Abstract
Oncolytic viruses have made their mark on the cancer world as a potential therapeutic option, with the possible advantages of reduced side effects and strengthened treatment efficacy due to higher tumor selectivity. Results have been so promising, that oncolytic viral treatments have now been approved for clinical trials in several countries. However, clinical studies may benefit from the ability to noninvasively and serially identify sites of viral targeting via molecular imaging in order to provide safety, efficacy, and toxicity information. Furthermore, molecular imaging of oncolytic viral therapy may provide a more sensitive and specific diagnostic technique to detect tumor origin and, more importantly, presence of metastases. Several strategies have been investigated for molecular imaging of viral replication broadly categorized into optical and deep tissue imaging, utilizing several reporter genes encoding for fluorescence proteins, conditional enzymes, and membrane protein and transporters. Various imaging methods facilitate molecular imaging, including computer tomography, magnetic resonance imaging, positron emission tomography, single photon emission CT, gamma-scintigraphy, and photoacoustic imaging. In addition, several molecular probes are used for medical imaging, which act as targeting moieties or signaling agents. This review will explore the preclinical and clinical use of in vivo molecular imaging of replication-competent oncolytic viral therapy.
Despite advances in conventional therapy, 12 million people worldwide will be diagnosed with cancer this year, and 7 million people will die from cancer-related causes.1 Therefore, developing novel therapies, which may also work synergistically in combination with conventional treatment options, is vital. Oncolytic viral therapies have made their mark on the cancer research world as a potential therapeutic option, with the possible advantages of lesser side effects and strengthened treatment efficacy due to higher tumor selectivity.2
Results have been so promising that oncolytic viral treatments have now been approved for clinical trials in several countries.3 However, clinical studies may benefit from the ability to noninvasively and serially identify sites of viral targeting via molecular imaging, and to measure the level of viral infection in order to provide important safety, efficacy, and toxicity information.4–6 Such real-time tracking would also provide useful viral dose and administration schedule information for optimization of therapy, and would obviate the need for multiple and repeated tissue biopsies. With this information, molecular imaging may lead to an improvement in both vector design and clinical protocols for future personalized treatments.7,8 Furthermore, molecular imaging of oncolytic viral therapy may provide a more sensitive and specific diagnostic technique to detect tumor origin and, more importantly, presence of metastases.
This review will explore the preclinical and clinical uses of in vivo molecular imaging of replication-competent oncolytic viral therapy.
Why Oncolytic Viruses for Gene Delivery?
Oncolytic viruses refer to those that are able to preferentially and selectively propagate in cancer cells, and consequently destroy tumor tissues leaving noncancerous tissues unharmed (Figure 1).9 The idea that viruses may be able to treat cancer was born almost by chance, when in the early 20th century patients with malignancies who experienced viral infection or received rabies vaccinations were noted to experience transient remissions.10,11 These early discoveries led to several viruses being tested in both preclinical and clinical settings during the late 1940s, 50s, and 60s.12 While early studies and clinical trials were considered groundbreaking, interest in viruses as potential antineoplastic therapies was abandoned due to unimpressive and short-lived success, as well as unacceptable side effects that eventually ended trials.13 It is only in the last two decades or so that the fervor of viruses as a strategy against cancer has been reignited with the advancements in scientific knowledge and technology. We now possess tools that enable us to develop more targeted and effective viruses.14 Examples of oncolytic viruses studied to date include adenovirus, herpes simplex virus (HSV), measles virus, vesicular stomatitis virus (VSV), and vaccinia virus (VACV), among others. The ultimate goal of replication-competent anticancerous viral therapy is to produce a safe and effective therapeutic index with minimal toxicity and side effects.14
Figure 1.
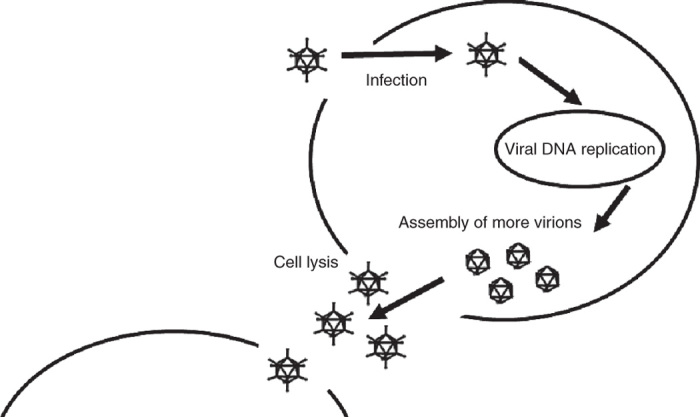
Mechanism of oncolytic viral therapy. Oncolytic viruses specifically target cancer cells, replicate within them, eventually causing cell lysis and death, thereby releasing progenies that subsequently infect adjacent cancer cells.
In addition to their oncotropic and oncolytic effects, advantages of using replication-selective oncolytic viruses for transgene delivery are several.15 In addition to their oncolytic capabilities, replicating viruses can deliver therapeutic transgenes to image and enhance the probability of tumor eradication through multiple avenues. Replication-selective viral systems can employ endogenous viral gene expression control signals (promoter/enhancer, polyadenylation, and splice signals) for transgene expression. Eliminating the need for exogenous promoters often needed in replication-deficient systems and polyadenylation signals is an economical use of the often limited transgene capacity afforded a replicating viral agent. Using endogenous viral promoters may also allow more predictable and controlled transgene expression. Further, in contrast to foreign or exogenous promoters, the promoters of the replicating agent are optimized for expression in the infected cell.
Viral gene expression is also highly regulated.16 Based on the expression of endogenous viral genes, it may be possible to predict the expression kinetics (timing and expression levels) of the transgene(s) carried by the replicating agent. Furthermore, when multiple transgenes are inserted into a single virus, their expression may be orchestrated to occur simultaneously or serially, at levels that will maximize their therapeutic benefits.
Noninvasive In Vivo Molecular Imaging of Replication-Competent Oncolytic Viruses
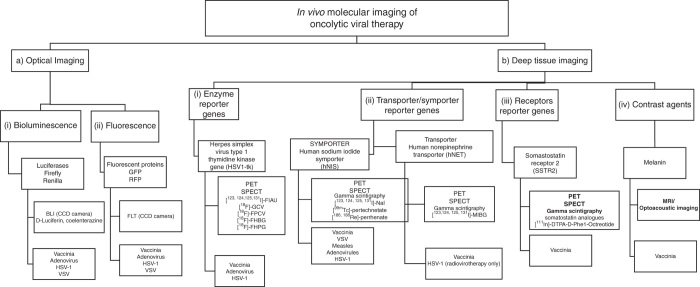
Consequently, several strategies have been investigated for molecular imaging of viral replication within cancer (Figure 2). Molecular imaging is defined as the ability to visualize and quantitatively measure the function of biological and cellular processes in vivo.7,8 These are categorized broadly into optical imaging and deep-tissue imaging, and utilize several reporter genes such as fluorescence proteins, conditional enzymes, and membrane proteins and transporters. Various imaging methods are utilized for molecular imaging, including computed tomography (CT), magnetic resonance imaging (MRI), positron-emission tomography (PET), single photon-emission CT (SPECT), γ-scintigraphy, and photoacoustic imaging.7,8,17–21 In addition, several molecular probes are used for medical imaging applications, such as radioactive iodine species, technecium, and 18F-FIAU, which act as targeting moieties or signaling agents.
Figure 2.
Overview of current oncolytic viruses genetically encoding reporter genes utilized for in vivo molecular imaging. (A) Optical imaging: (i) genes encoding for luciferases (Firefly and Renilla) mediated bioluminescence imaging via CCD cameras utilizing D-luciferin or coelenterazine substrates; (ii) genes encoding for fluorescent proteins (GFP and RFP) mediated fluorescence imaging via CCD cameras. Viruses encoding genes mediating both optical imaging modalities so far are vaccinia, adenovirus, HSV-1 and VSV. (B) Deep tissue imaging: (i) the enzyme reporter gene HSV-1 thymidine kinase is activated by several 18F tagged substrates such as ganciclovir (18F-GCV) and may be imaged via PET and SPECT with viruses encoding this gene so far being vaccinia, HSV-1 and adenovirus; (ii) the human sodium iodide symporter hNIS is one of the most promising reporter genes investigated so far, facilitating deep tissue imaging via uptake of several species of carrier free radionuclide probes such as radioiodide, technecium, and rhenium with viruses designed to encode this symporter so far being vaccinia, VSV, measles, adenovirus, and HSV-1; the human norepinephrine transporter hNET facilitates specific uptake of radiolabeled MIBG with viruses encoding this gene so far being a vaccinia and HSV-1 strain. Both reporter genes enable imaging of viral replication via PET, SPECT, and gamma scintigraphy; (iii) the somatostatin SSRT2 receptor reporter gene facilitates uptake of somatostatin analogues usually radiolabelled with indium ([111In]-DTPA-D-Phe1-Octreotide) and may be imaged via PET, SPECT and gamma scintigraphy with viruses encoding this gene so far being only a vaccinia strain; (iv) a vaccinia virus has been genetically engineered to carry the gene encoding for melanin, which may be used as a contrast agent and facilitated imaging of viral replication via MRI and optoacoustic imaging.
Optical imaging
Optical detection methods mainly include fluorescence, such as green fluorescent protein (GFP), enhanced GFP (eGFP), and red fluorescent protein (RFP), and bioluminescence imaging (BLI), which utilizes luciferases.8,19 Optical detection methods have the advantage of short acquisition times (for fluorescence imaging few milliseconds to several seconds, for bioluminescence a few seconds to several minutes) and high spatial resolution. The major disadvantage of optical imaging is its inability to perform deep-tissue imaging due to autofluorescence, light scattering, and the opacity of tissues below 600 nm due to absorbance of light by hemoglobin. Nevertheless, optical imaging in preclinical tests remains a very important tool to follow the distribution of oncolytic viruses.
Fluorescence.
The most widely used reporter protein is GFP, and most of the GFP variants are derived from the jellyfish Aequorea victoria. Many synthetic variants, including an eGFP, have been developed for improving the stability and brightness of fluorescence without altering their fluorescence or the function of fusion protein.25 In vivo molecular imaging of fluorescent proteins is usually achieved with special adapted charged-couple device cameras to detect fluorescence.
Several virus strains have been equipped with genes encoding fluorescent proteins such as GFPs.22–31 Moreover, a GFP-encoding vaccinia virus strain (GLV-1h68) is currently in phase 1 and 2 clinical trials, in which GFP is being investigated for the confirmation and monitoring of virus colonization of near-surface tumors and metastases.32,33 A number of viruses encoding RFPs have also been developed.34 For example, a VACV encoding for RFP has been shown to regress xenograft and syngeneic models of colorectal peritoneal carcinomatosis.35
Bioluminescence.
BLI is a very effective technique for use in small animal experimental models.8–10 It relies on the detection of very weak, visible light emitted by the oxidation of D-luciferin produced by the luciferase reporter gene in the presence of oxygen and ATP. This weak light is detected by very sensitive charged-couple device cameras that amplify the specific signal-to-noise ratio.11 Firefly luciferase, Renilla luciferase, and Gaussia luciferase are the most commonly used luciferase genes. Their corresponding substrates are luciferin for firefly luciferase and coelenterazine for Renilla and Gaussia luciferases. Bioluminescence systems depend on the delivery of a specific substrate to the reporter expressing cells and the presence of oxygen. Bioluminescence reporter genes have been more widely used than fluorescence reporter genes for whole-body imaging due to the advantages of higher sensitivity and lower background luminescence.26 However, a major disadvantage of BLI is its low spatial resolution, and thus its use is limited in larger subjects, and it thus remains experimental.
The use of BLI in several virus strains has been equipped with genes encoding luciferases,25,26,28,30,31,36–39 and this molecular imaging method in small animals has been shown to allow accurate tracking of gene expression, of the development or regression of tumor metastases, and of viral infection as well as assessment of gene therapy.
Deep-tissue imaging modalities
To date, three types of deep-tissue reporter genes have been explored with oncolytic viral therapies, those encoding for: enzymes (e.g., herpes simplex virus-thymidine kinase (HSV1-tk)), contrast agents (melanin), and receptors (such as somatostatin receptor) or symporter/transport proteins (hNET, hNIS). The deep-tissue imaging methods have been explored with oncolytic viral therapies utilizing instrumentation such as PET, SPECT, and γ-scintigraphy, which have also been combined with CT for additional anatomic correlation.7,17,18,40 SPECT technology detects individual photons emitted by radioisotopes such as iodine-123 (123I) or 99m-technecium pertechtenate (99mTcO4).40 PET devices detect the gamma rays produced when positrons emitted from the radioisotope collide with local neighboring electrons.4,5,8,17,18,41 Despite their relatively limited spatial resolution (3–5 mm for PET and 8–10 mm for SPECT), both imaging modalities provide molecular and functional information with a high sensitivity, especially when compared with optical imaging. This high degree of sensitivity may allow the use of relatively weak promoters and the imaging of relatively low levels of gene expression. Comparatively, PET provides higher sensitivity than SPECT.42 The repeatability, quantifiability, and high sensitivity of deep-tissue reporter gene systems may lead to rapid advancements in our understanding of oncolytic viral therapies.41 Consequently, PET and SPECT currently offer the greatest potential for translation to clinical use.
Photoacoustic technologies have also emerged, which may be combined with MRI for advanced imaging.7,40,43 MRI offers unlimited depth of detection and is capable of high spatial resolution (in the submillimeter range). Optoacoustic imaging utilizes short light energy pulses, which are absorbed by tissues and converted to thermal energy and subsequently to ultrasound signals that allow high-resolution whole-body imaging. However, its general applicability is limited due to its inferior sensitivity relative to PET or SPECT. Generation of higher contrast between the target and background could reduce such limitations, such as with the use of melanin. Compared with fluorescent proteins, melanin has the advantage of being visible by noninvasive deep-tissue imaging with multi-spectral tomography (MSPT) and MRI, due to its absorption in the near-infrared (IR) spectrum.43 Because the transduced cells construct the MRI contrast agent, imaging of gene expression is rendered possible without the need for an exogenous contrast agent.
Enzymes
Thymidine kinase (tk).
HSV1-tk is a viral enzyme that has a relaxed substrate specificity compared with its mammalian counterpart. The HSV1-tk gene is transferred into tumors, usually by infection with recombinant viruses, leading to the expression of the enzyme within tumor tissues.6,42 Tracer doses of radiolabeled prodrugs (usually an acycloguanosine, such as ganciclovir), administered systemically to the patient, are phosphorylated by the transgene (and only poorly by the endogenous tk). This phosphorylation converts the membrane-permeable prodrug into a polar product, which is trapped in the cell, thus enabling imaging. The most commonly used radioprobes are fialuridine, 1-(2-deoxy-2-fluoro-1-D-arabinofuranosyl)-5-iodouracil (FIAU), and [18F] 9-(4-fluoro-3-hydroxymethylbutyl) guanine (FHBG). This strategy has been validated using PET imaging in numerous experimental models, by accurately monitoring viral delivery and replication.18 However, thymidine analogue tracers are investigational drugs and add a level of regulatory complexity that has prevented their clinical usage. Furthermore, the nonhuman origin of HSV1-tk also raises concerns over their potential immunogenicity.41
Using replication-competent HSV-1 oncolytic viruses with tk under the control of different promoters, our lab was the first to demonstrate the time- and dose-dependent replication of an oncolytic virus.42 Viral-colonized human colorectal tumors were detected by 124I–FIAU PET imaging, with the PET signal intensity significantly increasing at 48 hours compared with that at 8 hours after viral injection, demonstrating that PET scanning can detect changes in TK activity resulting from local viral proliferation.
Contrast agents
Melanin.
A recent study by Stritzker et al.43 explored the use of a VACV encoding for melanin production. The oncolytic virus-mediated production of melanin and its optical absorption in the near-IR spectrum enabled the imaging of A549 tumors and metastases via utilization of MRI and MSPT. The ubiquitous presence of melanin in all kingdoms of life suggests that introduction of melanin synthesis as a diagnostic/theranostic marker is possible in most species.
Receptors
Human somatostatin receptor 2 (hSSRT2).
The SSTR2 is targeted by the high-affinity synthetic peptide pentetreotide, which is commonly used for receptor imaging after being radiolabeled with indium-111 (111In)44. This receptor is normally expressed in human kidney cells and neuroendocrine tumors, and gene therapy approaches to deliver the SSTR2 to tumors have also been attempted using adenoviral vectors.44,45 In a study by McCart et al.,46 nude mice bearing subcutaneous murine colon CA xenografts were injected intraperitoneally with an SSTR2-expressing VACV or control and imaged 6 days later with 111In-pentetreotide-mediated SPECT. Tumors infected with the SSTR2-expressing VACV accumulated higher concentrations of radioactivity compared with tumors in animals receiving the control virus. Further, the SSTR2-infected tumors were visible on imaging 6 days after VACV injection and could be visualized for up to 3 weeks post viral injection using repeat radiotracer injections. A limitation of the SSRT2 receptor is that radiotracers for SSRT2 require prior radiolabeling for the accumulation of radioprobes. Furthermore, unlike transporters such as the human norepinephrin transporter (hNET) and the human sodium iodide symporter (hNIS), SSTR2 is a receptor that usually has a 1:1 binding relationship with radiolabeled ligands and is therefore not capable of providing significant amplification through transport-mediated concentrative intracellular accumulation of substrate.
Human norepinephrin transporter (hNET).
Another deep-tissue reporter gene investigated in oncolytic viral strains is the hNET. hNET is a cell-surface human protein, which belongs to a family of Na+/Cl-dependent transporters that contain multiple transmembrane domains and mediates the transport of norepinephrine, dopamine, and epinephrine across the cell membrane. It can mediate molecular imaging via SPECT or PET using the radiotracer meta-iodobenzylguanidine (MIBG).47,48 The use of the hNET–MIBG reporter imaging is attractive since it is of human origin and will unlikely induce an immune response, as well as its limited expression in the central and peripheral sympathetic nervous systems.49 Moreover, MIBG is a standard radiopharmaceutical approved by the US Food and Drug Administration. Previously, attempts to image gene transfer therapies by PET using hNET–MIBG reporter imaging were successful in transduced cells and xenografts.47
Chen et al.50 were the first to report on an oncolytic virus encoding for hNET, GLV-1h99, a derivative of VACV GLV-1h68 also encoding for GFP and RLuc. The hNET protein did not hinder viral replication and in vitro or in vivo treatment efficacy, and was expressed at high levels on the membranes of infected cells. GLV-1h99-mediated expression of hNET resulted in specific uptake of the radiotracer 131I–MIBG. In mice, GLV-1h99-infected pancreatic tumors were readily imaged by PET using 124I–MIBG. This virus was further investigated by Brader et al.51 for the imaging of an orthotopic mouse model of human malignant mesothelioma using both 123I–MIBG-mediated SPECT imaging and 124I–MIBG-mediated PET imaging. Another group investigated the use of the hNET gene utilizing an oncolytic herpes virus, but did not investigate its use for imaging.52 A limitation of the hNET symporter is the requirement for MIBG for radiotracer labeling. The question then remained: is there a potential deep-tissue imaging reporter gene of human origin, which facilitates concentrative intracellular uptake of carrier-free radioprobes, which can facilitate high-resolution PET imaging? This led to the interest in hNIS.
Human sodium iodide symporter (hNIS).
This hNIS is an intrinsic plasma membrane protein, which mediates the active transport and concentration of iodide in the thyroid gland cells and some extra thyroidal tissues, in particular, the lactating mammary gland.53 Discovery and cloning of the rat NIS, by Carrasco and colleagues,54 and of the human NIS, by Jhiang and colleagues,55 mediating iodide uptake opened an exciting and extensive new field of research, including exploring the pathophysiological implications of NIS in thyroid disease, the therapeutic potential of the gene, and its imaging and diagnostic potentials. hNIS gene transfer via viral vector may allow infected tumor cells to concentrate several easily attainable, commercially available, and relatively inexpensive, carrier-free radioisotopes such as 123I, 124I, 125I, 131I, 99mTcO4, rhenium, and astatine for noninvasive imaging of NIS expression, all of which have long been approved for human use. hNIS use has also been shown to be comparable to the commonly used HSV1-tk reporter gene56 and correlated with 99mTcO4.57 This can be very useful for viral distribution with SPECT or PET scanning during and after viral therapy, and may allow for correlation with efficacy and toxicity during clinical trials, thus offering potential clinical translation of this dual therapy.
The oncolytic viruses that have been investigated so far in relation to hNIS are adenoviruses, measles, vesicular stomatitis, vaccinia, and most recently a HSV. Merron et al.58 were the first to insert the hNIS reporter gene in a wild-type replicating oncolytic adenovirus (AdlP1) and in an adenovirus in which a promoter from the human telomerase gene (RNA component) drives E1 expression (AdAM6). The virus-induced functional hNIS expression and replication in cell culture and kinetics of spread of the different viruses in colorectal tumor xenografts were visualized in vivo using 99mTcO4-mediated SPECT. Hakkarainen et al.59 created the adenovirus Ad5/3-Δ24-hNIS, a Rb-p16 pathway-selective infectivity-enhanced oncolytic adenovirus encoding hNIS. Ad5/3-Δ24-hNIS replication effectively regressed prostate cancer PC-3MM2 tumors with viral replication visualized via 123I-mediated γ-scintigraphy, and this virus is now in clinical trials.3 Another replication-competent oncolytic virus armed with two suicide genes and the NIS gene, Ad5-yCD/mutTK(SR39)rep-hNIS, was injected into contralateral lobes of the dog prostate and hNIS activity was monitored in live animals using γ-scintigraphy, allowing for dynamic monitoring of reporter gene expression within the prostate as well as biodistribution throughout the body.60 Several other adenoviruses, which carry the hNIS gene under several promoters in various cancers, have also been constructed using either intratumoral, intravenous, or intraperitoneal virus administration. A significant disadvantage was the reduced replication capacity of adenovirus.61–64
Several groups have also attempted hNIS transfer via oncolytic measles vectors. Dingli et al.65 were the first to show the noninvasive imaging and therapy potential of a measles virus encoding for hNIS (MV-NIS) by 123I–γ-scintigraphy imaging of the intratumoral spread of various multiple myeloma xenografts. Hasegawa et al.66 tested MV-NIS in combination with another viral construct, MV-CEA, to treat ovarian cancer SKOV3ip.1 xenografts. MV-NIS propagation was again mapped by serial radioiodine imaging via gamma scanner by systemic injection of 99mTcO4. However, γ-scintigraphy was considerably less sensitive than the plasma CEA marker for monitoring virus infection. Further, Liu et al.67 demonstrated that antitumor activity of MV-NIS was evident only in measles naive mice-bearing disseminated myeloma, and not in passively immunized mice, which may limit the clinical application of this virus. Several other studies have investigated the use of measles virus carrying the NIS gene.68–70
In mice with established subcutaneous 5TGM1 myeloma tumors, virus replication and spread of a vesicular stomatitis virus, VSV (51)-NIS, were also monitored noninvasively by serial 123I–γ-scintigraphy imaging after systemic or intratumoral injection. Dosimetry calculations based on these images pointed to the feasibility of combination radiovirotherapy with VSV(51)-NIS and 131I.
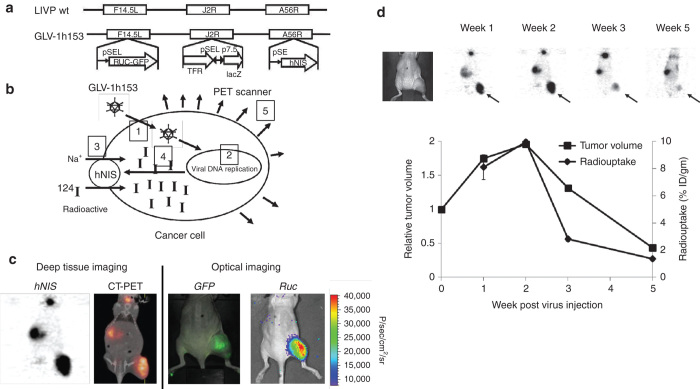
Our lab was the first to investigate a VACV carrying the hNIS, GLV-1h153, which is also a derivative of GLV-1h68.71 The virus also encoded for a GFP and the RLuc gene, and was found to be successful in the fluorescent, bioluminescent, and deep-tissue image monitoring of viral replication and therapy (Figure 3).72,73 Moreover, GLV-1h153 successfully regressed several tumor types in preclinical models including pancreatic cancer, triple-negative breast cancer, gastric cancer, and malignant pleural mesothelioma.74–78
Figure 3.
Molecular imaging of oncolytic vaccinia virus GLV-1h153. (a) GLV-1h153 construct. GLV-1h153 was derived from LIVP-wt virus, by replacing the gusA expression cassette at the A56R locus with the hNIS expression cassette through homologous recombination. The virus also contains RUC-GFP and lacZ expression cassettes at the F14.5L and J2R loci, respectively. PE, PE/L, P11, and P7.5 are VACV synthetic early, synthetic early/late, 11K, and 7.5K promoters, respectively. TFR is human transferrin receptor inserted in the reverse orientation with respect to the promoter PE/L. (b) GLV-1h153 specifically infects cancer cells, replicates within them, mediating hNIS gene expression on the cell surface subsequently causing intracellular radiotracer accumulation and signal detection via PET. (c) GFP, bioluminescence, and hNIS signal could be detected in GLV-1h153-infected tumors. Fusion of PET and CT images correlated hNIS-mediated uptake signal anatomically to location of thyroid, stomach, bladder (due to intrinsic hNIS expression), and tumor (due to virus-mediated hNIS expression), whereas GFP- and Ruc-mediated bioluminescence signals only to location of tumor. (d) hNIS-mediated enhanced radio-uptake in GLV-1h153-infected tumors was serially monitored via 124I-PET, being strongest at weeks 1 and 2 after virus injection and declining gradually by weeks 3 and 5. PET signal appeared to reflect tumor response to therapy and remained strong during initial tumor growth. However, when tumor growth began entering plateau or retardation phase, PET signal began to decrease and almost disappeared by week 5, when tumors entered regression phase.
Transporters and symporters.
Transporter-based reporter gene systems, as opposed to receptors that have a 1:1 binding relationship with radiolabeled ligands, provide signal amplification through transport-mediated concentrative intracellular accumulation of substrate. Furthermore, the human origin of most investigated transporters thus far likely renders them less immunogenic than other foreign reporter genes such as TK.
Clinical Trials with OVs Encoding for Reporter Genes
Preclinical imaging and therapy success with oncolytic viruses and reporter genes have led to several completed and ongoing clinical trials (Table 1).3 The clinical application of gene delivery will require the generation and investigation of safe and efficient gene-delivery systems, as well as the ability for systemic administration and regulated gene expression. Only a very small number of gene expression imaging studies have been carried out to date in humans utilizing oncolytic viral therapies.
Table 1. Clinical trials investigating deep-tissue molecular imaging of oncolytic viruses.
| Intervention | Condition | Phase | Sponsor | Status |
|---|---|---|---|---|
| Biological: adenovirus (Ad5-yCD/utTKSR39rep-hNIS) | Prostate cancer | Phase 1 | Henry Ford Health System, Michigan | Completed (ref. 70) |
| Biological: adenovirus (Ad5-CMV-NIS) | Chemotherapy-refractory cervical cancer | Phase 1 | Mayo Clinic, Rochester | Recruiting (ref. 3) |
| Biological: adenovirus (Ad5-CMV-NIS) | Radiation therapy refractory prostate cancer | Phase 1 | Mayo Clinic, Rochester | Recruiting (ref. 3) |
| Biological: measles virus (MV-NIS) | Recurrent ovarian cancer | Phase 1 | Mayo Clinic, Rochester | Recruiting (ref. 3) |
| Refractory multiple myeloma | ||||
| Malignant pleural mesothelioma | ||||
| Recurrent or metastatic squamous cell carcinoma of the head and neck cancer | ||||
| Biological: measles virus (MV-NIS) | Multiple myeloma | Phase 2 | University of Arkansas | Not yet recruiting (ref. 3) |
The NIS-based strategy was the first approach. Men with clinically localized prostate cancer were administered an intraprostatic injection of the replication-competent adenovirus Ad5-yCD/utTKSR39rep-hNIS for the imaging of prostate cancer.79 NIS gene expression was imaged noninvasively by the uptake of 99mTcO4 in infected cells using SPECT. Therapy was shown to be safe with 98% of the adverse events of grade 1 or 2. Enhanced radio-uptake was detected in the prostate in seven of nine patients. Further, the expression was monitored and found to increase to around 18% of the total prostate volume following injection of virus, which peaked 1–2 days post adenovirus injection and was detectable in the prostate for up to 7 days. No evidence of extraprostatic dissemination of the adenovirus was evident via SPECT imaging. The results demonstrated for the first time in humans the noninvasive imaging capability of hNIS utilizing an oncolytic viral vector.79 Other trials are also underway investigating the potential of Ad5-CMV-NIS for gene therapy and combination with radioactive iodine in treating patients with locally recurrent radiotherapy-resistant prostate cancer that did and recurrent cervical cancer.3
Several clinical trials have also been carried out with MV-NIS. The first was a dose-escalation phase 1 trial of MV-NIS when given with or without cyclophosphamide in treating patients with recurrent or refractory multiple myeloma.3 The time course of viral gene expression and viral elimination, as well as the biodistribution of virally infected cells at various time points after treatment with these regimens using 123I–γ-scintigraphy imaging, will also be determined. In addition, several other groups are recruiting patients for clinical trials in order to investigate the therapeutic effects of various MV-NIS with various cancers alongside SPECT/CT imaging of the course of viral gene expression, elimination, and biodistribution at various time points after treatment. The first phase 2 clinical trial utilizing oncolytic viruses and NIS will begin recruiting utilizing MV-NIS for multiple myeloma.
Therapeutic Potential with Reporter Genes
Although self-replicating virotherapy encoding reporter genes has shown great promise for treating cancer and for the noninvasive molecular imaging and monitoring of viral therapy, the additive or synergistic effects by combining targeted therapeutic systemic therapies, including prodrugs and radiotherapy, may aid in the elimination of tumors and virus-resistant tumors.
For example, the prodrug acycloguanosine, such as ganciclovir, is phosphorylated by the HSV1-tk transgene (and only poorly by the endogenous tk). This phosphorylation converts the membrane-permeable prodrug into a polar product, which is trapped in the cell. This phosphorylated product is then incorporated into the DNA where it acts as a chain terminator, inducing cell death. Apart from the transduced cancer cells, neighboring, nontransfected cells can also be killed through a mechanism referred to as the bystander effect.
Oncolytic viruses encoding transporter genes such as hNIS have also been found to have a synergistic antitumor effect when combined with ionizing radiation, such as 131I.80–82 One mechanism for such synergy appears to be radiation-induced upregulation of certain cellular DNA repair genes that result in promoting viral replication.83,84 Furthermore, a bystander effect may be possible as 131I undergoes α-particle decay with a path length of 0.2–2.4 mm.85 It is important to note that 131I is already approved in patients for multiple ailments, including thyroid cancers and palliation of bone metastasis, and is found to be safe with relatively few side effects.86 In addition, 131I is not a gene therapy and thus involves less risk and complications as compared with gene therapy of cancer. If additive or synergistic effects are found, patients may be more safely treated with combinations of lower doses of virus and radioiodine. The application of carrier-free radioiodine would thus be extended, and the extensive experience with radioiodine in thyroid cancer management will undoubtedly be helpful in the treatment of other NIS-transfected tumors. Our lab demonstrated an enhanced effect of oncolytic viral therapy with GLV-1h153 when combined with radiotherapy 131I in both pancreatic and breast cancer xenografts. The VACV encoding the gene for SSTR2 combined with the radiotherapeutic 111In-DOTATOC was also shown to be more effective than either alone at decreasing the growth rate of human embryonic kidney HEK-293 cells or colorectal MC-38 cells in monolayer.87 Furthermore, Ad5/3-Δ24-hNIS with radioiodide was significantly more effective than virus alone in mice with prostate cancer xenografts.
The contrast agent melanin may facilitate near-IR–assisted thermotherapy in addition to oncolytic virotherapy.43 A near-IR laser was utilized to specifically transfer energy to melanin-induced cells, with the transferred energy consequently converted to thermal energy, eventually heating the melanin-producing cells and cells in their vicinity to temperatures causing protein denaturation and cell death, therefore enabling thermotherapy. Stritzker et al. demonstrated that aliquots containing cells infected with VACV-encoding melanin achieved a higher temperature exposed to laser light, with near-complete kill of all cells within those aliquots as compared with mock infected cells. They also demonstrated that lung cancer xenografts on tumor-bearing mice treated with the melanin-inducing VACV had significantly enhanced regression when using a single 2-minute laser treatment, compared with tumors that were not exposed to the laser light, demonstrating an additive effect.
Conclusions
Noninvasive in vivo molecular imaging of oncolytic viral therapy has experienced a significant surge in preclinical and clinical studies, which may eventually aid in the design and improvement of early cancer detection, patient-specific treatment selection, targeted delivery, and therapy-specific monitoring of gene therapy, in order to better treat and cure diseases with high mortality. Preclinical research has resulted in the identification of a large number of reporter genes, many of which have already been utilized with several strains of oncolytic viruses, in addition to the development of novel molecular imaging contrast agents and imaging devices, hardwares, and software technologies. The capacity to quantatively and precisely visualize and monitor the in vivo regulation of viral replication and biodistribution and kinetics of gene expression may also lead to advances in the safety and efficacy of oncolytic viruses as vectors for gene therapy. Among the safety issues, cell-specific targeting is an important consideration of all cancer gene therapy trials. With preclinical success and further clinical trials, molecular imaging of oncolytic viral gene therapies will likely play an increasing role in the clinical treatment of cancer. Most current applications use PET and SPECT imaging, and other molecular imaging techniques, including MRI/MRS, optical (Raman), molecular US, and photoacoustic tomography, warrant further investigation.
The authors declare no conflict of interest.
References
- World Health Organization (2008). The global burden of cancer. In: World Cancer Report. http://www.iarc.fr/en/publications/pdfs-online/wcr/2008/index.php.
- Chen, NG and Szalay AA (2010). Oncolytic vaccinia virus: a theranostic agent for cancer. Future Virol 5: 763–784. [Google Scholar]
- The Clinical Trials Database. https://www.clinicaltrials.gov.
- Serganova, I and Blasberg, R (2005). Reporter gene imaging: potential impact on therapy. Nucl Med Biol 32: 763–780. [DOI] [PubMed] [Google Scholar]
- Serganova, I, Ponomarev, V and Blasberg, R (2007). Human reporter genes: potential use in clinical studies. Nucl Med Biol 34: 791–807. [DOI] [PubMed] [Google Scholar]
- Kuruppu, D, Brownell, AL, Zhu, A, Yu, M, Wang, X, Kulu, Y et al. (2007). Positron emission tomography of herpes simplex virus 1 oncolysis. Cancer Res 67: 3295–3300. [DOI] [PubMed] [Google Scholar]
- Pysz, MA, Gambhir, SS and Willmann, JK (2010). Molecular imaging: current status and emerging strategies. Clin Radiol 65: 500–516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuchmiy, AA, Efimov, GA and Nedospasov, SA (2012). Methods for in vivo molecular imaging. Biochemistry Mosc 77: 1339–1353. [DOI] [PubMed] [Google Scholar]
- Parato, KA, Senger, D, Forsyth, PA and Bell, JC (2005). Recent progress in the battle between oncolytic viruses and tumours. Nat Rev Cancer 5: 965–976. [DOI] [PubMed] [Google Scholar]
- Sinkovics, J (2007). The effect of oxygenation on the biological behaviour of tumours. Orv Hetil 148: 2056–2057. [DOI] [PubMed] [Google Scholar]
- Dock, G (1904). Rabies virus vaccination in a patient with cervical carcinoma. Amer J Med Sci 127: 563. [Google Scholar]
- Vähä-Koskela, MJ, Heikkilä, JE and Hinkkanen, AE (2007). Oncolytic viruses in cancer therapy. Cancer Lett 254: 178–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinkovics, JG and Horvath, JC (2008). Natural and genetically engineered viral agents for oncolysis and gene therapy of human cancers. Arch Immunol Ther Exp (Warsz) 56 (suppl. 1): 3s–59s. [DOI] [PubMed] [Google Scholar]
- Woo, Y, Adusumilli, PS and Fong, Y (2006). Advances in oncolytic viral therapy. Curr Opin Investig Drugs 7: 549–559. [PubMed] [Google Scholar]
- Hermiston, T (2000). Gene delivery from replication-selective viruses: arming guided missiles in the war against cancer. J Clin Invest 105: 1169–1172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolonen, N, Doglio, L, Schleich, S and Krijnse Locker, J (2001). Vaccinia virus DNA replication occurs in endoplasmic reticulum-enclosed cytoplasmic mini-nuclei. Mol Biol Cell 12: 2031–2046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youn, H and Chung, JK (2013). Reporter gene imaging. AJR Am J Roentgenol 201: W206–W214. [DOI] [PubMed] [Google Scholar]
- Sharma, V, Luker, GD and Piwnica-Worms, D (2002). Molecular imaging of gene expression and protein function in vivo with PET and SPECT. J Magn Reson Imaging 16: 336–351. [DOI] [PubMed] [Google Scholar]
- Hickson, J (2009). In vivo optical imaging: preclinical applications and considerations. Urol Oncol 27: 295–297. [DOI] [PubMed] [Google Scholar]
- Briat, A and Vassaux, G (2006). Preclinical applications of imaging for cancer gene therapy. Expert Rev Mol Med 8: 1–19. [DOI] [PubMed] [Google Scholar]
- Alberti, C (2012). From molecular imaging in preclinical/clinical oncology to theranostic applications in targeted tumor therapy. Eur Rev Med Pharmacol Sci 16: 1925–1933. [PubMed] [Google Scholar]
- Deng, H, Tang, N, Stief, AE, Mehta, N, Baig, E, Head, R et al. (2008). Oncolytic virotherapy for multiple myeloma using a tumour-specific double-deleted vaccinia virus. Leukemia 22: 2261–2264. [DOI] [PubMed] [Google Scholar]
- Zhang, Q, Yu, YA, Wang, E, Chen, N, Danner, RL, Munson, PJ et al. (2007). Eradication of solid human breast tumors in nude mice with an intravenously injected light-emitting oncolytic vaccinia virus. Cancer Res 67: 10038–10046. [DOI] [PubMed] [Google Scholar]
- Lun, XQ, Jang, JH, Tang, N, Deng, H, Head, R, Bell, JC et al. (2009). Efficacy of systemically administered oncolytic vaccinia virotherapy for malignant gliomas is enhanced by combination therapy with rapamycin or cyclophosphamide. Clin Cancer Res 15: 2777–2788. [DOI] [PubMed] [Google Scholar]
- Gentschev, I, Stritzker, J, Hofmann, E, Weibel, S, Yu, YA, Chen, N et al. (2009). Use of an oncolytic vaccinia virus for the treatment of canine breast cancer in nude mice: preclinical development of a therapeutic agent. Cancer Gene Ther 16: 320–328. [DOI] [PubMed] [Google Scholar]
- Le Boeuf, F, Diallo, JS, McCart, JA, Thorne, S, Falls, T, Stanford, M et al. (2010). Synergistic interaction between oncolytic viruses augments tumor killing. Mol Ther 18: 888–895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gentschev, I, Donat, U, Hofmann, E, Weibel, S, Adelfinger, M, Raab, V et al. (2010). Regression of human prostate tumors and metastases in nude mice following treatment with the recombinant oncolytic vaccinia virus GLV-1h68. J Biomed Biotechnol 2010: 489759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tysome, JR, Briat, A, Alusi, G, Cao, F, Gao, D, Yu, J et al. (2009). Lister strain of vaccinia virus armed with endostatin-angiostatin fusion gene as a novel therapeutic agent for human pancreatic cancer. Gene Ther 16: 1223–1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly, KJ, Woo, Y, Brader, P, Yu, Z, Riedl, C, Lin, SF et al. (2008). Novel oncolytic agent GLV-1h68 is effective against malignant pleural mesothelioma. Hum Gene Ther 19: 774–782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly, KJ, Brader, P, Woo, Y, Li, S, Chen, N, Yu, YA et al. (2009). Real-time intraoperative detection of melanoma lymph node metastases using recombinant vaccinia virus GLV-1h68 in an immunocompetent animal model. Int J Cancer 124: 911–918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thorne, SH, Negrin, RS and Contag, CH (2006). Synergistic antitumor effects of immune cell-viral biotherapy. Science 311: 1780–1784. [DOI] [PubMed] [Google Scholar]
- Savona, MR, Cruz, WP, Thornton, JA and Danaher, PJ (2007). Comparison of a semipermeable dressing bonded to an absorbent pad and a semipermeable dressing over a separate gauze pad for containment of vaccinia virus at the vaccination site. Infect Control Hosp Epidemiol 28: 1339–1343. [DOI] [PubMed] [Google Scholar]
- Zhou, X, Liu, B, Yu, X, Zha, X, Zhang, X, Wang, X et al. (2008). Controlled release of PEI/DNA complexes from PLGA microspheres as a potent delivery system to enhance immune response to HIV vaccine DNA prime/MVA boost regime. Eur J Pharm Biopharm 68: 589–595. [DOI] [PubMed] [Google Scholar]
- Lin, MZ, McKeown, MR, Ng, HL, Aguilera, TA, Shaner, NC, Campbell, RE et al. (2009). Autofluorescent proteins with excitation in the optical window for intravital imaging in mammals. Chem Biol 16: 1169–1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottolino-Perry, K, Tang, N, Head, R, Ng, C, Arulanandam, R, Angarita, FA et al. (2014). Tumor vascularization is critical for oncolytic vaccinia virus treatment of peritoneal carcinomatosis. Int J Cancer 134: 717–730. [DOI] [PubMed] [Google Scholar]
- Hung, CF, Tsai, YC, He, L, Coukos, G, Fodor, I, Qin, L et al. (2007). Vaccinia virus preferentially infects and controls human and murine ovarian tumors in mice. Gene Ther 14: 20–29. [DOI] [PubMed] [Google Scholar]
- Chang, CL, Ma, B, Pang, X, Wu, TC and Hung, CF (2009). Treatment with cyclooxygenase-2 inhibitors enables repeated administration of vaccinia virus for control of ovarian cancer. Mol Ther 17: 1365–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirn, DH, Wang, Y, Liang, W, Contag, CH and Thorne, SH (2008). Enhancing poxvirus oncolytic effects through increased spread and immune evasion. Cancer Res 68: 2071–2075. [DOI] [PubMed] [Google Scholar]
- Thorne, SH, Hwang, TH, O’Gorman, WE, Bartlett, DL, Sei, S, Kanji, F et al. (2007). Rational strain selection and engineering creates a broad-spectrum, systemically effective oncolytic poxvirus, JX-963. J Clin Invest 117: 3350–3358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richard-Fiardo, P, Franken, PR, Harrington, KJ, Vassaux, G and Cambien, B (2011). The use of molecular imaging of gene expression by radiotracers in gene therapy. Expert Opin Biol Ther 11: 1273–1285. [DOI] [PubMed] [Google Scholar]
- MacLaren, DC, Toyokuni, T, Cherry, SR, Barrio, JR, Phelps, ME, Herschman, HR et al. (2000). PET imaging of transgene expression. Biol Psychiatry 48: 337–348. [DOI] [PubMed] [Google Scholar]
- Bennett, JJ, Tjuvajev, J, Johnson, P, Doubrovin, M, Akhurst, T, Malholtra, S et al. (2001). Positron emission tomography imaging for herpes virus infection: Implications for oncolytic viral treatments of cancer. Nat Med 7: 859–863. [DOI] [PubMed] [Google Scholar]
- Stritzker, J, Kirscher, L, Scadeng, M, Deliolanis, NC, Morscher, S, Symvoulidis, P et al. (2013). Vaccinia virus-mediated melanin production allows MR and optoacoustic deep tissue imaging and laser-induced thermotherapy of cancer. Proc Natl Acad Sci USA 110: 3316–3320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slooter, GD, Mearadji, A, Breeman, WA, Marquet, RL, de Jong, M, Krenning, EP et al. (2001). Somatostatin receptor imaging, therapy and new strategies in patients with neuroendocrine tumours. Br J Surg 88: 31–40. [DOI] [PubMed] [Google Scholar]
- Zinn, KR, Buchsbaum, DJ, Chaudhuri, TR, Mountz, JM, Grizzle, WE and Rogers, BE (2000). Noninvasive monitoring of gene transfer using a reporter receptor imaged with a high-affinity peptide radiolabeled with 99mTc or 188Re. J Nucl Med 41: 887–895. [PubMed] [Google Scholar]
- McCart, JA, Mehta, N, Scollard, D, Reilly, RM, Carrasquillo, JA, Tang, N et al. (2004). Oncolytic vaccinia virus expressing the human somatostatin receptor SSTR2: molecular imaging after systemic delivery using 111In-pentetreotide. Mol Ther 10: 553–561. [DOI] [PubMed] [Google Scholar]
- Moroz, MA, Serganova, I, Zanzonico, P, Ageyeva, L, Beresten, T, Dyomina, E et al. (2007). Imaging hNET reporter gene expression with 124I-MIBG. J Nucl Med 48: 827–836. [DOI] [PubMed] [Google Scholar]
- Shulkin, BL, Shapiro, B, Tobes, MC, Shen, SW, Wieland, DM, Meyers, LJ et al. (1986). Iodine-123-4-amino-3-iodobenzylguanidine, a new sympathoadrenal imaging agent: comparison with iodine-123 metaiodobenzylguanidine. J Nucl Med 27: 1138–1142. [PubMed] [Google Scholar]
- Axelrod, J and Kopin, IJ (1969). The uptake, storage, release and metabolism of noradrenaline in sympathetic nerves. Prog Brain Res 31: 21–32. [DOI] [PubMed] [Google Scholar]
- Chen, N, Zhang, Q, Yu, YA, Stritzker, J, Brader, P, Schirbel, A et al. (2009). A novel recombinant vaccinia virus expressing the human norepinephrine transporter retains oncolytic potential and facilitates deep-tissue imaging. Mol Med 15: 144–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brader, P, Kelly, KJ, Chen, N, Yu, YA, Zhang, Q, Zanzonico, P et al. (2009). Imaging a genetically engineered oncolytic vaccinia virus (GLV-1h99) using a human norepinephrine transporter reporter gene. Clin Cancer Res 15: 3791–3801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorensen, A, Mairs, RJ, Braidwood, L, Joyce, C, Conner, J, Pimlott, S et al. (2012). In vivo evaluation of a cancer therapy strategy combining HSV1716-mediated oncolysis with gene transfer and targeted radiotherapy. J Nucl Med 53: 647–654. [DOI] [PubMed] [Google Scholar]
- Riesco-Eizaguirre, G and Santisteban, P (2006). A perspective view of sodium iodide symporter research and its clinical implications. Eur J Endocrinol 155: 495–512. [DOI] [PubMed] [Google Scholar]
- Dai, G, Levy, O and Carrasco, N (1996). Cloning and characterization of the thyroid iodide transporter. Nature 379: 458–460. [DOI] [PubMed] [Google Scholar]
- Smanik, PA, Liu, Q, Furminger, TL, Ryu, K, Xing, S, Mazzaferri, EL et al. (1996). Cloning of the human sodium lodide symporter. Biochem Biophys Res Commun 226: 339–345. [DOI] [PubMed] [Google Scholar]
- Moon, DH, Lee, SJ, Park, KY, Park, KK, Ahn, SH, Pai, MS et al. (2001). Correlation between 99mTc-pertechnetate uptakes and expressions of human sodium iodide symporter gene in breast tumor tissues. Nucl Med Biol 28: 829–834. [DOI] [PubMed] [Google Scholar]
- Moon, DH, Lee, SJ, Park, KY, Park, KK, Ahn, SH, Pai, MS et al. (2001). Correlation between 99mTc-pertechnetate uptakes and expressions of human sodium iodide symporter gene in breast tumor tissues. Nucl Med Biol 28: 829–834. [DOI] [PubMed] [Google Scholar]
- Merron, A, Peerlinck, I, Martin-Duque, P, Burnet, J, Quintanilla, M, Mather, S et al. (2007). SPECT/CT imaging of oncolytic adenovirus propagation in tumours in vivo using the Na/I symporter as a reporter gene. Gene Ther 14: 1731–1738. [DOI] [PubMed] [Google Scholar]
- Hakkarainen, T, Rajecki, M, Sarparanta, M, Tenhunen, M, Airaksinen, AJ, Desmond, RA et al. (2009). Targeted radiotherapy for prostate cancer with an oncolytic adenovirus coding for human sodium iodide symporter. Clin Cancer Res 15: 5396–5403. [DOI] [PubMed] [Google Scholar]
- Barton, KN, Tyson, D, Stricker, H, Lew, YS, Heisey, G, Koul, S et al. (2003). GENIS: gene expression of sodium iodide symporter for noninvasive imaging of gene therapy vectors and quantification of gene expression in vivo. Mol Ther 8: 508–518. [DOI] [PubMed] [Google Scholar]
- Trujillo, MA, Oneal, MJ, Davydova, J, Bergert, E, Yamamoto, M and Morris, JC 3rd (2009). Construction of an MUC-1 promoter driven, conditionally replicating adenovirus that expresses the sodium iodide symporter for gene therapy of breast cancer. Breast Cancer Res 11: R53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peerlinck, I, Merron, A, Baril, P, Conchon, S, Martin-Duque, P, Hindorf, C et al. (2009). Targeted radionuclide therapy using a Wnt-targeted replicating adenovirus encoding the Na/I symporter. Clin Cancer Res 15: 6595–6601. [DOI] [PubMed] [Google Scholar]
- Merron, A, Baril, P, Martin-Duque, P, de la Vieja, A, Tran, L, Briat, A et al. (2010). Assessment of the Na/I symporter as a reporter gene to visualize oncolytic adenovirus propagation in peritoneal tumours. Eur J Nucl Med Mol Imaging 37: 1377–1385. [DOI] [PubMed] [Google Scholar]
- Trujillo, MA, Oneal, MJ, McDonough, S, Qin, R and Morris, JC (2010). A probasin promoter, conditionally replicating adenovirus that expresses the sodium iodide symporter (NIS) for radiovirotherapy of prostate cancer. Gene Ther 17: 1325–1332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dingli, D, Peng, KW, Harvey, ME, Greipp, PR, O’Connor, MK, Cattaneo, R et al. (2004). Image-guided radiovirotherapy for multiple myeloma using a recombinant measles virus expressing the thyroidal sodium iodide symporter. Blood 103: 1641–1646. [DOI] [PubMed] [Google Scholar]
- Hasegawa, K, Pham, L, O’Connor, MK, Federspiel, MJ, Russell, SJ and Peng, KW (2006). Dual therapy of ovarian cancer using measles viruses expressing carcinoembryonic antigen and sodium iodide symporter. Clin Cancer Res 12: 1868–1875. [DOI] [PubMed] [Google Scholar]
- Liu, C, Russell, SJ and Peng, KW (2010). Systemic therapy of disseminated myeloma in passively immunized mice using measles virus-infected cell carriers. Mol Ther 18: 1155–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Msaouel, P, Iankov, ID, Allen, C, Aderca, I, Federspiel, MJ, Tindall, DJ et al. (2009). Noninvasive imaging and radiovirotherapy of prostate cancer using an oncolytic measles virus expressing the sodium iodide symporter. Mol Ther 17: 2041–2048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blechacz, B, Splinter, PL, Greiner, S, Myers, R, Peng, KW, Federspiel, MJ et al. (2006). Engineered measles virus as a novel oncolytic viral therapy system for hepatocellular carcinoma. Hepatology 44: 1465–1477. [DOI] [PubMed] [Google Scholar]
- Penheiter, AR, Wegman, TR, Classic, KL, Dingli, D, Bender, CE, Russell, SJ et al. (2010). Sodium iodide symporter (NIS)-mediated radiovirotherapy for pancreatic cancer. AJR Am J Roentgenol 195: 341–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddad, D, Chen, NG, Zhang, Q, Chen, CH, Yu, YA, Gonzalez, L et al. (2011). Insertion of the human sodium iodide symporter to facilitate deep tissue imaging does not alter oncolytic or replication capability of a novel vaccinia virus. J Transl Med 9: 36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddad, D, Chen, CH, Carlin, S, Silberhumer, G, Chen, NG, Zhang, Q et al. (2012). Imaging characteristics, tissue distribution, and spread of a novel oncolytic vaccinia virus carrying the human sodium iodide symporter. PLoS ONE 7: e41647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haddad, D, Chen, N, Zhang, Q, Chen, CH, Yu, YA, Gonzalez, L et al. (2012). A novel genetically modified oncolytic vaccinia virus in experimental models is effective against a wide range of human cancers. Ann Surg Oncol 19 Suppl 3: S665–S674. [DOI] [PubMed] [Google Scholar]
- Belin, LJ, Ady, JW, Lewis, C, Marano, D, Gholami, S, Mojica, K et al. (2013). An oncolytic vaccinia virus expressing the human sodium iodine symporter prolongs survival and facilitates SPECT/CT imaging in an orthotopic model of malignant pleural mesothelioma. Surgery 154: 486–495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gholami, S, Chen, CH, Belin, LJ, Lou, E, Fujisawa, S, Antonacci, C et al. (2013). Vaccinia virus GLV-1h153 is a novel agent for detection and effective local control of positive surgical margins for breast cancer. Breast Cancer Res 15: R26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gholami, S, Chen, CH, Lou, E, Belin, LJ, Fujisawa, S, Longo, VA et al. (2014). Vaccinia virus GLV-1h153 in combination with 131I shows increased efficiency in treating triple-negative breast cancer. FASEB J 28: 676–682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gholami, S, Chen, CH, Lou, E, De Brot, M, Fujisawa, S, Chen, NG et al. (2012). Vaccinia virus GLV-1h153 is effective in treating and preventing metastatic triple-negative breast cancer. Ann Surg 256: 437–445. [DOI] [PubMed] [Google Scholar]
- Gholami, S, Haddad, D, Chen, CH, Chen, NG, Zhang, Q, Zanzonico, PB et al. (2011). Novel therapy for anaplastic thyroid carcinoma cells using an oncolytic vaccinia virus carrying the human sodium iodide symporter. Surgery 150: 1040–1047. [DOI] [PubMed] [Google Scholar]
- Barton, KN, Stricker, H, Brown, SL, Elshaikh, M, Aref, I, Lu, M et al. (2008). Phase I study of noninvasive imaging of adenovirus-mediated gene expression in the human prostate. Mol Ther 16: 1761–1769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Advani, SJ, Sibley, GS, Song, PY, Hallahan, DE, Kataoka, Y, Roizman, B et al. (1998). Enhancement of replication of genetically engineered herpes simplex viruses by ionizing radiation: a new paradigm for destruction of therapeutically intractable tumors. Gene Ther 5: 160–165. [DOI] [PubMed] [Google Scholar]
- Kim, SH, Wong, RJ, Kooby, DA, Carew, JF, Adusumilli, PS, Patel, SG et al. (2005). Combination of mutated herpes simplex virus type 1 (G207 virus) with radiation for the treatment of squamous cell carcinoma of the head and neck. Eur J Cancer 41: 313–322. [DOI] [PubMed] [Google Scholar]
- Blank, SV, Rubin, SC, Coukos, G, Amin, KM, Albelda, SM and Molnar-Kimber, KL (2002). Replication-selective herpes simplex virus type 1 mutant therapy of cervical cancer is enhanced by low-dose radiation. Hum Gene Ther 13: 627–639. [DOI] [PubMed] [Google Scholar]
- Adusumilli, PS, Chan, MK, Hezel, M, Yu, Z, Stiles, BM, Chou, TC et al. (2007). Radiation-induced cellular DNA damage repair response enhances viral gene therapy efficacy in the treatment of malignant pleural mesothelioma. Ann Surg Oncol 14: 258–269. [DOI] [PubMed] [Google Scholar]
- Mezhir, JJ, Advani, SJ, Smith, KD, Darga, TE, Poon, AP, Schmidt, H et al. (2005). Ionizing radiation activates late herpes simplex virus 1 promoters via the p38 pathway in tumors treated with oncolytic viruses. Cancer Res 65: 9479–9484. [DOI] [PubMed] [Google Scholar]
- Boyd, M, Sorensen, A, McCluskey, AG and Mairs, RJ (2008). Radiation quality-dependent bystander effects elicited by targeted radionuclides. J Pharm Pharmacol 60: 951–958. [DOI] [PubMed] [Google Scholar]
- Chatal, JF and Hoefnagel, CA (1999). Radionuclide therapy. Lancet 354: 931–935. [DOI] [PubMed] [Google Scholar]
- Akinlolu, O, Ottolino-Perry, K, McCart, JA and Reilly, RM (2010). Antiproliferative effects of 111In- or 177Lu-DOTATOC on cells exposed to low multiplicity-of-infection double-deleted vaccinia virus encoding somatostatin subtype-2 receptor. Cancer Biother Radiopharm 25: 325–333. [DOI] [PubMed] [Google Scholar]