Abstract
Composite transdermal films of chitosan (CS)/montmorillonite K 10 (MMT) clay were prepared for the delivery of curcumin. CS/MMT films were evaluated for various physicochemical parameters. The films were characterized by Fourier transform infrared spectroscopy and X-ray diffraction analysis. Water uptake and swelling ratio of the films was found to decrease with increase in concentration of clay. Mechanical properties of the films were assessed in terms of tensile strength and extensibility using texture analyzer. Increase in tensile strength and reduction in extensibility was reported with increase in the clay content. This was attributed to the formation of intercalated structure and restriction in mobility of CS polymeric chains with the inclusion of clay particles. In vitro drug release study on transdermal films indicated pronounced sustained release of curcumin by the incorporation of clay particles in the CS polymer matrix. Stability study indicated no significant effect on physicochemical properties of films kept at 40°C and 75% RH for 3 months. Overall CS/MMT composite transdermal films exhibited improved mechanical and sustained drug release properties.
Keywords: Chitosan, chitosan/montmorillonite composites, curcumin, montmorillonite K 10, trasdermal fims
INTRODUCTION
Transdermal drug delivery systems are the dosage forms designed to deliver a therapeutically effective amount of drug across the skin. In order to deliver therapeutic agents through the human skin for systemic effects, the comprehensive morphological, biophysical, and physicochemical properties of the skin are to be considered.[1] Transdermal delivery provides a cutting edge over injectables and oral routes by increasing patient compliance and avoiding first pass metabolism. Transdermal delivery not only provides controlled administration of the drug, but also allows continuous input of drugs with short biological half-lives and eliminates pulsed entry into systemic circulation, which often causes undesirable side-effects. Hence transdermal system for drug delivery provide advantages of elimination of hepatic first pass metabolism, enhancement of therapeutic efficiency and maintenance of steady plasma level of the drug.[2] First transdermal system, transderm-SCOP was approved by Food and Drug Administration in 1979 for the prevention of nausea and vomiting associated with travel, particularly by sea. The evidence of percutaneous drug absorption may be found through measurable blood levels of the drug, detectable excretion of the drug and its metabolites in the urine and through the clinical response of the patient to the administered drug therapy.[3]
Chitosan (CS) has been widely studied for several decades for molecular separation, food packaging film, artificial skin, bone substitutes, water engineering and so on, owing to its good mechanical properties, biodegradability, biocompatibility, multiple functional groups as well as solubility in aqueous medium. However, poor thermal stability, hardness and gas barrier properties of CS are limiting its applicability. The chemical structure of CS containing multiple functional groups (hydroxyl, carbonyl, carboxyl, amine, amid) creates possibility for new bonding between CS chain and nano-filler particles, like clay (bentonite, montmorillonite [MMT]), silica (SiO2), and carbon nanotubes.[4]
Montmorillonite is 2:1 type aluminosilicate clay. Its crystalline structure presents an alumina octahedral between two tetrahedral layers of silica. The isomorphous substitution of Al3+ for Si4+ in the tetrahedral layer and Mg2+ for Al3+ in the octahedral layer results in a net negative surface charge on the clay. The imbalance of the negative charges at the surface is compensated by exchangeable cations (typically Na+ and Ca2+).[5]
Biopolymer/clay transdermal films as controlled drug-delivery vehicles have drawn much attention owing to their unique structure and properties. The synergistic effect of biopolymer and clay as well as the strong interfacial interactions between them (e.g., electrostatic and hydrogen bonding interaction) could advance the mechanical properties, swelling behavior, drug-loading efficiency, and controlled release behavior of the pristine biopolymer matrices.[6]
Curcumin is an orange-yellow crystalline powder obtained from rhizome of Curcuma longa (Zingiberaceae). Curcumin was first isolated in 1815, obtained in crystalline form in 1870 and identified as 1,6-heptadiene-3,5-dione-1,7-bis (4-hydroxy-3-methoxyphenyl)-(1E,6E) or diferuloyl methane. The feruloylmethane skeleton of curcumin was subsequently confirmed in 1910 by the initial work and synthesis by Lampe. Curcumin is a yellow–orange powder that is, insoluble in water and ether but soluble in ethanol, dimethylsulfoxide and acetone. Curcumin (also known as curcumin I) occurs naturally in the rhizome of C. longa, which is grown commercially and sold as turmeric, a yellow–orange dye. Turmeric contains curcumin along with other chemical constituents known as the “curcuminoids.”[7]
The present study was designed to prepare transdermal films of CS and MMT polymer composites. The transdermal films were evaluated in terms of thickness, weight variation, tensile strength, extensibility, surface pH, moisture content, percent moisture absorption, drug content, folding endurance, swelling ratio, in vitro dissolution studies and in vitro skin permeation studies. The transdermal films were also characterized by attenuated total reflectance/Fourier transform infrared spectroscopy (ATR-FTIR) and X-ray diffraction (XRD) techniques. Stability studies were also performed on the films.
MATERIALS AND METHODS
Materials
Chitosan of medium molecular weight (average molecular weight 92,700 g/mol) and MMT K10 were procured from Sigma-Aldrich Chemicals, USA. Curcumin was supplied as gift sample by Sanat Products Ltd., New Delhi, India. All other chemicals and reagents were of analytical grade.
Methods
Preparation of transdermal films
The transdermal films were prepared using CS, MMT and poly vinyl pyrollidone. CS solution was prepared by dissolving CS in a 2% (v/v) aqueous acetic acid solution, followed by centrifuging to remove the insoluble material. MMT was first swelled in 2% (v/v) aqueous acetic acid solution and then added to CS solution with continuous stirring (300 rpm) at 60°C for 4 h. Polyvinylpyrollidone was added as a film forming agent. Transdermal films were prepared using different ratios of CS and MMT [Table 1]. These were coded as CS/MMT 11, CS/MMT 41 AND CS/MMT 14 representing different ratios of CS and MMT that is, 1:1, 4:1 and 1:4 respectively. The formulation codes for different transdermal films are shown in Table 1.
Table 1.
Formulation and formula codes for transdermal films

Evaluation of transdermal films
Thickness
The thickness of transdermal film was measured using micrometer screw gauge (Mitutoyo, Japan) from three different points. The average and the standard deviation (SD) of the three readings were recorded.
Drug content
Preweighed sample of transdermal film was placed in 100 ml of phosphate buffer (pH 7.4) and agitated on mechanical shaker for 6 h. Solution was filtered and analyzed spectrophotometrically at 422 nm.[8]
Folding endurance test
The number times the films could be folded at the same place without breaking gave the value of the folding endurance. The films were folded at same place repeatedly either to break the films or to develop visible curves.
Weight variation test
Three films from each batch were weighed individually. The study was carried out for understanding inter-intra batch variation. The result of each batch was reported as average ± SD.
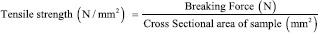
Tensile strength
The mechanical properties of CS/MMT transdermal films were evaluated using texture analyzer (TA XT plus, Stable Microsystems, Godalming Surrey, UK) equipped with 5 kg of load cell. Film strip in the dimension of 10 mm by 50 mm and free from air bubbles or imperfections, was held between two clamps positioned at a distance of 10 mm. The film was pulled by top clamp at a rate of 50 mm/min. The tensile strength and percent elongation were measured when the film broke.[9] The tensile strength formula is shown as below:
Extensibility
Extensibility was measured along with the tensile strength. The extensibility in the length of the exposed strip was calculated using a Vernier Caliper on addition of each weight until the strip broke. Extensibility is measured by the formula:
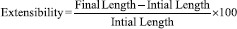
Surface pH determination
For the determination of surface pH of the patch a small area of the film was cut and was allowed to swell by keeping it in distilled water for 1 h in glass tubes. The surface pH was then noted by bringing a combined glass electrode near the surface of the film and allowing it to equilibrate for 1 min.
Moisture content analysis
The films were weighed individually and kept in desiccators containing 10 g of calcium chloride dehydrate as desiccant at 37°C for 24 h. The films were weighed individually until it showed a constant weight.[10] The percentage of the moisture content was calculated as:
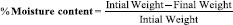
The studies were carried out in triplicates for each batch. The average and SD was determined.
Percentage moisture absorption
The films were weighed accurately and placed in a desiccator where a humidity condition of 75% RH was maintained by using saturated solution of sodium chloride. The films were taken out periodically and weighed for a period of 72 h. The percentage of moisture uptake was calculated as difference between final and initial weight of the film with respect to the initial weight:
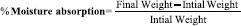
The studies were carried out in triplicates for each batch and the average and SD was determined.
Water uptake
The water uptake of the films was measured by immersing the films in phosphate buffer (pH 7.4) at room temperature for 24 h. Excess buffer on the surface of films was removed by blotting the surface with a tissue paper and the weight was then recorded.[11] This process was repeated at least three times for each sample. The water uptake of different samples was calculated using formula:
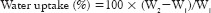
Where, W1 is the weight of completely dried sample and W2 is the weight of swelled sample in the phosphate buffer 7.4 at 37°C for every 30 min up to 24 h.
Swelling ratio
The completely dried, preweighed CS/MMT transdermal films were equilibrated in 250 ml of phosphate buffer (pH 7.4) at 25°C. The water uptake of the films is measured after 24 h using analytical balance. The swelling ratio (Q) of the films is calculated using the following equation:
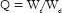
Where, Ws is the weight of the swollen film at different time intervals and Wd is the weight of dry film.
Attenuated total reflectance-Fourier transform infrared spectroscopy
Drug polymer interactions were checked by ATR-FTIR spectroscopy. ATR-FTIR spectra of the blank films and the optimized formulation was carried out using ATR-FTIR) spectrophotometer (Alpha, Bruker, Japan) in the reflectance mode with the wave number region 4000-650 cm−1.
X-ray diffraction
X-ray diffraction was used to investigate the crystallization of polymers and the model drugs as well as the effect of the plasticizer. The samples for the XRD studies were prepared by cutting films to fit the square tiles of the holder, mounted on the sample cell and scanned between 2 theta of 0-60° with counting time of 0.1 s step size. The X-ray patterns of the films were obtained with XPERT-PRO equipment (PANalytical, The Netherland) having an XRD commander programme.
In vitro drug release studies
Films with a predetermined surface area containing 25 mg curcumin were applied to a glass slide and covered with a stainless-steel mesh screen and clamped together. The assembly was placed at the bottom of the USP dissolution tester (Labindia DS 8000, India). In vitro drug release studies were carried out according to the paddle method. The height of the paddle from the surface of the assembly was adjusted to 2.5 cm. The vessel contained 900 ml phosphate buffer solution (pH = 7.4) and ethanol (50:50 v/v), the temperature was adjusted to 32°C and the speed was set at 50 r/min. Aliquots of 5 ml were withdrawn from the release medium at intervals through a sintered glass filter and replaced with equivalent amounts of buffer solution. The amount of drug released was determined spectrophotometrically (2202 systronics, India) at 422 nm. Blank experiments were performed simultaneously. Each experiment was carried out in triplicate.
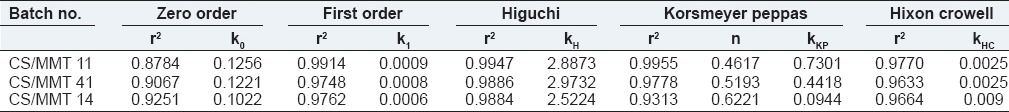
The in vitro drug release data of transdermal film formulations was fitted to various models such as zero-order (cumulative % drug release vs. time), first-order (log cumulative % drug remaining vs. time), Higuchi (cumulative % drug release vs. square root of time), Korsmeyer–Peppas (log cumulative % drug release vs. log time) and Hixon and Crowell (cube root of cumulative % drug remaining vs. time) models to ascertain the possible mechanism of drug release from the formulation.
In vitro skin permeation studies
In vitro drug permeation studies were carried out using all glass modified Franz diffusion cell having a volume of 20 ml in the receptor compartment. Full thickness rat skin was utilized for the permeation studies.
A full thickness rat skin was excised from the dorsal site of dead rat, and skin was washed with water. The fatty tissue layer was removed by using a spatula. The hair on the outer site of the skin was removed carefully using a scissor without damaging the stratum corneum. The skin was washed repeatedly with phosphate buffer, which would be used in the receptor medium and after 5 washings the absorbance of the washing buffer was taken to check any leaching of proteins from the skin. The skin was applied over the receptor compartment only after the absorbance of the washing buffer approached zero.
After applying the skin on the receptor compartment a film was applied over the skin which was fixed in its place by affixing the donor compartment over the receiver compartment. 1 ml samples were taken from the sampling port at predetermined intervals and the required amount of buffer was refilled back too. The diffusion cell consists of a receptor compartment of 20 ml volume. The diameter of the permeation area was 1 cm2 and area of 0.785 cm2. Phosphate buffer pH 7.2 was used as receptor solution. The volume of the diffusion cell was 20 ml and stirred with Teflon coated magnetic bead. The temperature was maintained at 32°C ± 2°C with the magnetic stirrer cum hot plate. The diffusion was carried out for 24 h and 1 ml of sample was withdrawn at time intervals of 1, 2, 4, 6, 8, 12, 16, 20 and 24 h. The same volume of phosphate buffer (pH 7.4) was added to receptor compartment to maintain sink conditions and the samples were analyzed at 422 nm wavelength using ultraviolet/visible spectrophotometer.
Stability studies
Stability studies were carried out for the optimized formulations under accelerated testing conditions for a period of 3 months. For stability study, the films were taken, wrapped with aluminum foil and placed in sample containers. The samples were withdrawn after 7, 15, and 30, 60 and 90 days. The transdermal films were evaluated for their physicochemical properties such as moisture content, tensile strength, and drug content.
RESULTS AND DISCUSSION
The prepared CS/MMT transdermal films were compact, smooth and without pores or imperfections. A synergistic effect of MMT for improving thermal stability, mechanical, and barrier properties of CS has been proposed. The evaluation of CS/MMT composite transdermal films in terms of various physicochemical properties, characterization FTIR and DSC studies and stability testing is discussed hereunder.
Thickness, drug content, weight variation, moisture content, moisture absorption, folding endurance and pH of transdermal films
Table 2 depicts various physicochemical properties of the prepared CS/MMT transdermal films. The polymeric combination of CS with MMT exhibited good film forming properties and the method of casting of films was found to produce good films. The thickness of the films vary from 0.13 ± 0.02 to 0.16 ± 0.03 mm while the drug content in the transdermal films range from 98.81% ± 1.02% to 99.69% ± 2.01%. The weight of the films ranged from 0.36 ± 0.04 to 0.39 ± 0.07 g. The results indicated that the method selected for the preparation of films was capable of producing films with uniform weight, content and minimal film variability. The folding endurance was found to be ranging between 250 ± 1.11 and 275 ± 2.50 indicating that the formulated films would maintain their integrity when applied to the skin. The pH of the films varied between 7.3 and 7.4.
Table 2.
Physicochemical properties of transdermal films
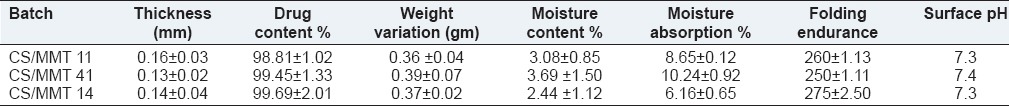
Low moisture content is useful for the long term stability of the transdermal films. It also reduces the brittleness and protects the formulation from microbial contamination. Moisture content and moisture uptake studies indicated that the increase in the concentration of hydrophilic polymer was directly proportional to the increase in moisture content and moisture uptake of the films, whereas increase in the concentration of hydrophobic polymer lead to the decrease in moisture content and moisture uptake of the films.
Water uptake
Water uptake capacity curves of the CS/MMT composite films and of pristine CS and MMT in phosphate buffer (pH 7.4) and 0.1N HCl (pH 1.2) at different time internals are depicted in Figures 1 and 2. It was observed that when the polymer films were treated in acidic pH, the CS/MMT 41 due to high content of CS showed highest water uptake capacity of 243% ± 3.79%. CS/MMT 14 having the lowest concentration of CS showed fairly less water uptake capacity of 92% ± 2.04%. Similarly, the water uptake capacity of CS/MMT 41 and CS/MMT 14 films in basic pH was 95% ± 2.13% and 48% ± 1.75% respectively. The MMT clay particles are dispersed in the CS polymer matrix leading to the formation of intercalated structures. Hence reduction is water uptake capacity of CS with the incorporation of MMT could be related to narrowing of pore channels and increase in diffusional path length of the polymer matrix. Kasirga et al. 2012 reported that there is a decrease in water vapor permeability with the incorporation of MMT in CS. This was proposed to be due to increase in diffusional path length and tortuous path because of dispersion of clay platelets in the polymer matrix.[12]
Figure 1.
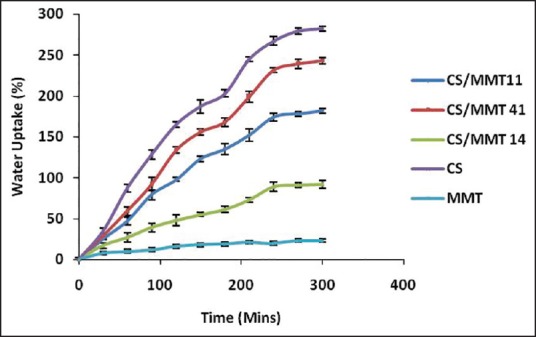
Water uptake capacity of chitosan/montmorillonite K 10 transdermal films in 0.1N HCl (pH 1.2)
Figure 2.
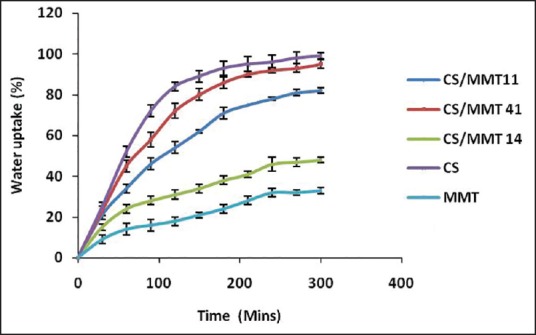
Water uptake capacity of chitosan/montmorillonite K transdermal films in phosphate buffer (pH 7.4)
Moreover, electrostatic interaction between protonated amine group of CS and negatively charged a site in the clay structure may also contribute toward a reduction in the water uptake capacity of CS with the incorporation of MMT clay particle. Similar findings of a reduction in swelling capacity and water uptake profile of CS with the incorporation of clay were reported by Salcedo et al.[13] and Depan et al.[14]
Swelling ratio
The swelling ratio studies performed on the transdermal films are shown in Figure 3. Similar to the water uptake studies, the swelling studies also showed a decrease in the swelling of the transdermal films with a decrease in the CS concentration. The swelling ratio was reported to be in the order CS/MMT 41> CS/MMT 11> CS/MMT 14. The behavior could be explained in terms that the MMT clay particles occupy the free space volume in the CS polymeric network thereby decreasing the volume available for swelling.[15]
Figure 3.
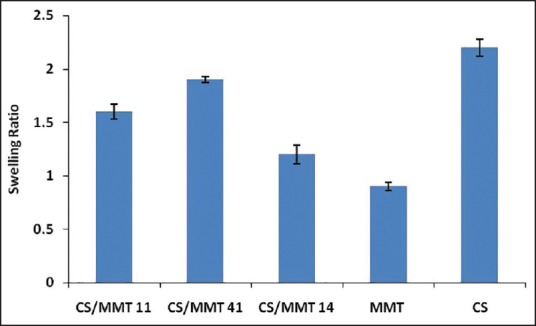
Swelling ratio of chitosan/montmorillonite K transdermal films
Mechanical properties
The mechanical strength of a CS/MMT films were described in terms its tensile strength and percentage of elongation to break that is, extensibility [Table 3]. A significant influence of the MMT concentration on the mechanical properties of the films was observed. Tensile strength was observed to increase with increase in the MMT content in the films. It was reported [Figure 4] that the tensile strength ranged from 23.12 ± 1.22 N/mm2 (pure CS film) to 54.11 ± 1.25 N/mm2 (CS/MMT 14 film). The improvement in tensile strength of the transdermal films might be attributed to the high-aspect ratio and rigidity which results from the strong affinity between the biopolymer and clay.[16] As reported evident from literature, tensile strength values of CS/MMT films increased significantly with increasing clay concentration due to a possible strain-induced alignment of the clay particle layers in the polymer matrix.[17]
Table 3.
Tensile strength and extensibility of CS/MMT transdermal films
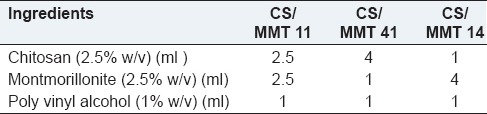
Figure 4.
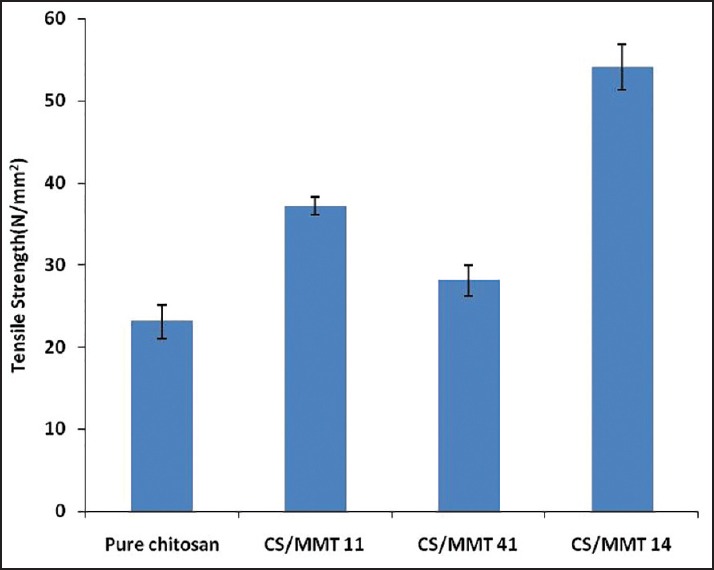
Tensile strength of transdermal films
The extensibility [Figure 5] ranged from 10.21 ± 2.01 (CS/MMT 14 film) to 16.17% ± 0.96% (CS/MMT 41 film), whereas the pure CS film recorded an extensibility of 22.60% ± 1.76%. The effect of MMT on the extensibility shows a significant decrease in elongation to break as the MMT concentration increases. This might be a result of more interlayer spacing and intercalated structure of the clay as observed from XRD. The decrease in the extensibility could also be attributed to the restriction of mobility of polymer chains in the presence of clay particles. The results were found to be in good agreement with the findings reported by Svoboda et al.,[18] Hasegawa et al.[19] and Bangyekan et al.[20]
Figure 5.
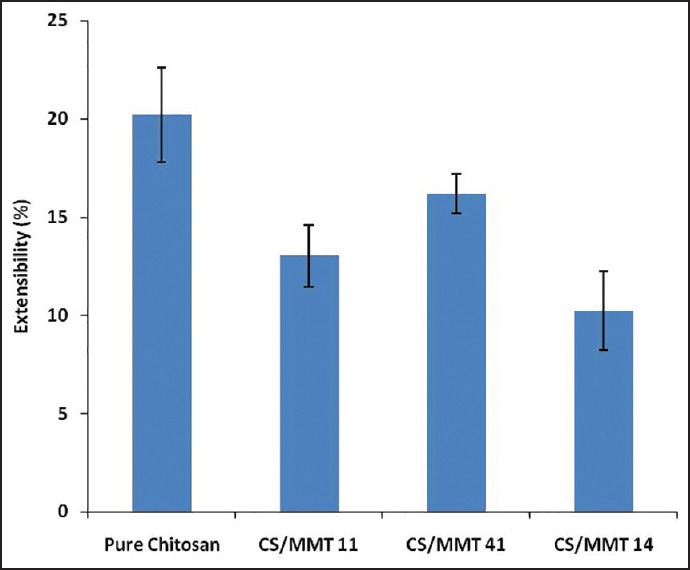
Extensibility of transdermal films
Fourier transform infrared spectroscopy analysis
The FTIR-ATR spectroscopy was used to analyze the interaction between CS and MMT in the transdermal film containing curcumin. Figure 6 shows that the peak at 2924, 2857 cm−1 are typical CS stretching vibrations, while the peaks at 1660, 1551 and 1309 cm−1 are amide 1, 2 and 3 respectively. The sharp peak at 1382 corresponds to the CH3 symmetrical deformation in the CS group. The peak at 897 cm−1 represents the saccharide group of CS.
Figure 6.
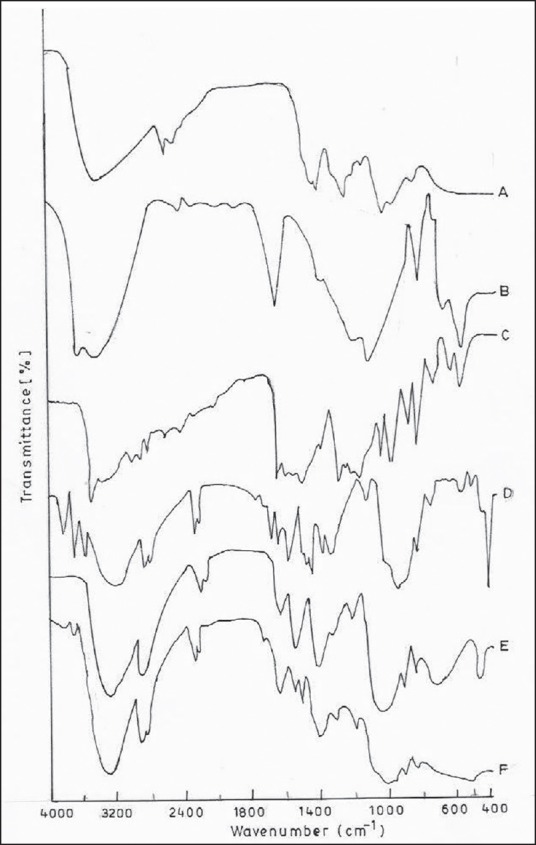
Fourier transform infrared spectroscopy spectra of (a) chitosan (CS) (b) montmorillonite (MMT) (c) curcumin (d) CS/MMT 11 (e) CS/MMT 41 and F) CS/MMT 14
The 3619 cm−1 peak corresponds to the OH stretching vibration in MMT. The H-O-H bending of H2O is indicated by the peak at 1633 cm−1. The peak at 1134 cm−1 indicates the Si-O stretching vibrations while the peak at 520 cm−1 is an indication of Si-O bending. The 919 cm−1 peak corresponds to the Al-O vibrations.
The peaks at 2363, 1613 and1512 cm−1 confirm the purity of curcumin in the films. The peaks around 3413, 1650 and 1081 depict that CS is overlapped with the bands of silicate while at 1647 cm−1 the vibration band of silicate is unaffected. The soluble parts of biopolymer containing OH and NH3 may form a hydrogen bond with MMT. The presence of amide group of CS visible in 1200-850 cm−1 range can be related to the addition of MMT. The vibration band at 1599 cm−1 corresponds to the deformation vibration of protonated amine group in the CS film, this group is shifted towards the lower frequency value 1517 cm−1 in the film which further indicates the electrostatic interaction between such groups and the negatively charged sites in the clay structure.
X-ray diffraction analysis
Figure 7 shows the XRD pattern of the CS/MMT polymer containing transdermal film. The CS shows a peak around 20.95° and the MMT peak is seen at 2θ = 8.54° and a d-spacing 4.3 A° the interaction of CS with MMT is seen at the peak 2θ = 7.9° and a d-spacing of 10.13 A°. This shifting of the peak to a lower angle is due to the intercalation between the CS and MMT layers. The increase of 4.3 A° to 13.78 A° in the interlayer spacing of the transdermal film further indicates the electrostatic interactions between the protonated amine group of the CS to the negatively charged sites in the clay structure. This leads to increase in crystallinity in the CS/MMT transdermal film as compared to individual CS and MMT structure.[21]
Figure 7.
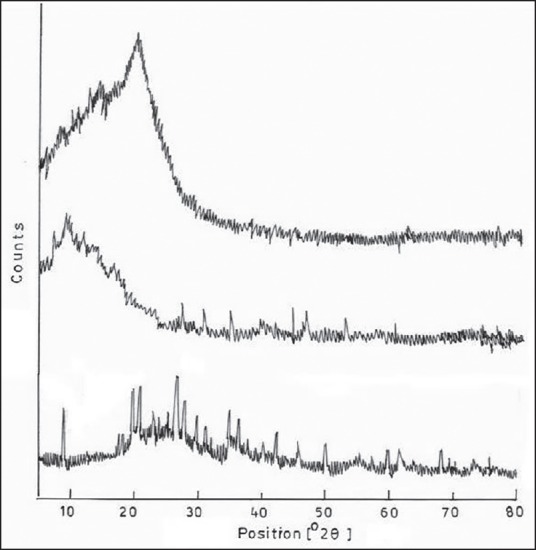
X-ray diffraction spectra pattern of (a) chitosan (CS), (b) montmorillonite (MMT) and (c) CS/MMT transdermal film
In vitro release study
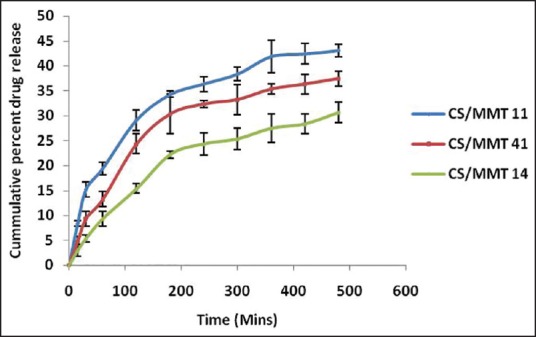
The results obtained from the in vitro dissolution study of the transdermal films are presented in Figure 8. The order of increased drug dissolution using the different approaches was as follows; CS/MMT 14> CS/MMT 41> CS/MMT 11. It was observed that during the first 60 min of the dissolution study, the CS/MMT 14 transdermal film showed just 19.41% ± 0.78% drug release as while the CS/MMT 41 and CS/MMT 11 transdermal films showed a release of 26.92% ± 1.03% and 33.43% ± 1.34% respectively. Results of various transdermal film tests indicate that among the three samples used for the preparation of CS/MMT transdermal films, CS/MMT 14 and CS/MMT 41 were more effective method than CS/MMT 11. This is quite evident from the drug release behavior of the CS/MMT transdermal films after 8 h of dissolution study which showed 53.34% ± 1.26%, 63.23% ± 1.56% and 73.27% ± 1.71% drug release for CS/MMT 14, 41 and 11 respectively. CS/MMT 14 and 41 showed better sustained release effects. This kind of behavior is a result of the strong electrostatic interaction between the cationic charges of CS and the anionic charges of MMT.
Figure 8.
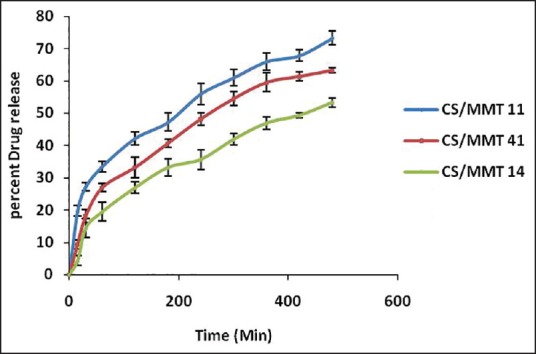
In vitro drug release of chitosan/montmorillonite K transdermal films
Determination of release kinetics
The regression coefficient (r2) values of Higuchi equation for CS/MMT transdermal films CS/MMT 11, CS/MMT 41 and CS/MMT 14 were found to be 0.9947, 0.9886 and 0.9884 respectively. To further confirm the mechanism of drug release from the CS/MMT transdermal films, the in vitro dissolution data were subjected to the Korsmeyer's Peppas equation. The values of the release exponent (n) were found to be ranging between 0.46 and 0.62 indicating a nonfickian (anomalous) drug release behavior. Due to the swelling ability of the polymer, there is an opening of the pore channels in between the polymer matrix which helps with the diffusion of the drug through the polymer matrix chains, thus, releasing the drug from the transdermal films. Table 4 depicts the values of various release kinetics parameters for the transdermal films.
Table 4.
Release kinetic parameters of transdermal films of curcumin
In vitro permeation study
Figure 9 presents the results obtained from the in vitro permeation study of the transdermal films. The order of increased drug dissolution using the different approaches was as follows; CS/MMT 14>CS/MMT 41> CS/MMT 11. Similar to the in vitro drug release effects, these results also clearly show that the CS/MMT 14 show the most sustained release effects as the release of the drug after 8 h of permeation study was 30.63% ± 2.04%, 37.45% ± 1.49% and 43.03% ± 1.09% for the transdermal films CS/MMT 14, CS/MMT 41 and CS/MMT 11 respectively. This phenomenon confirms that an ionic exchange reaction occurred between CS and MMT and that consequently CS was intercalated into the MMT structure.
Figure 9.
In vitro permeation study of the chitosan/montmorillonite K transdermal films
Stability study
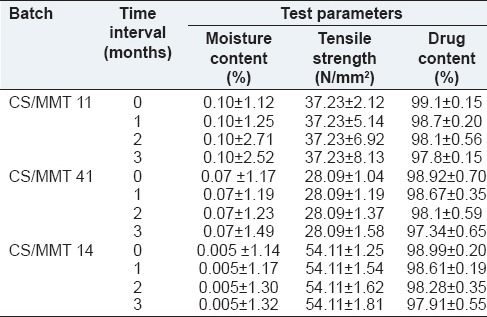
The prepared transdermal films were evaluated for stability studies. During the full duration of the study, temperature and relative humidity of about 40°C ± 2.20°C and 75% RH respectively were maintained. The transdermal films were analyzed at 0 day, 1, 2 and 3-month time interval for moisture content, tensile strength and drug content. To achieve required humidity, a saturated solution of sodium chloride was kept at the bottom of the desiccator. The performed stability studies indicated that there was no significant change in moisture content, tensile strength and drug content of the transdermal films as evident from Table 5.
Table 5.
Stability testing data for the prepared transdermal films
CONCLUSION
The transdermal films of Curcumin were formulated using solvent casting method employing the CS/MMT polymer composite and were evaluated for physical parametric tests, tensile strength, moisture content, swelling, in vitro dissolution studies and stability testing. FTIR results show that CS molecules and clay particles mainly interact on the surface each other. Similar type of intercalated and stacked, structures of CS and MMT were characterized according to XRD diffraction patterns. A great synergistic effect of MMT for improving thermal stability, mechanical, and barrier properties of CS matrix has been observed. This could be understood as due to a formation of filler network of MMT within the CS polymeric chains which forms a unique structure which provides larger tortuosity and obstacle for the transport of the drug. Increase in the concentration of MMT improved barrier properties and added to the polymer matrix of the gas permeability of clay, such as extension of the positive features, but also have gained a more opaque appearance that they have led to the produced films. The values of tensile strength of CS/MMT film increased significantly with increasing MMT concentration, while the values of extensibility decreased for high values of MMT concentration. In conclusion, the prepared curcumin loaded CS/MMT composite films can be of immense importance in the drug delivery through the transdermal route. The combination of biodegradable polymeric chains and clay reinforcement can be applied to achieve the desired combination of properties (mechanical, swelling and controlled release) of materials used for diverse biomedical and food applications.
ACKNOWLEDGMENTS
The authors gratefully acknowledge Dr. Madhu Chitkara, Vice Chancellor and Dr. Sandeep Arora, Dean, Chitkara College of Pharmacy, Chitkara University, Punjab, India, for support and institutional facilities.
Footnotes
Source of Support: Nil.
Conflict of Interest: None declared.
REFERENCES
- 1.Patel D, Chaudhary SA, Parmar B, Bhura N. Transdermal drug delivery system: A review. Pharm Innov. 2012;1:78–87. [Google Scholar]
- 2.Singh MC, Naik AS, Sawant SD. Transdermal drug delivery systems with major emphasis on transdermal patches: A review. J Pharm Res. 2010;3:2537–43. [Google Scholar]
- 3.Sakalle P, Dwivedi S, Dwivedi A. Design, evaluation, parameters and marketed products of transdermal patches: A review. J Pharm Res. 2010;3:235–40. [Google Scholar]
- 4.Paluszkiewicz C, Stodolak E, Hasik M, Blazewicz M. FT-IR study of montmorillonite-chitosan nanocomposite materials. Spectrochim Acta A Mol Biomol Spectrosc. 2011;79:784–80. doi: 10.1016/j.saa.2010.08.053. [DOI] [PubMed] [Google Scholar]
- 5.Khedr MA, Waly AI, Hafez AA. Synthesis of modified chitosan-montmorillonite nanocomposite. Aust J Basic Appl Sci. 2012;6:216–26. [Google Scholar]
- 6.Yang H, Wang W, Zhang J, Wang A. Preparation, characterization, and drug-release behaviors of a pH-Sensitive composite hydrogel bead based on guar gum, attapulgite, and Sodium Alginate. Int J Polym Mater Polym Biomater. 2012;62:369–76. [Google Scholar]
- 7.Anand P, Kunnumakkara AB, Newman RA, Aggarwal BB. Bioavailability of curcumin: Problems and promises. Mol Pharm. 2007;4:807–18. doi: 10.1021/mp700113r. [DOI] [PubMed] [Google Scholar]
- 8.Deodr SD, Sethi R, Srimal RC. Preliminary study on antirheumatic activity of curcumin (diferyloyl methane) Indian J Med Res. 1980;71:632–40. [PubMed] [Google Scholar]
- 9.Elsabee MZ, Abdou ES. Chitosan based edible films and coatings: A review. Mater Sci Eng C Mater Biol Appl. 2013;33:1819–41. doi: 10.1016/j.msec.2013.01.010. [DOI] [PubMed] [Google Scholar]
- 10.Ubaidulla U, Reddy MV, Ruckmani K, Ahmad FJ, Khar RK. Transdermal therapeutic system of carvedilol: Effect of hydrophilic and hydrophobic matrix on in vitro and in vivo characteristics. AAPS Pharm Sci Tech. 2007;8:2. doi: 10.1208/pt0804081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bahrami SB, Kordestani SS, Mirzadeh H, Mansoori P. Poly (vinyl alcohol) – Chitosan blends: Preparation, mechanical and physical properties. Iran Polym J. 2013;12:139–46. [Google Scholar]
- 12.Kasirga Y, Oral A, Caner C. Preparation and characterization of chitosan/montmorillonite-K10 nanocomposites films for food packaging applications. Polym Compos. 2012;33:1874–82. [Google Scholar]
- 13.Salcedo I, Aguzzi C, Sandri G, Bonferoni MC, Mori M, Cerezo P, et al. In vitro biocompatibility and mucoadhesion of montmorillonite chitosan nanocomposite: A new drug delivery. Appl Clay Sci. 2012;55:131–37. [Google Scholar]
- 14.Depan D, Kumar AP, Singh RP. Cell proliferation and controlled drug release studies of nanohybrids based on chitosan-g-lactic acid and montmorillonite. Acta Biomater. 2009;5:93–100. doi: 10.1016/j.actbio.2008.08.007. [DOI] [PubMed] [Google Scholar]
- 15.Cojocariu A, Porfire L, Cheaburu C, Vasile C. Chitosan/montmorillonite composites as matrices for prolonged delivery of some novel nitric oxide donor compounds based on theophylline and paracetamol. Cellulose Chem Technol. 2012;46:35–43. [Google Scholar]
- 16.Sothornvit R, Hong SI, An DJ, Rhim JW. Effect of clay content on the physical and antimicrobial properties of whey protein isolate/organo-clay composite films. Food Sci Technol. 2010;43:279–84. [Google Scholar]
- 17.Ray SS, Okamoto M. Polymer/layered silicate nanocomposites, structure, development and processing operations. Prog Polym Sci. 2003;28:1539–43. [Google Scholar]
- 18.Svoboda P, Zeng CC, Wang H, Lee LJ, Tomasko DL. Morphology and mechanical properties of polypropylene organoclay nanocomposites. J Appl Polym Sci. 2002;85:1562–70. [Google Scholar]
- 19.Hasegawa N, Okamoto H, Kato M, Usuki A. Preperation and properties of ethylene propylene rubber (EPR)-clay nanocomposites based on maleic anhydride-modified EPR and organophilic clay. J Appl Polym Sci. 2000;78:1918–23. [Google Scholar]
- 20.Bangyekan C, Aht-Ong D, Srikulkit K. Preparation and properties evaluation of chitosan-coated cassava starch films. Carbohydr Polym. 2006;63:61–71. [Google Scholar]
- 21.Liu H, Nakagawa K, Chaudhary D, Asakuma Y, Tadé MO. Freeze-dried macroporous foam prepared from chitosan/xanthan gum/montmorillonite nanocomposites. Chem Eng Res Des. 2011;89:2356–64. [Google Scholar]


