Abstract
Of the chronic diseases affecting grade-school children, asthma is the most common and accounts for the greatest number of school days missed. Moreover, it can influence family dynamics and function in other ways, and unfortunately, it can also be associated with mortality, particularly in the inner-city environments of the United States. Thus understanding factors that lead to its development in early life is essential in developing strategies aimed at primary prevention. Two risk factors that have been identified by a number of investigators include the development of allergic sensitization and wheezing respiratory tract illnesses caused by viruses and bacteria, either alone or in combination. Both of these factors appear to exert their influences within the first few years of life, such that asthma becomes established before the child enters grade school at age 5 to 6 years. Therefore, because both allergic sensitization and viral and bacterial illnesses can occur in children who do not have asthma, it is paramount to identify genetic and environmental factors that activate, interact with, and/or direct the immune system and components of the respiratory tract along pathways that allow asthma to become established and expressed clinically.
Key words: Asthma, viruses, bacteria, childhood, rhinovirus, respiratory syncytial virus, allergic sensitization, inflammation
Abbreviations used: COAST, Childhood Origins of Asthma; OR, Odds ratio; RSV, Respiratory syncytial virus
Information for Category 1 CME Credit
Credit can now be obtained, free for a limited time, by reading the review articles in this issue. Please note the following instructions.
Method of Physician Participation in Learning Process: The core material for these activities can be read in this issue of the Journal or online at the JACI Web site: www.jacionline.org. The accompanying tests may only be submitted online at www.jacionline.org. Fax or other copies will not be accepted.
Date of Original Release: March 2016. Credit may be obtained for these courses until February 28, 2017.
Copyright Statement: Copyright © 2016-2017. All rights reserved.
Overall Purpose/Goal: To provide excellent reviews on key aspects of allergic disease to those who research, treat, or manage allergic disease.
Target Audience: Physicians and researchers within the field of allergic disease.
Accreditation/Provider Statements and Credit Designation: The American Academy of Allergy, Asthma & Immunology (AAAAI) is accredited by the Accreditation Council for Continuing Medical Education (ACCME) to provide continuing medical education for physicians. The AAAAI designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™. Physicians should claim only the credit commensurate with the extent of their participation in the activity.
List of Design Committee Members: Daniel J. Jackson, MD, James E. Gern, MD, and Robert F. Lemanske, Jr, MD
Disclosure of Significant Relationships with Relevant Commercial
Companies/Organizations: D. J. Jackson has received research support from the National Institutes of Health (NIH) and has received consultancy fees from Vectura. J. E. Gern has received research support from the NIH, GlaxoSmithKline, and Merck and has received consultancy fees from GlaxoSmithKline, AstraZeneca, Boehringer Ingelheim, Genentech, Amgen, and Novartis. R. F. Lemanske has received travel support to attend board meetings; is employed by the University of Wisconsin; has received research support from the National Heart, Lung, and Blood Institute (Program Project Grant [COAST], AsthmaNet, Lung Development Grant [RO-1]) and Pharmaxis; has received consultancy fees from Merck, Sepracor, SA Boney and Associates, GlaxoSmithKline, the American Institute of Research, Genentech, Double Helix Development, and Boehringer Ingelheim; has received speakers' honoraria from Michigan Public Health, Allegheny General Hospital, AAP, West Allegheny Health, California Chapter 4, the Colorado Allergy Society, the Pennsylvania Allergy Society, Howard Pilgrim Health, the California Society of Allergy, the NYC Allergy Society, the World Allergy Organization, APAPARI, the Kuwait Allergy Society, Lurie Children's Hospital, Boston Children's Hospital, LA Children's Hospital, Northwestern University, the Western Society of Allergy, Asthma, and Immunology, and the Asthma and Allergy Foundation of America, Alaska Chapter; has received payment for manuscript preparation from the American Academy of Allergy, Asthma & Immunology; receives royalties from Elsevier and UpToDate; and has receives consultancy fees from Health Star Communications.
Activity Objectives:
-
1.
To understand that exposure to viral and bacterial pathogens is associated with recurrent wheezing and increased asthma risk in preschool-aged children.
-
2.
To determine the population at risk of developing asthma during childhood.
-
3.
To assess the effect of respiratory syncytial virus (RSV) infections on the future risk for asthma, and identify which patients would benefit from RSV prophylaxis.
-
4.
To understand the mechanisms of allergy-virus interactions and the association with allergic inflammation and asthma.
Recognition of Commercial Support: This CME activity has not received external commercial support.
List of CME Exam Authors: Jennifer Fergeson, DO, Adeeb Bulkhi, MD, Andrew Cooke, MD, Chen Hsing Lin, MD, Sultan Alandijani, MD, Peter Ricketti, DO, and Richard F. Lockey, MD.
Disclosure of Significant Relationships with Relevant Commercial
Companies/Organizations: The exam authors disclosed no relevant financial relationships.
Discuss this article on the JACI Journal Club blog: www.jaci-online.blogspot.com.
Of the various environmental risk factors demonstrated to be influential in the inception of childhood asthma, the development of allergic sensitization in early life,1 followed by wheezing respiratory tract illnesses2 caused by infection with various respiratory pathogens, appear to be the most consistent and reproducible.3 It has been shown that allergic sensitization has its greatest effect on recurrent wheezing and asthma when the process occurs in the first few years of life4, 5, 6 and when it involves development of sensitization to multiple aeroallergens.5, 6 Thus the strength of its influence appears not to be binary but more quantitative and developmental in nature.
Preschool wheezing illnesses with both viral7, 8, 9 and bacterial10, 11, 12 pathogens are also associated with recurrent wheezing and increased asthma risk. Historically, Respiratory syncytial virus (RSV) has been considered the most important respiratory pathogen in producing these long-term lower respiratory sequelae,13 and more recent data provide additional support for this relationship.14, 15 Furthermore, animal studies provide mechanistic evidence by which RSV can cause asthma.16 Treatment trials in preterm infants with palivizumab, an anti-RSV mAb, have demonstrated significant reductions in the development of recurrent wheezing.17
With the advent of molecular virology technology during the past few decades, human rhinovirus has been increasingly recognized as a major etiologic factor in preschool wheezing illnesses and a highly significant link with the risk of childhood asthma. Indeed, rhinovirus-induced wheezing illnesses are associated with an approximately 10-fold increase in asthma risk by 6 years of age,7 and potential mechanisms underlying these developments have been described.18 Certain rhinovirus species (eg, A and C > B) might be more virulent, thereby causing more lower respiratory tract illnesses19, 20, 21, 22 and increased morbidity, which result in emergency department visits and hospitalization.20, 23 Moreover, the development of recurrent lower respiratory tract symptoms after rhinovirus C infections can occur more frequently in subjects with demonstrable allergic sensitization.24
Airway bacteria in early life also influence asthma risk. Colonization with common respiratory pathogens, such as Streptococcus pneumoniae, Moraxella catarrhalis, and Haemophilus influenzae, in early life signals an increased risk for recurrent wheeze and early childhood asthma, and these same pathogens are associated with acute wheezing illnesses in young children. New tools to assess the airway microbiome are shedding additional light on relationships between beneficial versus pathogenic microbes and the risk for acute wheezing and asthma.
This article will expand on the contributions of allergic sensitization and viral and bacterial respiratory tract wheezing illnesses on increased risk for both recurrent preschool wheezing and childhood asthma. When possible, we will emphasize data generated in long-term longitudinal studies that have facilitated etiologic and epidemiologic characterizations.
Viruses, bacteria, and asthma development
Early-life viral infections and childhood asthma
Our understanding of the role of viral infections in wheezing has expanded with the development and evolution of molecular viral diagnostics. Results from studies using PCR and sequenced-based tests indicate that there is a long list of viruses that can cause upper and lower respiratory tract illnesses (Table I ),8, 25, 26, 27, 28, 29 and the clinical manifestations of infections with these different viruses are quite similar. Infections can lead to no symptoms at all, cold symptoms, or lower respiratory syndromes, such as pneumonitis, bronchiolitis, and acute wheezing illnesses. The risk of asthma seems to be most closely associated with the occurrence and frequency of virus-induced wheezing illnesses.7 Other clinical manifestations of acute viral illnesses that are associated with increased risk of asthma include fever and pneumonitis.4
Table I.
Viruses that can cause wheezing illnesses
| Virus | Major antigenic groups |
|---|---|
| Rhinovirus | A, B, C |
| RSV | A, B |
| Coronaviruses | NL63, HKU1, OC43, 229E |
| Metapneumovirus | A, B |
| Parainfluenza viruses | I, II, III, IV |
| Enterovirus | D68, others |
| Influenza viruses | A, B, C |
| Bocavirus | |
| Adenovirus | Serotypes 1, 2, 3, 5, 6, 7 |
| Polyomavirus | WU, KI, Malawi |
Nearly all wheezing illnesses in the first few years occur during viral respiratory tract infections, and the viruses most frequently associated with wheezing illnesses are RSV and rhinoviruses.28 Other viruses that are commonly detected during wheezing illnesses include metapneumovirus, coronaviruses, parainfluenza viruses, and bocaviruses. Coinfections with more than 1 virus are most commonly confirmed in infants and can be associated with more severe symptoms.8, 30 Prolonged illnesses can be caused by sequential infections with more than 1 virus, infections with a virus followed by a bacterial pathogen, or, less commonly, prolonged infection with a single virus.31 Adenoviruses are more likely than other respiratory viruses to cause prolonged viral shedding.32
RSV
RSV is an enveloped, nonsegmented, negative-strand RNA virus of the family Paramyxoviridae, and RSV infections are ubiquitous in infancy.33 RSV has 2 main antigenic groups (A and B). In general, RSV-B infections tend to cause less severe illnesses, but virulence varies considerably with individual strains of RSV, even within the antigenic groups. Most RSV infections cause mild or asymptomatic illnesses; it is estimated that only 9% of infections lead to outpatient clinic visits, and 5% or less of infections lead to hospitalization.34 Even so, RSV infections are the most common cause of bronchiolitis in the first year of life. Risk factors for RSV-induced bronchiolitis include prematurity; comorbid conditions affecting the heart, lungs, or immune system; and age and season of birth.35 The peak age for susceptibility to RSV lower respiratory tract infection is at 2 to 3 months of age after neutralizing antibody acquired transplacentally from the mother has waned.
RSV replicates in epithelial cells lining the upper airways and bronchioles and in type I pneumocytes. RSV infections that extend into the lower airways can cause airway narrowing and closure by causing epithelial cell necrosis and inducing cellular inflammation and mucus hypersecretion.36
There are several lines of evidence to suggest that more severe RSV infections in early life can contribute to the risk for childhood asthma. First, approximately one third of children with RSV-induced bronchiolitis have recurrent wheezing episodes. In turn, children with recurrent wheezing are at increased risk for subsequent asthma, especially if they have atopic parents or other atopic features.37 Furthermore, in a large Tennessee Medicaid database children who were born approximately 120 days before the peak RSV season were at increased risk for bronchiolitis and had a corresponding increase in the risk for asthma by age 3.5 to 5.5 years.15 Similarly, analysis of RSV-induced bronchiolitis in birth cohort studies indicates a positive association with the risk for asthma but not allergic sensitization.38, 39 Finally, analysis of the Tucson Children's Respiratory cohort through young adulthood revealed an interaction between RSV lower respiratory tract infection in infancy and smoking with respect to asthma. Active smoking was a significant risk factor (relative risk, 1.7; 95% CI, 1.2-2.3; P = .003) for asthma only in young adults with a history of RSV lower respiratory tract infection during infancy.40
Treatment with mAbs specific for RSV F protein (palivizumab and motavizumab) can reduce the risk of RSV-induced bronchiolitis, and several interventional studies have tested whether prevention of RSV-induced bronchiolitis in infancy leads to a reduction in recurrent wheezing and asthma. A prospective nonrandomized study of palivizumab administration to preterm infants in Canada and Europe reported found an 80% reduction in the risk of recurrent wheezing from ages 2 to 5 years in nonatopic children but no effect in atopic children.41 In a multicenter study conducted in The Netherlands, healthy preterm infants born at 33 to 35 weeks' gestation were enrolled in a double-blind, placebo-controlled trial of palivizumab prophylaxis and were monitored for 1 year to determine effects on recurrent wheeze. Palivizumab treatment led to a 61% relative reduction in total number of wheezing days in the first year of life.17 Similarly, a case-control study of palivizumab in Japanese infants with mild prematurity demonstrated reduced recurrent wheezing in the treated versus untreated groups (6.4% vs 18.9%, P < .001).42 The most recent study evaluated effects of motavizumab on 2127 healthy full-term Navajo infants, an ethnic group at high risk for bronchiolitis. In a double-blind, placebo-controlled randomized trial involving infants aged 6 months or less, motavizumab reduced by 87% the relative risk of hospital admission for bronchiolitis.43 However, motavizumab did not reduce subsequent rates of medically attended wheezing between the ages of 1 and 3 years (14.9% vs 14.0% in the placebo group).
Thus passive immunization against RSV certainly reduces acute bronchiolitis in preterm infants and likely reduces the rates of recurrent wheezing for premature infants. Whether RSV prophylaxis can reduce recurrent wheezing in term infants is unclear. Additional studies are needed to more clearly understand who might benefit from prophylaxis to reduce recurrent wheezing and also whether this approach can prevent atopic asthma. Given that children who become polysensitized to allergens in infancy are at greatest risk for persistent wheezing and reduced lung function,5, 17, 44 the latter question is of great clinical importance.
Rhinoviruses
Rhinoviruses consist of more than 160 individual types that are classified into 3 species (A, B, and C) based on viral genetics.45 Rhinoviruses are the pathogens most often associated with upper respiratory tract infections (common colds) and can also cause lower respiratory tract infection and wheezing illnesses.23, 46, 47 They are the second most common viruses associated with bronchiolitis and are the most common viruses detected in association with wheezing illnesses in children by the age of 1 to 2 years.48, 49, 50 The species of rhinovirus affects virulence: rhinoviruses A and C cause more severe illnesses in infants21, 24 and are more likely to cause lower respiratory illnesses.20, 23 Cadherin-related family member 3 (CDHR3) was recently identified as an entry factor on airway epithelial cells for rhinovirus C.51 Interestingly, a CDHR3 polymorphism is associated with the risk for childhood asthma, and the risk allele is associated with more CDHR3 on cell surfaces.52 This finding suggests the possibility that rhinovirus C infections in early life could contribute to asthma pathogenesis.
For years, it was mistakenly assumed that rhinovirus infections were confined to the upper airway. This assumption was based on early findings that rhinoviruses replicated best at 33°C to 35°C,53 and it was thought that higher temperatures in the lower airways would preclude lower airway infection. However, temperatures in the large and medium conducting airways are cooler than core temperature and are ideal for rhinovirus replication.54 Furthermore, some rhinovirus types, including C-species viruses, replicate equally well at 33°C and 37°C.55, 56 Finally, rhinovirus can be detected in sputum and bronchial biopsy specimens after experimental inoculation of the upper airway,57, 58, 59 which is concurrent with peak symptoms and reductions in peak expiratory flow.60 Rhinoviruses have also been detected in lower airway biopsy specimens from infants with recurrent wheezing.61 These findings provide strong evidence that rhinovirus can infect the lower airways, especially in young children.
Rhinovirus-induced wheezing illnesses in the first 2 to 3 years of life are closely associated with the risk of subsequent asthma. This relationship was first recognized in long-term studies of Finnish infants hospitalized for acute wheezing illnesses. Rhinovirus was the predominant virus detected during wheezing illnesses after 5 months of age. When reassessed 5 years later, 60% of children who wheezed with rhinovirus in the first 2 years of life had asthma.62 Similar findings were obtained at subsequent follow-up of these children in adolescence.63 Birth cohort studies have provided definitive evidence of a strong relationship between rhinovirus-induced wheezing and asthma. In the Childhood Origins of Asthma (COAST) study outpatient rhinovirus-induced wheezing illnesses during the first year of life were a significant predictor of recurrent wheezing through age 3 years.8 Furthermore, viral cause of wheezing illnesses differentially affected the risk for subsequent asthma; the risk of asthma was greater for children who wheezed with rhinovirus (odds ratio [OR], 9.8) or children who wheezed with rhinovirus and RSV (OR, 10.0) compared with that in children who wheezed only with RSV (OR, 2.6).7 Similar findings were reported in the Childhood Asthma Study in Perth: early-life infections with rhinovirus or RSV were associated with an increased risk of asthma but only in children who had aeroallergen sensitization by 2 years of age.4 In a study conducted in The Netherlands, rhinovirus-induced wheezing episodes were positively related to asthma risk. This relationship was partially explained by lower lung function in infancy being a risk factor for rhinovirus wheezing.64 Finally, in a collaborative study involving 2 birth cohorts (COAST and Copenhagen Prospective Studies on Asthma in Childhood), the 17q21 genotype strongly influenced both the risk of rhinovirus-induced wheezing and the probability that an infant with rhinovirus-induced wheezing would go on to have asthma.65 Thus there are unresolved questions as to whether rhinovirus-induced illnesses in childhood promote the development of asthma, reveal the presence of pre-existing asthma, or both.
Bacteria and wheezing illnesses
The previous sections reviewed information linking viral infection with wheezing illnesses and subsequent asthma. Remarkably, the association between airway bacteria and wheezing illnesses might be similarly robust,10 and this relationship could begin quite early in life. In a Danish birth cohort study colonization of the nasopharynx with S pneumoniae, M catarrhalis, and H influenzae at 1 month of age was associated with increased incidence of recurrent wheeze and asthma in early childhood.66
Viral infections can lead to changes in the composition of the airway microbiome and overgrowth of respiratory pathogens, and it is likely that both the virus and secondary changes in airway bacteria contribute respiratory symptoms and perhaps airway obstruction.11, 67, 68, 69 These findings suggest a potential role for antibiotic treatment for wheezing illnesses in early life; however, the evidence base for this approach appears to be mixed. On one hand, serious bacterial infections accompanying bronchiolitis appear to be rare,70 and a recent meta-analysis found that antibiotics, such as azithromycin or ampicillin, were not efficacious for treatment of bronchiolitis.71 On the other hand, Bacharier et al72 conducted a randomized multicenter clinical trial involving 607 preschool children with a history of recurrent wheezing illnesses who were then treated with azithromycin or placebo at the beginning of respiratory symptoms. Azithromycin treatment reduced the risk of progression to severe lower respiratory illness (hazard ratio, 0.64; 95% CI, 0.41-0.98; P = .04). The mechanism of effect could be related to either the antimicrobial or anti-inflammatory activities of macrolide antibiotics. Additional studies are needed to more clearly identify children most likely to respond to azithromycin therapy given the risks of selecting for antibiotic-resistant organisms and the theoretical risks of disturbing the microbiome during infancy.
Although bacterial pathogens are positively associated with wheezing and asthma, environmental exposure to a broad range of bacteria might protect against wheezing illnesses. For example, children exposed to animal sheds on farms are less likely to have wheezing in early life, including transient wheezing of presumed viral origin.73 These beneficial effects appear to be related to exposure to diverse environmental microbes. Indeed, animal models indicate that gastrointestinal administration of selected bacteria can provide protection against signs of illness after challenge with RSV.74 Furthermore, exposures in urban environments were related to the risk of recurrent wheeze in the Urban Environment and Childhood Asthma birth cohort study.75 Dust samples obtained during the first year of life were analyzed for both bacteria and allergen content. Infants with wheezing and atopy by age 3 years were most likely to grow up in homes with reduced exposure to both microbes and specific allergens (cat, mouse, and cockroach). These findings suggest that early-life exposures to bacteria and perhaps animal proteins modulate immune responses that are important determinants of respiratory illness severity.
Interactions between allergy and infections
Allergic sensitization, most notably to perennial aeroallergens, has been defined as a pivotal risk factor for the development of asthma.1 Recently, it has become evident that both the timing of allergic sensitization and the quantity of sensitization are important prognostic indicators. Simpson et al5 have identified an atopy phenotype in children they termed “multiple early sensitization.” In those children sensitized to multiple aeroallergens at an early age, Simpson et al reported a remarkable increase in risk for asthma inception, severe exacerbations leading to hospitalization, and impaired lung function. This link between early-life sensitization to multiple allergens and increased asthma risk has been replicated in additional cohort studies.6
This observed link between early sensitization to aeroallergens and asthma risk is in part due to synergy between allergic inflammation and viral infections, most commonly rhinovirus. Indeed, children in the COAST study who were sensitized to aeroallergens and wheezed with rhinovirus during the first 3 years of life had the greatest risk for asthma inception.7 This led to the question of whether viral illnesses in early life lead to allergic sensitization or whether the converse was true. Using a longitudinal multistate Markov model in the COAST study, Jackson et al2 identified a sequential relationship whereby allergic sensitization leads to viral wheezing. This relationship was strongest for rhinovirus-induced wheezing, and there was no evidence that viral wheezing led to sensitization.
There is also strong evidence to implicate allergic sensitization and exposure as a risk factor for wheezing with common cold infections later in childhood. In emergency department studies detection of a respiratory tract virus, most commonly rhinovirus, with detectable allergen-specific IgE and/or the presence of eosinophilic inflammation were all identified as risk factors for acute wheezing episodes.25, 76, 77 Notably, viral infections and allergic inflammation synergistically enhanced the risk of wheezing, and higher levels of allergen-specific IgE conferred the greatest risk. This synergism might be particularly notable for patients with rhinovirus C, in whom aeroallergen sensitization was recently reported as a risk factor for recurrent severe exacerbations leading to emergency department visits and hospitalization.24
Mechanisms of allergy-virus interactions
There are multiple mechanisms by which viral infections are thought to interact with allergic inflammation to lead to lower respiratory airway dysfunction, wheezing, and asthma exacerbations.78 First, underlying allergic inflammation can directly enhance airway responsiveness to rhinovirus infection.79 Additionally, viral infections can damage the barrier function of the airway epithelium, leading to enhanced absorption of aeroallergens across the airway wall and enhanced inflammation, whereas underlying allergic inflammation might also lead to enhanced viral replication.80, 81 Of interest, both rhinovirus infections60 and allergens82 can enhance airway epithelial cell production of IL-33, a recently identified innate cytokine, which promotes type 2 airway inflammation and remodeling. This steroid-resistant pathway has been reported to be upregulated in children with difficult-to-control asthma.82 Interestingly, IL-33 polymorphisms have been linked with intermediate and late-onset wheezing, which are strongly linked to early-life allergic sensitization.83 Another innate epithelial cytokine, IL-25, is also induced by rhinovirus and is likely to accentuate allergic airway inflammation in the context of rhinovirus infections in allergic subjects.84
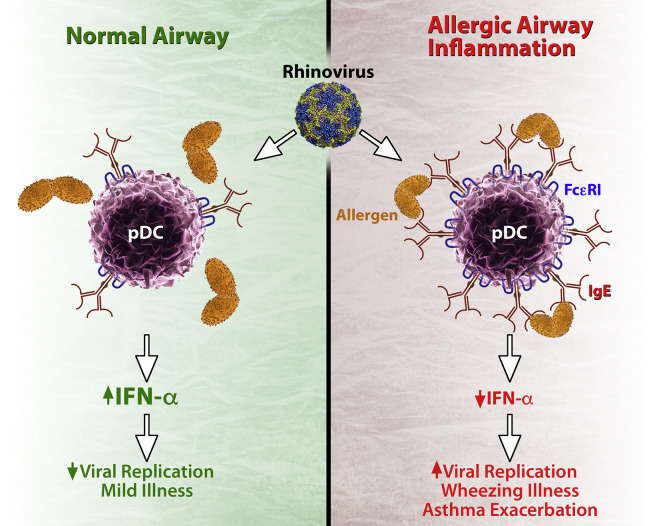
Finally, there is significant evidence that children with allergic asthma have impaired antiviral responses.85, 86, 87 Indeed, allergen exposure and high-affinity IgE receptor cross-linking has been shown to impair virus-induced type I and III interferon production in peripheral blood cells (Fig 1 ).88, 89 The result would be both enhanced viral replication and enhanced type 2 inflammation in the airway.90, 91
Fig 1.
Interplay between FcεRI and antiviral responses. In normal airways plasmacytoid dendritic cells (pDCs) are major sources of type I and type III interferons in response to viral infections. These cells also have low-level expression of FcεRI. In the context of allergy, pDCs express greater FcεRI, which is inversely related to interferon responses. In addition, cross-linking of FcεRI further reduces interferon responses. Consequences of suppressed interferon responses to viral infection could include increased viral replication, more severe illnesses with wheezing, and exacerbation of pre-existing asthma.
The most direct evidence to support the importance of allergen-virus interactions in patients with virus-induced wheezing and asthma exacerbations comes from a recent clinical trial of omalizumab (anti-IgE) to prevent seasonal virus-induced asthma exacerbations.92 In this trial virus-induced exacerbations were significantly reduced by omalizumab, and this reduction coincided with an enhanced type I interferon response ex vivo in rhinovirus-stimulated mononuclear cells. Of note, those participants who had larger increases in type I interferon response with omalizumab treatment had the greatest protection from virus-induced exacerbations.
Conclusion
Two key risk factors for the development of childhood asthma are the development of allergic sensitization in early life and wheezing respiratory tract illnesses caused primarily by viruses but also by bacteria either alone or as coinfections accompanying illnesses of viral cause. Therapies directed at these 2 risk factors, either alone or in combination, appear to be essential components to target for development of effective strategies for the primary prevention of asthma in children.
What do we know?
-
•
Early-life aeroallergen sensitization and viral wheezing illnesses are independent and synergistic risk factors for asthma inception.
-
•
Bacterial pathogens have recently emerged as an additional important contributor to asthma risk, either alone or as cofactors with viral infections.
What is still unknown?
-
•
Whether the prevention of allergic sensitization, antiviral strategies, immunostimulants, or other strategies that augment immune responses to airway pathogens will prevent the development of asthma.
Footnotes
Series editors: Donald Y. M. Leung, MD, PhD, and Dennis K. Ledford, MD
Supported by National Institutes of Health grants PO1HL070831, UM1AI114271, and U19AI104317.
References
- 1.Sly P.D., Boner A.L., Bjorksten B., Bush A., Custovic A., Eigenmann P.A. Early identification of atopy in the prediction of persistent asthma in children. Lancet. 2008;372:1100–1106. doi: 10.1016/S0140-6736(08)61451-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jackson D.J., Evans M.D., Gangnon R.E., Tisler C.J., Pappas T.E., Lee W.M. Evidence for a causal relationship between allergic sensitization and rhinovirus wheezing in early life. Am J Respir Crit Care Med. 2012;185:281–285. doi: 10.1164/rccm.201104-0660OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Holt P.G., Strickland D.H., Sly P.D. Virus infection and allergy in the development of asthma: what is the connection? Curr Opin Allergy Clin Immunol. 2012;12:151–157. doi: 10.1097/ACI.0b013e3283520166. [DOI] [PubMed] [Google Scholar]
- 4.Kusel M.M., de Klerk N.H., Kebadze T., Vohma V., Holt P.G., Johnston S.L. Early-life respiratory viral infections, atopic sensitization, and risk of subsequent development of persistent asthma. J Allergy Clin Immunol. 2007;119:1105–1110. doi: 10.1016/j.jaci.2006.12.669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Simpson A., Tan V.Y., Winn J., Svensen M., Bishop C.M., Heckerman D.E. Beyond atopy: multiple patterns of sensitization in relation to asthma in a birth cohort study. Am J Respir Crit Care Med. 2010;181:1200–1206. doi: 10.1164/rccm.200907-1101OC. [DOI] [PubMed] [Google Scholar]
- 6.Stoltz D.J., Jackson D.J., Evans M.D., Gangnon R.E., Tisler C.J., Gern J.E. Specific patterns of allergic sensitization in early childhood and asthma & rhinitis risk. Clin Exp Allergy. 2013;43:233–241. doi: 10.1111/cea.12050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jackson D.J., Gangnon R.E., Evans M.D., Roberg K.A., Anderson E.L., Pappas T.E. Wheezing rhinovirus illnesses in early life predict asthma development in high-risk children. Am J Respir Crit Care Med. 2008;178:667–672. doi: 10.1164/rccm.200802-309OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lemanske R.F., Jr., Jackson D.J., Gangnon R.E., Evans M.D., Li Z., Shult P.A. Rhinovirus illnesses during infancy predict subsequent childhood wheezing. J Allergy Clin Immunol. 2005;116:571–577. doi: 10.1016/j.jaci.2005.06.024. [DOI] [PubMed] [Google Scholar]
- 9.Jackson D.J. Early-life viral infections and the development of asthma: a target for asthma prevention? Curr Opin Allergy Clin Immunol. 2014;14:131–136. doi: 10.1097/ACI.0000000000000047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bisgaard H., Hermansen M.N., Bonnelykke K., Stokholm J., Baty F., Skytt N.L. Association of bacteria and viruses with wheezy episodes in young children: prospective birth cohort study. BMJ. 2010;341:c4978. doi: 10.1136/bmj.c4978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kloepfer K.M., Lee W.M., Pappas T.E., Kang T.J., Vrtis R.F., Evans M.D. Detection of pathogenic bacteria during rhinovirus infection is associated with increased respiratory symptoms and asthma exacerbations. J Allergy Clin Immunol. 2014;133:1301–1307. doi: 10.1016/j.jaci.2014.02.030. e1-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Mills K.H. Prior exposure to bacteria attenuates viral disease of the respiratory tract: a role for IL-17 and innate immune memory? Am J Respir Crit Care Med. 2014;189:126–128. doi: 10.1164/rccm.201312-2158ED. [DOI] [PubMed] [Google Scholar]
- 13.Sigurs N., Bjarnason R., Sigurbergsson F., Kjellman B. Respiratory syncytial virus bronchiolitis in infancy is an important risk factor for asthma and allergy at age 7. Am J Respir Crit Care Med. 2000;161:1501–1507. doi: 10.1164/ajrccm.161.5.9906076. [DOI] [PubMed] [Google Scholar]
- 14.James K.M., Gebretsadik T., Escobar G.J., Wu P., Carroll K.N., Li S.X. Risk of childhood asthma following infant bronchiolitis during the respiratory syncytial virus season. J Allergy Clin Immunol. 2013;132:227–229. doi: 10.1016/j.jaci.2013.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu P., Dupont W.D., Griffin M.R., Carroll K.N., Mitchel E.F., Gebretsadik T. Evidence of a causal role of winter virus infection during infancy in early childhood asthma. Am J Respir Crit Care Med. 2008;178:1123–1129. doi: 10.1164/rccm.200804-579OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Krishnamoorthy N., Khare A., Oriss T.B., Raundhal M., Morse C., Yarlagadda M. Early infection with respiratory syncytial virus impairs regulatory T cell function and increases susceptibility to allergic asthma. Nat Med. 2012;18:1525–1530. doi: 10.1038/nm.2896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Blanken M.O., Rovers M.M., Molenaar J.M., Winkler-Seinstra P.L., Meijer A., Kimpen J.L. Respiratory syncytial virus and recurrent wheeze in healthy preterm infants. N Engl J Med. 2013;368:1791–1799. doi: 10.1056/NEJMoa1211917. [DOI] [PubMed] [Google Scholar]
- 18.Jackson D.J. The role of rhinovirus infections in the development of early childhood asthma. Curr Opin Allergy Clin Immunol. 2010;10:133–138. doi: 10.1097/ACI.0b013e3283352f7c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Annamalay A.A., Khoo S.K., Jacoby P., Bizzintino J., Zhang G., Chidlow G. Prevalence of and risk factors for human rhinovirus infection in healthy aboriginal and non-aboriginal Western Australian children. Pediatr Infect Dis J. 2012;31:673–679. doi: 10.1097/INF.0b013e318256ffc6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bizzintino J., Lee W.M., Laing I.A., Vang F., Pappas T., Zhang G. Association between human rhinovirus C and severity of acute asthma in children. Eur Respir J. 2011;37:1037–1042. doi: 10.1183/09031936.00092410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee W.M., Lemanske R.F., Jr., Evans M.D., Vang F., Pappas T., Gangnon R. Human rhinovirus species and season of infection determine illness severity. Am J Respir Crit Care Med. 2012;186:886–891. doi: 10.1164/rccm.201202-0330OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Miller E.K., Williams J.V., Gebretsadik T., Carroll K.N., Dupont W.D., Mohamed Y.A. Host and viral factors associated with severity of human rhinovirus-associated infant respiratory tract illness. J Allergy Clin Immunol. 2011;127:883–891. doi: 10.1016/j.jaci.2010.11.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Iwane M.K., Prill M.M., Lu X., Miller E.K., Edwards K.M., Hall C.B. Human rhinovirus species associated with hospitalizations for acute respiratory illness in young US children. J Infect Dis. 2011;204:1702–1710. doi: 10.1093/infdis/jir634. [DOI] [PubMed] [Google Scholar]
- 24.Cox D.W., Bizzintino J., Ferrari G., Khoo S.K., Zhang G., Whelan S. Human rhinovirus species C infection in young children with acute wheeze is associated with increased acute respiratory hospital admissions. Am J Respir Crit Care Med. 2013;188:1358–1364. doi: 10.1164/rccm.201303-0498OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Heymann P.W., Carper H.T., Murphy D.D., Platts-Mills T.A., Patrie J., McLaughlin A.P. Viral infections in relation to age, atopy, and season of admission among children hospitalized for wheezing. J Allergy Clin Immunol. 2004;114:239–247. doi: 10.1016/j.jaci.2004.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kusel M.M., de Klerk N.H., Holt P.G., Kebadze T., Johnston S.L., Sly P.D. Role of respiratory viruses in acute upper and lower respiratory tract illness in the first year of life: a birth cohort study. Pediatr Infect Dis J. 2006;25:680–686. doi: 10.1097/01.inf.0000226912.88900.a3. [DOI] [PubMed] [Google Scholar]
- 27.Allander T., Jartti T., Gupta S., Niesters H.G., Lehtinen P., Osterback R. Human bocavirus and acute wheezing in children. Clin Infect Dis. 2007;44:904–910. doi: 10.1086/512196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jartti T., Lehtinen P., Vuorinen T., Osterback R., van den H.B., Osterhaus A.D. Respiratory picornaviruses and respiratory syncytial virus as causative agents of acute expiratory wheezing in children. Emerg Infect Dis. 2004;10:1095–1101. doi: 10.3201/eid1006.030629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rockett R.J., Bialasiewicz S., Mhango L., Gaydon J., Holding R., Whiley D.M. Acquisition of human polyomaviruses in the first 18 months of life. Emerg Infect Dis. 2015;21:365–367. doi: 10.3201/eid2102.141429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Papadopoulos N.G., Moustaki M., Tsolia M., Bossios A., Astra E., Prezerakou A. Association of rhinovirus infection with increased disease severity in acute bronchiolitis. Am J Respir Crit Care Med. 2002;165:1285–1289. doi: 10.1164/rccm.200112-118BC. [DOI] [PubMed] [Google Scholar]
- 31.Jartti T., Lee W.M., Pappas T., Evans M., Lemanske R.F., Jr., Gern J.E. Serial viral infections in infants with recurrent respiratory illnesses. Eur Respir J. 2008;32:314–320. doi: 10.1183/09031936.00161907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kalu S.U., Loeffelholz M., Beck E., Patel J.A., Revai K., Fan J. Persistence of adenovirus nucleic acids in nasopharyngeal secretions: a diagnostic conundrum. Pediatr Infect Dis J. 2010;29:746–750. doi: 10.1097/INF.0b013e3181d743c8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ruuskanen O., Ogra P.L. Respiratory syncytial virus. Curr Probl Pediatr. 1993;23:50–79. doi: 10.1016/0045-9380(93)90003-U. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Carroll K.N., Wu P., Gebretsadik T., Griffin M.R., Dupont W.D., Mitchel E.F. The severity-dependent relationship of infant bronchiolitis on the risk and morbidity of early childhood asthma. J Allergy Clin Immunol. 2009;123:1055–1061. doi: 10.1016/j.jaci.2009.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Geevarghese B., Simoes E.A. Antibodies for prevention and treatment of respiratory syncytial virus infections in children. Antivir Ther. 2012;17:201–211. doi: 10.3851/IMP2061. [DOI] [PubMed] [Google Scholar]
- 36.Habibi M.S., Openshaw P.J. Benefit and harm from immunity to respiratory syncytial virus: implications for treatment. Curr Opin Infect Dis. 2012;25:687–694. doi: 10.1097/QCO.0b013e32835a1d92. [DOI] [PubMed] [Google Scholar]
- 37.Castro-Rodriguez J.A., Holberg C.J., Wright A.L., Martinez F.D. A clinical index to define risk of asthma in young children with recurrent wheezing. Am J Respir Crit Care Med. 2000;162:1403–1406. doi: 10.1164/ajrccm.162.4.9912111. [DOI] [PubMed] [Google Scholar]
- 38.Stein R.T., Sherrill D., Morgan W.J., Holberg C.J., Halonen M., Taussig L.M. Respiratory syncytial virus in early life and risk of wheeze and allergy by age 13 years. Lancet. 1999;354:541–545. doi: 10.1016/S0140-6736(98)10321-5. [DOI] [PubMed] [Google Scholar]
- 39.Henderson J., Hilliard T.N., Sherriff A., Stalker D., Al Shammari N., Thomas H.M. Hospitalization for RSV bronchiolitis before 12 months of age and subsequent asthma, atopy and wheeze: a longitudinal birth cohort study. Pediatr Allergy Immunol. 2005;16:386–392. doi: 10.1111/j.1399-3038.2005.00298.x. [DOI] [PubMed] [Google Scholar]
- 40.Voraphani N., Stern D.A., Wright A.L., Guerra S., Morgan W.J., Martinez F.D. Risk of current asthma among adult smokers with respiratory syncytial virus illnesses in early life. Am J Respir Crit Care Med. 2014;190:392–398. doi: 10.1164/rccm.201311-2095OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Simoes E.A., Carbonell-Estrany X., Rieger C.H., Mitchell I., Fredrick L., Groothuis J.R. The effect of respiratory syncytial virus on subsequent recurrent wheezing in atopic and nonatopic children. J Allergy Clin Immunol. 2010;126:256–262. doi: 10.1016/j.jaci.2010.05.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yoshihara S., Kusuda S., Mochizuki H., Okada K., Nishima S., Simoes E.A. Effect of palivizumab prophylaxis on subsequent recurrent wheezing in preterm infants. Pediatrics. 2013;132:811–818. doi: 10.1542/peds.2013-0982. [DOI] [PubMed] [Google Scholar]
- 43.O'Brien K.L., Chandran A., Weatherholtz R., Jafri H.S., Griffin M.P., Bellamy T. Efficacy of motavizumab for the prevention of respiratory syncytial virus disease in healthy Native American infants: a phase 3 randomised double-blind placebo-controlled trial. Lancet Infect Dis. 2015;15:1398–1408. doi: 10.1016/S1473-3099(15)00247-9. [DOI] [PubMed] [Google Scholar]
- 44.Havstad S., Johnson C.C., Kim H., Levin A.M., Zoratti E.M., Joseph C.L. Atopic phenotypes identified with latent class analyses at age 2 years. J Allergy Clin Immunol. 2014;134:722–727.e2. doi: 10.1016/j.jaci.2014.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Gern J.E. The ABCs of rhinoviruses, wheezing, and asthma. J Virol. 2010;84:7418–7426. doi: 10.1128/JVI.02290-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Peltola V., Jartti T., Putto-Laurila A., Mertsola J., Vainionpaa R., Waris M. Rhinovirus infections in children: a retrospective and prospective hospital-based study. J Med Virol. 2009;81:1831–1838. doi: 10.1002/jmv.21590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gern J.E., Palmenberg A.C. Rhinoviruses. In: Knipe D.M., Howley P., editors. Fields virology. Lippencott Williams and Wilkins; Philadelphia: 2013. pp. 531–549. [Google Scholar]
- 48.Turunen R., Koistinen A., Vuorinen T., Arku B., Soderlund-Venermo M., Ruuskanen O. The first wheezing episode: respiratory virus etiology, atopic characteristics and illness severity. Pediatr Allergy Immunol. 2014;25:796–803. doi: 10.1111/pai.12318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Hasegawa K., Mansbach J.M., Camargo C.A., Jr. Infectious pathogens and bronchiolitis outcomes. Expert Rev Anti Infect Ther. 2014;12:817–828. doi: 10.1586/14787210.2014.906901. [DOI] [PubMed] [Google Scholar]
- 50.Miller E.K., Gebretsadik T., Carroll K.N., Dupont W.D., Mohamed Y.A., Morin L.L. Viral etiologies of infant bronchiolitis, croup and upper respiratory illness during 4 consecutive years. Pediatr Infect Dis J. 2013;32:950–955. doi: 10.1097/INF.0b013e31829b7e43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bochkov Y.A., Watters K., Ashraf S., Griggs T.F., Devries M.K., Jackson D.J. Cadherin-related family member 3, a childhood asthma susceptibility gene product, mediates rhinovirus C binding and replication. Proc Natl Acad Sci U S A. 2015;112:5485–5490. doi: 10.1073/pnas.1421178112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bonnelykke K., Sleiman P., Nielsen K., Kreiner-Moller E., Mercader J.M., Belgrave D. A genome-wide association study identifies CDHR3 as a susceptibility locus for early childhood asthma with severe exacerbations. Nat Genet. 2014;46:51–55. doi: 10.1038/ng.2830. [DOI] [PubMed] [Google Scholar]
- 53.Stott E.J., Killington R.A. Rhinoviruses. Annu Rev Microbiol. 1972;26:503–524. doi: 10.1146/annurev.mi.26.100172.002443. [DOI] [PubMed] [Google Scholar]
- 54.McFadden E.R., Jr., Pichurko B.M., Bowman H.F., Ingenito E., Burns S., Dowling N. Thermal mapping of the airways in humans. J Appl Physiol. 1985;58:564–570. doi: 10.1152/jappl.1985.58.2.564. [DOI] [PubMed] [Google Scholar]
- 55.Ashraf S., Brockman-Schneider R., Bochkov Y.A., Pasic T.R., Gern J.E. Biological characteristics and propagation of human rhinovirus-C in differentiated sinus epithelial cells. Virology. 2013;436:143–149. doi: 10.1016/j.virol.2012.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Papadopoulos N.G., Sanderson G., Hunter J., Johnston S.L. Rhinoviruses replicate effectively at lower airway temperatures. J Med Virol. 1999;58:100–104. doi: 10.1002/(sici)1096-9071(199905)58:1<100::aid-jmv16>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 57.Gern J.E., Galagan D.M., Jarjour N.N., Dick E.C., Busse W.W. Detection of rhinovirus RNA in lower airway cells during experimentally-induced infection. Am J Respir Crit Care Med. 1997;155:1159–1161. doi: 10.1164/ajrccm.155.3.9117003. [DOI] [PubMed] [Google Scholar]
- 58.Papadopoulos N.G., Bates P.J., Bardin P.G., Papi A., Leir S.H., Fraenkel D.J. Rhinoviruses infect the lower airways. J Infect Dis. 2000;181:1875–1884. doi: 10.1086/315513. [DOI] [PubMed] [Google Scholar]
- 59.Mosser A.G., Vrtis R., Burchell L., Lee W.M., Dick C.R., Weisshaar E. Quantitative and qualitative analysis of rhinovirus infection in bronchial tissues. Am J Respir Crit Care Med. 2005;171:645–651. doi: 10.1164/rccm.200407-970OC. [DOI] [PubMed] [Google Scholar]
- 60.Jackson D.J., Makrinioti H., Rana B.M., Shamji B.W., Trujillo-Torralbo M.B., Footitt J. IL-33-dependent type 2 inflammation during rhinovirus-induced asthma exacerbations in vivo. Am J Respir Crit Care Med. 2014;190:1373–1382. doi: 10.1164/rccm.201406-1039OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Malmstrom K., Pitkaranta A., Carpen O., Pelkonen A., Malmberg L.P., Turpeinen M. Human rhinovirus in bronchial epithelium of infants with recurrent respiratory symptoms. J Allergy Clin Immunol. 2006;118:591–596. doi: 10.1016/j.jaci.2006.04.032. [DOI] [PubMed] [Google Scholar]
- 62.Kotaniemi-Syrjanen A., Vainionpaa R., Reijonen T.M., Waris M., Korhonen K., Korppi M. Rhinovirus-induced wheezing in infancy—the first sign of childhood asthma? J Allergy Clin Immunol. 2003;111:66–71. doi: 10.1067/mai.2003.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hyvarinen M.K., Kotaniemi-Syrjanen A., Reijonen T.M., Korhonen K., Korppi M.O. Teenage asthma after severe early childhood wheezing: an 11-year prospective follow-up. Pediatr Pulmonol. 2005;40:316–323. doi: 10.1002/ppul.20273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.van der Gugten A.C., van der Zalm M.M., Uiterwaal C.S., Wilbrink B., Rossen J.W., van der Ent C.K. Human rhinovirus and wheezing: short and long-term associations in children. Pediatr Infect Dis J. 2013;32:827–833. doi: 10.1097/INF.0b013e318290620e. [DOI] [PubMed] [Google Scholar]
- 65.Caliskan M., Bochkov Y.A., Kreiner-Moller E., Bonnelykke K., Stein M.M., Du G. Rhinovirus wheezing illness and genetic risk of childhood onset asthma. N Engl J Med. 2013;368:1398–1407. doi: 10.1056/NEJMoa1211592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bisgaard H., Hermansen M.N., Buchvald F., Loland L., Halkjaer L.B., Bonnelykke K. Childhood asthma after bacterial colonization of the airway in neonates. N Engl J Med. 2007;357:1487–1495. doi: 10.1056/NEJMoa052632. [DOI] [PubMed] [Google Scholar]
- 67.Franz A., Adams O., Willems R., Bonzel L., Neuhausen N., Schweizer-Krantz S. Correlation of viral load of respiratory pathogens and co-infections with disease severity in children hospitalized for lower respiratory tract infection. J Clin Virol. 2010;48:239–245. doi: 10.1016/j.jcv.2010.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lehtinen P., Jartti T., Virkki R., Vuorinen T., Leinonen M., Peltola V. Bacterial coinfections in children with viral wheezing. Eur J Clin Microbiol Infect Dis. 2006;25:463–469. doi: 10.1007/s10096-006-0166-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Esposito S., Zampiero A., Terranova L., Ierardi V., Ascolese B., Daleno C. Pneumococcal bacterial load colonization as a marker of mixed infection in children with alveolar community-acquired pneumonia and respiratory syncytial virus or rhinovirus infection. Pediatr Infect Dis J. 2013;32:1199–1204. doi: 10.1097/INF.0b013e31829ec274. [DOI] [PubMed] [Google Scholar]
- 70.Librizzi J., McCulloh R., Koehn K., Alverson B. Appropriateness of testing for serious bacterial infection in children hospitalized with bronchiolitis. Hosp Pediatr. 2014;4:33–38. doi: 10.1542/hpeds.2013-0073. [DOI] [PubMed] [Google Scholar]
- 71.Farley R., Spurling G.K., Eriksson L., Del Mar C.B. Antibiotics for bronchiolitis in children under two years of age. Cochrane Database Syst Rev. 2014;10:CD005189. doi: 10.1002/14651858.CD005189.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Bacharier L.B., Guilbert T.W., Mauger D.T., Boehmer S., Beigelman A., Fitzpatrick A.M. Early administration of azithromycin and prevention of severe lower respiratory tract illnesses in preschool children with a history of such illnesses: a randomized clinical trial. JAMA. 2015;314:2034–2044. doi: 10.1001/jama.2015.13896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Loss G.J., Depner M., Hose A.J., Genuneit J., Karvonen A.M., Hyvarinen A. The early development of wheeze: environmental determinants and genetic susceptibility at 17q21. Am J Respir Crit Care Med. 2015 doi: 10.1164/rccm.201507-1493OC. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 74.Fujimura K.E., Demoor T., Rauch M., Faruqi A.A., Jang S., Johnson C.C. House dust exposure mediates gut microbiome Lactobacillus enrichment and airway immune defense against allergens and virus infection. Proc Natl Acad Sci U S A. 2014;111:805–810. doi: 10.1073/pnas.1310750111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lynch S.V., Wood R.A., Boushey H., Bacharier L.B., Bloomberg G.R., Kattan M. Effects of early-life exposure to allergens and bacteria on recurrent wheeze and atopy in urban children. J Allergy Clin Immunol. 2014;134:593–601.e12. doi: 10.1016/j.jaci.2014.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Soto-Quiros M., Avila L., Platts-Mills T.A., Hunt J.F., Erdman D.D., Carper H. High titers of IgE antibody to dust mite allergen and risk for wheezing among asthmatic children infected with rhinovirus. J Allergy Clin Immunol. 2012;129:1499–1505.e5. doi: 10.1016/j.jaci.2012.03.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Green R.M., Custovic A., Sanderson G., Hunter J., Johnston S.L., Woodcock A. Synergism between allergens and viruses and risk of hospital admission with asthma: case-control study. BMJ. 2002;324:763. doi: 10.1136/bmj.324.7340.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kloepfer K.M., Gern J.E. Virus/allergen interactions and exacerbations of asthma. Immunol Allergy Clin North Am. 2010;30:553–563. doi: 10.1016/j.iac.2010.08.002. vii. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Gern J.E., Calhoun W., Swenson C., Shen G., Busse W.W. Rhinovirus infection preferentially increases lower airway responsiveness in allergic subjects. Am J Respir Crit Care Med. 1997;155:1872–1876. doi: 10.1164/ajrccm.155.6.9196088. [DOI] [PubMed] [Google Scholar]
- 80.Jakiela B., Brockman-Schneider R., Amineva S., Lee W.M., Gern J.E. Basal cells of differentiated bronchial epithelium are more susceptible to rhinovirus infection. Am J Respir Cell Mol Biol. 2008;38:517–523. doi: 10.1165/rcmb.2007-0050OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lachowicz-Scroggins M.E., Boushey H.A., Finkbeiner W.E., Widdicombe J.H. Interleukin-13-induced mucous metaplasia increases susceptibility of human airway epithelium to rhinovirus infection. Am J Respir Cell Mol Biol. 2010;43:652–661. doi: 10.1165/rcmb.2009-0244OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Saglani S., Lui S., Ullmann N., Campbell G.A., Sherburn R.T., Mathie S.A. IL-33 promotes airway remodeling in pediatric patients with severe steroid-resistant asthma. J Allergy Clin Immunol. 2013;132:676–685.e13. doi: 10.1016/j.jaci.2013.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Savenije O.E., Mahachie John J.M., Granell R., Kerkhof M., Dijk F.N., de Jongste J.C. Association of IL33-IL-1 receptor-like 1 (IL1RL1) pathway polymorphisms with wheezing phenotypes and asthma in childhood. J Allergy Clin Immunol. 2014;134:170–177. doi: 10.1016/j.jaci.2013.12.1080. [DOI] [PubMed] [Google Scholar]
- 84.Beale J., Jayaraman A., Jackson D.J., Macintyre J.D., Edwards M.R., Walton R.P. Rhinovirus-induced IL-25 in asthma exacerbation drives type 2 immunity and allergic pulmonary inflammation. Sci Transl Med. 2014;6:256ra134. doi: 10.1126/scitranslmed.3009124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Contoli M., Ito K., Padovani A., Poletti D., Marku B., Edwards M.R. Th2 cytokines impair innate immune responses to rhinovirus in respiratory epithelial cells. Allergy. 2015;70:910–920. doi: 10.1111/all.12627. [DOI] [PubMed] [Google Scholar]
- 86.Contoli M., Message S.D., Laza-Stanca V., Edwards M.R., Wark P.A., Bartlett N.W. Role of deficient type III interferon-lambda production in asthma exacerbations. Nat Med. 2006;12:1023–1026. doi: 10.1038/nm1462. [DOI] [PubMed] [Google Scholar]
- 87.Wark P.A., Johnston S.L., Bucchieri F., Powell R., Puddicombe S., Laza-Stanca V. Asthmatic bronchial epithelial cells have a deficient innate immune response to infection with rhinovirus. J Exp Med. 2005;201:937–947. doi: 10.1084/jem.20041901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Durrani S.R., Montville D.J., Pratt A.S., Sahu S., Devries M.K., Rajamanickam V. Innate immune responses to rhinovirus are reduced by the high-affinity IgE receptor in allergic asthmatic children. J Allergy Clin Immunol. 2012;130:489–495. doi: 10.1016/j.jaci.2012.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gill M.A., Bajwa G., George T.A., Dong C.C., Dougherty I.I., Jiang N. Counterregulation between the FcepsilonRI pathway and antiviral responses in human plasmacytoid dendritic cells. J Immunol. 2010;184:5999–6006. doi: 10.4049/jimmunol.0901194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pritchard A.L., Carroll M.L., Burel J.G., White O.J., Phipps S., Upham J.W. Innate IFNs and plasmacytoid dendritic cells constrain Th2 cytokine responses to rhinovirus: a regulatory mechanism with relevance to asthma. J Immunol. 2012;188:5898–5905. doi: 10.4049/jimmunol.1103507. [DOI] [PubMed] [Google Scholar]
- 91.Lynch J.P., Mazzone S.B., Rogers M.J., Arikkatt J.J., Loh Z., Pritchard A.L. The plasmacytoid dendritic cell: at the cross-roads in asthma. Eur Respir J. 2014;43:264–275. doi: 10.1183/09031936.00203412. [DOI] [PubMed] [Google Scholar]
- 92.Teach S.J., Gill M.A., Togias A., Sorkness C.A., Arbes S.J., Jr., Calatroni A. Preseasonal treatment with either omalizumab or an inhaled corticosteroid boost to prevent fall asthma exacerbations. J Allergy Clin Immunol. 2015;136:1476–1485. doi: 10.1016/j.jaci.2015.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]