Abstract
Cocaine addiction continues to be a major health and social problem in the United States and other countries. Currently used pharmacological agents for treating cocaine abuse have proved inadequate, leaving few treatment options. An alternative is to use protein-based therapeutics that can eliminate the load of cocaine, thereby attenuating its effects. This approach is especially attractive because the therapeutic agents exert no pharmacodynamic action of their own and therefore have little potential for side effects. The effectiveness of these agents, however, is limited by their inability to act directly within the CNS. Bacteriophage have the capacity to penetrate the CNS when administered intranasally. Here, a method is presented for engineering filamentous bacteriophage to display cocaine-binding proteins on its surface that sequester cocaine in the brain. These antibody-displaying constructs were examined by using a locomotor activity rodent model to assess the ability of the phage-displayed proteins to block the psychoactive effects of cocaine. Results presented demonstrate a strategy in the continuing efforts to find effective treatments for cocaine addiction and suggest the application of this protein-based treatment for other drug abuse syndromes.
Cocaine is highly addictive and may be the most reinforcing of all drugs of abuse (1–3). Despite intensive efforts, effective therapies for cocaine craving and addiction remain elusive. Unlike the historically successful methadone treatment for heroin addiction, there is no proven pharmacotherapy for cocaine abuse (4). A number of medications acting as agonists, antagonists, or antidepressants have been evaluated in both animal models and humans, with only limited success (5–11). In the absence of a single highly effective drug, available pharmacological agents must be part of a comprehensive approach toward treatment.
Unquestionably, an improved pharmacotherapy would increase the effectiveness of such programs, and alternative strategies for treating cocaine addiction are needed if progress is to be made. One such strategy is to use protein-based therapeutics, whereby proteins are designed to bind cocaine, thereby blocking its effects, and/or degrade cocaine via hydrolysis of the benzoyl ester, thus rendering it less psychoactive (12). Over the last decade, several groups have reported the successful blocking of the psychostimulatory effects of cocaine by anticocaine antibodies with both active and passive immunization in rodent models. These results demonstrate that anticocaine antibodies bind to cocaine in circulation, retarding its ability to enter the brain (13–17). Both strategies reduce cocaine-induced locomotor activity and self-administration in rats. A different antibody-based approach to cocaine addiction treatment uses catalytic antibodies specific for cocaine and the cleavage of its benzoyl ester (18–23). The efficacy of catalytic antibodies has been demonstrated in rodent models of cocaine overdose and reinforcement, but kinetic constants for all reported antibody catalysts are marginal and, thus, improved rates will be required before clinical development is warranted (24). Finally, groups using butyrylcholinesterase (BChE), the major cocaine-metabolizing enzyme present in the plasma of humans and other mammals (25, 26) have reported that i.v. pretreatment with either wild-type or genetically engineered BChE can mitigate the behavioral and physiological effects of cocaine and accelerate its metabolism (27–29). One drawback common to all of these protein-based approaches is that none can act directly within the CNS; thus, their success depends solely on peripheral contact between the enzyme or antibody with ingested cocaine.
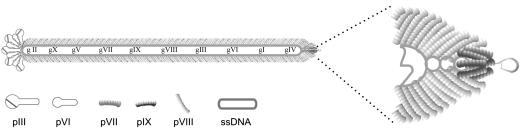
Bacteriophage are viruses that infect bacteria and are distinct from animal and plant viruses in that they lack intrinsic tropism for eukaryotic cells (30). Filamentous bacteriophage fd can be produced at high titer in bacterial culture, making production simple and economical. Furthermore, phage are extremely stable to a variety of harsh conditions, such as extremes in pH and treatment with nucleases or proteolytic enzymes (30). However, perhaps the most significant importance is the genetic flexibility of filamentous phage. In 1985, Smith reported a method that physically linked genotype and phenotype in a protein display system, and this technology has become known as phage display (31); it allows a wide variety of proteins, antibodies, and peptides to be displayed on the phage coat (Fig. 1).
Fig. 1.
Filamentous phage fd architecture.
Advances in filamentous phage display for in vitro application have been described wherein phage displaying a random peptide library were intravenously injected into mice and subsequently rescued from the internal organs, showing that the integrity of the phage was not compromised (32, 33); and a report in which filamentous phage were shown to penetrate the CNS has been published (34). In this later study, Solomon and coworkers (34) were able to deliver phage-displayed anti-β-amyloid antibodies via intranasal administration into the brains of mice. This paper is significant because it provides the following findings: (i) filamentous phage can access the CNS, (ii) phage can display foreign proteins on its surface and still penetrate the CNS, and (iii) bacteriophage can be injected multiple times into the same animal without visible toxic effects.
Previously, we have shown that sequestering of cocaine by anticocaine antibodies can suppress the psychomotor and reinforcing actions of the drug (13–15). We note the emergence from these studies of a murine monoclonal antibody termed GNC 92H2 that has exquisite affinity and specificity to cocaine (Kd = 40 nM) and benzoyl ecgonine (Kd = 1.4 μM) (35). We have also demonstrated that antibody libraries can be displayed on the filamentous phage coat proteins pIII, pVII, and pIX for the selection of high-affinity antibodies to a wide variety of antigens (36, 37). Herein, we detail the therapeutic potential of a phage-displayed protein (GNC 92H2-pVIII) that is designed to be not only highly specific for binding of cocaine but that can also access and act directly within the CNS as an additional mode of drug abuse therapy.
Materials and Methods
Preparation of Phage-Display Vectors. The phage-display vector pCGMT-p8 that was used in this study was derived from the phagemid pCGMT (38). The DNA sequence encoding the C terminus of the coat protein III (pIII) gene in pCGMT was replaced with the major coat protein VIII (pVIII) gene. The vector also contains the cloning site for the single chain (scFv) genes with two SfiI restriction sites. The genes for the scFv antibodies GNC 92H2 and RCA6028 were amplified by using PCR methodology (36, 37). The PCR products were agarose gel purified, recovered (Qiagen, Valencia, CA), digested with the restriction enzyme SfiI (New England Biolabs), and ligated into pCGMT-p8. DNA sequencing was used to confirm the construct.
Preparation and Purification of Phage Particles Displaying scFv GNC 92H2. Escherichia coli TG1 cells (Stratagene) were transformed with the phagemid encoding the appropriate scFv antibody. E. coli TG1 cultures were grown in 2 × 0.5 liters of 2YT broth in the presence of the antibiotic carbenicillin (100 μg/ml). Upon an optical density at a wavelength at 600 nm (OD600) of 0.8, the cells were infected with 0.5 ml of VCS M13 helper phage (Stratagene) (1012 plaque-forming units/ml). After 30 min incubation at room temperature, the culture was grown for 2 h at 37°C. Kanamycin/isopropyl β-d-thiogalactoside were then added to a final concentration of 70 μg/ml, and the culture was grown overnight at 30°C. After growth overnight, the bacterial cells were removed by centrifugation, and phage particles were harvested from the supernatant by precipitation with NaCl (3% wt/vol) and polyethylene glycol (PEG) 8000 (4% wt/vol). The phage pellet was resuspended in sterile endotoxin-free PBS (Invitrogen) and precipitated again. Upon resuspension of the pellet in 4 ml of PBS, the phage solution was filtered through a pyrogen-free 0.45-μm cellulose-acetate filter to remove any remaining bacterial cells. The phage preparation was titered, i.e., the number of colony forming units was determined according to standard protocols (39).
Affinity Measurements of Phage-Displayed Proteins. Equilibrium dialysis was performed by using [3H]cocaine as the ligand and phage scFv GNC 92H2-pVIII; helper phage VCS M13 and RCA6028-pVIII were also measured and used as controls. Phage samples were serially diluted on a 96-well microtiter plate (80 μl per well). Wells (12 per sample) were then filled with another 80 μl of [3H]cocaine in PBS (2 nM per well). A second plate was prepared with 2 × 12 wells containing just PBS (160 μl per well). The two plates were tightly connected with filled wells facing each other and separated with a dialysis membrane (cutoff 6,000–8,000 Da). The plates were attached vertically to a shaker and were shaken at high frequency for 24 h at room temperature, after which they were carefully separated. The membrane was discarded, and from each well 100 μl was transferred to a scintillation vial. Five milliliters of scintillation fluid was added to each vial, and radiation was counted for each sample for 5 min. The experiment was repeated twice for each serum sample. The average in differences in dosage per minute (DPM) between opposite wells was determined for each dilution of phage particles. The number of phage particles was determined spectrophotometrically (A320 subtracted from A266) by using a molar extinction coefficient of 1.006 × 104 M–1·cm–1, and a genome size of 3,722 bases for the modified phage.
Animals. Male Wistar rats (n = 16; Charles River Breeding Laboratories), weighing 200–225 g on arrival, were housed in groups of two in a humidity- and temperature-controlled (22°C) vivarium on a 12 h light/dark cycle (lights on at 10 p.m.) with free access to food and water. All behavioral procedures were performed during the light cycle. Before behavioral testing, each rat was handled by the experimenter for 10 min. All procedures were conducted in strict adherence to the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Intranasal Phage Administration Protocols. Animals were anesthetized with a bolus i.p. injection of 60 mg/kg pentobarbital (sodium salt, Sigma) diluted in physiological saline. Rats were placed dorsally with heads positioned to maximize residency of exogenous substances on the olfactory epithelium. Intranasal (i.n.) administration of GNC 92H2-pVIII (1.0 × 1014 plaque-forming units/ml) diluted in PBS (50 μl per naris) was administered over 15–30 min by using a Hamilton microsyringe (100 μl) with scilastic tubing. During i.n. administration, the opposite naris was closed to induce natural aspiration of the injected substance. Control animals received 50 μl of phage/sc RCA6028-pVIII, a single chain antibody that binds to RCA60, ricin. Injections were delivered twice per day for 3 consecutive days.
Detection and Identification of Filamentous Phage in Rat Brain. Brain samples were obtained in a time continuum throughout the phage-infusion regimen, testing days, and postchallenge days as to ascertain the time frame of introduction, residence and clearance of phage in the neural tissue. To this end, brains were collected on days 2–4, 7, 8, 10, 13, 15, and 17 after onset of phage infusion. Animals were deeply anesthetized with halothane vapor and rapidly decapitated. Brains were harvested from cerebellum to olfactory tubercule and immediately ground to a fine, homogenous consistency, and washed with and incubated in 3 ml of PBS (pH 7.4) for 1 h at room temperature. After the incubation time, the tubes were spun down at 1,000 rpm in a benchtop GS-6R centrifuge (Beckman). E. coli TG1 cells were infected with serial dilutions of the supernatant for 1 h at room temperature and plated onto LB broth agar plates containing carbenicillin (100 μg/ml). Colonies were counted the following day, and the titer was calculated based on serial dilution. Phagemids isolated from phage particles on days 4 and 7 were analyzed by using DNA sequencing at the Protein and Nucleic Acid Core facility of The Scripps Research Institute to positively identify the phage particles as well as to confirm the presence of the antibody gene.
Detection of Antifilamentous Phage Antibodies in Rat Serum. Blood was drawn from the jugular vein for serum IgG measurements after phage injections at day 28. Filamentous phage particles displaying scFv GNC 92H2 or RCA6028 on pVIII were coated on 96-well microtiter plates (NuncMaxiSorp) overnight at 4°C. Wells were washed five times and blocked with 5% (wt/vol) BLOT-QuickBlocker (Oncogene) in PBS (pH 7.4) at ambient temperature for 1 h. After subsequent washing, rat serum (serially diluted) was added into the wells and incubated for 1 h at room temperature. Repeated washing of the plate was followed by the addition of goat-anti-rat IgG-horseradish peroxidase (HRP) (Pierce) and goat-anti-rat IgM-HRP (Pierce), which was diluted 1:5,000 in blocking solution and was added followed by incubation for 1 h at ambient temperature. The plates were copiously washed, and HRP substrate (3,3′,5,5′-tetramethyl benzidine and hydrogen peroxide) was added according to the manufacturer's instructions (Pierce). Color reactions were read at a wavelength of 450 nm by using a Thermomax ELISA plate reader (Molecular Devices).
Behavioral Procedures. Locomotor activity was measured in a bank of 16 wire cages; each cage was 20 cm high × 25 cm wide × 36 cm long, with two horizontal infrared beams across the long axis 2 cm above the floor. Total photocell beam interruptions and crossovers, the number of times breaking the photocell beam followed directly by breaking the other photocell beam, were recorded by a computer every 10 min; background noise was provided by a white noise generator.
Before the phage-treatment regime, each rat was habituated to the photocell cages overnight, and before drug injection the rats were habituated again to the photocell cages for 90 min. To determine preimmunization drug response (baseline), animals received an i.p. injection of 15 mg/kg cocaine·HCl mixed in saline solution (bolus 1 ml/kg), and their locomotor responses were measured during a 90-min session. Based on locomotor activity scores, animals were assigned to the experimental (n = 8) or control group (n = 8) in ranking order; a between subjects design was implemented by using three different experimental groups: 10 mg/kg, n = 16; 15 mg/kg, n = 16; and 30 mg/kg, n = 16.
Stereotypic behavior (sniffing and rearing) was rated for 10 s every 10 min as described (40). Data were arranged in contingency tables in the following way: (i) for each response category and for each 10-min interval, the number of rats showing a particular category was tabulated; (ii) the degree of heterogeneity in each contingency table was then calculated by a likelihood ratio method (see Statistical Analysis).
On challenge days, animals received an i.p. injection of isotonic saline (bolus 1 mg/kg) and were habituated for 90 min before the drug injection. Locomotor activity was measured during habituation and the testing session as described above. The experimental design consisted of a 2 × 3 between-subjects design, where two different phage infusions and three cocaine doses were administered. Animals received either RCA6028-pVIII or GNC 92H2-pVIII, the anticocaine mAb-displaying phage. Cocaine doses ranged from 10, 15, and 30 mg/kg i.p. In all experiments, animals were challenged with their corresponding dose of cocaine at the fourth day from the onset of phage infusions, that is, the next day after the last phage treatment. Animals were subjected to cocaine challenges for 3 consecutive days.
Statistical Analysis. Locomotor activity data were analyzed by subjecting 10-min total means for locomotor activity to a two-factor ANOVA (group × time) with repeated measures on the within-group factor, time. Individual means comparisons for the main treatment effects were analyzed by using a Newman–Keuls a posteriori test. Stereotyped behavior data were analyzed by a likelihood ratio method, the “information statistic” (41, 42).
Results
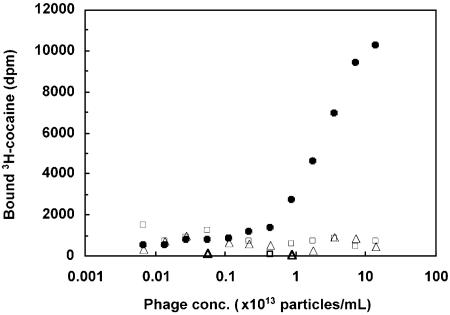
Validation of GNC 92H2-pVIII Affinity for Cocaine. The affinity of GNC 92H2-pVIII for cocaine was determined by using a RIA based on equilibrium dialysis with tritium-labeled cocaine and serial dilutions of phage. A comparison was made between the cocaine binding ability of GNC 92H2-pVIII with VCS M13 helper phage and with RCA6028-pVIII; the latter two phage constructs, in theory, are not expected to bind cocaine. As shown in Fig. 2, phage GNC 92H2-pVIII clearly bind cocaine, whereas control phage do not. Based on this binding curve, we estimate the Kd-avg of phage GNC 92H2-pVIII to be between 50 nM and 5 μM, depending on the number of scFv 92H2 antibodies displayed on each phage particle.
Fig. 2.
GNC 92H2-pVIII affinity for cocaine. The affinity of phage-displaying GNC 92H2-pVIII for cocaine, as determined by using equilibrium dialysis with [3H]cocaine and serial dilutions of phage. GNC 92H2-pVIII (filled circles), RCA6028-pVIII (open squares), and VCS M13 (open triangles).
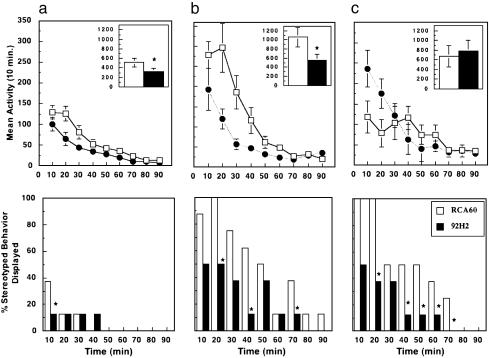
Psychomotor Response to Systemic Cocaine. The average weight of the animals upon completion of the studies was 365 ± 42 g (n = 16). The photocell cage habituation procedure resulted in consistent patterns of activity after saline injection: transient arousal (<20 min) followed by typically low levels of ambulation. Cocaine injection pretreatment baseline values were: RCA6028-pVIII, 523 ± 98.6; GNC 92H2-pVIII, 594 ± 121.5. Intranasal administration of phage required an average of 20 min of infusion time per naris. Spillage due to sneezing or abrupt movement was monitored by degree of wetness on tissue rostral drape. Substantial wetness merited dosing de novo. Fig. 3 shows the psychomotor response to cocaine after the intranasal phage administration regime. At the low dose of 10 mg/kg, cocaine elicited a significant motor differential between groups [Fig. 3a Upper, RCA6028-pVIII, 513 ± 94.29; GNC 92H2-pVIII, 317.25 ± 78.95; F(1,14) = 5.3, P < 0.05]. A time-dependent effect was reflected in the stereotypy levels displayed by animals at this dose (Fig. 3a Lower: 2Î = 76.2, df = 1, 9). In a different group, 15 mg/kg cocaine resulted in a highly pronounced difference in both psychomotor measures [locomotor: Fig. 3b Upper, RCA6028-pVIII, 1,064.375 ± 213.52; GNC 92H2-pVIII, 550.125 ± 135.89; F(1,14) = 6.875, P < 0.05; with significant main effects of treatment × time interaction: F(1,8) = 4.268, P < 0.001; stereotypy: Fig. 3b Lower, 2Î = 82.2, df = 1, 9; P < 0.05]. According to simple main effects analysis, differences between groups were greater from the 10- to 40-min time points of the session [time 10–20: F(1,8) = 7.27, P < 0.017; time 20–30: F(1,8) = 9.03, P < 0.009; time 30–40: F(1,8) = 4.18, P < 0.05]. The group receiving the higher dose of cocaine (30 mg/kg) displayed the classic pattern of behavior observed with psychomotor stimulant. Thus, at a high dose, locomotor activity was decreased because of a high amount of stereotyped behavior in relation to the former groups in the locomotor category (Fig. 3c Upper). Although the overall ANOVA test statistic did not reach significance, there were treatment × time interaction main effects [F(1,8) = 4.81; P < 0.001], with simple main effects during the first 10 min of the session [time 0–10: F(1,8) = 4.12, P < 0.05; and a marginal effect at time 10–20; F(1,8) = 3.996, P < 0.06). Lastly, a dramatic difference was observed in the stereotypy measure at this group as shown in Fig. 3c Lower (2Î = 91.7, df = 1, 9; P < 0.01).
Fig. 3.
Locomotor activity (crossovers; Upper) and stereotyped behavior (sniffing and rearing; Lower) after i.p. injection of cocaine after nasal immunization with GNC 92H2-pVIII (filled circles) or RCA6028-pVIII (open squares). The figure shows the response to postnasal immunization cocaine challenge at 10 (a), 15 (b), and 30 (c) mg/kg. (Upper) Values represent means ± SEM of 16 animals (n = 8). *, P < 0.05 ANOVA, significant difference between groups. (Lower) Data represent the percentage of incidence of the observed behavior. *, P < 0.05.
Subsequent challenges with cocaine resulted in sustained suppressive effects in some animals, albeit not significant as a group. Fig. 4 depicts the pattern of mean activity as a total 90-min session in a 2-within × 2-between subject's design where time (90 min) and cocaine challenge day (1 or 4) are the within-subjects factors, and cocaine dose (10, 15, or 30 mg/kg) and treatment (RCA6028-pVIII or GNC 92H2-pVIII) are the between-subjects factors. The significance achieved in this study was time-dependent with regards to days postphage infusion (cocaine challenge day 1 = day 4 after infusion; cocaine challenge day 4 = day 7 after infusion). As evidenced in Fig. 4, and complementarily with Fig. 3, psychomotor effects were significantly blocked by phage treatment only on the cocaine challenge day 1 for 10 and 15 mg/kg doses of the drug (left-most columns on Fig. 4 a and b) but not 30 mg/kg (right-most columns of Fig. 4 a and b) [day 1/10 mg/kg, RCA6028-pVIII: 513.75 ± 94.29; GNC 92H2-pVIII: 317.25 ± 78.95; day 1/15 mg/kg, RCA6028-pVIII: 1,064.38 ± 213.52; GNC 92H2-pVIII: 550.13 ± 213.52; day 1/30 mg/kg, RCA6028-pVIII: 675.1 ± 222.74, GNC 92H2-pVIII: 778.25 ± 225.71; day 4/10 mg/kg, RCA6028-pVIII: 592.64 ± 82.51; GNC 92H2-pVIII: 622.41 ± 105.43; day 4/15 mg/kg, RCA6028-pVIII: 862.25 ± 235.28; GNC 92H2-pVIII: 1391.38 ± 255.25; day 4/30 mg/kg, RCA6028-pVIII: 592.25 ± 299.5, GNC 92H2-pVIII: 839.75 ± 235.77].
Fig. 4.
Ambulatory behavior (crossovers) elicited by increasing doses of systemic cocaine (i.p.): 10 (a), 15 (b), and 30 (c) mg/kg in a between-subject design. Data are represented as total mean activity ± SEM of 48 animals (n = 8) from cocaine challenges on days 1 and 4 after phage infusion (days 4 and 7 after initial infusion). *, P < 0.05, ANOVA, significant difference between groups.
Analysis of Filamentous Phage Found in the Brain. To investigate the capability of filamentous phage to enter the CNS, the amount of phage that accumulates, and its duration of stay in the CNS, a phage titer experiment was conducted. Thus, on days 1–3, 1 × 1015 phage were administered intranasally twice daily to each rat. Whole brains were removed, washed, serially diluted, and allowed to infect bacteria. Phage were counted, and all numbers reported in Table 1 are based on a total of four brains used per day and an average number was calculated from this total. The threshold of phage detection was 105 colony-forming units; phage were not detected until day 3, and the highest level of phage was found on day 4. Phage-titer dropped off rapidly by day 7 but was persistent until day 13. Phagemids isolated from phage particles on days 4 and 7 were analyzed by using DNA sequencing, and the presence of the scFv antibody genes was confirmed. However, no phage was detected on day 17 or under the same experimental conditions with the brains of rats unimmunized.
Table 1. Phage titer detected in the rat brains after intranasal treatment of GNC92H2-pVIII.
| Day* | Phage titer† |
|---|---|
| 2 | None detected |
| 3 | 2.8 × 109 |
| 4 | 2.5 × 1013 |
| 7 | 1.3 × 1010 |
| 8 | 4.6 × 109 |
| 10 | 2.6 × 1011 |
| 13 | 2.1 × 1011 |
| 15 | 6.6 × 107 |
| 17 | None detected |
On days 1-3, an average of 1 × 1015 phage were administered intranasally into each rat.
A total of four rat brains was used for each day examined, and the titer was estimated based on the total counts divided by four.
Discussion
To assess the efficacy of immunization with phage-display antibodies within the CNS the psychostimulant effects of cocaine were measured in the rat. This psychostimulant effect is a dose-dependent increase in locomotor activity and stereotyped behavior as a result of cocaine's actions on dopaminergic neurons in the brain. Male Wistar rats were tested in photocell cages after treatment with i.p. cocaine (15 mg/kg) to determine preimmunization drug response. Three different doses of cocaine were chosen: 10, 15, and 30 mg/kg. These doses of cocaine represent a broad range of both locomotor and behavioral responses. Thus, the lowest dose produces little locomotor and virtually no stereotyped behavior, the medium dose produces a significant locomotor activation and modest stereotyped behavior, and the highest dose produces less locomotor activity but more robust stereotyped behavior.
Animals were administered intranasally twice per day for 3 consecutive days with phage-displaying single chain antibodies on their pVIII surfaces, including GNC 92H2-pVIII and RCA6028-pVIII (Figs. 1 and 2). The pVIII gene contains 2,800 copies, and as such was anticipated to provide an overall higher concentration of protein on the phage surface versus the more common display gene pIII, which can only exhibit up to five copies on its surface (39). Protein surface concentration was considered to be a key element in success of our approach because the antibodies displayed were not catalytic; hence, sequestering was the only means of inhibiting cocaine from reaching its target. Monoclonal antibody GNC 92H2 has previously been shown to have excellent avidity and specificity to cocaine and has yielded outstanding results in previous passive immunization behavioral studies (13–15). RCA6028 is a single chain antibody that has excellent affinity (400 nM) and selectivity to RCA60 (Ricinus Communis Agglutinin, ricin) and thus was considered a control (36).
Animals received three consecutive daily cocaine challenges of one of three doses of the drug 4 days after the onset of the phage-infusion regime. Intranasal administration of phage GNC 92H2-pVIII versus RCA6028-pVIII resulted in significant psychomotor differences between groups in response to cocaine (Fig. 3). At the 10 mg/kg dose, a 30% reduction in ambulatory behavior (crossovers) compared to baseline values was observed in the GNC 92H2-pVIII group but not in controls (Fig. 3a Upper), and this difference was reflected in the stereotypy measurement during the first 10 min of the session (Fig. 3b Lower). This modest difference in ambulation is not surprising given the tenuous hypermotility elicited at this dose. Furthermore, the paucity of the observed stereotypy is consistent with the reported negligible presence of this behavior at the low dose of cocaine (43). In contrast, a marked 47% decrease in locomotor activity was measured in GNC 92H2-pVIII-treated animals versus baseline values (15 mg/kg), whereas controls increased their overall responses by 11% (Fig. 3b Upper). This quantitative trend was also observed in the percent stereotypy displayed by this 15 mg/kg-treated group, where the behavior was rated in controls up to 70 min into the session, as opposed to 50 min in the GNC 92H2-pVIII group (Fig. 3b Lower). These results bear a striking similarity to those previously reported by our group using both active immunization with two different cocaine conjugates (13–15) and passive immunization with the mAb GNC 92H2 (15). This similarity probably is contingent on two main experimental factors. First, the same cocaine dose was used, therefore, the patterns of hyperactivity are congruent. Second, the cocaine-blocking mechanism, albeit central versus peripheral, still obeys an immune-mediated dynamic, which is subject to the same elements of affinity and titer as depicted by Fig. 3c. At the 30 mg/kg cocaine dose, a reversal of behavioral profile was obtained, whereby control animals showed diminishing levels of locomotion because of increasing stereotyped behavior compared to both their own baseline values and GNC 92H2-pVIII-treated rats during the first 30 min of the session (Fig. 3c Upper). Stereotyped behavior was sustained by controls significantly longer and at higher percentages than by the GNC 92H2-pVIII group (Fig. 3c Lower) reflecting the typical emergence of increased levels of this measure at the higher doses of cocaine (43). Therefore, the apparent absence of a blunting effect in locomotor activity in the GNC 92H2-pVIII-treated animals versus controls may instead be interpreted as a shift to the right of the cocaine dose/response function (see Fig. 3) as their repetitive (stereotypic) behavior increased and endured.
To confirm the basis of the behavioral suppression, we determined the presence of phage particles in the brain before, during, and after the time span of animal behavioral studies (Table 1). The earliest time point at which phage were observed in the brain was at day 3, whereas the highest titer of phage observed in the brain was at day 4. We note that high phage titers dropped precipitously from day 5 to day 7 (103), but were relatively constant at this number until day 15 and were not detected by day 17. Thus, upon subsequent challenges, i.e., day 4, there were no significant differences in either motor measure between groups. Fig. 4 provides comparative analysis between RCA6028- and GNC 92H2-pVIII-treated animals at day 4 after infusion-regime completion. Although no statistical significant differences were reached, the large error bars at the two higher doses denote sustained hypermotor suppression in three of the eight GNC 92H2-pVIII-treated rats (Fig. 4 b and c). It would thus appear that a threshold of phage-displaying protein must be present in the brain for a full-blunted behavioral response to be observed. We anticipate that this amount may be reduced if catalytic proteins for the drug of abuse are displayed on the appropriate gene's surface.
In understanding the role of our nasal vaccine, we felt it was important to investigate potential limitations. The CNS is considered an immune privileged site; however, the possibility of phage entering the periphery cannot be ruled out. Filamentous phage in itself, and with displayed proteins on its surface, comprises a foreign entity to the immune system. Additionally, there is a growing body of research wherein nasal vaccination has become increasingly popular (44, 45). Gratifyingly, ELISA analysis of rat serum from vaccinated animals showed no appreciable titer to phage and thus provides further evidence that potential toxic side effects are not being manifested in animals that were administered filamentous phage (32, 34).
We have shown a promising strategy in the continuing effort to find effective treatments for cocaine addiction. Whereas previous protein-based treatments have relied on peripheral drug-protein interactions, our approach delivers the therapeutic protein agent directly into the CNS, the site of drug action. Thus, convergence of phage display and immunopharmacotherapy has enabled us to investigate how a protein-based therapeutic acting within the CNS can influence the effects of cocaine in animal models. Future investigations should include the combination of this phage-based approach with either passive or active immunization protocols to determine whether any synergistic benefits can be obtained. Other tantalizing scenarios might comprise the display of two different proteins of interest on the phage using one protein to target the phage to a specific area of the brain and another protein to provide the actual therapeutic function, effectively increasing the concentration of the therapeutic protein in specific regions in the CNS. The application of this protein-based treatment for cocaine abuse may also serve as a therapeutic for other drug abuse syndromes, as well as any xenobiotic intoxication in which areas of the CNS are targeted.
Acknowledgments
We thank Drs. Beka Solomon and Louis G. Hom for initial helpful comments and Mike Arends for editorial assistance. This research was supported in part by National Institutes on Drug Abuse Grant DA015700, The Skaggs Institute for Chemical Biology, and the Pearson Center for Alcoholism and Addiction Research.
References
- 1.Rosecan, J. S. & Spitz, H. I. (1987) Cocaine Abuse: New Directions in Treatment and Research, eds. Spitz, H. I. & Rosecan, J. S. (Brunner/Mazel, New York).
- 2.Withers, N. W., Pulvirenti, L., Koob, G. F. & Gillin, J. C. (1995) J. Clin. Psychopharmacol. 15, 63–78. [DOI] [PubMed] [Google Scholar]
- 3.Mendelson, J. H. & Mello, N. K. (1996) New Engl. J. Med. 334, 965–972. [DOI] [PubMed] [Google Scholar]
- 4.Hall, W. C., Talbert, R. L. & Ereshefsky, L. (1990) Pharmacotherapy 10, 47–65. [PubMed] [Google Scholar]
- 5.Tennant, F. S., Jr., & Sagherian, A. A. (1987) Arch. Internal Med. 147, 109–112. [PubMed] [Google Scholar]
- 6.Berger, P., Gawin, F. & Kosten, T. R. (1989) Lancet 1, 283. [DOI] [PubMed] [Google Scholar]
- 7.Hubner, C. B. & Koob, G. F. (1990) Neuropsychopharmacology 3, 101–108. [PubMed] [Google Scholar]
- 8.Dackis, C. A., Gold, M. S., Sweeney, D. R., Byron, J. P., Jr., & Climko, R. (1987) Psychiatry Res. 20, 261–264. [DOI] [PubMed] [Google Scholar]
- 9.Kosten, T. R., Kleber, H. D. & Morgan, C. (1989) Biol. Psychiatry 26, 637–639. [DOI] [PubMed] [Google Scholar]
- 10.Mello, N. K., Lukas, S. E., Kamien, J. B., Mendelson, J. H., Drieze, J. & Cone, E. J. (1992) J. Pharmacol. Exp. Therapeutics 260, 1185–1193. [PubMed] [Google Scholar]
- 11.Arndt, I. O., Dorozynsky, L., Woody, G. E., McLellan, A. T. & O'Brien, C. P. (1992) Arch. Gen. Psychiatry 49, 888–893. [DOI] [PubMed] [Google Scholar]
- 12.Cashman, J. R. (1997) NIDA Res. Monogr. 173, 225–258. [PubMed] [Google Scholar]
- 13.Carrera, M. R. A., Ashley, J. A., Parsons, L. H., Wirsching, P., Koob, G. F. & Janda, K. D. (1995) Nature 378, 727–730. [DOI] [PubMed] [Google Scholar]
- 14.Carrera, M. R. A., Ashley, J. A., Zhou, B., Wirsching, P., Koob, G. F. & Janda, K. D. (2000) Proc. Natl. Acad. Sci. USA 97, 6202–6206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carrera, M. R. A., Ashley, J. A., Wirsching, P., Koob, G. F. & Janda, K. D. (2001) Proc. Natl. Acad. Sci. USA 98, 1988–1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fox, B. S., Kantak, K. M., Edwards, M. A., Black, K. M., Bollinger, B. K., Botka, A. J., French, T. L., Thompson, T. L., Schad, V. C., Greenstein, J. L., et al. (1996) Nat. Med. 2, 1129–1132. [DOI] [PubMed] [Google Scholar]
- 17.Kantak, K. M., Collins, S. L., Lipman, E. G., Bond, J., Giovanoni, K. & Fox, B. S. (2000) Psychopharmacology 148, 251–262. [DOI] [PubMed] [Google Scholar]
- 18.Landry, D. W., Zhao, K., Yang, G. X.-P., Glickman, M. & Georgiadis, T. M. (1993) Science 259, 1899–1901. [DOI] [PubMed] [Google Scholar]
- 19.Cashman, J. R., Berkman, C. E. & Underiner, G. E. (2000) J. Pharm. Exp. Ther. 293, 952–961. [PubMed] [Google Scholar]
- 20.Yang, G., Chun, J., Arakawa,-Uramoto, H., Wang, X., Gawinowicz, M. A., Zhao, K. & Landry, D. W. (1996) J. Am. Chem. Soc. 118, 5881–5890. [Google Scholar]
- 21.Baird, T. J., Deng, S.-X., Landry, D. W., Winger, G. & Woods, J. H. (2000) J. Pharmacol. Exp. Ther. 295, 1127–1134. [PubMed] [Google Scholar]
- 22.Matsushita, M., Hoffman, T. Z., Ashley, J. A., Zhou, B., Wirsching, P. & Janda, K. D. (2001) Bioorg. Med. Chem. Lett. 11, 87–90. [DOI] [PubMed] [Google Scholar]
- 23.Isomura, S., Hoffman, T. Z., Wirsching, P. & Janda, K. D. (2002) J. Am. Chem. Soc. 124, 3661–3668. [DOI] [PubMed] [Google Scholar]
- 24.Meijler, M. M., Matsushita, M., Wirsching, P. & Janda, K. D. (2004) Curr. Drug Discovery Technol. 1, 77–89. [DOI] [PubMed] [Google Scholar]
- 25.Gorelick, D. A. (1997) Drug Alcohol Dependence 48, 159–165. [DOI] [PubMed] [Google Scholar]
- 26.Mattes, C. E., Belendiuk, G. W., Lynch, T. J., Brady, R. O. & Dretchen, K. L. (1998) Addiction Biol. 3, 171–188. [DOI] [PubMed] [Google Scholar]
- 27.Nachon, F., Nicolet, Y., Viquie, N., Masson, P., Fontecilla-Camps, J. C. & Lockridge, O. (2002) Eur. J. Biochem. 269, 630–637. [DOI] [PubMed] [Google Scholar]
- 28.Mattes, C. E., Lynch, T. J., Singh, A., Bradley, R. M., Kellaris, P. A., Brady, R. O. & Dretchen, K. L. (1997) Tox. Appl. Pharmacol. 145, 372–380. [DOI] [PubMed] [Google Scholar]
- 29.Sun, H., Pang, Y.-P., Lockridge, O. & Brimijoin, S. (2002) Mol. Pharmacol. 62, 220–224. [DOI] [PubMed] [Google Scholar]
- 30.Larocca, D., Burg, M. A., Jensen-Pergakes, K., Ravey, E. P., Gonzales, A. M. & Baird, A. (2002) Curr. Pharm. Biotechnol. 3, 45–57. [DOI] [PubMed] [Google Scholar]
- 31.Smith, G. P. (1985) Science 228, 1315–1317. [DOI] [PubMed] [Google Scholar]
- 32.Pasqualini, R. & Ruoslahti, E. (1996) Nature 380, 364–366. [DOI] [PubMed] [Google Scholar]
- 33.Essler, M. & Ruoslahti, E. (2002) Proc. Natl. Acad. Sci. USA 99, 2252–2257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Frenkel, D. & Solomon, B. (2002) Proc. Natl. Acad. Sci. USA 99, 5675–5679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Moss, J. A., Coyle, A. R., Ahn, J.-M., Meijler, M. M., Offer, J. & Janda, K. D. (2003) J. Immunol. Methods 281, 143–148. [DOI] [PubMed] [Google Scholar]
- 36.Gao, C., Mao, S., Lo, C.-H. L., Wirsching, P., Lerner, R. A. & Janda, K. D. (1999) Proc. Natl. Acad. Sci. USA 96, 6025–6030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gao, C., Mao, S., Kaufmann, G., Wirsching, P., Lerner, R. A. & Janda, K. D. (2002) Proc. Natl. Acad. Sci. USA 99, 12612–12616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gao, C., Lin, C.-H., Lo, C.-H. L., Mao, S., Wirsching, P., Lerner, R. A. & Janda, K. D. (1997) Proc. Natl. Acad. Sci. USA 94, 11777–11782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Barbas, C. F., Burton, D. R., Scott, J. K. & Silverman, G. J. (2001) Phage Display: A Laboratory Manual (Cold Spring Harbor Lab. Press, Plainview, NY).
- 40.Fray, P. J., Sahakian, B. J., Robbins, T. W., Koob, G. F. & Iversen, S. D. (1980) Psychopharmacology 69, 253–259. [DOI] [PubMed] [Google Scholar]
- 41.Robbins, T. W. (1977) in Principles of Behavioral Pharmacology, Handbook of Psychopharmacology, eds. Iversen, L., Iversen, S. D. & Snyder, S. H. (Plenum, New York), Vol. 7, pp. 37–82. [Google Scholar]
- 42.Kullback, S. (1968) Information Theory and Statistics (Dover, New York).
- 43.Lyon, M. & Robbins, T. W. (1975) in Current Developments in Psychopharmacology, eds. Essman, W. & Valzelli, L. (Spectrum, New York), Vol. 2, pp. 89–163. [Google Scholar]
- 44.Illum, L. & Davis, S. S. (2001) Adv. Drug Delivery Rev. 51, 1–3. [PubMed] [Google Scholar]
- 45.Jones, N. (2001) Adv. Drug Delivery Rev. 51, 5–19. [DOI] [PubMed] [Google Scholar]