Abstract
Objective: Set within the context of the 2015 International Year of Light and Light-Based Technologies,and of a growing and aging world population with ever-rising healthcare needs, this perspective and mini-review focuses on photobiomodulation (PBM) therapy as an emerging, cost-effective, treatment option for cancer (i.e., solid tumors) and other complex diseases, particularly, of the eye (e.g., age-related macular degeneration, diabetic retinopathy, glaucoma, retinitis pigmentosa) and the central nervous system (e.g., Alzheimer's and Parkinson's disease). Background data: Over the last decades, primary and secondary mechanisms of PBM have been revealed. These include oxygen-dependent and oxygen-independent structural and functional action pathways. Signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a given set of parameters. Methods: This study was a perspective and nonsystematic literature mini-review. Results: Studies support what we describe as a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photo-cceptors to restore physiologic function. Conclusions: Based on existing evidence, it is argued that PBM therapy can raise the standard of care and improve the quality of life of patients for a fraction of the cost of many current approaches. PBM therapy can, therefore,benefit large, vulnerable population groups, including the elderly and the poor, whilehaving a major impact on medical practice and public finances.
Introduction
The United Nations declared 2015 to be the International Year of Light and Light-Based Technologies (IYL 2015) in recognition of the vital role of light-based systems in our daily lives, and their growing importance to meeting the world's challenges in areas as diverse as energy, education, telecommunication, agriculture, and health.1 Although our perception of light is often limited to the visible band of the electromagnetic (EM) spectrum,2 both lower and shorter wavelengths are increasingly used in new medical technologies3 including soft, injectable, and bioresorbable electronics.4 Described as an imperative cross-cutting discipline of in the twenty-first century, light science has already revolutionized the physical sciences and industry. The control of light at the nanoscale has unveiled a plethora of phenomena, leading to powerful new applications and setting high expectations for years to come.5 In particular, light's ability to control materials and transport coded signals forms the bases for many new photonic devices and systems, wherein photons act as tailor-made EM energy packets that can perform various functions.
Here, we describe a paradigm shift or “quantum leap” in the understanding and use of light and its interaction with water and other relevant photoacceptors to control biologic function in medicine through photobiomodulation (PBM) therapy. We propose that progress will lead to the imminent inception of PBM therapy as a mainstream treatment for multiple complex diseases, including solid tumors, as well as neurodegenerative diseases (NDs) of the eye and central nervous system (CNS)6–10 (Fig. 1). PBM therapy can raise the standard of care and improve the quality of life of patients at a fraction of the cost of many current approaches. Thus, a “quantum leap” in PBM therapy will benefit large and vulnerable population groups, including the elderly and the poor, while having a major impact on medical practice and public finances.11 This is particularly important because the high price of drug therapies, which can reach hundreds of thousands of dollars per year,12 as well as a growing and aging world population, are putting a severe strain on family and public finances around the world.13
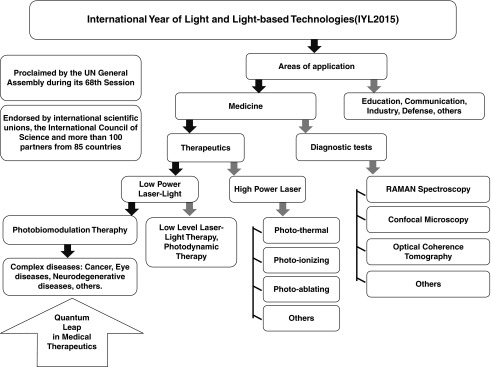
FIG. 1.
Flow chart illustrating fields of light-based technologies, highlighting photobiomodulation (PBM) therapy applied to complex diseases as a quantum leap in medical therapeutics.
Origin, Trajectory and Myriad Relationships in PBM's “Quantum Leap” in Medicine
Concurrent with progress in PBM therapy, a long history of discoveries has put medicine at the brink of a revolution in the use of light–water interactions for the treatment of complex diseases.7,8,10,14 Long ago, Albert Szent-Gyorgyi postulated that water was at the core of energy transfer in biological systems (i.e., quantum biology), and that that explained how energy from biomolecules could be translated into free energy for cells.15–17 Ling further elaborated on the physical state of water in living cells,18 and proposed on theoretical grounds that ordered layers of water could extend infinitely under ideal conditions.19,20 Later, Huber proposed a structural basis of light energy and electron transfer in biology.21 More recently, Zewail and others showed that, with rapid laser techniques, it is possible to “see” how atoms in a molecule move during a chemical reaction.22 Light science has now reached microscales at the limit of recordable physical observation (e.g., resonant intermolecular transfer of vibrational energy in water at −100 fs)23,24 showing, for example, the memory of persistent correlations in water structures within 50 fs, which is important in stabilizing biological systems.25 These and other tremendous achievements have changed our view of water, from a merely passive medium to an integral active player in the physiology of life, and have opened the gates to both direct measurement and control of physiological processes via light–water interaction.
State of the Art in PBM
In 2016, PBM therapy will be added to the MeSH database as an entry term for records spanning five decades of research.26 As argued by Anders et al., this is a key step, as it distinguishes PBM therapy from light-based devices used for heating of tissues, such as near infrared (NIR) lamps or other applications that rely on thermal effects for all or part of their mechanisms of action.26 In contrast, PBM therapy employs low-level monochromatic or quasimonochromatic light, currently from visible blue (∼400 nm) to far-infrared (FIR ∼3200 nm), to induce nonthermal (≤0.01°C) photochemical and photophysical effects. Nonlinear processes through which PBM therapy can stimulate or inhibit; that is, modulate, physiological activity depend upon signal-to-noise rate and target cell/tissue parameters.27–29 Thus, signal and target characteristics determine biological outcome, which is optimal (or even positive) only within a narrow set of parameters.13
Over the last decades, primary and secondary mechanisms of PBM at the tissue, cellular, and molecular levels have been revealed. These include two major structural and functional action pathways. The first, or classic, action pathway relates to oxygen-dependent mechanisms operated by oxidation-reduction enzymes of the respiratory chain, particularly cytochrome c oxidase (CcO), which is partly responsible for light energy absorption and transfer to cells and tissues.30 This pathway is associated to cofactors, pigments, metals, and proteins that act as key redox centers within the body's bioenergetic rack mechanism described by Huber.21 Nitric oxide (NO), as a first-level player, also has an activation and modulation role in the oxygen-dependent pathway.31–33
The second, or oxygen-independent, action pathway centers on the vital role of water not only as the prevalent medium of life but as an active molecule, capable of absorbing radiant energy (e.g., IR light) and transporting/transducing it along extended biological surfaces, from bulk water to confined water in nanoscopic tissue and cell spaces. Light–water dynamics precede/coexist with the classic oxygen-dependent action pathway and complement and facilitate energy transfer for increased adenosine triphosphate (ATP) production.29,34,35 As a point of comparison, correlated internal electron- and proton-transfer reactions have been tracked in real time into the oxidized enzyme (CcO), revealing an overall real time of 3.46 ms.36 This relay is slower by several orders of magnitude than total energy transport through water dynamics from bulk liquid water to confined spaces.34
Oxygen-independent light–water interactions may further power and modulate molecular signaling pathways and gene transcription factors via multiple nonmetabolic pathways.10,35 For examle, the energy of the drive force wave of an infrared pulsed laser device (IPLD) used in our group's previous studies (NIR 0.27 eV) is within the range of the strength of hydrogen bonds,29,37 and the IPLD carrier wave oscillates at a frequency (3x 10e6 Hz) that enters in vibrational resonance with the rate of electron transfer through the DNA double helix.29,37 Theoretical evidence suggests that these wave properties promote the activation of open state dynamics,38,39 allowing the activation of complex chaotic dynamics as well as the regulation of DNA replication and transcription, because the existence of open states in one place of the chain can influence the dynamics of other distant open states.29,34,35 Resulting effects match reported reductions in the frequency of chromosome aberrations induced by that low-energy laser irradiation,40 as well as theoretical,38,39 experimental,27,28 and clinical studies.41–48 These and other oxygen-independent PBM effects are channeled through metabolic control levels to regulate the energy-dependent path from the genotype to the phenotype.49,50
Light–Water Interactions and the Quantum Leap in PBM
We propose that the key to understanding and controlling the biophysics and biochemistry of higher-order organisms stems from their dual aqueous and energy-dependent nature. Water represents ∼70% by mass of an adult human body, or nearly 99% of total molecules by number, given water's low molecular weight. In addition, high-order organisms, including humans, can be represented as complex electrochemical (semiconducting) systems that comprise a vast array of energy-sensitive materials and machinery, such as ion pumps (e.g., chemically driven electron pumping through molecular wires, such as the D pathway in CcO),34 molecular motors (e.g., ATP synthase and Brownian biomotors), transistors-capacitors (e.g., cell membrane), liquid crystals (e.g., membrane structure), and rechargeable electrolytic biological batteries (e.g., hydrophilic interface in cells/tissues). Life system's double nature, whose two main structural and functional pillars are energy and water joined to biomolecules, has, therefore, tremendous consequences for life and health.
Water's permittivity, calculated considering the system as a plane capacitor, is generally high. Therefore, radiant energy can penetrate and be absorbed by tissues to provide powerful tools in medicine.51 One example is the exclusion zone (EZ) described by Pollack.52 High-energy EZ water forms along hydrophilic surfaces (e.g., tissue interfaces) in response to radiant energy.53 Remarkably, EZ water can separate and store electrical charges, and can release up to 70% of such charges when it is perturbed, such as by injury-induced redox potentials.54 We have argued that supplied energy can power and modulate cellular work and signaling pathways, even when the metabolic energy pathway has been compromised, steering cells toward or away from programmed cell death.34 EZ water may, thus, act as an electrolytic bio-battery,35 which can efficiently and selectively transfer energy to sites expressing redox injury potentials, as found in cancer and other complex diseases, triggering reparative and regenerative mechanisms that can lead to restoring homeostasis/homeokinesis and, ultimately, health.29,34,35
Experimentally, IR energy absorption by water has been recently modeled in a porcine model, confirming that absorption depends upon fluence and wavelength. Further, the higher the concentration of water in tissues, the higher IR energy absorption will be.55 This is consistent with controlled clinical studies in solid tumors and complex ophthalmic and neurologic diseases,9,46,56 as well as molecular, biochemical, biophysical, and metabolic mechanistic support for a quantum leap in medical therapeutics based on the simple, but powerful, idea that properly tailored light can power and modulate physiologically reparative mechanisms.30,57–62
Cancer and Tumor Microenvironments
The bases of our understanding of cancer are constantly being questioned and revised, leading to new treatment goals. In a paradigm-changing editorial, Prendergast recently argued that “disorders in microenvironment and peripheral systems that control cancer might increasingly be viewed as primary rather than secondary factors in the root nature of cancer as a clinical disease.” This constitutes “a crucial and radical distinction from prevailing thought, since it implies that cancer may be a symptom of an underlying clinical disorder, rather than the root problem itself that needs to be addressed.” 6,63
Prendergast further suggested that “effective treatment of cancer may not necessarily entail understanding or addressing this complexity, but mastering the use of tissue or systemic systems that have the inherent ability to do so.” Hence, a common thread linking emerging perspectives in oncology and PBM therapy may well be the restitution of tissue homeostasis-homeokinesis via light-energy supplementation, a microenvironment effect that comprises and extends the Warburg effect previously discussed by our group.57,64–67
Photobiomodulation and Cancer
As far back as 1964–1966, McGuff et al. showed 64,65 that “laser energy has a selective effect on certain malignant tumors, resulting in their progressive regression and ultimate dissolution.” Following years of controversy,66,67 editorials by Karu68 and Lanzafame11,69 now stress evidence supporting the potential anticancer effects of PBM.11,68,69 New data confirm that PBM under certain parameters is safe for use in cancer patients.60 This is in accord with clinical results from our group using the abovementioned proof of concept IPLD.4,44
A phase I trial in patients with advanced neoplasias demonstrated that the IPLD studied was safe for clinical use and improved performance status and quality of life.41 Antitumor activity was observed in 88.23% of patients with 10 years of follow-up.41
In that series, T2-weighted MRI data showed increased water content of tumor heterogeneities42,44 preceding tumor-volume reduction and a therapeutic anticancer effect.42,44 Structural, kinetic, and thermodynamic implications of these changes in water dynamics have been analyzed at the tissue, cell, and interstitial levels.27 In conjunction, selective activation of programmed cellular death [i.e., apoptosis, necrosis, and anoikis (cell death by loss of cell adhesion)] and cytomorphologic modification (e.g., reduced size, increased roundness, increased vacuoles) were documented in neoplastic cells, but not in peripheral tissues.8,42 Modulation of cluster of differentiation (CD)4 CD45RA+, CD25 activated, tumor necrosis factor alpha (TNF-α), and soluble interleukin (IL)-2 receptor (sIL-2R) was further documented.43 These hallmark results, supported by independent data,70–72 demonstrate that PBM therapy can modulate antitumor effects,6,8 in sharp contrast with long-held views.45,73,74 This evidence is also consistent with growing experimental and clinical reports from multiple other authors.60,75–82
PBM and Ophthalmic and Neurodegenerative Disorders
Recent evidence underscores common mechanisms between cancer and NDs of the eye and CNS. Research suggests that oxidative proteome damage may be the most likely cause of aging and age-related maladies such as cancer and other complex diseases, including NDs.83 Findings also show “common mechanisms of onset,” with a focus on genes such as DJ-1 and Myc-Modulator 1 (MM-1) and signaling pathways that contribute to the onset and pathogenesis of cancer and NDs such as retinitis pigmentosa (RP), Parkinson disease (PD), and cerebellar atrophy.”84 Finally, both disease groups are profoundly energetic in nature, featuring prominent deterioration of metabolic energy pathways.10
External light energy supplementation has been shown to generate neuroprotective, vasoprotective, baroprotective, immunomodulatory, and regenerative effects (Fig. 2). 47 We have documented that such effects may be activated and modulated locally and/or remotely via oxygen-dependent and oxygen-independent pathways that can encompass extended biologic surfaces and may even reach avascular eye tissues (i.e., cornea, lens, aqueous humor, and vitreous) noninvasively. Although a full elucidation of involved mechanisms escapes the scope of this perspective and mini-review, a very brief discussion of results from multiple authors is given subsequently.
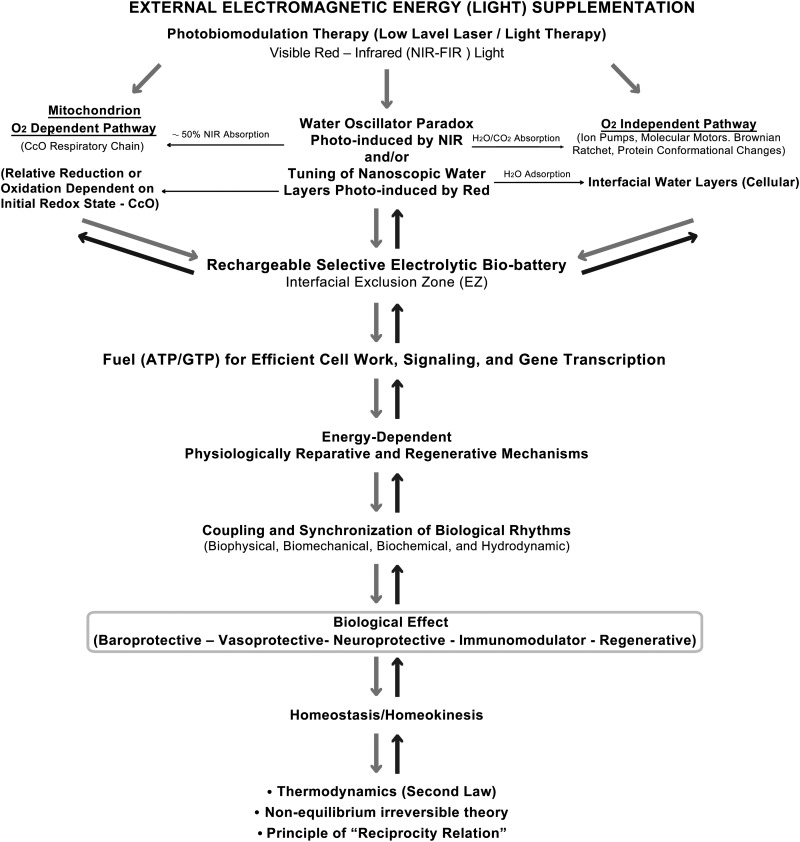
FIG. 2.
Electromagnetic (light) energy supplementation based on water–light interactions. Upper left side shows classic oxygen (O2) dependent pathways by which light energy generates adenosine triphosphate (ATP)/ guanosine-5′-triphosphate (GTP) and other high-energy molecules. Upper right side shows O2 independent pathways by which photoinduced, nonlinear, oscillations in water provide energy for cellular work, signaling, and gene transcription. Top center shows interfacial exclusion zone (EZ) water, which acts as a selective rechargeable electrolytic bio-battery. Together, these pathways activate and modulate physiologically reparative mechanisms which, at appropriate irradiation parameters, can generate neuroprotective, vasoprotective, baroprotective, immunomodulator, and regenerative effects locally and remotely, promoting homeostasis/homeokinesis through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by the second law of thermodynamics. Arrows point to the sequence and direction of events. (Updated from reference 47. Authors retained copyright.)
PBM has shown promise in the treatment of diabetic retinopathy (DR),85,86 age-related macular degeneration (AMD),46 glaucoma,47 RP,87 Stargardt disease,88 Leber's hereditary optic neuropathy,89 Alzheimer's disease (AD), and PD, 90,91 among other conditions.89 Strikingly, although each of these NDs has different etiologies and pathogeneses, “they frequently induce a set of cell signals that lead to well-established and similar morphological and functional changes, including programmed cell death. Furthermore, oxidative stress, activation of apoptotic pathways and inflammatory response, are common features in all these diseases.”92
Remarkably, PBM can modulate apoptosis as well as necrosis.42,45,47 PBM can also be both pro-oxidant in the short term, but antioxidant in the long term,93 thus modulating reactive oxygen species (ROS) generation. We also found clinical evidence of immune regulatory effects over inflammation during treatment of solid tumors with the IPLD, a NIR diode laser pulsed at a frequency of 3 MHz.43 These results are in agreement with the regulating role of the vagal reflex on the inflammatory reflex reported by Tracey, using an electronic device that stimulated nerves to treat inflammation.71,72
In addition, PBM has been shown to protect against retinal dysfunction and photoreceptor cell death in rodent models of retinal injury and retinal degeneration.94 PBM has been further reported to attenuate oxidative stress and inflammation in primary astrocytes induced by amyloid β peptide (Aβ),95 and to reduce Aβ-induced apoptosis,96 which is thought to play a major role in AD. Nevertheless, it has been argued that red to NIR light cannot be transmitted through the scalp to the brain more than a few centimeters,97 which makes it nearly impossible to noninvasively treat AD with PBM 98 using conventional (direct) delivery systems/methods. Similarly, although an absence of adverse effects from 670 and 830 nm PBM applied to the retina in Sprague Dawley albino rats has been reported,94 extreme care must be taken to avoid photodamage of the eye99 from direct PBM procedures.
Conversely, we published an interventional case report of a patient with bilateral geographic atrophic AMD (gaAMD) and associated neurologic disease treated noninvasively, indirectly, and at a distance (i.e., remotely) from ocular structures and the CNS with the above-referenced IPLD/photo-infrared pulsed bio-modulation (PIPBM).46 Results showed neurologic improvement, transitory color vision, enhanced visual acuity, full-field electroretinogram (ERG) modifications toward a normal rhythm, drusen mobilization, decreased lens opacity, and lower intraocular pressure (IOP), in accord with a retrospective noncomparative data analysis from the phase I trial of patients with advanced cancer treated with the IPLD,41 which showed statistically significant evidence of a therapeutic hypotensor effect over IOP,47 and they are consistent with the positive neurological evolution of two trial patients.
Moreover, although trial participants did not develop media opacity, one pre-existing incipient cataract in the right eye of a patient (transitional meningioma) became denser and slightly smaller 3 months post-treatment, and remained unchanged 1 year post-treatment. The left eye lens of the same patient was unaffected. Although the finding could be part of the natural history of the cataract, we stressed that possible deterministic effects related to the initial metabolic or biochemical state of lens opacities should be studied.41
In accordance with the what was described, a robust body of evidence suggests that protein misfolding, insolubility, and aggregation are at the root of both cataracts and other diseases including AD, PD, and Huntington's disease,100 and that external EM energy (light) supplementation can have reparative effect on protein misfolding, activating and modulating metabolic control levels of protein folding/unfolding.10,34 In addition, PBM effects on targets such as heat shock proteins (α crystalline), enzymes of the antioxidative system, Na+-K+-ATPase, Ca +2-ATPase, aquaporins (AQPs), and ion pumps have been referred to as part of mechanisms that could have influenced the response observed in the lens on the cases studied.46 We further proposed that, among other effects, PBM can stimulate and/or substitute ATP production via water dynamics, which is vital for the activation and inactivation kinetics in phototransduction.46 PBM can also affect the synthesis of molecules in a liquid crystalline (LC) state (e.g., self-assembly of lipids, water, and other biomolecules such as proteins and sterols, which are sensitive to temperature and/or electric fields) If confirmed, the latter may have multidisciplinary applications in medicine and biology in areas such as photovision, in which LCs are essential functional components.28
A first rapid communication referring to the retina and optic nerve additionally showed first evidence of EZ water as a selective rechargeable bio-battery applicable to PBM, suggesting a new understanding of the eye's energetic environment, which may have deep implications in ocular physiology as well as in the pathophysiology, diagnosis, and treatment of blinding diseases using light-based therapies.48 Therefore, as a promising alternative to drug therapies,101 or in combination with other treatments, PBM therapy may be developed into a viable therapeutic approach with multidisciplinary applications in ophthalmology and neuroscience,46 inducing and modulating physiologically reparative and regenerative effects that can favor homeostasis/homeokinesis27–29 through the coupling and synchronization of biophysical, biochemical, biomechanical, and hydrodynamic oscillators, as guided by thermodynamics.
Treatment Costs and Availability
At the 2015 American Society of Clinical Oncology (ASCO) annual meeting, Dr. Leonard Saltz, chief of gastrointestinal oncology at Memorial Sloan Kettering Cancer Center, discussed the high cost of cancer drugs. He argued that “the unsustainably high prices of cancer drugs is a big problem, and it's our problem,” citing as examples the cost of nivolumab ($28.78/mg) and ipilimumab ($157.46/mg), which is “approximately 4000 times the cost of gold.”102 Previously, >100 oncologists had protested the high price of cancer drugs, also calling them economically “unsustainable.” They noted that, of 12 cancer drugs approved in 2012, 11 were priced > $100,000 per year,103 with multiple drugs often being required for extended periods. Such high prices and their impact on families, governments, and society at large are leading some to propose that cost should be considered a “financial toxicity” to be assessed with other toxicities when treatments are considered by doctors and patients.104 In contrast, although it has been estimated the cost of developing new drug therapies can run up to USD $1.3–$1.7 billion,105,106 the development cost of new photonics devices can be substantially lower, which can lower therapy costs and increase treatment availability. For the same reasons, PBM can also offer a noninvasive and cost-effective therapeutic option for patients with NDs of the retina, brain, and beyond.56, 85
Conclusions
The celebration of the IYL 2015 by the United Nations1 is a fitting time to announce what we describe here as a “quantum leap” in PBM therapy. It is also a good opportunity to ensure that policy makers and the medical community become aware of and embrace the immense potential of light-based medical technologies, especially PBM therapy, as an emerging treatment option for cancer and other complex diseases.107 Although not all tissues respond to PBM therapy,69 in vitro and in vivo xenografts and evidence from clinical studies does suggest that it is time to begin considering PBM therapy as a potential drug equivalent.11,108 In addition, PBM therapy may have minimal or no adverse effects, improve quality of life and functional status and raise the current standard of care for many cancer patients when used alone or in combination with other therapies.9 PBM therapy further represents a novel hope for the treatment of numerous eye and neurologic diseases. And as stated, PBM may be developed at a lower cost than many current treatments,8,10 which can help meet the healthcare needs of an increasing and aging world population. As such, this perspective and mini-review focuses on the large potential tangible contributions of light-based therapies for large demographic segments of the population, such as aging “baby boomers” who are expected to face a higher incidence of diseases such as cancer, AMD, DR, glaucoma, RP, AD, and PD, as well as other neurologic diseases in the next 15 years. In light of the growing costs of drugs and their impact on developed and developing countries, we propose that PBM therapy may offer a novel, safe, and effective therapy choice that would be more accessible to large vulnerable groups, such as the poor and the elderly.
Concurrently with the United Nations' declaration of 2015 as the year of light and light-based technologies, PBM therapy stands at the brink of delivering a new generation of treatments for complex diseases. New PBM therapies will preserve quality of life and raise standard of care in an efficient and cost-efficient manner. This will particularly benefit the most vulnerable demographic sectors, such as the elderly and the poor, and reduce the strain of growing healthcare costs in both industrialized and developing countries. We propose that such developments and their imminent impact represent a paradigm shift or “quantum leap” in PBM therapy and medicine at large.
Acknowledgments
We thank Jesús Alberto Santana-Rodríguez for reviewing and editing this article, and Luis Rafael Santana-Rodríguez for design and technical support. This study was supported by Fundalas, Foundation for Interdisciplinary Research and Development.
Author Disclosure Statement
No competing financial interests exist.
References
- 1.Lightsource.org United Nations Educational, Scientific and Cultural Organization: International Year of Light, 2015. Available at: http://www.light2015.org/Home/About.html (Last accessed July26, 2015)
- 2.Osborne I. Light and optics. frontiers in light & optics. Introduction. Science 2015;348:514–515 [DOI] [PubMed] [Google Scholar]
- 3.Walmsley IA. Quantum optics: science and technology in a new light. Science 2015;348:525–330 [DOI] [PubMed] [Google Scholar]
- 4.Rogers JA. Electronics for humans body. JAMA 2015;313:561–562 [DOI] [PubMed] [Google Scholar]
- 5.Koenderink AF, Alù A, Polman A. Nanophotonics: shrinking light-based technology. Science 2015;348:516–521 [DOI] [PubMed] [Google Scholar]
- 6.Santana-Blank L, Rodríguez-Santana E, Santana Rodríguez KE. Concurrence of emerging developments in photobiomodulation and cancer. Photomed Laser Surg 2012;30:615–616 [DOI] [PubMed] [Google Scholar]
- 7.Rodríguez-Santana E, Santana-Blank L. Laser photobiomodulation as a potential multihallmark therapy for age-related macular degeneration. Photomed Laser Surg 2013;31:409–410 [DOI] [PubMed] [Google Scholar]
- 8.Santana-Blank L, Rodríguez-Santana E, Reyes H, Santana-Rodríguez JA, Santana-Rodríguez KE. Water–light interaction: a novel pathway for multi hallmark therapy in cancer. Int J Cancer Ther Oncol 2014;2:02012 [Google Scholar]
- 9.Santana-Blank L, Rodríguez-Santana E, Santana-Rodríguez JA, Santana-Rodríguez KE, Reyes H. Laser photobiomodulation as a potential multi-target anticancer therapy–review. J Solid Tumors 2013;3:50–62 [Google Scholar]
- 10.Santana-Blank L, Rodríguez-Santana E, Santana-Rodríguez KE, Santana-Rodríguez JA, Reyes H. Water's many roles in laser photobiomodulation. J Cancer Res Treat 2015;3:1–5 [Google Scholar]
- 11.Lanzafame RJ. One man's light: mechanistic convergence of photobiomodulation and biological effects. Photomed Laser Surg 2014;32:243–244 [DOI] [PubMed] [Google Scholar]
- 12.Lab-express-scripts.com. Express Script Holding Company, 2015. Available at: http://lab.express-scripts.com/drug-trendreport/introduction/year-in-review (Last accessed July26, 2015)
- 13.Santana-Blank L, Rodríguez-Santana E, Reyes H, Santana-Rodríguez JA, Santana-Rodríguez KE. Laser photobiomodulation: a new promising player for the multi–hallmark treatment of advanced cancer. Int J Cancer Ther Oncol 2013;1:6–8 [Google Scholar]
- 14.Rodríguez-Santana E, Santana-Blank L. Emerging evidence on the crystalline waterlight interface in ophthalmology and therapeutic implications in photobiomodulation: first communication. Photomed Laser Surg 2014;32:240–242 [DOI] [PubMed] [Google Scholar]
- 15.Szent-Györgyi A. Biology and pathology of water. Perspect Biol Med 1971;14:239–249 [DOI] [PubMed] [Google Scholar]
- 16.Szent-Györgyi A. Bioenergetics. Science 1956;124:873–987 [DOI] [PubMed] [Google Scholar]
- 17.Szent-Györgyi A. Biological oxidation and vitamins: Harvey Lecture, May 18, 1939. Bull N Y Acad Med 1939;15:456–468 [PMC free article] [PubMed] [Google Scholar]
- 18.Ling GN. The physical state of water in living cell and model systems. Ann N Y Acad Sci 1965;125:401–417 [DOI] [PubMed] [Google Scholar]
- 19.Ling GN. A new theoretical foundation for the polarized-oriented multilayer theory of cell water and for inanimate system demonstrating long–range dynamic structuring of eater molecules. Physiol Chem Phys Med NMR 2003;35:91–130 [PubMed] [Google Scholar]
- 20.Tychinsky V. High electric susceptibility is the signature of structure of structured water in water-containing objects. Water 2011;3:95–99 [Google Scholar]
- 21.Huber R. A structural basis of light energy and electrom transfer in biology. Nobel Lecture. Biosci Rep 1989;9:635–673 [DOI] [PubMed] [Google Scholar]
- 22.Nobelprize.org. Nobel Media AB, 1999. Available at: http://www.nobelprize.org/nobel_prizes/chemistry/laureates/1999/zewail-lecture.pdf (Last accessed July26, 2015)
- 23.Woutersen S, Bakker HJ. Resonant intermolecular transfer of vibrational energy in liquid water. Nature 1999;402:507–509
- 24.Liu L, Bakker HJ. Vibrational excitation induced proton transfer in hydrated Nafion membranes. J Phys Chem B 2015;119:2628–2637 [DOI] [PubMed] [Google Scholar]
- 25.Cowan ML, Bruner BD, Huse N, et al. Ultrafast memory loss and energy redistribution. Nature 2005;434:199–202 [DOI] [PubMed] [Google Scholar]
- 26.Anders JJ, Lanzafame RJ, Arany PR. Low-level light therapy versus photobiomodulation therapy. Photomed Laser Surg 2015;33:183–184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Santana-Blank LA, Rodríguez-Santana E, Scott-Algara D, Hunger M, Santana-Rodríguez KE, Orellana R. Short–term bioeffects of an infrared pulsed laser device on burned rat skin monitored by transverse relaxation times (NMR). Lasers Surg Med 2000;27:411–119 [DOI] [PubMed] [Google Scholar]
- 28.Rodríguez-Santana E, Santana-Blank LA, Reyes H, et al. H-NMR spin-lattice and correlation times of burned soft-tissue after treatment with an infrared pulsed laser device. Lasers Surg Med 2003;33:190–198 [DOI] [PubMed] [Google Scholar]
- 29.Santana-Blank LA, Rodríguez-Santana E, Santana-Rodríguez KE. Photo-infrared pulsed bio-modulation (PIPBM): a novel mechanism for the enhancement of physiologically reparative responses. Photomed Laser Surg 2005;24:416–424 [DOI] [PubMed] [Google Scholar]
- 30.Passarella S, Karu T. Absorption of monochromatic and narrow band radiation in the visible and near IR by both mitochondrial and non-mitochondrial photoacceptors results in photobiomodulation. J Photochem Photobiol B 2014;140:344–358 [DOI] [PubMed] [Google Scholar]
- 31.Unitt DC, Hollis VS, Palacios-Callender M, Frakich N, Moncada S. Inactivation of nitric oxide by cytochrome c oxidase under steady-state oxygen conditions. BiochimBiophys Acta 2010;1797:371–377 [DOI] [PubMed] [Google Scholar]
- 32.Ball KA, Castello PR, Poyton RO. Low intensity light stimulates nitrite-dependent nitric oxide synthesis but not oxygen consumption by cytochrome c oxidase: implications for phototherapy. J Photochem Photobiol B 2011;102:182–191 [DOI] [PubMed] [Google Scholar]
- 33.Lane N. New light on medicine. Sci Am 2003;288:38–45 [DOI] [PubMed] [Google Scholar]
- 34.Santana-Blank L, Rodríguez-Santana E, Santana-Rodríguez KE. Theoretic, experimental, clinical bases of the water oscillator hypothesis in near-infrared photobiomodulation. Photomed Laser Surg 2010;28:S41–S52 [DOI] [PubMed] [Google Scholar]
- 35.Santana-Blank L, Rodríguez-Santana E, Santana-Rodríguez KE. Photobiomodulation of aqueous interfaces as selective rechargeable bio-batteries in complex diseases: personal view, Photomed Laser Surg 2012;30:242–249 [DOI] [PubMed] [Google Scholar]
- 36.Belevich I, Bloch DA, Belevich N, Wikström M, Verkhovsky MI. Exploring the proton pump mechanism of cytochrome c oxidase in real time. Proc Natl Acad Sci U S A 2007;108:2685–2690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Porath D, Bezryadin A, De Vries S, Dekker C. Direct measurement of electrical transport through DNA molecules. Nature 2000;403:635–637 [DOI] [PubMed] [Google Scholar]
- 38.González JA, Martín-Landrove M. Solitons in a nonlinear DNA model. Phys Lett 1994;191:409–415 [Google Scholar]
- 39.González JA, Martin-Landrove M, Carbó JR, Chacón M. Bifurcations and chaos of DNA solitonic dynamics. Trieste: International Centre For Theoretical Physics, International Atomic Energy Agency, 1994 [Google Scholar]
- 40.Joyce KM, Downes CS, Hannigan BM. Radioadaptation in Indian muntjac fibroblast cells induced by low intensity laser irradiation. Mutat Res 1999;435:35–42 [DOI] [PubMed] [Google Scholar]
- 41.Santana-Blank LA, Rodríguez-Santana E, Vargas F, et al. Phase I trial of an infrared pulsed laser device in patients with advanced neoplasias. Clin Cancer Res 2002;8:3082–3091 [PubMed] [Google Scholar]
- 42.Santana-Blank LA, Rodríguez-Santana E, Vargas F, Santana-Rodríguez KE. Photoinduced cytomorphologic changes in an advanced cancer phase I clinical trial. Lasers Surg Med 2002;30:18–25 [DOI] [PubMed] [Google Scholar]
- 43.Santana-Blank LA, Castes M, Rojas ME, Vargas F, Scott-Algara D. Evaluation of serum levels of tumour necrosis factor-alpha (TNF-alpha) and soluble IL-2 receptor (sIL-2R) and CD4, CD8 and natural killer (NK) populations during infrared pulsed laser device (IPLD) treatment. Clin Exp Immunol 1992;90:43–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Santana-Blank LA, Reyes H, Rodríguez–Santana E, Santana-Rodríguez KE. Microdensitometry of T2-weighted magnetic resonance (MR) images from patients with advanced neoplasias in a phase I clinical trial of an infrared pulsed laser device (IPLD). Lasers Surg Med 2004;34:398–406 [DOI] [PubMed] [Google Scholar]
- 45.Santana-Blank L. Effects of an infrared pulsed laser device (IPLD) over apoptosis in cancer cells. Bioelectromagnetics 2005;26:523–524 [DOI] [PubMed] [Google Scholar]
- 46.Rodríguez-Santana E, Reyes H, Santana-Rodríguez KE, Santana-Blank L. Photoinfrared pulsed biomodulation in age-related macular degeneration associated to neurological disease: one interventional case report and mini-review. J Chin Clin Med 2008;3:470–477 [Google Scholar]
- 47.Rodríguez-Santana E, Santana-Blank LA, Santana-Rodríguez KE. Hypotensor effect of photo-infrared pulsed biomodulation over intraocular pressure. J Chin Clin Med 2011;6:147–157 [Google Scholar]
- 48.Rodríguez-Santana E, Santana-Blank L. Emerging evidence on the crystalline waterlight interface in ophthalmology and therapeutic implications in photobiomodulation: first communication. Photomed Laser Surg 2014;32:240–242 [DOI] [PubMed] [Google Scholar]
- 49.Strohman R. Maneuvering in the complex path from genotype to phenotype. Science 2002;296:701–703 [DOI] [PubMed] [Google Scholar]
- 50.Strohman R. Thermodynamics—old laws in medicine and complex disease. Nat Biotechnol 2003;21:477–479 [DOI] [PubMed] [Google Scholar]
- 51.Pendry JB, Luo Y, Zhao R. Transforming the optical landscape. Science 2015;348:521–524 [DOI] [PubMed] [Google Scholar]
- 52.Musumeci F, Pollack GH. High electrical permittivity of ultrapure water at the waterplatinum interface. Chem Phys Lett 2014;613:19–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pollack GH. Cell electrical properties: reconsidering the origin of the electrical potential. Cell Biol Int 2015;39:237–242 [DOI] [PubMed] [Google Scholar]
- 54.Pollack GH. Charging the water battery. In: The Enigma of Interfacial Water in the Fourth Phase of Water: Beyond Solid, Liquid, and Vapor. Pollack GH. (ed.). Seattle: Ebner and Sons, 2013, pp. 85–101 [Google Scholar]
- 55.Kim S, Shin S, Jeong S. Effects of tissue water content on the propagation of laser light during low-level laser therapy. J Biomed Opt 2015;20:051027. [DOI] [PubMed] [Google Scholar]
- 56.Rodríguez-Santana E, Santana-Blank L. Laser photobiomodulation as a potential multihallmark therapy for age-related macular degeneration. Photomed Laser Surg 2013;31:409–410 [DOI] [PubMed] [Google Scholar]
- 57.Santana-Blank L, Rodríguez-Santana E, Santana-Rodríguez JA, Santana-Rodríguez KE. Solid tumors and photobiomodulation: a novel approach to induce physiologically reparative homeostasis/homeokinesis-review. J Solid Tumors 2012;2:23–35 [Google Scholar]
- 58.Kim HP. Lightening up light therapy: activation of retrograde signaling pathway by photobiomodulation. Biomol Ther 2014;22:491–496 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vilar JMG, Rubi JM. Stochastic multiresonance. Phys Rev Lett 1997;78:2882 [Google Scholar]
- 60.Tanaka Y, Matsuo K, Yuzuriha S, Yan H, Nakayama J. Non-thermal cytocidal effect of infrared irradiation on cultured cancer cells using specialized device. Cancer Sci 2010;101:1396–1402 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rojas JC, Bruchey AK, Gonzalez–Lima F. Low-level light therapy improves cortical metabolic capacity and memory retention. J Alzheimers Dis 2012;32:741–752 [DOI] [PubMed] [Google Scholar]
- 62.Gonzalez-Lima F, Barksdale BR, Rojas JC. Mitochondrial respiration as a target for neuroprotection and cognitive enhancement. Biochem Pharmacol 2014;88:584–593 [DOI] [PubMed] [Google Scholar]
- 63.Prendergast GC. Perspectives on emerging trends in cancer research. Cancer Res 2011;71:2027–2028 [DOI] [PubMed] [Google Scholar]
- 64.McGuff PE, Deterling RA, Jr, Gottlieb LS. Tumoricidal effect of laser energy on experimental and human malignant tumors. N Engl J Med 1965;273:490–492 [DOI] [PubMed] [Google Scholar]
- 65.McGuff PE, Deterling RA, Jr, Gottlieb LS, Fahimi HD, Bushnell D, Roeber F. The laser treatment of experimental malignant tumours. Can Med Assoc J 1964;91:1089–1095 [PMC free article] [PubMed] [Google Scholar]
- 66.Basford JR. Editorial: low-energy laser therapy: controversies and new research findings. Lasers Surg Med 1989;9:1–5 [DOI] [PubMed] [Google Scholar]
- 67.Basford JR. Low-intensity laser therapy: still not an established clinical tool. Lasers Surg Med 1995;16:331–342 [DOI] [PubMed] [Google Scholar]
- 68.Karu T. Mitochondrial mechanisms of photobiomodulation in context of new data about multiple roles of ATP. Photomed Laser Surg 2010;28:159–160 [DOI] [PubMed] [Google Scholar]
- 69.Lanzafame RJ. Photobiomodulation: an enlightened path emerges. Photomed Laser Surg 2013;31:299–300 [DOI] [PubMed] [Google Scholar]
- 70.Coussens LM, Zitvogel L, Palucka AK. Neutralizing tumor-promoting chronic inflammation: a magic bullet? Science 2013;339:286–291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tracey KJ. The inflammatory reflex. Nature 2002;420:853–859 [DOI] [PubMed] [Google Scholar]
- 72.Tracey KJ. Shock medicine. Stimulation of the nervous system could replace drugs for inflammatory and acetoimmune conditions. Sci Am 2015;312:28–35 [Google Scholar]
- 73.Santana-Blank L. Contraindications in noninvasive laser therapy: truth and fiction. Photomed Laser Surg 2004;22:442. [DOI] [PubMed] [Google Scholar]
- 74.Lanzafame RJ. Photobiomodulation and cancer and other musings. Photomed Laser Surg 2011;29:3–4 [DOI] [PubMed] [Google Scholar]
- 75.Traitcheva N, Angelova P, Radeva M, Berg H. ELF fields and photooxidation yielding lethal effects on cancer cells. Bioelectromagnetics 2003;24:148–150 [DOI] [PubMed] [Google Scholar]
- 76.Radeva M, Berg H. Differences in lethality between cáncer cells and human lymphocytes caused by LF-electromagnetic fields. Bioelectromagnetics 2004;25:503–507 [DOI] [PubMed] [Google Scholar]
- 77.Wang F, Chen TS, Xing D, Wang JJ, Wu YX. Measuring dynamics of caspase-3 activity in living cells using FRET technique during apoptosis induced by high fluence low power laser irradiation. Lasers Surg Med 2005;36:2–7 [DOI] [PubMed] [Google Scholar]
- 78.Lane N. Power games. Nature 2006;443:901–903 [DOI] [PubMed] [Google Scholar]
- 79.Tata DB, Fahey M, Mitra K, Anders J, Waynant RW. Near-IR induced suppression of metabolic activity in aggressive cancers. Proc SPIE 2007;6428:64280E [Google Scholar]
- 80.Tata DB, Waynant RW. Laser light induced modulations in metabolic activities in human brain cancer. Proc SPIE 2008;6846:684607 [Google Scholar]
- 81.Zimin AA, Zhevago NA, Buĭniakova AI, Samoĭlova KA. Application of low-power visible and near infrared radiation in clinical oncology. Vopr Kurortol Fizioter Lech Fiz Kult 2000;6:49–52 [PubMed] [Google Scholar]
- 82.Myakishev–Rempel M, Stadler I, Brondon P, et al. A preliminary study of the safety of red light phototherapy of tissues harboring cancer. Photomed Laser Surg 2012;30:551–558 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell 2011;144:646–674 [DOI] [PubMed] [Google Scholar]
- 84.Ariga H. Common mechanisms of onset of cancer and neurodegenerative diseases. Biol Pharm Bull 2015;38:795–808 [DOI] [PubMed] [Google Scholar]
- 85.Tang J, Herda AA, Kern TS. Photobiomodulation in the treatment of patients with noncenter-involving diabetic macular oedema. Br J Ophthalmol 2014;98:1013–1015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Tang J, Du Y, Lee CA, Talahalli R, Eells JT, Kern TS. Low-intensity far-red light inhibits early lesions that contribute to diabetic retinopathy: in vivo and in vitro. Invest Ophthalmol Vis Sci 2013;54:3681–3690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gopalakrishnan S, Abroe B, Schmitt HM, et al. Different treatment paradigms of 830 nm photobiomodulation are retinoprotective in a rodent model of retinitis pigmentosa Invest Ophthalmol Vis Sci 2014;55:57–60 [Google Scholar]
- 88.Scalinci SZ, Milone F, Magnifico M, et al. Photobiomodulation (phototherapy) of retinal tissue in Stargardt disease. Program Number: 5665. Poster Board Number: C0055. ARVO Annual Meeting, Denver, Colorado, May 3–7, 2015 [Google Scholar]
- 89.Eells JT, Wong–Riley MT, VerHoeve J, et al. Mitochondrial signal transduction in accelerated wound and retinal healing by near-infrared light therapy. Mitochondrion 2004;4:559–567 [DOI] [PubMed] [Google Scholar]
- 90.Peoples C, Spana S, Ashkan K, et al. Photobiomodulation enhances nigral dopaminergic cell survival in a chronic MPTP mouse model of Parkinson's disease. Parkinsonism Relat Disord 2012;18:469–476 [DOI] [PubMed] [Google Scholar]
- 91.De Taboada L, Yu J, El-Amouri S, et al. Transcranial laser therapy attenuates amyloid–beta peptide neuropathology in amyloid–beta protein precursor transgenic mice. J Alzheimers Dis 2011;23:521–535 [DOI] [PubMed] [Google Scholar]
- 92.Cuenca N, Fernández–Sánchez L, Campello L, et al. Cellular responses following retinal injuries and therapeutic approaches for neurodegenerative diseases. Prog Retin Eye Res 2014;43:17–75 [DOI] [PubMed] [Google Scholar]
- 93.Chen A C-H, Huang Y-Y, Arany PR, Hamblin MR. Role of reactive oxygen species in low level light therapy. Proc SPIE 2009;7165:716502 [Google Scholar]
- 94.Eells JT, Abroe B, Schmitt HM, et al. NIR Photobiomodulation does not alter retinal function or morphology in nondystrophic Sprague Dawley rats. Control ID: 1896776. ARVO Annual Meeting, Denver, Colorado, May3–7, 2015 [Google Scholar]
- 95.Yang X, Askarova S, Sheng W, et al. Low energy laser light (632.8 nm) suppresses amyloid-β peptide-induced oxidative and inflammatory responses in astrocytes. Neuroscience 2010;171:859–868 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Liang J, Liu L, Xing D. Photobiomodulation by low-power laser irradiation attenuates Aβ-induced cell apoptosis through the Akt/GSK3β/β-catenin pathway. Free Radic Biol Med 2012;53:1459–1467 [DOI] [PubMed] [Google Scholar]
- 97.Haeussinger FB, Heinzel S, Hahn T, Schecklmann M, Ehlis A-C, Fallgatter AJ. Simulation of near infrared light absorption considering individual head and prefrontal cortex anatomy: implications for optical neuroimaging. PLoS ONE 2011;6:e26377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Farfara D, Tuby H, Trudler D, et al. Low-level laser therapy ameliorates disease progression in a mouse model of Alzheimer's disease. J Mol Neurosci 2015;55:430–436 [DOI] [PubMed] [Google Scholar]
- 99.Hamblin MR, Pires de Sousa MV, Arany PR, Carroll JD, Patthoff D. Low level laser (light) therapy and photobiomodulation: the path forward. Proc SPIE 2015;9309:930902 [Google Scholar]
- 100.Cetinel S, Unsworth L, Montemagno C. Peptide-based treatment strategies for cataract. J Glaucoma 2014;23:S73–S76 [DOI] [PubMed] [Google Scholar]
- 101.Purushothuman S, Johnstone DM, Nandasena C, Mitrofanis J, Stone J. Photobiomodulation with near infrared light mitigates Alzheimer's disease-related pathology in cerebral cortex – evidence from two transgenic mouse models. Alzheimers Res Ther 2014;6:2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Healio.com Healio, 2015. Available at: http://www.healio.com/hematology-oncology/practice management/news/online/%7Bc43d35c7-a65a-48ec-be2b-2f6434360412%7D/speakercost-of-cancer-drugs-a-major-problem (Last accessed July26, 2015)
- 103.Experts in Chronic Myeloid Leukemia. The price of drugs for chronic myeloid leukemia (CML) is a reflection of the unsustainable prices of cancer drugs: from the perspective of a large group of CML experts. Blood 2013;121:4439–4442 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Am.asco.org American Society of Clinical Oncology, 2015. Available at: https://am.asco.org/cost-cancer-drugs-should-bepart- treatment-decisions (Last accessed July26, 2015)
- 105.Collier R. Drug development cost estimates hard to swallow. CMAJ 2009;180:279–280 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Light DW, Lexchin JR. Pharmaceutical research and development: what do we get for all that money? Br Med J 2012;344:1–5 [DOI] [PubMed] [Google Scholar]
- 107.Lanzafame RJ. Janusian perenigrations and progress. Photomed Laser Surg 2015;33:1–2 [DOI] [PubMed] [Google Scholar]
- 108.Karu T. Is it time to consider photobiomodulation as a drug equivalent? Photomed Laser Surg 2013;31:189–191 [DOI] [PMC free article] [PubMed] [Google Scholar]