Abstract
Objective:
Nucleus accumbens (NAcc) has a role in addiction and ingestive behavior. In order to assess orexinergic system involved in this, we infused Orexin A antagonist and assessed the effect on food intake fluid intake and alcohol preference in Wistar rats.
Materials and Methods:
Inbred Wistar rats (n = 54) were divided into control and experimental groups (low dose and high dose). Using stereotaxic method, guide cannula was set in place bilaterally to reach NAcc. Low dose (3 ng) and high dose (6 ng) of Orexin A antagonist (SB-334867) was infused, and the food consumption, water intake and alcohol intake, and two bottle free choice preference test for alcohol were carried out in the experimental group. The control group received saline infusion and rest of the methods followed were same. The measurements were carried out immediately after the infusion, at 1 h, 2 h, 4 h, and for the whole day and represented in the figure and tables.
Results:
A decrease in water intake observed immediately after the infusion in 1st h (P < 0.05) and 2nd h (P < 0.01), which was more in high dose group compared to low dose and controls. Alcohol intake was also following the same pattern. In two bottle free choice, rats did not show any specific preference to alcohol.
Conclusion:
There was dose dependent reduction in intake of food and fluids in treated rats. This suggested a possible role for orexinergic system in ingestive behavior. However, Orexin A may not have a role in modulation of alcohol addiction by the rewarding center NAcc.
KEY WORDS: Nucleus accumbens, food, Orexin-A antagonist (SB-334867), water and alcohol
Introduction
accumbens (NAcc) is known to affect the ingestive behavior and addiction to substances.[1,2] It has also been implicated in reward and motivation.[3] Two distinct sub regions have been described in NAcc, and there appeared a lot of overlap in the functioning of these two regions.[4] Microinjection of addictive drugs into NAcc showed a dose-dependent increase in dopamine levels. Dopamine is a neurochemical involved in reward circuitry. Intranuclear self-administration of amphetamine showed an increased reward feeling.[5] Other parts in subcortical region too influence ingestive behavior, which may be closely linked to accumbens. Food intake and water intake were shown to be influenced by several parts of the basal brain, such as lateral hypothalamus,[6] paraventricular nucleus, septum pellucidum,[7] and basolateral amygdala.[8] There is extensive neural networking in the regulation of ingestive behavior. NAcc has been shown to be well connected to the major subcortical centers, such as ventral tegmental area (VTA);[9] basolateral amygdala and other centers.[10] Major neurotransmitters involved in the modulation of alcohol intake, reward, and food intake were reported from the neural circuits in this region.[11]
Recently, hypocretins, a group of heterogeneous peptides, have been identified from the hypothalamus, hypocretin 1 and 2 (Orexin A and B), peptides having 32 aa and 29 aa, respectively.[12] Originally, these substances have been implicated in the regulation of sleep wakeful cycle, as evidenced by the increased Orexin A found in hypothalamus during the active phase of wakefulness.[13] In our laboratory, we found that Orexin A increased food and fluid intake when injected into NAcc.[14] The centrally administered orexin antibody inhibits food intake dose-dependently in 24 h fasted rats, and intraperitoneal administration of orexin antibody failed to suppress food consumption, represents that orexin antibody acts in the central nervous system but not in the peripheral tissue.[15] Further, they confirmed that endogenous orexins have physiological role on feeding behavior. Though initially Orexins were postulated to affect ingestive behavior, the evidence of role for Orexin A in feeding behavior and the alcohol intake and also alcohol preference are sketchy. In our previous study, we have demonstrated that Orexin A increased food and fluid intake in fasted rats.[14] In order to elucidate the role of Orexin A in the ingestive activity unequivocally, we carried out this set of experiments in male Wistar rats fasted overnight, by infusing the Orexin A antagonist SB-334867 into the NAcc.[16] We also tested the alcohol preference in treated rats to elucidate the effect of Orexin A antagonist on alcohol intake. The results of the experiments on food, water and alcohol intake, and alcohol preference are discussed here.
Materials and Methods
Fifty-four male Wistar albino rats (n = 54) weighing (250 ± 10 g), 3-4 months old were selected for study. They were divided into three groups viz. Water group, alcohol group and two bottle free choice group (n = 18 each). They were subdivided into three subgroups, viz. Group 1 - Control (Saline infusion); Group 2 - Low dose of SB-334867 (3 ng); Group 3 - High dose of SB-334867 (6 ng, n = 6 each). Food and fluid was provided to all groups ad lib, except where mentioned for overnight fasting.
All animals were housed individually in polypropylene cages, with proper husk bedding and maintained on a 12-h light/dark cycle in temperature controlled environment. Animals were maintained according to the guidelines of committee for the purpose of control and supervision of experiments on animals and guidelines of the Government of India for the use of laboratory animals. Institutional Animal Ethical Committee has approved this study protocol.
Drugs and Apparatus
Saline 0.9%, SB-334867 (From: Tocris bioscience) was dissolved in 2% cyclodextran in sterile water. When not in use, solutions were stored at 4°C up to 3 weeks. Harvard Pico plus (USA) infusion pump was used to deliver drug. Tap water was provided in Plastic drinking bottles, and rat food pellets (Hindustan Unilever Ltd.,) were provided. Ethyl alcohol (Absolute) was procured (Hayman Ltd., Eastways Park, Witham, Essex, CM83YE, UK) and diluted to make a 10% alcohol (This concentration was selected on the basis of a pilot study on the preference of alcohol concentration). Ketamine (NEON Laboratories limited, Thane, M.S) and xylazine (Indian immunological Ltd., Hyderabad) were used for anesthesia.
Surgical Procedure
Male Wistar albino rats were anesthetized by injecting a mixture of ketamine hydrochloride (60 mg/kg), xylazine hydrochloride (6 mg/kg) and mounted on a stereotaxic apparatus (Inco, India). An incision was made on the scalp, thoroughly disinfected with surgical spirit. The area was cleaned up with cotton and hydrogen peroxide. Coordinate points were marked on the skull in corresponding areas to reach NAcc, with the reference of Paxinos and Watson brain atlas[17] (From Bregma: Anteroposterior +2.2 mm, lateral ±1 mm and vertical 7.4 mm). Burr hole was made, and stainless steel guide cannula (22 gauze) was implanted according to the stereotaxic coordinates. Once the cannula is in place, it is secured with the help of screws and dental acrylic. Guide cannula was fitted with stylet, and rats were allowed to recover for at least 7 days prior to the experimentation. Infusion cannula (internal cannula) was fabricated from Septoject stainless steel dental needle of 30 gauges which has a hub, convenient for handling.[18] Infusion cannula extends 1 mm beyond the respective guide cannula. Prior to the start of the experiments all rats received two training sessions in which they were kept on 24 h fasting then received food, water, and 10% alcohol. During this session, rats learnt fasting.
Experimental Procedure
Normal saline and SB-334867 (in two doses) were infused, respectively, in separate groups of rats after 24 h fasting; unanesthetized (free moving) rats through the secured guide cannula. The infusion was carried out by 10 μl Hamilton syringe connected with polyethylene tube and internal cannula. This syringe was fitted to the Harvard pump. Then the stylet placed in the guide cannula was removed. The internal cannula was inserted into the guide cannula and secured. Then the pump was started to deliver the solution into the right and left side of NAcc one after the other, 1 μl/min (after infusion internal cannula left for about 10 s to allow drug diffusion). Two doses of SB-334867 were infused at 3 ng (low dose) and 6 ng (high dose). At the end of infusion, the internal cannula was removed, and stylet was placed back in position and time was noted. Post infusion, immediately premeasured amount of food, water, and 10% alcohol were provided in respective groups. The effect of SB-334867 on consumption was measured and noted, meticulously at infusion time intervals 1, 2, 4, and 24 h, respectively. Left over pellets, water, and alcohol were removed, and the amount consumed was calculated (Amount consumed = Premeasured quantity-left over quantity, e.g. at the end of 1 h).
Following the completion of the study, the rats were sacrificed by lethal dose of anesthesia and the brain was dissected out and preserved for histological processing. Seven micron sections were sliced and stained with cresyl violet to confirm the site of infusion [Figure 1].
Figure 1.
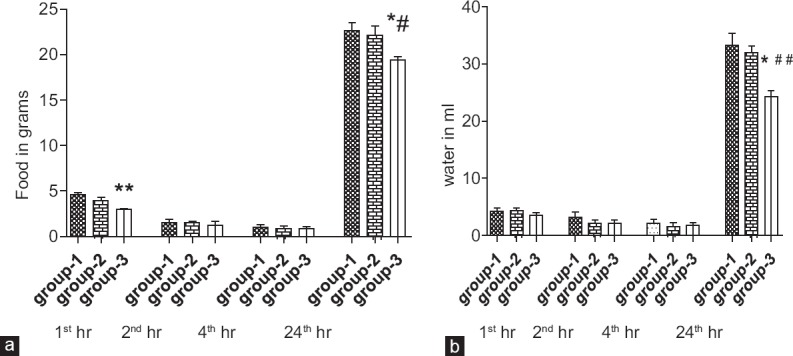
Orexin A antagonist (SB-334867) in the nucleus accumbens. Bars represent, (a) water and (b) food intake of rats injected with SB-334867 in the nucleus accumbens, at 1st, 2nd, 4th and 24 h time period with dose of 0 (0.9% saline = Group 1) and 3 ng SB-334867 (Group 2) or 6 ng SB-334867 (Group 3), each bar represents the mean ± standard error of mean, (*) indicates significant difference between Group 1 versus Group 2 or Group 3, (#) indicates, significant difference between Group 2 versus Group 3. Analysis by ANOVA significance (post hoc Tukey highly significant difference, n = 18), 1st h food: Group 1 versus Group 3** P < 0.002, 24 h food: Group 1 versus Group 3** P < 0.028; Group 2 versus Group 3 #P < 0.05, 24 h water: Group 1 versus Group 3** P < 0.002; Group 2 versus Group 3 ##P < 0.006
Statistical Analysis
Analysis of the data was done using the statistical software SPSS version - 16 (SPSS for Windows, Version 16.0. Chicago, SPSS Inc. USA); one-way ANOVA was done to compare the consummatory behavior in between the groups. Inter comparison was done by post-hoc Tukey's test (Hourly consumption compared separately, e.g. 1 h control food intake vs. 1 h SB-334867 treated food intake). Data were expressed as mean ± standard error of mean P < 0.05, was considered significant.
Results
Experiment I
Food and water consumption were measured (n = 18) in this group, NAcc cannulated animals (n = 18), were divided into subgroups, Group 1 (0.9% saline infusion), Group 2 (SB-334867-3 ng), Group 3 (SB-334867-6 ng). Drugs were injected bilaterally into NAcc [Data showed in Table 1 and Figure 1a, b].
Table 1.
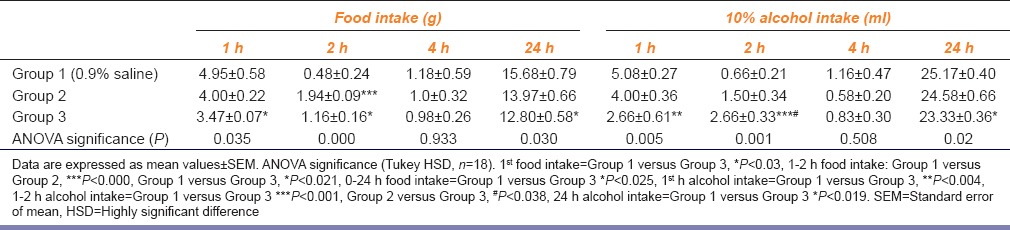
Effect of SB-334867 on food and 10% alcohol intake at 1, 2, 4 and 24 h time period (n=6 in each group)
Food intake
Compared to control at 1 h after SB-334867 treatment showed significant decrease (F[2, 15] =9.171 p = 0.003) in the food intake (i.e. Group 1 vs. Group 3, p < 0.002); whereas, no significant change was noticed at 2 h (F[2, 15] = 0.190 p = 0.829); 4 h (F[2, 15] = 0.160 p = 0.854); 24 h postinfusion time intervals (F[2, 15] = 4.873 p = 0.023) (Group 1 vs. Group 3, p < 0.028; Group 2 vs. Group 3, p < 0.05).
Water intake
SB-334867 treatment showed no effect on water intake at 1 h (F[2, 15] =0.957 p = 0.406); 2 h (water 2 h F[2, 15] = 0.773 p = 0.479); 4 h (F[2, 15] =0.288 p = 0.753) postinfusion time intervals; but total 24 h water intake was decreased (F[2, 15] = 10.688 p = 0.001) compared to control (Group 1 vs. Group 3, p < 0.002; Group 2 vs. Group 3, p < 0.006).
Experiment II
Alcohol (10%) and food consumption were measured [n = 18, data in Table 2 and Figure 2].
Table 2.
Effects of SB-334867 on food, water, and 10% alcohol intake (two bottle preference) at 1, 2, 4, and 24 h time period
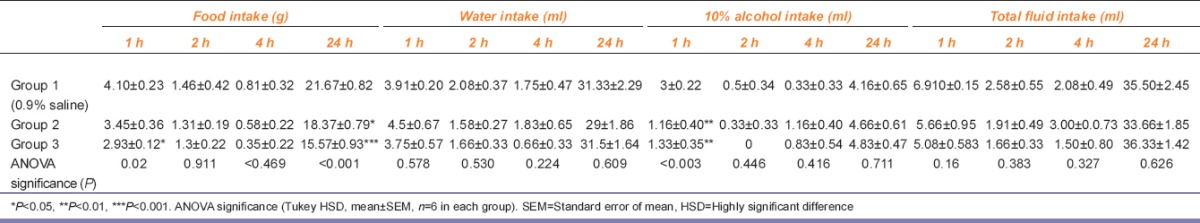
Figure 2.
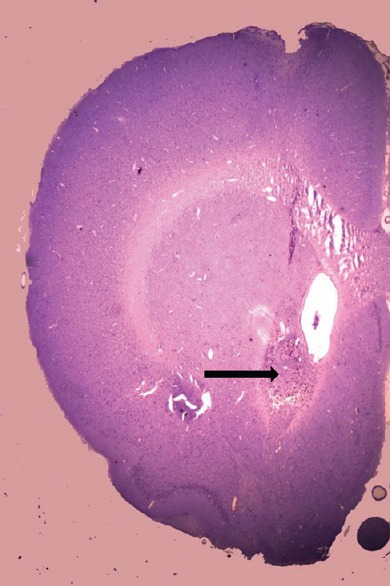
Histological section of injected site: Cresyl Violet stained section (7 μ) of rat brain showing infusion site (black arrow) (×2.5)
NAcc cannulated rats were divided into three subgroup, Group 1 (0.9% saline n = 6), Group 2 (SB-334867-3 ng, n = 6), and Group 3 (SB-334867-6 ng, n = 6).
10% alcohol intake results
At 1 h and 2 h SB-334867 treatment significantly attenuated alcohol consumption at 1st h (F[2, 15] = 4.457 p = 0.030), (Group 1 vs. Group 3, p < 0.004), 2nd h (F[2, 15] = 11.122 p = 0.001) (Group 1 vs. Group 3, p < 0.001; Group 2 vs. Group 3, p < 0.038). However, there was no significant change in alcohol consumption at 4 h (F[2, 15] = 0.709 p = 0.508) and 24 h (F[2, 15] = 2.631 p = 0.105) intervals, respectively.
Food intake
At 1 h and 2 h SB-334867 treatment significantly (F[2, 15] = 4.230 p = 0.035) attenuated food intake (Group 1 vs. Group 3, p < 0.03); (F[2, 15] = 16.558 p = 0.000) (Group 1 vs. Group 2, p < 0.000; Group 2 vs. Group 3, p < 0.021), respectively, compared to control. No significant changes were noticed at 4 h (F[2, 15] = 0.070 p = 0.933). Whereas, total food intake was decreased (0–24 h) (F[2, 15] = 4.457 p = 0.030) (Group 1 vs. Group 3, p < 0.025).
Experiment III
Food, 10% alcohol, and water [two bottle preference, Table 2] consumption were measured. NAcc cannulated rats were sub grouped as Group 1 (0.9% saline, n = 6), Group 2 (SB-334867-3 ng, n = 6), and Group 3 (SB-334867-6 ng, n = 6), were injected.
Food intake
SB-334867 treatment at 1 h (F[2, 15] = 5.111, p = 0.02) attenuated food intake (Group 1 vs. Group 3, p < 0.011) but no significant difference was observed at 2 h, 4 h (F[2, 15] = 0.093 p = 0.911), (F[2, 15] = 0.797 p = 0.469), respectively, and at 24 h both dose of antagonist showed decrease in food intake (F[2, 15] = 12.698 p = 0.001) (Group 1 vs. Group 2 and Group 3, p < 0.039, p < 0.000, respectively), compared to control group.
Water intake
SB-334867 treatment produced no change in water intake in any group with time interval at 1 h (F[2, 15] = 0.584 p = 0.578), 2 h (F[2, 15] = 0.662 p = 0.530), 4 h(F[2, 15] = 1.655 P = 0.224) and 24 h (F[2, 15] = 0.513 p = 0.609).
Alcohol (10%) intake
SB-334867 treatment attenuated alcohol intake at 1 h (F[2, 15] = 9.098 p = 0.003), (Group 1 vs. Group 2 and Group 3, p <0.004, p < 0.008, respectively). No significance change in any group at 2 h (F[2, 15] = 0.854 p = 0.446), 4 h (F[2, 15] = 0.931 p = 0.416) and 24 h (F[2, 15] = 0.349 p = 0.711), respectively.
Total fluid intake
No significant change in any of the groups, at 1st h (F[2, 15] = 2.064 p = 0.161), 2nd h (F[2, 15] =1.023 p = 0.383), 4th h (F[2, 15] =1.205 p = 0.327) and 24 h (F[2, 15] =0.484, p = 0.626).
Discussion
Modulation of food and water intake by various neurochemicals has been under scrutiny. Among several candidate molecules which have been shown to affect the feeding behavior, including alcohol intake, Orexins also are implicated.[19] Orexins were originally believed to be stimulants of the food intake and regulation of ingestive activity; later they were found to affect the sleep and wakeful states.[20] Dube et al. have demonstrated that central administration of Orexins has modulatory role in ingestive behavior, primarily centered in hypothalamus.[21] In our present experiment, we tested the effect of Orexin A antagonist (SB-334867) on NAcc. NAcc has been assigned a pivotal role in addiction and feeding-related activities.[22] Orexins also have been implicated in mediation of this action of NAcc.[4] However, NAcc showed two histologically distinct districts,[23] which may have functional differences[24] and their functions appeared to overlap significantly.[25] In our previous experiments, we found that infusion of Orexin A into NAcc using microinjection technique increased food and water intake in the hours immediately after infusion, but there was no particular preference to alcohol when tested by two bottle free choice.[14] Therefore, we attempted to inject Orexin A antagonist into NAcc and analyzed the food intake, water intake and alcohol intake in rats, which were fasted overnight.
The consumption in 1st h declined in orexin antagonist-treated animals significantly. Our study further proves the role of Orexin A in feeding behavior. Orexin receptor type 1 (OX1R) antagonist SB-334867 infusion attenuates the feeding and drinking. Orexin A consistently showed the stimulatory effect on feeding and drinking. OX1R antagonist has 10 times greater affinity for Orexin A than B.[26,27]
Orexinergic neurons project to the AccSh and both orexin receptors (OX1R and OX2R) are present in the NAcc, with OX2R expressed to a greater extent.[28,29] Orexin A increased GABAergic currents and decreased N-methyl-D-aspartate currents in isolated accumbens neurons.[30] In addition, orexins excite dopaminergic VTA neurons.[31] Since dopaminergic VTA neurons innervate and excite AccSh GABAergic (inhibitory) neurons, orexin signaling could further increase local inhibition in the Acc by increasing neuronal activity in the VTA, resulting in further enhancement of consummatory behavior. But this was contradicted by Baldo and Kelley,[32] who found no effect on feeding or locomotor activity with intra-AccSh Orexin A.
We tested the possibility of Orexin A acting as a modulator of alcohol consumption [Table 2] along with food. From our previous study, we established that rats preferred to consume alcohol at 10% solution which has been confirmed in our own previous study.[33] Therefore in this study, we provided alcohol in this dilution following the infusion of drug into NAcc. We found a significant decrease in the alcohol intake in the hours immediately following the infusion of Orexin A antagonist. The decline in food and water intake was lower in the low dose (3 ng) infusion group while it was higher in the high dose (6 ng) group. In order to test the alcohol preference, we provided the rats with two bottle choice condition, where one bottle of water and another containing 10% alcohol were provided simultaneously. Following infusion of the Orexin A antagonist produced a significant decline in food intake and alcohol intake. This type of decline was found both in low and high dose but restricted to 1st h after the infusion. However, the decline in food intake was more marked compared to the water or alcohol. This evidence gives support for the involvement of the Orexin A in the regulation of either food intake but does not support the possibility of Orexin A involvement in the preference for alcohol.
Financial Support and Sponsorship
Department of Biotechnology, Part of DBT funded project, Ref: Ref: BT/PR14012/MED/30/315/2010 dated 30.09.2010 Government of India.
Conflicts of Interest
There are no conflicts of interest.
Acknowledgements
The authors are thankful to the Department of Biotechnology, Government of India, for the financial support. Kasturba Medical College, Mangalore, Manipal University, for the facilities provided.
References
- 1.Trojniar W, Plucinska K, Ignatowska-Jankowska B, Jankowski M. Damage to the nucleus accumbens shell but not core impairs ventral tegmental area stimulation-induced feeding. J Physiol Pharmacol. 2007;58(Suppl 3):63–71. [PubMed] [Google Scholar]
- 2.Marty VN, Spigelman I. Long-lasting alterations in membrane properties, KC currents, and glutamatergic synaptic currents of nucleus accumbens medium spiny neurons in a rat model of alcohol dependence. Front Neurosci. 2012;6:86. doi: 10.3389/fnins.2012.00086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Henderson MB, Green AI, Bradford PS, Chau DT, Roberts DW, Leiter JC. Deep brain stimulation of the nucleus accumbens reduces alcohol intake in alcohol-preferring rats. Neurosurg Focus. 2010;29:E12. doi: 10.3171/2010.4.FOCUS10105. [DOI] [PubMed] [Google Scholar]
- 4.Stratford TR, Kelley AE. GABA in the nucleus accumbens shell participates in the central regulation of feeding behavior. J Neurosci. 1997;17:4434–40. doi: 10.1523/JNEUROSCI.17-11-04434.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hernandez L, Lee F, Hoebel BG. Simultaneous microdialysis and amphetamine infusion in the nucleus accumbens and striatum of freely moving rats: Increase in extracellular dopamine and serotonin. Brain Res Bull. 1987;19:623–8. doi: 10.1016/0361-9230(87)90047-5. [DOI] [PubMed] [Google Scholar]
- 6.Hernandez L, Hoebel BG. Feeding and hypothalamic stimulation increase dopamine turnover in the accumbens. Physiol Behav. 1988;44:599–606. doi: 10.1016/0031-9384(88)90324-1. [DOI] [PubMed] [Google Scholar]
- 7.Maejima Y, Sakuma K, Santoso P, Gantulga D, Katsurada K, Ueta Y, et al. Oxytocinergic circuit from paraventricular and supraoptic nuclei to arcuate POMC neurons in hypothalamus. FEBS Lett. 2014;588:4404–12. doi: 10.1016/j.febslet.2014.10.010. [DOI] [PubMed] [Google Scholar]
- 8.Ganaraja B, Jeganathan PS. Effect of basolateral amygdala and ventromedial hypothalamic lesions on ingestion and taste preference in rat. Indian J Med Res. 2000;112:65–70. [PubMed] [Google Scholar]
- 9.Narayanan NS, Guarnieri DJ, DiLeone RJ. Metabolic hormones, dopamine circuits, and feeding. Front Neuroendocrinol. 2010;31:104–12. doi: 10.1016/j.yfrne.2009.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wise RA. Dopamine, learning and motivation. Nat Rev Neurosci. 2004;5:483–94. doi: 10.1038/nrn1406. [DOI] [PubMed] [Google Scholar]
- 11.Koob GF. Neurocircuitry of alcohol addiction: Synthesis from animal models. Handb Clin Neurol. 2014;125:33–54. doi: 10.1016/B978-0-444-62619-6.00003-3. [DOI] [PubMed] [Google Scholar]
- 12.Sakurai T, Amemiya A, Ishii M, Matsuzaki I, Chemelli RM, Tanaka H, et al. Orexins and orexin receptors: A family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell. 1998;92:573–85. doi: 10.1016/s0092-8674(00)80949-6. [DOI] [PubMed] [Google Scholar]
- 13.Liu Y, Zhao Y, Ju S, Guo L. Orexin A upregulates the protein expression of OX1R and enhances the proliferation of SGC-7901 gastric cancer cells through the ERK signaling pathway. Int J Mol Med. 2015;35:539–45. doi: 10.3892/ijmm.2014.2038. [DOI] [PubMed] [Google Scholar]
- 14.Mayannavar S, Rashmi KS, Rao YD, Yadav S, Ganaraja B. Effect of orexin-A infusion in to the nucleus accumbens on consummatory behaviour and alcohol preference in male Wistar rats. Indian J Physiol Pharmacol. 2014;58:319–26. [PubMed] [Google Scholar]
- 15.Yamada H, Okumura T, Motomura W, Kobayashi Y, Kohgo Y. Inhibition of food intake by central injection of anti-orexin antibody in fasted rats. Biochem Biophys Res Commun. 2000;267:527–31. doi: 10.1006/bbrc.1999.1998. [DOI] [PubMed] [Google Scholar]
- 16.Smart D, Sabido-David C, Brough SJ, Jewitt F, Johns A, Porter RA, et al. SB-334867-A: The first selective orexin-1 receptor antagonist. Br J Pharmacol. 2001;132:1179–82. doi: 10.1038/sj.bjp.0703953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paxinos G, Watson C. London: Academic Press; 1998. The Rat Brain in Stereotaxic Coordinates. [Google Scholar]
- 18.Mayannavar S, Rashmi KS, Deshpande K, Pai SR, Ganaraja B. Preparation of durable cannula for intracranial micro-infusion of neuroactive substances in small animals. Int J Innov Res Sci Eng Technol. 2013;2:6032–8. [Google Scholar]
- 19.Willie JT, Chemelli RM, Sinton CM, Yanagisawa M. To eat or to sleep?. Orexin in the regulation of feeding and wakefulness. Annu Rev Neurosci. 2001;24:429–58. doi: 10.1146/annurev.neuro.24.1.429. [DOI] [PubMed] [Google Scholar]
- 20.Thorpe AJ, Kotz CM. Orexin A in the nucleus accumbens stimulates feeding and locomotor activity. Brain Res. 2005;1050:156–62. doi: 10.1016/j.brainres.2005.05.045. [DOI] [PubMed] [Google Scholar]
- 21.Dube MG, Kalra SP, Kalra PS. Food intake elicited by central administration of orexins/hypocretins: Identification of hypothalamic sites of action. Brain Res. 1999;842:473–7. doi: 10.1016/s0006-8993(99)01824-7. [DOI] [PubMed] [Google Scholar]
- 22.Kelley AE. Ventral striatal control of appetitive motivation: Role in ingestive behavior and reward-related learning. Neurosci Biobehav Rev. 2004;27:765–76. doi: 10.1016/j.neubiorev.2003.11.015. [DOI] [PubMed] [Google Scholar]
- 23.Salgado S, Kaplitt MG. The Nucleus Accumbens: A Comprehensive Review. Stereotact Funct Neurosurg. 2015;93:75–93. doi: 10.1159/000368279. [DOI] [PubMed] [Google Scholar]
- 24.Ramaswamy C, Ghosh S, Vasudev R. Alteration of preference of food substances in terms of taste and nutritional value following the lesion of two subdistricts of nucleus accumbens. Indian J Med Res. 1998;108:139–44. [PubMed] [Google Scholar]
- 25.Stratford TR, Kelley AE. GABA in the nucleus accumbens shell participates in the central regulation of feeding behavior. J Neurosci. 1997;17:4434–40. doi: 10.1523/JNEUROSCI.17-11-04434.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Haynes AC, Jackson B, Overend P, Buckingham RE, Wilson S, Tadayyon M, et al. Effects of single and chronic intracerebroventricular administration of the orexins on feeding in the rat. Peptides. 1999;20:1099–105. doi: 10.1016/s0196-9781(99)00105-9. [DOI] [PubMed] [Google Scholar]
- 27.Muroya S, Funahashi H, Yamanaka A, Kohno D, Uramura K, Nambu T, et al. Orexins (hypocretins) directly interact with neuropeptide Y, POMC and glucose-responsive neurons to regulate Ca 2+signaling in a reciprocal manner to leptin: Orexigenic neuronal pathways in the mediobasal hypothalamus. Eur J Neurosci. 2004;19:1524–34. doi: 10.1111/j.1460-9568.2004.03255.x. [DOI] [PubMed] [Google Scholar]
- 28.Cluderay JE, Harrison DC, Hervieu GJ. Protein distribution of the orexin-2 receptor in the rat central nervous system. Regul Pept. 2002;104:131–44. doi: 10.1016/s0167-0115(01)00357-3. [DOI] [PubMed] [Google Scholar]
- 29.Lu XY, Bagnol D, Burke S, Akil H, Watson SJ. Differential distribution and regulation of OX1 and OX2 orexin/hypocretin receptor messenger RNA in the brain upon fasting. Horm Behav. 2000;37:335–44. doi: 10.1006/hbeh.2000.1584. [DOI] [PubMed] [Google Scholar]
- 30.Martin G, Fabre V, Siggins GR, de Lecea L. Interaction of the hypocretins with neurotransmitters in the nucleus accumbens. Regul Pept. 2002;104:111–7. doi: 10.1016/s0167-0115(01)00354-8. [DOI] [PubMed] [Google Scholar]
- 31.Nakamura T, Uramura K, Nambu T, Yada T, Goto K, Yanagisawa M, et al. Orexin-induced hyperlocomotion and stereotypy are mediated by the dopaminergic system. Brain Res. 2000;873:181–7. doi: 10.1016/s0006-8993(00)02555-5. [DOI] [PubMed] [Google Scholar]
- 32.Baldo BA, Kelley AE. Amylin infusion into rat nucleus accumbens potently depresses motor activity and ingestive behavior. Am J Physiol Regul Integr Comp Physiol. 2001;281:R1232–42. doi: 10.1152/ajpregu.2001.281.4.R1232. [DOI] [PubMed] [Google Scholar]
- 33.Mayannavar SK, Shiva RK, Aithal K, Bhat RM, Ganaraja B. Effect of bilateral lesions of the nucleus accumbens on consummatory behaviour in Wistar rats. J Pharm Res. 2013;7:263–6. [Google Scholar]


