To the Editor
Posttraumatic stress disorder (PTSD) is a heterogeneous condition comprised of threat/fear (e.g., intrusions, avoidance, hypervigilance) and loss/dysphoria (e.g., numbing, dysphoric arousal) symptoms.1,2 Contemporary scientific efforts in psychiatry, such as the National Institute of Mental Health Research Domain Criteria (RDoC) project, are encouraging studies of the neurobiological and behavioral underpinnings of transdiagnostic aspects of mental disorders, such as threat and loss symptoms, with the goal of developing novel, mechanism-based classifications of psychopathology, which can be used to develop more targeted treatments.3
In line with such efforts, we recently found that PTSD is associated with greater cannabinoid type 1 (CB1) receptor availability,4 and that greater CB1 receptor availability in the amygdala was associated with increased threat, but not loss, symptoms in trauma survivors.5 Variation in the cannabinoid receptor type 1 (CNR1) gene may also contribute to risk for PTSD, with the minor (A) allele of rs1049353 associated with increased likelihood of PTSD.6 Additionally, rs1049353 has been found to interact with childhood physical abuse (CPA), one type of trauma that might impact the endocannabinoid system, to predict anhedonia.7 However, no study has examined associations between rs1049353 genotype—alone or interactively with CPA—and the phenotypic expression of PTSD symptoms.
Using data from the Detroit Neighborhood Health Study (DNHS), an epidemiologic study of predominantly African-American adults from urban Detroit,8,9 we examined how rs1049353 genotype—alone and interactively with CPA—relates to severity of posttraumatic threat and loss symptoms. We hypothesized that rs1049353 genotype would specifically underlie threat, but not loss, symptoms.5 The University of Michigan Institutional Review Board approved the DNHS and participants provided written informed consent.
Method
A total 487 adults (mean age=53.3 [SD=15.6] years; 57.7% female; 82.7% African-American) provided valid data for rs1049353 and completed the PTSD Checklist10 and Conflict Tactics Scale11 measure of CPA. We conducted a multivariate analysis of covariance (MANCOVA) to examine the relation between rs1049353 genotype (AA/AG vs. GG) and CPA (alone and in interaction) as predictors of threat (sum of intrusion, avoidance, and anxious arousal) and loss (sum of numbing and dysphoric arousal) symptoms. Age, sex, lifetime trauma burden, and the first two principal components from a multidimensional scaling analysis of genome-wide data were included as covariates.
Results
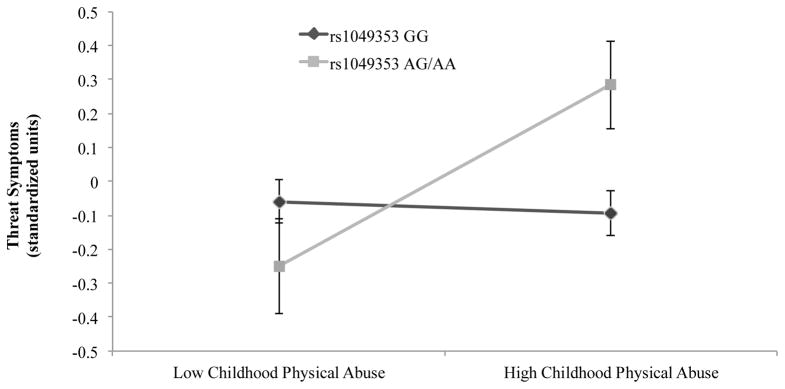
CPA predicted threat symptoms (F[1,454]=5.59, p=.018), but main effects of rs1049353 genotype were non-significant (threat: F[1,454]=0.69, p=0.41; loss: F[1,454]=0.51, p=0.47). There was also a significant rs1049353 genotype × CPA interaction for threat symptoms only (F[1,454]=7.57, p=.006); minor (A) allele carriers with high levels of CPA reported greater threat symptoms (Figure 1). In a separate MANCOVA of the five PTSD symptom dimensions (re-experiencing, avoidance, anxious arousal, numbing, dysphoric arousal), this interaction also emerged for two of three dimensions of threat symptoms: avoidance (F[1,454]=6.13, p=0.014) and re-experiencing (F[1,454]=6.13, p=0.014).
Figure 1.
Association of CNR1 SNP rs1049353, childhood physical abuse, and posttraumatic threat symptoms (N=487)
Note. Error bars represent standard error (SE) of the mean. Low and high Childhood Physical Abuse (CPA) was based on summing the items to the Conflict Tactics Scale and dichotomizing based on a median split. GG/low CPA n=203, GG/high CPA n=194, AG/AA/low CPA n=43, AG/AA/high CPA n=47. Genotype frequencies were as follows: A/A (n=7 [1.4%]), A/G: (n=83 [17.0%]), G/G: (n=397 [81.6%]); they did not deviate from Hardy-Weinberg equilibrium, χ2(1)=1.20, p=.27. Results of a MANCOVA revealed a significant rs1049353 genotype × CPA interaction in predicting threat symptoms only (F[1,454]=7.57, p=.006).
An additional CNR1 SNP—rs806368—which has been implicated in PTSD,6 was also examined alone and interactively with CPA in relation to the severity of threat and loss symptoms. Although there was a marginal main effect of rs806368 genotype on severity of threat symptoms (F[1,453]=4.37, p=.037), it did not withstand the correction for multiple comparisons of two SNPs (p=.025). rs806368 genotype was also not related to loss symptoms (F[1,453]=2.99, p=.085), nor was the interaction of this polymorphism and CPA related to threat or loss symptoms (F’s<2.47, p’s>.11).
Discussion
The current findings extend our prior work implicating the endocannabinoid system in PTSD.4,5 The rs1049353 SNP has been found to interact with CPA to contribute to decreased anhedonia,7 and here we demonstrate that rs1049353 genotype interacts with CPA to increase severity of threat/fear, but not loss/dysphoria, symptoms of PTSD. The rs1049353 SNP is exonic, but synonymous, and may cause alterations in CNR1 protein formation during translation.7 Notably, a previous study7 observed a protective effect of the minor A allele in rs1049353 and CPA interaction in relation to anhedonia symptoms. One explanation for these results is differing demographic (e.g., racial) compositions or trauma exposure characteristics of the samples, or that the rs1049353 genotype interacts with level of CPA to predict posttraumatic threat symptoms in some CPA survivors, and anhedonic symptoms in other samples of CPA survivors. Future research is needed to replicate these results and investigate underlying mechanisms.
Footnotes
Funding and Disclosure
This research was funded by National Institutes of Health grants (R01DA022720, R01DA022720-S1 [PhenX], R01DA022720-S1 [Supplement], and RC1MH088283). Preparation of this report was supported in part by the US Department of Veterans Affairs National Center for Posttraumatic Stress Disorder and a private donation. Dr. Neumeister has received consulting fees from Pfizer. Dr. Pietrzak is a scientific consultant to Cogstate. The authors declare no conflicts of interest.
References
- 1.Armour C, Carragher N, Elhai JD. Assessing the fit of the Dysphoric Arousal model across two nationally representative epidemiological surveys: The Australian NSMHWB and the United States NESARC. J Anxiety Disord. 2013;27:109–115. doi: 10.1016/j.janxdis.2012.10.006. [DOI] [PubMed] [Google Scholar]
- 2.Pietrzak RH, Rosenheck RA, Cramer JA, et al. Elucidating the transdiagnostic dimensional structure of trauma-related psychopathology: Findings from VA cooperative study 504 – risperidone treatment for military service related chronic post traumatic stress disorder. J Affect Disord. 2014;172C:331–336. doi: 10.1016/j.jad.2014.10.025. [DOI] [PubMed] [Google Scholar]
- 3.Cuthbert BN. The RDoC framework: Facilitating transition from ICD/DSM to dimensional approaches that integrate neuroscience and psychopathology. World Psychiatry. 2014;13:28–35. doi: 10.1002/wps.20087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Neumeister A, Normandin MD, Pietrzak RH, et al. Elevated brain cannabinoid CB1 receptor availability in post-traumatic stress disorder: a positron emission tomography study. Mol Psychiatry. 2013;18:1034–1040. doi: 10.1038/mp.2013.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pietrzak RH, Huang Y, Corsi-Travali S, et al. Cannabinoid type 1 receptor availability in the amygdala mediates threat processing in trauma survivors. Neuropsychopharmacology. 2014;39:2519–2528. doi: 10.1038/npp.2014.110. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 6.Lu AT, Ogdie MN, Jarvelin M-R, et al. Association of the cannabinoid receptor gene (CNR1) with ADHD and post-traumatic stress disorder. Am J Med Genet B Neuropsychiatr Genet. 2008;8:1488–1494. doi: 10.1002/ajmg.b.30693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Agrawal A, Nelson EC, Littlefield AK, et al. Cannabinoid receptor (CNR1) genotype moderates the effects of childhood physical abuse on anhedonia and depression. Arch Gen Psychiatry. 2012;69:732–740. doi: 10.1001/archgenpsychiatry.2011.2273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Uddin M, Aiello AE, Wildman DE, et al. Epigenetic and immune function profiles associated with posttraumatic stress disorder. Proc Natl Acad Sci USA. 2010;107:9470–9475. doi: 10.1073/pnas.0910794107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lowe SR, Meyers JL, Galea S, et al. RORA and posttraumatic stress trajectories: main effects and interactions with childhood physical abuse history. Brain and Behavior. 2015;5(4):e00323. doi: 10.1002/brb3.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weathers F, Litz B, Herman D, et al. The PTSD Checklist [PCL]: reliability, validity, and diagnostic utility. Paper presented at the Annual Convention of the International Society for Traumatic Stress Studies; San Antonio, TX. 1993. [Google Scholar]
- 11.Straus MA. Measuring intrafamily conflict and violence: the Conflict Tactics (CT) Scales. J Marriage Fam. 1979;41:75–88. [Google Scholar]