Abstract
Eravacycline is a novel broad-spectrum fluorocycline with potent Gram-negative activity, including for multidrug-resistant strains. Among 472 Escherichia coli clinical isolates from 24 Veterans Affairs medical centers (in 2011), divided equally as susceptible versus resistant to fluoroquinolones, broth microdilution eravacycline MICs were distributed unimodally, ranging from 0.03 to 1.0 μg/ml (MIC50 of 0.125 μg/ml, MIC90 of 0.25 μg/ml). Eravacycline MICs were ∼2-fold higher among fluoroquinolone-resistant, gentamicin-resistant, multidrug-resistant, and sequence type 131 (ST131) isolates (P < 0.01 for each comparison).
TEXT
Sequence type 131 (ST131) of Escherichia coli causes most antimicrobial-resistant E. coli infections in the United States today, especially among veterans (1, 2). It is closely associated with fluoroquinolone resistance, thanks to its H30 subclone (1–5), and, with extended-spectrum beta-lactamase (ESBL) production, thanks to the H30 subclone's H30Rx subclone (6–8).
Eravacycline is a novel, broad-spectrum fluorocycline with potent Gram-negative activity, including against multidrug-resistant (MDR) strains (9–15). In phase 2 and 3 clinical trials for complicated intra-abdominal infections, in which E. coli predominated, eravacycline was as effective as ertapenem (11, 16). Eravacycline activity against ST131 is undefined (9). Here, for E. coli clinical isolates from Veterans Affairs medical centers (VAMCs) across the United States (1), we determined MICs to eravacycline and 8 comparator agents, and we compared susceptibility results with ST131, H30, and H30Rx genotype.
Isolates.
Sets of ∼20 each of consecutive E. coli clinical isolates were collected in 2011 from 24 widely distributed Veterans Affairs medical centers, as ∼10 each of fluoroquinolone-susceptible and -resistant isolates per center (1). ST131 and its H30 and H30Rx subclones were detected by lineage-specific PCR assays (4, 8).
Susceptibility testing.
Broth microdilution MICs were determined for eravacycline, minocycline, tigecycline, levofloxacin, piperacillin-tazobactam, gentamicin, ceftazidime, ceftriaxone, and meropenem using 2015 Clinical and Laboratory Standards Institute (CLSI)-specified procedures, reference strains, and breakpoints (17, 18). The median of 2 to 5 total MIC determinations was used. Categorical susceptibility to noneravacycline comparator agents was defined per CLSI criteria (18). The new susceptibility data, plus existing data for other agents (1), were used to classify isolates as MDR based on nonsusceptibility to ≥1 member each of ≥3 drug classes. Extended-spectrum beta-lactamase (ESBL) production was determined by a standard double-disk method, using cefotaxime and ceftazidime, with and without clavulanate (17, 18).
Statistical analysis.
Comparisons of proportions and MIC values were tested using a chi-square test and the Mann-Whitney U test, respectively.
Eravacycline and comparator agent MICs versus fluoroquinolone phenotype.
Among the 472 study isolates, eravacycline MICs were distributed unimodally from 0.03 to 1.0 mg/liter (MIC50 of 0.125 mg/liter, MIC90 of 0.25 mg/liter) (Fig. 1). They were statistically significantly but only 2-fold higher among fluoroquinolone-resistant than fluoroquinolone-susceptible isolates (Table 1; Fig. 1). Comparator agent MICs followed the same pattern, being significantly higher, although only slightly so, among fluoroquinolone-resistant than fluoroquinolone-susceptible isolates (Table 1). Categorical susceptibility exhibited a similar trend, being less common among fluoroquinolone-resistant than fluoroquinolone-susceptible isolates.
FIG 1.
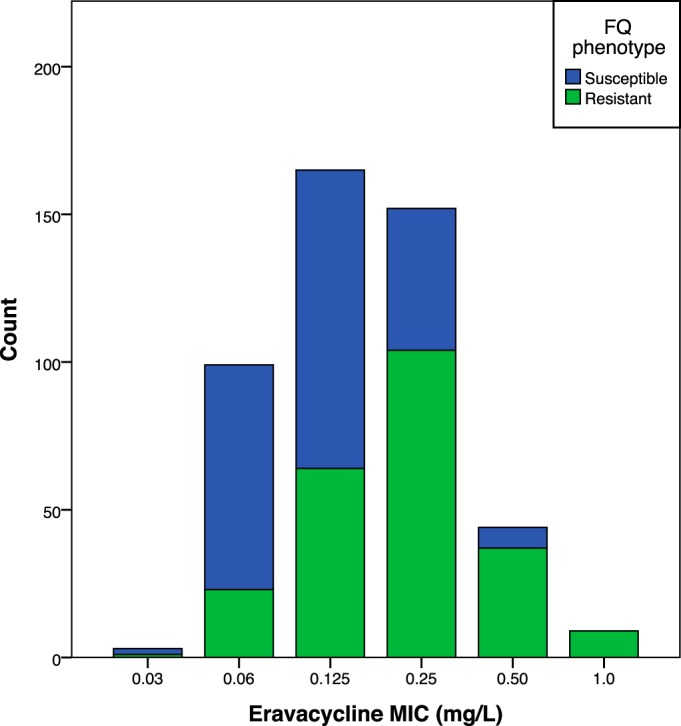
Distribution of eravacycline MICs among 472 Escherichia coli isolates from U.S. veterans, overall and in relation to fluoroquinolone (FQ) phenotype. Isolates were considered FQ resistant if the ciprofloxacin MIC was ≥2 mg/liter.
TABLE 1.
MICs for eravacycline and 8 other antimicrobial agents among 472 Escherichia coli isolates from veterans (in 2011)
| Drug | MIC (mg/liter) of: |
No. (column %) susceptible of: |
||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| FQ-Sa isolates (n = 234) |
FQ-Rb isolates (n = 238) |
P valuec | FQ-S isolatesa (n = 234) | FQ-R isolatesb (n = 238) | P valuec | |||||
| Range | MIC50 | MIC90 | Range | MIC50 | MIC90 | |||||
| Eravacycline | 0.03 to 0.50 | 0.13 | 0.25 | 0.03 to 1.0 | 0.25 | 0.50 | <0.001 | Undefined | Undefined | n.a.d |
| Minocycline | 0.25 to 256 | 1 | 8 | 0.50 to 256 | 2 | 8 | <0.001 | 204 (87) | 199 (84) | |
| Piperacillin-tazobactame | ≤1 to > 128 | 2 | 8 | ≤1 to > 128 | 2 | 32 | <0.001 | 222 (95) | 204 (86) | 0.001 |
| Gentamicin | 0.06 to > 16 | 2 | 2 | 0.25 to > 16 | 2 | > 16 | <0.001 | 227 (97) | 157 (66) | <0.001 |
| Levofloxacin | ≤0.06 to 1 | 0.32 | 0.25 | 4 to >8 | >8 | >8 | <0.001 | 234 (100) | 0 (0) | <0.001 |
| Ceftazidime | ≤0.13 to > 16 | 0.50 | 1 | ≤0.13 to > 16 | 1 | 2 | <0.001 | 231 (99) | 219 (92) | <0.001 |
| Ceftriaxone | ≤0.03 to >4 | 0.06 | 0.25 | ≤0.03 to >4 | 0.13 | 1 | <0.001 | 230 (98) | 215 (90) | <0.001 |
| Meropenem | ≤0.03 to 0.13 | <0.03 | <0.03 | ≤0.03 to 2 | ≤0.03 | ≤0.03 | 0.02 | 234 (100) | 237 (99.6) | |
| Tigecycline | 0.01 to 1 | 0.50 | 0.50 | 0.25 to 4 | 0.50 | 1 | <0.001 | 234 (100) | 237 (99.6) | |
FQ-S, fluoroquinolone susceptible.
FQ-R, fluoroquinolone resistant.
P values (by a two-tailed Mann-Whitney U test [MIC data] or by Fisher's exact test [% susceptible]) for comparisons of fluoroquinolone-susceptible versus fluoroquinolone-resistant isolates are shown where the P value is <0.05.
n.a., not applicable (no data to compare).
MIC data shown are for piperacillin. The tazobactam concentration was 4 mg/liter throughout.
Eravacycline MICs versus resistance to comparator agents.
Eravacycline MICs varied significantly in relation to resistance to comparator agents only for fluoroquinolones (Table 1) and gentamicin (MIC50 for gentamicin-susceptible versus gentamicin-resistant isolates, 0.125 mg/liter versus 0.25 mg/liter; P = 0.003). The difference for gentamicin disappeared with stratification by fluoroquinolone phenotype (not shown), consistent with it reflecting the association of gentamicin resistance and fluoroquinolone resistance (Table 1).
Eravacycline MICs versus MDR and ESBL status.
By class, the prevalence of resistance declined as follows: penicillins (57%), fluoroquinolones (50%), folate inhibitors (31%), cephalosporins (22%), aminoglycosides (19%), tetracyclines (15%), nitrofurans (7%), carbapenems (0.4%), and glycylcyclines (0.2%). Overall, 41% of isolates qualified as MDR. Eravacycline MICs were approximately 2-fold higher among MDR isolates (MIC50 of 0.25 mg/liter; range, 0.06 to 1.0 mg/liter) than non-MDR isolates (MIC50 of 0.125 mg/liter; range, 0.03 to 1.0 mg/liter) (P < 0.001).
Of the 28 ceftriaxone and/or ceftazidime-resistant isolates, 19 (68%) were ESBL producers. Eravacycline MICs were similar among these 19 isolates (MIC50 of 0.125 mg/liter, MIC90 of 0.50 mg/liter) and the remaining nine cephalosporin-resistant isolates (MIC50 of 0.25 mg/liter, MIC90 of 0.50 mg/liter).
ST131.
ST131 and its H30 subclone accounted for over three-fourths of fluoroquinolone-resistant isolates but for only 2% to 6% of fluoroquinolone-susceptible isolates; the H30Rx subclone was restricted to and accounted for 15% of fluoroquinolone-resistant isolates (Table 2). These lineages were associated with slightly (by approximately 2-fold) but statistically significantly higher eravacycline MICs than other isolates (Table 3). These associations weakened or disappeared when the comparisons were stratified by fluoroquinolone phenotype (Table 4, left-hand P value column), suggesting that they resulted mostly from the association of ST131 with fluoroquinolone resistance (Table 2), which in turn was associated with higher eravacycline MICs (Table 1). Accordingly, associations of the eravacycline MIC with fluoroquinolone resistance persisted despite stratification for ST131, H30, and H30Rx status (Table 4, right-hand P value column).
TABLE 2.
Prevalence of ST131 and its clonal subsets in relation to fluoroquinolone resistance among 472 Escherichia coli isolates from U.S. veterans
| Clonal subset | Prevalence of subset (no. [column %]) in: |
P valuea | |
|---|---|---|---|
| Fluoroquinolone-susceptible isolates (n = 234) | Fluoroquinolone-resistant isolates (n = 238) | ||
| ST131 (n = 201) | 14 (6) | 187 (79) | <0.001 |
| H30 (n = 185) | 5 (2) | 180 (76) | <0.001 |
| H30Rx (n = 35) | 0 (0) | 35 (15) | <0.001 |
P values are by Fisher's exact test (two-tailed).
TABLE 3.
Association of eravacycline MIC with ST131 subsets
| ST131 subset | Specific group | No. of isolates | MIC (mg/liter) data |
P valuea for ST131 subset vs others | ||
|---|---|---|---|---|---|---|
| Range | MIC50 | MIC90 | ||||
| ST131 | ST131 | 201 | 0.03–1.0 | 0.25 | 0.50 | <0.001 |
| Others | 271 | 0.03–1.0 | 0.13 | 0.25 | ||
| H30 | H30 | 185 | 0.03–1.0 | 0.25 | 0.50 | <0.001 |
| Others | 287 | 0.03–1.0 | 0.13 | 0.25 | ||
| H30Rx | H30Rx | 35 | 0.06–1.0 | 0.25 | 0.50 | 0.001 |
| Others | 437 | 0.03–1.0 | 0.13 | 0.25 | ||
P values are by the Mann-Whitney U test (two-tailed).
TABLE 4.
Association of eravacycline MIC with ST131 genotype, stratified by fluoroquinolone phenotype
| Fluoroquinolone phenotype (no.) | ST131 subset | Group | ST131 subset members and MIC data |
P valuea |
||||
|---|---|---|---|---|---|---|---|---|
| No. of isolates | MIC range, mg/liter | MIC50, mg/liter | MIC90, mg/liter | ST131 subset vs others | Susceptible vs resistant isolates | |||
| Susceptible (234) | ST131 | ST131 | 14 | 0.06 to 0.50 | 0.13b | 0.50 | 0.04 | 0.17 |
| Others | 220 | 0.03 to 0.50 | 0.13b | 0.25 | <0.001 | |||
| H30 | H30 | 5 | 0.06 to 0.25 | 0.13 | 0.25 | 0.78 | 0.06 | |
| Others | 229 | 0.03 to 0.50 | 0.13 | 0.25 | <0.001 | |||
| H30Rx | H30Rx | 0 | n.a.c | n.a. | n.a. | n.a. | n.a. | |
| Others | 234 | 0.03 to 0.50 | 0.13 | 0.25 | <0.001 | |||
| Resistant (238) | ST131 | ST131 | 187 | 0.03 to 1.0 | 0.25 | 0.50 | 0.27 | |
| Others | 51 | 0.03 to 1.0 | 0.13 | 0.50 | ||||
| H30 | H30 | 180 | 0.03 to 1.0 | 0.25 | 0.50 | 0.20 | ||
| Others | 58 | 0.06 to 1.0 | 0.25 | 0.50 | ||||
| H30Rx | H30Rx | 35 | 0.06 to 1.0 | 0.25 | 0.50 | 0.28 | ||
| Others | 203 | 0.03 to 1.0 | 0.13 | 0.50 | ||||
P values (by two-tailed Mann-Whitney U test) are for comparison of ST131 group members versus others (same fluoroquinolone phenotype) or of fluoroquinolone-susceptible versus fluoroquinolone-resistant isolates (same ST131 genotype). Bold font represents significant difference (P < 0.05).
Mean MIC (fluoroquinolone-susceptible isolates): ST131, 0.21 mg/liter; others, 0.14 mg/liter.
n.a., not applicable (no fluoroquinolone-susceptible H30Rx isolates).
Comment.
Our findings support four main conclusions. First, eravacycline was broadly active, with MICs of ≤1.0 mg/liter irrespective of coresistance phenotype and ST131 status. Second, fluoroquinolone resistance was associated with higher MICs for all agents tested, although, for eravacycline, this difference was small. Third, eravacycline MICs varied with additional coresistance only for gentamicin and MDR status, likely due to associations with fluoroquinolone resistance. Fourth, although ST131 and its clonal subsets exhibited higher eravacycline MICs than other isolates, these differences also were small and likely reflected fluoroquinolone resistance. Thus, eravacycline should prove useful for treating multidrug-resistant E. coli infections among veterans, regardless of coresistance and ST131 genotype.
The present eravacycline MICs for E. coli from veterans are consistent with previously reported values from nonveterans (9, 11), suggesting that they are widely generalizable. Their unimodal distribution indicates the absence of a naturally resistant subpopulation that could be selected for by widespread eravacycline use. Whether such a resistant subpopulation might emerge with future extensive use remains to be seen.
The increment in eravacycline MICs observed among fluoroquinolone-resistant isolates was statistically significant but small (∼2-fold), so it is of doubtful clinical importance. Conceivably, a common resistance mechanism (e.g., specific efflux pumps) may reduce susceptibility to both fluoroquinolones and eravacycline. The similar minor (∼2-fold) but statistically significant eravacycline MIC elevations noted among gentamicin-resistant and MDR isolates disappeared with stratification by fluoroquinolone resistance, suggesting that they resulted from confounding by fluoroquinolone phenotype, consistent with the strong observed associations of gentamicin resistance and MDR status with fluoroquinolone resistance.
The ST131 isolates, and specifically those representing the H30 and H30Rx subclones, had slightly (∼2×) but significantly higher eravacycline MICs than other isolates. These differences likewise disappeared with stratification by fluoroquinolone phenotype. Thus, the overall higher eravacycline MICs of ST131 isolates likely reflected the higher prevalence of fluoroquinolone resistance in ST131 isolates, not some distinctive aspect of the ST131 clonal background. The small size of these ST131-associated MIC differences suggests that eravacycline should be comparably effective for treating E. coli infections regardless of the ST131 status of a strain.
This study is limited by its in vitro nature and the paucity of isolates from sources other than urine. Study strengths include the geographically dispersed strain collection, the comparisons with ST131 genotype, and the comparisons with key coresistance phenotypes, including MDR status and ESBL production.
Summary.
Among clinical E. coli isolates from U.S. veterans (in 2011), eravacycline MICs largely mirrored those reported previously for nonveteran patient populations (9, 11), with a symmetrical unimodal distribution centered around 0.125 to 0.25 mg/liter. Eravacycline MICs were statistically significantly higher, but only slightly so, in association with fluoroquinolone, gentamicin, and multidrug resistance and among ST131, H30, and H30Rx isolates, all of which were associations that tracked with fluoroquinolone resistance. Thus, eravacycline should be equally useful for treating extraintestinal E. coli infections in veterans regardless of coresistance phenotype and ST131 status.
ACKNOWLEDGMENTS
This material is based on work supported by an investigator-initiated grant from Tetraphase and by the Office of Research and Development, Medical Research Service, Department of Veterans Affairs, grant I01 CX000192 01 (both to J.R.J.). The sponsors had no involvement in the conduct of the study, data analysis, manuscript preparation, or decision to publish.
J.R.J. has received contracts, grants, or consultancies from Actavis, ICET, Jannsen, Merck, and Tetraphase and has patent applications pertaining to tests for specific Escherichia coli strains. The other authors report no conflicts of interest.
REFERENCES
- 1.Colpan A, Johnston B, Porter S, Clabots C, Anway R, Thao L, Kuskowski MA, Tchesnokova V, Sokurenko EV, Johnson JR, VICTORY (Veterans Influence of Clonal Types On Resistance: Year 2011) investigators . 2013. Escherichia coli sequence type 131 (ST131) as an emergent multidrug-resistant pathogen among U.S. veterans. Clin Infect Dis 57:1256–1265. doi: 10.1093/cid/cit503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Johnson JR, Johnston B, Clabots C, Kuskowski MA, Castanheira M. 2010. Escherichia coli sequence type ST131 as the major cause of serious multidrug-resistant E. coli infections in the United States (2007). Clin Infect Dis 51:286–294. doi: 10.1086/653932. [DOI] [PubMed] [Google Scholar]
- 3.Johnson JR, Tchesnokova V, Johnston B, Clabots C, Roberts PL, Billig M, Riddel K, Rogers P, Qin X, Butler-Wu S, Price LB, Aziz M, Nicolas-Chanoine M, Debroy C, Robicsek A, Hansen G, Urban C, Platell JL, Trott DJ, Zhanel G, Weissman SJ, Cookson B, Fang F, Limaye AP, Scholes D, Chattopadhyay S, Hooper DC, Sokurenko E. 2013. Abrupt emergence of a single dominant multidrug-resistant strain of Escherichia coli. J Infect Dis 207:919–928. doi: 10.1093/infdis/jis933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Price LB, Johnson JR, Aziz M, Clabots C, Johnston B, Tchesnokova V, Nordstrom L, Billig M, Chattopadhyay S, Stegger M, Andersen PS, Pearson T, Riddell K, Rogers P, Scholes D, Kahl B, Keim P, Sokurenko EV. 2013. The epidemic of ESBL-producing Escherichia coli ST131 is driven by a single highly virulent subclone, H30Rx. mBio 6:e00377-13. doi: 10.1128/mBio.00377-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nicolas-Chanoine M, Bertrand X, Madec J-Y. 2014. Escherichia coli ST131, an intriguing clonal group. Clin Microbiol Rev 27:543–574. doi: 10.1128/CMR.00125-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Johnson JR, Urban C, Weissman SJ, Jorgensen JH, Lewis JSI, Hansen G, Edelstein P, Robicsek A, Cleary T, Adachi J, Paterson DL, Lolans K, Quinn J, Hanson ND, Johnston BD, Clabots C, Kuskowski MA, AMERECUS investigators . 2012. Molecular epidemiology of Escherichia coli sequence type ST131 (O25:H4) and blaCTX-M-15 among extended-spectrum cephalosporinase-producing E. coli in the United States (2000-2009). Antimicrob Agents Chemother 56:2364–2370. doi: 10.1128/AAC.05824-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Banerjee R, Robicsek A, Kuskowski M, Porter S, Johnston B, Sokurenko E, Tchesnokova V, Price L, Johnson JR. 2013. Molecular epidemiology of Escherichia coli sequence type ST131 and its H30 and H30Rx subclones among extended-spectrum beta-lactamase-positive and -negative E. coli clinical isolates from the Chicago region (2007-2010). Antimicrob Agents Chemother 57:6385–6388. doi: 10.1128/AAC.01604-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Olesen B, Hansen DS, Nilsson F, Frimodt-Møller J, Leihof RF, Struve C, Scheutz F, Johnston B, Krogfelt KA, Johnson JR. 2013. Prevalence and characteristics of the epidemic multiresistant Escherichia coli ST131 clonal group among extended-spectrum beta-lactamase (ESBL)-producing E. coli in Copenhagen. J Clin Microbiol 51:1779–1785. doi: 10.1128/JCM.00346-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sutcliffe JA, O'Brian W, Fyfe C, Grossman TH. 2013. Antibacterial activity of eravacycline (TP-434), a novel fluorocycline, against hospital and community pathogens. Antimicrob Agents Chemother 57:5548–5558. doi: 10.1128/AAC.01288-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grossman TH, Murphy TM, Slee AM, Lofland D, Sutcliffe JA. 2015. Eravacycline (TP-434) is efficacious in animal models of infection. Antimicrob Agents Chemother 59:2567–2571. doi: 10.1128/AAC.04354-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Solomkin JS, Ramesh MK, Cesnauskas G, Novikovs N, Stefanova P, Sutcliffe JA, Walpole SM, Horn PT. 2014. Phase 2, randomized, double-blind study of the efficacy and safety of two dose regimens of eravacycline versus ertapenem for adult community-acquired complicated intraabdominal infections. Antimicrob Agents Chemother 58:1847–1854. doi: 10.1128/AAC.01614-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grossman TH, O'Brian W, Kerstein KO, Sutcliffe JA. 2015. Eravacycline (TP-434) is active in vitro against biofilms formed by uropathogenic Escherichia coli. Antimicrob Agents Chemother 59:2446–2449. doi: 10.1128/AAC.04967-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Grossman TH, Pillar CM, Sahm DF, Sutcliffe JA. 2015. In vitro susceptibility testing of eravacycline is unaffected by medium age and nonstandard assay parameters. Antimicrob Agents Chemother 59:2426–2427. doi: 10.1128/AAC.04727-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Abdallah M, Olafisoye O, Cortes C, Urban C, Landman D, Quale J. 2015. Activity of eravacycline against Enterobacteriaceae and Acinetobacter baumannii, including multidrug-resistant isolates, from New York City. Antimicrob Agents Chemother 59:1802–1805. doi: 10.1128/AAC.04809-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Morissey I, Sutcliffe JA, Hackel M, Hawser S. 2015. Assessment of eravacycline against a recent global collection of 4,462 Enterobacteriaceae clinical isolates (2013-2014). Joint 55th Intersci Conf Antimicrob Agents Chemother and 28th International Congress of Chemotherapy Meeting, San Diego, CA. [Google Scholar]
- 16.Solomkin J, Evans D, Slepavicius A, Marsh A, Tsai L, Horn P. 2015. Results of IGNITE1: a phase 3 study to evaluate the efficacy and safety of eravacycline versus ertapenem in complicated intra-abdominal infections, abstr LBOS0239d European Conference of Clinical Microbiology and Infectious Diseases, Copenhagen, Denmark. [Google Scholar]
- 17.Clinical and Laboratory Standards Institute. 2015. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically; approved standard—10th ed CLSI document M07-A10. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]
- 18.Clinical and Laboratory Standards Institute. 2015. Performance standards for antimicrobial susceptibility testing; twenty-fifth informational supplement. CLSI M100-S25. Clinical and Laboratory Standards Institute, Wayne, PA. [Google Scholar]


