Abstract
Background
To evaluate the correlation between variations of CA 19.9 blood levels and the entity of necrosis at CT after radiofrequency ablation (RFA) of unresectable pancreatic adenocarcinoma.
Methods
In this study, from June 2010 to February 2014, patients with diagnosis of unresectable and not metastatic pancreatic ductal adenocarcinoma, expressing tumor marker CA 19.9, treated with RFA procedure were included. All these patients underwent RFA. CT study was performed 1 week after RFA. The dosage of CA 19.9 levels was performed 1 month after RFA. Features of necrosis at CT, as mean entity, density and necrosis percentages compared to the original lesion, were evaluated and compared by using t-test with CA 19.9 blood levels variations after RFA procedure.
Results
In this study were included 51 patients with diagnosis of unresectable and not metastatic pancreatic ductal adenocarcinoma, expressing tumor marker CA 19.9, treated with RFA procedure and with CT study and CA 19.9 available for analysis. After the procedure, CA 19.9 blood levels reduced in 24/51 (47%), remained stable in 10/51 (20%) and increased in 17/51 (33%). In patients with CA 19.9 levels reduced, the tumor marker were reduced less than 20% in 4/24 (17%) and more than 20% in 20/24 (83%); instead the tumor marker were reduced less than 30% in 8/24 (33%) and more than 30% in 16/24 (67%). At CT scan necrotic area density difference was not statistically significant. Also there was no statistically significant difference among the mean area, the mean volume and the mean ablation volume in percentage related to the treated tumor among the three different groups of patients divided depending on the CA 19.9 blood levels. But a tendency to a statistically significant difference was found in comparing the mean percentage of ablation volume between two subgroups of patients with a decrease of CA 19.9 levels with less or more than 20% reduction of tumor markers and between two subgroups with less or more than 30% reduction of CA 19.9 levels.
Conclusions
RFA of unresectable pancreatic adenocarcinoma induces reduction of CA 19.9 blood levels in about half of the cases.
Keywords: Radiofrequency ablation (RFA), pancreatic cancer, tumor marker, pancreatic adenocarcinoma
Introduction
Radiofrequency ablation (RFA) of pancreatic cancer is a technique based on the positive effect in treating liver tumors and in giving palliative cares in case of lung, kidney, adrenal gland, prostate, breast, bone and more recently also cholangiocarcinoma intrahepatic tumors (1-3).
RFA, among others ablation techniques, is reported to be a possible palliative treatment for unresectable pancreatic ductal adenocarcinoma locally advanced but not metastatic (4-7). The palliative therapeutic effect is intuitively linked to the development of intratumoral necrosis and so to the neoplastic mass cytoreduction resulting but also argumentations on post-ablation amplification of tumor-induced immune response are possible (8).
In the follow up of these patients CT study and tumor markers dosage are always required. The CT study can be considered the reference imaging method in the follow-up of patients treated with RFA (4,5). CA 19.9 is a tumor marker that, although its low specificity and not optimal sensitivity in pancreatic adenocarcinoma diagnosis, is useful in the evaluation of patients with pancreatic tumor at time of diagnosis or during treatment follow-up, mainly to estimate pancreatic tumor extension or therapeutic response (9-11).
Aim of this study is to evaluate the immediately effect on tumor marker of RFA of pancreatic cancer, evaluating the CA 19.9 levels variations in short-term and comparing the results with post-ablation area features at CT study. To our knowledge in literature exist data about feasibility of pancreatic cancer RFA technique (4), complications (12,13) and patients survival (6,14), but a correlation between CT aspects of post RFA necrotic area and variation of tumor marker has not been reported so far.
Materials and methods
Patients
In this study, from June 2010 to February 2014, patients with diagnosis of unresectable and not metastatic pancreatic ductal adenocarcinoma, expressing tumoral marker CA 19.9, treated with RFA procedure were included. Patients were recruited in two different hospitals (University Hospital G.B. Rossi in Verona and Casa di Cura Dott. Pederzoli in Peschiera del Garda). Consensus from ethics board was obtained for RFA of locally advanced pancreatic cancer in the surgical departments. Inclusion criteria were: (I) patients with diagnosis of unresectable and not metastatic pancreatic ductal adenocarcinoma, treated with RFA; (II) availability of CT study and tumor marker CA 19.9 data for analysis.
RFA technique
In all patients RFA was performed intra-operative under general anaesthesia and laparotomy. Generator system RITA 1500 X (AngioDynamics, USA) was used and different needles (StarBurst XL, Talon or UniBlate) based on lesion size and location. All the procedure was performed under ultrasound guidance. Intraoperative ultrasound was used to confirm presence and unresectability of the tumor, the feasibility of the procedure in terms of lesion visualization and possible needle insertion and positioning, excluding every possible risk due to contiguous vascular and digestive structures, especially the duodenum. During the treatment, needle tip and electrodes were kept at least 5 mm away from susceptible structures, as duodenum and peri-pancreatic vessels (12,15). Moreover the necrotic area must not exceed the lesions margins, securing the ablation/necrotic area totally contained by the lesion and keeping safety margins in respect to vascular and digestive structures. Needle and electrodes correct positioning are followed by parameters regulation on the machine for the procedure. The RFA technique used has been already described in literature (4). Time varied from 5 to 10 min in respect to lesion size. Since proteins denaturation starts at 50°-60°, to obtain a homogeneous necrosis it is necessary to use higher temperature during the procedure but, on the other hand, too high temperature increases complications risk as reported (4). Therefore the temperature fixedly applied in all cases was 90° (4). The ablation was monitored by using ultrasound observing gradually the hyperechoic area due to gas formation appearing inside the lesion.
CT study
All patients received a CT study 1 week after RFA procedure. CT was done utilizing CT multislice equipment (Siemens 16 detectors and Philips 64 detectors). Scans were realized both before and after 120-140 mL contrast media not ionic injection intravenous (Ultravist 370, Schering, Berlino), with a flow rate of 3-4 mL/s. For all the scans were applied the following parameters: collimation of 5 mm, ratio between table progress and tube rotation of 7.5/12 mm and reconstruction interval of 5/3 mm. Utilizing bolus test, were acquired images in arterial phase, on average 25-30 s from contrast medium injection and in portal phase at 60-70 s.
At CT the ablated area (thermo-lesion/necrotic area) was evaluated in terms of density (Figure 1) and dimensions (Figure 2).
Figure 1.
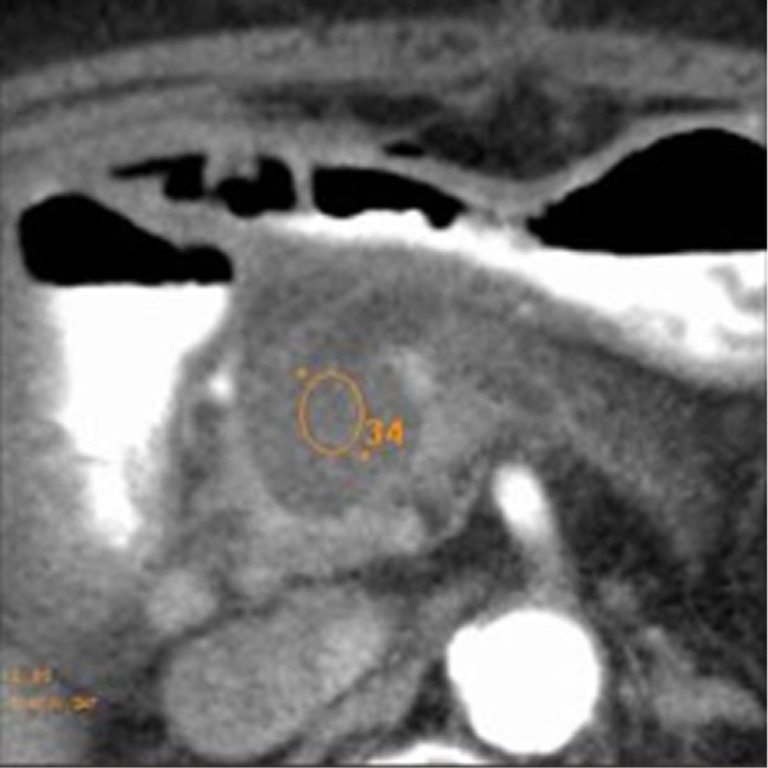
Measurement in Hounsfield Unit of thermo-lesion mean density.
Figure 2.
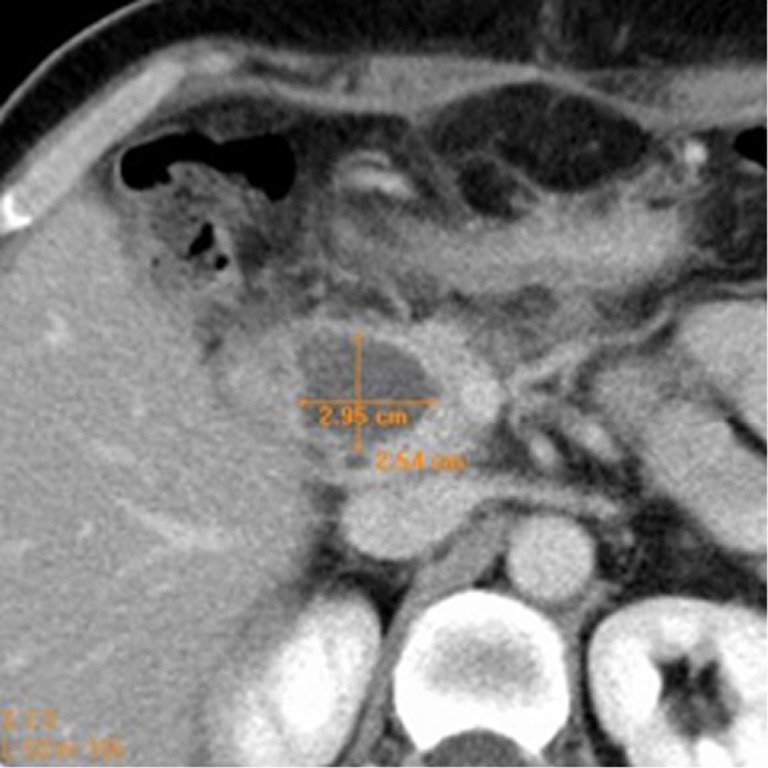
Measurement in centimeters of necrotic area.
We evaluated the density, measured in Hounsfield Unit (HU), of necrosis in the pre-contrastographic phase and post-contrastographic, arterial and venous, phases. In each dynamic phase three regions of interest (ROI) with a diameter at least of 1 cm for density calculation were placed inside the necrosis: first, ROI localized inside the necrosis; second, ROI in the immediately cranial scan; and third, ROI in the immediately caudal scan. These ROI were placed exactly in the same positions in different scans. The mean density among the different ROI was calculated. As regards the size of the thermo-lesion, we assessed in each lesion three axes (anteroposterior, transverse and longitudinal) thus calculating the estimated area for necrosis. With the same calculation the estimated area of the tumor was measured. We also calculated the estimated volume of the primary tumors and of the thermo-lesions. In every case a percentage of post RFA necrosis in respect to the original tumor was determined.
Tumor marker CA 19.9
Tumor marker CA 19.9 blood level was dosed in Units (U/mL) in all patients included in this study previously and further (1 month later) RFA procedure. The CA 19.9 value after the procedure was considered reduced (decrease ≥10 Units; patient group A), stable (equal value or increase <10 Units or decrease <10 Units; patient group B), or increased (increase ≥10 Units; patient group C). The group A then was divided into two subgroups depending on the CA 19.9 variations were more or less 20% or 30% respectively.
Comparisons and data analysis
We compared thermo-lesion features at CT study and CA 19.9 levels variation after the procedures. In particular, the mean density of thermo-lesions and mean necrosis percentages at CT were divided in different groups of variation of CA 19.9 levels and were evaluated and compared by using student t-test.
Results
In this study, from June 2010 to February 2014, 51 patients (27 men and 24 women, with mean age of 64 years in an age-range from 41 to 87 years) were included with diagnosis of unresectable and not metastatic pancreatic ductal adenocarcinoma, expressing tumoral marker CA 19.9, treated with RFA procedure and with CT and CA 19.9 available for analysis. Patients were recruited in the two different hospitals as follow: 16 patients from University Hospital G.B. Rossi in Verona and 35 patients from Casa di Cura Dott. Pederzoli in Peschiera del Garda.
At CT study the treated tumors had a mean area and volume of 1,036 mm2 and 28,696 mm3, respectively. At CT study the intratumoral thermo-lesions had a mean area and volume of 365 mm2 and 6,567 mm3, respectively. The mean ablation volume in percentage related to the treated tumor was 24%. At CT the mean density was 28 [16-37], 35 [19-58] and 38 [22-70] HU respectively in pre-contrastographic, arterial and portal phases.
CA 19.9 blood levels after RFA procedure were reduced (group A) in 24/51 patients (47%) (Table 1), were stable (group B) in 10/51 patients (20%) (Table 2) and were increased (group C) in 17/51 patients (33%) (Table 3).
Table 1. CA 19.9 blood levels before and after the RFA procedure in group A, consisting of patients with CA 19.9 values reduced more than 10 Units.
| Patients with CA 19.9 values reduced more than 10 Units | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 504 | 204 |
| 2 | 2,697 | 1,561 |
| 3 | 2,554 | 1,810 |
| 4 | 253 | 207 |
| 5 | 156 | 51 |
| 6 | 106 | 30 |
| 7 | 1,212 | 979 |
| 8 | 277 | 158 |
| 9 | 238 | 117 |
| 10 | 649 | 160 |
| 11 | 2,555 | 2,000 |
| 12 | 599 | 446 |
| 13 | 233 | 144 |
| 14 | 2,229 | 185 |
| 15 | 266 | 198 |
| 16 | 769 | 176 |
| 17 | 234 | 160 |
| 18 | 948 | 39 |
| 19 | 39 | 24 |
| 20 | 72 | 59 |
| 21 | 253 | 165 |
| 22 | 4,843 | 2,650 |
| 23 | 163 | 103 |
| 24 | 112 | 102 |
RFA, radiofrequency ablation.
Table 2. CA 19.9 blood levels before and after the RFA procedure in group B, consisting of patients with stable CA 19.9 values or with oscillations of these levels between ±10 Units.
| Patients with stable CA 19.9 values (variations of ±10 Units) | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 740 | 747 |
| 2 | 28 | 26 |
| 3 | 0.6 | 0.6 |
| 4 | 21 | 22 |
| 5 | 19 | 20 |
| 6 | 0.8 | 0.6 |
| 7 | 6 | 6 |
| 8 | 238 | 237 |
| 9 | 161 | 166 |
| 10 | 225 | 223 |
RFA, radiofrequency ablation.
Table 3. CA 19.9 blood levels before and after the RFA procedure in group C, consisting of patients with CA 19.9 values increased more than 10 Units.
| Patients of group A (values reduced more than 10 Units) with CA 19.9 values reduced more than 20% | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 504 | 204 |
| 2 | 2,697 | 1,561 |
| 3 | 2,554 | 1,810 |
| 4 | 156 | 51 |
| 5 | 106 | 30 |
| 6 | 277 | 158 |
| 7 | 238 | 117 |
| 8 | 649 | 160 |
| 9 | 2,555 | 2,000 |
| 10 | 599 | 446 |
| 11 | 233 | 144 |
| 12 | 2,229 | 185 |
| 13 | 266 | 198 |
| 14 | 769 | 176 |
| 15 | 234 | 160 |
| 16 | 948 | 39 |
| 17 | 39 | 24 |
RFA, radiofrequency ablation.
Then patients of group A were divided into respectively four subgroups depending on the percentages of CA 19.9 levels decrease: the tumor marker were reduced less than 20% in 4/24 (17%) (Table 4) and more than 20% in 20/24 (83%) (Table 5); instead the tumor markers were reduced less than 30% in 8/24 (33%) (Table 6) and more than 30% in 16/24 (67%) (Table 7).
Table 4. CA 19.9 blood levels before and after the RFA procedure in subgroups of group A, consisting of patients with CA 19.9 values reduced more than 20%.
| Patients of group A (values reduced more than 10 Units) with CA 19.9 values reduced more than 20% | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 504 | 204 |
| 2 | 2,697 | 1,561 |
| 3 | 2,554 | 1,810 |
| 4 | 156 | 51 |
| 5 | 106 | 30 |
| 6 | 277 | 158 |
| 7 | 238 | 117 |
| 8 | 649 | 160 |
| 9 | 2,555 | 2,000 |
| 10 | 599 | 446 |
| 11 | 233 | 144 |
| 12 | 2,229 | 185 |
| 13 | 266 | 198 |
| 14 | 769 | 176 |
| 15 | 234 | 160 |
| 16 | 948 | 39 |
| 17 | 39 | 24 |
| 18 | 253 | 165 |
| 19 | 4,843 | 2,650 |
| 20 | 163 | 103 |
RFA, radiofrequency ablation.
Table 5. CA 19.9 blood levels before and after the RFA procedure in subgroups of group A, consisting of Patients with CA 19.9 values reduced less than 20%.
| Patients of group A (values reduced more than 10 Units) with CA 19.9 values reduced less than 20% | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 253 | 207 |
| 2 | 1,212 | 979 |
| 3 | 72 | 59 |
| 4 | 112 | 102 |
RFA, radiofrequency ablation.
Table 6. CA 19.9 blood levels before and after the RFA procedure in subgroups of group A, consisting of patients with CA 19.9 values reduced more than 30%.
| Patients of group A (values reduced more than 10 Units) with CA 19.9 values reduced more than 30% | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 504 | 204 |
| 2 | 2,697 | 1,561 |
| 3 | 156 | 51 |
| 4 | 106 | 30 |
| 5 | 277 | 158 |
| 6 | 238 | 117 |
| 7 | 649 | 160 |
| 8 | 233 | 144 |
| 9 | 2,229 | 185 |
| 10 | 769 | 176 |
| 11 | 234 | 160 |
| 12 | 948 | 39 |
| 13 | 39 | 24 |
| 14 | 253 | 165 |
| 15 | 4,843 | 2,650 |
| 16 | 163 | 103 |
RFA, radiofrequency ablation.
Table 7. CA 19.9 blood levels before and after the RFA procedure in subgroups of group A, consisting of patients with CA 19.9 values reduced less than 30%.
| Patients of group A (values reduced more than 10 Units) with CA 19.9 values reduced less than 30% | CA 19.9 values pre | CA 19.9 values post |
|---|---|---|
| 1 | 2,554 | 1,810 |
| 2 | 253 | 207 |
| 3 | 1,212 | 979 |
| 4 | 2,555 | 2,000 |
| 5 | 599 | 446 |
| 6 | 266 | 198 |
| 7 | 72 | 59 |
| 8 | 112 | 102 |
RFA, radiofrequency ablation.
The thermo-lesion mean CT density in pre-contrastographic, arterial and portal phases in group A was respectively 28 [16-37], 37 [19-57] and 39 [25-70] HU; in group B was respectively 26 [21-31], 31 [20-43] and 35 [26-55] HU; and in the group C was respectively 30 [19-36], 37 [25-58] and 39 [22-58] HU. Thermo-lesion mean CT density difference in all phases was not statistically significant among the three groups (P>0.05).
In group A tumors had a mean area and volume of 1,004 mm2 and 28,192 mm3, respectively; while the intratumoral thermo-lesions had a mean area and volume of 379 mm2 and 6,696 mm3, respectively. Therefore the mean ablation volume in percentage related to the treated tumor was 26%.
In group B tumors had a mean area and volume of 1,000 mm2 and 27,521 mm3, respectively; while the intratumoral thermo-lesions had a mean area and volume of 389 mm2 and 8,252 mm3, respectively. The mean ablation volume in percentage related to the treated tumor was 24%.
In group C tumors had a mean area and volume of 1,080 mm2 and 29,565 mm3, respectively; while the intratumoral thermo-lesions had a mean area and volume of 325 mm2 and 5,498 mm3, respectively. The mean ablation volume in percentage related to the treated tumor was 22%.
There was no statistically significant difference among the mean area, the mean volume and the mean ablation volume in percentage related to the treated tumor between the three groups (P>0.05).
Patients with CA 19.9 levels decrease were then divided in subgroups based on reduction of CA 19.9 levels less or more than 20% and 30% after RFA procedure. A tendency to a statistically significant difference was found in comparing the mean ablation volume in percentage related to the treated tumor between the two subgroups with less or more than 20% reduction of CA 19.9 levels (P=0.03) and between the two subgroups with less or more than 30% reduction of CA 19.9 levels (P=0.13). Correlating the percentage of necrosis volume and the percentage of CA 19.9 reduction in patients of group A, moderate linear correlation was in fact found in the present study (Figure 3). Finally, the thermo-lesion mean CT density does not differ statistically among all subgroups respectively in all phases of CT study.
Figure 3.
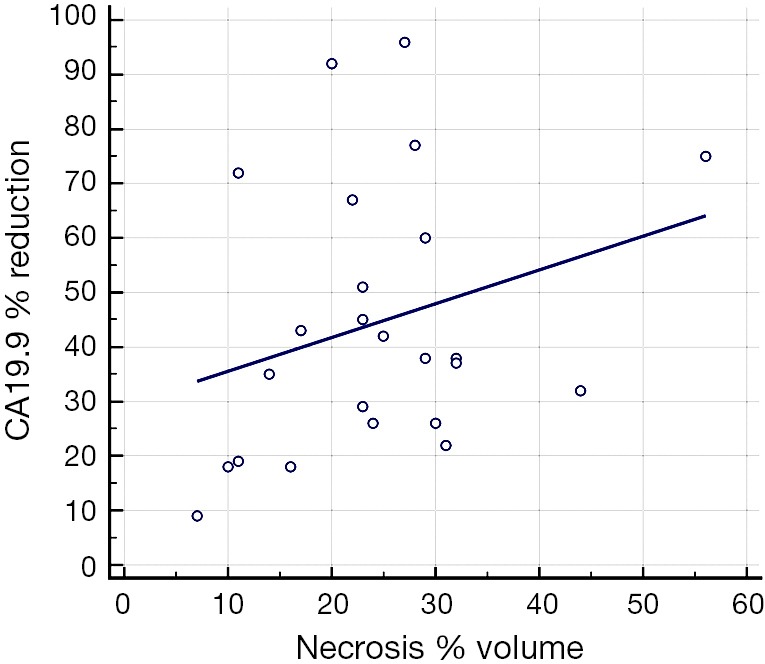
Scatter diagram with regression line of percentage of necrosis volume and the percentage of CA 19.9 reduction.
Discussion
RFA can be performed in non-metastatic locally advanced pancreatic tumor and currently it is performed under intra-operative ultrasound-guidance in the operating room during a laparotomy, with the immediate palliative aim to obtain a tumor reduction in a combined therapeutic program (4,16-21). Pancreatic tumors RFA is restricted only to locally advanced tumors, not resectable but not metastatic. It is reported that the necrotic area does not have to exceed the lesions margins, keeping safety margins in respect to vascular and digestive structures (4,5), so the ablation consequently will necessarily result to be incomplete compared to the whole lesion in every cases. Pancreatic ductal adenocarcinoma is usually markedly hypovascular, so after procedure the identification of necrotic area compared to remaining tumor tissue can be difficult. As a result of the present study it is important to evaluate the post-RFA necrotic area extension, while in pancreatic cancer RFA the persistency of remaining tumor at the periphery of treated area is an intrinsic aspect of the procedure (4,5). And therefore the RFA is a part of treatment that has to be necessarily completed by chemotherapy and/or radiotherapy (RT). RFA is become a palliative treatment that can be included in a combined therapeutic plane (6,16-18). Feasibility of this procedure was proved by Girelli et al. that standardized the technique and decreased the temperature applied thus obtaining reduction of complication rate (4).
Regarding patients survival after RFA, in literature there are controversial results (6,14,16,18).
Spiliotis et al. conclude that RFA may slow the tumor growth resulting in long-term survival, as in one of our patients, who lived 15 months after surgery without evidence of disease progression (14). In Casadei et al. experience, RFA remains a feasible procedure, but it presented a very high rate of postoperative complications with data, even if only on seven patients, suggesting no impact on survival (16). A recent review concludes that RFA seems to be feasible and safe when used with the correct temperature and it appears to have a positive impact on survival (6).
In our study we demonstrated that RFA of unresectable pancreatic adenocarcinoma induces reduction of CA 19.9 in 47% of the patients. The reduction of CA 19.9 was important and quantifiable in a percentage superior to 30% in 16 of 24 patients, while it was superior to 20% in 20 of 24 patients.
The lack of a statistically significant difference in the thermo-lesion mean CT density in all contrast-enhanced phases among the three groups proves that the RFA produced necrosis inside the tumors, so the stability or the increase of CA 19.9 blood levels in 27 of 51 patients were not caused by the lack of necrosis due to a failure of the RFA procedure; on the contrary the cause can be related to the systemic spread of the pancreatic cancer, so probably in these 27 patients the disease was diffuse out of pancreas even if not visible at pre-procedure staging. Accordingly, it must be stressed the importance of an accurate patients selection for RFA procedure. In the 24 of 51 patients that showed a decrease of CA 19.9 values, this study proves that there is a tendency to a correlation between that the larger is the ablated area the more important is the CA 19.9 levels reduction. This result that can appear intuitive, however, has not been previously proved for RFA of pancreatic cancer.
These results bring us to reassess critically the argumentations of possible therapeutic effect of RFA of pancreatic cancer based on immune stimulation independently on the size of the ablation area. This study demonstrated that the first therapeutic effect of RFA of the pancreatic cancer is based on the cytoreduction and is depends strictly from the size of the thermo-lesion. Many authors correlated the heat therapy applied to tumors with the potential trigger of an antitumor immunity cascade mechanism (8,22-25). Specifically in pancreatic cancer RFA this concept could lead to keep the ablation area as small as possible so to obtain a safer procedure cancelling the risk of damage of sensitive structures in peri-pancreatic area. This study proves instead, as expected, the importance of the size of the ablated area for the amount of tumor cytoreduction that remains the first therapeutic effect of the procedure. This concept in a future perspective should lead to the development of pancreatic RFA techniques in order to produce a greater tumoral necrosis, increasing protection mechanisms of surrounding structures.
Regarding patients with CA 19.9 levels increased after RFA in the present study it could be argue that a systemic disease was present at the time of the diagnosis not visible at standard imaging staging studies.
Commenting the results of the CT study during the follow up, it does not being an absolute density value of coagulative necrosis post-RFA of pancreatic cancer. The thermo-lesion, even if necrotic, on CT can show a little density increase during post-contrastographic phase. We considered this phenomenon an intrinsic behavior of intralesional coagulative necrosis that in a sub-acute phase (1 week) has still preserved tissue architecture (26) and that could disappear when necrosis evolves with lysosomal enzymes digestion. Some studies have revealed this temporary coagulative necrosis behavior in other tissue type (27,28).
At the end of this study, it has been proved that reduction of tumor marker in patient affected by unresectable pancreatic adenocarcinoma is possible by using RFA and that the entity of the reduction in related to the size of the thermo-lesion. Further studies remain absolutely necessary for clinical validation.
Conclusions
In the present study RFA induces reduction of CA 19.9 of unresectable pancreatic adenocarcinoma in about half of the cases. If present, there is a tendency of relationship between the entity of the reduction of CA 19.9 and the size of RFA necrotic area. Multicentre randomized trials are necessary to confirm the effect size of RFA and to assess the clinical impact minimizing selection and publication biases.
Acknowledgements
None.
Footnotes
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- 1.McWilliams JP, Lee EW, Yamamoto S, et al. Image-guided tumor ablation: emerging technologies and future directions. Semin Intervent Radiol 2010;27:302-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Goldberg SN, Grassi CJ, Cardella JF, et al. Image-guided tumor ablation: standardization of terminology and reporting criteria. J Vasc Interv Radiol 2009;20:S377-90. [DOI] [PubMed] [Google Scholar]
- 3.Carrafiello G, Laganà D, Cotta E, et al. Radiofrequency ablation of intrahepatic cholangiocarcinoma: preliminary experience. Cardiovasc Intervent Radiol 2010;33:835-9. [DOI] [PubMed] [Google Scholar]
- 4.Girelli R, Frigerio I, Salvia R, et al. Feasibility and safety of radiofrequency ablation for locally advanced pancreatic cancer. Br J Surg 2010;97:220-5. [DOI] [PubMed] [Google Scholar]
- 5.D'Onofrio M, Barbi E, Girelli R, et al. Radiofrequency ablation of locally advanced pancreatic adenocarcinoma: an overview. World J Gastroenterol 2010;16:3478-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fegrachi S, Besselink MG, van Santvoort HC, et al. Radiofrequency ablation for unresectable locally advanced pancreatic cancer: a systematic review. HPB (Oxford) 2014;16:119-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Keane MG, Bramis K, Pereira SP, et al. Systematic review of novel ablative methods in locally advanced pancreatic cancer. World J Gastroenterol 2014;20:2267-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dromi SA, Walsh MP, Herby S, et al. Radiofrequency ablation induces antigen-presenting cell infiltration and amplification of weak tumor-induced immunity. Radiology 2009;251:58-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hayes DF, Bast RC, Desch CE, et al. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst 1996;88:1456-66. [DOI] [PubMed] [Google Scholar]
- 10.Boeck S, Haas M, Laubender RP, et al. Application of a time-varying covariate model to the analysis of CA 19-9 as serum biomarker in patients with advanced pancreatic cancer. Clin Cancer Res 2010;16:986-94. [DOI] [PubMed] [Google Scholar]
- 11.Haas M, Heinemann V, Kullmann F, et al. Prognostic value of CA 19-9, CEA, CRP, LDH and bilirubin levels in locally advanced and metastatic pancreatic cancer: results from a multicenter, pooled analysis of patients receiving palliative chemotherapy. J Cancer Res Clin Oncol 2013;139:681-9. [DOI] [PubMed] [Google Scholar]
- 12.Wu Y, Tang Z, Fang H, et al. High operative risk of cool-tip radiofrequency ablation for unresectable pancreatic head cancer. J Surg Oncol 2006;94:392-5. [DOI] [PubMed] [Google Scholar]
- 13.Pezzilli R, Ricci C, Serra C, et al. The problems of radiofrequency ablation as an approach for advanced unresectable ductal pancreatic carcinoma. Cancers (Basel) 2010;2:1419-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Spiliotis JD, Datsis AC, Michalopoulos NV, et al. Radiofrequency ablation combined with palliative surgery may prolong survival of patients with advanced cancer of the pancreas. Langenbecks Arch Surg 2007;392:55-60. [DOI] [PubMed] [Google Scholar]
- 15.Tang Z, Wu YL, Fang HQ, et al. Treatment of unresectable pancreatic carcinoma by radiofrequency ablation with 'cool-tip needle': report of 18 cases. Zhonghua Yi Xue Za Zhi 2008;88:391-4. (in Chinese) [PubMed] [Google Scholar]
- 16.Casadei R, Ricci C, Pezzilli R, et al. A prospective study on radiofrequency ablation locally advanced pancreatic cancer. Hepatobiliary Pancreat Dis Int 2010;9:306-11. [PubMed] [Google Scholar]
- 17.Ikuta S, Kurimoto A, Iida H, et al. Optimal combination of radiofrequency ablation with chemoradiotherapy for locally advanced pancreatic cancer. World J Clin Oncol 2012;3:12-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Frigerio I, Girelli R, Giardino A, et al. Short term chemotherapy followed by radiofrequency ablation in stage III pancreatic cancer: results from a single center. J Hepatobiliary Pancreat Sci 2013;20:574-7. [DOI] [PubMed] [Google Scholar]
- 19.Cantore M, Girelli R, Mambrini A, et al. Combined modality treatment for patients with locally advanced pancreatic adenocarcinoma. Br J Surg 2012;99:1083-8. [DOI] [PubMed] [Google Scholar]
- 20.Girelli R, Frigerio I, Giardino A, et al. Results of 100 pancreatic radiofrequency ablations in the context of a multimodal strategy for stage III ductal adenocarcinoma. Langenbecks Arch Surg 2013;398:63-9. [DOI] [PubMed] [Google Scholar]
- 21.Giardino A, Girelli R, Frigerio I, et al. Triple approach strategy for patients with locally advanced pancreatic carcinoma. HPB (Oxford) 2013;15:623-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Basu S, Binder RJ, Suto R, et al. Necrotic but not apoptotic cell death releases heat shock proteins, which deliver a partial maturation signal to dendritic cells and activate the NF-kappa B pathway. Int Immunol 2000;12:1539-46. [DOI] [PubMed] [Google Scholar]
- 23.Baronzio G, Gramaglia A, Fiorentini G. Hyperthermia and immunity. A brief overview. In Vivo 2006;20:689-95. [PubMed] [Google Scholar]
- 24.Milani V, Noessner E. Effects of thermal stress on tumor antigenicity and recognition by immune effector cells. Cancer Immunol Immunother 2006;55:312-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Teng LS, Jin KT, Han N, et al. Radiofrequency ablation, heat shock protein 70 and potential anti-tumor immunity in hepatic and pancreatic cancers: a minireview. Hepatobiliary Pancreat Dis Int 2010;9:361-5. [PubMed] [Google Scholar]
- 26.Vinay Kumar, Abul K. Abbas, Nelson Fausto, et al. Robbins & Cotran Pathologic Basis of Disease, 8th Edition. Saunders: Elsevier, 2010. [Google Scholar]
- 27.Lee YJ, Lu DS, Osuagwu F, et al. Irreversible electroporation in porcine liver: acute computed tomography appearance of ablation zone with histopathologic correlation. J Comput Assist Tomogr 2013;37:154-8. [DOI] [PubMed] [Google Scholar]
- 28.Javadi S, Ahrar JU, Ninan E, et al. Characterization of contrast enhancement in the ablation zone immediately after radiofrequency ablation of renal tumors. J Vasc Interv Radiol 2010;21:690-5. [DOI] [PubMed] [Google Scholar]


