Abstract
Aim:
To assess the effects of adding nano-titanium dioxide (nano-TiO2) and nano-silicon dioxide (nano-SiO2) and their mixture to poly (methyl methacrylate) (PMMA) to induce antimicrobial activity in acrylic resins.
Materials and Methods:
Acrylic specimens in size of 20 mm × 20 mm × 1 mm of 0.5% and 1% of nano-TiO2 (21 nm) and nano-SiO2 (20 nm) and their mixture (TiO2/SiO2 nanoparticles) (1:1 w/w) were prepared from the mixture of acrylic liquid containing nanoparticles and acrylic powder. To obtain 0.5% and 1% concentration, 0.02 g and 0.04 g of the nanoparticles was added to each milliliter of the acrylic monomer, respectively. Antimicrobial properties of six specimens of these preparations, as prepared, were assessed against planktonic Lactobacillus acidophilus and Streptococcus mutans at 0, 15, 30, 45, 60, 75, and 90 min follow-up by broth dilution assay. The specimens of each group were divided into three subgroups: Dark, daylight, or ultraviolet A (UVA). The percent of bacterial reduction is found out from the counts taken at each time point.
Statistical Analysis:
Data were analyzed using one-way analysis of variance and Tukey's post hoc analysis.
Results:
Exposure to PMMA containing the nanoparticles reduced the bacterial count by 3.2–99%, depending on the nanoparticles, bacterial types, and light conditions. Planktonic cultures of S. mutans and L. acidophilus exposed to PMMA containing 1% of TiO2/SiO2 nanoparticles showed a significant decrease (P < 0.001) (98% and 99%, respectively) in a time-dependent manner under UVA. The S. mutans and L. acidophilus counts did not significantly decrease in PMMA containing 0.5% nano-TiO2 and PMMA containing 0.5% nano-SiO2 in the dark. No statistically significant reduction (P > 0.05) was observed in the counts of S. mutans and L. acidophilus in PMMA without the nanoparticles exposed to UVA.
Conclusions:
PMMA resins incorporated with TiO2/SiO2 nanoparticles showed strong antimicrobial activity against the cariogenic bacteria.
Keywords: Antimicrobial activity, cariogenic bacteria, nano-silicon dioxide, nano-titanium dioxide, poly(methyl methacrylate)
INTRODUCTION
Plaque accumulation followed by enamel demineralization and gingivitis is a well-known complication in orthodontic therapy when fixed or removable appliances are used. The removable appliances are mostly made of poly(methyl methacrylate) (PMMA).[1] The challenging problem is susceptibility to plaque accumulation on these appliances due to the food retentive configuration of the acrylic materials. This may be attributed to the intrinsic porosity of PMMA, which enhances biofilm formation caused by the activities of cariogenic oral flora.[2]
Being used as orthodontic appliances or removable partial denture, the acrylic plates are placed in contact with teeth, and thus the resulting plaque accumulation may increase the risk of dental caries and gingival problems. In cold-cured acrylic resins (ARs), the problem is even more prominent, because they display more surface porosities than the heat-cured ones.[3,4] These porosities hamper the complete removal of dental plaques, such that mechanical cleaning often turns out to be inadequate. Therefore, some biocompatible antimicrobial agents are required.[5,6]
Several strategies have been proposed to reduce the risk of dental caries during orthodontic treatment. A recent method to prevent microorganism-induced oral diseases is to manufacture self-cleaning dental materials. The use of nanoparticles has been suggested to incorporate antimicrobial activity into orthodontic materials. Application of fluoride-releasing composites containing nanofillers, antimicrobial orthodontic band cement with silver nanoparticles, and nanosilver-containing orthodontic adhesive primers has been shown to be effective in inhibiting the bacterial growth around fixed appliances.[7,8] For removable appliances, addition of nanoparticles to PMMA can impart antimicrobial activity to acrylic plates. Among the available nanoparticles, titanium dioxide (TiO2) serves as a good example, because its antibacterial properties have been demonstrated in various biomaterials.[9,10] Moreover, composites containing TiO2 have been produced and assessed with regard to their antimicrobial effectiveness.[11] Among the orthodontic materials, orthodontic wires coated with TiO2 nanoparticles have demonstrated antimicrobial effects against Streptococcus mutans.[12] However, the effects of these particles on ARs have not yet received much attention.
The antimicrobial activities of TiO2 against Escherichia coli, Staphylococcus aureus, Pseudomonas aeruginosa, Candida albicans, Giardia lamblia, and Lactobacillus acidophilus have already been reported,[12,13,14] including its role as a cancer cell killer.[15] Evidently, TiO2 also has photocatalytic effects that enable it to remove pollutants in water via oxidation or reduction mechanisms. Different reactive oxygen-containing species such as H2O2, OH˙, and O2˙ that damage the bacterial cells are produced by the photo-initiating chemical reactions.[16,17]
Photocatalysis is a promising green technology for the inactivation of microorganisms because it does not cause adverse biological and environmental effects.[18] However, because of the wide band gap (approximately 3.2 eV), TiO2 nanoparticles are effective only in the UV region of the solar spectrum.[16] Besides its antibacterial effects, TiO2 has other advantages, which include its white color and high stability, as well as its low cost and low toxicity.[19,20,21] Therefore, it would be an appropriate substance that can be incorporated into the dental materials.
In order for antimicrobial agents to become more safe and durable and be released gradually, application of inorganic carriers such as apatite, zeolite, and phosphate has been suggested.[22] Silicon oxides are considered to be more appropriate carriers because of their porous structures and better adsorption properties. Nano-silicon dioxide (nano-SiO2) has the advantage of having extremely high surface activity, which enables it to absorb various ions and molecules.[23] The combination of nano-TiO2 and nano-SiO2 has been studied in some recent investigations.[24] We believe that by their incorporation into ARs, one could induce the desired photocatalytic and self-sterilizing properties to acrylic appliances. Because S. mutans and L. acidophilus are the most important cariogenic oral bacteria,[25] we included them in our study. Hence, this study was conducted for evaluating the antimicrobial activity of PMMA ARs containing TiO2 and SiO2 nanoparticles against S. mutans and L. acidophilus.
MATERIALS AND METHODS
Specimen Preparation
Acrylic liquid and powder (Selecta Plus, Dentsply Company, UK) were mixed, resulting in three-dimensional (20 mm × 20 mm × 1 mm) acrylic specimens. To compensate for the setting shrinkage of acryl, a larger metal mold was employed. Then, nano-TiO2 (21 nm) and/or nano-SiO2 (20 nm) (Degussa Company, Germany) particles were mixed with the monomeric acrylic liquid by stirring. To obtain 0.5% concentration, 0.02 g of the nanoparticles was added to each milliliter of the acrylic monomer. For 1% concentration, 0.04 g of the nanoparticles was added to each milliliter of the monomer.
For preparing specimens of the mixed groups (ARs containing both nano-TiO2 and nano-SiO2), they were mixed at a 1:1 weight ratio. The mixtures were sonicated (1 h for TiO2- containing monomers and 2–3 min for SiO2 -containing monomers), after which the powder and the acrylic liquid were mixed. Neat monomer containing no nanoparticles was used as the control group. The ratio of 3 g of powder to 1 ml of liquid was adopted, according to the manufacturer's instructions. The powder was gradually added to the liquid, and as it reached the dough stage, the prepared nano-metal oxide doped-ARs were introduced into the mold and allowed to set thoroughly. The specimens were polished to afford the aforementioned dimensions. Finally, they were sterilized by gamma rays, with at least 25 kilogray dosages.
Microorganism Tests and Growth Conditions
Lyophilized L. acidophilus and S. mutans (ATCC cultures 4356 and 25,175, respectively, obtained from Rayen Biotechnology Co., Ltd., Tehran, Iran) were rehydrated in brain heart infusion (BHI) broth (Merck, Darmstadt, Germany) and incubated in an anaerobic atmosphere at 37°C for 48 h.
Testing of Antimicrobial Activity by Broth Dilution Assay
After culturing the planktonic bacteria overnight, the bacterial density was adjusted to 0.2–0.3 at optical density 600 nm. The bacterial suspension was then diluted with BHI broth to 1 × 108 colony-forming units per milliliter (CFU/ml), and 100 μl of this bacterial suspension was pipetted into the acrylic samples. The specimens of each group were divided into three subgroups according to three different light conditions: Dark, daylight, or ultraviolet A (UVA). The samples were illuminated with 2 × 15 W UVA light (Philips Electronics, Seoul, Korea), daylight, and black light (300–500 nm emission). The light source was placed 10 cm above the samples. In the control group, the light source was covered with a black cloth to verify the viability of bacterial counts during the exposure. The samples were placed in dishes filled with cold water to prevent them from drying.
To determine the time dependence on photocatalysis, the samples were exposed to light sources for 0, 15, 30, 45, 60, 75, and 90 min. After illumination, at each time point, the samples were added into individual falcon tubes containing the bacterial suspensions. Then, the tubes were incubated in an anaerobic chamber (5% CO2, 10% H2, and 85% N2) at 37°C for 48 h. CFU/ml were assessed in the culture, before the exposure and after 0, 15, 30, 45, and 90 min exposure to test conditions. To determine the CFU/ml, the cells were serially diluted with a salt solution (0.9% NaCl) in microtiter plates and enumerated using the drop-plate method,[26] in which 20 μl of each dilution was added to BHI agar for L. acidophilus and S. mutans. After the inoculation, the CFUs/ml were counted to determine the inhibitory effect of PMMA containing nano-TiO2 and PMMA containing nano-SiO2. All the tests were performed in triplicate. The CFUs/ml for L. acidophilus and S. mutans were counted and the percent of bacterial reduction is found out from the counts taken at each time point. The cultures were checked for contamination throughout the experimental period.[27,28]
Statistical Analysis
The results of the assays were analyzed using one-way analysis of variance and Tukey's post hoc analysis using SPSS software (version 13, Chicago, IL, USA). In all the analyses, the confidence level was set at P < 0.05.
RESULTS
The reduction percentages of bacterial content (in CFU/ml) up to 90 min exposure to three different light conditions were compared and are shown in Table 1. Antibacterial activity of PMMA containing nanoparticles, at two concentrations (0.5% and 1%) of nano-TiO2 and nano-SiO2 and their mixture (TiO2/SiO2 nanoparticles) (1:1 w/w), against L. acidophilus and S. mutans cultures was time-dependent, such that extending the length of nanoparticles exposure from 15 to 90 min enhanced the inhibitory effects of nanoparticles on these microorganisms.
Table 1.
Bactericidal effects of poly (methyl methacrylate) containing nanoparticles under ultraviolet A illumination, visible light, and dark conditions
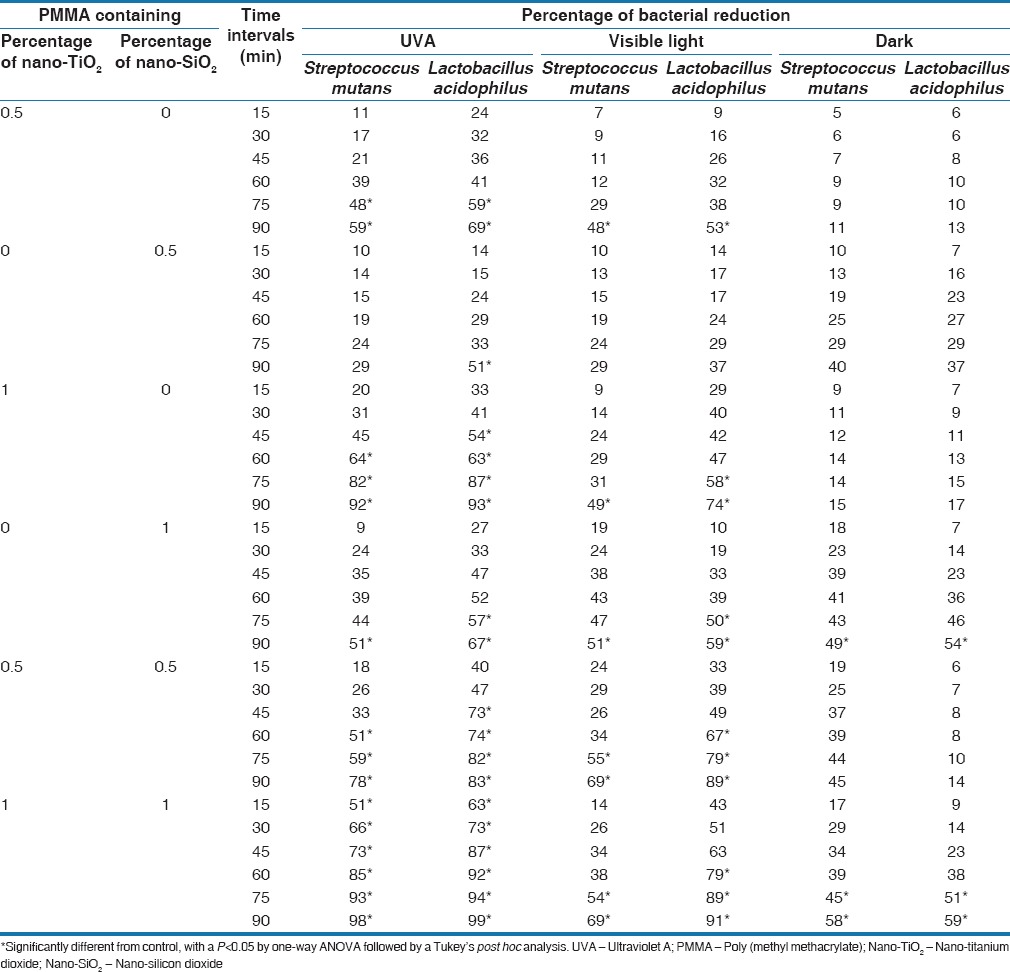
In general, the antimicrobial activity of nano-TiO2 was significantly higher than the activity of nano-SiO2 under UVA illumination (P < 0.05) and visible light (P < 0.05) [Table 1]. After a 90 min UVA exposure, 0.5% nano-TiO2 reduced culture viability of L. acidophilus and S. mutans by 69% and 59%, respectively. The differences between the antibacterial effects of samples containing 0.5% nano-TiO2 versus those with PMMA alone were statistically significant (P < 0.05). Although the 90 min time point showed reductions in the viability of L. acidophilus and S. mutans cultures treated with 0.5% nano-TiO2 in the dark, these reductions were not significant (P > 0.05) [Table 1]. On the other hand, 1% nano-TiO2 reduced L. acidophilus and S. mutans viability by 93% and 92% under UVA illumination, respectively, suggesting more antibacterial effect on these bacterial cultures than 0.5% nano-TiO2.
In addition, 1% nano-SiO2 had a bactericidal effect on both L. acidophilus and S. mutans cultures in a time-dependent manner. After a 90 min exposure, 1% nano-SiO2 reduced the viability of L. acidophilus and S. mutans by 59% and 51%, respectively, with more antibacterial effect at the earlier time points. The antibacterial activity of 0.5% nano-SiO2 ranged from modest to none. Culture of L. acidophilus showed the highest susceptibility to 0.5% nano-SiO2 under UVA illumination, which reduced bacterial viability by 51%. Furthermore, 0.5% nano-SiO2 did not inhibit the growth of S. mutans cultures at any of the test conditions and time points, similar to those treated with PMMA alone. Among the groups containing the nanoparticles, the group containing 0.5% nano-SiO2 showed the lowest reduction of 5 ± 5.0% for S. mutans, under dark conditions, although the difference was not statistically significant compared with the control group (P = 0.43). Most notably, at all time points of the assay, 1% of both nano-TiO2 and nano-SiO2 under UVA illumination showed a higher inhibitory activity than PMMA alone (P < 0.001), which did not inhibit the growth of any test culture.
Regarding the three light conditions, the highest reduction in bacterial population (in CFU/ml) occurred in the UVA group (P < 0.05), followed by the visible light group (P < 0.05), and the least percentage of reduction was observed in the group exposed to dark (P > 0.05). In the control group, reduction in bacterial population was not observed under UVA exposure, which actually showed 4.3% increase in L. acidophilus CFU/ml. Overall, under UVA illumination, the potential to reduce bacterial cells was as follows: Mixed group (TiO2/SiO2) > TiO2 > SiO2. In visible light condition, both TiO2 and SiO2 exhibited similar antimicrobial effects against S. mutans at both 1% and 0.5% concentrations (P > 0.05); however, TiO2 (whether 0.5% or 1%) showed a higher reduction of L. acidophilus under UVA illumination. SiO2 (0.5% or 1%) showed a higher reduction percentage of CFU/ml for both S. mutans and L. acidophilus than that by TiO2 under dark condition.
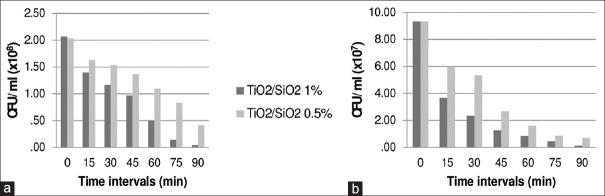
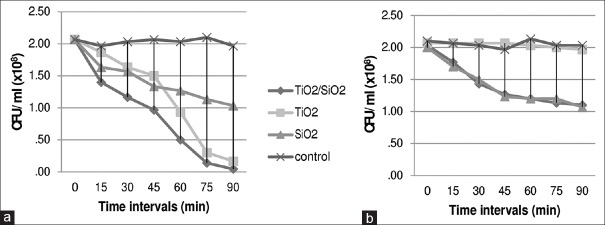
The antimicrobial effect was more obvious in higher concentrations. For example, at the last follow-up in 1% TiO2 [Table 1], the reduction percent of L. acidophilus under UVA (93%) was valuable while, for 0.5% TiO2, the reduction was 69%. The same pattern was observed for SiO2 and TiO2/SiO2. Figure 1 illustrates the comparison of the different concentrations of the mixed groups. A comparison of the effect of different nanoparticles and the bacterial reduction trend in ARs containing TiO2, SiO2, and TiO2/SiO2 in dark and under UVA against S. mutans are illustrated in Figure 2.
Figure 1.
Colony-forming units per milliliter of Streptococcus mutans (a) and Lactobacillus acidophilus (b) on acrylic resins contained 1% and 0.5% titanium dioxide/silicon dioxide nanoparticles in different time intervals (min = minutes)
Figure 2.
Colony-forming units per milliliter of Streptococcus mutans, on acrylic resins contained titanium dioxide/silicon dioxide 1%, titanium dioxide 1%, silicon dioxide 1% and no nanoparticle (control) in two different light conditions: (a) Ultraviolet A illumination and (b) dark, in different follow-ups (min = minutes)
DISCUSSION
One of the recent approaches in preventive dentistry is to apply self-sterilizing dental materials. This study demonstrated that adding nanoparticles to acrylic specimens could reduce bacterial growth and population. A significant reduction in the number of bacterial CFU/ml was observed for L. acidophilus in acrylic specimens containing 1% of both nano-TiO2 and nano-SiO2, exposed to UVA for 90 min (98.6 ± 0.4%). Under similar conditions, the S. mutans population was reduced by 97.9 ± 0.3%, while that of the control group increased [Table 1]. Regarding the nanoparticles, it may be concluded that the higher the concentration, the higher the antimicrobial activity. Hence, a concentration of 1% turned out to be more bactericidal than 0.5%, when nano-TiO2, nano-SiO2, or their mixtures are employed [Table 1]. This is consistent with the findings of other investigators;[14,29] however, no threshold has been suggested till date, which calls for further studies.
The antibacterial activity of nano-ARs appears to be much higher in UVA than in dark [Figure 2]. However, a quick glimpse of the control group results reveals that in the absence of nanoparticles, UVA does not show any antimicrobial activity [Table 1]. By comparing the results of the nano groups exposed to UVA versus those in dark and visible light, it can be concluded that although TiO2 nanoparticles (whether 1% or 0.5%) could reduce the bacterial population in dark and visible light, utilizing UVA would enhance their antimicrobial activity [Table 1]. These findings are in harmony with those previously reported.[30]
According to Fujishima et al.,[16] the intensity of UVA is directly related to the photocatalytic activity of nanoparticles, due to the fact that a higher intensity leads to the formation of more reactive oxygen species. The photocatalytic activity of TiO2 was evident even in visible light. Thus, the bacterial growth inhibition was more prominent in the visible light than in dark light [Table 1]. However, in the ARs containing nano-SiO2 alone, the antimicrobial activity in visible light did not differ from that in dark, implying that light has no significant effect in improving the bactericidal properties of SiO2. Our data showed that, as can be seen in dark, the behavior of ARs containing nano-TiO2 resembles that of the control group, but the ARs containing nano-SiO2, whether alone or in combination with TiO2, led to reduction in the CFU/ml over time. These findings are consistent with those of Adams et al.,[29] who showed that the toxicity of nano-sized SiO2 against Bacillus subtilis and E. coli was displayed even in dark. This result suggests the possibility of another mechanism for the antimicrobial activity of SiO2 nanoparticles, which deserves further scrutiny.
Although nano-SiO2 was not as effective as nano-TiO2 under UVA, this study showed that in contrast to previous studies, nano-sized SiO2 was not an inert substance and had some antibacterial effects. The fact that ARs containing both TiO2 and SiO2 nanoparticles had the highest bacterial growth inhibition suggests that when used as a carrier of TiO2, SiO2 can enhance the antimicrobial capacity of nano-sized TiO2. This can be attributed to a probable synergistic effect of TiO2 and SiO2 nanoparticles or the capacity of nano-SiO2 to improve the photocatalytic properties of TiO2. Recent studies have shown that the combination of TiO2/SiO2 nanoparticles possesses higher stability than pure TiO2. This enhances the photocatalytic activity of nanoparticles due to the higher specific surface area, larger pore volumes, higher band gap energy, and smaller crystallite structure.[31] Thus, nano-SiO2 can be used as the carrier of TiO2, as was performed in this study.
The results of this study also show that the antimicrobial activities of nanoparticles increased with time because the last follow-up (after 90 min) demonstrated the least CFU/ml [Figures 1 and 2]. Thus, a longer follow-up is needed to detect at what time the reduction trend reaches a plateau. Choi et al.[12] claimed that the maximum antimicrobial activity of orthodontic wires coated with TiO2 nanoparticles was observed in 20–30 min exposure to UVA, after which a reduction ensued. The same pattern was reported by Watts et al.[32] while a two-step decay dynamics of the photokilling process was reported by Sunada et al.[33] The latter pattern comprises a very low-rate bactericidal step followed by a higher rate step. However, in the present study, a steady decline in the number of microorganisms in the ARs containing TiO2 and SiO2 nanoparticles was observed up to the last follow-up (after 90 min).
In the current study, the catalytic effect of UVA was slightly more prominent on L. acidophilus growth suppression than that on S. mutans (99% compared to 98% reduction in CFU/ml) [Table 1]. In other words, L. acidophilus exhibited more sensitivity to the antimicrobial effects of the nanoparticles than S. mutans. This can be attributed to the different thickness and complexity of the cell walls of the microorganism. Several theories have been proposed for the photokilling mechanism of TiO2 under UV irradiation. Direct photocatalytic oxidation of co-enzyme A or pre-oxidation of the membrane phospholipids[17] has been suggested.
To benefit from the advantages of UVA, the patients should be advised to keep their acrylic orthodontic appliances containing TiO2 and SiO2 nanoparticles for about 1 h in outdoors under sun exposure or under UVA lamps with an intensity of 1 mW/cm2. The solar UVA intensity is about 4 mW/cm2 on sunny days and drops by about 10 times on cloudy days.[34] For denture base material, the patients should be advised to keep their dentures under an appropriate irradiation source during the night, but for orthodontic appliances that critically require nocturnal wearing, daytime exposure to UVA is more prudent. Thus, if these instructions are followed, there will be lesser microbial load in the mouth, which in turn would result in the prevention of bacterial oral diseases such as dental caries and gingivitis. However, one should notice that besides the antimicrobial activities of TiO2 nanoparticles, a high concentration of nano-TiO2 can affect the appearance and mechanical properties of ARs by acting as impurities.[11]
The effect of adding TiO2 and SiO2 nanoparticles on the flexural strength of ARs was earlier evaluated by our research team.[35] Thus, further studies are needed to compare the different nanoparticles separately or in combination with TiO2, with respect to their antimicrobial activities and effects on the mechanical properties of the acrylic material. Further studies also are required to evaluate the flexural strength and the physical properties of the new mixed material at varying percentage concentrations as well as further clinical evaluations are required to substantiate these results as well as the biocompatibility of the acrylic material. A limitation of the study was the short duration of the experiment and longer period is needed to extrapolating the results of this study to the anticariogenic effects of ARs incorporated with nanoparticles. Moreover, in light of the importance of flexural strength as an important mechanical property of acrylic material, the lack of evaluation of the effect of nanoparticles on the flexural strength of PMMA ARs can be a limitation in this study. To address this issue, further studies should be considered.
CONCLUSIONS
Adding TiO2 and SiO2 nanoparticles to PMMA can impart antimicrobial activity to the resins, which is more efficient under UVA exposure, owing to the photocatalytic properties of nano-TiO2. Using this method, self-sterilizing acrylic appliances can be applied to prevent bacterial oral diseases in patients who wear them.
Financial Support and Sponsorship
This research has been supported by Tehran University of Medical Sciences & health Services grant No. 8778-69-02-89.
Conflicts of Interest
There are no conflicts of interest.
Acknowledgment
The authors would like to appreciate all persons helped us in this project.
REFERENCES
- 1.Faltermeier A, Rosentritt M, Müssig D. Acrylic removable appliances: Comparative evaluation of different postpolymerization methods. Am J Orthod Dentofacial Orthop. 2007;131:301.e16–22. doi: 10.1016/j.ajodo.2006.07.019. [DOI] [PubMed] [Google Scholar]
- 2.Ghorbanzadeh R, Pourakbari B, Bahador A. Effects of baseplates of orthodontic appliances with in situ generated silver nanoparticles on cariogenic bacteria: A randomized, double-blind cross-over clinical trial. J Contemp Dent Pract. 2015;16:291–8. doi: 10.5005/jp-journals-10024-1678. [DOI] [PubMed] [Google Scholar]
- 3.Morgan TD, Wilson M. The effects of surface roughness and type of denture acrylic on biofilm formation by Streptococcus oralis in a constant depth film fermentor. J Appl Microbiol. 2001;91:47–53. doi: 10.1046/j.1365-2672.2001.01338.x. [DOI] [PubMed] [Google Scholar]
- 4.Duymus ZY, Yanikoglu ND. The investigation of solubility values, water sorption and bond strength of auto-polymerising and heat-polymerising acrylic resin materials. Eur J Prosthodont Restor Dent. 2006;14:116–20. [PubMed] [Google Scholar]
- 5.Borzabadi-Farahani A, Borzabadi E, Lynch E. Nanoparticles in orthodontics, a review of antimicrobial and anti-caries applications. Acta Odontol Scand. 2014;72:413–7. doi: 10.3109/00016357.2013.859728. [DOI] [PubMed] [Google Scholar]
- 6.Melo MA, Morais WA, Passos VF, Lima JP, Rodrigues LK. Fluoride releasing and enamel demineralization around orthodontic brackets by fluoride-releasing composite containing nanoparticles. Clin Oral Investig. 2014;18:1343–50. doi: 10.1007/s00784-013-1073-5. [DOI] [PubMed] [Google Scholar]
- 7.Moreira DM, Oei J, Rawls HR, Wagner J, Chu L, Li Y, et al. A novel antimicrobial orthodontic band cement with in situ-generated silver nanoparticles. Angle Orthod. 2015;85:175–83. doi: 10.2319/022314-127.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blöcher S, Frankenberger R, Hellak A, Schauseil M, Roggendorf MJ, Korbmacher-Steiner HM. Effect on enamel shear bond strength of adding microsilver and nanosilver particles to the primer of an orthodontic adhesive. BMC Oral Health. 2015;15:42. doi: 10.1186/s12903-015-0024-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Su W, Wei SS, Hu SQ, Tang JX. Preparation of TiO (2)/Ag colloids with ultraviolet resistance and antibacterial property using short chain polyethylene glycol. J Hazard Mater. 2009;172:716–20. doi: 10.1016/j.jhazmat.2009.07.056. [DOI] [PubMed] [Google Scholar]
- 10.Yao Y, Ohko Y, Sekiguchi Y, Fujishima A, Kubota Y. Self-sterilization using silicone catheters coated with Ag and TiO2 nanocomposite thin film. J Biomed Mater Res B Appl Biomater. 2008;85:453–60. doi: 10.1002/jbm.b.30965. [DOI] [PubMed] [Google Scholar]
- 11.Feng XX, Zhang LL, Chen JY, Guo YH, Zhang HP, Jia CI. Preparation and characterization of novel nanocomposite films formed from silk fibroin and nano-TiO2. Int J Biol Macromol. 2007;40:105–11. doi: 10.1016/j.ijbiomac.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 12.Choi JY, Kim KH, Choy KC, Oh KT, Kim KN. Photocatalytic antibacterial effect of TiO (2) film formed on Ti and TiAg exposed to Lactobacillus acidophilus. J Biomed Mater Res B Appl Biomater. 2007;80:353–9. doi: 10.1002/jbm.b.30604. [DOI] [PubMed] [Google Scholar]
- 13.Shibata T, Hamada N, Kimoto K, Sawada T, Sawada T, Kumada H, et al. Antifungal effect of acrylic resin containing apatite-coated TiO2 photocatalyst. Dent Mater J. 2007;26:437–44. doi: 10.4012/dmj.26.437. [DOI] [PubMed] [Google Scholar]
- 14.Lee JH, Kang M, Choung SJ, Ogino K, Miyata S, Kim MS, et al. The preparation of TiO2 nanometer photocatalyst film by a hydrothermal method and its sterilization performance for Giardia lamblia. Water Res. 2004;38:713–9. doi: 10.1016/j.watres.2003.10.011. [DOI] [PubMed] [Google Scholar]
- 15.Blake DM, Maness P, Huang Z, Wolfrum EJ, Huang J. Application of the photocatalytic chemistry of titanium dioxide to disinfection and the killing of cancer cells. Sep Purif Methods. 1999;28:1–50. [Google Scholar]
- 16.Fujishima A, Rao TN, Tryk DA. Titanium dioxide photocatalysis. J Photoch Photobio C. 2000;1:1–21. [Google Scholar]
- 17.Fonseca SM, Barker AL, Ahmed S, Kemp TJ, Unwin PR. Direct observation of oxygen depletion and product formation during photocatalysis at a TiO2 surface using scanning electrochemical microscopy. Chem Commun (Camb) 2003;21:1002–3. doi: 10.1039/b212222g. [DOI] [PubMed] [Google Scholar]
- 18.Wan Y, Zhang D, Wang Y, Qi P, Wu J, Hou B. Vancomycin-functionalised Ag/TiO2 phototoxicity for bacteria. J Hazard Mater. 2011;186:306–12. doi: 10.1016/j.jhazmat.2010.10.110. [DOI] [PubMed] [Google Scholar]
- 19.Linsebigler AL, Lu GQ, Yates JT. Photocatalysis on TiO2 surfaces: Principles, mechansms, and selected results. Chem Rev. 1995;95:735–58. [Google Scholar]
- 20.Mu R, Xu Z, Li L, Shao Y, Wan H, Zheng S. On the photocatalytic properties of elongated TiO2 nanoparticles for phenol degradation and Cr (VI) reduction. J Hazard Mater. 2010;176:495–502. doi: 10.1016/j.jhazmat.2009.11.057. [DOI] [PubMed] [Google Scholar]
- 21.Pant HR, Pandeya DR, Nam KT, Baek WI, Hong ST, Kim HY. Photocatalytic and antibacterial properties of a TiO2/nylon-6 electrospun nanocomposite mat containing silver nanoparticles. J Hazard Mater. 2011;189:465–71. doi: 10.1016/j.jhazmat.2011.02.062. [DOI] [PubMed] [Google Scholar]
- 22.Jia H, Hou W, Wei L, Xu B, Liu X. The structures and antibacterial properties of nano-SiO2 supported silver/zinc-silver materials. Dent Mater. 2008;24:244–9. doi: 10.1016/j.dental.2007.04.015. [DOI] [PubMed] [Google Scholar]
- 23.Matsunami N, Hosono H. Colloid formation effects on depth profile of implanted Ag in SiO2 glass. Appl Phys Lett. 1993;63:2050–2. [Google Scholar]
- 24.Fujishima M, Takatori H, Tada H. Interfacial chemical bonding effect on the photocatalytic activity of TiO2 -SiO2 nanocoupling systems. J Colloid Interface Sci. 2011;361:628–31. doi: 10.1016/j.jcis.2011.06.024. [DOI] [PubMed] [Google Scholar]
- 25.Van Houte J, Aasenden R, Peebles TC. Lactobacilli in human dental plaque and saliva. J Dent Res. 1981;60:2–5. doi: 10.1177/00220345810600010401. [DOI] [PubMed] [Google Scholar]
- 26.Bahador A, Lesan S, Kashi N. Effect of xylitol on cariogenic and beneficial oral streptococci: A randomized, double-blind crossover trial. Iran J Microbiol. 2012;4:75–81. [PMC free article] [PubMed] [Google Scholar]
- 27.Fekrazad R, Khoei F, Hakimiha N, Bahador A. Photoelimination of Streptococcus mutans with two methods of photodynamic and photothermal therapy. Photodiagnosis Photodyn Ther. 2013;10:626–31. doi: 10.1016/j.pdpdt.2013.07.002. [DOI] [PubMed] [Google Scholar]
- 28.Bahador A, Pourakbari B, Bolhari B, Hashemi FB. In vitro evaluation of the antimicrobial activity of nanosilver-mineral trioxide aggregate against frequent anaerobic oral pathogens by a membrane-enclosed immersion test. Biomed J. 2015;38:77–83. doi: 10.4103/2319-4170.132901. [DOI] [PubMed] [Google Scholar]
- 29.Adams LK, Lyon DY, McIntosh A, Alvarez PJ. Comparative toxicity of nano-scale TiO2, SiO2 and ZnO water suspensions. Water Sci Technol. 2006;54:327–34. doi: 10.2166/wst.2006.891. [DOI] [PubMed] [Google Scholar]
- 30.Maneerat C, Hayata Y. Antifungal activity of TiO2 photocatalysis against Penicillium expansum in vitro and in fruit tests. Int J Food Microbiol. 2006;107:99–103. doi: 10.1016/j.ijfoodmicro.2005.08.018. [DOI] [PubMed] [Google Scholar]
- 31.Mahyar A, Behnajady MA, Modirshahla N. Enhanced photocatalytic degradation of C.I. basic violet 2 using TiO2-SiO2 composite nanoparticles. Photochem Photobiol. 2011;87:795–801. doi: 10.1111/j.1751-1097.2011.00930.x. [DOI] [PubMed] [Google Scholar]
- 32.Watts RJ, Kong S, Orr MP, Miller GC, Henry BE. Photocatalytic inactivation of coliform bacteria and viruses in secondary wastewater effluent. Water Res. 1995;29:95–100. [Google Scholar]
- 33.Sunada K, Watanabe T, Hashimoto K. Studies on photokilling of bacteria on TiO2 thin film. J Photochem Photobiol A Chem. 2003;156:227–33. [Google Scholar]
- 34.Akhavan O. Lasting antibacterial activities of Ag-TiO2/Ag/a-TiO2 nanocomposite thin film photocatalysts under solar light irradiation. J Colloid Interface Sci. 2009;336:117–24. doi: 10.1016/j.jcis.2009.03.018. [DOI] [PubMed] [Google Scholar]
- 35.Sodagar A, Bahador A, Khalil S, Shahroudi AS, Kassaee MZ. The effect of TiO2 and SiO2 nanoparticles on flexural strength of poly (methyl methacrylate) acrylic resins. J Prosthodont Res. 2013;57:15–9. doi: 10.1016/j.jpor.2012.05.001. [DOI] [PubMed] [Google Scholar]