Abstract
Established prognostic tools in patients with myelodysplastic syndromes (MDS) were largely derived from untreated patient cohorts. Although azanucleosides are standard therapies for higher-risk (HR)-MDS, the relative prognostic performance of existing prognostic tools among patients with HR-MDS receiving azanucleoside therapy is unknown. In the MDS Clinical Research Consortium database, we compared the prognostic utility of the International Prognostic Scoring System (IPSS), revised IPSS (IPSS-R), MD Anderson Prognostic Scoring System (MDAPSS), World Health Organization-based Prognostic Scoring System (WPSS) and the French Prognostic Scoring System (FPSS) among 632 patients who presented with HR-MDS and were treated with azanucleosides as the first-line therapy. Median follow-up from diagnosis was 15.7 months. No prognostic tool predicted the probability of achieving an objective response. Nonetheless, all five tools were associated with overall survival (OS, P = 0.025 for the IPSS, P = 0.011 for WPSS and P < 0.001 for the other three tools). The corrected Akaike Information Criteria, which were used to compare OS with the different prognostic scoring systems as covariates (lower is better) were 4138 (MDAPSS), 4156 (FPSS), 4196 (IPSS-R), 4186 (WPSS) and 4196 (IPSS). Patients in the highest-risk groups of the prognostic tools had a median OS from diagnosis of 11 – 16 months and should be considered for up-front transplantation or experimental approaches.
INTRODUCTION
Myelodysplastic syndromes (MDS) comprise a group of heterogeneous hematopoietic stem cell malignancies with dysregulated differentiation leading to peripheral cytopenias, prominent dysplastic cell morphology changes and a variable propensity for leukemic progression.1,2 MDS also exhibit wide heterogeneity in the clinical course and outcomes of individual patients.3–5 Accurate risk stratification is thus extremely important for informed counseling of patients and guiding recommendations for therapeutic interventions.6 Since the publication of the first widely used prognostic tool in 1997, the International Prognostic Scoring System (IPSS),7 several other risk stratification models have been developed and validated to address some of the important limitations of the IPSS.8 The dynamic WHO (World Health Organization) classification-based Prognostic Scoring System (WPSS),9 which incorporated transfusion needs, was published in 2007 and the global MD Anderson Prognostic Scoring System (MDAPSS),10 which included secondary MDS and proliferative overlap disorders, shortly followed in 2008. In addition, the IPSS itself was revised and the revised version (IPSS-R),11 which gave more weight to cytogenetic abnormalities and degrees of cytopenias, was published in 2012.
Despite the introduction of other models and increased understanding of the utility of integrating some of the newly discovered prognostic recurrent molecular mutations in prognostic systems, these four prognostic tools (IPSS, WPSS, MDAPSS and IPSS-R) remain the most widely used risk stratification models in clinical practice.12 In practice, these systems generally separate MDS patients into two broad prognostic groups: those with lower-risk disease and those with higher-risk (HR)-MDS. Whenever possible, patients with HR-MDS are treated with aggressive interventions such as allogeneic hematopoietic stem cell transplantation and/or azanucleoside therapy with the goal of altering the natural history of the disease and prolonging survival. The azanucleosides azacitidine and decitabine were approved for treatment of MDS by the US Food and Drug Administration in 2004 and 2006, respectively. Although azacitidine remains the only drug demonstrated to prolong survival in patients with HR-MDS in a randomized phase 3 study,13 both azacitidine and decitabine have been shown to result in objective hematologic responses in 40–60% of patients including complete remission (CR) in 10–20% of patients, delay progression to acute myeloid leukemia (AML) and improve quality of life.13–20
Importantly, many patients with HR-MDS who receive azanucleosides do not derive clinical benefit. In addition, neither drug is curative and most responding patients lose their response within two years.21 Moreover, patients with HR-MDS who do not respond or lose initial response after azanucleoside therapy have a dismal prognosis with a median overall survival (OS) of <6 months.22,23 For all these reasons, extensive research has been dedicated to identify clinical or laboratory predictors for clinical benefit from azanucleoside therapy in patients with HR-MDS.24 Unfortunately, no such reliable predictors have been discovered that would enable patients with lower probability of benefit to avoid prolonged, expensive, ineffective and potentially toxic azanucleoside therapy, and to be considered for more intensive or experimental treatment approaches early in the disease course.25,26 The survival estimates provided by the prognostic tools for MDS are usually factored in the decision of whether to recommend azanucleoside therapy for individual patients. However, three of the commonly used tools (IPSS, WPSS and IPSS-R) were developed from patient cohorts who had not received azanucleoside therapy, while the MDAPSS was published shortly after the approval of azanucleosides and included only a small proportion of patients treated with these agents.7,9–11
In this analysis, we sought to compare the relative prognostic discriminatory power of the four commonly used prognostic models (IPSS, WPSS, MDAPSS and IPSS-R) and the French Prognostic Scoring System (FPSS), which was developed specifically for azacitidine-treated HR-MDS patients,27,28 in a large multicenter cohort of HR-MDS patients who received azanucleosides at one of the centers in the MDS Clinical Research Consortium.
METHODS
Study cohort
In this multi-institution study, a combined MDS database obtained from six institutions in the MDS Clinical Research Consortium (H. Lee Moffitt Cancer Center and Research Institute, Cleveland Clinic, MD Anderson Cancer Center, Dana-Farber Cancer Institute, Weill Medical College of Cornell University and the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins) was used to identify patients. Eligible patients had a pathologically confirmed diagnosis of MDS and HR-disease defined by an IPSS risk score of 1.5 or more (that is, IPSS intermediate-2 [INT-2] and high-risk groups) at presentation to the institutions and subsequently received azanucleoside therapy (azacitidine or decitabine) as first-line therapy. Data were collected for each eligible patient locally and then imported in the Consortium database. The study was approved by the Investigational Review Board in each institution.
Calculation of risk status using the five prognostic tools
The prognostic scores were calculated and risk categories were determined for every patient using all five risk stratification models (IPSS, IPSS-R, MDAPSS, WPSS and the FPSS) as previously described.7,9–11,27,28 The IPSS and the IPSS-R were calculated using the baseline bone marrow blast percentage, cytogenetics and the number (also severity for IPSS-R) of cytopenias.7,11 The IPSS has four risk categories: low, INT-1, INT-2 and high, while the IPSS-R has five risk categories (very low, low, INT, high and very high). The MDAPSS was calculated using seven baseline parameters (age, Eastern Cooperative Cancer Group performance status, platelet count, hemoglobin level, bone marrow blast percentage, cytogenetics and red blood cell (RBC) transfusion dependency) to categorize patients into four prognostic groups (low INT-1, INT-2 and high).10 The WPSS was calculated using the baseline WHO class, cytogenetics and RBC transfusion dependency and used five risk categories (very low, low, INT, high and very high).9 A non-applicable risk category was added to the WPSS for patients in whom the WPSS score could not be calculated because of missing WHO classification. The FPSS was calculated using four baseline clinical criteria: Eastern Cooperative Cancer Group performance status, cytogenetics, presence of peripheral blood blasts and RBC transfusion dependency to group patients into three risk categories (low, INT and high).27,28
Response and survival determination
Responses to azanucleoside therapy were defined as per the International Working Group 2006 (IWG-2006) criteria.29 According to the best achieved response, patients were categorized into responders (CR, partial response, hematologic improvement and marrow CR) and non-responders (stable disease and progressive disease (PD)). Overall response rate (ORR) was defined as the sum of patients who were CR, partial response and hematologic improvement. OS was calculated from the time of diagnosis of MDS to the time of death or last known follow-up. Leukemia-free survival (LFS) was calculated from the time of diagnosis of MDS to the time of death, diagnosis of acute myeloid leukemia (≥30% bone marrow blasts) or last known follow-up.
Statistical considerations
Multiple imputation using the chained equation approach was used to impute all missing data by creating a number of complete data sets and using plausible values to account for variables that would otherwise be missing. Restrictions were placed on the imputations that prevented out of range values from being included in the imputed data set. Multivariate imputation by chained equations with random forest per-variable imputation was used to impute 100 data sets. The 100 imputations were generated from a model with 97 variables using random forest imputation for missing continuous data and logistic/polytomous imputation for missing categorical data. All statistical analyses were performed on the 100 completed data sets, then combined using the standard multiple imputation combining rules. P-values and 95% confidence intervals (95% CIs) were calculated from complete data estimates using the standard multiple imputation combining rules to reflect the increased uncertainty due to missing data. The fraction of missing information, which represents the impact the missing data have on the quantity of interest was also estimated for every analysis.
In survival analyses, observations were censored for patients who were last known to be alive. Median OS was estimated using the Kaplan – Meier method, and differences for subgroups were assessed using the log-rank test. Stratified (by institution) Cox proportional hazards models were fit to assess association of prognostic systems with OS. Corrected Akaike Information Criteria (AICc) were used to compare these fitted models. The AIC is a calculated value that is used to inform model selection when there are a number of competing models to estimate the data set by providing a quantitative estimation of the quality of each model relative to each of the other models. A smaller AIC value is considered as an indicator of a better fit of the model to the data. Demographics and baseline characteristics were summarized. Medians and inter-quartile ranges were used to summarize continuous data and counts and percentages for categorical data. Logistic regression models were fitted and tested for association of prognostic risk categories with ORR. All analyses were two tailed and performed using an alpha significance threshold of 0.05. R version 3.1.1 (2014-07-10; R Core Team (2014, Vienna, Austria; http://www.R-project.org/)) was used for all analyses.
RESULTS
Baseline demographics and characteristics
The demographics and baseline characteristics of the 632 patients included in the study are shown in Table 1. The median age of the cohort was 70 years (inter-quartile ranges: 64, 76 years) with 86.2% ≥ 60 years of age, 88.4% white, 66.5% male and 28.6% with therapy-related (t)-MDS. Reflecting the disease severity at baseline, 82% of patients had >5% bone marrow blasts, 62.8% of patients were RBC transfusion dependent, 45.3% were platelet transfusion dependent and 57.6% had poor-risk IPSS cytogenetics. As stratified by the IPSS risk group, 69.6% of patients were in the INT-2 group while the rest (30.4%) were in the high-risk group. The median duration of follow-up was 15.7 months (95% CI: 14.6, 16.8). The median time from diagnosis to azanucleoside initiation was 0.95 months (95% CI: 0.86, 1.06). The majority of patients (90.3%) had an Eastern Cooperative Cancer Group performance status of 0 or 1. Not a single center accounted for more than 39% of patients.
Table 1.
Demographics and baseline characteristics of the study cohort (N = 632)
| Baseline characteristics (N = 632) | N (%) |
|---|---|
| Gender | |
| Male | 420 (66.5%) |
| Female | 212 (33.5%) |
| Race | |
| White | 559 (88.4%) |
| African America | 27 (4.3%) |
| Hispanic | 10(1.6%) |
| Other/unknown | 36 (5.7%) |
| Age at diagnosis | |
| <60 years | 87 (13.8%) |
| ≥60 years | 545 (86.2%) |
| Therapy-related MDS | |
| Yes | 181 (28.6%) |
| No | 451 (71.4%) |
| WHO classification | |
| RA or RCUD | 19 (3.0%) |
| RARS | 12 (1.9%) |
| RCMD | 74 (11.7%) |
| RAEB-1 | 167 (26.5%) |
| RAEB-2 | 349 (55.2%) |
| MDS-NOS | 10 (1.6%) |
| 5q-MDS | 1 (0.1%) |
| IPSS risk group | |
| Intermediate-2 | 440 (69.6%) |
| High | 192 (30.4%) |
| IPSS cytogenetic risk group | |
| Good | 140 (22.2%) |
| Intermediate | 128 (20.2%) |
| Poor | 364 (57.6%) |
| RBC transfusion dependence | |
| Yes | 397 (62.8%) |
| No | 235 (37.2%) |
| Platelet transfusion dependence | |
| Yes | 286 (45.3%) |
| No | 346 (54.7%) |
| ECOG PS | |
| 0 | 131 (20.7%) |
| 1 | 440 (69.6%) |
| 2 | 57 (9.0%) |
| 3 | 3 (0.5%) |
| 4 | 1 (0.2%) |
| Institution | |
| H. Lee Moffitt Cancer Center | 245 (38.8%) |
| Cleveland Clinic | 87 (13.8%) |
| Dana-Farber Cancer Institute | 39 (6.2%) |
| Weill Medical College of Cornell University | 66 (10.4%) |
| MD Anderson Cancer Center | 170 (26.9%) |
| Johns Hopkins University | 25 (4.0%) |
| Type of azanucleoside | |
| Azacitidine | 429 (67.9%) |
| Decitabine | 203 (32.1%) |
Abbreviations: ECOG PS, Eastern Cooperative Cancer Group performance status; IPSS, International Prognostic Scoring System; MDS, myeldysplastic syndrome; NOS, non-otherwise-specified; RA, refractory anemia; RAEB, refractory anemia with excess blasts; RARS, refractory anemia with ring sideroblasts; RBC, red blood cells; RCMD, refractory cytopenia(s) with multilineage dysplasia; RCUD, refractory cytopenia with unilineage dysplasia; WHO, World Health Organization; 5q-, deletion of chromosome 5q.
Of the 632 patients, 429 (67.9%) received azacitidine while 203 (32.1%) received decitabine. The median number of administered cycles of azanucleoside therapy was 5.0 (inter-quartile ranges: 3.0, 8.0), with 456 (72.2%) of the patients receiving 4 cycles of therapy or more. By the end of follow-up, 517 patients had died (81.8%), of whom 360 were IPSS INT-2 at diagnosis (57.0%) while 157 were IPSS high risk (24.8%). The median OS from diagnosis for the entire cohort was 17.0 months (95% CI: 15.8, 18.4, Figure 1a). The median OS from diagnosis was 18.0 months (95, % CI: 16.3, 19.8) for the INT-2 group compared with 16.1 months (95% CI: 13.9, 18.7) for the high-risk group (P = 0.029). When stratified by the type of azanucleoside received, there was no statistically significant difference in OS for azacitidine-treated patients (median OS, 16.4 months; 95% CI: 15.0, 17.9) compared with that of decitabine-treated patients (median OS, 18.8 months; 95% CI: 16.2, 21.8, P-value: 0.11, Figure 1b) in unadjusted analysis.
Figure 1.
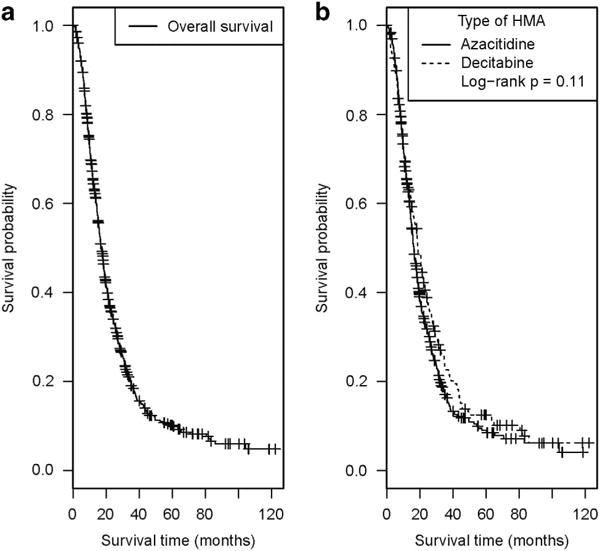
Unadjusted Kaplan – Meier estimates for overall survival from the time of diagnosis for the entire cohort (a) and as stratified by the type of azanucleoside received (b).
Overall Survival as stratified by the risk groups within the prognostic schemes
Table 2 summarizes the median OS and the 95% CI for each risk group within the five prognostic tools, while Figure 2 depicts the Kaplan–Meier estimates for OS of the cohort as stratified by risk groups within each prognostic tool. As noted, each of the risk schemes separated patients in groups with overall significantly different OS. The highest-risk group in each prognostic system identified subsets of patients with extremely poor prognosis despite azanucleoside therapy (median OS (95% CI); IPSS high: 16.1 months (13.9, 18.7); IPSS-R very high: 13.8 months (12.3, 15.4); FPSS high: 10.9 months (9.4, 12.7); MDAPSS high: 14.3 months (13.0, 15.7); and WPSS very high: 14.9 months (13.4, 16.5)).
Table 2.
Overall survival of the azanucleoside-treated cohort of patients with higher-risk myelodysplastic syndromes (n = 632) as stratified by the risk groups within each of the five prognostic systems
| IPSS
|
IPSS-R
|
FPSS
|
MDAPSS
|
WPSS
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk group | N (%) | Median OS in months (95% CI) | Risk group | N (%) | Median OS in months (95% CI) | Risk group | N (%) | Median OS in months (95% CI) | Risk group | N (%) | Median OS in months (95% CI) | Risk group | N (%) | Median OS in months (95% CI) |
| Low | 0 (0) | — | Very lowa | 0 (0) | — | Low | 40 (6.3) | 36.89 (23.50, 57.92); fmi = 0.179 | Lowa | 10 (1.6) | — | Very lowa | 1 (0.1) | — |
| INT-1 | 0 (0) | — | Low | 6 (0.9) | 56.71 (25.80, 124.67); fmi = 0.152 | INT | 490 (77.6) | 17.90 (16.48, 19.44); fmi = 0.115 | INT-1 | 54 (8.5) 31.95 | (19.26, 53.00); fmi = 0.161 | Lowa | 5 (0.8) | — |
| INT-2 | 440 (69.6) | 17.95 (16.30, 19.76); fmi = 0.0687 | INT | 68 (10.8) | 35.04 (23.66, 51.88); fmi = 0.175 | High | 102 (16.1) | 10.91 (9.40, 12.66); fmi = 0.142 | INT-2 | 184 (29.1) | 20.91 (17.84, 24.50); fmi = 0.039 | INT | 20 (3.2) | 19.66,(10.06, 38.40); fmi = 0.449 |
| High | 192 (30.4) | 16.13 (13.92, 18.70); fmi = 0.0273 | High | 213 (33.7) | 20.76 (18.21, 23.66); fmi = 0.080 | High | 384 (60.8) | 14.29 (12.98, 15.73); fmi = 0.026 | High | 323 (51.1) | 19.42 (17.33, 21.77); fmi = 0.235 | |||
| Very high | 345 (54.6) | 13.77 (12.30, 15.41); fmi = 0.060 | Very high | 273 (43.2) | 14.85 (13.37, 16.50); fmi = 0.156 | |||||||||
| N/Ab | 10 (1.6) | 13.96 (7.69, 25.34); fmi = 0.627 | ||||||||||||
Abbreviations: CI, confidence interval; fmi, fraction of missing information; FPSS, French Prognostic Scoring System; IPSS, International Prognostic Scoring System; IPSS-R, revised IPSS; MDAPSS, MD Anderson Prognostic Scoring System; N/A, non-applicable; OS, overall survival; WPSS, World Health Organization classification-based Prognostic Scoring System. The study by design enrolled only patients with IPSS INT-2 and high, and therefore there were no patients with IPSS low or INT-1 risk. The log-rank P-values for overall differences of OS among different groups within each of the prognostic schemes were all statistically significant (all o0.03).
Not estimable owing to small cell count.
Only patients with specific WHO classifications could have a WPSS score calculated. Patients who had an ineligible WHO classification were grouped into a ‘Not Applicable’ category, so they would not be excluded from the sample.
Figure 2.
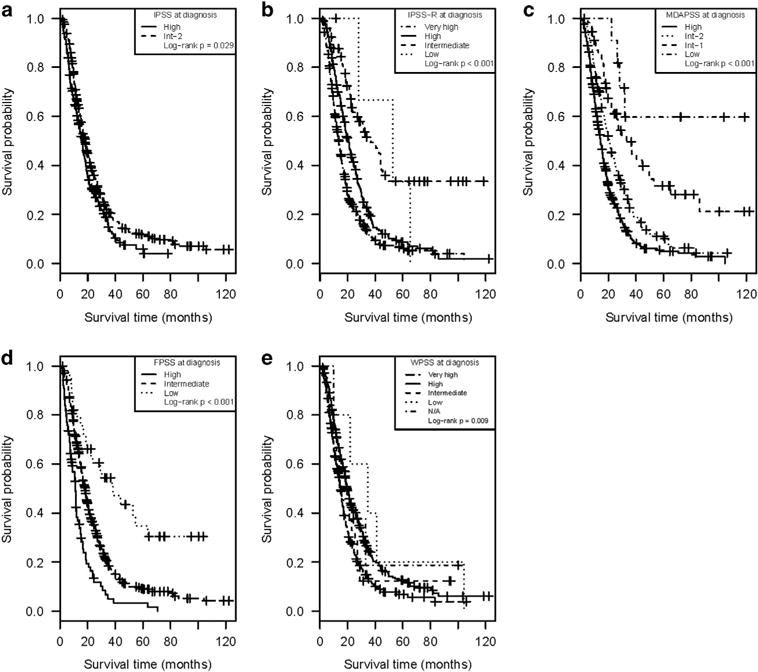
Kaplan–Meier estimates for overall survival from time of diagnosis as stratified by the risk groups within each of the five prognostic tools: (a) IPSS, (b) IPSS-R, (c) MDAPSS, (d) FPSS and (e) WPSS.
LFS as stratified by the risk groups within the prognostic schemes
The median LFS for the entire cohort was 13.0 months (95% CI: 11.9, 14.1). Table 3 summarizes the median LFS and the 95% CI for each risk group as defined using the five prognostic tools, while Figure 3 depicts the Kaplan–Meier LFS estimates for the cohort, by risk groups, for each prognostic tool. As can be observed, each of the five prognostic tools separated patients in risk groups with overall significantly different LFS (Table 3 and Figure 3).
Table 3.
Leukemia-free survival of the azanucleoside-treated cohort of patients with higher-risk myelodysplastic syndromes (n = 632) as stratified by the risk groups within each of the five prognostic systems
| IPSS
|
IPSS-R
|
FPSS
|
MDA PSS
|
WPSS
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk group | N (%) | Median LFS in months (95% CI) | Risk group | N (%) | Median LFS in months (95% CI) | Risk group | N (%) | Median LFS in months (95% CI) | Risk group | N (%) | Median LFS in months (95% CI) | Risk group | N (%) | Median LFS In months (95% CI) |
| Low | 0 (0) | — | Very lowa | 0 (0) | — | Low | 40 (6.3) | 24.3 (16.1, 36.8); fmi = 0.252 | Lowa | 10 (1.6) | — | Very lowa | 1 (0.1) | — |
| INT-1 | 0 (0) | — | Low | 6 (0.9) | 25.5 (5.8, 112.4); fmi = 0.595 | INT | 490 (77.6) | 13.3 (12.2, 14.6); fmi = 0.088 | INT-1 | 54 (8.5) | 19.1 (14.2, 25.8); fmi = 0.118 | Lowa | 5 (0.8) | — |
| INT-2 | 440 (69.6) | 13.7 (12.2, 15.3); fmi = 0.078 | INT | 68 (10.8) | 22.5 (16.9, 29.9); fmi = 0.007 | High | 102 (16.1) | 9.6 (7.8, 11.7); fmi = 0.176 | INT-2 | 184 (29.1) | 15.4 (12.3, 19.3); fmi = 0.123 | INT | 20 (3.2) | 16.9 (8.6, 33.1); fmi = 0.382 |
| High | 192 (30.4) | 10.9 (8.5, 13.8); fmi = 0.119 | High | 213 (33.7) | 14.4 (11.9, 17.4); fmi = 0.066 | High | 384 (60.8) | 11.8 (10.7, 13.1); fmi = 0.061 | High | 323 (51.1) | 14.7 (12.3, 17.6); fmi = 0.168 | |||
| Very high | 345 (54.6) | 10.7 (9.2, 12.4); fmi = 0.047 | Very high | 273 (43.2) | 11.2 (9.5, 13.4); fmi = 0.240 | |||||||||
| N/Ab | 10 (1.6) | 13.4 (7.0, 25.5); fmi = 0.534 | ||||||||||||
| P = 0.076 | P < 0.001 | P < 0.001 | P < 0.001 | P = 0.003 | ||||||||||
Abbreviations: CI, confidence interval; fmi, fraction of missing information; FPSS, French Prognostic Scoring System; IPSS, International Prognostic Scoring System; IPSS-R, revised IPSS; LFS, leukemia-free survival; MDAPSS, MD Anderson Prognostic Scoring System; N/A, non-applicable; WPSS, World Health Organization classification-based Prognostic Scoring System. The study by design enrolled only patients with IPSS INT-2 and high, and therefore there were no patients with IPSS low or INT-1 risk. The log-rank P-values for overall differences of LFS among different groups within each of the prognostic schemes were all statistically significant (except for IPSS where it was 0.076).
Not estimable owing to small cell count.
Only patients with specific WHO classifications could have a WPSS score calculated. Patients who had an ineligible WHO classification were grouped into a ‘Not Applicable’ category, so they would not be excluded from the sample.
Figure 3.
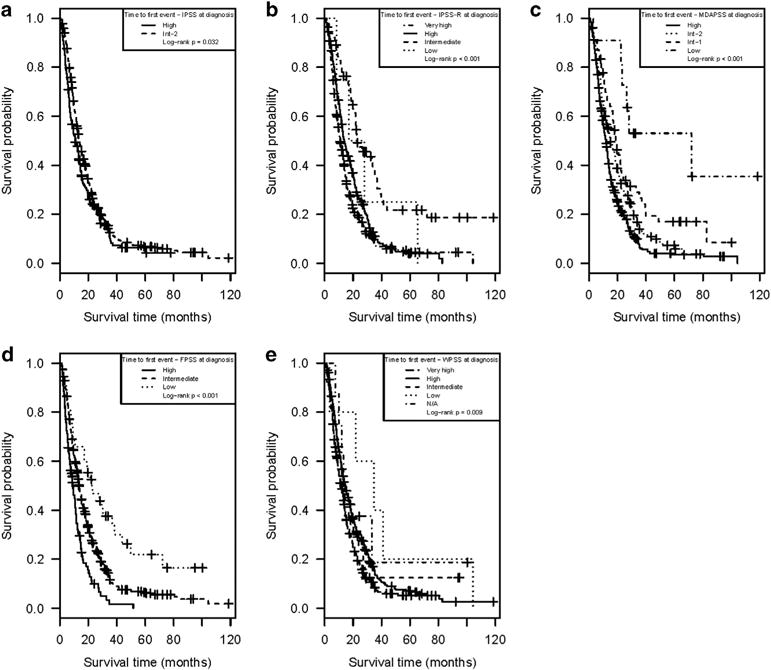
Kaplan – Meier estimates for leukemia-free survival from time of diagnosis as stratified by the risk groups within each of the 5 prognostic tools: (a) IPSS, (b) IPSS-R, (c) MDAPSS, (d) FPSS and (e) WPSS.
ORR as stratified by the risk groups within the prognostic schemes
The ORR for the entire cohort to azanucleoside therapy when best objective response was evaluated was 42.1% (CR, 21.0%; partial response, 9.8%; hematologic improvement, 11.3%), whereas in 57.9% no objective response was achieved (stable disease, 38.4%; PD, 19.5%). Table 4 summarizes the ORR by the risk groups for each of the five prognostic tools. As can be seen in Table 4, none of the five prognostic tools were able to predict the achievement of an objective response.
Table 4.
Overall response rate of the azanucleoside-treated cohort of patients with higher-risk myelodysplastic syndromes (n = 632) by the risk groups within each of the five prognostic systems
| IPSS
|
IPSS-R
|
FPSS
|
MDAPSS
|
WPSS
|
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk group | N (%) | ORR (%) | Risk group | N (%) | ORR (%) | Risk group | N (%) | ORR (%) | Risk group | N (%) | ORR (%) | Risk group | N (%) | ORR (95% CI) |
| Low | 0 (0) | — | Very lowa | 0 (0) | — | Low | 40 (6.3) | 57.5 | Lowa | 10 (1.6) | 70.0 | Very lowa | 1 (0.1) | 0.0 |
| INT-1 | 0 (0) | — | Low | 6 (0.9) | 50.0 | INT | 490 (77.6) | 41.4 | INT-1 | 54 (8.5) | 37.0 | Lowa | 5 (0.8) | 40.0 |
| INT-2 | 440 (69.6) | 39.8 | INT | 68 (10.8) | 48.5 | High | 102 (16.1) | 39.2 | INT-2 | 184 (29.1) | 43.5 | INT | 20 (3.2) | 40.0 |
| High | 192 (30.4) | 43.1 | High | 213 (33.7) | 37.6 | High | 384 (60.8) | 41.4 | High | 323 (51.1) | 42.4 | |||
| Very high | 345 (54.6) | 43.4 | Very high | 273 (43.2) | 42.5 | |||||||||
| N/Ab | 10 (1.6) | 30.0 | ||||||||||||
| P = 0.51 | P = 0.39 | P = 0.24 | P = 0.46 | P = 0.93 | ||||||||||
Abbreviations: FPSS, French Prognostic Scoring System; IPSS, International Prognostic Scoring System; IPSS-R, revised IPSS; MDAPSS, MD Anderson Prognostic Scoring System; N/A, non-applicable; ORR, overall response rate; WPSS, World Health Organization classification-based Prognostic Scoring System. The ORR was defined as the sum of patients with complete response, partial response and hematologic improvement rates as the best achieved response. The P-values come from the χ2 test and suggests that prognostic scoring group is not associated with ORR group, for any of the prognostic scoring systems.
Not estimable owing to small cell count.
Only patients with specific WHO classifications could have a WPSS score calculated. Patients who had an ineligible WHO classification were grouped into a ‘Not Applicable’ category, so they would not be excluded from the sample.
Comparison of the prognostic utility for survival among the five prognostic tools
The AICcs were used to compare the univariable Cox models containing prognostics scoring systems as covariates (lower is better). The AICcs for the OS models were 4138.4 (MDAPSS), 4185.6 (WPSS), 4196.1 (IPSS-R), 4155.9 (FPSS) and 4196.3 (IPSS; Table 5). To assess if the relative prognostic discrimination for the five prognostic tools was influenced by gender (male vs female) or type of azanucleoside used (azacitidine vs decitabine), we calculated the AIC scores for the five prognostic tools in each of these four subgroups (Table 5). The MDAPSS appeared to have the best relative prognostic discrimination (lowest AIC) in patients who have received either azanucleoside and among females, while the IPSS-R had the lowest AIC among males.
Table 5.
Calculated AICc scores (from OS models) for the different prognostic tools (lower is better) in the entire cohort and in gender subgroups (male and female) and based on the type of azanucleoside used (azacitidine and decitabine)
| Entire cohort | ||||
| MDAPSS | FPSS | WPSS | IPSS-R | IPSS |
| 4138.4 | 4155.9 | 4185.6 | 4196.1 | 4196.3 |
| Female subgroup | ||||
| MDAPSS | FPSS | IPSS-R | WPSS | IPSS |
| 1040.0 | 1051.8 | 1056.1 | 1066.9 | 1069.6 |
| Male subgroup | ||||
| IPSS-R | MDAPSS | FPSS | WPSS | IPSS |
| 2458.2 | 2462.1 | 2469 | 2484 | 2488.0 |
| Azacitidine subgroup | ||||
| MDAPSS | IPSS-R | FPSS | WPSS | IPSS |
| 2587.4 | 2591.5 | 2595.7 | 2616.9 | 2623.1 |
| Decitabine subgroup | ||||
| MDAPSS | FPSS | IPSS-R | WPSS | IPSS |
| 1058.5 | 1062.8 | 1067.5 | 1071.1 | 1074.0 |
Abbreviations: IPSS, International Prognostic Scoring System; IPSS-R, revised IPSS; MDAPSS, MD Anderson Prognostic Scoring System; WPSS, World Health Organization classification-based Prognostic Scoring System.
We also conducted an analysis of the subcohort of patients who have received four or more cycles of azanucleoside therapy (Supplementary Material). The AICcs for the OS models (lower is better) were 2730.1 (MDAPSS), 2733.9 (IPSS-R), 2742 (FPSS), 2763.9 (WPSS) and 2775.4 (IPSS).
DISCUSSION AND CONCLUSIONS
MDS represent a group of hematopoietic stem cell malignancies that are biologically and clinically heterogonous.3–5 Consequently, accurate risk stratification is a fundamental component of the recommended management strategy.6,30 This study is the first to comprehensively compare the prognostic utility of the most commonly used MDS prognostic tools in a large cohort of well-defined HR-MDS patients treated with azanucleosides.
The median age of the patients in our cohort, high proportion of those 60 years or older, and the gender and race distributions closely mirror large population-based surveillance, epidemiology, and end results-Medicare linked database analyses of MDS,31–34 indicating our sample closely reflects the MDS population in the US. In contrast, the relatively higher prevalence of t-MDS (28.6%) than previously reported, probably reflects a tertiary center referral pattern, a more comprehensive ascertainment of prior chemotherapy or radiation exposure, or possibly the stronger association of t-MDS with adverse prognostic that features the characteristic of HR-MDS.
Several conclusions can be drawn from our study. First, and reassuringly, all the prognostic tools functioned well to separate azanucleoside-treated patients with HR-MDS into risk groups with overall significantly different survivals. Second, none of the prognostic tools predicted the probability of achieving an objective response with azanucleosides, further emphasizing the complex relationship between response and survival benefit after azanucleoside therapy and highlighting the limitations of relying on response as the sole marker of clinical benefit in MDS trials using azanucleosides.35,36
Third, the median OS from diagnosis of the azacitidine-treated patients (16.4 months) was significantly shorter than the 24.5 months median OS observed in the landmark randomized AZA-001 trial of azacitidine in HR-MDS.13 This is consistent with the survival outcomes noted in surveillance, epidemiology, and end results-Medicare population-based analyses in the USA and non-trial data from registries and compassionate-use programs in Europe.27,37–39 For example, a report from the Spanish Compassionate use registry of 200 azacitidine-treated patients with all IPSS risk groups estimated an overall median OS of 16.5 months (95% CI, 12.4 – 19.1 months).38 A report from the Dutch compassionate patient-named program of 90 azacitidine-treated patients estimated a median OS of 13 months.39 The Groupe Francophone des Myelodysplasies reported a median OS of 13.5 months for 282 patients with HR-MDS treated with azacitidine in the French compassionate, patient-named program.27 Although the reasons behind the discrepancy in survival outcomes between the randomized trial and the real-life setting are unknown, the unselected nature of patients in real-life analyses compared the highly selected individuals enrolled in randomized trials could account, at least partly, for this observation. It should be noted here that, although we did not observe a statistically significant differences in the OS of azacitidine-treated compared with decitabine-treated patients, this was an unadjusted analysis and there could be significant differences in the baseline characteristics of the patients, the severity of MDS, and selection bias in the choice of the azanucleoside that could have influenced these findings. Nonetheless, when counseling patients with HR-MDS regarding expected survival with azacitidine therapy, a median survival of 16 – 17 months might be a more realistic estimate than the AZA-001 clinical trial estimate of 24.5 months.
Fourth, in accordance with prior published reports we observed an ORR of 40 – 50% across the different risk groups. It has been well documented that many patients with HR-MDS do not derive clinical benefit from azanucleosides therapy. This observation, combined with the need of a therapeutic trial of 4 – 6 months (with all the associated potential toxicity, cost and wasted time) before achievement of a clinical response is ruled out, has generated significant interest in development of clinical prediction tools or discovery of biomarkers that can predict clinical benefit from azanucleosides.40 As none of the commonly used prognostic tools were developed to predict the response of MDS patients to any specific treatment modality, the FPSS was developed purposefully to stratify outcomes in azacitidine-treated patients with HR-MDS and was subsequently validated in external cohorts of North American and European patients.26–28,39,41
In developing the FPSS, the French group performed an analysis of 282 such patients enrolled in a compassionate, patient-named program who received a median of six cycles of azacitidine therapy.27 Performance status ≥ 2, intermediate- and poor-risk cytogenetics, presence of peripheral blood blasts and RBC transfusion dependency (defined as ≥ 4 units/8 weeks) independently predicted for worse OS. These four easily obtainable parameters were used to develop the FPSS, which separated patients into three risk groups with significantly different median OS at 32, 15 and 6 months, respectively.27,28 Despite this, a recent analysis of the randomized phase 2 E1905 study has shown that while the FPSS separated azacitidine-treated patients into 3 different risk groups with significantly different median OS, the FPSS did not improve on the prognostic utility of the IPSS-R among azacitidine-treated MDS patients.26 In our current analysis, the highest-risk group in the FPSS had the worst median OS among the highest-risk groups of the five different prognostic tools (11 months). The MDAPSS had a lower AICc than the FPSS indicating improved prognostic performance in this setting, although the MDAPSS was not specifically developed for use in azanucleosides-treated patients.
Despite recent promising advances in identifying molecular markers associated with a higher likelihood of predicting clinical benefit from azanucleosides, such as TET2 and DNMT3A mutations, no molecular or clinical pattern identifies patients with such a low probability of achieving clinical benefit from azanucleosides that it would justify withholding these drugs.42,43 Nonetheless, we found that the median OS of azanucleoside-treated patients in the highest-risk group in each of the IPSS-R, WPSS, FPSS and the MDAPSS was only 11 – 16 months from time diagnosis. These observations highlight that every patient with HR-MDS, especially those belonging to the highest-risk groups in the prognostic tools, should be strongly considered for allogeneic hematopoietic stem cell transplantation or evaluation for clinical trial enrollment, and not simply be given azanucleosides without awareness or consideration of these expected poor outcomes. There remains a significant clinical and research need for clinical tools or biomarkers that can identify patients with low probability of benefit (or survival) and/or high probability of azanucleosides treatment failure, which would be useful to select such patients for alternative aggressive approaches or experimental therapies such as azanucleosides-based combinations or clinical trials.42,44 This approach would spare such patients the possible side effects, costs, and wasted 4 – 6 months of ineffective azanucleoside therapy before lack of benefit is documented.25
Our study has important limitations. As with any large, retrospective analysis, our results are subject to bias resulting from missing data and tertiary center referral bias. For example, the high proportion of t-MDS might have affected the relative performance of some of the prognostic tools more than others. In addition, the details of routes and doses of azanucleoside therapy were not collected, though previous studies have shown little difference in response rates with variation in these parameters.20,38,45 Compared with this analysis, other studies have calculated survival from date of initiation of the azanucleoside rather than from date of diagnosis. However, since this analysis was limited to patients who presented with HR-MDS, most of whom were symptomatic and were started on azanucleoside therapy soon after diagnosis. Indeed, the median time from diagnosis to azanucleoside initiation for the entire cohort was only 0.95 months (95% CI: 0.86, 1.06). Finally, the relative prognostic utility might be different depending on the type of the azanucleoside used and might require separate comparisons of the prognostic tools in these subsets of patients (that is, azacitidine-treated patients separately from decitabine-treated ones). Nonetheless, subgroup analyses did not identify major differences in performance of these instruments depending on therapy type, though. This factor might have affected the performance of the FPSS more than the other tools as the FPSS was specifically developed for azacitidine-treated patients.
In conclusion, in this multicenter cohort of azanucleoside-treated patients with HR-MDS, the commonly used prognostic tools maintained prognostic utility for OS and LFS. The MDAPSS and FPSS appeared superior to the other tools for OS prediction. Patients in the highest-risk groups identified by each prognostic tool had a median OS of 11 – 16 months with azanucleoside therapy and should be strongly considered for up-front allogeneic hematopoietic stem cell transplantation or experimental approaches.
Supplementary Material
Acknowledgments
We would like to thank the patients who contributed to this large database and their families, as well the research coordinators in the six sites who worked to construct the database. The authors would also like to thank the Evans Foundation and the Aplastic Anemia and Myelodysplastic Syndromes Foundation for supporting the research activities of the MDS Clinical Research Consortium. This research was presented in part at the American Society of Hematology 56th Annual meeting in San Francisco, CA, USA in December 2014. AMZ received funding through MDS Clinical Research Consortium and the American Society of Oncology Young Investigator Award.
Footnotes
CONFLICT OF INTEREST
The authors declare no conflict of interest.
AUTHOR CONTRIBUTIONS
AMZ conceived the study concept, analyzed the data and wrote the manuscript. MAS, GG-M, DPS and RK contributed data to the study, analyzed the data and revised the manuscript. KZ and JB analyzed the data, performed the statistical analyses and contributed to manuscript writing. NAA and CZ collected data and reviewed the manuscript. GR, AZ, AN, EJ, HK, SDG, JPM and AL contributed data to the study and reviewed the manuscript. All the authors reviewed the manuscript and approved the final version.
References
- 1.Tefferi A, Vardiman JW. Myelodysplastic syndromes. N Engl J Med. 2009;361:1872–1885. doi: 10.1056/NEJMra0902908. [DOI] [PubMed] [Google Scholar]
- 2.Vardiman JW, Thiele J, Arber DA, Brunning RD, Borowitz MJ, Porwit A, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114:937–951. doi: 10.1182/blood-2009-03-209262. [DOI] [PubMed] [Google Scholar]
- 3.Raza A, Galili N. The genetic basis of phenotypic heterogeneity in myelodysplastic syndromes. Nat Rev Cancer. 2012;12:849–859. doi: 10.1038/nrc3321. [DOI] [PubMed] [Google Scholar]
- 4.Malcovati L, Papaemmanuil E, Ambaglio I, Elena C, Galli A, Della Porta MG, et al. Driver somatic mutations identify distinct disease entities within myeloid neoplasms with myelodysplasia. Blood. 2014;124:1513–1521. doi: 10.1182/blood-2014-03-560227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cazzola M, Della Porta MG, Malcovati L. The genetic basis of myelodysplasia and its clinical relevance. Blood. 2013;122:4021–4034. doi: 10.1182/blood-2013-09-381665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zeidan AM, Linhares Y, Gore SD. Current therapy of myelodysplastic syndromes. Blood Rev. 2013;27:243–259. doi: 10.1016/j.blre.2013.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Greenberg P, Cox C, LeBeau MM, Fenaux P, Morel P, Sanz G, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–2088. [PubMed] [Google Scholar]
- 8.Zeidan AM, Gore SD, Padron E, Komrokji RS. Current state of prognostication and risk stratification in myelodysplastic syndromes. Curr Opin Hematol. 2015;22:146–154. doi: 10.1097/MOH.0000000000000110. [DOI] [PubMed] [Google Scholar]
- 9.Malcovati L, Germing U, Kuendgen A, Della Porta MG, Pascutto C, Invernizzi R, et al. Time-dependent prognostic scoring system for predicting survival and leukemic evolution in myelodysplastic syndromes. J Clin Oncol. 2007;25:3503–3510. doi: 10.1200/JCO.2006.08.5696. [DOI] [PubMed] [Google Scholar]
- 10.Kantarjian H, O’Brien S, Ravandi F, Cortes J, Shan J, Bennett JM, et al. Proposal for a new risk model in myelodysplastic syndrome that accounts for events not considered in the original International Prognostic Scoring System. Cancer. 2008;113:1351–1361. doi: 10.1002/cncr.23697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Greenberg PL, Tuechler H, Schanz J, Sanz G, Garcia-Manero G, Sole F, et al. Revised international prognostic scoring system for myelodysplastic syndromes. Blood. 2012;120:2454–2465. doi: 10.1182/blood-2012-03-420489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zeidan AM, Komrokji RS. There’s risk, and then there’s risk: the latest clinical prognostic risk stratification models in myelodysplastic syndromes. Curr Hematol Malig Rep. 2013;8:351–360. doi: 10.1007/s11899-013-0172-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fenaux P, Mufti GJ, Hellstrom-Lindberg E, Santini V, Finelli C, Giagounidis A, et al. Efficacy of azacitidine compared with that of conventional care regimens in the treatment of higher-risk myelodysplastic syndromes: a randomised, open-label, phase III study. Lancet Oncol. 2009;10:223–232. doi: 10.1016/S1470-2045(09)70003-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Silverman LR, Demakos EP, Peterson BL, Kornblith AB, Holland JC, Odchimar-Reissig R, et al. Randomized controlled trial of azacitidine in patients with the myelodysplastic syndrome: a study of the cancer and leukemia group B. J Clin Oncol. 2002;20:2429–2440. doi: 10.1200/JCO.2002.04.117. [DOI] [PubMed] [Google Scholar]
- 15.Kornblith AB, Herndon JE, 2nd, Silverman LR, Demakos EP, Odchimar-Reissig R, Holland JF, et al. Impact of azacytidine on the quality of life of patients with myelodysplastic syndrome treated in a randomized phase III trial: a Cancer and Leukemia Group B study. J Clin Oncol. 2002;20:2441–2452. doi: 10.1200/JCO.2002.04.044. [DOI] [PubMed] [Google Scholar]
- 16.Silverman LR, McKenzie DR, Peterson BL, Holland JF, Backstrom JT, Beach CL, et al. Further analysis of trials with azacitidine in patients with myelodysplastic syndrome: studies 8421, 8921, and 9221 by the Cancer and Leukemia Group B. J Clin Oncol. 2006;24:3895–3903. doi: 10.1200/JCO.2005.05.4346. [DOI] [PubMed] [Google Scholar]
- 17.Kantarjian H, Oki Y, Garcia-Manero G, Huang X, O’Brien S, Cortes J, et al. Results of a randomized study of 3 schedules of low-dose decitabine in higher-risk myelodysplastic syndrome and chronic myelomonocytic leukemia. Blood. 2007;109:52–57. doi: 10.1182/blood-2006-05-021162. [DOI] [PubMed] [Google Scholar]
- 18.Lubbert M, Suciu S, Baila L, Ruter BH, Platzbecker U, Giagounidis A, et al. Low-dose decitabine versus best supportive care in elderly patients with intermediate- or high-risk myelodysplastic syndrome (MDS) ineligible for intensive chemotherapy: final results of the randomized phase III study of the European Organisation for Research and Treatment of Cancer Leukemia Group and the German MDS Study Group. J Clin Oncol. 2011;29:1987–1996. doi: 10.1200/JCO.2010.30.9245. [DOI] [PubMed] [Google Scholar]
- 19.Steensma DP, Baer MR, Slack JL, Buckstein R, Godley LA, Garcia-Manero G, et al. Multicenter study of decitabine administered daily for 5 days every 4 weeks to adults with myelodysplastic syndromes: the alternative dosing for outpatient treatment (ADOPT) trial. J Clin Oncol. 2009;27:3842–3848. doi: 10.1200/JCO.2008.19.6550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lyons RM, Cosgriff TM, Modi SS, Gersh RH, Hainsworth JD, Cohn AL, et al. Hematologic response to three alternative dosing schedules of azacitidine in patients with myelodysplastic syndromes. J Clin Oncol. 2009;27:1850–1856. doi: 10.1200/JCO.2008.17.1058. [DOI] [PubMed] [Google Scholar]
- 21.Zeidan AM, Kharfan-Dabaja MA, Komrokji RS. Beyond hypomethylating agents failure in patients with myelodysplastic syndromes. Curr Opin Hematol. 2014;21:123–130. doi: 10.1097/MOH.0000000000000016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Prebet T, Gore SD, Esterni B, Gardin C, Itzykson R, Thepot S, et al. Outcome of high-risk myelodysplastic syndrome after azacitidine treatment failure. J Clin Oncol. 2011;29:3322–3327. doi: 10.1200/JCO.2011.35.8135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jabbour E, Garcia-Manero G, Batty N, Shan J, O’Brien S, Cortes J, et al. Outcome of patients with myelodysplastic syndrome after failure of decitabine therapy. Cancer. 2010;116:3830–3834. doi: 10.1002/cncr.25247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Steensma DP. Can hypomethylating agents provide a platform for curative therapy in myelodysplastic syndromes? Best Pract Res Clin Haematol. 2012;25:443–451. doi: 10.1016/j.beha.2012.10.007. [DOI] [PubMed] [Google Scholar]
- 25.Zeidan AM, Lee JW, Prebet T, Greenberg P, Sun Z, Juckett M, et al. Platelet count doubling after the first cycle of azacitidine therapy predicts eventual response and survival in patients with myelodysplastic syndromes and oligoblastic acute myeloid leukaemia but does not add to prognostic utility of the revised IPSS. Br J Haematol. 2014;167:62–68. doi: 10.1111/bjh.13008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zeidan AM, Lee JW, Prebet T, Greenberg P, Sun Z, Juckett M, et al. Comparison of the prognostic utility of the revised International Prognostic Scoring System and the French Prognostic Scoring System in azacitidine-treated patients with myelodysplastic syndromes. Br J Haematol. 2014;166:352–359. doi: 10.1111/bjh.12884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Itzykson R, Thepot S, Quesnel B, Dreyfus F, Beyne-Rauzy O, Turlure P, et al. Prognostic factors for response and overall survival in 282 patients with higher-risk myelodysplastic syndromes treated with azacitidine. Blood. 2011;117:403–411. doi: 10.1182/blood-2010-06-289280. [DOI] [PubMed] [Google Scholar]
- 28.Itzykson R, Thepot S, Quesnel B, Dreyfus F, Recher C, Wattel E, et al. Long-term outcome of higher-risk MDS patients treated with azacitidine: an update of the GFM compassionate program cohort. Blood. 2012;119:6172–6173. doi: 10.1182/blood-2012-04-422204. [DOI] [PubMed] [Google Scholar]
- 29.Cheson BD, Greenberg PL, Bennett JM, Lowenberg B, Wijermans PW, Nimer SD, et al. Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia. Blood. 2006;108:419–425. doi: 10.1182/blood-2005-10-4149. [DOI] [PubMed] [Google Scholar]
- 30.Faltas B, Zeidan A, Gergis U. Myelodysplastic syndromes: toward a risk-adapted treatment approach. Expert Rev Hematol. 2013;6:611–624. doi: 10.1586/17474086.2013.840997. [DOI] [PubMed] [Google Scholar]
- 31.Ma X, Does M, Raza A, Mayne ST. Myelodysplastic syndromes: incidence and survival in the United States. Cancer. 2007;109:1536–1542. doi: 10.1002/cncr.22570. [DOI] [PubMed] [Google Scholar]
- 32.Ma X. Epidemiology of myelodysplastic syndromes. Am J Med. 2012;125:S2–S5. doi: 10.1016/j.amjmed.2012.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zeidan AM, Gore SD, McNally DL, Baer MR, Hendrick F, Mahmoud D, et al. Lenalidomide performance in the real world: patterns of use and effectiveness in a Medicare population with myelodysplastic syndromes. Cancer. 2013;119:3870–3878. doi: 10.1002/cncr.28298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sekeres MA. Epidemiology, natural history, and practice patterns of patients with myelodysplastic syndromes in 2010. J Natl Compr Cancer Netw. 2011;9:57–63. doi: 10.6004/jnccn.2011.0006. [DOI] [PubMed] [Google Scholar]
- 35.Gore SD, Fenaux P, Santini V, Bennett JM, Silverman LR, Seymour JF, et al. A multivariate analysis of the relationship between response and survival among patients with higher-risk myelodysplastic syndromes treated within azacitidine or conventional care regimens in the randomized AZA-001 trial. Haematologica. 2013;98:1067–1072. doi: 10.3324/haematol.2012.074831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Itzykson R, Fenaux P. Predicting the outcome of patients with higher-risk myelodysplastic syndrome treated with hypomethylating agents. Leuk Lymphoma. 2012;53:760–762. doi: 10.3109/10428194.2011.651618. [DOI] [PubMed] [Google Scholar]
- 37.Wang R, Gross CP, Frick K, Xu X, Long J, Raza A, et al. The impact of hypomethylating agents on the cost of care and survival of elderly patients with myelodysplastic syndromes. Leuk Res. 2012;36:1370–1375. doi: 10.1016/j.leukres.2012.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Garcia-Delgado R, de Miguel D, Bailen A, Gonzalez JR, Bargay J, Falantes JF, et al. Effectiveness and safety of different azacitidine dosage regimens in patients with myelodysplastic syndromes or acute myeloid leukemia. Leuk Res. 2014;38:744–750. doi: 10.1016/j.leukres.2014.03.004. [DOI] [PubMed] [Google Scholar]
- 39.van der Helm LH, Alhan C, Wijermans PW, van Marwijk Kooy M, Schaafsma R, Biemond BJ, et al. Platelet doubling after the first azacitidine cycle is a promising predictor for response in myelodysplastic syndromes (MDS), chronic myelomonocytic leukaemia (CMML) and acute myeloid leukaemia (AML) patients in the Dutch azacitidine compassionate named patient programme. Br J Haematol. 2011;155:599–606. doi: 10.1111/j.1365-2141.2011.08893.x. [DOI] [PubMed] [Google Scholar]
- 40.Lee EJ, Zeidan AM. Genome sequencing in myelodysplastic syndromes: can molecular mutations predict benefit from hypomethylating agent therapy? Expert Rev Hematol. 2015;8:155–158. doi: 10.1586/17474086.2015.1016905. [DOI] [PubMed] [Google Scholar]
- 41.Breccia M, Loglisci G, Cannella L, Finsinger P, Mancini M, Serrao A, et al. Application of French prognostic score to patients with International Prognostic Scoring System intermediate-2 or high risk myelodysplastic syndromes treated with 5-azacitidine is able to predict overall survival and rate of response. Leuk Lymphoma. 2012;53:985–986. doi: 10.3109/10428194.2011.643408. [DOI] [PubMed] [Google Scholar]
- 42.Bejar R, Lord A, Stevenson K, Bar-Natan M, Perez-Ladaga A, Zaneveld J, et al. TET2 mutations predict response to hypomethylating agents in myelodysplastic syndrome patients. Blood. 2014;124:2705–2712. doi: 10.1182/blood-2014-06-582809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Traina F, Visconte V, Elson P, Tabarroki A, Jankowska AM, Hasrouni E, et al. Impact of molecular mutations on treatment response to DNMT inhibitors in myelodysplasia and related neoplasms. Leukemia. 2014;28:78–87. doi: 10.1038/leu.2013.269. [DOI] [PubMed] [Google Scholar]
- 44.Bejar R. Clinical and genetic predictors of prognosis in myelodysplastic syndromes. Haematologica. 2014;99:956–964. doi: 10.3324/haematol.2013.085217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Grinblatt DL, Sekeres MA, Komrokji RS, Swern AS, Sullivan KA, Narang M. Patients with myelodysplastic syndromes treated with azacitidine in clinical practice: the AVIDA registry. Leuk Lymphoma. 2015;56:887–895. doi: 10.3109/10428194.2014.935366. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


