Abstract
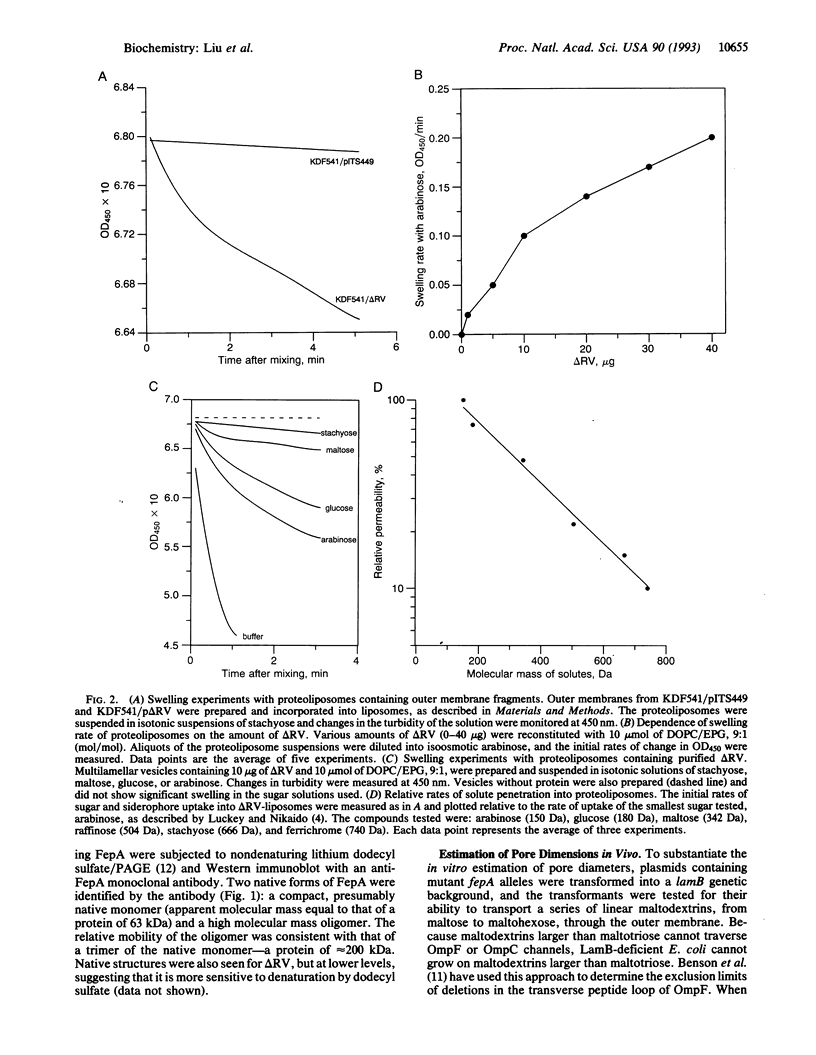
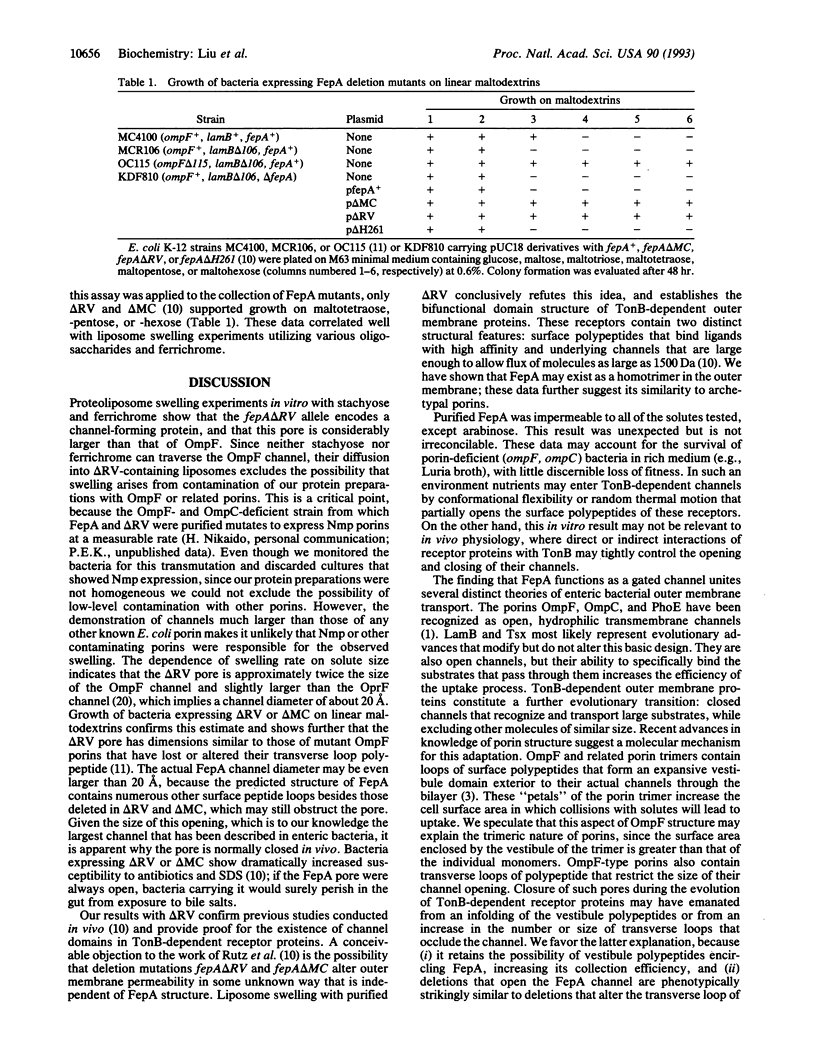
FepA is an Escherichia coli outer membrane receptor protein for the siderophore ferric enterobactin. Prior studies conducted in vivo suggested that FepA and other TonB-dependent outer membrane proteins transport ligands by a gated-channel mechanism. To corroborate and extend these findings we have determined the permeability properties of the FepA channel in vitro, by measuring the diffusion rates of hydrophilic nonelectrolytes through the FepA channel in liposome swelling experiments. Like porins, the FepA deletion mutant delta RV showed a size-dependent permeability to oligosaccharides, indicating that it forms a nonspecific, hydrophilic pore. Unlike OmpF and other E. coli porins, however, delta RV proteoliposomes transported stachyose (666 Da) and ferrichrome (740 Da). These data, and other uptake results with a series of maltodextrins of increasing size, confirm the existence of a channel domain within FepA that is considerably larger than OmpF-type pores. These results represent a reconstitution of the channel function of a TonB-dependent receptor protein and establish that FepA contains the largest channel that has been characterized in the E. coli outer membrane.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Benson S. A., Occi J. L., Sampson B. A. Mutations that alter the pore function of the OmpF porin of Escherichia coli K12. J Mol Biol. 1988 Oct 20;203(4):961–970. doi: 10.1016/0022-2836(88)90121-0. [DOI] [PubMed] [Google Scholar]
- Cowan S. W., Schirmer T., Rummel G., Steiert M., Ghosh R., Pauptit R. A., Jansonius J. N., Rosenbusch J. P. Crystal structures explain functional properties of two E. coli porins. Nature. 1992 Aug 27;358(6389):727–733. doi: 10.1038/358727a0. [DOI] [PubMed] [Google Scholar]
- Fiss E. H., Stanley-Samuelson P., Neilands J. B. Properties and proteolysis of ferric enterobactin outer membrane receptor in Escherichia coli K12. Biochemistry. 1982 Aug 31;21(18):4517–4522. doi: 10.1021/bi00261a050. [DOI] [PubMed] [Google Scholar]
- Guterman S. K., Dann L. Excretion of enterochelin by exbA and exbB mutants of Escherichia coli. J Bacteriol. 1973 Jun;114(3):1225–1230. doi: 10.1128/jb.114.3.1225-1230.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jalal M. A., van der Helm D. Purification and crystallization of ferric enterobactin receptor protein, FepA, from the outer membranes of Escherichia coli UT5600/pBB2. FEBS Lett. 1989 Jan 30;243(2):366–370. doi: 10.1016/0014-5793(89)80163-2. [DOI] [PubMed] [Google Scholar]
- Kadner R. J. Vitamin B12 transport in Escherichia coli: energy coupling between membranes. Mol Microbiol. 1990 Dec;4(12):2027–2033. doi: 10.1111/j.1365-2958.1990.tb00562.x. [DOI] [PubMed] [Google Scholar]
- Klebba P. E., Benson S. A., Bala S., Abdullah T., Reid J., Singh S. P., Nikaido H. Determinants of OmpF porin antigenicity and structure. J Biol Chem. 1990 Apr 25;265(12):6800–6810. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Luckey M., Nikaido H. Specificity of diffusion channels produced by lambda phage receptor protein of Escherichia coli. Proc Natl Acad Sci U S A. 1980 Jan;77(1):167–171. doi: 10.1073/pnas.77.1.167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McIntosh M. A., Earhart C. F. Coordinate regulation by iron of the synthesis of phenolate compounds and three outer membrane proteins in Escherichia coli. J Bacteriol. 1977 Jul;131(1):331–339. doi: 10.1128/jb.131.1.331-339.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
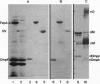
- Murphy C. K., Kalve V. I., Klebba P. E. Surface topology of the Escherichia coli K-12 ferric enterobactin receptor. J Bacteriol. 1990 May;172(5):2736–2746. doi: 10.1128/jb.172.5.2736-2746.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Nikaido K., Harayama S. Identification and characterization of porins in Pseudomonas aeruginosa. J Biol Chem. 1991 Jan 15;266(2):770–779. [PubMed] [Google Scholar]
- Nikaido H., Rosenberg E. Y. Effect on solute size on diffusion rates through the transmembrane pores of the outer membrane of Escherichia coli. J Gen Physiol. 1981 Feb;77(2):121–135. doi: 10.1085/jgp.77.2.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Rosenberg E. Y. Porin channels in Escherichia coli: studies with liposomes reconstituted from purified proteins. J Bacteriol. 1983 Jan;153(1):241–252. doi: 10.1128/jb.153.1.241-252.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Saier M. H., Jr Transport proteins in bacteria: common themes in their design. Science. 1992 Nov 6;258(5084):936–942. doi: 10.1126/science.1279804. [DOI] [PubMed] [Google Scholar]
- Postle K. TonB and the gram-negative dilemma. Mol Microbiol. 1990 Dec;4(12):2019–2025. doi: 10.1111/j.1365-2958.1990.tb00561.x. [DOI] [PubMed] [Google Scholar]
- Rutz J. M., Liu J., Lyons J. A., Goranson J., Armstrong S. K., McIntosh M. A., Feix J. B., Klebba P. E. Formation of a gated channel by a ligand-specific transport protein in the bacterial outer membrane. Science. 1992 Oct 16;258(5081):471–475. doi: 10.1126/science.1411544. [DOI] [PubMed] [Google Scholar]
- Schnaitman C. A. Outer membrane proteins of Escherichia coli. I. Effect of preparative conditions on the migration of protein in polyacrylamide gels. Arch Biochem Biophys. 1973 Aug;157(2):541–552. doi: 10.1016/0003-9861(73)90673-5. [DOI] [PubMed] [Google Scholar]
- Sugawara E., Nikaido H. Pore-forming activity of OmpA protein of Escherichia coli. J Biol Chem. 1992 Feb 5;267(4):2507–2511. [PubMed] [Google Scholar]
- Wang C. C., Newton A. An additional step in the transport of iron defined by the tonB locus of Escherichia coli. J Biol Chem. 1971 Apr 10;246(7):2147–2151. [PubMed] [Google Scholar]
- Weiss M. S., Abele U., Weckesser J., Welte W., Schiltz E., Schulz G. E. Molecular architecture and electrostatic properties of a bacterial porin. Science. 1991 Dec 13;254(5038):1627–1630. doi: 10.1126/science.1721242. [DOI] [PubMed] [Google Scholar]