Abstract
Background
Despite the efficacy of decitabine treatment in myelodysplastic syndrome (MDS), no definite predictor of response is known. In this study, we investigated whether the expression levels of human equilibrative nucleoside transporter 1 (hENT1), hENT2, deoxycytidine kinase (DCK) and cytidine deaminase (CDA) genes could predict response to decitabine in MDS.
Methods
We performed quantitative real-time PCR in marrow mononuclear cells to examine the expression of hENT1, hENT2, DCK, and CDA prior to therapy in 98 MDS patients initially treated with decitabine. Response and overall survival of patients treated with decitabine were analyzed according to gene expression levels. HENT1 knockdown was performed by shRNA in the SKM-1 cell line, and the effect of this on the demethylation ability of decitabine on long interspersed nucleotide element 1 (LINE1) was investigated.
Results
Patients responding to decitabine presented with significantly higher hENT1 expression levels than non-responders (p = 0.004). Overall response, complete response, and cytogenetic complete response rate in patients with high hENT1 expression (79.4, 41.3, and 43.8 %) were significantly higher than those in patients with low hENT1 expression (48.6, 20.0, and 5.9 %, respectively) (p = 0.004, 0.033, and 0.006, respectively). In higher-risk MDS, patients with high hENT1 expression showed prolonged survival compared with those with low hENT1 expression (22.0 vs 14.0 months; p = 0.027). However, the expression levels of hENT2, DCK, and CDA did not affect response rate. Knockdown of hENT1 in SKM-1 cells weakened the demethylation effect on LINE1 induced by decitabine.
Conclusions
High expression of hENT1 appears to predict a good response to decitabine and a prolonged survival in higher-risk MDS patients treated with decitabine. HENT1 expression knockdown weakens the demethylation effect of decitabine.
Electronic supplementary material
The online version of this article (doi:10.1186/s12967-016-0817-9) contains supplementary material, which is available to authorized users.
Keywords: Myelodysplastic syndrome, Decitabine, Human equilibrative nucleoside transporter 1, Long interspersed nuclear element
Background
Myelodysplastic syndromes (MDS) are recognized as among the most common hematological neoplasms [1], comprising a heterogeneous group of clonal hematopoietic stem/progenitor cell disorders characterized by ineffective hematopoiesis, dysplastic cell morphology, and the potential for clonal evolution [2]. Approximately 50–60 % MDS patients have abnormal karyotypes, frequently with 5q-, +8, −7(q) or 20q- [1]. Decitabine is one of the drugs approved for treatment of MDS, and low-dose decitabine has been proven to be effective for some MDS patients [3, 4]. However, a cohort of patients do not respond to this treatment initially [4, 5], and there are almost no effective factors predicting which patients will respond to decitabine before starting the therapy.
It is likely that genetic variability of key enzymes in decitabine transport and metabolism may have an impact on the treatment response of decitabine. Thus, the expression levels of genes related to decitabine transport [including human equilibrative nucleoside transporter 1 (hENT1) and hENT2], and metabolism [including deoxycytidine kinase (DCK) and cytidine deaminase (CDA)] may influence the therapeutic effect of the drug.
Because the biochemical targets of decitabine are intracellular, the mandatory first step in the production of cytotoxicity is permeation across the cell membrane. Decitabine and physiologic nucleosides are hydrophilic, and diffusion through the cell membrane lipid layer is slow. Therefore, efficient cellular uptake requires the presence of specialized integral membrane nucleoside transporter proteins [6]. Two general processes of nucleoside transport have been identified: the equilibrative bi-directional facilitators (hENT1 and 2) and the concentrative sodium/nucleoside symporters hCNT1 and 3. The major routes for transporting decitabine are hENT1 and hENT2, and, to a lesser extent, hCNT1 and hCNT3 [6]. When transporting into the cell, decitabine is phosphorylated into monophosphorylated derivative 5-azadCMPby deoxycytidine (DCK). Then, 5-aza-dCMP is phosphorylated to 5-aza-dCTP, which is the active form of decitabine and incorporated into DNA, where it acts by demethylation. Catabolizing enzymes such as cytidine deaminase (CDA) can catalyze decitabine metabolites to uridine and deoxyuridine, therefore decreasing the amount of 5-aza-dCTP that can be formed.
In solid tumors such as pancreatic ductal adenocarcinoma, hENT1 expression was used to identify subgroups of patients with different disease progression risk, while DCK expression was shown to be most relevant to mortality risk [7]. In cancer cell lines, those most resistant to decitabine had a combination of low expression of DCK, hENT1 and high CDA expression [8]. Furthermore, a high CDA/DCK ratio has been reported to predict response to decitabine [9]. A recent study by Wu et al. [10] showed that high hENT1 expression is a predictor for response to decitabine in MDS. However, the samples involved in their study were insufficiently large, and the effect of hENT1 expression on patient survival was not addressed. Furthermore, to our knowledge, no in vitro studies have shown the direct effect of hENT1 expression on the demethylation of decitabine in MDS cells.
Here, we enrolled a total of 98 patients treated with decitabine to further determine whether hENT1, hENT2, DCK, and CDA can affect the response and prognosis in MDS patients treated with decitabine. We also investigated the alteration of hypomethylation and the subsequent biological actions induced by decitabine in the SKM-1 cell line following knockdown of hENT1 expression.
Methods
Patients
All patients enrolled in this study had been diagnosed with MDS at our department according to French–American–British (FAB)/World Health Organization (WHO) classification criteria [11, 12]. Bone marrow (BM) samples were obtained at diagnosis of MDS before the initiation of any treatment between September 2009 and December 2013 following approval by the local ethics committee and according to institutional guidelines. Each of the 98 MDS patients treated with decitabine provided written informed consent. The characteristics of the MDS patients are listed in Table 1. As of December 2014, the median follow-up period was 24 months (range 2–62 months). This study was approved by the Ethics Committee of Shanghai Jiao Tong University Affiliated Sixth People’s Hospital.
Table 1.
Characteristics of the 98 MDS patients treated with decitabine
| Characteristics | Category |
|---|---|
| Median age (range) | 62 (20–82) |
| Sex, n (%) | |
| Male | 69 (70) |
| Female | 29 (30) |
| WHO/FAB classification, n (%) | |
| RCMD | 19 (20) |
| RAS | 2 (3) |
| RAEB-1 | 25 (22) |
| RAEB-2 | 29 (30) |
| RAEB-t | 14 (17) |
| CMML | 9 (8) |
| Karyotypes, n(%) | |
| Normal | 49 (50) |
| +8 | 7 (7) |
| −7/7q- | 9 (9) |
| −5/5q- | 2 (2) |
| 20q- | 5 (5) |
| Complex | 13 (13) |
| Others | 13 (13) |
| International prognostic scoring system, n (%) | |
| Low | 4 (4) |
| Intermediate-1 | 33 (34) |
| Intermediate-2 | 34 (35) |
| High | 27 (27) |
MDS myelodysplastic syndrome, FAB French-American-British, WHO world health organization, RCMD refractory cytopenia with multilineage dysplasia, RAS refractory anemia with ringed sideroblasts, RAEB refractory anemia with excess of blasts, RAEB-t refractory anemia with excess of blasts in transformation, CMML chronic myelomonocytic leukemia
Real-time quantitative polymerase chain reaction
Total RNA was extracted from 2 ml of BM mononuclear cells (BMNCs) using the RNeasy Mini Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. cDNA was synthesized by random priming from 10 µL of total RNA using the RevertAid™ First Strand cDNA Synthesis Kit (Fermentas, Burlington, Canada) according to the manufacturer’s instructions. PCR was performed in a fluorescent quantitation PCR cycler (LightCycler, Roche, Switzerland) in a final volume of 10 μL, including 1 µL of cDNA, 0.5 μM of each primer, 4 mM MgCl2, and 2 μL of the supplied enzyme mix containing the reaction buffer, FastStart Taq DNA polymerase, and DNA double-strand-specific SYBR Green I dye for PCR product detection. Primer sequences were as follows: hENT1 (NM_001078177.1): 5′-TCTCCAACTCTCAGCCCACCAA-3′ (sense) and 5′-CCTGCGATGCTGGACTTGACCT-3′ (antisense); hENT2 (NM_001532.2): 5′-ACCATGCCCTCCACCTACAG-3′ (sense), 5′-GGGCCTGGGATGATTTATTG-3′ (antisense); DCK (NM_000788.2): 5′-GGCCGCCACAAGACTAAGGA-3′ (sense) and 5′-CACCATCTGGCAACAGGTTCA-3′ (antisense); and CDA (NM_001785.2): 5′-CCTGCAGGCAAGTCATGAGAG-3′ (sense); 5′-ACCATCCGGCTTGGTCATGTA-3′ (antisense). The primer sequences for GAPDH were as follows: 5′-GCACCGTCAAGGCTGAGAAC-3′ (sense) and 5′-GTGGTGAAGACGCCAGTGGA-3′ (antisense). PCR conditions were as follows: preincubation at 95 °C for 30 s, then 40 cycles of 95 °C for 15 s, 62 °C for 30 s, and 72 °C for 30 s. The threshold cycle (Ct) value was subsequently determined, and the relative quantification of mRNA expression was calculated using the comparative Ct method.
The relative quantification value of the target, which was normalized to that of an endogenous control (GAPDH) and relative to that of a calibrator (the mean expression level of normal controls), was expressed as 2−ΔΔCt (fold difference) where ΔCt = Ct of the target gene-Ct of the endogenous control gene (GAPDH) and ΔΔCt = ΔCt of the samples for the target gene −ΔCt of the calibrator for the target gene [13]. To analyze the characteristics of target gene expression levels in MDS treated with decitabine, the mean expression level of target genes in BM from 20 normal donors (median age, 60 years; male: female 2:1) was used as a control. Based on control mean expression levels of each gene, we divided the enrolled patients into two groups: a high gene expression group with 2−ΔΔCt values above mean levels, and a low gene expression group with 2−ΔΔCt values below mean levels.
Decitabine treatment
All patients were treated with decitabine (Johnson and Johnson Inc., New Brunswick, NJ), which was administered at a dose of 20 mg/m2 by continuous intravenous infusion for 1 h, and repeated daily for 5 days. The cycle was repeated every 4 weeks, depending on each patient’s recovery from myelosuppression. BM examinations were performed 4 weeks after decitabine treatment was completed to evaluate the response. The final treatment response was assessed after at least four cycles of decitabine therapy, except for patients who discontinued therapy because of disease progression when receiving fewer than four cycles of therapy.
Responses to decitabine therapy and overall survival
The primary observation parameter was the overall response rate (OR), which included the rates of complete remission (CR), marrow CR (mCR), hematologic improvements (HI), and cytogenetic CR (CCR) (only in patients with abnormal karyotypes before therapy). The treatment response was assessed using modified International Working Group (IWG 2006) response criteria [14]. Overall survival (OS) was calculated from the initial date of decitabine treatment to the date of death from any cause.
Cell line and culture
Decitabine was purchased from Sigma-Aldrich (St Louis, MO), dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich) at 10 mM, and stored at −20 °C. SKM-1 cells (Health Science Research Resources Bank, Osaka, Japan) were grown in RPMI-1640 medium (Gibco, Grand Island, NY) containing 10 % heat-inactivated fetal bovine serum, 100 IU/ml penicillin, and 100 µg/ml streptomycin in a humidified atmosphere of 5 % CO2 at 37 °C. Cells were seeded in 12-well plates at a density of 5 × 105 cells in 2 ml culture medium and treated with DMSO or decitabine every 24 h. After 24, 48, and 72 h, cells were harvested and DNA was isolated using DNA reagent (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions. As a solvent control, DMSO was added to a final concentration of 0.01 %.
Lentivirus-delivered small interfering (si)RNA gene knockdown
The siRNA sequence for hENT1 knockdown was 5′- ACCAATGAAAGCCACTCTA-3′, and the scrambled control siRNA sequence was 5′-TTCTCCGAACGTGTCACGT-3′. The siRNA sequences were cloned into the pLSLG lentiviral vector. This and the help vector (pLV-HELP) were transfected into 293FT cells for viral packaging. Virus was then collected for the infection of SKM-1 cells in the presence of 5 μg/ml polybrene (Genechem, Shanghai, China). After 4 days of culture, cells were used for further experiments.
Pyrosequencing to measure LINE-1 methylation
Genomic DNA was extracted using DNeasy Blood & Tissue Kit (QIAGEN, Hilden, Germany) according to the manufacturer’s instructions and then modified by treatment with sodium bisulfite using the Zymo EZ DNA Methylation kit (Zymo Research, Irvine, CA) following the manufacturer’s protocol. A modified method of PCR-based pyrosequencing was performed to quantify methylation of LINE-1 repetitive elements, as previously described [15]. The primers used in PCR were: forward primer (5′-TTTTTTGAGTTAGGTGTGGGATA-3′) and biotinylated reverse primer (Biotin-AAAAATCAAAAAATTCCCTTTCC-3′). PCR cycling conditions were: 45 cycles of 95 °C for 15 s, 50 °C for 20 s, and 72 °C for 30 s. The PCR product was bound to streptavidin Sepharose beads (Amersham Biosciences, Uppsala, Sweden) and then purified. The LINE-1-S3 pyrosequencing primer (5′-GGGTGGGAGTGAT-3′) was annealed to the purified single-stranded PCR product. Pyrosequencing was performed using the PSQ HS 96 Pyrosequencing System. The average relative amount of cytosine in the three CpG sites was used as the overall LINE-1 methylation level.
Statistical methods
Statistical analyses were performed using the Statistical Package for Social Sciences (SPSS 16.0; SPSS Inc., Chicago, IL). Two independent sample populations were compared using the Student’s t test. The Pearson χ2 or Fisher’s exact test was applied to compare the enumeration data between groups. The Kaplan–Meier method was used to estimate median OS. The log-rank test was used to compare Kaplan–Meier survival estimates between groups. Multivariate Cox proportional hazards models were used to calculate the hazard ratios and 95 % confidence intervals of the associations between risk factors and survival. p values <0.05 were considered statistically significant.
Results
Patient characteristics
A total of 98 patients were enrolled in this study. The median patient age was 62 years (range 20–82 years), and the male to female ratio was 2.38:1 (69:29). All patients in this study were diagnosed with de novo MDS. The FAB/WHO disease sub-types at study entry were refractory cytopenia with multilineage dysplasia (RCMD; n = 19), refractory anemia with ringed sideroblasts (RAS; n = 2), refractory anemia with excess blasts (RAEB-1; n = 25 and RAEB-2; n = 29), refractory anemia with excess blasts in transformation (RAEB-t; n = 14), and chronic myelomonocytic leukemia (CMML) (n = 9). The International Prognostic Scoring System (IPSS) [16] risk category was low in four, intermediate (INT)-1 in 33, INT-2 in 34, and high risk in 27 patients. A total of 49 patients (50.0 %) with abnormal karyotypes were determined by G-binding analysis [17]. Patient characteristics are listed in Table 1. The median number of decitabine therapy cycles was four (range 2–10). Eight patients discontinued treatment after two cycles of decitabine therapy because of disease progression. All patients were evaluable.
HENT1 expression levels were significantly elevated in responders to decitabine therapy
Among the 98 MDS patients, 67 (68.4 %) achieved a response, including 33 (33.7 %) with CR, 11 (11.2 %) with mCR only, 15 (15.3 %) with HI only, and eight (8.2 %) with mCR with HI. Thirty-one (31.6 %) patients showed no response to decitabine therapy. Among the 49 patients with abnormal karyotypes, 15 (30.6 %) reached CCR. Patients responding to decitabine showed significantly increased mean hENT1 expression levels compared with non-responders (1.79 ± 1.53-fold vs 0.94 ± 0.77-fold, p = 0.004) (Fig. 1a). Patients who achieved CR with decitabine also showed increased hENT1 expression levels compared with those who did not achieve CR (2.05 ± 1.86-fold vs 1.25 ± 1.00-fold, p = 0.007) (Fig. 1b). Increased hENT1 expression was also detected in patients who achieved CCR when compared with those who did not (2.97 ± 1.87-fold vs 1.16 ± 1.05-fold, p < 0.001) (Fig. 1c). HENT1 expression level also increased in patients achieved mCR or HI when compared to that in NR patients (Additional file 1). Expression levels of hENT2, DCK, and CDA did not affect response including (OR, CR, CCR, mCR or HI) rate in MDS patients treated with decitabine (Fig. 1a–c; Additional file 1a, b).
Fig. 1.
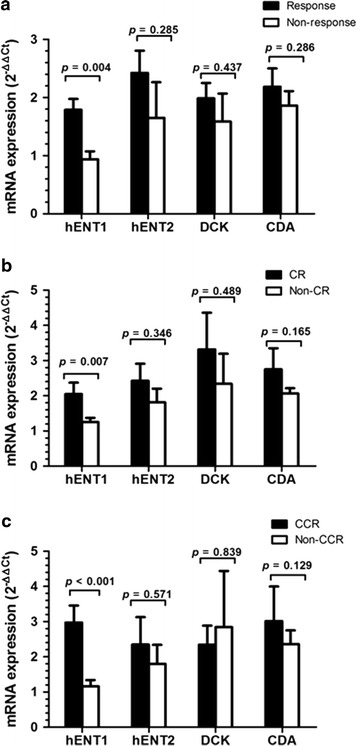
Comparison of hENT1, hENT2, DCK, and CDA expression levels between responders and non-responders; CR and non-CR; CCR and non-CCR. a Increased hENT1 in responders compared to non-responders. hENT2, DCK, and CDA mRNA expression levels did not differ between responders and non-responders. b Elevated hENT1 level in CR patients compared to non-CR patients. No significant difference of hENT2, DCK, and CDA mRNA level between CR and non-CR patients. c Increased hENT1 rather than hENT2, DCK, or CDA level in CCR patients compared to non-CCR patients
High expression of hENT1 confers response to decitabine therapy
The mean expression level of hENT1, hENT2, DCK, and CDA of 20 normal donors was used as a control. Compared with these, a total of 63 patients (64.3 %) showed high hENT1 expression and 35 (35.7 %) showed low hENT1 expression. Among the 63 high hENT1 expression patients, 50 (79.4 %) achieved a response, which was a significantly higher proportion than the 17/35 (48.6 %) low hENT1 expression patients (p = 0.004). Twenty-six of 63 (41.3 %) hENT1 high expression patients achieved CR, which was significantly higher than that of low hENT1 expression patients (7/35, 20.0 %) (p = 0.033). There was no difference in mCR or HI rate between the high and low hENT1 expression groups.
Among the 49 patients with abnormal karyotype, the CCR rate was significantly higher in the hENT1 high expression group (14/32, 43.8 %) than the low hENT1 expression group (1/17, 5.9 %) (p = 0.006) (Fig. 2a). There was no significant difference in OR, CR, CCR, mCR, or HI between patients with high and low hENT2 expression (Fig. 2b), high and low DCK (Fig. 2c), high and low CDA (Fig. 2d), or between the high and low CDA/DCK ratio (Fig. 2e).
Fig. 2.
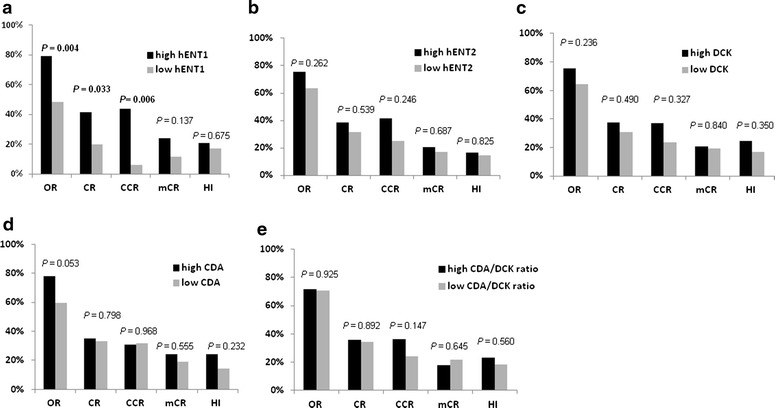
Comparison of response rate in patients treated with decitabine between those with a high and low hENT1 expression, b high and low hENT2 expression, c high and low DCK expression, d high and low CDA expression, and e high and low CDA/DCK ratio
Fewer treatment cycles were needed for patients with high hENT1 expression to achieve CR
We recorded the number of cycles needed for patients treated with decitabine to achieve CR. The median number of administered cycles was three (range 1–4) for all patients who achieved CR. For patients with high hENT1 expression levels, significantly fewer cycles (median, two (range 1–4) cycles) were needed to achieve CR compared with low hENT1 expression patients (median, three (range 2–4) cycles) (p = 0.035).
High hENT1 expression predicted prolonged survival in higher-risk MDS patients receiving decitabine treatment
During a median follow-up period of 24.0 months (range 2–62 months), the median OS of 98 patients was 18.0 months [95 % confidence interval (CI): 14.8–21.1 months]. The median OS between all MDS patients with high and low hENT1 expression was not significantly different (23.0 vs 17.0 months; p = 0.207) (Fig. 3a). However, among the 61 higher-risk (IPSS int-2 or high-risk) patients, the median OS of patients with high hENT1 expression was significantly longer than that of low hENT1 expression patients (22.0 vs 14.0 months; p = 0.027) (Fig. 3b). Univariate analysis showed hENT1 was a prognosis predictor in higher risk MDS patients treated with decitabine (p = 0.027). Furthermore, multivariate Cox analyses were conducted by integrating several risk factors including hemoglobin, platelet, BM blast, hENT1 expression level. In the multivariate analyses, low hENT1 [hazard ration (HR) = 2.363; 95 % confidence interval (CI) = 1.174–4.757; Likelihood ratio test, p = 0.031] appeared to be independent prognostic markers of adverse events in higher risk MDS treated with decitabine (Table 2).
Fig. 3.
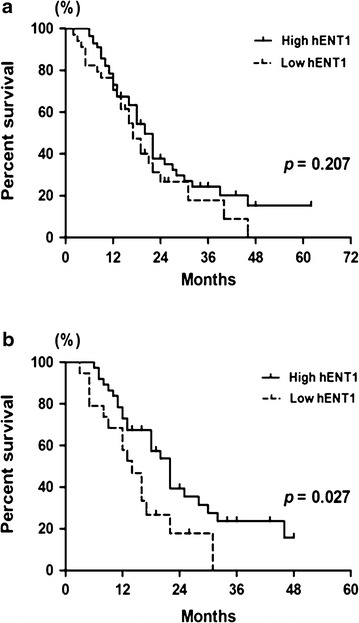
Comparison of overall survival of patients with high and low hENT1 expression treated with decitabine. a In all MDS patients, the median overall survival time was not significantly different between high hENT1 patients and low hENT1 patients. b In the higher-risk patients, a prolonged overall survival time was observed in patients with high hENT1 compared to those with low hENT1
Table 2.
Univariate and Multivariate analysis for survival in higher risk MDS patients treated with decitabine
| Criterion | Univariate P value | Multivariate P value | HR | 95.0 % CI for HR |
|---|---|---|---|---|
| Hemoglobin (>80 g/L vs ≤80 g/L) | 0.010 | 0.026 | 2.023 | 1.177–4.112 |
| Platelet (>50 × 109/L vs ≤50 × 109/L) | 0.008 | 0.022 | 2.134 | 1.113–4.091 |
| Bone marrow blasts percentage (5–10/10–20/>20 %) | 0.010 | 0.041 | 1.522 | 1.017–2.277 |
| hENT1 expression (high vs low) | 0.027 | 0.031 | 2.363 | 1.174–4.757 |
| Age (>60 years vs ≤60 years) | 0.796 | – | – | – |
| Gender (male vs female) | 0.569 | – | – | – |
| Neutrophils (>1.0 × 109/L vs ≤1.0 × 109/L) | 0.212 | – | – | – |
| Karyotype (low/intermediate/high) | 0.318 | – | – | – |
| hENT2 expression (high vs low) | 0.201 | – | – | – |
| DCK expression (high vs low) | 0.203 | – | – | – |
| CDA expression (high vs low) | 0.411 | – | – | – |
Lower expression of hENT1 in SKM-1 cells reduces the demethylation effect of decitabine
HENT1 mRNA expression levels were significantly decreased in hENT1-siRNA-lentivirus-transfected SKM-1 cells compared with control lentivirus-transfected SKM-1 cells (Fig. 4a). Before decitabine treatment, the LINE-1 methylation status did not differ significantly between hENT1-knockdown and control cells. However, after decitabine treatment (at final concentrations of 0.5, 1.0, and 5.0 µmol/l), LINE-1 methylation levels were lowered in both hENT1-knockdown and control cells. The lowest methylation status was achieved following treatment with 1.0 µmol/l decitabine in both groups of cells. However, compared with the control group, the demethylation effect of decitabine was significantly weakened in hENT1-knockdown SKM-1 cells (p = 0.0026) (Fig. 4b–f). The biggest gap of demethylation effect between the two groups was observed at a decitabine treatment of 0.5 µmol/l.
Fig. 4.
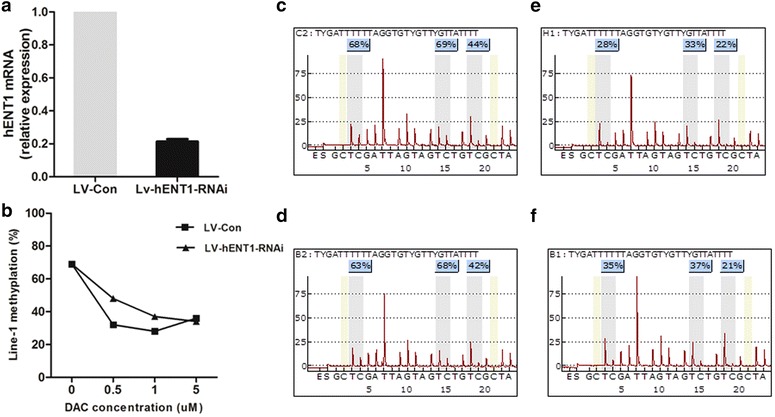
LINE-1 methylation level before and after decitabine treatment in hENT1-siRNA and control SKM-1 cells. a hENT1 mRNA expression in SKM-1 cells transfected with hENT1-siRNA-lentivirus and control lentivirus. b LINE-1 methylation levels changed with different concentrations of decitabine. c–f A pyrosequencing assay was used to measure LINE-1 methylation before decitabine treatment in control lentivirus-transfected (c) and hENT1-siRNA cells (d). LINE-1 methylation was also measured after 1 µM decitabine treatment in control (e) and hENT1-siRNA cells (f)
Discussion
Mechanisms of in vivo resistance to nucleoside analogs are complex and remain unresolved. One possibility may result from the presence of insufficient intracellular triphosphate, as seen for some nucleic acid analogs such as cytarabine and fludarabine [18].
Decitabine is administered to patients without knowledge of their genetic background, which may affect drug efficacy. The primary mechanism for insufficient intracellular concentrations of decitabine may result from a combination of multiple factors including inadequate uptake through membrane transporters, DCK deficiency, increased CDA, or high dNTP pools. HENT1 is the major membrane transporter for decitabine transporting into cell, which can affect the intracellular drug concentration and thus the efficacy of decitabine [19]. Studies both in cancer cell lines [8] and in MDS patients [9, 10] showed that resistant to decitabine had a low hENT1. In accordance with these findings, our results revealed significantly higher hENT1 expression levels in decitabine responders compared with non-responders. Moreover, in CR patients, even higher hENT1 levels were observed compared with non-CR patients. In patients with high hENT1 expression, significantly higher response rate and CR rate was observed, compared with those of low hENT1 expression patients. Therefore, high hENT1 expression predicts response to decitabine in MDS patients and a higher possibility to achieve CR.
For MDS patients with abnormal karyotypes, CCR means the deeper remission and prolonged relapse-free survival [20]. To our best knowledge, there is no effect indictor for predicting CCR in MDS patients received decitabine therapy. Of note, in the current study, results showed that patients achieved CCR had a significantly higher hENT1 expression, compared with non-CCR cases (2.97 ± 1.87-fold vs 1.16 ± 1.05-fold, p < 0.001). Among the 15 CCR patients, 14 presented with high hENT1 expression, and only one showed low hENT1 levels. Therefore, a high hENT1 level is a critical factor to achieve CCR for MDS patients treated with decitabine.
A high CDA or CDA/DCK ratio has previously been reported to contribute to decreased cytidine analog half-life and likely contributes to worse outcomes with decitabine therapy [9, 21]. However, in our study, this ratio did not differ significantly between responders and non-responders. It is conceivable that this discrepancy reflects differences in drug metabolism among diverse racial groups.
Unlike traditional cytotoxic therapies that induce rapid responses in MDS (mostly after one or two cycles) [22, 23], decitabine has a different pattern, in which a response is rare after one cycle but improves over time [4, 24]. Our results showed that patients with higher hENT1 expression achieved CR with fewer cycles of therapy than those with lower hENT1 expression. We speculate that higher hENT1 levels can result in higher intracellular concentrations for one dose, a subsequently improved drug efficacy for each cycle, and therefore fewer cycles to reach the best response.
The results also showed that high expression of hENT1 predicted a prolonged survival in higher-risk MDS patients receiving decitabine treatment. Multivariate regression analysis confirmed that the hENT1 expression level was an independent prognostic indicator in higher risk MDS patients treated with decitabine, regardless of BM blast percentage, platelet count or hemoglobin level. The prolonged survival in patients with high hENT1 expression may result from a better response compared with patients with low hENT1. Therefore, besides the prognosis predicting system such as IPSS/IPSS-Revision (IPSS-R) [25]/WHO prognostic scoring system (WPSS) [26], hENT1 expression level could serve as a complementary prognosis predictor for higher-risk MDS patients treated with decitabine.
The in vitro study showed that knockdown of hENT1 weakened the hypomethylation effect of decitabine treatment in an MDS cell line. Moreover, the biggest gap in the demethylation effect between the two groups was at a decitabine treatment concentration of 0.5 µmol/l, while the effect tended to close in the control group with increasing decitabine concentrations. Therefore, hENT1 expression levels appear to affect the decitabine efficacy at low concentrations. Increasing decitabine to a final concentration of 5 µM resulted in no difference between the hENT1-knockdown group and controls. Therefore, for MDS patients with low hENT1 expression, increasing the dosage of decitabine may be a good choice to achieve a clinical response. Drugs targeting to increase expression of hENT1 may reduce the primary resistance to decitabine and increase the response rate. Notably, other genes involved in the transcriptional down-regulation of hENT1 expression may also affect the response to decitabine. The hENT1 promoter region contains consensus sequences for several transcription factors, including the C/EBP homologous protein (CHOP), a member of the CCAAT/enhancer binding protein family [27, 28]. The CHOP may transcriptional regulate the expression of hENT1 and thus affect the response to decitabine, which will be evaluated in our later study.
Nevertheless, the hENT1 expression level is not the only factor that can predict response to decitabine. The expression of multidrug resistant protein 1 (MRP1) [29] may result to resistance to decitabine in MDS patients, which may partly account for the no response of the drug. Other factors including cytogenetic changes (monosomy 7 or complex karyotypes) [4, 20], and mutations in genes such as TET2 [30] and DNMT3a [31], have also been documented. In the future, analysis of hENT1 expression combined with other factors can serve as an effective response prediction system for decitabine therapy in MDS, which will benefit more patients.
Conclusions
In summary, high expression of hENT1 predicts a good response to decitabine and prolonged survival in higher-risk MDS patients treated with decitabine. Knockdown of hENT1 expression weakens the demethylation effect of decitabine. The hENT1 expression level could serve as an effective response predictor in MDS patients treated with decitabine.
Authors’ contributions
LW and XL conceived the idea for this study. LW, WS, FX, QH, and DW carried out data collection. LW, CC, and FX performed data analysis. LW, DW, JS, LZ, LS, CX, and ZZ provided patient material. LW, WS, and XL wrote the manuscript. XL and LW supervised the study. All authors read and approved the final manuscript.
Acknowledgements
This work was supported by the National Science Foundation of China (81200346) and Natural Science Foundation of Shanghai (12ZR1422400).
Competing interests
The authors declare that they have no competing interests.
Abbreviations
- CCR
cytogenetic complete response
- CDA
cytidine deaminase
- CMML
chronic myelomonocytic leukemia
- CR
complete response
- DCK
deoxycytidine kinase
- FAB
French–American–British
- hENT1
human equilibrative nucleoside transporter 1
- IPSS
international Prognostic Scoring System
- LINE1
long interspersed nucleotide elements 1
- MDS
myelodysplastic syndrome
- OR
overall response
- Q-PCR
quantitative real-time PCR
- RAEB-1
refractory anemia with excess blasts-1
- RAEB-2
refractory anemia with excess blasts-2
- RAEB-t
refractory anemia with excess of blasts in transformation
- RAS
refractory anemia with ringed sideroblasts
- RCMD
refractory cytopenia with multilineage dysplasia
- WHO
World Health Organization
Additional file
10.1186/s12967-016-0817-9 HENT1 expression level increased significantly in patients achieved mCR (a) or HI (b) when compared to that in NR patients, while expression levels of hENT2, DCK, and CDA was not significantly different between mCR (a) or HI (b) patients and NR.
Contributor Information
Lingyun Wu, Email: lincy2032@163.com.
Wenhui Shi, Email: shiwh0049@163.com.
Xiao Li, Email: lixiao3326@163.com.
Chunkang Chang, Email: changchunkang@sina.com.
Feng Xu, Email: fx3619@163.com.
Qi He, Email: he_qi2000@163.com.
Dong Wu, Email: wudong5242@163.com.
Jiying Su, Email: sujianxin2006@126.com.
Liyu Zhou, Email: zhouliyu2000@163.com.
Luxi Song, Email: songluxi@139.com.
Chao Xiao, Email: xch0216@sina.com.
Zheng Zhang, Email: david3321@126.com.
References
- 1.Bejar R, Steensma DP. Recent developments in myelodysplastic syndromes. Blood. 2014;124:2793–2803. doi: 10.1182/blood-2014-04-522136. [DOI] [PubMed] [Google Scholar]
- 2.Tefferi A, Vardiman JW. Myelodysplastic syndromes. N Engl J Med. 2009;361:1872–1885. doi: 10.1056/NEJMra0902908. [DOI] [PubMed] [Google Scholar]
- 3.Kantarjian H, Issa JP, Rosenfeld CS, et al. Decitabine improves patient outcomes in myelodysplastic syndromes: results of a phase III randomized study. Cancer. 2006;106:1794–1803. doi: 10.1002/cncr.21792. [DOI] [PubMed] [Google Scholar]
- 4.Steensma DP, Baer MR, Slack JL, et al. Multicenter study of decitabine administered daily for 5 days every 4 weeks to adults with myelodysplastic syndromes: the alternative dosing for outpatient treatment (ADOPT) trial. J Clin Oncol. 2009;27:3842–3848. doi: 10.1200/JCO.2008.19.6550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kadia TM, Jabbour E, Kantarjian H. Failure of hypomethylating agent-based therapy in myelodysplastic syndromes. Semin Oncol. 2011;38:682–692. doi: 10.1053/j.seminoncol.2011.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Damaraju VL, Mowles D, Yao S, et al. Role of human nucleoside transporters in the uptake and cytotoxicity of azacitidine and decitabine. Nucleosides, Nucleotides Nucleic Acids. 2012;31:236–255. doi: 10.1080/15257770.2011.652330. [DOI] [PubMed] [Google Scholar]
- 7.Tavano F, Fontana A, Pellegrini F, et al. Modeling interactions between Human Equilibrative Nucleoside Transporter-1 and other factors involved in the response to gemcitabine treatment to predict clinical outcomes in pancreatic ductal adenocarcinoma patients. J Transl Med. 2014;12:248. doi: 10.1186/s12967-014-0248-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Qin T, Jelinek J, Si J, et al. Mechanisms of resistance to 5-aza-2′-deoxycytidine in human cancer cell lines. Blood. 2009;113:659–667. doi: 10.1182/blood-2008-02-140038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Qin T, Castoro R, El Ahdab S, et al. Mechanisms of resistance to decitabine in the myelodysplastic syndrome. PLoS ONE. 2011;6:e23372. doi: 10.1371/journal.pone.0023372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wu P, Geng S, Weng J, et al. The hENT1 and DCK genes underlie the decitabine response in patients with myelodysplastic syndrome. Leuk Res. 2015;39:216–220. doi: 10.1016/j.leukres.2014.08.016. [DOI] [PubMed] [Google Scholar]
- 11.Bennett JM, Catovsky D, Daniel MT. Proposals for the classification of the acute leukaemias: French-American-British cooperative group. Br J Haematol. 1976;33:451–458. doi: 10.1111/j.1365-2141.1976.tb03563.x. [DOI] [PubMed] [Google Scholar]
- 12.Vardiman JW, Harris NL, Brunning RD. The World Health Organization (WHO) classification of the myeloid neoplasms. Blood. 2002;100:2292–2302. doi: 10.1182/blood-2002-04-1199. [DOI] [PubMed] [Google Scholar]
- 13.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 14.Tefferi A, Barosi G, Mesa RA, et al. International Working Group (IWG) consensus criteria for treatment response in myelofibrosis with myeloid metaplasia, for the IWG for myelofibrosis research and treatment (IWG-MRT) Blood. 2006;108:1497–1503. doi: 10.1182/blood-2006-03-009746. [DOI] [PubMed] [Google Scholar]
- 15.Yang AS, Estecio MR, Doshi K, et al. A simple method for estimating global DNA methylation using bisulfite PCR of repetitive DNA elements. Nucleic Acids Res. 2004;32:e38. doi: 10.1093/nar/gnh032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Greenberg P, Cox C, LeBeau MM, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–2088. [PubMed] [Google Scholar]
- 17.ISCN (International System for Human Cytogenetic Nomenclature) Guidelines for cancer cytogenetics. In: Mitelman F, editor. Supplement to an International System for Human Cytogenetic Nomenclature. Basel: Karger; 1991. pp. 1–53. [Google Scholar]
- 18.Galmarini CM, Mackey JR, Dumontet C. Nucleoside analogues: mechanisms of drug resistance and reversal strategies. Leukemia. 2001;15:875–890. doi: 10.1038/sj.leu.2402114. [DOI] [PubMed] [Google Scholar]
- 19.Damaraju VL, Mowles D, Yao S, et al. Role of human nucleoside transporters in the uptake and cytotoxicity of azacitidine and decitabine. Nucleosides, Nucleotides Nucleic Acids. 2012;31:236–255. doi: 10.1080/15257770.2011.652330. [DOI] [PubMed] [Google Scholar]
- 20.Li X, Chang C, He Q, et al. Cytogenetic response based on revised IPSS cytogenetic risk stratification and minimal residual disease monitoring by FISH in MDS patients treated with low-dose decitabine. Leuk Res. 2013;37:1516–1521. doi: 10.1016/j.leukres.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 21.Mahfouz RZ, Jankowska A, Ebrahem Q, et al. Increased CDA expression/activity in males contributes to decreased cytidineanalog half-life and likely contributes to worse outcomes with 5-azacytidine or decitabine therapy. Clin Cancer Res. 2013;19:938–948. doi: 10.1158/1078-0432.CCR-12-1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.de Witte T, Suciu S, Verhoef G, et al. Intensive chemotherapy followed by allogeneic or autologous stem cell transplantation for patients with myelodysplastic syndromes (MDSs) and acute myeloid leukemia following MDS. Blood. 2001;98:2326–2331. doi: 10.1182/blood.V98.8.2326. [DOI] [PubMed] [Google Scholar]
- 23.Wu L, Li X, Su J, et al. Efficacy and safety of CHG regimen (low-dose cytarabine, homoharringtonine with G-CSF priming) as induction chemotherapy for elderly patients with high-risk MDS or AML transformed from MDS. J Cancer Res Clin Oncol. 2011;137:1563–1569. doi: 10.1007/s00432-011-1020-2. [DOI] [PubMed] [Google Scholar]
- 24.Saunthararajah Y. Key clinical observations after 5-azacytidine and decitabine treatment of myelodysplastic syndromes suggest practical solutions for better outcomes. Hematology Am Soc Hematol Educ Program. 2013;2013:511–521. doi: 10.1182/asheducation-2013.1.511. [DOI] [PubMed] [Google Scholar]
- 25.Greenberg PL, Tuechler H, Schanz J, et al. Revised international prognostic scoring system (IPSS-R) for myelodysplastic syndromes. Blood. 2012;120:2454–2465. doi: 10.1182/blood-2012-03-420489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Malcovati L, Germing U, Kuendgen A, et al. Time-dependent prognostic scoring system for predicting survival and leukemic evolution in myelodysplastic syndromes. J Clin Oncol. 2007;25:3503–3510. doi: 10.1200/JCO.2006.08.5696. [DOI] [PubMed] [Google Scholar]
- 27.Ubeda M, Wang XZ, Zinszner H, et al. Stress-induced binding of the transcriptional factor CHOP to a novel DNA control element. Mol Cell Biol. 1996;16:1479–1489. doi: 10.1128/MCB.16.4.1479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Farías M, Puebla C, Westermeier F, et al. Nitric oxide reduces SLC29A1 promoter activity and adenosine transport involving transcription factor complex hCHOP-C/EBPalpha in human umbilical vein endothelial cells from gestational diabetes. Cardiovasc Res. 2010;86:45–54. doi: 10.1093/cvr/cvp410. [DOI] [PubMed] [Google Scholar]
- 29.Kurata M, Hasegawa M, Nakagawa Y, et al. Expression dynamics of drug resistance genes, multidrug resistance 1 (MDR1) and lung resistance protein (LRP) during the evolution of overt leukemia in myelodysplastic syndromes. Exp Mol Pathol. 2006;8:249–254. doi: 10.1016/j.yexmp.2006.01.004. [DOI] [PubMed] [Google Scholar]
- 30.Bejar R, Lord A, Stevenson K, et al. TET2 mutations predict response to hypomethylating agents in myelodysplastic syndrome patients. Blood. 2014;124:2705–2712. doi: 10.1182/blood-2014-06-582809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Traina F, Visconte V, Elson P, et al. mpact of molecular mutations on treatment response to DNMT inhibitors in myelodysplasia and related neoplasms. Leukemia. 2014;28:78–87. doi: 10.1038/leu.2013.269. [DOI] [PubMed] [Google Scholar]


