Abstract
Background
Postprandial glucose (PPG) and insulin responses play a role in carcinogenesis. We evaluated the association between dietary glycemic index (GI) and glycemic load (GL), markers of carbohydrate intake and PPG, and lung cancer risk in non-Hispanic whites.
Methods
GL and GI were assessed among 1,905 newly diagnosed lung cancer cases recruited from the University of Texas MD Anderson Cancer Center in Houston, Texas and 2,413 healthy controls recruited at Kelsey-Seybold Clinics in Houston. We assessed associations between quintiles of GI/GL and lung cancer risk and effect modification by various risk factors. Odds ratios (ORs) and 95% confidence intervals (CI) were estimated using multivariable logistic regression.
Results
We observed a significant association between GI (5th vs 1st quintile (Q)) OR = 1.49, 95% CI: 1.21–1.83, P-trend <0.001) and lung cancer risk and GIac (5th vs 1st Q OR = 1.48, 95% CI: 1.20–1.81, P-trend =0.001) and lung cancer risk. We observed a more pronounced association between GI and lung cancer risk among never smokers, (5th vs 1st Q OR=2.25, 95% CI: 1.42–3.57), squamous cell carcinomas (SCCs) (5th vs 1st Q OR=1.92, 95% CI: 1.30–2.83) and those with less than 12 years of education (5th vs 1st Q OR=1.75, 95% CI: 1.19–2.58, P-interaction = 0.02).
Conclusion
This study suggests that dietary glycemic index and other lung cancer risk factors may jointly and independently influence lung cancer etiology.
Impact
Understanding the role of glycemic index in lung cancer could inform prevention strategies and elucidate biological pathways related to lung cancer risk.
Keywords: glycemic index, glycemic load, lung cancer, joint effects
INTRODUCTION
Lung cancer is the second most commonly diagnosed cancer in the United States (1). Smoking is the most well characterized risk factor for lung cancer and accounts for approximately 85% of the population burden of lung cancer in developed nations (2). However, evidence suggests that select dietary factors may modulate lung cancer risk. Factors including vitamins A, C, and E and diets high in fruits and vegetables have been associated with reduced lung cancer risk, while intake of red meat, dairy products, saturated fat, and lipids have all been associated with an increased risk (3).
The type and amount of dietary carbohydrate are the main determinants of postprandial glucose and insulin responses (4) which have been shown play a role in promoting tumor growth and carcinogenesis (5, 6). The glycemic index (GI) is a classification of carbohydrate rich foods based on postprandial blood glucose responses, dependent on both the nature of the carbohydrate and the type and extent of the food processing. GI measures how quickly carbohydrates in food cause blood glucose levels to rise after eating (7). Elevated blood glucose levels stimulate the secretion of insulin. Insulin receptors activate signaling pathways in the cell that are mitogenic, suggesting that chronically elevated concentrations of insulin may influence the risk of cancer through indirect effects on the insulin-like growth factors (IGFs). IGFs have been shown to play a critical role in regulating cell proliferation and differentiation in cancer (8) and there is evidence to suggest that IGFs are elevated in lung cancer patients (9, 10).
Previous studies have investigated the association between GI, and the related measure glycemic load (GL), and a variety of cancers including colorectal (11–13), stomach (14–16), pancreas (17, 18), endometrial (7, 19–21), ovarian (22, 23), prostate (24, 25) and thyroid (26) but these studies are limited and results have been largely inconclusive. To date, only one smaller study has evaluated the association between dietary glycemic index and lung cancer risk in a case-control population in Uruguay (27). In a large study of newly diagnosed lung cancer patients and healthy controls, we investigated whether dietary GI and GL were associated with lung cancer risk in non-Hispanic whites, and whether these associations varied by known or suspected lung cancer risk factors, including smoking.
MATERIALS AND METHODS
Study population
The patients and control subjects were selected from an ongoing case-control study of lung cancer conducted in the Department of Epidemiology at The University of Texas MD Anderson Cancer Center. Newly diagnosed, histologically-confirmed cases who had not previously received treatment other than surgery were recruited from MD Anderson Cancer. Healthy control subjects, with no previous history of cancer (except non-melanoma skin cancer), were selected from individuals seen for routine care at Kelsey-Seybold Clinics; the largest physician group-practice plan in the Houston Metropolitan area. Controls were frequency matched to cases according to their age in 5 year categories, gender, and ethnicity. The study was approved by the Institutional Review Boards at The University of Texas MD Anderson Cancer Center and the Kelsey-Seybold Foundation.
Data Collection
All study participants completed an in-person interview by MD Anderson staff interviewers to obtain information on a variety of factors, including but not limited to, demographics, socioeconomic status (education), history of hypertension prior to diagnosis or recruitment (for control subjects), physical activity and smoking (including cigarettes per day) and alcohol history. An individual who had never smoked or had smoked less than 100 cigarettes in his or her lifetime was defined as a never smoker. An individual who had smoked at least 100 cigarettes in his or her lifetime but had quit at least 12 months prior to diagnosis (for cases) or interview (for controls) was classified as a former smoker. Current smokers were those who were currently smoking or quit less than 12 months before diagnosis (for cases) or before the interview (for controls).
Weight and height at diagnosis (for cases) or recruitment (for control subjects) was recorded. Body mass index (BMI; kg/m2) was derived from weight and adult height. BMI was categorized according to the standard classifications of the World Health Organization (WHO) (normal = <25kg/m2; overweight = 25–29.9kg/m2; obese = ≥30kg/m2). Participants also reported the average number of times they undertook each of five broad groups of activities in the year before the interview. Activities included active sports, physical exercises, swimming, walking (including walking for golf), cycling, gardening or yard work, hunting, housework and other strenuous exercises. A metabolic equivalent value (MET) was assigned on the basis of the energy cost of each activity group (28). Energy expenditure from physical activity was calculated as the MET value of each activity multiplied by the frequency of each activity and then summed across all activities.
We used a modified version of the National Cancer Institute Health Habits and History Questionnaire (29). The questionnaire includes a semi-quantitative food frequency list made up of food and beverage items and an open-ended section regarding dietary behaviors such as dining in restaurants and food preparation methods. Portion size was also queried. Total energy intake, total carbohydrate intake, total fiber intake, and grams per day of consumption for each food item were estimated using the USDA Food and Nutrient Database for Dietary Studies (FNDDS)(30). Total meat intake was calculated by adding total grams per day of each meat item in the FFQ. All nutrient and food variables of interest were energy adjusted for total caloric intake using the residual method (31).
Due to smaller numbers of minorities, the large number of GI/GL categories, and to reduce the residual confounding by population stratification, the present analysis is limited to non-Hispanic whites only.
Exclusions and Eligibility
A total of 4,644 non-Hispanic white cases and controls had complete dietary information for inclusion in the present analysis. We excluded individuals with outlying total energy intake by excluding individuals (N = 158) with values that fell outside the interval delimited by the 25th percentile minus 1.5 times the interquartile range and the 75th percentile plus 1.5 times the interquartile range based on the distribution of energy intake in the population, by gender. Individuals with missing BMI were excluded from the analysis since the distribution of missing data was not even among cases and controls (N=168). A total of 4,318 individuals were included in the present analysis (1,905 cases and 2,413 controls).
Glycemic Index/Load Calculations
We derived glycemic index values according to a method previously described (32, 33). Briefly, using published GI values compiled by Foster-Powell et al (32), we linked GI values (using a scale in which the GI for pure glucose =100) to each of the individual foods in the FFQ. The overall GL was calculated by taking the product of the carbohydrate content of a given food item by the quantity of that food item consumed per day and its GI value and then summing the values for all food items. The overall GI, which reflects the average quality of carbohydrate consumed, was calculated by dividing the total GL by total daily carbohydrate consumption. We also calculated total GI using total available carbohydrate (GIac), which was calculated using the same formula for GI, but by subtracting the fiber values from the carbohydrate values used in the GI calculations. GI exposures were energy-adjusted using the residual method (31).
Statistical Analysis
Physical activity levels (METs) and alcohol intake were categorized into tertiles based on the distribution in control subjects. Missing physical activity was consistent between cases and controls and therefore coded as a separate “unknown” category. Analyses limited to only the sample with complete physical activity information were consistent with the overall findings. Smoking status was categorized according to pack-years, calculated as the number of cigarettes per day multiplied by the number of years smoking, into the following categories: never smoker, former smoker <20 pack-years, former smoker ≥ 20 pack-years, current smoker < 20 pack-years and current smoker ≥ 20 pack-years. We additionally created a more granular categorization of smoking (never smoker, former smoker <10 pack-years, former smoker 10–20 pack-years, former smoker ≥ 20 pack-years, current smoker < 10 pack-years, current smoking 10–20 pack-years, and current smoker ≥ 20 pack-years) which yielded consistent results (not shown). Alcohol consumption was adjusted for total caloric intake using the residual method and divided into tertiles based on the distribution in the controls for males and females separately. GI and GL exposures were categorized into quintiles based on the distribution among the controls and by gender with the reference group comprised of individuals in the lowest category of intake.
Comparisons for case-control characteristics were performed using the Pearson χ2 test for categorical variables and Student t tests for continuous variables. Unconditional logistic regression was used to calculate odds ratios (OR) and 95% confidence intervals (CI) for the association between quintiles of GI and GL and lung cancer risk (overall and separately for two major histologic subtypes, adenocarcinoma (AC) and squamous cell carcinoma (SCC)). In calculating ORs and 95% CIs, we used two modeling approaches. In the first (minimally-adjusted) model we included age, gender, education (<12 years, 12–15 years, 16+ years), and smoking status. Matching variables were retained in these models to account for the possibility of residual confounding.
In the second approach (the fully-adjusted model) we additionally controlled for variables that were considered biologically and statistically relevant in the multivariable model. Variables were retained if they improved the fit and predictive power of the model and were statistically significant by the likelihood ratio test. The variables included physical activity (tertiles), BMI (WHO categories), total caloric intake (continuous), alcohol intake (tertiles), total meat intake (energy-adjusted continuous) and fiber intake (energy-adjusted continuous). Alcohol intake, meat intake and fiber intake were removed from the final model because they were not associated with lung cancer risk. Tests for trend were obtained by including an ordinal exposure variable in the model.
Stratified analyses for overall lung cancer risk were conducted by smoking status (never/ever), gender, years of education (<12, 12+), age (<60, 60+), and BMI (normal versus overweight and obese). Multiplicative interaction was assessed by including the cross-product term of dichotomous variable with the GI/GL exposure in the logistic regression model. Statistical significance was determined using the Wald statistic. We subsequently ran a sensitivity analysis on a dataset further matched on smoking status. Results of this analysis were qualitatively similar to the overall findings and are presented in the Supplementary Tables 1–3. Joint effects of smoking (ever/never) and GL/GI (median high/low, determined by the distribution in controls, by gender) were also assessed. Analyses were conducted with STATA version 13.0 (Stata Corp, College Station, Tex).
Results
Participant characteristics are described in Table 1. Cases were more likely to smoke, be heavier smokers and had lower BMI, physical activity levels, and years of education (P<0.001 each). Cases had higher daily values for both glycemic index variables (P<0.001 for each), lower total carbohydrate intake g/day (marginally significant P=0.06), and lower fiber intake (P<0.001).
Table 1.
Participant Characteristics
| Cases (N=1905) N(%) |
Controls (N=2413) N(%) |
P-value | |
|---|---|---|---|
| Age, Mean (SD)a | 60.69 (10.67) | 60.78 (10.43) | 0.79 |
| Gendera | |||
| Male | 1006 (52.81) | 1232 (51.06) | |
| Female | 899 (47.19) | 1181 (48.94) | 0.25 |
| Smoking Status | |||
| Never | 311 (16.33) | 593 (24.58) | |
| Former | 814 (42.73) | 988 (40.94) | |
| Current | 780 (40.94) | 832 (34.48) | <0.001 |
| Pack Years in Ever Smokers, Mean (SD) | 49.85 (31.00) | 42.32 (30.33) | <0.001 |
| BMI, kg/m2 | |||
| Underweight/Normal | 841 (44.15) | 774 (32.08) | |
| Overweight | 688 (36.12) | 955 (39.58) | |
| Obese | 376 (19.74) | 684 (28.35) | <0.001 |
| Physical Activityb | |||
| Low | 618 (39.34) | 666 (31.28) | |
| Medium | 460 (29.28) | 649 (30.48) | |
| High | 493 (31.38) | 814 (38.23) | <0.001 |
| Years of Educationb | |||
| Less than 12 | 792 (41.62) | 603 (25.03) | |
| 12 to 15 | 558 (29.32) | 825 (34.25) | |
| 16+ | 553 (29.06) | 981 (40.72) | <0.001 |
| Family History of Cancerb | |||
| No | 1442 (76.97) | 2016 (83.90) | |
| Yes | 456 (24.03) | 387 (16.10) | 0.001 |
| Emphysemab | |||
| No | 1547 (82.11) | 2250 (93.36) | |
| Yes | 337 (17.89) | 160 (6.64) | <0.001 |
| Pneumoniab | |||
| No | 1114 (58.94) | 1717 (71.30) | |
| Yes | 776 (41.06) | 691 (28.70) | <0.001 |
| Hay feverb | |||
| No | 1591 (84.63) | 1897 (78.68) | |
| Yes | 289 (15.37) | 514 (21.32) | <0.001 |
| Histologic Subtypec | |||
| Adenocarcinoma | 1071 (57.06) | NA | |
| Squamous Cell Carcinoma | 446 (23.76) | NA | |
| Other | 360 (19.18) | NA | |
| Dietary Factors | |||
| Glycemic Load, Mean (SD) | 136.64 (62.86) | 136.77 (56.86) | 0.94 |
| Glycemic Index, Mean (SD) | 53.75 (5.14) | 52.74 (4.30) | <0.001 |
| Glycemic Index, available carbohydrate, Mean (SD) | 58.33 (5.22) | 57.62 (4.31) | <0.001 |
| Carbohydrate g/day, Mean(SD) | 252.09(110.59) | 258.03 (101.65) | 0.06 |
| Fiber g/day, Mean (SD) | 19.19 (9.46) | 21.39 (10.05) | <0.001 |
| Meat intake g/day, Mean (SD) | 125.94 (81.08) | 127.09 (74.50) | 0.63 |
matching factors
Missing; years of education N=6, family history of cancer N=17, emphysema N=24, pneumonia N=20, hayfever N=27; unknown physical activity N=618
Unknown histologic subtype not included in the table (N=28)
Minimally- and fully-adjusted results for the association between GI and GL variables and lung cancer risk (overall, AC, and SCC) are presented in Table 2. GL was not associated with lung cancer risk in any model. Higher GI and GIac were significantly associated with an increased risk of lung cancer overall (GI 5th versus 1st quintile, fully adjusted OR = 1.49, 95% CI: 1.21–1.83, P-trend <0.001; GIac 5th versus 1st quintile, fully adjusted OR =1.48, 95% CI: 1.20–1.81, P-trend = 0.001). GIac was marginally significantly associated with AC risk (GIac 5th versus 1st quintile, fully adjusted OR = 1.31, 95% CI: 1.02–1.67, P-trend= 0.08). GI and GIac were significantly associated with SCC risk (GI 5th versus 1st quintile, fully adjusted OR = 1.92, 95% CI: 1.30–2.83, P-trend <0.001; GIac 5th versus 1st quintile, fully adjusted OR = 1.92, 95% CI: 1.31–2.82, P-trend <0.001), with more pronounced effect estimates for SCCs compared to ACs.
Table 2.
Associations Between Lung Cancer Risk and Energy -Adjusted Quintilesa of Dietary Glycemic Load, Glycemic Index and Glycemic Index (Available Carbohydrate) Intake
| Quintiles of Daily GI/GL | ||||||
|---|---|---|---|---|---|---|
|
| ||||||
| Q1 | Q2 | Q3 | Q4 | Q5 | P-trend | |
| Overall | ||||||
| Glycemic Load | ||||||
| Cases/Controls | 358/483 | 332 (36.73) | 409/477 | 353/479 | 430/488 | |
| Minimally Adjusted Model b | 1 (REF) | 1.00 (0.82–1.22) | 1.18 (0.97–1.44) | 1.03 (0.84–1.26) | 1.20 (0.98–1.45) | 0.08 |
| Fully Adjusted Model c | 1 (REF) | 1.04 (0.84–1.27) | 1.12 (0.90–1.38) | 1.04 (0.84–1.29) | 1.16 (0.94–1.42) | 0.19 |
| Glycemic Index | ||||||
| Cases/Controls | 399/488 | 315/474 | 361/492 | 374/482 | 554/473 | |
| Minimally Adjusted Model b | 1 (REF) | 1.08 (0.88–1.33) | 1.14 (0.93–1.39) | 1.11 (0.91–1.37) | 1.59 (1.30–1.93) | <0.001 |
| Fully Adjusted Model c | 1 (REF) | 1.11 (0.90–1.38) | 1.16 (0.94–1.43) | 1.16 (0.93–1.42) | 1.49 (1.21–1.83) | <0.001 |
| Glycemic Index (Available Carbohydrate) | ||||||
| Cases/Controls | 301/481 | 327/471 | 355/499 | 393/475 | 527/483 | |
| Minimally Adjusted Model b | 1 (REF) | 1.08 (0.88–1.33) | 1.09 (0.89–1.34) | 1.20 (0.98–1.47) | 1.49 (1.23–1.81) | <0.001 |
| Fully Adjusted Model c | 1 (REF) | 1.18 (0.95–1.46) | 1.11 (0.89–1.37) | 1.30 (1.05–1.60) | 1.48 (1.20–1.81) | 0.001 |
| By Histologic Subtype; Adenocarcinoma | ||||||
| Glycemic Load | ||||||
| Cases/Controls | 188/483 | 198/482 | 201/477 | 179/479 | 236/488 | |
| Minimally Adjusted Model b | 1(REF) | 1.05 (0.82–1.33) | 1.08 (0.85–1.38) | 0.97 (0.76–1.24) | 1.22 (0.97–1.24) | 0.2 |
| Fully Adjusted Model c | 1 (REF) | 1.07 (0.83–1.38) | 1.01 (0.79–1.30) | 0.98 (0.76–1.27) | 1.16 (0.91–1.47) | 0.43 |
| Glycemic Index | ||||||
| Cases/Controls | 179/488 | 189/474 | 177/492 | 184/482 | 273/473 | |
| Minimally Adjusted Model b | 1(REF) | 1.09 (0.86–1.39) | 0.94 (0.74–1.20) | 0.98 (0.76–1.25) | 1.40 (1.11–1.77) | 0.02 |
| Fully Adjusted Model c | 1 (REF) | 1.13 (0.8–1.46) | 0.98 (0.76–1.26) | 1.01 (0.78–1.30) | 1.30 (1.02–1.66) | 0.1 |
| Glycemic Index (Available Carbohydrate) | ||||||
| Cases/Controls | 176/481 | 196/471 | 170/499 | 204/475 | 256/483 | |
| Minimally Adjusted Model b | 1(REF) | 1.12 (0.88–1.42) | 0.91 (0.71–1.17) | 1.10 (0.87–1.17) | 1.32 (1.05–1.67) | 0.03 |
| Fully Adjusted Model c | 1 (REF) | 1.23 (0.96–1.58) | 0.93 (0.72–1.21) | 1.15 (0.90–1.48) | 1.31 (1.02–1.67) | 0.08 |
| By Histologic Subtype; Squamous Cell Carcinoma | ||||||
| Glycemic Load | ||||||
| Cases/Controls | 84/483 | 69/482 | 97/477 | 75/479 | 90/488 | |
| Minimally Adjusted Model b | 1(REF) | 0.79 (0.55–1.13) | 1.26 (0.90–1.77) | 0.92 (0.64–1.31) | 1.05 (0.75–1.49) | 0.54 |
| Fully Adjusted Model c | 1 (REF) | 0.82 (0.56–1.20) | 1.28 (0.89–1.83) | 0.97 (0.67–1.41) | 1.05 (0.73–1.49) | 0.65 |
| Glycemic Index | ||||||
| Cases/Controls | 48/488 | 55/474 | 95/492 | 87/482 | 130/473 | |
| Minimally Adjusted Model b | 1(REF) | 1.14 (0.74–1.73) | 1.79 (1.22–2.63) | 1.57 (1.06–2.32) | 2.08 (1.43–3.02) | <0.001 |
| Fully Adjusted Model c | 1 (REF) | 1.07 (0.69–1.65) | 1.68 (1.13–2.51) | 1.56 (1.04–2.32) | 1.92 (1.30–2.83) | <0.001 |
| Glycemic Index (Available Carbohydrate) | ||||||
| Cases/Controls | 49/481 | 60/471 | 91/499 | 89/475 | 126/483 | |
| Minimally Adjusted Model b | 1(REF) | 1.13 (0.75–1.71) | 1.61 (1.09–2.36) | 1.63 (1.11–2.40) | 1.94 (1.34–2.81) | <0.001 |
| Fully Adjusted Model c | 1 (REF) | 1.14 (0.75–1.75) | 1.56 (1.05–2.32) | 1.73 (1.16–2.58) | 1.92 (1.31–2.82) | <0.001 |
quintiles based on distribution in controls, by gender
Minimally Adjusted Model include adjustment for age, education, gender and smoking status N=6 missing education
Same as minimally adjusted, but also includes history of emphysema, pneumonia, hay fever, family history of lung cancer and physical activity, total energy intake and BMI
Fully-adjusted Missing; N=55; unknown physical activity N=618
Analyses stratified by age, gender, education, smoking status, and BMI were also conducted (Table 3). Effect estimates for GL, GI, and GIac were more pronounced for never smokers compared to ever smokers (GL 5th versus 1st quintile; never smokers OR = 1.81, 95% CI: 1.11–2.93, P-trend =0.02 versus ever smokers OR = 1.01, 95% CI: 0.80–1.26, P-trend =0.94, GI 5th versus 1st quintile; never smokers OR = 2.25, 95% CI: 1.42–3.59, P-trend =0.002 versus ever smokers OR = 1.31, 95% CI: 1.04–1.65, P-trend =0.02: GIac 5th versus 1st quintile, never smokers OR = 2.06, 95% CI: 1.30–3.27, P-trend =0.001 versus ever smokers OR = 1.36, 95% CI: 1.08–1.71, P-trend = 0.01). A significant interaction was observed between GL and smoking status (P-interaction = 0.04).
Table 3.
Associations Between Lung Cancer Risk and Energy -Adjusted Quintilesa of Dietary Glycemic Load, Glycemic Index and Glycemic Index (Total Available Carbohydrate) Intake, Stratified by Smoking Status and Years of Education
| Smoking Status | Years of Education | |||||||
|---|---|---|---|---|---|---|---|---|
| Never | Ever | <12 | 12 + | |||||
| Cases/Controls | OR (95%CI) | Cases/Controls | OR (95%CI) | Cases/Controls | OR (95%CI) | Cases/Controls | OR (95%CI) | |
| Glycemic Load | ||||||||
| Q1 | 39/101 | 1 (REF) | 317/382 | 1 (REF) | 128/114 | 1(REF) | 230/369 | 1(REF) |
| Q2 | 52/111 | 1.28 (0.77–2.15) | 293/365 | 0.97 (0.77–1.21) | 133/122 | 1.05 (0.71–1.55) | 220/360 | 1.05 (0.82–1.34) |
| Q3 | 62/122 | 1.39 (0.84–2.30) | 329/351 | 1.06 (0.84–1.33) | 174/124 | 1.30 (0.89–1.91) | 235/353 | 1.07 (0.83–1.37) |
| Q4 | 70/134 | 1.40 (0.85–2.29) | 280/343 | 0.96 (0.76–1.22) | 159/114 | 1.38 (0.94–2.03) | 194/365 | 0.91 (0.71–1.18) |
| Q5 | 85/122 | 1.81 (1.11–2.93) | 343/362 | 1.01 (0.80–1.26) | 198/129 | 1.55 (1.07–2.24) | 232/359 | 1.03 (0.81–1.32) |
| P-trend | 0.02 | 0.94 | 0.006 | 0.83 | ||||
| P-interaction = 0.04 | P-interaction = 0.04 | |||||||
| Glycemic Index | ||||||||
| Q1 | 49/131 | 1 (REF) | 241/356 | 1 (REF) | 85/93 | 1(REF) | 214/395 | 1(REF) |
| Q2 | 64/120 | 1.40 (0.88–2.23) | 250/352 | 1.01 (0.80–1.29) | 96/103 | 1.04 (0.67–1.61) | 219/371 | 1.13 (0.89–1.44) |
| Q3 | 61/127 | 1.34 (0.94–2.13) | 299/362 | 1.10 (0.87–1.40) | 123/125 | 1.11 (0.73–1.70) | 238/367 | 1.19 (0.93–1.52) |
| Q4 | 57/117 | 1.41 (0.87–2.28) | 315/359 | 1.09 (0.86–1.38) | 196/124 | 1.77 (1.18–2.65) | 178/358 | 0.93 (0.72–1.20) |
| Q5 | 77/95 | 2.25 (1.42–3.59) | 457/374 | 1.31 (1.04–1.65) | 292/158 | 1.75 (1.19–2.58) | 262/315 | 1.37 (1.07–1.75) |
| P-trend | 0.002 | 0.02 | <0.001 | 0.11 | ||||
| P-interaction = 0.37 | P-interaction = 0.02 | |||||||
| Glycemic Index (Available Carbohydrate) | ||||||||
| Q1 | 47/127 | 1 (REF) | 244/353 | 1 (REF) | 95/94 | 1(REF) | 206/387 | 1(REF) |
| Q2 | 55/124 | 1.21 (0.75–1.95) | 271/345 | 1.16 (0.91–1.47) | 101/116 | 0.99 (0.65–1.52) | 226/355 | 1.25 (0.98–1.60) |
| Q3 | 65/119 | 1.56 (0.97–2.49) | 288/372 | 1.01 (0.80–1.28) | 130/116 | 1.14 (0.75–1.73) | 225/383 | 1.09 (0.85–1.39) |
| Q4 | 63/111 | 1.68 (1.04–2.70) | 329/363 | 1.20 (0.95–1.52) | 189/120 | 1.71 (1.15–1.73) | 204/355 | 1.14 (0.88–1.46) |
| Q5 | 78/109 | 2.06 (1.30–3.27) | 430/370 | 1.36 (1.08–1.71) | 277/157 | 1.77 (1.21–2.60) | 250/326 | 1.33 (1.04–1.70) |
| P-trend | 0.001 | 0.01 | <0.001 | 0.09 | ||||
| P-interaction = 0.29 | P-interaction = 0.01 | |||||||
Fully Adjusted Model where appropriate Missing N = 55
quintiles based on distribution in controls, by gender
High GL, GI and GIac were significantly associated with lung cancer risk for individuals with less than 12 years of education (GL 5th versus 1st quintile OR = 1.55, 95% CI: 1.07–2.24, P-trend 0.006, GI 5th versus 1st quintile OR =1.75, 95% CI: 1.19–2.58, P-trend <0.001, GIac 5th versus 1st quintile OR =1.77, 95% CI: 1.21–2.60, P-trend <0.001) (Table 3). High GI and GIac were also associated with lung cancer risk in individuals with more than 12 years of education, but the effect estimates were attenuated and the trends were no longer significant. We observed interactions between GL (P-interaction =0.04), GI (P-interaction = 0.02), and GIac (P-interaction =0.01) and years of education on lung cancer risk. The remaining stratified analyses were consistent with the overall findings (not shown), with increased risk consistently associated with the highest quintile of GI across all subgroups; GI 5th versus 1st quintile; female =1.43, 95 % CI: 0.82–1.51, P-trend =0.01; GI 5th versus 1st quintile; male= 1.40, 95% CI: 1.05–1.88, P-trend =0.05; GI 5th versus 1st quintile; overweight/obese =1.53, 95% CI: 0.84–1.99, P-trend = 0.002; GI 5th versus 1st quintile; normal weight=1.67, 95% CI: 1.20–2.33, P-trend <0.001; GI 5th versus 1st quintile; Age <60 = 1.37, 95% CI: 1.00–1.87, P-trend =0.04; GI 5th versus 1st quintile; Age 60+ = 1.41, 95% CI: 1.07–1.86, P-trend =0.01. Results for GIac are consistent with these findings and therefore not shown here
Discussion
This is only the second study to suggest an independent association between GI and lung cancer risk and the first study to suggest that GI may influence lung cancer risk more profoundly in specific subgroups, including never smokers, individuals with low levels of education (<12 years), and those diagnosed with certain histologic subtypes of lung cancer, specifically SCC. In this case-control study, we observed a 49% increased risk of lung cancer associated with daily GI with consistent findings for GIac. Results for the two major histologic subtypes were consistent; however more pronounced effects were observed between GI and SCC and between GL/GI and lung cancer risk in never smokers. Additionally, we observed significant interactions between high GL, GI, and GIac and education.
A previous case-control study of 463 cases and 465 controls conducted in Uruguay found that glycemic index and sucrose-to-dietary fiber ratio were significantly associated with lung cancer risk (OR=2.77, 95%CI: 1.28–5.97 and OR=1.77, 95% CI: 1.11–2.83, respectively)(27). While the associations between glycemic index and glycemic load have not been extensively studied with regards to lung cancer risk, previous studies of the association between these factors and other cancers have suggested a role for increased dietary glycemic index and glycemic load in cancer etiology.
Diets high in GI result in higher levels of blood glucose and insulin, which promote glucose intolerance, insulin resistance and hyperinsulinemia (5, 6, 8). Insulin resistance is a pathological condition, and previous studies suggest that insulin resistance is associated with abnormally high levels of growth factors, adipokines, reactive oxygen species, adhesion factors, and proinflammatory cytokines, all of which have been associated with neoplastic tissue survival and cancer stem cell development (34–36). Circulating levels of insulin have also been associated with a variety of different cancers (37) and may modulate cancer risk via perturbations in the IGF axis.
The IGF system is an integral part of growth regulation by the body and abnormalities in all levels of the IGF system have been implicated in carcinogenesis and cellular transformation (38, 39). IGFs, such as IGF-1, play a pivotal role in regulating cell proliferation, differentiation and apoptosis. IGF-binding proteins normally inhibit the action of IGFs by blocking the binding of IGFs to their receptor (10). Lower levels of these binding proteins have also been associated with increased cancer risk (40). In a previous case-control study of 204 histologically confirmed primary lung cancer patients and 218 control subjects, higher plasma levels of IGF-I and lower levels of IGFBP-3, an IGF binding protein, were associated with an increased risk of lung cancer that persisted after adjustment for age, gender, BMI, smoking status, race and family history of any cancer (10). This study also showed a significant dose response relationship between levels of plasma IGF-1 and lung cancer risk. Results from animal experiments and cell cultures studies also suggest that IGF-1 is a potent mitogen for a variety of cancer cells including breast, prostate, lung, colon, and liver cells (41). The evidence for the association between the IGF system and lung cancer is inconsistent however, with several studies suggesting a null association (42, 43). Further research is necessary to understand the underlying mechanisms linking glycemic index/load, the insulin-like growth factor axis, and lung cancer risk in human populations.
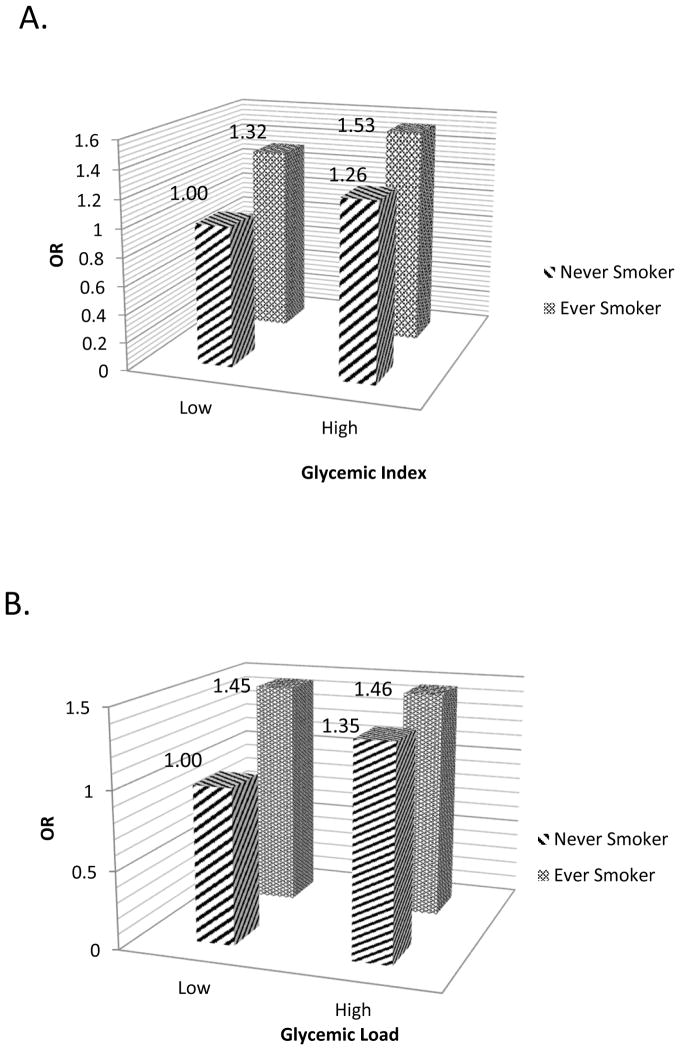
The stratified analyses by smoking status showed a more profound, independent association between dietary GI and lung cancer risk in individuals without traditional lung cancer risk factors (i.e smoking). Smoking is the most important risk factor for most lung cancers, therefore, it stands to reason that among smokers, GI might not play an overwhelming role in lung cancer risk. We did find, however that smokers with high dietary GI had slightly larger effect estimates compared to smokers with low GI in the joint effects analysis (Figure 1). Stratified analyses by other factors, such as years of education and histologic subtype, suggest possible joint or modifying effects between these risk factors and GI. Educational attainment is a proxy for socioeconomic status which has been linked with poor diet quality (including high intake of simple sugars and reduced intake of fiber) in various studies (44–46). Socioeconomic status is also closely linked with smoking behavior (47), therefore the associations between glycemic index and lung cancer risk in individuals with less than 12 years of formal education may represent the joint impact of low diet quality and smoking on lung cancer risk.
Figure 1. Joint Effect of GL/GI and Smoking Status.
Joint effect of A) glycemic index and B) glycemic load with smoking status in lung cancer risk (p-values for all ORs are significant). Low and High are defined by the median cutoff in control
Fully Adjusted Model where appropriate - Missing N = 55
SCC is the histologic subtype of lung cancer most closely linked with smoking behavior (48). Previous studies of dietary intake and lung cancer risk have suggested that the impact of dietary factors, such as fruit and vegetable intake, on lung cancer risk may be more pronounced in smokers and SCCs (49, 50). GI may have a more profound impact on SCC via insulin-like growth factor pathways. Smoking has been associated with expression of IGF-1 and insulin-like growth factor type 1 receptor (IGF-1R), particularly in SCCs of the lung (51–53). In our study, smokers and non-smokers differed by various characteristics (see Supplementary Table 4) including histologic subtype and education; 95% of SCC cases were smokers and nearly 50% had less than 12 years of education compared to the 75% of ACs that were smokers and only 35% had less than 12 years of education. These differences are consistent with the existing literature and could account for the differential associations between GI and lung cancer risk in various subgroups (54–56). Future research is necessary to elucidate the mechanisms underlying the interplay between socioeconomic status, smoking behavior, glycemic index, and lung cancer risk.
The present study has several strengths. It is the largest study of GL/GI and lung cancer risk to date and the first to be conducted in a U.S population. We utilized a large sample size of newly diagnosed, histologically confirmed lung cancer cases and included detailed data on many potential risk factors, including dietary information from a validated FFQ. Finally, we conducted a thorough analysis (overall, stratified, and joint) matched on smoking status to address potential residual confounding by smoking status and other factors.
While this study provides the first quantitative assessment of the association between glycemic index and risk of lung cancer in a US population, there are several limitations that should be addressed. It is a retrospective case-control study, meaning recall and reporting bias and confounding are important considerations. It is possible that cases report their dietary intake differently from healthy controls. For example, healthy controls are more likely to recall healthy dietary habits than patients, leading to biased effect estimates. However, it is unlikely that cases versus controls differentially reported dietary consumption based on glycemic index values. Prospective cohort studies are required for estimating the causal association between diet and lung cancer. Smoking has been established as a cause of type II diabetes (57), and recent studies suggest an association between diabetes, diabetic medication (such as metformin), and lung cancer risk (58–61). Information regarding diabetes, hypertension and heart disease were not collected within the scope of the present study until recently, and we do not currently have information regarding diabetic medication. Given the link between high glycemic index diets (via insulin) and diabetes, this association warrants further attention in future studies. Additionally, FFQs are subject to random and systematic error and are therefore not believed to accurately measure individual dietary intake, but rather, rank individuals well based on their relative intake. It is also possible that the association between glycemic index and lung cancer varies by race and that the results of the present study, limited to non-Hispanic whites, are not generalizable to other ethnic or racial subgroups. This study provides additional evidence that diet may independently, and jointly with other risk factors, impact lung cancer etiology. Further research is necessary to understand the exact underlying mechanism for the association between dietary glycemic index and lung cancer risk.
Supplementary Material
Acknowledgments
This work was supported in part by grants from the National Institutes of Health (P50 CA070907, R01 CA176568) (X. Wu), Cancer Prevention & Research Institute of Texas (RP130502) (X. Wu), The University of Texas MD Anderson Cancer Center institutional support for the Center for Translational and Public Health Genomics and the NCI R25T Postdoctoral Fellowship in Cancer Prevention (S.C. Melkonian).
Footnotes
No conflicts of interest to report
References
- 1.American Cancer Society. Cancer Facts & Figures 2015. Atlanta, Ga: American CancerSociety; 2015. [Google Scholar]
- 2.American Cancer Society. Global Cancer Facts & Figures. 2. Atlanta: American Cancer Society; 2011. [Google Scholar]
- 3.Dela Cruz CS, Tanoue LT, Matthay RA. Lung cancer: epidemiology, etiology, and prevention. Clinics in chest medicine. 2011;32:605–44. doi: 10.1016/j.ccm.2011.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Fedirko V, Lukanova A, Bamia C, Trichopolou A, Trepo E, Nothlings U, et al. Glycemic index, glycemic load, dietary carbohydrate, and dietary fiber intake and risk of liver and biliary tract cancers in Western Europeans. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2013;24:543–53. doi: 10.1093/annonc/mds434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Giovannucci E. Insulin, insulin-like growth factors and colon cancer: a review of the evidence. The Journal of nutrition. 2001;131:3109S–20S. doi: 10.1093/jn/131.11.3109S. [DOI] [PubMed] [Google Scholar]
- 6.Hu J, La Vecchia C, Augustin LS, Negri E, de Groh M, Morrison H, et al. Glycemic index, glycemic load and cancer risk. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2013;24:245–51. doi: 10.1093/annonc/mds235. [DOI] [PubMed] [Google Scholar]
- 7.Galeone C, Augustin LS, Filomeno M, Malerba S, Zucchetto A, Pelucchi C, et al. Dietary glycemic index, glycemic load, and the risk of endometrial cancer: a case-control study and meta-analysis. European journal of cancer prevention: the official journal of the European Cancer Prevention Organisation. 2013;22:38–45. doi: 10.1097/CEJ.0b013e328354d378. [DOI] [PubMed] [Google Scholar]
- 8.Klement RJ, Kammerer U. Is there a role for carbohydrate restriction in the treatment and prevention of cancer? Nutrition & metabolism. 2011;8:75. doi: 10.1186/1743-7075-8-75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spitz MR, Barnett MJ, Goodman GE, Thornquist MD, Wu X, Pollak M. Serum insulin-like growth factor (IGF) and IGF-binding protein levels and risk of lung cancer: a case-control study nested in the beta-Carotene and Retinol Efficacy Trial Cohort. Cancer epidemiology, biomarkers & prevention. 2002;11:1413–8. [PubMed] [Google Scholar]
- 10.Yu H, Spitz MR, Mistry J, Gu J, Hong WK, Wu X. Plasma levels of insulin-like growth factor-I and lung cancer risk: a case-control analysis. Journal of the National Cancer Institute. 1999;91:151–6. doi: 10.1093/jnci/91.2.151. [DOI] [PubMed] [Google Scholar]
- 11.Flood A, Peters U, Jenkins DJ, Chatterjee N, Subar AF, Church TR, et al. Carbohydrate, glycemic index, and glycemic load and colorectal adenomas in the Prostate, Lung, Colorectal, and Ovarian Screening Study. The American journal of clinical nutrition. 2006;84:1184–92. doi: 10.1093/ajcn/84.5.1184. [DOI] [PubMed] [Google Scholar]
- 12.Aune D, Chan DS, Lau R, Vieira R, Greenwood DC, Kampman E, et al. Carbohydrates, glycemic index, glycemic load, and colorectal cancer risk: a systematic review and meta-analysis of cohort studies. Cancer causes & control: CCC. 2012;23:521–35. doi: 10.1007/s10552-012-9918-9. [DOI] [PubMed] [Google Scholar]
- 13.Franceschi S, Dal Maso L, Augustin L, Negri E, Parpinel M, Boyle P, et al. Dietary glycemic load and colorectal cancer risk. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2001;12:173–8. doi: 10.1023/a:1008304128577. [DOI] [PubMed] [Google Scholar]
- 14.Larsson SC, Bergkvist L, Wolk A. Glycemic load, glycemic index and carbohydrate intake in relation to risk of stomach cancer: a prospective study. International journal of cancer Journal international du cancer. 2006;118:3167–9. doi: 10.1002/ijc.21753. [DOI] [PubMed] [Google Scholar]
- 15.Augustin LS, Gallus S, Negri E, La Vecchia C. Glycemic index, glycemic load and risk of gastric cancer. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2004;15:581–4. doi: 10.1093/annonc/mdh130. [DOI] [PubMed] [Google Scholar]
- 16.Lazarevic K, Nagorni A, Jeremic M. Carbohydrate intake, glycemic index, glycemic load and risk of gastric cancer. Central European journal of public health. 2009;17:75–8. doi: 10.21101/cejph.a3501. [DOI] [PubMed] [Google Scholar]
- 17.Jiao L, Flood A, Subar AF, Hollenbeck AR, Schatzkin A, Stolzenberg-Solomon R. Glycemic index, carbohydrates, glycemic load, and the risk of pancreatic cancer in a prospective cohort study. Cancer epidemiology, biomarkers & prevention: a publication of the American Association for Cancer Research, cosponsored by the American Society of Preventive Oncology. 2009;18:1144–51. doi: 10.1158/1055-9965.EPI-08-1135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Heinen MM, Verhage BA, Lumey L, Brants HA, Goldbohm RA, van den Brandt PA. Glycemic load, glycemic index, and pancreatic cancer risk in the Netherlands Cohort Study. The American journal of clinical nutrition. 2008;87:970–7. doi: 10.1093/ajcn/87.4.970. [DOI] [PubMed] [Google Scholar]
- 19.Coleman HG, Kitahara CM, Murray LJ, Dodd KW, Black A, Stolzenberg-Solomon RZ, et al. Dietary carbohydrate intake, glycemic index, and glycemic load and endometrial cancer risk: a prospective cohort study. American journal of epidemiology. 2014;179:75–84. doi: 10.1093/aje/kwt222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu WH, Xiang YB, Zhang X, Ruan Z, Cai H, Zheng W, et al. Association of dietary glycemic index and glycemic load with endometrial cancer risk among Chinese women. Nutrition and cancer. 2015;67:89–97. doi: 10.1080/01635581.2015.976319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Augustin LS, Gallus S, Bosetti C, Levi F, Negri E, Franceschi S, et al. Glycemic index and glycemic load in endometrial cancer. International journal of cancer Journal international du cancer. 2003;105:404–7. doi: 10.1002/ijc.11089. [DOI] [PubMed] [Google Scholar]
- 22.Nagle CM, Kolahdooz F, Ibiebele TI, Olsen CM, Lahmann PH, Green AC, et al. Carbohydrate intake, glycemic load, glycemic index, and risk of ovarian cancer. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2011;22:1332–8. doi: 10.1093/annonc/mdq595. [DOI] [PubMed] [Google Scholar]
- 23.Augustin LS, Polesel J, Bosetti C, Kendall CW, La Vecchia C, Parpinel M, et al. Dietary glycemic index, glycemic load and ovarian cancer risk: a case-control study in Italy. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2003;14:78–84. doi: 10.1093/annonc/dkg022. [DOI] [PubMed] [Google Scholar]
- 24.Augustin LS, Galeone C, Dal Maso L, Pelucchi C, Ramazzotti V, Jenkins DJ, et al. Glycemic index, glycemic load and risk of prostate cancer. International journal of cancer Journal international du cancer. 2004;112:446–50. doi: 10.1002/ijc.20416. [DOI] [PubMed] [Google Scholar]
- 25.Shikany JM, Flood AP, Kitahara CM, Hsing AW, Meyer TE, Willcox BJ, et al. Dietary carbohydrate, glycemic index, glycemic load, and risk of prostate cancer in the Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial (PLCO) cohort. Cancer causes & control: CCC. 2011;22:995–1002. doi: 10.1007/s10552-011-9772-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Randi G, Ferraroni M, Talamini R, Garavello W, Deandrea S, Decarli A, et al. Glycemic index, glycemic load and thyroid cancer risk. Annals of oncology: official journal of the European Society for Medical Oncology/ESMO. 2008;19:380–3. doi: 10.1093/annonc/mdm433. [DOI] [PubMed] [Google Scholar]
- 27.De Stefani E, Deneo-Pellegrini H, Mendilaharsu M, Ronco A, Carzoglio JC. Dietary sugar and lung cancer: a case-control study in Uruguay. Nutrition and cancer. 1998;31:132–7. doi: 10.1080/01635589809514692. [DOI] [PubMed] [Google Scholar]
- 28.Ainsworth BE, Haskell WL, Whitt MC, Irwin ML, Swartz AM, Strath SJ, et al. Compendium of physical activities: an update of activity codes and MET intensities. Medicine and science in sports and exercise. 2000;32:S498–504. doi: 10.1097/00005768-200009001-00009. [DOI] [PubMed] [Google Scholar]
- 29.Block G, Coyle LM, Hartman AM, Scoppa SM. Revision of dietary analysis software for the Health Habits and History Questionnaire. American journal of epidemiology. 1994;139:1190–6. doi: 10.1093/oxfordjournals.aje.a116965. [DOI] [PubMed] [Google Scholar]
- 30.USDA Food and Nutrient Database for Dietary Studies, 4.1. Beltsville, MD: Agricultural Research Service, Food Surveys Research Group; 2010. [Google Scholar]
- 31.Willett WC, Howe GR, Kushi LH. Adjustment for total energy intake in epidemiologic studies. The American journal of clinical nutrition. 1997;65:1220S–8S. doi: 10.1093/ajcn/65.4.1220S. discussion 9S–31S. [DOI] [PubMed] [Google Scholar]
- 32.Foster-Powell K, Holt SH, Brand-Miller JC. International table of glycemic index and glycemic load values: 2002. The American journal of clinical nutrition. 2002;76:5–56. doi: 10.1093/ajcn/76.1.5. [DOI] [PubMed] [Google Scholar]
- 33.Flood A, Subar AF, Hull SG, Zimmerman TP, Jenkins DJ, Schatzkin A. Methodology for adding glycemic load values to the National Cancer Institute Diet History Questionnaire database. Journal of the American Dietetic Association. 2006;106:393–402. doi: 10.1016/j.jada.2005.12.008. [DOI] [PubMed] [Google Scholar]
- 34.Djiogue S, Nwabo Kamdje AH, Vecchio L, Kipanyula MJ, Farahna M, Aldebasi Y, et al. Insulin resistance and cancer: the role of insulin and IGFs. Endocrine-related cancer. 2013;20:R1–R17. doi: 10.1530/ERC-12-0324. [DOI] [PubMed] [Google Scholar]
- 35.Sakurai T, Kudo M. Signaling pathways governing tumor angiogenesis. Oncology. 2011;81(Suppl 1):24–9. doi: 10.1159/000333256. [DOI] [PubMed] [Google Scholar]
- 36.Pollak M. The insulin and insulin-like growth factor receptor family in neoplasia: an update. Nature reviews Cancer. 2012;12:159–69. doi: 10.1038/nrc3215. [DOI] [PubMed] [Google Scholar]
- 37.Karlstad O, Starup-Linde J, Vestergaard P, Hjellvik V, Bazelier MT, Schmidt MK, et al. Use of insulin and insulin analogs and risk of cancer - systematic review and meta-analysis of observational studies. Current drug safety. 2013;8:333–48. doi: 10.2174/15680266113136660067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yu H, Rohan T. Role of the insulin-like growth factor family in cancer development and progression. Journal of the National Cancer Institute. 2000;92:1472–89. doi: 10.1093/jnci/92.18.1472. [DOI] [PubMed] [Google Scholar]
- 39.Moschos SJ, Mantzoros CS. The role of the IGF system in cancer: from basic to clinical studies and clinical applications. Oncology. 2002;63:317–32. doi: 10.1159/000066230. [DOI] [PubMed] [Google Scholar]
- 40.Ryan PD, Goss PE. The emerging role of the insulin-like growth factor pathway as a therapeutic target in cancer. The oncologist. 2008;13:16–24. doi: 10.1634/theoncologist.2007-0199. [DOI] [PubMed] [Google Scholar]
- 41.DiGiovanni J, Kiguchi K, Frijhoff A, Wilker E, Bol DK, Beltran L, et al. Deregulated expression of insulin-like growth factor 1 in prostate epithelium leads to neoplasia in transgenic mice. Proceedings of the National Academy of Sciences of the United States of America. 2000;97:3455–60. doi: 10.1073/pnas.97.7.3455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen B, Liu S, Xu W, Wang X, Zhao W, Wu J. IGF-I and IGFBP-3 and the risk of lung cancer: a meta-analysis based on nested case-control studies. Journal of experimental & clinical cancer research: CR. 2009;28:89. doi: 10.1186/1756-9966-28-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Cao H, Wang G, Meng L, Shen H, Feng Z, Liu Q, et al. Association between circulating levels of IGF-1 and IGFBP-3 and lung cancer risk: a meta-analysis. PloS one. 2012;7:e49884. doi: 10.1371/journal.pone.0049884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Raffensperger S, Kuczmarski MF, Hotchkiss L, Cotugna N, Evans MK, Zonderman AB. Effect of race and predictors of socioeconomic status on diet quality in the HANDLS Study sample. Journal of the National Medical Association. 2010;102:923–30. doi: 10.1016/s0027-9684(15)30711-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Kant AK, Graubard BI. Secular trends in the association of socio-economic position with self-reported dietary attributes and biomarkers in the US population: National Health and Nutrition Examination Survey (NHANES) 1971–1975 to NHANES 1999–2002. Public health nutrition. 2007;10:158–67. doi: 10.1017/S1368980007246749. [DOI] [PubMed] [Google Scholar]
- 46.McCabe-Sellers BJ, Bowman S, Stuff JE, Champagne CM, Simpson PM, Bogle ML. Assessment of the diet quality of US adults in the Lower Mississippi Delta. The American journal of clinical nutrition. 2007;86:697–706. doi: 10.1093/ajcn/86.3.697. [DOI] [PubMed] [Google Scholar]
- 47.Hiscock R, Bauld L, Amos A, Fidler JA, Munafo M. Socioeconomic status and smoking: a review. Annals of the New York Academy of Sciences. 2012;1248:107–23. doi: 10.1111/j.1749-6632.2011.06202.x. [DOI] [PubMed] [Google Scholar]
- 48.Sakurai H, Asamura H, Watanabe S, Suzuki K, Tsuchiya R. Clinicopathologic features of peripheral squamous cell carcinoma of the lung. The Annals of thoracic surgery. 2004;78:222–7. doi: 10.1016/j.athoracsur.2004.01.029. [DOI] [PubMed] [Google Scholar]
- 49.Christensen KY, Naidu A, Parent ME, Pintos J, Abrahamowicz M, Siemiatycki J, et al. The risk of lung cancer related to dietary intake of flavonoids. Nutrition and cancer. 2012;64:964–74. doi: 10.1080/01635581.2012.717677. [DOI] [PubMed] [Google Scholar]
- 50.Byers TE, Graham S, Haughey BP, Marshall JR, Swanson MK. Diet and lung cancer risk: findings from the Western New York Diet Study. American journal of epidemiology. 1987;125:351–63. doi: 10.1093/oxfordjournals.aje.a114542. [DOI] [PubMed] [Google Scholar]
- 51.Morgillo F, Kim WY, Kim ES, Ciardiello F, Hong WK, Lee HY. Implication of the insulin-like growth factor-IR pathway in the resistance of non-small cell lung cancer cells to treatment with gefitinib. Clinical cancer research: an official journal of the American Association for Cancer Research. 2007;13:2795–803. doi: 10.1158/1078-0432.CCR-06-2077. [DOI] [PubMed] [Google Scholar]
- 52.Pillai RN, Ramalingam SS. Inhibition of insulin-like growth factor receptor: end of a targeted therapy? Translational lung cancer research. 2013;2:14–22. doi: 10.3978/j.issn.2218-6751.2012.11.05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kaklamani VG, Linos A, Kaklamani E, Markaki I, Mantzoros C. Age, sex, and smoking are predictors of circulating insulin-like growth factor 1 and insulin-like growth factor-binding protein 3. Journal of clinical oncology: official journal of the American Society of Clinical Oncology. 1999;17:813–7. doi: 10.1200/JCO.1999.17.3.813. [DOI] [PubMed] [Google Scholar]
- 54.Pampel FC, Krueger PM, Denney JT. Socioeconomic Disparities in Health Behaviors. Annual review of sociology. 2010;36:349–70. doi: 10.1146/annurev.soc.012809.102529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Boyle RG, O’Connor P, Pronk N, Tan A. Health behaviors of smokers, ex-smokers, and never smokers in an HMO. Preventive medicine. 2000;31:177–82. doi: 10.1006/pmed.2000.0699. [DOI] [PubMed] [Google Scholar]
- 56.Kvaavik E, Meyer HE, Tverdal A. Food habits, physical activity and body mass index in relation to smoking status in 40–42 year old Norwegian women and men. Preventive medicine. 2004;38:1–5. doi: 10.1016/j.ypmed.2003.09.020. [DOI] [PubMed] [Google Scholar]
- 57.The Health Consequences of Smoking-50 Years of Progress: A Report of the Surgeon General. Atlanta (GA): 2014. [Google Scholar]
- 58.Sakoda LC, Ferrara A, Achacoso NS, Peng T, Ehrlich SF, Quesenberry CP, Jr, et al. Metformin use and lung cancer risk in patients with diabetes. Cancer prevention research. 2015;8:174–9. doi: 10.1158/1940-6207.CAPR-14-0291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hall GC, Roberts CM, Boulis M, Mo J, MacRae KD. Diabetes and the risk of lung cancer. Diabetes care. 2005;28:590–4. doi: 10.2337/diacare.28.3.590. [DOI] [PubMed] [Google Scholar]
- 60.Lee JY, Jeon I, Lee JM, Yoon JM, Park SM. Diabetes mellitus as an independent risk factor for lung cancer: a meta-analysis of observational studies. European journal of cancer. 2013;49:2411–23. doi: 10.1016/j.ejca.2013.02.025. [DOI] [PubMed] [Google Scholar]
- 61.Tseng CH. Diabetes but not insulin increases the risk of lung cancer: a Taiwanese population-based study. PloS one. 2014;9:e101553. doi: 10.1371/journal.pone.0101553. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.