Abstract
The molecular tool kit for producing flat-bladed photosynthetic structures evolved in marine and terrestrial plants during the middle Paleozoic, but it took a further 20 million years before leaves suddenly spread throughout land floras. This delay has long been difficult to explain, given the apparent advantage of leaves for photosynthetic primary production. Theory and experiments predict that exceptionally high atmospheric CO2 levels in the middle Paleozoic delayed the origin of leaves by restricting stomatal development. This would have limited evaporative cooling, leading to lethal overheating of leaves absorbing large quantities of solar energy. Here we test the central prediction of this argument with a morphometric analysis of 300 plant fossils from major European collections. We show a 25-fold enlargement of leaf blades in two phylogenetically independent clades as atmospheric CO2 levels fell during the late Paleozoic. Furthermore, preliminary data suggest that the first abrupt increase in leaf size was accompanied by an 8-fold rise in stomatal density. These evolutionary patterns support the relaxation of biophysical constraints on leaf area predicted by theory and point to a significant role for CO2 in plant evolution.
The origin of megaphyll leaves was a major event in vascular land plant evolutionary history that proceeded through the developmental modification of lateral branch systems (1, 2). However, leaves did not become widespread in fossil floras until 50 million years after the emergence of vascular plants (3–5). This inordinately long delay is genuinely puzzling. Marine algae developed laminate photosynthetic organs some 10 mm wide as early as the Ordovician (6), and tiny megaphyll leaves are known from the rare Early Devonian vascular land plant Eophyllophyton bellum (7). Fossil evidence therefore demonstrates that the genes required for laminate structures had already evolved in independent groups, and that no intrinsic genetic or developmental barriers delayed the widespread appearance of leaves. Theory suggests an environmental barrier to leaf evolution, involving evaporative and convective cooling, leaf size, and CO2-mediated stomatal evolution (8). This mechanism draws attention to the fact that low stomatal frequencies of early land plants (9, 10) in the high CO2 atmosphere of the middle Paleozoic (Fig. 1A) placed a tight physical limit on evaporative energy loss (8). Large leaves developing in these plants would therefore have been prone to lethal overheating because of greater interception of solar energy and lower convective heat dissipation than a leafless stem (8). As atmospheric CO2 levels subsequently declined (11) (Fig. 1 A), stomatal numbers in land plants rose (9), and greater evaporative cooling ensued, alleviating the requirement for convective heat loss. Because the capacity for convective heat loss decreases with increasing leaf size, it follows that leaves became progressively larger as stomatal numbers rose and atmospheric CO2 declined in the late Paleozoic, a prediction that can be tested against observations from the fossil record.
Fig. 1.
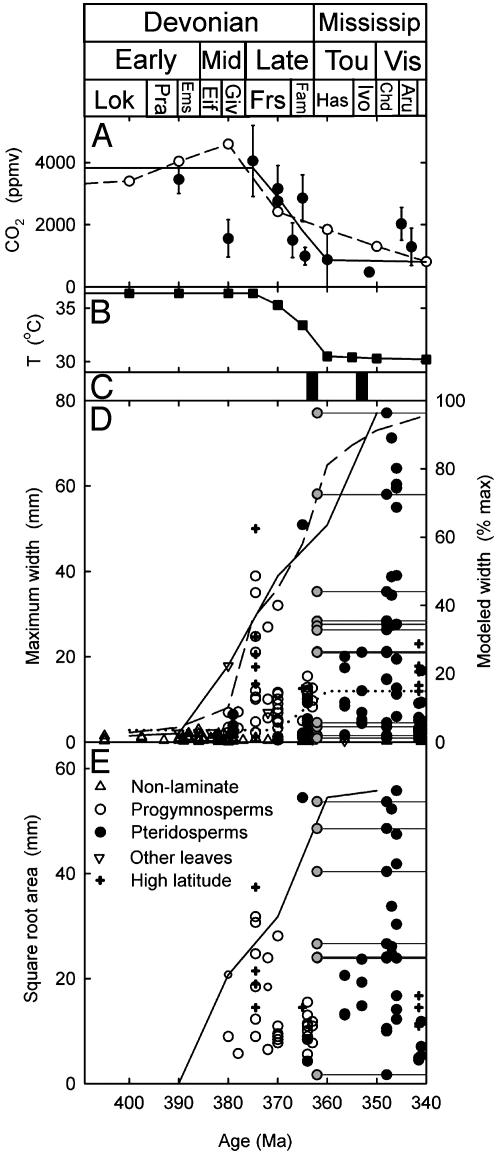
Leaf and atmospheric evolution in the Devonian and Mississippian. (A) Modeled (-○-) (11) or paleosol (•) [CO2], with a piecewise linear fit (solid line) and error bars indicating range of uncertainty (11). (B) Tropical air temperatures, calculated by assuming radiative forcing by CO2 (25) above a baseline value of 30°C. (C) Geologic evidence for continental glaciation (solid bars) (11, 19). (D) Maximum leaf or branch width. Range of uncertainty in the age of leaves classified only as Mississippian (Tou-Vis) (gray points and horizontal lines) and maximum size for 10-million-year intervals (solid line). Modeled leaf size with an increasing stomatal density, normalized as a percentage of its maximum value in the Visean (dashed line) and with stomatal density fixed at the mean Early Devonian value (dotted line). (E) Square root of leaf area, with symbols and lines as in D. Stratigraphic abbreviations follow the standard usage and geologic time scale of ref. 26.
We tested this theoretical linkage between plant and atmospheric evolution on geological time scales by compiling and analyzing an extensive dataset of 300 leaf fossil species from Devonian and Mississippian floras. Trends in leaf size were investigated by measuring maximum width and the square root of the lamina area, taken as an integrative measure of average leaf width. These dimensions together characterize the path length of air flow across the leaf, which is a major determinant of the boundary layer resistance to convective heat loss (12). The principal European fossil collections for the time interval of interest are highly influenced by coal exploration and mining and are therefore biased toward the paleotropics. However, rare specimens from higher paleolatitudes were included, adding a global dimension to the database. These allowed an assessment of the effects of a lower sun angle and cooler temperatures on leaf size.
Methods
Sampling for Devonian and Mississippian leaf fossils was extensive and designed to include well-preserved specimens for a wide range of species and collection localities (see Table 1, which is published as supporting information on the PNAS web site, for full details). The specimens were archived in European museum and university collections at the British Geological Survey (Keyworth, U.K.), Cardiff University (Cardiff, U.K.), National Museums of Scotland (Edinburgh), Natural History Museum (London), National Museum of Wales (Cardiff, U.K.), Royal Belgian Institute of Natural Sciences (Brussels), Senckenberg Institute (Frankfurt), Swedish Museum of Natural History (Stockholm), and University of Liège (Liège, Belgium). Our analysis excluded measurements for the rare morpho-taxa of detached fan-shaped leaves assigned to the artificial Order Palaeophyllales (13). The taxonomic affinity and ecology of these leaves are unknown (14), and their status as vascular plants remains uncertain in many cases, with the potential for confusion with laminate algae.
Measurements of the maximum leaf width and lamina area were made from high-resolution digital photographs of each specimen (Coolpix 995, Nikon), using image analysis and morphometrics software (tpsdig, Ver. 1.39; F. James Rohlf, Morphometrics, State University of New York, Stony Brook). Where important specimens of fossil species were not represented in museum collections, measurements were made by using published photographs or camera lucida drawings of fossils but not from interpretive reconstructions of the fossil plant. Maximum leaf width was defined as the largest intact dimension of the lamina or terminal nonlaminate branch, measured perpendicular to the rachis. The lamina corresponded to a pinnule in the case of pinnate fronds and the entire leaf in simple (nonpinnate) forms. The area was determined only for intact laminae with a maximum width >5 mm. The square root of lamina area is a measure of the path length for air flow over the leaf in any direction.
Cuticles are preserved very rarely in Devonian fossils of the earliest laminate leaves. However, cuticle preparations were examined for Archaeopteris macilenta (15), together with isolated cuticles showing close morphological affinity to Archaeopteris, found in association with Archaeopteris halliana and Archaeopteris obtusa (16). These dated to the Frasnian and therefore represented the first widespread appearance of megaphyll leaves in the plant fossil record. Stomatal density was determined by using randomly selected areas for each (n = 7, A. macilenta and n = 1, cf. Archaeopteris). Counts of stomatal frequency were made by using a light microscope (DMLB, Leica Microsystems, Milton Keynes, U.K.) on 13 fields of view (0.0625 mm2) for the former species and 7 for the latter. Guard cell length was measured by using an image analysis system (quantiment 500, Leica Microsystems) as an indicator of pore size by sampling 24 guard cells for A. macilenta and 5 for cf. Archaeopteris.
The resistance of fully open stomata to water vapor efflux (rs) was calculated following diffusion theory (12), as
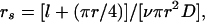 |
where l is the estimated depth of the stomatal pore (10 μm), r is the pore radius, estimated as 1/3 of the guard cell length, D is the diffusion coefficient for water vapor in air, and v is the density of stomata per unit leaf area.
Full simulations of the leaf energy budget were made following the method given in ref. 8. The maximum viable leaf size was calculated by adjusting the modeled path length for air flow over the leaf until a leaf temperature of 45°C was reached. At this point, lethal overheating was just avoided through the dissipation of heat by evaporation and convection. Two scenarios were considered. The first simulated changes in atmospheric CO2 and temperature, together with the observed evolution of stomatal density and guard cell size. The second considered only atmospheric change, with stomatal characters fixed at values typical for early leafless land plants.
Results and Discussion
Our analyses indicate a clear trend of rising maximum (Fig. 1D) and average (Fig. 1E) megaphyll leaf width between 380 and 340 million years ago that broadly coincides with the drop in atmospheric CO2 (Fig. 1 A) and cooling of tropical temperatures (Fig. 1 B and C). These temporal trends show that leaf blades became longer and broader, not simply less dissected, and are the result of two distinct phases of megaphyll evolution. The first widespread appearance of leaves occurs in the Progymnosperms during the Mid-Late Devonian (Fig. 1 D and E), with the evolutionary radiation of Archaeopteris (e.g., Fig. 2 A and B). Laminae become progressively larger through this interval (Fig. 1 D and E), reaching maximum widths of 5 mm in the Givetian (e.g., Fig. 2 A) and 39 mm in the Frasnian (e.g., Fig. 2B). This initial radiation of megaphylls is soon followed by a phylogenetically independent (5) origin and diversification of leaves within the Pteridosperms (e.g., Fig. 2C) during the Famennian and Mississippian (Fig. 1 D and E). The Pteridosperms are characterized by large multipinnate megaphylls (“fronds”), and the leaflets of this group are significantly larger in our analysis than the older Progymnosperm leaves, attaining a maximum width of 77 mm (Fig. 1D) only after atmospheric CO2 fell to its lowest level (Fig. 1 A). Overall, this pattern of change argues for a biophysical rather than a developmental or phylogenetic driver of the trend.
Fig. 2.
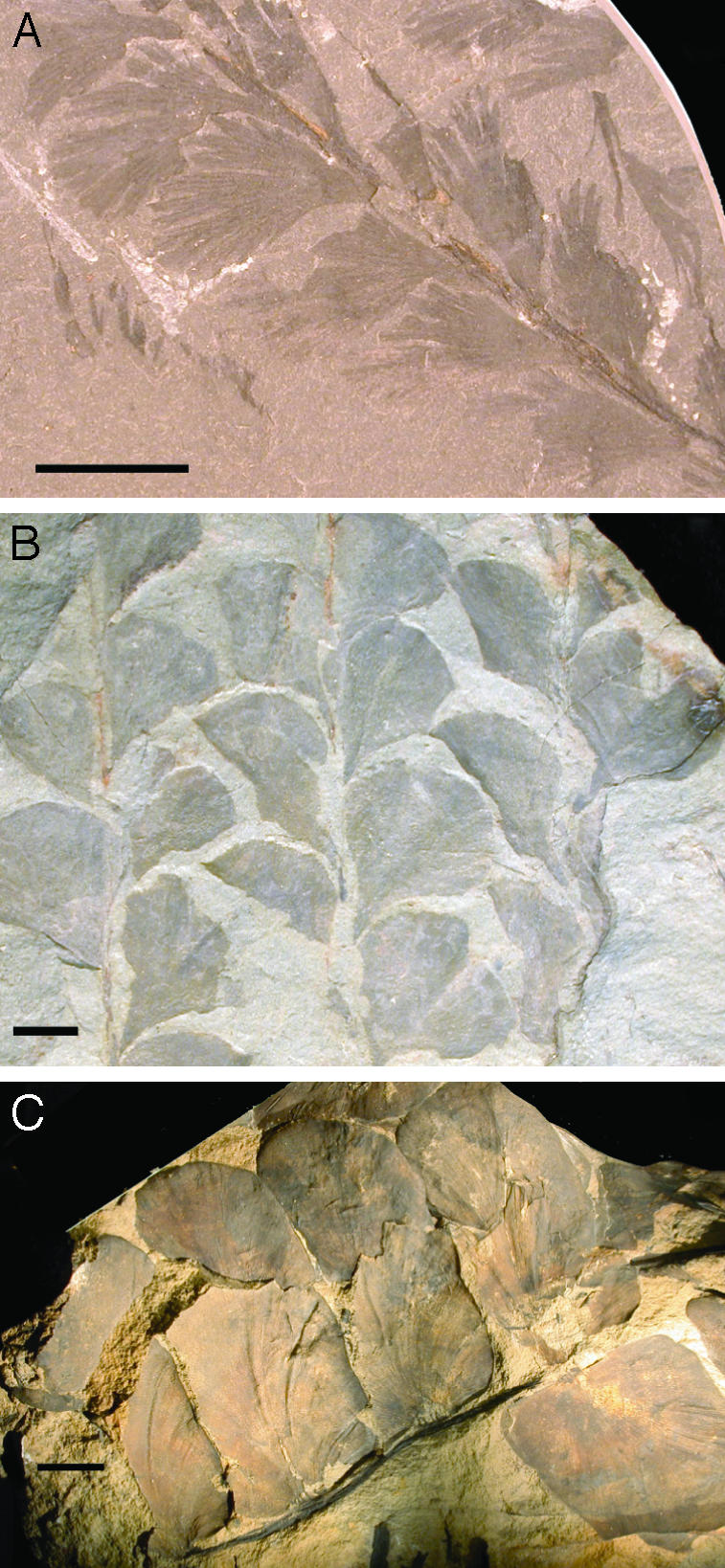
Examples of significant Devonian and Mississippian leaf fossils. (A) A. macilenta (Progymnospermopsida, Heist-op-den-Berg, Belgium; Royal Belgian Institute of Natural Sciences, Brussels, catalogue no. 99.730). (B) A. obtusa (Progymnospermopsida, Youngsville, NY; Senckenberg Institute, Frankfurt, catalogue no. SM B16765). (C) Fryopsis frondosa (Lagenostomopsida, Niederburbach, Germany; Natural History Museum, London, catalogue no. V52540). [Bars = 10 mm (A) and 15 mm (B and C).]
Until now, stomatal frequencies have not been reported for leaves during their initial radiation, making it difficult to establish a direct linkage between leaf size and the potential for evaporative cooling. However, our examination of fossil cuticles (see Methods) belonging to two Frasnian Archaeopteris species (15, 16) shows stomatal densities (A. macilenta 32.0 ± 0.8 mm–2 and cf. Archaeopteris 36.6 ± 3.0 mm–2) eight times greater than those of axial Early Devonian plants (4.5 ± 0.4 mm–2) (9, 10). The rise in density accompanied a 20-fold increase in the width of the photosynthetic organ (Fig. 1D). Moreover, the guard cells of Archaeopteris were large (62 ± 3 μmin A. macilenta and 57 ± 4 μm in cf. Archaeopteris), approaching the maximum observed in any modern or fossil plants (17). These evolutionary shifts in stomatal anatomy are consistent with the effects of rising CO2 on modern land plants (18) and would have lowered the resistance of fully open stomata to water vapor (see Methods) by >10 times compared with their axial ancestors (12).
The major Late Paleozoic decline in atmospheric CO2 levels was accompanied by significant climatic cooling (19), which partially alleviated the risk of overheating in large primitive leaves. In a mathematical analysis, we isolated and quantified the relative roles of climate and stomata by simulating changes in leaf energy balance (8, 20) and either varying or holding constant stomatal characteristics and climate. The analysis showed that cooling without changes in stomatal density allowed maximum leaf size to increase by only three times (Fig. 1D). Intriguingly, measurements of leaf size for scarce Archaeopteris fossils from two high paleolatitude Devonian localities in South America (21) support this result. These show maximum widths ≈30% larger than low latitude plants of equivalent age (Fig. 1D). Repeating the energy balance calculations with the observed increases in stomatal numbers allowed simulated tropical leaf size to rise 25 times, in agreement with the fossil evidence (Fig. 1 D and E). Our analysis therefore indicates that stomatal density plays a much greater relative role than climatic cooling in allowing progressive evolution of larger leaves.
We have shown that the origin and subsequent diversification of leaves tracks independent evidence for declining atmospheric CO2 levels. The observed trends are entirely consistent with the relaxation of an energetic constraint mediated by CO2-driven stomatal evolution. However, we recognize that once flat leaves had evolved, in concert with root and vascular systems (22, 23), they would have quickly propagated throughout terrestrial ecosystems by conferring significant selective benefits in competition for light and space. This competition is manifested in the fossil record by the well-documented evolution of plant height during the same interval (22). Viewed in this context, declining CO2 levels may have lifted a barrier to the evolution of leaves, much as rising O2 may have done for the Cambrian invertebrate explosion (24).
Supplementary Material
Acknowledgments
This work was supported by a Leverhulme Trust research grant (to D.J.B. and W.G.C.) and by Royal Society University Research Fellowships (to C.P.O. and D.J.B.). We thank P. Kenrick (Natural History Museum, London), V. Wilde (Senckenberg Institute, Frankfurt), M. Howe and P. Taylor (British Geological Survey, Nottingham, U.K.), E. M. Friis and Th. Denk (Swedish Museum of Natural History, Stockholm), C. Berry (Cardiff University, Cardiff, U.K.), M. Fairon-Demaret and P. Gerrienne (University of Liège, Liège, Belgium), F. Damblon (Royal Belgian Institute of Natural Sciences, Brussels), and J. Hilton (National Museums of Scotland, Edinburgh) for kind assistance and access to fossil collections. We thank K. Niklas, M. A. Gandolfo (Cornell University, Ithaca, NY), and P. G. Gensel (University of North Carolina, Chapel Hill) for loans of fossil cuticles; D. Royer, C. Wellman, and M. C. Press for comments on the manuscript; and F. J. Rohlf for advice on morphometric analyses.
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Cronk, Q. C. B. (2001) Nat. Rev. Genet. 2, 607–619. [DOI] [PubMed] [Google Scholar]
- 2.Zimmermann, W. (1930) Die Phylogenie der Pflanzen (Fischer, Jena, Germany).
- 3.Gensel, P. G. & Andrews, H. N. (1984) Plant Life in the Devonian (Praeger Scientific, New York).
- 4.Kenrick, P. & Crane, P. R. (1997) Nature 389, 33–39. [Google Scholar]
- 5.Boyce, C. K. & Knoll, A. H. (2002) Paleobiology 28, 70–100. [Google Scholar]
- 6.Fry, W. L. (1983) Rev. Pal. Pal. 39, 313–341. [Google Scholar]
- 7.Hao, S. G., Beck, C. B. & Wang, D. M. (2003) Int. J. Plant Sci. 164, 71–75. [Google Scholar]
- 8.Beerling, D. J., Osborne, C. P. & Chaloner, W. G. (2001) Nature 410, 352–354. [DOI] [PubMed] [Google Scholar]
- 9.McElwain, J. C. & Chaloner, W. G. (1995) Ann. Bot. 76, 389–395. [Google Scholar]
- 10.Edwards, D. (1998) Phil. Trans. R. Soc. London B 353, 141–157. [Google Scholar]
- 11.Crowley, T. J. & Berner, R. A. (2001) Science 292, 870–872. [DOI] [PubMed] [Google Scholar]
- 12.Nobel, P. S. (1991) Physicochemical and Environmental Physiology (Academic, San Diego).
- 13.Arber, E. A. N. (1912) Trans. Linn. Soc. London Bot. 7, 391–407. [Google Scholar]
- 14.Høeg, O. A. (1967) in Traité de Paléobotanique, ed. Boureau, E. (Masson, Paris), Vol. 2, pp. 362–399. [Google Scholar]
- 15.Carlucchio, L. M., Hueber, F. M. & Banks, H. P. (1966) Am. J. Bot. 53, 719–730. [Google Scholar]
- 16.Gensel, P. G. & Barnett-Lawrence M. (1996) in Devonian Fishes and Plants of Miguasha, Quebec, Canada, eds. Schultze, H.-P. & Cloutier, R. (Pfeil, Munich,), pp. 79–90.
- 17.Hetherington, A. M. & Woodward, F. I. (2003) Nature 424, 901–908. [DOI] [PubMed] [Google Scholar]
- 18.Woodward, F. I. (1987) Nature 327, 617–618. [Google Scholar]
- 19.Royer, D. L., Berner, R. A., Montañez, I. P., Tabor, N. J. & Beerling, D. J. (2004) GSA Today 14, 10.1130/1052-5173.
- 20.Beerling, D. J. & Woodward, F. I. (1997) Bot. J. Linn. Soc. 124, 137–153. [Google Scholar]
- 21.Berry, C. M., Morel, E., Mojica, J. & Villarroel, C. (2000) Geol. Mag. 137, 257–268. [Google Scholar]
- 22.Chaloner, W. G. & Sheerin, A. (1979) in The Devonian System, eds. House, M. R., Scrutton, C. T. & Bassett, M. G. (Palaeontological Society Special Paper in Palaeontology, no. 23), pp. 145–161.
- 23.Algeo, T. J. & Scheckler, S. E. (1998) Philos. Trans. R. Soc. London B 353, 113–130. [Google Scholar]
- 24.Knoll, A. H. & Carroll, S. B. (1999) Science 284, 2129–2137. [DOI] [PubMed] [Google Scholar]
- 25.Kothavala, Z., Oglesby, R. J. & Saltzman, B. (1999) Geophys. Res. Lett. 26, 209–212. [Google Scholar]
- 26.Harland W. B., Armstrong, R. L., Cox, A. V., Craig, L. E., Smith, A. G. & Smith, D. G. (1990) A Geologic Time Scale (Cambridge Univ. Press, Cambridge, U.K.).
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.