desorption ionization-time of flight mass spectrometry-based functional assay for rapid detection of resistance against β-lactam antibiotics. J Clin Microbiol 50:927–937. doi: 10.1128/JCM.05737-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Burckhardt I, Zimmermann S. 2011. Using matrix-assisted laser desorption ionization-time of flight mass spectrometry to detect carbapenem resistance within 1 to 2.5 hours. J Clin Microbiol 49:3321–3324. doi: 10.1128/JCM.00287-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Johansson A, Ekelöf J, Giske CG, Sundqvist M. 2014. The detection and verification of carbapenemases using ertapenem and matrix assisted laser desorption ionization−time of flight. BMC Microbiol 14:89. doi: 10.1186/1471-2180-14-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Papagiannitsis CC, Študentová V, Izdebski Oikonomou RO, Pfeifer Y, Petinaki E, Hrabák J. 2015. MALDI-TOF MS meropenem hydrolysis assay with NH4HCO3, a reliable tool for the direct detection of carbapenemase activity. J Clin Microbiol 53:1731–1735. doi: 10.1128/JCM.03094-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Oviaño M, Fernández B, Fernández A, Barba MJ, Mouriño C, Bou G. 2014. Rapid detection of Enterobacteriaceae producing extended spectrum beta-lactamases directly from positive blood cultures by matrix-assisted laser desorption ionization−time of flight mass spectrometry. Clin Microbiol Infect 20:1146–1157. doi: 10.1111/1469-0691.12729. [DOI] [PubMed] [Google Scholar]
- 11.Livermore DM, Tulkens PM. 2009. Temocillin revived. J Antimicrob Chemother 63:243–245. doi: 10.1093/jac/dkn511. [DOI] [PubMed] [Google Scholar]
- 12.Huang TD, Poirel L, Bogaerts P, Berhin C, Nordmann P, Glupczynski Y. 2014. Temocillin and piperacillin/tazobactam resistance by disc diffusion as antimicrobial surrogate markers for the detection of carbapenemase-producing Enterobacteriaceae in geographical areas with a high prevalence of OXA-48 producers. J Antimicrob Chemother 69:445–450. doi: 10.1093/jac/dkt367. [DOI] [PubMed] [Google Scholar]
- 13.Hartl R, Widhalm S, Kerschner H, Apfalter P. 2013. Temocillin and meropenem to discriminate resistance mechanisms leading to decreased carbapenem susceptibility with focus on OXA-48 in Enterobacteriaceae. Clin Microbiol Infect 19:E230−E232. doi: 10.1111/1469-0691.12146. [DOI] [PubMed] [Google Scholar]
- 14.Oteo J, Saez D, Bautista V, Fernández-Romero S, Hernández-Molina JM, Pérez-Vázquez M, Aracil B, Campos J, Spanish Collaborating Group for the Antibiotic Resistance Surveillance Program . 2013. Carbapenemase-producing Enterobacteriaceae in Spain in 2012. Antimicrob Agents Chemother 57:6344–6347. doi: 10.1128/AAC.01513-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Pérez-Pérez FJ, Hanson ND. 2002. Detection of plasmid-mediated AmpC β-lactamase genes in clinical isolates by using multiplex PCR. J Clin Microbiol 40:2153–2162. doi: 10.1128/JCM.40.6.2153-2162.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.van Dijk K, Voets GM, Scharringa J, Voskuil S, Fluit AC, Rottier WC, Leverstein-Van Hall MA, Cohen Stuart JW. 2014. A disc diffusion assay for detection of class A, B and OXA-48 carbapenemases in Enterobacteriaceae using phenyl boronic acid, dipicolinic acid and temocillin. Clin Microbiol Infect 20:345–349. doi: 10.1111/1469-0691.12322. [DOI] [PubMed] [Google Scholar]
- 17.Tijet N, Boyd D, Patel S, Mulvey M, Melano R. 2013. Evaluation of the Carba NP test for rapid detection of carbapenemase-producing Enterobacteriaceae and Pseudomonas aeruginosa. Antimicrob Agents Chemother 57:4578–4580. doi: 10.1128/AAC.00878-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Chong P, McCorrister S, Unger M, Boyd D, Mulvey M, Westmacott G. 2015. MALDI-TOF MS detection of carbapenemase activity in clinical isolates of Enterobacteriaceae spp., Pseudomonas aeruginosa, and Acinetobacter baumannii compared against the Carba-NP assay. J Microbiol Methods 111:21–23. doi: 10.1016/j.mimet.2015.01.024. [DOI] [PubMed] [Google Scholar]
- 19.Hoyos-Mallecot Y, Cabrera-Alvargonzalez JJ, Miranda-Casas C, Rojo-Martín MD, Liebana-Martos C, Navarro-Marí JM. 2014. MALDI-TOF MS, a useful instrument for differentiating metallo-β-lactamases in Enterobacteriaceae and Pseudomonas spp. Lett Appl Microbiol 58:325–329. doi: 10.1111/lam.12203. [DOI] [PubMed] [Google Scholar]
- 20.Álvarez-Buylla A, Picazo JJ, Culebras E. 2013. Optimized method for Acinetobacter species carbapenemase detection and identification by matrix-assisted laser desorption ionization−time of flight mass spectrometry. J Clin Microbiol 51:1589–1592. doi: 10.1128/JCM.00181-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sauget M, Cabrolier N, Manzoni M, Bertrand X, Hocquet D. 2014. Rapid, sensitive and specific detection of OXA-48-like-producing Enterobacteriaceae by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J Microbiol Methods 105:88–91. doi: 10.1016/j.mimet.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 22.Jung J, Popp C, Sparbier K, Lange C, Kostrzewa M, Schubert S. 2014. Evaluation of matrix-assisted laser desorption ionization-time of flight mass spectrometry for rapid detection of β-lactam resistance in Enterobacteriaceae derived from blood cultures. J Clin Microbiol 52:924–930. doi: 10.1128/JCM.02691-13. [DOI] [PMC free article] [PubMed] [Google Scholar]